Published online Aug 28, 2025. doi: 10.4329/wjr.v17.i8.110868
Revised: July 16, 2025
Accepted: August 8, 2025
Published online: August 28, 2025
Processing time: 72 Days and 5.4 Hours
Signet ring cell carcinoma originating from the appendix is extremely rare, and the lack of specific clinical symptoms and imaging features makes preoperative diagnosis particularly challenging.
We report a case of a 49-year-old woman who presented with irregular vaginal bleeding lasting more than five months. Computed tomography (CT) and mag
Imaging showed appendiceal wall thickening and marked thickening of the uterine myometrium, with lesions demonstrating progressive enhancement after contrast administration. These findings should raise suspicion for the implan
Core Tip: Signet ring cell carcinoma originating from the appendix is relatively rare, and its imaging features are seldom reported, making preoperative diagnosis extremely challenging. This study retrospectively analyzes a case of Signet ring cell carcinoma originating from the appendix and implantation to the entire uterus. The imaging characteristics of both the appendix and the uterus were analyzed using computed tomography and magnetic resonance imaging, with the aim of enhancing awareness of this disease.
- Citation: Liu JM, Li Z, Qi LH, Chu BL, Deng ZX, Tang FY. Imaging features of appendiceal signet ring cell carcinoma with uterine implantation: A case report. World J Radiol 2025; 17(8): 110868
- URL: https://www.wjgnet.com/1949-8470/full/v17/i8/110868.htm
- DOI: https://dx.doi.org/10.4329/wjr.v17.i8.110868
Signet ring cell carcinoma is a distinct clinical and pathological subtype of colorectal cancer, characterized by aggressive behavior and a low incidence of approximately 0.6%-2.3%[1–6]. Signet ring cell carcinoma originating from the appendix is even rarer, accounting for about 16% of all Signet ring cell carcinoma cases[6]. Approximately 93% of appendiceal signet ring cell carcinomas have metastasized to adjacent organs, lymph nodes, or the peritoneal cavity at diagnosis[6]. Early detection and intervention are therefore essential for improving patient outcomes[7]. Accurate preoperative diagnosis remains challenging because of nonspecific clinical symptoms and a lack of characteristic imaging findings. This report presents a rare case of appendiceal Signet ring cell carcinoma with implantation to the entire uterus, emphasizing its computed tomography (CT) and magnetic resonance imaging (MRI) characteristics to improve disease recognition.
A 49-year-old woman presented with more than 5 months of irregular vaginal bleeding.
Her last menstrual period occurred on April 15, 2023, and was initially accompanied by heavy bleeding. She sought care at a local hospital and received unspecified medical therapy that reduced the bleeding; intermittent spotting persisted. She denied significant abdominal pain, dizziness, headache, palpitations, or anal fullness.
No notable past medical history.
There was no significant personal and family history.
Vital signs were as follows: Temperature, 36.8 °C; heart rate, 75 beats per minute; respiratory rate, 19 breaths per min; and blood pressure, 150/82 mmHg. She was alert and appeared well, with no jaundice of the skin or sclera. Cardiopulmonary auscultation was unremarkable. The liver and spleen were not palpable below the costal margins, and there was no edema of the lower extremities.
Laboratory results were as follows: Hemoglobin 94 g/L (reference range: 115–150 g/L); carcinoembryonic antigen 6.2 ng/mL (≤ 5 ng/mL); cancer antigen 125 60.2 U/mL (≤ 35 U/mL); cancer antigen 19-9 3.2 U/mL (≤ 37 U/mL); human epididymis protein 4 50.1 pmol/L (≤ 70 pmol/L for premenopausal women); and risk of ovarian malignancy algorithm index 8.11% (reference range for premenopausal women, 0%–7.4%.
Enhancement rate measurement method: A round region-of-interest (20-40 mm2) was set in the solid component of lesion. Plain scan CT value, arterial phase CT value, venous phase CT value and lag phase CT value were measured on the same level, and each CT value was measured three times. The means were recorded as the final results. CT enhancement rate of arterial phase: (Arterial phase CT value-plain scan CT value)/plain scan CT value. Enhancement rate of venous phase: (Venous phase CT value-plain scan CT value)/plain scan CT value. Enhancement rate of lag phase: (Lag phase CT value-plain scan CT value)/plain scan CT value[8]. The enhancement rate of lesion in MR referred to the CT measurement method. The MR enhancement rate of arterial phase: (Arterial phase MR value-plain scan MR value)/plain scan MR value. Enhancement rate of venous phase: (Venous phase MR value-plain scan MR value)/plain scan MR value. Enhancement rate of lag phase: (Lag phase MR value-plain scan MR value)/plain scan MR value.
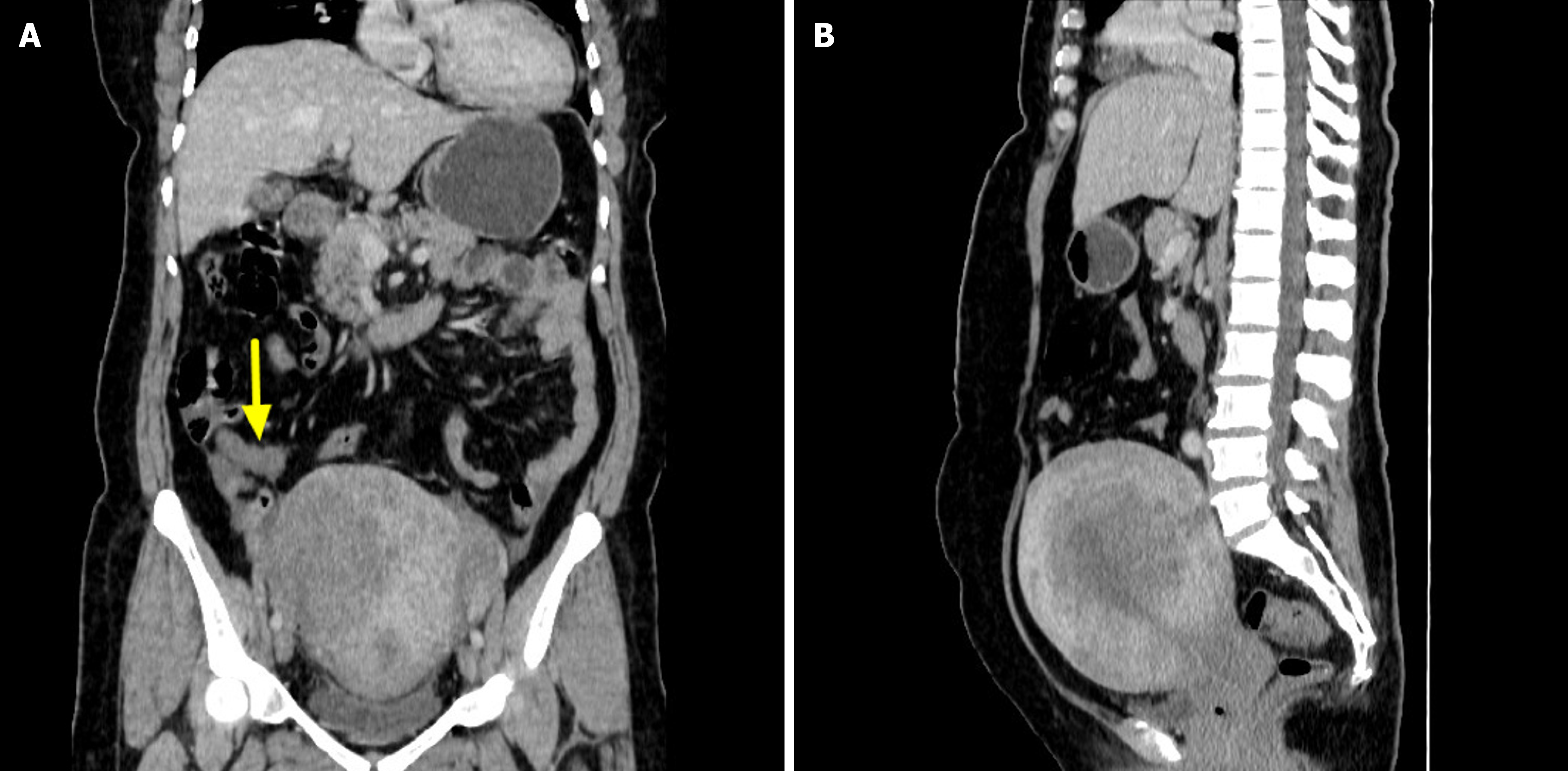
CT findings: The appendix was enlarged with concentric wall thickening and lacked significant intraluminal gas or fecalith-like density. Following contrast administration, the appendiceal wall demonstrated gradual and progressive enhancement, with enhancement rates of 17.8% in the arterial phase, 21.4% in the venous phase, and 46.4% in the delayed phase (Figure 1A). The lesion exhibited a clear boundary from the surrounding tissues, with no evidence of exudative changes.
The uterus appeared uniformly enlarged with an uneven myometrial texture and a smooth perimetrium. It also showed continuous progressive enhancement after contrast administration (Figure 1B), with enhancement rates of 25.0% in the arterial phase, 36.8% in the venous phase, and 55.3% in the delayed phase. The uterine lesion maintained a well-defined border from adjacent structures. No obvious pelvic lymphadenopathy was observed.
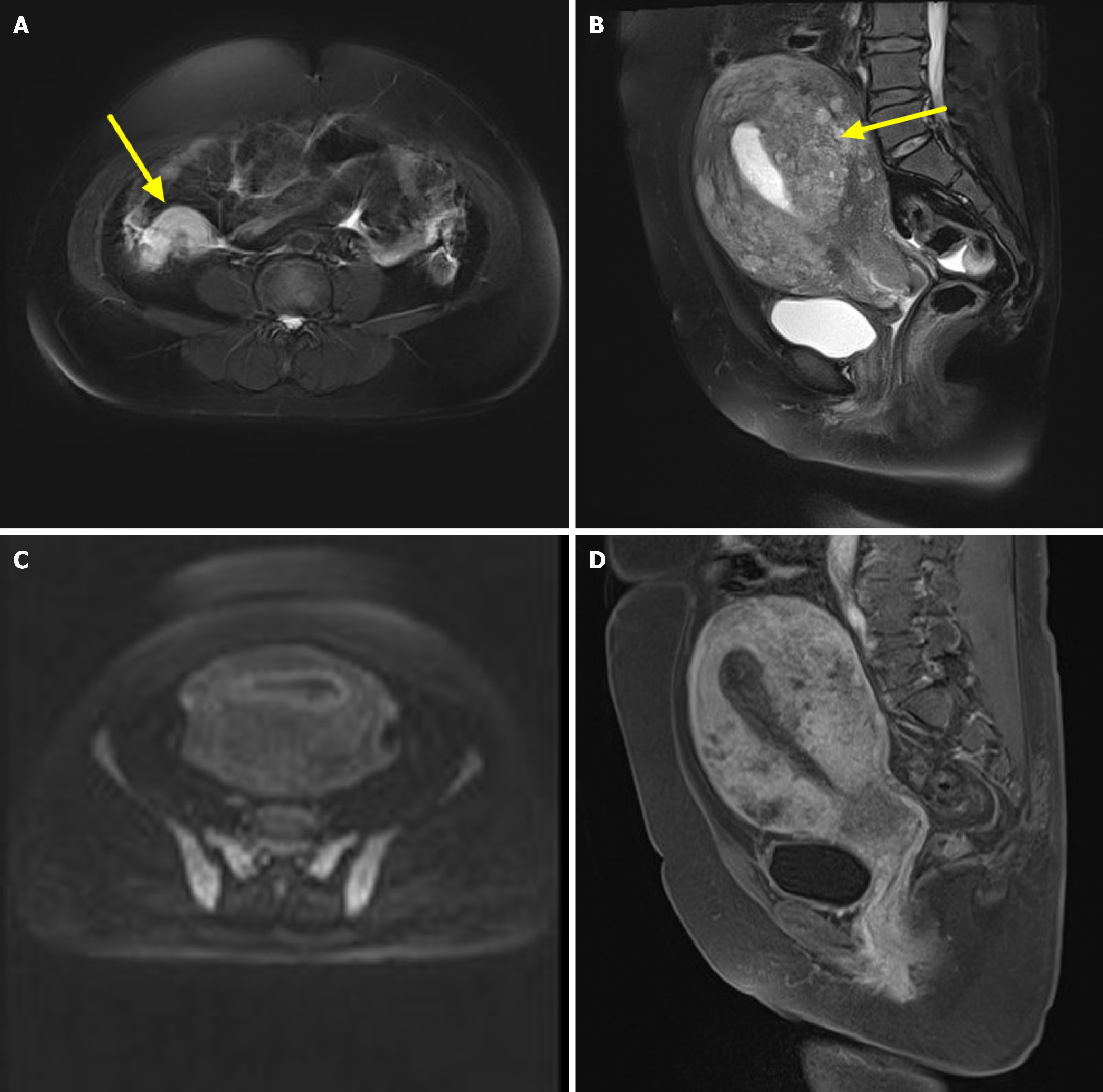
MRI findings: The appendix appeared enlarged, demonstrating isointensity on T1-weighted imaging (T1WI) and slight hyperintensity on T2-weighted imaging (T2WI) (Figure 2A). Diffusion-weighted imaging (DWI) showed mildly hyperintense signals, with no significant reduction in the apparent diffusion coefficient value. After contrast administration, the lesion exhibited gradual and progressive enhancement, with enhancement rates of 35.4% in the arterial phase, 60.6% in the venous phase, and 71.6% in the delayed phase. The lesion was well demarcated from adjacent tissues.
The uterine junctional zone was indistinct, with significant thickening of the myometrium and cervix. The lesion showed isointensity on T1WI and iso- to hyperintense signals on T2WI (Figure 2B). DWI revealed mild hyperintensity (Figure 2C), and the ADC value showed no obvious reduction. Post-contrast images demonstrated gradual and progressive enhancement of the uterus and cervix (Figure 2D), with enhancement rates of 35.8% in the arterial phase, 76.6% in the venous phase, and 97.5% in the delayed phase.
Both ovaries were clearly visualized, showing no abnormal signal on DWI and no significant enhancement post-contrast. A small amount of free fluid was noted in the pelvic cavity.
CT diagnosis: Enlarged appendix noted; differential diagnosis requires further evaluation. Significant uterine enlar
MRI diagnosis: Thickened appendix; specialist consultation recommended for comprehensive assessment. Marked heterogeneous thickening of the myometrium and cervix, suggestive of diffuse adenomyosis; additional evaluation warranted. Small volume of free pelvic fluid.
Preliminary clinical diagnosis: Abnormal uterine bleeding; mild anemia; uterine adenomyosis; appendiceal tumor?
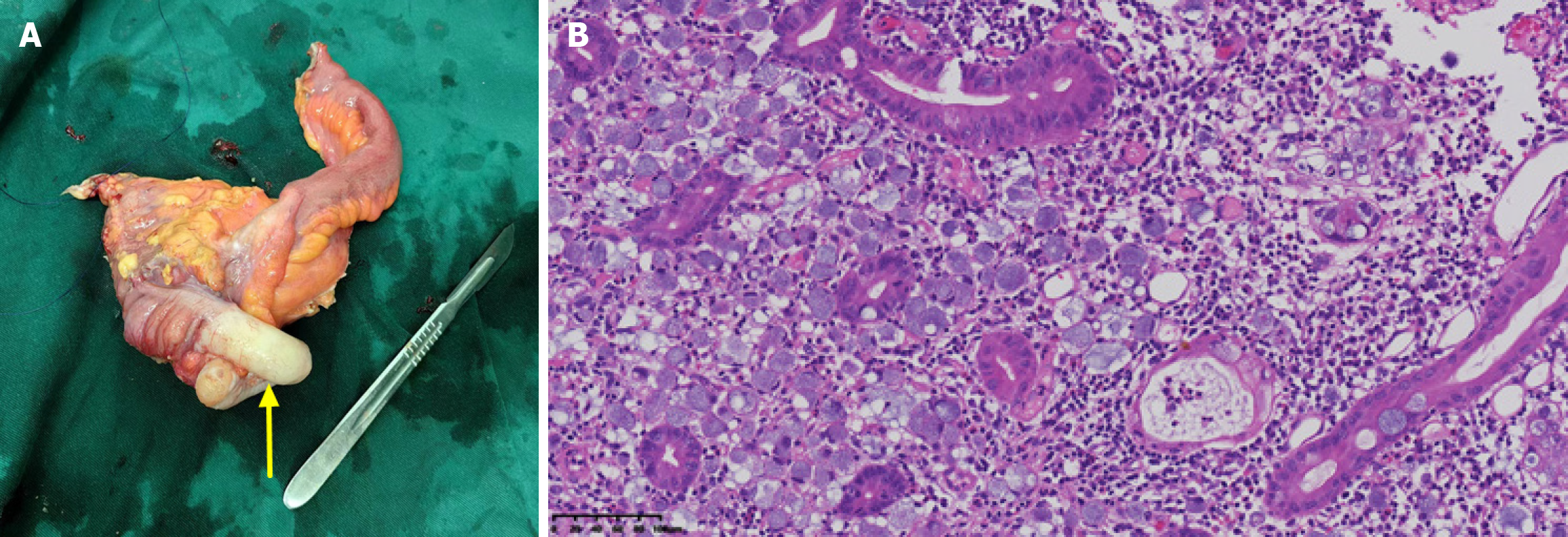
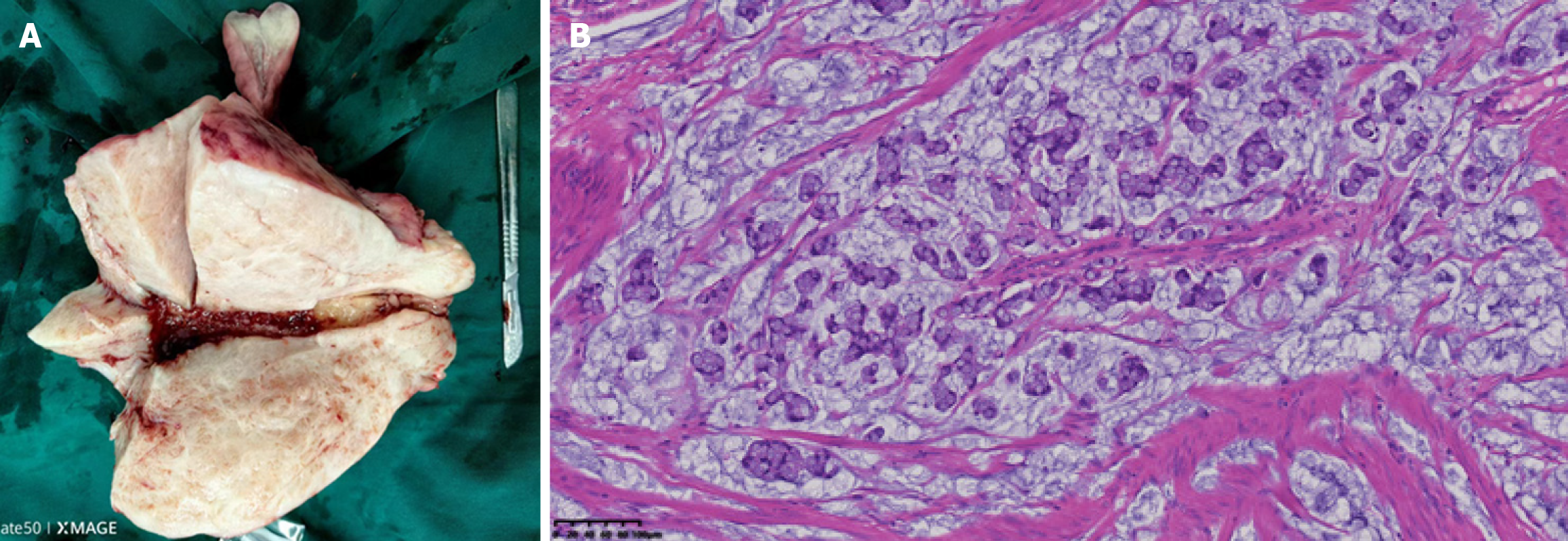
Postoperative pathological diagnosis: Appendiceal Signet ring cell carcinoma involved the entire appendix and serosa, extending into the cecal muscular layer and serosa. Signet ring cell carcinoma was identified in the endometrium, myometrium, and serosa. Signet ring cell carcinoma also involved the cervical stroma.
Appendiceal Signet ring cell carcinoma involved the entire appendix and serosa (Figure 3), extending into the cecal muscle layer, serosa. Signet ring cell carcinoma was identified in the endometrium, myometrium, and serosa (Figure 4). Signet ring cell carcinoma also involved the cervical stroma.
The patient received adjuvant chemotherapy after surgery.
Has been followed for two years. Regular monitoring with tumor markers and imaging has revealed no evidence of recurrence, and follow-up is ongoing.
Appendiceal Signet ring cell carcinoma often presents with nonspecific clinical manifestations and may be asymptomatic or mimic acute appendicitis[7,9]. The tumor commonly causes obstruction of the appendiceal lumen, leading to increased intraluminal pressure or compression of the appendiceal mesentery. This results in ischemia, congestion, and potentially secondary bacterial infection. These pathological changes manifest as appendicitis, influenced by the appendix’s ana
In this case, the appendix was enlarged and exhibited concentric wall thickening, consistent with the findings reported by Cho et al[14]. Both CT and MRI demonstrated gradual progressive enhancement following contrast administration, resembling the enhancement pattern described by Wang et al[16]. The uterus and cervix, affected by tumor implantation, showed diffuse thickening with an indistinct junctional zone. The uterine serosal layer remained intact, and the uterine lesions exhibited gradual progressive enhancement similar to the primary appendiceal lesion.
Based on this case and a review of the literature, several insights can be summarized: (1) Following implantation of Signet ring cell carcinoma to the uterus, diffuse thickening of the uterine myometrium may occur, often involving the cervix; (2) MRI characteristics of both primary and implantation lesions typically include iso-intensity on T1WI, iso- to hyperintensity on T2WI, slightly increased signal on DWI, with both CT and MRI showing gradual progressive enhancement after contrast administration; and (3) A small amount of ascitic fluid may be present in the pelvic cavity.
Preoperative accurate diagnosis is critical, as it may necessitate modifications in the surgical approach and the extent of resection[18]. Patients with appendiceal malignancy often present with symptoms and signs that mimic acute ap
Retrospective analysis revealed that the Signet ring cell carcinoma had metastasized to the uterus, and several features distinguished it from uterine adenomyosis: (1) Adenomyosis commonly presents with dysmenorrhea[19], whereas this patient had no history of abdominal pain; (2) Adenomyosis typically involves the uterine myometrium and rarely affects the cervix, while in this case, both the uterus and cervix were involved; and (3) Typical MRI findings of adenomyosis include high signal intensity in the myometrium on T2WI and focal high signal on T1WI in cases of hemorrhage[20]. In contrast, this case demonstrated significant myometrial thickening with an indistinct junctional zone and lacked focal high signal on both T1WI and T2WI.
The differential diagnosis of appendiceal signet ring cell carcinoma should include endometrial carcinoma, cervical cancer, and Krukenberg tumors. Endometrial carcinoma is one of the most common gynecologic malignancies[21], with lesions typically confined to the endometrial cavity. While it may invade the myometrium, the extent is usually limited. On DWI, endometrial carcinoma generally exhibits high signal intensity. In contrast, uterine implantation from SRCC tend to involve the uterus diffusely, with DWI signals that are less intense than those of endometrial carcinoma. Cervical cancer is the second most common malignancy in women[22], often originating in the cervix and potentially spreading upward to the uterus or downward to the vagina. It usually presents as a high-intensity signal on DWI. However, signet ring cell carcinoma implantation to the uterus show diffuse thickening of both the uterus and cervix, with lower DWI signal intensity than cervical cancer. Krukenberg tumors—metastatic ovarian tumors commonly originating from the gastrointestinal tract—affect both ovaries in approximately 80% of cases[23]. MRI findings typically include well-defined margins, predominantly solid components, and marked heterogeneous enhancement following contrast administration. Ascites is frequently present[24]. In comparison, signet ring cell carcinoma implantation to the uterus demonstrate lower degrees of enhancement and ascites is rarely observed.
Appendiceal Signet ring cell carcinoma is relatively rare, and preoperative diagnosis remains challenging. However, when imaging reveals thickening of the appendiceal wall alongside marked thickening of the uterine myometrium, with lesions demonstrating progressive enhancement after contrast administration, implantation appendiceal signet ring cell carcinoma involving the uterus should be considered.
| 1. | Hugen N, Verhoeven RH, Lemmens VE, van Aart CJ, Elferink MA, Radema SA, Nagtegaal ID, de Wilt JH. Colorectal signet-ring cell carcinoma: benefit from adjuvant chemotherapy but a poor prognostic factor. Int J Cancer. 2015;136:333-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 102] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 2. | Nitsche U, Friess H, Agha A, Angele M, Eckel R, Heitland W, Jauch KW, Krenz D, Nüssler NC, Rau HG, Ruppert R, Schubert-Fritschle G, Wilhelm D, Werner J, Engel J. Prognosis of mucinous and signet-ring cell colorectal cancer in a population-based cohort. J Cancer Res Clin Oncol. 2016;142:2357-2366. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 54] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 3. | Benesch MGK, Mathieson A. Epidemiology of Signet Ring Cell Adenocarcinomas. Cancers (Basel). 2020;12:1544. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 4. | Thota R, Fang X, Subbiah S. Clinicopathological features and survival outcomes of primary signet ring cell and mucinous adenocarcinoma of colon: retrospective analysis of VACCR database. J Gastrointest Oncol. 2014;5:18-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 25] [Reference Citation Analysis (0)] |
| 5. | Wu X, Lin H, Li S. Prognoses of different pathological subtypes of colorectal cancer at different stages: A population-based retrospective cohort study. BMC Gastroenterol. 2019;19:164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 6. | Enblad M, Egerszegi PP, Birgisson H, Sjöblom T, Glimelius B, Folkesson J. Signet Ring Cell Colorectal and Appendiceal Cancer: A Small Signet Ring Cell Component Is Also Associated with Poor Outcome. Cancers (Basel). 2023;15:2497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 7. | Andjelkovic B, Stojanovic B, Stojanovic MD, Milosevic B, Cvetkovic A, Spasic M, Jakovljevic S, Cvetkovic D, Stojanovic BS, Milosev D, Mitrovic M, Stankovic V. Appendiceal Signet Ring Cell Carcinoma: An Atypical Cause of Acute Appendicitis-A Case Study and Review of Current Knowledge. Diagnostics (Basel). 2023;13:2359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 8. | An YY, Yang GZ, Lin B, Zhang N, Hou HT, Zhu FM, Tian FJ, Wang J. Differentiation of lipid-poor adenoma from pheochromocytoma on biphasic contrast-enhanced CT. Abdom Radiol (NY). 2021;46:4353-4361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 9. | Caesar-Peterson S, Tulla K, Southall C, Lin Y, Genelus-Dominique E. A rare case of signet ring cell carcinoma of the appendix. J Surg Case Rep. 2020;2020:rjaa139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 10. | Kulkarni RV, Ingle SB, Siddiqui S. Primary signet ring cell carcinoma of the appendix: A rare case report. World J Clin Cases. 2015;3:538-541. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 7] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 11. | Hyngstrom JR, Hu CY, Xing Y, You YN, Feig BW, Skibber JM, Rodriguez-Bigas MA, Cormier JN, Chang GJ. Clinicopathology and outcomes for mucinous and signet ring colorectal adenocarcinoma: analysis from the National Cancer Data Base. Ann Surg Oncol. 2012;19:2814-2821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 266] [Article Influence: 20.5] [Reference Citation Analysis (1)] |
| 12. | Vukovic J, Vrebalov Cindro P, Tomic S, Tonkic A. Signet Ring Carcinoma of the Appendix Presenting as Crohn's Disease in a Young Male. Case Rep Gastroenterol. 2018;12:277-285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 13. | Turaga KK, Pappas SG, Gamblin T. Importance of histologic subtype in the staging of appendiceal tumors. Ann Surg Oncol. 2012;19:1379-1385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 153] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 14. | Cho YJ, Kim HJ, Jang SK, Yeon JW, Kim KH, Paik SY. Signet-ring cell carcinoma of the appendix: a case report with an emphasis on sonographic findings. Ultrasonography. 2016;35:164-167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 15. | Ina EA, Sobczak A, Drzymalski K, Biglione A. Appendiceal Signet Ring Cell Carcinoma Presenting As Acute Appendicitis: A Case Report. Cureus. 2024;16:e59137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 16. | Wang F, Tao Y, Liu Y, Tang G. Primary signet ring cell carcinoma of the appendix: An interesting case. Am J Med Sci. 2022;364:e10-e11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 17. | Okumura K, Oura S. A Metastatic Breast Tumor of an Appendiceal Signet Ring Cell Carcinoma. Case Rep Oncol. 2023;16:267-272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 18. | Kwag KS, Kim HJ, Jang SK, Yeon JW, Paik S, Jeon BG, Kim KH, Park JH, Shin E. Sonographic Findings of Malignant Appendix Tumors in Seven Cases. J Med Ultrasound. 2018;26:52-55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 19. | Habiba M, Benagiano G. Classifying Adenomyosis: Progress and Challenges. Int J Environ Res Public Health. 2021;18:12386. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 20. | Harmsen MJ, Trommelen LM, de Leeuw RA, Tellum T, Juffermans LJM, Griffioen AW, Thomassin-Naggara I, Van den Bosch T, Huirne JAF. Uterine junctional zone and adenomyosis: comparison of MRI, transvaginal ultrasound and histology. Ultrasound Obstet Gynecol. 2023;62:42-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 21. | Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359-E386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20108] [Cited by in RCA: 20515] [Article Influence: 2051.5] [Reference Citation Analysis (20)] |
| 22. | Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18694] [Cited by in RCA: 21370] [Article Influence: 2137.0] [Reference Citation Analysis (3)] |
| 23. | Xie H, Erickson BJ, Sheedy SP, Yin J, Hubbard JM. The diagnosis and outcome of Krukenberg tumors. J Gastrointest Oncol. 2021;12:226-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 24. | Kakushima N, Kamoshida T, Hirai S, Hotta S, Hirayama T, Yamada J, Ueda K, Sato M, Okumura M, Shimokama T, Oka Y. Early gastric cancer with Krukenberg tumor and review of cases of intramucosal gastric cancers with Krukenberg tumor. J Gastroenterol. 2003;38:1176-1180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 47] [Article Influence: 2.2] [Reference Citation Analysis (0)] |