Published online Mar 26, 2020. doi: 10.12998/wjcc.v8.i6.1150
Peer-review started: November 26, 2019
First decision: January 19, 2020
Revised: February 1, 2020
Accepted: February 28, 2020
Article in press: February 28, 2020
Published online: March 26, 2020
Processing time: 120 Days and 15.4 Hours
Invaginated teeth pose greater challenges in clinical management because of their complex configuration. With advancements in equipment and materials, such as the dental operation microscope, cone-beam computed tomography and mineral trioxide aggregate, the preservation rate of type III dens invaginatus could be greatly increased.
This case report presented a 31-year-old woman with complaints of spontaneous swelling and pain in the right maxillary lateral tooth. With the aid of cone-beam computed tomography, type III dens invaginatus with apical periodontitis was diagnosed and confirmed. Three-visit endodontic treatment was performed. In the first visit, the invagination was carefully removed under the dental operation microscope, and chemomechanical preparation was done. In the second visit, mineral trioxide aggregate apical barrier surgery was performed in this tooth. In the third visit, the canal was finally obturated with thermoplastic gutta-percha to recover the crown morphology. A 26-mo follow-up revealed a satisfied outcome both in the radiographic and oral examinations.
In this case, removal of the entire abnormal structure provided great convenience for the follow-up treatment. When confronted with the same clinical case in the future, we can take a similar approach to address it.
Core tip: Type III dens invaginatus poses great challenges when conducting clinical operations. This article reported an upper right lateral incisor with type III dens invaginatus using the method that removed the invagination under the dental operation microscope, and then mineral trioxide aggregate apical barrier was made for the convenience of next treatment. The 26-mo follow-up revealed a satisfactory therapeutic effect.
- Citation: Liu J, Zhang YR, Zhang FY, Zhang GD, Xu H. Microscopic removal of type III dens invaginatus and preparation of apical barrier with mineral trioxide aggregate in a maxillary lateral incisor: A case report and review of literature. World J Clin Cases 2020; 8(6): 1150-1157
- URL: https://www.wjgnet.com/2307-8960/full/v8/i6/1150.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v8.i6.1150
Dens invaginatus (DI) is a developmental variation resulting from an infolding of the outer enamel surface of a tooth into the interior[1]. It is caused by deepening or invagination of the enamel organ into the dental papilla before biological mineralization takes place[2]. After the tooth erupts, a cystic deep cavity can appear on the tooth surface. It usually occurs in maxillary lateral incisors and occasionally in maxillary central incisors or canines[1,3]. According to the depth of the invagination and anatomic variation, it can be divided into the deformed lingual sulcus, deformed root sulcus, malformed lingual tip, and dens in dente. Dens in dente is rare but the most severe condition in clinical practice[1]. The classification advised by Oehlers is the most commonly used, which classifies DI into three types on the basis of penetration depth and communication with periapical tissue[4]. Type I: Invagination confined inside the crown and not extending beyond the cementoenamel junction. Type II: Invagination extending beyond the cementoenamel junction but does not reach periapical tissue and remains as a blind sac. It may or may not communicate with the pulp. Type III: Invagination penetrates through the entire root, perforating the apical area and having a second foramen in the apical or periodontal area; there is no immediate connection with the pulp. A study on the proportion of various Oehlers types found that type I accounted for 81.25%, type II accounted for 6.25% and type III accounted for 12.5%[5]. This article reports and discusses a case of non-surgical treatment of a maxillary lateral incisor diagnosed as type III DI with apical periodontitis. Treatment was performed with the guidance of cone-beam computed tomography (CBCT) imaging by removing the invagination with ultrasonic equipment and preparing an apical barrier with mineral trioxide aggregate (MTA) under a dental operation microscope (DOM).
A 31-year-old woman presented to the Department of Conservative Dentistry and Endodontics with complaints of spontaneous swelling and pain in an upper right anterior tooth for several days.
The patient felt spontaneous swelling and pain in an upper right anterior tooth for several days. The tooth was insensitive to cold and heat, but the patient felt uncomfortable when chewing. The tooth and its periodontal tissue had swollen before.
The patient recalled that the tooth had swelled before, and it was relieved after oral anti-inflammatory drugs.
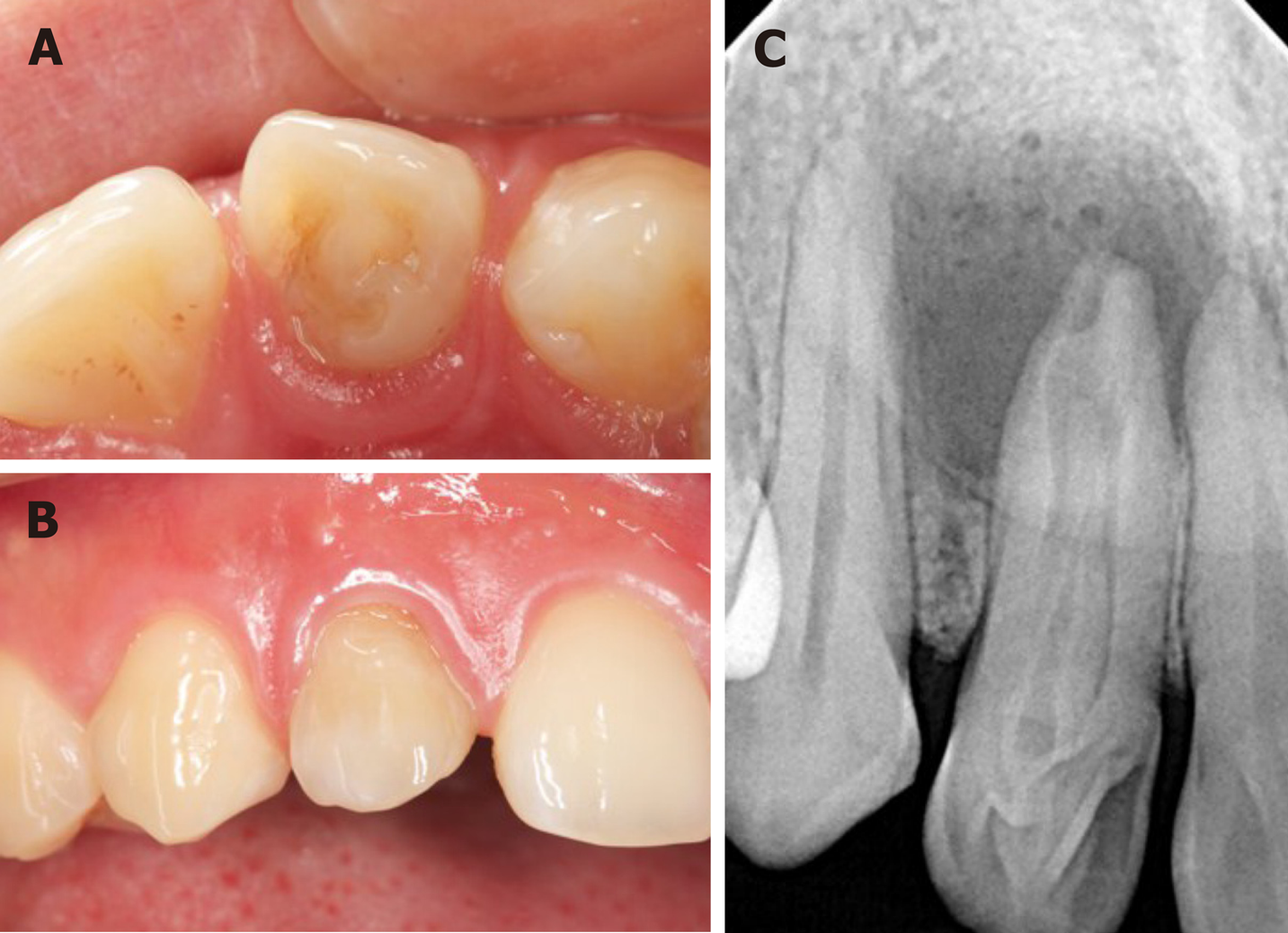
Intra-oral examination disclosed the upper right lateral incisor with unusual morphological features, in which a deep sulcus of the palatal aspect was presented (Figure 1A). The crown also had a little change in color when compared to the adjacent tooth (Figure 1B). The tooth did not respond to thermal and cold tests, but it was sensitive to percussion. There was a swelling in the apical mucosa area, and no obvious fluctuation was detected. The electric pulp vitality test indicated pulp necrosis.
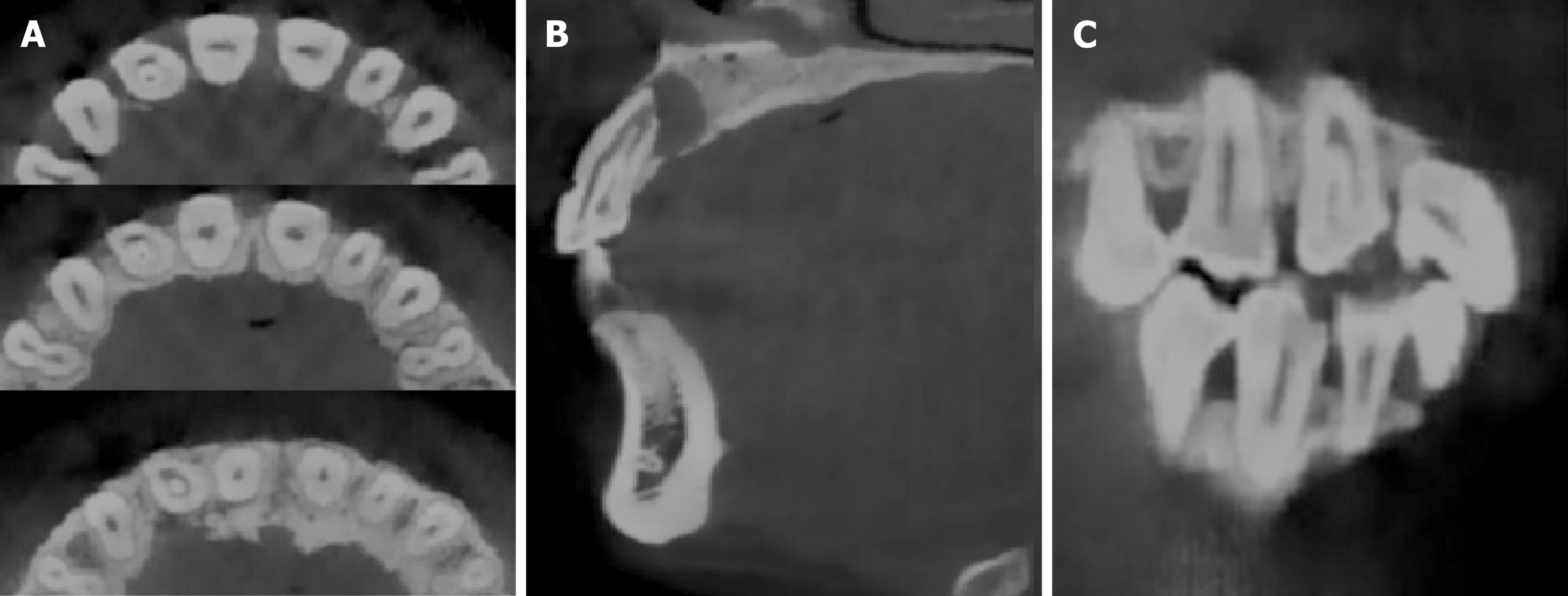
A preoperative radiographic examination (Figure 1C) showed the presence of dens invagination in the upper right lateral incisor and a large periapical radiolucency appearing around the apex. Because there was an aberrant anatomic structure and a periapical radiolucency of the tooth, a CBCT scan was taken as a complementary examination (Figure 2). The CBCT showed that the distal, mesial and palatal portions of the upper-middle section of the invagination was connected to the external teeth, and the lower section was separated from the external teeth.
Based on the subjective, objective and radiographic finding, a diagnosis of DI with apical periodontitis was made in the right maxillary lateral incisor.
The proposed treatment protocol was as follows: The first protocol was extracting the tooth and planting; the second protocol was doing apical surgery after inflammatory control; and the third protocol was trying to remove the invaginated tooth and making an apical barrier under the DOM and deciding whether to perform apical surgery, depending on the apical healing situation.
After consideration of the advantages, disadvantages, prognosis, and cost of every treatment protocol, the patient chose the third protocol, which tried to remove the invaginated tooth and prepare an MTA apical barrier under the DOM. The difficulty of the operation was evaluated preoperatively and the possibility of extracting the invagination was assessed by CBCT images. The informed consent of treatment was provided for the patient.
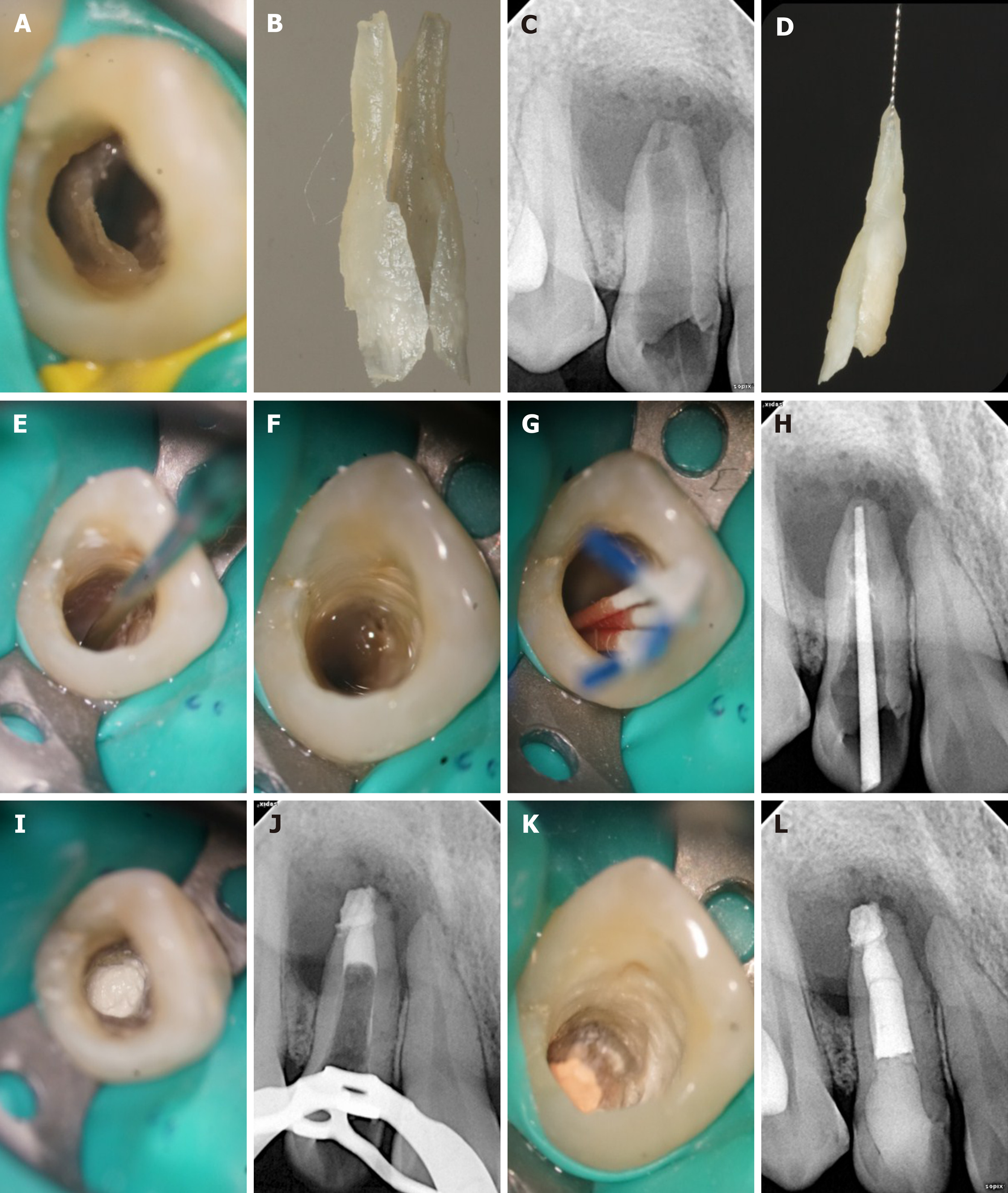
At the first appointment, local anesthesia consisting of articaine with 1:100000 epinephrine was administered, and access to the cavity was gained under rubber dam isolation. Using the DOM (OPMI PROergo, Carl Zeiss Meditec AG, Jena, Germany), the root canal of the invagination was visualized, just as shown by CBCT, and the labial side was separated from the external tooth. The abnormally invaginated tissue was carefully removed from the root trunk using ET20 and ET40 ultrasonic tips until completely separated (Figure 3A) and removed by medical forceps (Figure 3B). Afterwards, an intraoperative film was taken to confirm that the invagination had been completely removed (Figure 3C and 3D). Then, an ultrasonic tip was used to activate the canal soaked with sodium hypochlorite (Figure 3E). After drying with a sterile absorbent paper point, the canal was filled with calcium hydroxide paste and sealed temporarily. The patient was given a prescription for anti-inflammatory drugs with instructions to take for pain only if needed and to inform the doctor immediately if there was any discomfort. The patient was scheduled for MTA apical barrier surgery 1 wk later.
At the second appointment, the pain of the upper right lateral tooth had been significantly relieved. The examination found that the tooth was not sensitive to percussion, and no tenderness but slight bulging was present in the labial apical mucosa. Under rubber dam isolation, the calcium hydroxide in the root canal was removed by ultrasound. Then, 1% sodium hypochlorite was flushed into the canal and activated with ultrasonic tips, saline was used in the final irrigation (Figure 3F), and the canal was dried with absorbent paper points (Figure 3G). The working length was determined by an electronic apex locator and confirmed radiographically (Figure 3H). An apical barrier of 4 mm in thickness was created with MTA (Figure 3I and 3J), where a wet cotton pellet was placed above. Then the tooth was sealed with Ceivitron (GC Corporation, Tokyo, Japan).
At the third appointment, which was set for 1 wk after the second appointment, the patient felt better. There was no obvious discomfort in the upper right lateral tooth, and the apical bulging in the labial mucosa was reduced. Rubber dam isolation was carried out to remove the temporary materials and cotton pellet of the root canal. Under the DOM, the barrier was probed firmly with a DG-16 probe (Hu-Friedy, Chicago, IL, United States) to confirm that the MTA was completely coagulated. The remaining space was then obturated with thermoplastic gutta-percha (SybronEndo, Orange, CA, United States) and AH-Plus (Dentsply Sirona, York, PA, United States) sealer, using a vertical condensation technique (Figure 3K). Thereafter, the access cavity was sealed with composite resin (ESPE Z350; 3M Science, Maplewood, MN, United States) (Figure 3L).
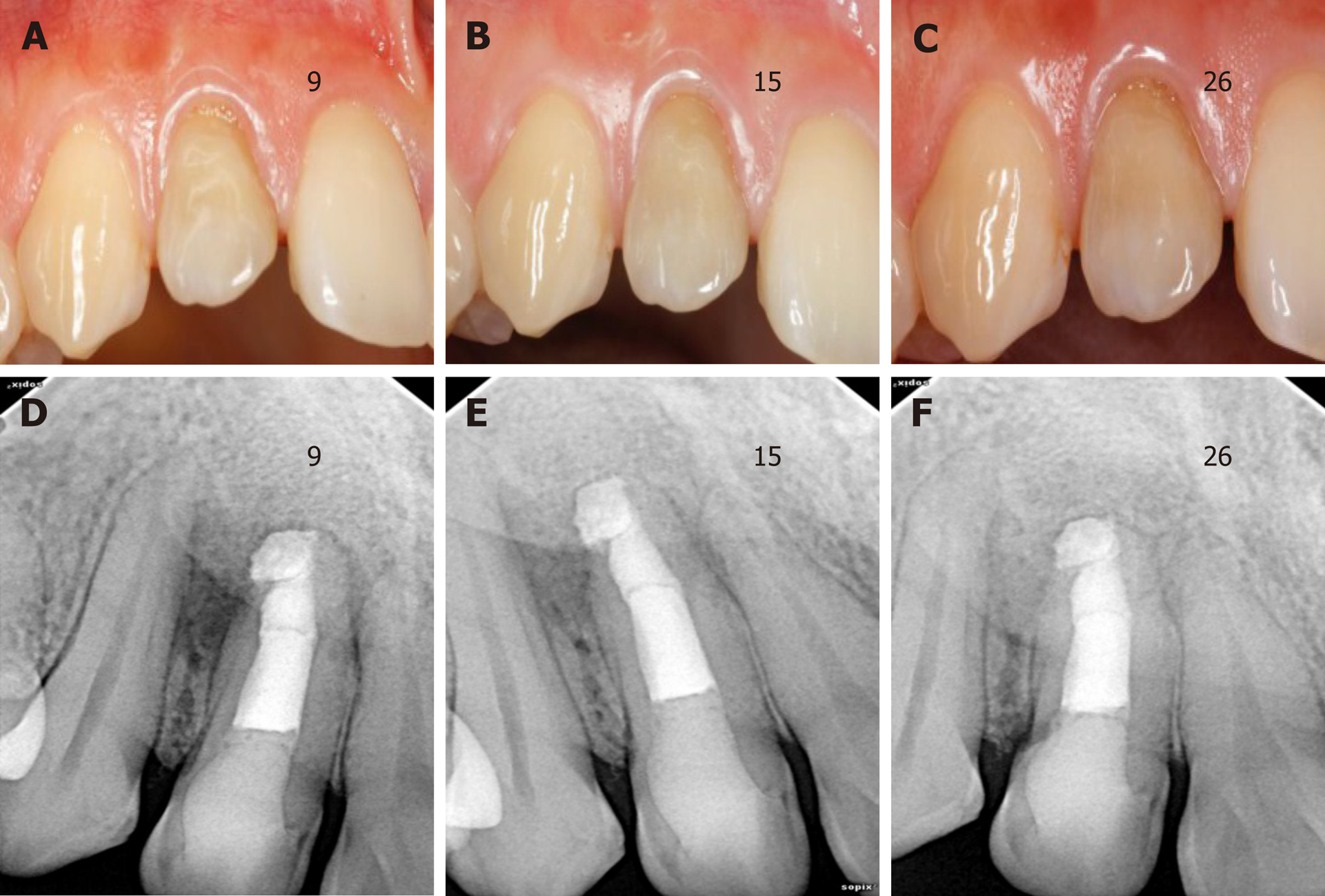
At the 9-, 15- and 26-mo follow-up appointments, the tooth remained asymptomatic. Chewing function could be exercised normally. However, the color of the crown did not significantly change until 26 mo (Figure 4A-C). There was no tenderness or obvious bulging in the labial apical mucosa. Radiographic examination at 9-, 15- and 26-mo postoperatively (Figure 4D-F) showed ongoing periapical repair, in which the apical radiolucency had been reduced and the bone trabecular structure was visible.
Dens in dente is the most serious type of dens invagination[1]. It has been reported that the incidence of DI ranges from 0.04% to 10%, with the maxillary lateral incisors most susceptible to involvement[6-8]. Depending on the severity of the infection and the complexity of its anatomy, several treatment methods for the invagination have been described. They included filling treatment, endodontic treatment[9], apical surgery[10,11], intentional reimplantation[12], and finally extraction. A case of invagination removal had also been reported in other literature[13].
In this case, after careful preoperative CBCT analysis, it was decided to use the method of removing the invagination and preparing the MTA apical barrier under a DOM to preserve the tooth. According to American Association of Endodontists commendations, CBCT should be considered the preferred treatment of teeth with the potential of suspected complex canal morphology or dental anomalies[14]. As a non-invasive diagnostic tool, CBCT has the advantages of increased accuracy and higher resolution. Moreover, CBCT offers significant scan-time reduction, radiation dose reduction, and reduced cost for the patient[15-17]. Compared with traditional 2-dimensional radiographs, CBCT produces undistorted three-dimensional information, which clearly displays images of the facial skeleton and the teeth and their surrounding tissues[18]. The induction and widespread adoption of the DOM is the most revolutionary in non-surgical and surgical endodontic treatment, which offers various high- and low-power magnifications. DOM offers homogeneous illumination without shadows and a 3-dimensional view, which combine to make the examination site clearer[19,20].
Despite the help of CBCT and the DOM, there were still some challenges in this case. One of the difficulties was how to successfully remove the invagination and reduce the damage to the main root canal wall. We were inspired by orthognathic surgery to place the ultrasound tip in the root canal of the invaginated tooth. It was expected that the force of ultrasonic vibration would be conducted to the joint between the invagination and external tooth so that the invagination could be removed. Finally, the invagination was taken out in two steps, which basically conformed to the preoperative plan.
Another difficulty was the preparation of the MTA apical barrier. The tooth was characterized by absorption of the apex and destruction of the apical foramen. An apical barrier is used in teeth with necrotic pulps, open apices, periapical radiolucencies and root resorption[21]. Materials including collagen, calcium-enriched mixture cement, Biodentine and Portland cement have been reportedly used to make apical barriers[21]. However, several studies with varying follow-up periods revealed that MTA as an apical barrier has a high success rate in teeth with necrotic pulps, open apices, and apical lesions[22-26]. MTA is characterized by hard tissue conductivity[27] and biocompatibility[28] and has been recognized as a bioactive material. The main components of MTA are tricalcium silicate, tricalcium aluminate, calcium silicate, and tetracalcium aluminoferrite[29]. MTA is a type of hydraulic cement. Its powder contains small hydrophilic particles, which can interact with the natural fluids present in tissues[30]. The MTA barrier also has the advantages of reducing clinical time cost and early safe tooth restoration[21]. In this case, we chose MTA as the apical barrier material. The pulsatile exudation of the apex could be seen under the DOM, which made placement of the MTA apical barrier difficult. During the apical barrier operation, the exudation liquid was aspirated with a syringe while filling the MTA. Finally, the material was compacted by ultrasonic vibration to improve the sealing performance. At the same time, the depth of the thermoplastic gutta-percha was controlled at the level of the alveolar ridge, to leave enough depth and space for the resin to make up for the loss of dentin. Therefore, the resistance of the tooth could be improved. The follow-up examinations demonstrated that the treatment of the invagination was successful because radiographs showed that the apical radiolucency had been reduced and the bone trabecular structure was visible.
The article reported a type III DI treated by removing the invagination combined with an MTA apical barrier. A follow-up of 26 mo revealed satisfactory healing of the soft tissue and bone of the periapical lesion, but the long-term efficacy still should be observed.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, research and experimental
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Dereci O, Muhvić-Urek M S-Editor: Dou Y L-Editor: Filipodia E-Editor: Zhang YL
| 1. | Shafer WG, Hine MK, Levy BM. A Text-Book of Oral Pathology. 4th ed. Philadelphia: W.B. Saunders Company, 1983: 2. |
| 2. | Hülsmann M. Dens invaginatus: aetiology, classification, prevalence, diagnosis, and treatment considerations. Int Endod J. 1997;30:79-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 94] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 3. | Alani A, Bishop K. Dens invaginatus. Part 1: classification, prevalence and aetiology. Int Endod J. 2008;41:1123-1136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 121] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 4. | OEHLERS FA. Dens invaginatus (dilated composite odontome). I. Variations of the invagination process and associated anterior crown forms. Oral Surg Oral Med Oral Pathol. 1957;10:1204-18 contd. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 283] [Cited by in RCA: 274] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 5. | Cakici F, Celikoglu M, Arslan H, Topcuoglu HS, Erdogan AS. Assessment of the prevalence and characteristics of dens invaginatus in a sample of Turkish Anatolian population. Med Oral Patol Oral Cir Bucal. 2010;15:e855-e858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 6. | Rotstein I, Stabholz A, Heling I, Friedman S. Clinical considerations in the treatment of dens invaginatus. Endod Dent Traumatol. 1987;3:249-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 56] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 7. | Jaramillo A, Fernández R, Villa P. Endodontic treatment of dens invaginatus: a 5-year follow-up. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101:e15-e21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 8. | Hovland EJ, Block RM. Nonrecognition and subsequent endodontic treatment of dens invaginatus. J Endod. 1977;3:360-362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 89] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 9. | Tsurumachi T, Hayashi M, Takeichi O. Non-surgical root canal treatment of dens invaginatus type 2 in a maxillary lateral incisor. Int Endod J. 2002;35:310-314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 10. | Nallapati S. Clinical management of a maxillary lateral incisor with vital pulp and type 3 dens invaginatus: a case report. J Endod. 2004;30:726-731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 33] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 11. | Ortiz P, Weisleder R, Villareal de Justus Y. Combined therapy in the treatment of dens invaginatus: case report. J Endod. 2004;30:672-674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Nedley MP, Powers GK. Intentional extraction and reimplantation of an immature invaginated central incisor. ASDC J Dent Child. 1997;64:417-420. [PubMed] |
| 13. | Girsch WJ, McClammy TV. Microscopic removal of dens invaginatus. J Endod. 2002;28:336-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 45] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 14. | Special Committee to Revise the Joint AAE/AAOMR Position Statement on use of CBCT in Endodontics. AAE and AAOMR Joint Position Statement: Use of Cone Beam Computed Tomography in Endodontics 2015 Update. Oral Surg Oral Med Oral Pathol Oral Radiol. 2015;120:508-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 194] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 15. | Scarfe WC, Farman AG, Sukovic P. Clinical applications of cone-beam computed tomography in dental practice. J Can Dent Assoc. 2006;72:75-80. [PubMed] |
| 16. | Yajima A, Otonari-Yamamoto M, Sano T, Hayakawa Y, Otonari T, Tanabe K, Wakoh M, Mizuta S, Yonezu H, Nakagawa K, Yajima Y. Cone-beam CT (CB Throne) applied to dentomaxillofacial region. Bull Tokyo Dent Coll. 2006;47:133-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 17. | Ziegler CM, Woertche R, Brief J, Hassfeld S. Clinical indications for digital volume tomography in oral and maxillofacial surgery. Dentomaxillofac Radiol. 2002;31:126-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 51] [Reference Citation Analysis (0)] |
| 18. | Baratto Filho F, Zaitter S, Haragushiku GA, de Campos EA, Abuabara A, Correr GM. Analysis of the internal anatomy of maxillary first molars by using different methods. J Endod. 2009;35:337-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 134] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 19. | Zafersoy-Akarslan Z, Erten H, Uzun O, Semiz M. Reproducibility and agreement of clinical diagnosis of occlusal caries using unaided visual examination and operating microscope. J Can Dent Assoc. 2009;75:455. [PubMed] |
| 20. | Carr GB, Murgel CA. The use of the operating microscope in endodontics. Dent Clin North Am. 2010;54:191-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 60] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 21. | Torabinejad M, Parirokh M, Dummer PMH. Mineral trioxide aggregate and other bioactive endodontic cements: an updated overview - part II: other clinical applications and complications. Int Endod J. 2018;51:284-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 239] [Article Influence: 29.9] [Reference Citation Analysis (0)] |
| 22. | Mente J, Leo M, Panagidis D, Ohle M, Schneider S, Lorenzo Bermejo J, Pfefferle T. Treatment outcome of mineral trioxide aggregate in open apex teeth. J Endod. 2013;39:20-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 63] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 23. | Pace R, Giuliani V, Nieri M, Di Nasso L, Pagavino G. Mineral trioxide aggregate as apical plug in teeth with necrotic pulp and immature apices: a 10-year case series. J Endod. 2014;40:1250-1254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 43] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 24. | Bonte E, Beslot A, Boukpessi T, Lasfargues JJ. MTA versus Ca(OH)2 in apexification of non-vital immature permanent teeth: a randomized clinical trial comparison. Clin Oral Investig. 2015;19:1381-1388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 57] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 25. | Moore A, Howley MF, O'Connell AC. Treatment of open apex teeth using two types of white mineral trioxide aggregate after initial dressing with calcium hydroxide in children. Dent Traumatol. 2011;27:166-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 59] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 26. | Annamalai S, Mungara J. Efficacy of mineral trioxide aggregate as an apical plug in non-vital young permanent teeth: preliminary results. J Clin Pediatr Dent. 2010;35:149-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 27. | Moretton TR, Brown CE, Legan JJ, Kafrawy AH. Tissue reactions after subcutaneous and intraosseous implantation of mineral trioxide aggregate and ethoxybenzoic acid cement. J Biomed Mater Res. 2000;52:528-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 28. | Enkel B, Dupas C, Armengol V, Akpe Adou J, Bosco J, Daculsi G, Jean A, Laboux O, LeGeros RZ, Weiss P. Bioactive materials in endodontics. Expert Rev Med Devices. 2008;5:475-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 36] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 29. | Islam I, Chng HK, Yap AU. X-ray diffraction analysis of mineral trioxide aggregate and Portland cement. Int Endod J. 2006;39:220-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 64] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 30. | Gancedo-Caravia L, Garcia-Barbero E. Influence of humidity and setting time on the push-out strength of mineral trioxide aggregate obturations. J Endod. 2006;32:894-896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 79] [Article Influence: 4.2] [Reference Citation Analysis (0)] |