Published online Mar 6, 2024. doi: 10.12998/wjcc.v12.i7.1346
Peer-review started: December 12, 2023
First decision: December 31, 2023
Revised: January 9, 2024
Accepted: February 6, 2024
Article in press: February 6, 2024
Published online: March 6, 2024
Processing time: 79 Days and 22.8 Hours
Radicular cyst is a lesion of odontogenic origin that arises from epithelial remains due to periapical periodontitis caused by inflammatory reactions generated at the apex of affected teeth with infected or necrotic pulps. The therapeutic mana
A 68-year-old patient presented with a radicular cyst from an upper lateral incisor. The microsurgical management used was aimed at enucleating the chemical membrane, performing apicoectomy of the tooth along with careful and precise retrograde filling, and implementing GTR technique using a resorbable collagen membrane and bovine xenograft. The diagnosis of radicular cyst was confirmed using histopathological analysis. The patient underwent follow-up evaluations at 10 and 30 d postoperatively. At 4 months postoperative evaluation, she remained asymptomatic, and radiographs showed significant periapical healing with adequate bone formation.
These results suggest that microsurgical management using the GTR technique with collagen membrane and xenograft, contributes to bone regeneration.
Core Tip: The radicular cyst is a lesion of odontogenic origin whose therapeutic management is controversial. However, the present clinical case describes the management of a radicular cyst using a microsurgical approach, and the performance of an apicoectomy of the tooth associated with the entity, followed by the guided tissue regeneration technique, implementation of a resorbable type I bovine collagen membrane, and bovine xenograft. The diagnosis was based on clinical findings that were confirmed with histopathology analysis. This case underlines the importance of clinical, periapical radiographic, and cone-beam computed tomography evaluation as guides for an accurate diagnosis in order to enhance the development of an appropriate treatment plan for a radicular cyst.
- Citation: Gómez Mireles JC, Martínez Carrillo EK, Alcalá Barbosa K, Gutiérrez Cortés E, González Ramos J, González Gómez LA, Bayardo González RA, Lomelí Martínez SM. Microsurgical management of radicular cyst using guided tissue regeneration technique: A case report. World J Clin Cases 2024; 12(7): 1346-1355
- URL: https://www.wjgnet.com/2307-8960/full/v12/i7/1346.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v12.i7.1346
Radicular cysts, also known as apical periodontal cysts, are the most common inflammatory odontogenic cysts of the jaw[1]. They are chronic lesions arising from pre-existing periapical granulomas triggered by inflammatory reactions generated in the apex of the affected teeth due to infected or necrotic pulps, although they may also be located on the lateral surfaces of roots associated with accessory canals[2]. Generally, radicular cysts have a predilection for the male gender, and they occur between the third and sixth decades of life[1]. A radicular cyst is clinically characterized by a slight increase in volume that is soft and fluctuating during palpation[1]. The lesion may present as a hard and irregular bony swelling[3]. It is usually asymptomatic, and sometimes presents mild pain and pain on percussion[4]. Extra-orally, an oval, diffuse, painless swelling is observed in the affected area. Radicular cysts are often slow-growing, and may cause sensitivity, mobility, root resorption and displacement of the affected teeth. When the cystic entity is infected, it can also cause pain and inflammation[1].
The treatment of radicular cyst is aimed at eradicating the lesion and enhancing the healing of the periapical tissue so as to improve the outcome. The modality chosen is usually the one that grants the lowest possible risk of recurrence and minimum morbidity. Some of the factors that determine the treatment modality for radicular cysts are the origin and extension of the lesion, its relationship with important anatomical structures, clinical characteristics, and patient collaboration, amongst others[5,6]. When the lesions are small, the first line of treatment is endodontic therapy which is a conservative method. In contrast, marsupialization and enucleation have been used as surgical treatment methods of choice when the cysts are large and/or there are invariable changes in the peri-radicular tissue that cannot be treated with non-surgical endodontic procedure[4,5]. Moreover, different conventional or microsurgical endodontic and periodontal surgical techniques such as apicectomy and guided tissue regeneration (GTR) have been described for complementing the enucleation treatment. However, despite the different modalities and techniques, the management of radicular cysts is controversial. The present clinical case describes the management of a radicular cyst using a microsurgical approach, and the performance of an apicectomy of the tooth associated with the entity, followed by GTR technique, implementation of a resorbable membrane of type I bovine collagen, and bovine xenograft.
A 68-year-old female patient without systemic engagement, was referred by a general dentist to a private practice with finding of a radiolucent zone in the periapical area of the dental organ (OD) 22 and displacement of the adjacent roots.
The patient mentioned having received endodontic treatment in the previous 6 years, without clinical or radiographic follow-up.
The patient did not mention presence of any systemic disease, and she was not undergoing any medical treatment.
Mother was diagnosed with high blood pressure in 2017.
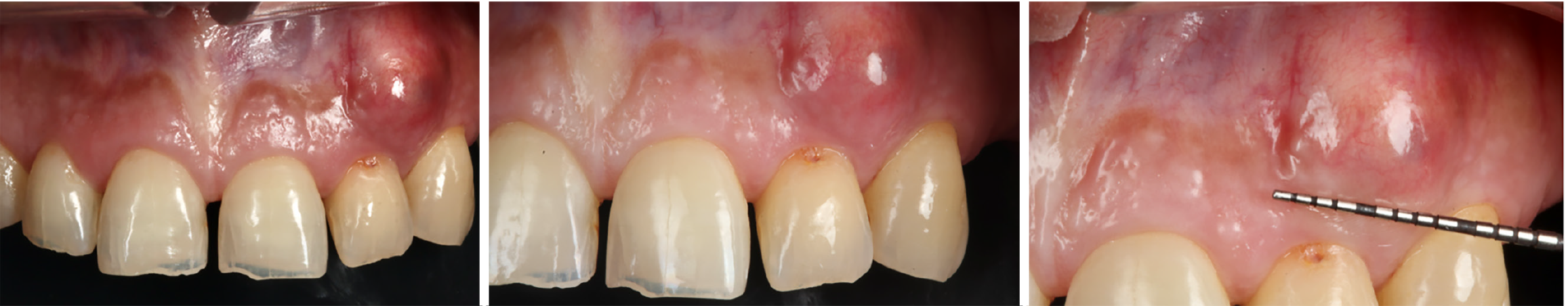
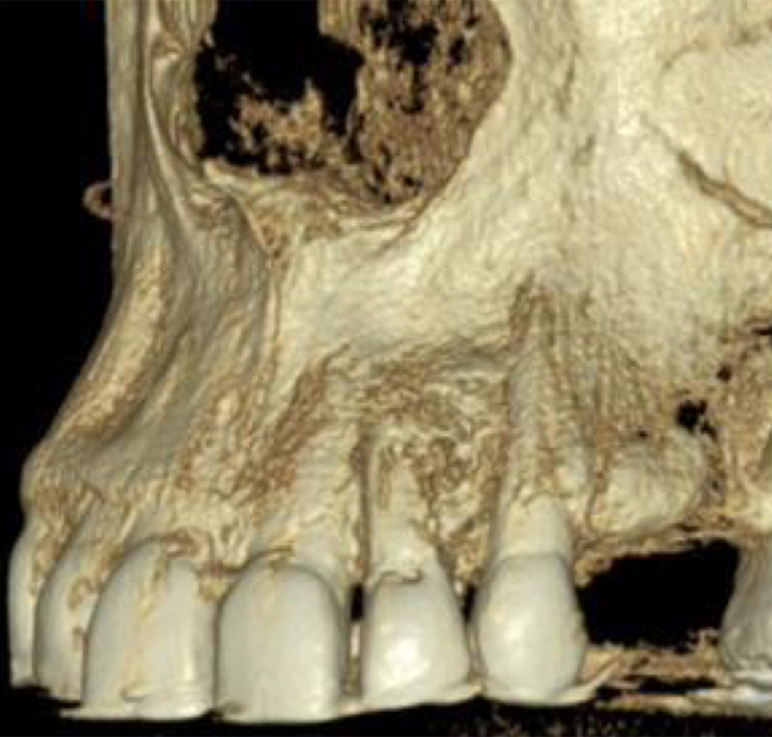
During the clinical examination, a bulge was identified in the vestibular area corresponding to OD 22, with a soft, painless consistency (Figure 1). This OD did not present grade of mobility, supra- and sub-gingival calculus or probing depth.
Hemoglobin, neutrophil and platelet levels were within normal limits.
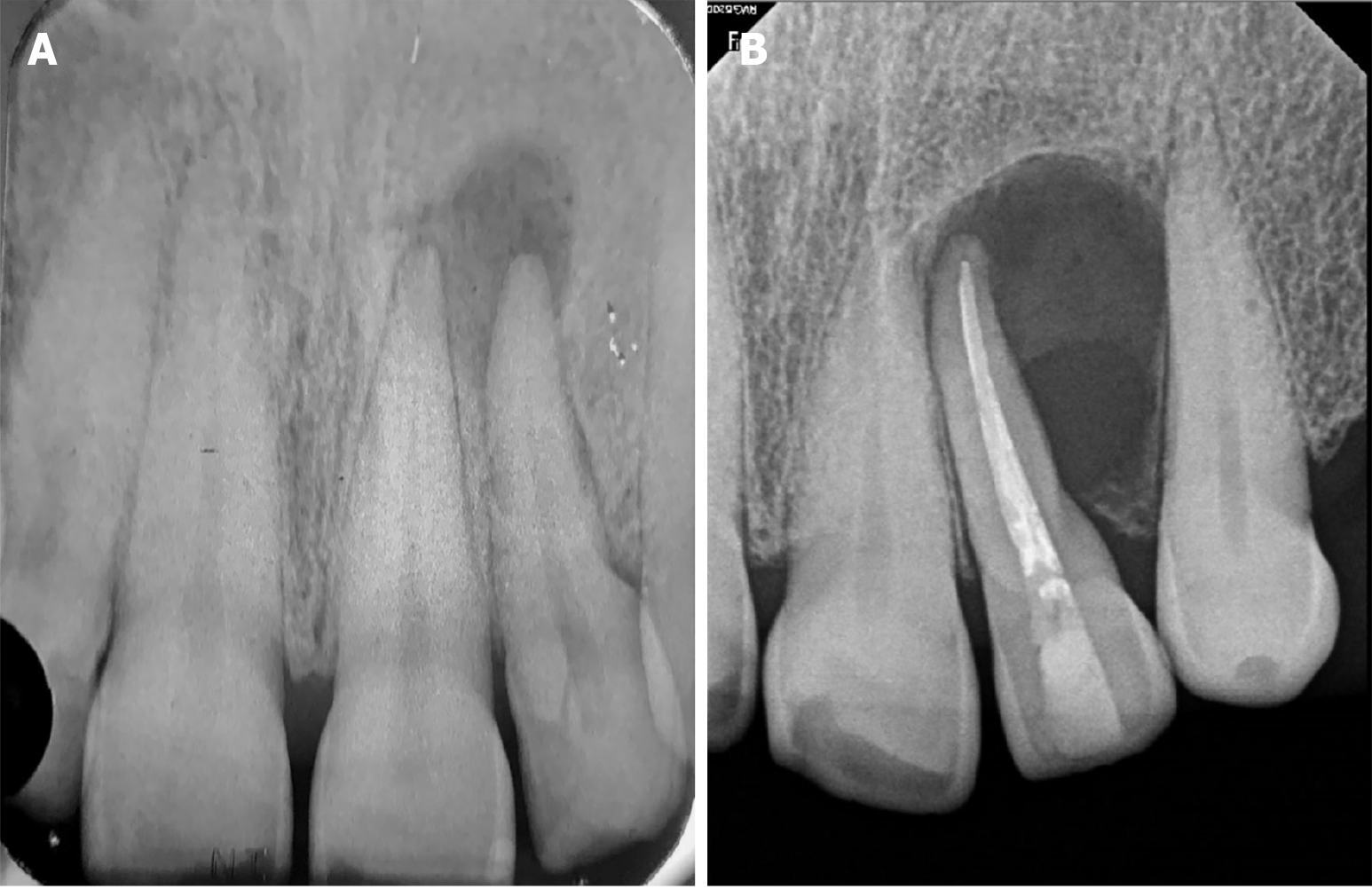
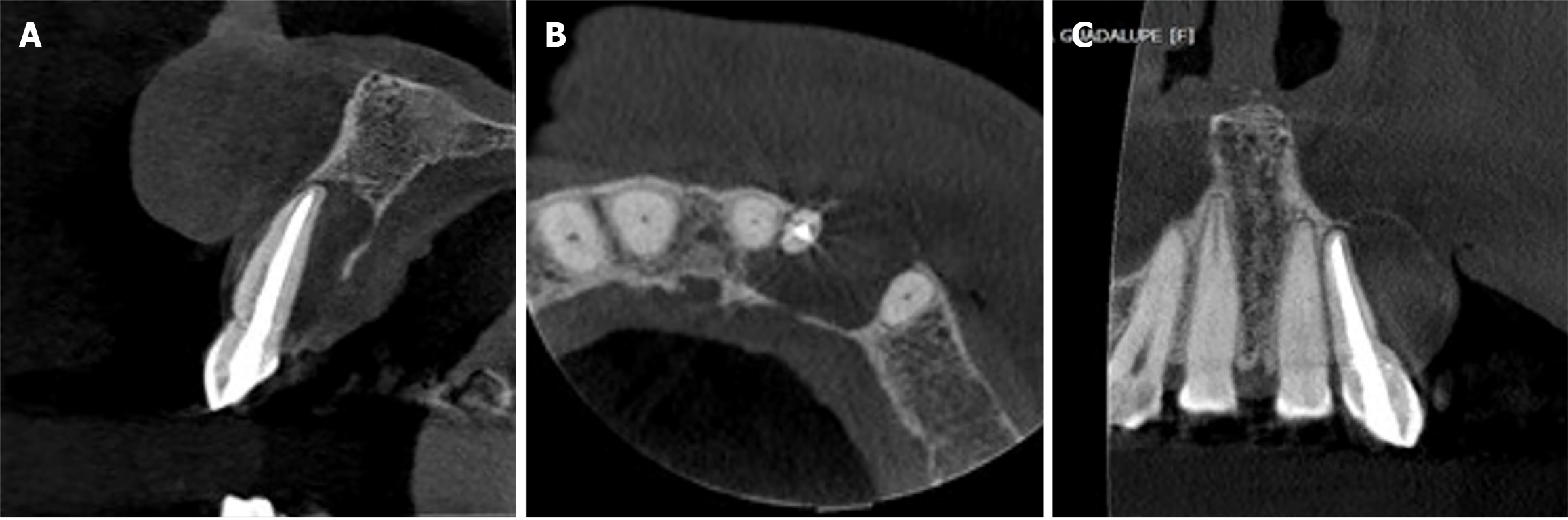
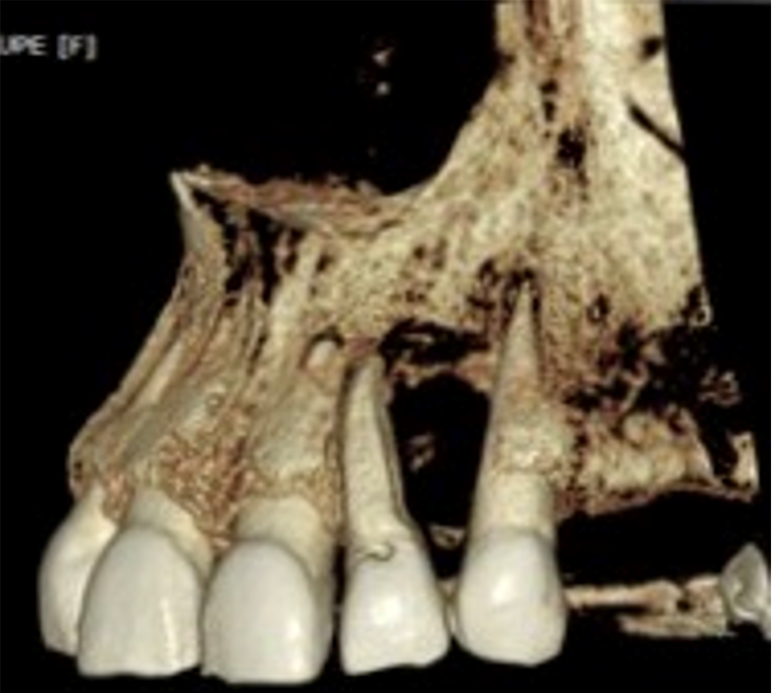
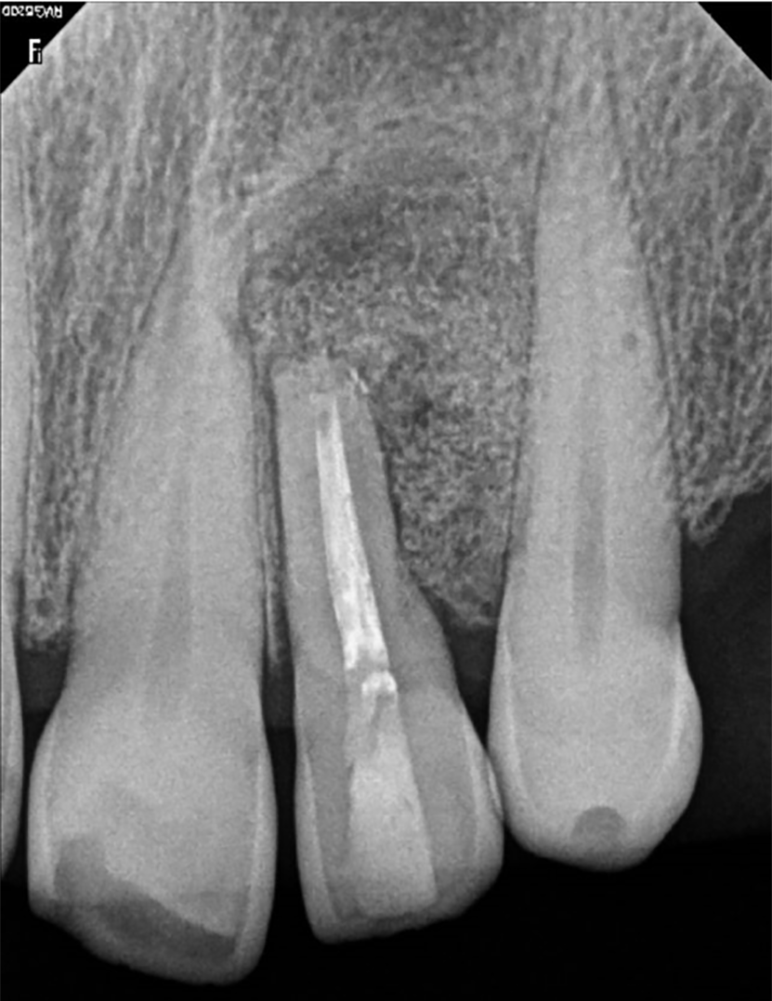
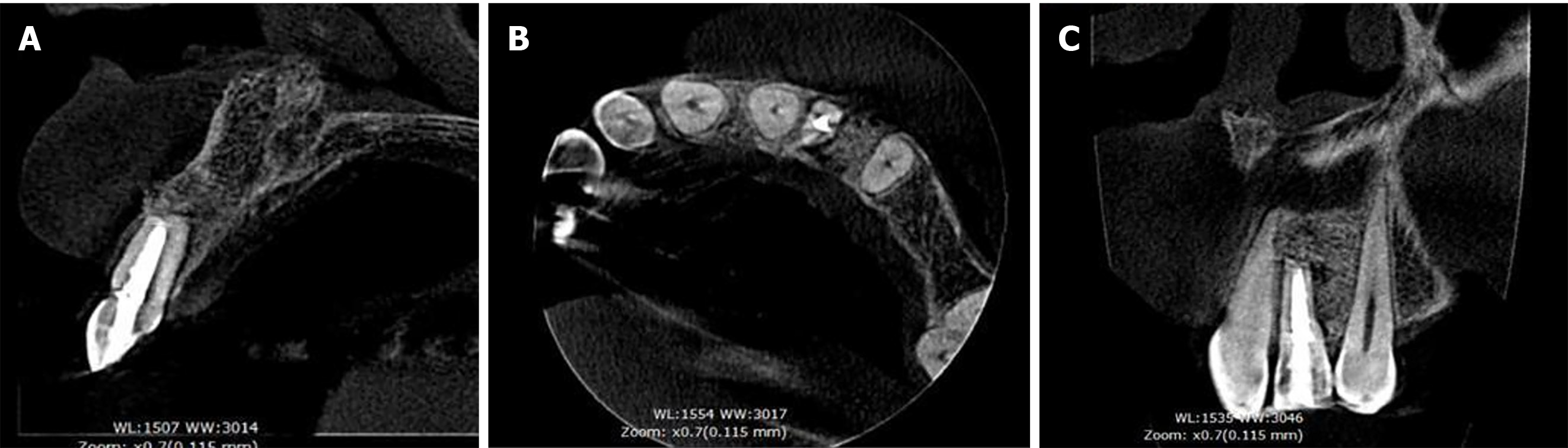
Periapical radiograph revealed a well-defined, completely asymptomatic and circumferential radiolucent area (Figure 2). Evaluation using computed tomography revealed the absence of vestibular cortex, with the dimensions of the loss being 6 mm wide × 16 mm high and 5.5 mm deep (Figure 3). In the three-dimensional reconstruction, the extent and extension of the lesion can be observed. This evaluation was performed with the OnDemand computed tomography Viewer (Figure 4).
After the evaluation, analysis and interdisciplinary discussion were carried out with specialists in endodontics, periodontics and oral pathology, with respect to the possible treatment options. However, based on the patient's clinical history and her detachment from treatment, as evidenced by not attending any dental consultation for 6 years, including the recommended radiographic follow-up after root canal treatment, a micro-surgical approach was decided upon. This approach would enucleate the radicular cyst and carry out GTR to improve the treatment prognosis with the least possible trauma to the soft tissues. The treatment of choice and the associated risks were thoroughly explained to the patient, and she gave signed informed consent. Surgical enucleation was the chosen treatment, as well as apicoectomy of the piece involved, followed finally with GTR.
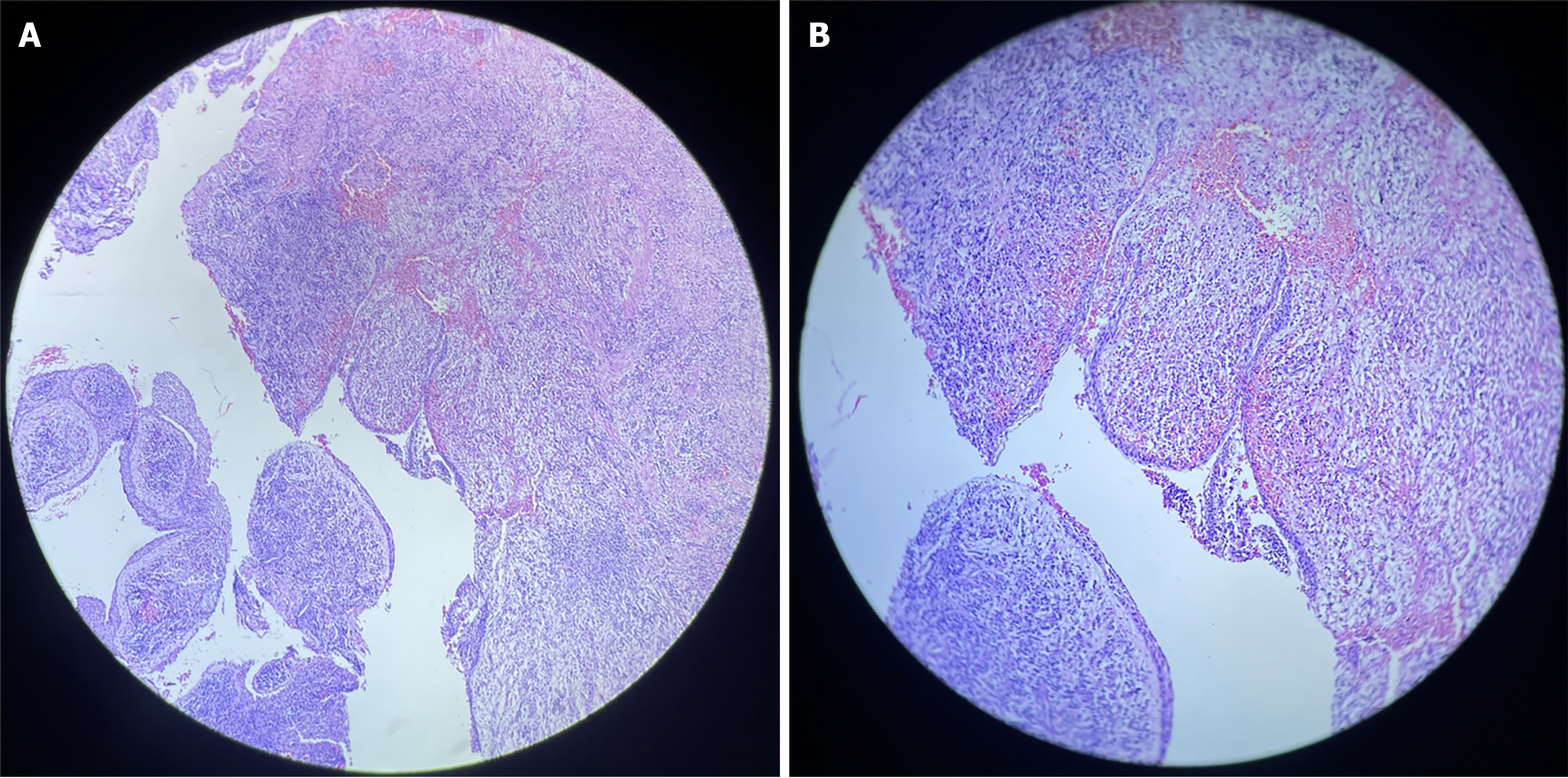
The results from histological analysis revealed a cavity lined by non-keratinized odontogenic epithelium with intracapsular projections and acanthosis. Moreover, intense mixed inflammatory infiltrates and foamy macrophages were present in the fibrous capsule, in addition to erythrocyte extravasation. These features were consistent with diagnosis of radicular cyst (Figure 5).
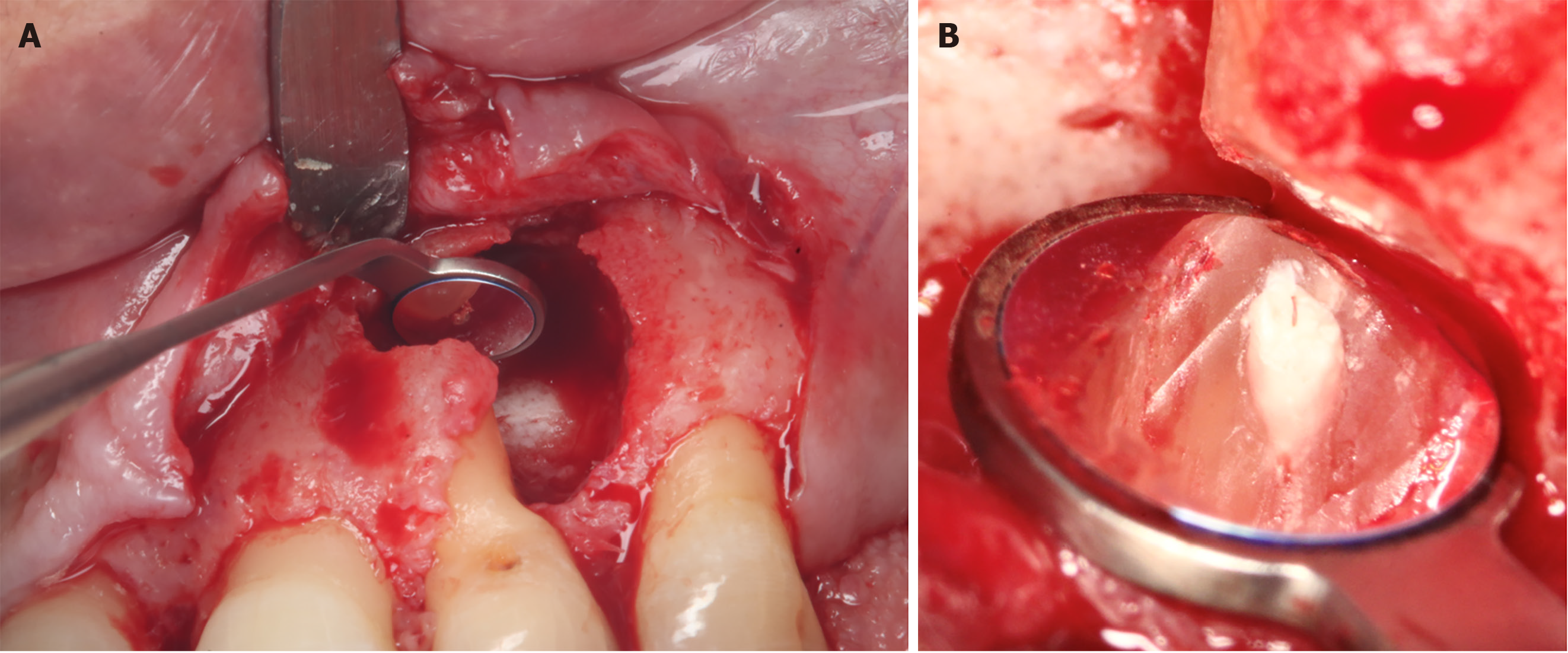
The patient was scheduled for the surgery, during which specialists in the three previously mentioned specialties were involved. The procedure was performed under the magnification of a Zeiss Extaro 300 clinical microscope. The area was anesthetized with articaine: Epinephrine bi-tartrate (1:100000). Following anesthesia, a time lag of 20 min was allowed for adequate vasoconstriction. Then, initial intra-surgical incisions and a vertical incision were made in order to have a clear view of the lesion. A full-thickness flap was raised to expose the lesion which was carefully dissected completely using curettes. The lesion was placed in a container with 40% formalin (universal percentage for fixing histopathological samples). Following enucleation, the cavity was examined, washed and enlarged by means of a high-speed electric piece in order to have better control of the instruments during the apicoectomy. Using methylene blue, the root was inspected for possible fractures. The root apex was cut with an endo zekrya bur in a 45º turbine at 3 mm coronal to the apex, and the apex was again inspected with the help of magnification and micromirrors to locate any microfractures in the root canal. Retro-instrumentation was performed with diamond ultrasound tips. The cavity was dried and inspected for the presence of internal debris. Then, biodentine (Septodont) retro-preparation was placed in the canal (Figure 6). The excess was removed with the help of a carrier and burnisher, and using indirect vision, effective sealing of the canal was confirmed. Once the apicoectomy was completed, it was washed with cold saline, and a xenograft (Biooss by Geistlish) previously hydrated with physiological saline was used to fill the cavity. Then, a Bio-Gide® collagen membrane (Geistlisch) was placed and adapted over the bone window (Figure 7). The flap was repositioned, making sure that the closure was first intention and tension-free (Figure 8A). The suture chosen was 6-0 polypropylene due to ease of handling and excellent biocompatibility. Conventional post-operative care was used. The antibiotic used was Amoxicillin (750 mg; 1 tablet every 12 h), while the analgesics were sodium naproxen 275 mg: Paracetamol 300 mg (2 tablets every 8 h). Additionally, 0.20% chlorhexidine gel was applied in the area for 2 wk. Ice application in the area was done for the first 72 h.
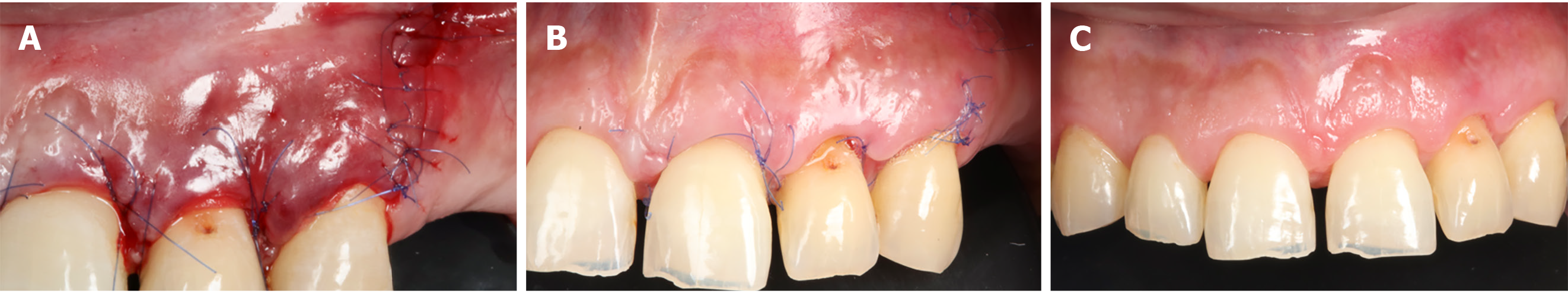
At the 4-month follow-up, gingival clinical examination revealed that the affected area which was subjected to GTR recovered its normal volume and color (Figure 8B and C). The periapical radiograph demonstrated total bone filling in the area adjacent to the dental piece up to the apicoectomy level of the tooth root. Two-dimensional (2D) radiographic healing was judged incomplete by two independent reviewers and calibrated according to the Molven criteria. Tomographic evaluation showed complete healing in the area, with beginnings of vestibular cortical formation (Figures 9 and 10).
The radicular cyst represents 55% of odontogenic cysts, and 52%-68% of all cysts of the jaws[7]. This chronic entity is the result of multiple inflammatory reactions initially guided by various proinflammatory interleukins and growth factors which cause proliferation of epithelial cells derived from epithelial rests of Malassezz in granulomatous tissue, followed cyst formation and growth[2,8]. However, since events associated with the molecular biology of the radicular cyst are still unclear, there is need for more research in this area[2]. The therapeutic management of radicular cysts is controversial. To our knowledge, in the literature, there is only one case report of enucleation of a radicular cyst managed with microsurgery and apicoectomy, but without the use of the GTR technique in the same surgical procedure. The present clinical case describes the management of a radicular cyst with microsurgical approach, performance of an apicoectomy of the tooth associated with the entity, application of GTR technique, use of a resorbable membrane of type I bovine collagen, and bovine xenograft.
In recent years, the incorporation of magnification into dental practice has resulted in improved visual acuity, ergonomic benefits, decreased patient morbidity, rapid healing, and increased patient acceptance[4,9]. Endodontic microsurgery becomes the recommended procedure when initial root canal therapy or subsequent retreatments fail to prevent or heal apical periodontitis, and the lesion near the tooth's apex remains unrepaired[4]. In the meta-analysis of Setzer et al[10], the probability of success of conventional endodontic surgery vs microsurgery, and the probability of success for the 2 techniques were compared. The authors concluded that the success rates of conventional surgeries and microsurgeries were 59% and 94%, respectively[11]. In a review article, it was reported that endodontic microsurgery had an overall success rate of 91.5% at 5 years, and 93.3% at 10 years. These data indicate that the microsurgical approach has resulted in a significant increase in the success rate of endodontic surgeries, since this technique is minimally invasive, and it allows for greater precision. In the present case, the apicoectomy with retrodental filling through a microsurgical approach using biodentine was carried out with the aim of removing the periapical lesion and generating an environment conducive to healing in both hard and soft tissues. The use of biodentine for retrograde obturation after apicoectomy has been described by other authors[12,13]. Biodentine is a biologically active cement based on calcium silicate. It has a variety of dental applications due to its physicochemical characteristics, biocompatibility and ease of handling which account for its current wide use, particularly in the endodontic area[14,15]. For several years, Mineral Trioxide Aggregate (MTA) was considered the first treatment option in many surgical and non-surgical dental procedures such as root perforations, direct pulp capping, apexification processes, periapical lesions, retrograde obturation, and filling/temporary padding. This ceramic material possesses biocompatibility and sealing capacity, and it is not adversely affected by contact with blood and peri-radicular fluids. However, it has the disadvantage of long setting time, high cost and difficulty in handling[11]. To compensate for the deficiencies of MTA, biodentine gained prominence due to its excellent sealing capacity. It does not shrink or reabsorb, and it hardens quickly and is easy to handle. Biodentine has several advantages over MTA, such as greater antibacterial and antifungal activity, greater adhesion strength, and greater biocompatibility[12,14,15]. Due to these benefits, biodentine is an excellent option for use as a retro-filling material.
Reports have shown that GTR has been implemented as an adjunct to radicular cyst enucleation and apicoectomy for enhancement of periapical tissue healing, prevention of soft tissue collapse, and improvement of treatment outcomes[11,16,17]. Full recovery of the area around the apex of a tooth includes the regeneration of alveolar bone, periodontal ligament cells and cementum. However, within the bone defect, the surrounding connective tissues may grow, thereby preventing bone healing. GTR is a technique that uses a bone substitute material along with a membrane that serves both as a biological and physical shield to prevent cells that do not contribute to bone growth, such as epithelial cells, from invading the area. Moreover, GTR allows the migration of bone-forming cells from slow migration to bad sites, thereby ensuring selective cell re-population and guiding the proliferation of various tissues during the healing process[17,18]. Numerous studies have described this technique, with respect to the application of a wide range of materials and treatment protocols, all of which achieved clinical effectiveness. Zubizarreta-Macho et al[16] have described a meta-analysis of clinical evidence on the efficacy of the GTR technique in bone defects after endodontic surgical treatment[16]. These authors highlighted the importance of membranes and demonstrated that employing a membrane by itself, without the addition of a bone graft, was 1.02 times more effective compared to endodontic surgery without the aid of GTR techniques. It was also found to surpass the efficacy of methods involving platelet-rich plasma. Nonetheless, the synergy of both membrane and bone grafting proved to be substantially more advantageous, achieving a success rate that was 3.6 times higher than using the membrane in isolation. On the other hand, the success rate of the combination of membrane and bone graft was 3.7 times higher than that of endodontic surgery alone (control). It was concluded in the meta-analysis that GTR techniques in which bone grafts and membranes were used as complements to surgical endodontic treatment, promoted complete healing of the periapical bone with enhanced success rate, and improved the prognosis of endodontic surgery. The determinants of the success of GTR are the type of bone regenerative biomaterial and the barriers applied. In a systematic review and meta-analysis, Sumangali et al[19] analyzed the success of GTR in periapical surgeries based on clinical, histological, and radiographic evaluations with different materials, and reported that the use of xenograft in combination with collagen membrane produced a superior result. In the present case, in order to improve the healing of the periapical tissue and avoid tissue collapse due to bone defect, a resorbable type I bovine collagen membrane was used to cover the particulate bovine xenograft after the enucleation of the cyst and the apicoectomy. The membrane is 100% natural and multi-porous. The GTR was performed by Morillo et al[20] using cortico-cancellous bone substitute and fibrin-rich plasma (PRF) membrane, after the removal of a radicular cyst[20]. The latter is a natural blood-derived structure associated with the immune system and platelet concentrate that exhibits several vital properties such as cell attachment, cell migration, cell proliferation, and cell differentiation. Additionally, it contains multiple growth factor-like platelet-derived growth factor, transforming growth factor β1, insulin-like growth factor, and vascular endothelial growth factor, all of which enhance stimulation of the healing of surgical wounds in big bones[17]. The PRF was also implemented as a post-enucleation membrane of a radicular cyst by Govindaraju et al[6], but without the addition of bone filler biomaterials[6]. Despite the multiple and beneficial properties of PRF, its main disadvantage is that it resorbs within approximately 7-28 d, which is substantially shorter than the 4-6 wk required for most periodontal regeneration applications[17], and also shorter than the 16 to 24 wk required for bone enhancement[18]. Furthermore, its capacity to maintain space is compromised by its high rate of reabsorption[17]. In the present study, the use of PRF membrane was ruled out in favor of a collagen membrane. Tseng et al[21] have reported that after enucleation of radicular cyst with apicoectomy and retrograde obturation of the apical root canal, micro-macroporous biphasic calcium phosphate bio-ceramic was placed in the bone defect. However, they did not use any membrane. On the other hand, there are reports of clinical cases in which no biomaterials were used for bone regeneration after the enucleation of the radicular cyst[4,7,22,23]. For instance, Elhakim et al[4] made the complete enucleation of a periapical cyst without sacrificing the vitality of the adjacent teeth, and used no bone filler material or membrane: They argued that the bone graft material might have compromised the integrity of the neurovascular bundles of vital teeth and limited their repair potential.
The microsurgical approach with GTR is associated with better illumination and enlargement of the surgical field, thereby providing adequate access to the intraosseous defect and debridement thereof with greater precision and minimal trauma[4,9,24]. This approach allows for obtaining and maintaining primary closure at the edges of the wound, and minimizing bacterial contamination, thereby ensuring favorable conditions for regeneration[4]. Likewise, this microsurgical modality is characterized by minimal marginal tissue recession and very limited intraoperative and postoperative morbidity[4,9,25]. Due to these fundamental factors, the microsurgical approach for the treatment of intra-bony defects has been proposed by several authors[26]. In the present case, the magnification allowed for a greater precision in the management of the enucleation of the lesion, as well as primary closure without tension, thereby favoring an adequate evolution of the wound with minimal discomfort. Additionally, at 6 months of follow-up in the clinical and radiographic evaluation, successful results were observed which were attributed to adequate surgical management and the use of correct regeneration biomaterials.
Finally, it is important to highlight that the management of radicular cysts requires a multidisciplinary approach that involves several dental specialists, in order to obtain accurate diagnosis and safe and effective treatment plans. Regular follow-up of endodontically treated teeth is essential for verification of apical healing, and if a periapical lesion develops, it is identified at an early stage. Early detection and accurate diagnosis are essential for adequate treatment and prevention of possible complications.
The microsurgical management of a radicular cyst using the GTR technique, resorbable membrane of type I bovine collagen and bovine xenograft after apicoectomy of the associated tooth with retrograde biodentine seal, allowed for successful removal of the periapical lesion and generated an environment conducive to healing in both hard and soft tissues. The successful outcome of this case confirms the efficacy of microsurgical management in GTR using correctly-chosen biomaterials for the enucleation of the radicular cyst. Therefore, dentists should be aware of the potential benefits of these biomaterials that may be used to provide quality treatments for their patients.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Medicine, research and experimental
Country/Territory of origin: Mexico
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Ghannam WM, Egypt; He CY, China S-Editor: Liu H L-Editor: A P-Editor: Li X
| 1. | Deshmukh J, Shrivastava R, Bharath KP, Mallikarjuna R. Giant radicular cyst of the maxilla. BMJ Case Rep. 2014;2014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 2. | Rios Osorio N, Caviedes-Bucheli J, Mosquera-Guevara L, Adames-Martinez JS, Gomez-Pinto D, Jimenez-Jimenez K, Avendano Maz H, Bornacelly-Mendoza S. The Paradigm of the Inflammatory Radicular Cyst: Biological Aspects to be Considered. Eur Endod J. 2023;8:20-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 3. | Shetty S, Angadi PV, Rekha K. Radicular cyst in deciduous maxillary molars: a rarity. Head Neck Pathol. 2010;4:27-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 4. | Elhakim A, Kim S, Kim E, Elshazli AH. Preserving the vitality of teeth adjacent to a large radicular cyst in periapical microsurgery: a case report with 4-year follow-up. BMC Oral Health. 2021;21:382. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 5. | Vega Llauradó A, Ayuso Montero R, Teixidor Olmo I, Salas Enric J, Marí Roig A, López López J. Opcionesterapéuticas en quistes odontogénicos. Revisión. Av Odontoestomatol. 2013;29:81-93. |
| 6. | Govindaraju L, Antony DP, S P. Surgical Management of Radicular Cyst With the Application of a Natural Platelet Concentrate: A Case Report. Cureus. 2023;15:e33992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 7. | Noda A, Abe M, Shinozaki-Ushiku A, Ohata Y, Zong L, Abe T, Hoshi K. A Bilocular Radicular Cyst in the Mandible with Tooth Structure Components Inside. Case Rep Dent. 2019;2019:6245808. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 8. | Lin LM, Huang GT, Rosenberg PA. Proliferation of epithelial cell rests, formation of apical cysts, and regression of apical cysts after periapical wound healing. J Endod. 2007;33:908-916. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 113] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 9. | Yadav VS, Salaria SK, Bhatia A, Yadav R. Periodontal microsurgery: Reaching new heights of precision. J Indian Soc Periodontol. 2018;22:5-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 10. | Setzer FC, Shah SB, Kohli MR, Karabucak B, Kim S. Outcome of endodontic surgery: a meta-analysis of the literature--part 1: Comparison of traditional root-end surgery and endodontic microsurgery. J Endod. 2010;36:1757-1765. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 279] [Cited by in RCA: 208] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 11. | Montero-Miralles P, Ibáñez-Barranco R, Cabanillas-Balsera D, Areal-Quecuty V, Sánchez-Domínguez B, Martín-González J, Segura-Egea JJ, Jiménez-Sánchez MC. Biomaterials in periapical regeneration after microsurgical endodontics: A narrative review. J Clin Exp Dent. 2021;13:e935-e940. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Barzuna M, Cárdenas T. Use of a bioceramic (BiodentineTM) in retrodental filling in apicoectomy report of a clinical case. Vital Dent. 2014;20:69-74. |
| 13. | Alvarez HA, Santillana CH, Portocarrero JP. Quiste odontogénico inflamatorio: Reporte de caso. Salud Vida Sipanense. 2020;132-143. [DOI] [Full Text] |
| 14. | Malkondu Ö, Karapinar Kazandağ M, Kazazoğlu E. A review on biodentine, a contemporary dentine replacement and repair material. Biomed Res Int. 2014;2014:160951. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 162] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 15. | Rajasekharan S, Martens LC, Cauwels RGEC, Anthonappa RP. Biodentine™ material characteristics and clinical applications: a 3 years literature review and update. Eur Arch Paediatr Dent. 2018;19:1-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 63] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 16. | Zubizarreta-Macho Á, Tosin R, Tosin F, Velasco Bohórquez P, San Hipólito Marín L, Montiel-Company JM, Mena-Álvarez J, Hernández Montero S. Influence of Guided Tissue Regeneration Techniques on the Success Rate of Healing of Surgical Endodontic Treatment: A Systematic Review and Network Meta-Analysis. J Clin Med. 2022;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 17. | Agrawal AA. Platelet rich fibrin is not a barrier membrane! Or is it? World J Clin Cases. 2023;11:2396-2404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 18. | Ren Y, Fan L, Alkildani S, Liu L, Emmert S, Najman S, Rimashevskiy D, Schnettler R, Jung O, Xiong X, Barbeck M. Barrier Membranes for Guided Bone Regeneration (GBR): A Focus on Recent Advances in Collagen Membranes. Int J Mol Sci. 2022;23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 91] [Reference Citation Analysis (0)] |
| 19. | Sumangali A, Naik AC, Mohan N, Gautam N, Abrol S, Mustafa M, Tiwari H. Bone Regenerative Biomaterials in Periapical Surgery: A Systemic Review and Meta-Analysis. J Pharm Bioallied Sci. 2021;13:S933-S937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 20. | Morillo ML, Peralta HV, Vásquez VM, Guzmán DJ, Harris RJ. Guided bone regeneration in the treatment of radicular cyst: Case report. Den J. 2023;26:99-103. [DOI] [Full Text] |
| 21. | Tseng SK, Tsai YL, Li UM, Jeng JH. Radicular cyst with actinomycotic infection in an upper anterior tooth. J Formos Med Assoc. 2009;108:808-813. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 22. | Molgatini S, Rey E, Basilaki J, Mosca C, Galante R, Gliosca L. [Isolation of Pseudomonas stutzeri from an odontogenic inflammatory cyst: Diagnostic relevance]. Rev Argent Microbiol. 2017;49:32-38. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 23. | Nilesh K, Dadhich A. Unusually large radicular cyst presenting in the maxillary sinus. BMJ Case Rep. 2020;13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 24. | Cortellini P, Tonetti MS. Minimally invasive surgical technique and enamel matrix derivative in intra-bony defects. I: Clinical outcomes and morbidity. J Clin Periodontol. 2007;34:1082-1088. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 61] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 25. | Ribeiro FV, Nociti Júnior FH, Sallum EA, Sallum AW, Casati MZ. Use of enamel matrix protein derivative with minimally invasive surgical approach in intra-bony periodontal defects: clinical and patient-centered outcomes. Braz Dent J. 2010;21:60-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 26. | Aimetti M, Ferrarotti F, Mariani GM, Romano F. A novel flapless approach versus minimally invasive surgery in periodontal regeneration with enamel matrix derivative proteins: a 24-month randomized controlled clinical trial. Clin Oral Investig. 2017;21:327-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 45] [Article Influence: 5.0] [Reference Citation Analysis (0)] |