Published online Feb 28, 2016. doi: 10.4329/wjr.v8.i2.183
Peer-review started: September 18, 2015
First decision: October 30, 2015
Revised: November 8, 2015
Accepted: December 17, 2015
Article in press: December 18, 2015
Published online: February 28, 2016
Processing time: 163 Days and 4.8 Hours
Xanthogranulomatous cholecystitis (XGC) is an uncommon variant of chronic cholecystitis characterized by xanthogranulomatous inflammation of the gallbladder. Intramural accumulation of lipid-laden macrophages and acute and chronic inflammatory cells is the hallmark of the disease. The xanthogranulomatous inflammation of the gallbladder can be very severe and can spill over to the neighbouring structures like liver, bowel and stomach resulting in dense adhesions, perforation, abscess formation, fistulous communication with adjacent bowel. Striking gallbladder wall thickening and dense local adhesions can be easily mistaken for carcinoma of the gallbladder, both intraoperatively as well as on preoperative imaging. Besides, cases of concomitant gallbladder carcinoma complicating XGC have also been reported in literature. So, we have done a review of the imaging features of XGC in order to better understand the entity as well as to increase the diagnostic yield of the disease summarizing the characteristic imaging findings and associations of XGC. Among other findings, presence of intramural hypodense nodules is considered diagnostic of this entity. However, in some cases, an imaging diagnosis of XGC is virtually impossible. Fine needle aspiration cytology might be handy in such patients. A preoperative counselling should include possibility of differential diagnosis of gallbladder cancer in not so characteristic cases.
Core tip: A pre-operative diagnosis of xanthogranulomatous cholecystitis always comes handy for the surgeons. Diagnosing atypical cases can be challenging and acknowledge of pathological changes occurring in the disease along with the spectrum of imaging findings can be a useful armoury in hands of the radiologist. So we have tried to give a concise review of this entity emphasizing on radiological and pathological aspects. Few points in differential diagnosing with other entities especially carcinoma gallbladder have also been entailed.
- Citation: Singh VP, Rajesh S, Bihari C, Desai SN, Pargewar SS, Arora A. Xanthogranulomatous cholecystitis: What every radiologist should know. World J Radiol 2016; 8(2): 183-191
- URL: https://www.wjgnet.com/1949-8470/full/v8/i2/183.htm
- DOI: https://dx.doi.org/10.4329/wjr.v8.i2.183
Xanthogranulomatous cholecystitis (XGC) is a chronic inflammatory disease of the gallbladder characterized by focal or diffuse destructive inflammatory process followed by marked proliferative fibrosis along with infiltration of macrophages and foamy cells[1].
The nomenclature was done by McCoy et al[2] in 1976 though it was first described in 1970 by Christensen et al[3]. Christensen et al[3] and Amazon et al[4] had noted a pseudotumoral form of chronic cholecystitis that was characterized by the presence of xanthoma-like foam cells and scarring and that contained ceroid (wax-like) nodules in an inflammed gallbladder wall. They used the terms fibroxanthogranulomatous inflammation and ceroid granulomas of the gallbladder, respectively, which are now known as synonyms of XGC[3,4].
Albeit a common gallbladder pathology with an incidence rate of 0.7% to 10 %, it has been sparsely described in literature and is poorly understood[1]. It occurs in a wide range of age groups but the incidence is higher in the sixth and seventh decades of life. It’s occurrence in a two month old infant has also been described in literature[5]. Male preponderance has been reported with a male to female ratio of 2:1[6]. However one of the Indian studies found a marked female preponderance with a male to female ratio of 1:9[7]. The most important association of XGC is with gall stones which are seen in as many as 80% of cases[8].
Patients can present with features of acute cholecystitis (22%), chronic cholecystitis (88%), pain (95%), obstructive jaundice (22%), cholangitis (2%) and palpable mass (5%)[6]. On examination, a palpable mass or positive Murphy’s sign can be localised. However, these clinical features are not specific for XGC and often no clinical difference between patients with XGC and carcinoma gallbladder can be found[9].
Leukocytosis has been observed though there is no specific biochemical test or liver function discordance pointing towards the diagnosis of XGC. Complications are present in 32% of cases and include perforation, abscess formation, fistulous tracts to the duodenum or skin, and extension of the inflammatory process to the liver, colon, or surrounding soft tissues[10].
Yu et al[11] found that elevation of tumor biomarkers is frequent in XGC which further creates confusion in differentiating the disease with carcinoma of gallbladder.
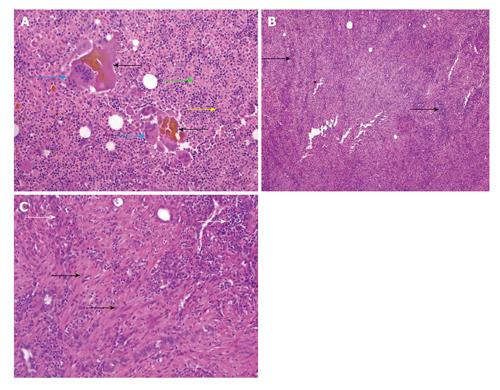
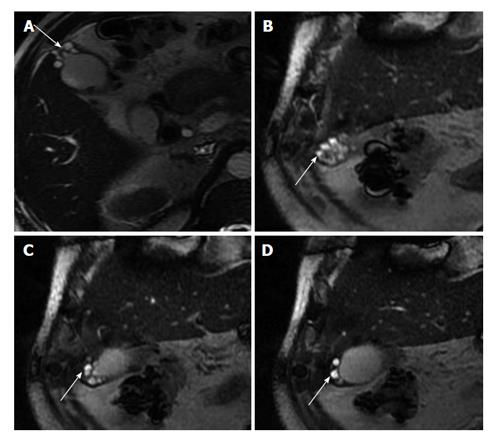
XGC is characterized pathologically by the presence of greyish-yellow nodules or streaks in the gallbladder wall which are mainly caused by lipid laden macrophages. The exact etiopathogenesis is unclear. One proposed theory behind the xanthogranulomatous etiology is mucosal ulceration or rupture of Rokitansky-Aschoff sinuses due to increased intraluminal pressure secondary to gallbladder or cystic duct obstruction which leads to entry of bile in gallbladder wall. This intramural bile is incompletely engulfed by the macrophages leading to chronic granulomatous inflammatory response (Figure 1A).
The histological diagnosis is based on diffuse or focal mural changes in the form of xanthoma cells (foamy histiocytes containing lipids and bile pigment), giant multinucleate histiocytes and acute or chronic inflammatory cells. These histiocytes are positive for CD68 on immunohistochemistry (Figures 1B, C and 2). Microabscesses also tend to form in the gallbladder wall and finally a fibrous reaction and scarring results from healing of the inflammatory reaction[12]. Rupture of gallbladder serosal lining and spread of the inflammatory response leads to adhesions with adjacent liver, duodenum and transverse colon (Figure 3).
XGC is associated with gallbladder carcinoma in 8.5% to 30.5% cases[13]. Gallbladder carcinoma may provide route for bile entry into the stroma owing to its greater degree of tissue destruction[14]. Obstruction of the cystic duct by a neoplasm may initiate the histiocytic inflammatory process of XGC[15]. The association is important because when both lesions are present in the same specimen, there is a possibility of overlooking the carcinoma altogether[16].
Possibility of coexisting infection has also been proposed. Howard et al[17] have reported that intraoperative cultures of the bile and gallbladder have been positive usually for Escherichia coli, Klebsiella, Enterococcus and, less frequently, for Pseudomonas, Serratia and Staphylococcus aureus.
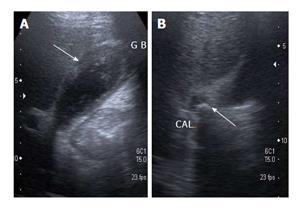
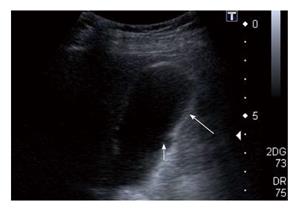
The sonographic findings include the presence of gallstones or sludge and moderate to marked focal or diffuse thickening of the gallbladder wall. Parra et al[18] observed that the wall thickening was hyperechoic in comparison to the liver in 100% of patients (Figure 4). Presence of hypoechoic nodules or bands in the thickened wall can occasionally be seen, the presence of which is considered a characteristic finding. Hypoechoic nodules on sonography have been observed in 15% and 73% cases by Parra et al[18] and Kim et al[19] respectively. Hypoechoic band has been observed in around 19% cases of XGC[18,20]. Xanthogranulomatous nodules behave as well-defined hypoechoic areas on sonography (Figure 5). Hypoechoic bands might be caused by a more generalized involvement of the mucosa[18]. Complications like perforation, abscess and hepatic infiltration can also be seen on sonography[18,19].
Computed tomography (CT) findings of patients presenting with acute symptoms and patients presenting with chronic symptoms are usually not much different[21]. CT findings include - diffuse or focal wall thickening, intramural hypoattenuating nodules in thickened walls, luminal surface enhancement (LSE) with continuous mucosal lines or mucosal lines with focal breach. Cholelithiasis and choledocholithiasis are often seen associated with XGC.
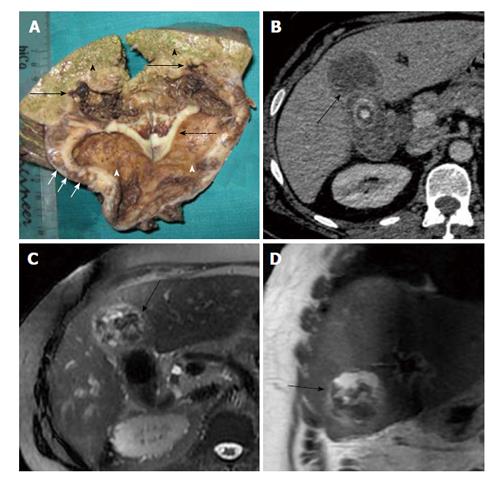
Gallbladder wall thickening can range from 4.0 mm to 18.5 mm and is usually diffuse in nature[21]. Diffuse gallbladder (GB) wall thickening has been observed in 88.9% and 87.8% of patients by two independent researchers Goshima et al[22] and Zhao et al[21] respectively. Focal thickening is less commonly seen in XGC, and is more likely to be associated with carcinoma of gallbladder. Diffuse thickening associated with XGC is usually symmetrical but diffuse asymmetrical thickening has also been described with XGC in 22.2% cases[21,22].To the best of our knowledge, XGC presenting as mass replacing gallbladder, intra-luminal mass or polypoidal mass-like thickening has not yet been described in literature.
The intramural nodules detected on imaging studies (85.7% and 61.1% by Zhao et al[21] and Goshima et al[22] respectively) are either xanthogranulomas or abscesses. Occupation of a large area of the thickened gallbladder wall by intramural nodules is highly suggestive of XGC[23] (Figure 6). Xanthogranulomas are more often revealed on imaging than abscesses though the later cause more clinical complications[15]. In acute inflammatory phase intramural nodules were abscesses in contrast to xanthogranulomas in the latter phase[15].
A continuous mucosal lining is more often observed with XGC (66.7% of cases) compared to a disrupted mucosal lining (33.3%)[22]. XGC is pathology of gallbladder wall and hence mucosal surface is intact or only focally denuded. On the contrary, carcinoma of gallbladder arises from the gallbladder epithelium and causes mucosal disruption in majority of the cases. Mucosal line disruption has been observed in 82.2% cases of carcinoma of gallbladder. Mucosal disruption in XGC is only seen with diffuse thickening of the gallbladder wall and patients with disrupted mucosal lining are more likely to have complications[21].
LSE is defined as enhancement of the gallbladder wall predominantly at the luminal surface. This finding was noted in 85.7% of cases by Zhao et al[21] and 70% cases by Shuto et al[24]. LSE noted in XGC is more apparent in the portal venous phase and represents preservation of the epithelial layer[21,24]. This further points towards the intramural location of the disease process with an intact overlying mucosa as opposed to the disrupted mucosa in gallbladder carcinoma.
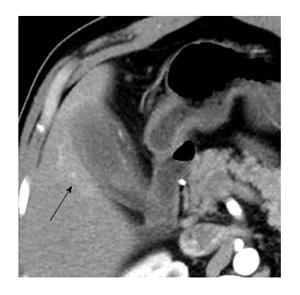
Cholelithiasis, choledocholithiasis (Figure 7), hepaticolithiasis have all been described frequently with XGC. Resultant gallbladder distention and biliary dilatation can be observed (Figure 7).
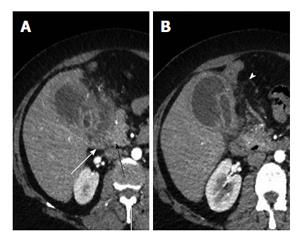
The infiltration of adjacent structurescan manifest as pericholecystic fat strandings, blurring of interface between gallbladder and liver, early enhancement of liver (or transient hepatic attenuation difference) (Figure 8), infiltration of bowel (duodenum/colon) (Figure 9), infiltration of stomach and invasion of abdominal wall (Figure 10). While pericholecystic fat stranding and blurring of interface between gallbladder and liver are quite frequent, the other findings are sparsely observed. Liver infiltration can result in an early enhancement of parenchyma in 40% of cases[21] (Figure 8). Other complications include gallbladder perforation, abscess formation (Figure 11) or fistulous communications. Involvement of the biliary tree by the inflammatory process (xanthogranulomatous choledochitis) can also be seen[25]. However, the absence of intrahepatic biliary dilatation is more frequently observed in XGC and is an important finding in differentiating it from carcinoma of gallbladder.
Occurrence of lymphadenopathy (> 10 mm in short axis diameter) has been variably described by different researchers. While Zhao et al[21] have described an incidence of 10.2%, Goshima et al[22] found an incidence of 90%. But both the researchers were of the opinion that loco-regional lymphadenopathy can be useful in differentiation from carcinoma of gallbladder. Only 41% patients with gallbladder carcinoma showed homogeneous enhancement of enlarged nodes compared to 100% patients with XGC[22].
Associated gallbladder or biliary malignancies can also be visualized on CT[13]. Notably, Indian researchers, Krishnani et al[13] have described a high coexistence of carcinoma gallbladder with XGC (19.6% of cases). Although the causative mechanism behind the association between the two entities is unclear, both are complications of cholelithiasis and cholecystitis of a particular duration. XGC may obscure the adenocarcinoma[13]. Also, the extent of carcinoma may be considerably overestimated or underestimated, especially since XGC is also known to form adhesions to other organs; features conventionally attributed to malignancies[9].
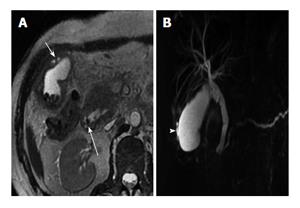
In-phase and opposed-phase chemical shift imaging is helpful in demonstrating fat within the thickened gallbladder wall in patients with XGC[26] (Figure 12). Zhao et al[21] subjected intramural nodules to chemical shift imaging. Seventy-seven point seven percent of XGC nodules showed reduced signal intensity on out-of-phase images. This variable nature of the intramural nodule on magnetic resonance imaging (MRI) can be attributed to the presence of diverse contents like foamy histiocytes, lymphocytes, plasma cells, polymorphonuclear leucocytes, fibrosis, giant cells, microabscess and necrosis within these nodules[22]. The researchers also observed that few nodules (2 out of 11) were detected only on CT and not seen on MRI and attributed this to a lower spatial resolution of MRI. Areas of iso- to slightly high signal intensity on T2-weighted images, showing slight enhancement at early phase and strong enhancement during delayed phase of dynamic study, corresponded with areas of abundant xanthogranulomas[24]. Areas with very high signal intensity on T2-weighted images without enhancement corresponded with necrosis and/or abscesses[24].
LSE of gallbladder wall represented preservation of the epithelial layer[24]. The early-enhanced areas of the liver bed on dynamic CT and MR images can be seen sometimes associated with XGC which correspond with accumulation of inflammatory cells and abundant fibrosis[24].
Kang et al[27] have revealed the benefit of diffusion-weighted magnetic resonance imaging (DWI) in differentiating XGC from the wall-thickening type of gallbladder cancer. Diffusion restriction was more frequently seen in gallbladder cancer (68%) than in XGC (7%). They also found out that the mean ADC value of XGC was higher than that of the wall-thickening type of gallbladder cancer with statistical significance (1.637 × 10-3 mm2/s vs 1.076 × 10-3 mm2/s respectively, P = 0.005)[27]. The authors concluded that the addition of DWI to conventional MRI improves discrimination between XGC and the wall-thickening type of gallbladder cancer.
Sawada et al[28] have described positive uptake of XGC on 18F-FDG positron emission tomography (PET) scan which again adds to the confusion. They have described the expression of GLUT1 and GLUT3 receptors in XGC to be the causative factor behind the false positive PET scan[28]. Radiological feature of XGC have been summarized in Table 1.
| Findings |
| Diffuse or focal mural thickening |
| Luminal surface enhancement |
| Intramural fat |
| Hypodense/hypoechoic nodules or bands |
| Associations |
| Cholelithiasis |
| Choledocholithiasis |
| Gallbladder carcinoma |
| Complications |
| Gallbladder perforation |
| Abscesses |
| Adhesions and fistulas to liver, duodenum, gatric outlet, colon (hepatic flexure/transverse colon) and anterior abdominal wall |
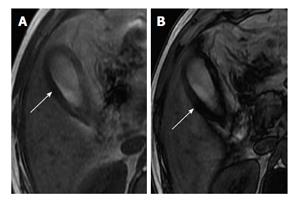
Gallbladder adenomyomatosis is a process of diffuse epithelial and smooth muscle proliferation likely in response to chronic gallbladder obstruction. Dilated Rokitansky-Aschoff sinuses contribute to formation of intramural diverticula that may contain bile, cholesterol, sludge or stones. Cholesterol crystals show characteristic reverberation or V-shaped comet tail artefacts on sonography. On T2-weighted MRI a characteristic “pearl necklace sign” is noted (Figure 13). Intramural foci of adenomyomatosis are often small and aligned in a linear fashion[29]. Presence of intramural nodules covering a large area of thickened gallbladder wall is specific for XGC[30]. Rate of complications is higher in XGC compared to adenomyomatosis. Inflammatory changes outside the gallbladder should raise suspicion of XGC over adenomyomatosis[31].
Although accurate pre-operative differentiation of XGC from carcinoma purely on the basis of radiological and clinical features may be difficult, there are some pointers on imaging that have been found to be helpful. In a study done by Goshima et al[22], five CT findings showed significant difference between XGC and carcinoma of gallbladder. These findings were diffuse gallbladder wall thickening, continuous mucosal lining, intramural hypo-attenuating nodules in the thickened walls, absence of macroscopic hepatic invasion and absence of intrahepatic bile duct dilatation[22]. They reported that the diagnostic accuracy of XGC increases with the presence of three or more of the above mentioned findings. Besides, presence of regional lymphadenopathy is more prevalent in carcinoma compared to XGC. While 58.9% cases with gallbladder carcinoma had retroperitoneal lymph nodes enlargement, only 10.2% cases of XGC had mild lymph node enlargement (1-1.5 cm in diameter)[21].
Actinomycosis of the gallbladder presenting as a mass with extensive infiltration into surrounding structures by sonography and CT is very difficult to differentiate from gallbladder carcinoma and XGC, as radiologic features overlap considerably[32]. In cases where a correct diagnosis is impossible, either close clinical and radiological follow up or imaging-guided aspiration or biopsy may be useful.
Fine needle aspiration cytology (FNAC) plays an important role in making pre-operative differentiation between carcinoma and XGC or in co-existent lesions.Percutaneous as well as endoscopic needle biopsy can be done in patients suspicious of malignancy. In personal experience of the authors, approaching the lesion with a transhepatic route can minimise the risk of potential complications. The sensitivity of detecting malignancy is around 80% when adenocarcinoma is associated with XGC and overall sensitivity and specificity of detecting carcinoma is approximately 90% and 94% respectively[14].
Most gallbladder carcinomas associated with XGC occur in the GB neck region which is due to increased pressure within the gallbladder. Thus, careful observation of the GB neck and cystic duct region by endoscopic ultrasound (EUS) and adequate sampling from this region can greatly reduce the incidence of false negative diagnosis of co-existing gallbladder carcinomas. Hijioka et al[33] have reported an accuracy of 93.3% using EUS guided FNAC.
At operation,XGC may give appearance of an advanced gallbladder carcinoma due to wall thickening and local destructive spread of inflammation[12]. A carcinoma may be masked by the severe inflammation[12]. Intraoperative frozen section investigation or FNAC has been suggested to confirm the diagnosis. In cases with no invasion of adjacent organs these tools are indicated because they can change the surgical strategy (e.g., simple cholecystectomy vs associated liver resection).
Complete resection of the gallbladder is not always possible especially due to poor visualization of the Calot’s triangle. Prolonged operating time and technical difficulties are noted along with a high conversion rate (laparoscopic to open cholecystectomy) of upto 80%[34].The complication rates may be as high as 20% and length of hospital stay is also generally longer[12].
XGC can be a diagnostic dilemma and a correct pre-operative diagnosis can be aided by awareness of characteristic findings on CT and MRI. In some cases diagnosing this entity only on imaging can be extremely challenging and FNAC may be helpful in pre-operative diagnosis.
P- Reviewer: Cha SH, Lipar M S- Editor: Ji FF L- Editor: A E- Editor: Liu SQ
| 1. | Rammohan A, Cherukuri SD, Sathyanesan J, Palaniappan R, Govindan M. Xanthogranulomatous cholecystitis masquerading as gallbladder cancer: can it be diagnosed preoperatively? Gastroenterol Res Pract. 2014;2014:253645. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 2. | McCoy JJ, Vila R, Petrossian G, McCall RA, Reddy KS. Xanthogranulomatous cholecystitis. Report of two cases. J S C Med Assoc. 1976;72:78-79. [PubMed] |
| 3. | Christensen AH, Ishak KG. Benign tumors and pseudotumors of the gallbladder. Report of 180 cases. Arch Pathol. 1970;90:423-432. [PubMed] |
| 4. | Amazon K, Rywlin AM. Ceroid granulomas of the gallbladder. Am J ClinPathol. 1980;73:123-127. [PubMed] |
| 5. | Kim SH, Kim HY, Jung SE, Park KW, Choi YH, Kim WS, Park SH. Xanthogranulomatous cholecystits in 2-month-old infant. J Korean Surg Soc. 2013;85:191-194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 6. | Guzmán-Valdivia G. Xanthogranulomatous cholecystitis in laparoscopic surgery. J Gastrointest Surg. 2005;9:494-497. [PubMed] |
| 7. | Balagué C, Targarona EM, Sugrañes G, Rey MJ, Arce Y, Viella P, Trias M. [Xanthogranulomatous cholecystitis simulating gallbladder neoplasm: therapeutic implications]. Gastroenterol Hepatol. 1996;19:503-506. [PubMed] |
| 8. | Ros PR, Goodman ZD. Xanthogranulomatous cholecystitis versus gallbladder carcinoma. Radiology. 1997;203:10-12. [PubMed] |
| 9. | Roberts KM, Parsons MA. Xanthogranulomatous cholecystitis: clinicopathological study of 13 cases. J Clin Pathol. 1987;40:412-417. [PubMed] |
| 10. | Houston JP, Collins MC, Cameron I, Reed MW, Parsons MA, Roberts KM. Xanthogranulomatous cholecystitis. Br J Surg. 1994;81:1030-1032. [PubMed] |
| 11. | Yu H, Yu TN, Cai XJ. Tumor biomarkers: help or mislead in the diagnosis of xanthogranulomatous cholecystitis?-analysis of serum CA 19-9, carcinoembryonic antigen, and CA 12-5. Chin Med J (Engl). 2013;126:3044-3047. [PubMed] |
| 12. | Srinivas GN, Sinha S, Ryley N, Houghton PW. Perfidious gallbladders - a diagnostic dilemma with xanthogranulomatous cholecystitis. Ann R Coll Surg Engl. 2007;89:168-172. [PubMed] |
| 13. | Krishnani N, Shukla S, Jain M, Pandey R, Gupta RK. Fine needle aspiration cytology in xanthogranulomatous cholecystitis, gallbladder adenocarcinoma and coexistent lesions. Acta Cytol. 2000;44:508-514. [PubMed] |
| 14. | Benbow EW. Xanthogranulomatous cholecystitis associated with carcinoma of the gallbladder. Postgrad Med J. 1989;65:528-531. [PubMed] |
| 15. | Kim PN, Lee SH, Gong GY, Kim JG, Ha HK, Lee YJ, Lee MG, Auh YH. Xanthogranulomatous cholecystitis: radiologic findings with histologic correlation that focuses on intramural nodules. AJR Am J Roentgenol. 1999;172:949-953. [PubMed] |
| 16. | Benbow EW, Taylor PM. Simultaneous xanthogranulomatous cholecystitis and primary adenocarcinoma of gallbladder. Histopathology. 1988;12:672-675. [PubMed] |
| 17. | Howard TJ, Bennion RS, Thompson JE. Xanthogranulomatous cholecystitis: a chronic inflammatory pseudotumor of the gallbladder. Am Surg. 1991;57:821-824. [PubMed] |
| 18. | Parra JA, Acinas O, Bueno J, Güezmes A, Fernández MA, Fariñas MC. Xanthogranulomatous cholecystitis: clinical, sonographic, and CT findings in 26 patients. AJR Am J Roentgenol. 2000;174:979-983. [PubMed] |
| 19. | Kim PN, Ha HK, Kim YH, Lee MG, Kim MH, Auh YH. US findings of xanthogranulomatous cholecystitis. Clin Radiol. 1998;53:290-292. [PubMed] |
| 20. | Casas D, Pérez-Andrés R, Jiménez JA, Mariscal A, Cuadras P, Salas M, Gómez-Plaza MC. Xanthogranulomatous cholecystitis: a radiological study of 12 cases and a review of the literature. Abdom Imaging. 1996;21:456-460. [PubMed] |
| 21. | Zhao F, Lu PX, Yan SX, Wang GF, Yuan J, Zhang SZ, Wang YX. CT and MR features of xanthogranulomatous cholecystitis: an analysis of consecutive 49 cases. Eur J Radiol. 2013;82:1391-1397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 46] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 22. | Goshima S, Chang S, Wang JH, Kanematsu M, Bae KT, Federle MP. Xanthogranulomatous cholecystitis: diagnostic performance of CT to differentiate from gallbladder cancer. Eur J Radiol. 2010;74:e79-e83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 63] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 23. | Chun KA, Ha HK, Yu ES, Shinn KS, Kim KW, Lee DH, Kang SW, Auh YH. Xanthogranulomatous cholecystitis: CT features with emphasis on differentiation from gallbladder carcinoma. Radiology. 1997;203:93-97. [PubMed] |
| 24. | Shuto R, Kiyosue H, Komatsu E, Matsumoto S, Kawano K, Kondo Y, Yokoyama S, Mori H. CT and MR imaging findings of xanthogranulomatous cholecystitis: correlation with pathologic findings. Eur Radiol. 2004;14:440-446. [PubMed] |
| 25. | Goldar-Najafi A, Khettry U. Xanthogranulomatous choledochitis: a previously undescribed mass lesion of the hepatobiliary and ampullary region. Semin Liver Dis. 2003;23:101-106. [PubMed] |
| 26. | Hatakenaka M, Adachi T, Matsuyama A, Mori M, Yoshikawa Y. Xanthogranulomatous cholecystitis: importance of chemical-shift gradient-echo MR imaging. Eur Radiol. 2003;13:2233-2235. [PubMed] |
| 27. | Kang TW, Kim SH, Park HJ, Lim S, Jang KM, Choi D, Lee SJ. Differentiating xanthogranulomatous cholecystitis from wall-thickening type of gallbladder cancer: added value of diffusion-weighted MRI. Clin Radiol. 2013;68:992-1001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 52] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 28. | Sawada S, Shimada Y, Sekine S, Shibuya K, Yoshioka I, Matsui K, Okumura T, Yoshida T, Nagata T, Uotani H. Expression of GLUT-1 and GLUT-3 in xanthogranulomatous cholecystitis induced a positive result on 18F-FDG PET: report of a case. Int Surg. 2013;98:372-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 29. | Yoshimitsu K, Irie H, Aibe H, Tajima T, Nishie A, Asayama Y, Matake K, Yamaguchi K, Matsuura S, Honda H. Well-differentiated adenocarcinoma of the gallbladder with intratumoral cystic components due to abundant mucin production: a mimicker of adenomyomatosis. Eur Radiol. 2005;15:229-233. [PubMed] |
| 30. | Shetty GS, Abbey P, Prabhu SM, Narula MK, Anand R. Xanthogranulomatous cholecystitis: sonographic and CT features and differentiation from gallbladder carcinoma: a pictorial essay. Jpn J Radiol. 2012;30:480-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 31. | Cecava ND, Andrews R. Case report of xanthogranulomatous cholecystitis, review of its sonographic and magnetic resonance findings, and distinction from other gallbladder pathology. J Radiol Case Rep. 2011;5:19-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 32. | Lee YH, Kim SH, Cho MY, Rhoe BS, Kim MS. Actinomycosis of the gallbladder mimicking carcinoma: a case report with US and CT findings. Korean J Radiol. 2007;8:169-172. [PubMed] |
| 33. | Hijioka S, Mekky MA, Bhatia V, Sawaki A, Mizuno N, Hara K, Hosoda W, Shimizu Y, Tamada K, Niwa Y. Can EUS-guided FNA distinguish between gallbladder cancer and xanthogranulomatous cholecystitis? Gastrointest Endosc. 2010;72:622-627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 50] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 34. | Guzmán-Valdivia G. Xanthogranulomatous cholecystitis: 15 years’ experience. World J Surg. 2004;28:254-257. [PubMed] |