Published online Aug 28, 2025. doi: 10.4329/wjr.v17.i8.110307
Revised: June 24, 2025
Accepted: July 23, 2025
Published online: August 28, 2025
Processing time: 85 Days and 11.5 Hours
Esophageal cancer (EC) is one of the most prevalent malignant gastrointestinal tumors; accurate prediction of EC staging has high significance before treatment.
To explore a rational radiomic approach for predicting preoperative staging of EC based on magnetic resonance imaging (MRI).
This retrospective study included 210 patients with pathologically confirmed EC, randomly divided into a primary cohort (n = 147) and a validation cohort (n = 63) in a ratio of 7:3. All patients underwent a preoperative MRI scan from the neck to the abdomen. High-throughput and quantitative radiomics features were extr
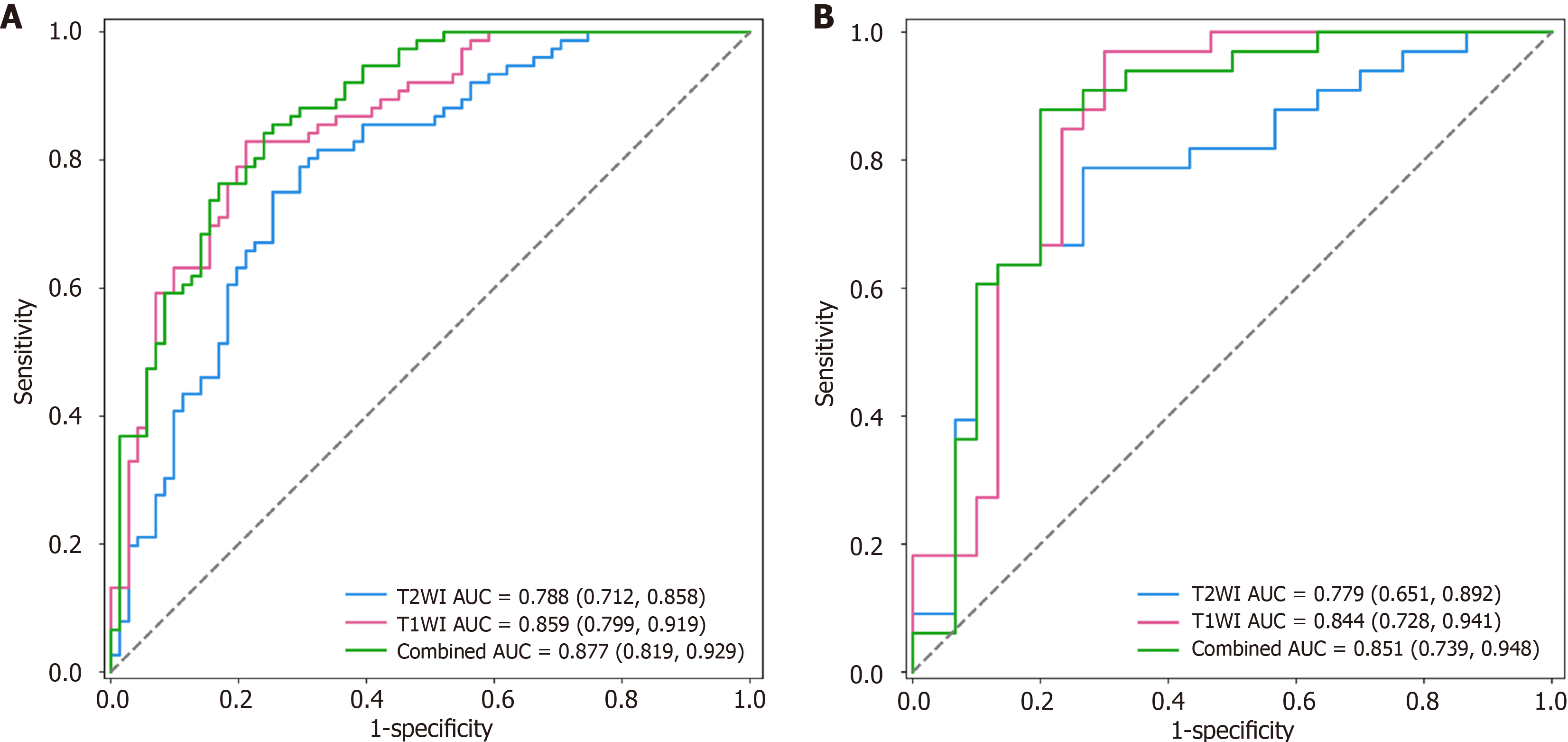
A total of 214 radiomics features were extracted. Following feature dimension reduction, the T1WI and T2WI sequences were retained, and 14 features from the T1WI sequence and 3 features from the T2WI sequence were selected to construct radiomics signatures. The radiomics signature combining T2WI with T1WI-Gd demonstrated superior discrimination of stages in the validation cohort (AUC: 0.851; SEN: 0.697; SPE: 0.793), which outperformed single-sequence models (AUC: 0.779, 0.844; SEN: 0.667, 0.636; SPE: 0.8, 0.8).
MRI-based radiomics signatures could identify EC stages before treatment, which could serve as a noninvasive and quantitative approach aiding personalized treatment planning.
Core Tip: This study developed a novel magnetic resonance imaging-based radiomics approach to noninvasively predict preoperative stage of esophageal cancer (EC). By integrating quantitative features from T2-weighted imaging and contrast-enhanced T1-weighted imaging sequences in 210 EC patients, a logistic regression model achieved high accuracy in distinguishing early-stage (I-II) from advanced-stage (III-IV) disease. This multimodal radiomics signature outperformed single-sequence models and offers a promising tool for guiding personalized treatment strategies.
- Citation: Yang RH, Lin ZP, Dong T, Fan WX, Qin HD, Jiang GH, Dai HY. Magnetic resonance imaging-based radiomics signature for predicting preoperative staging of esophageal cancer. World J Radiol 2025; 17(8): 110307
- URL: https://www.wjgnet.com/1949-8470/full/v17/i8/110307.htm
- DOI: https://dx.doi.org/10.4329/wjr.v17.i8.110307
Esophageal cancer (EC) poses a significant health burden in China, ranking sixth in incidence and fourth in cancer mortality, with a dismal 5-year survival of 19%[1,2]. Early-stage (I-II) disease typically undergoes endoscopic or surgical resection, whereas advanced (III-IV) EC requires neoadjuvant chemoradiation prior to surgery or definitive chemoradiation[3]. Lymph node (LN) metastasis correlates strongly with reduced survival[4], underscoring the imperative for accurate pretreatment staging. The accurate prediction of EC staging prior to treatment is clinically critical.
Radiomics is increasingly recognized as a critical tool in cancer research. High-throughput mining enables the extraction of quantitative image features from digitally encoded medical images. These robust image-based signatures could potentially enhance precision diagnosis and treatment strategies[5,6]. Recent radiomics research has demonstrated the possibility of using magnetic resonance imaging (MRI) to substantially improve the detection and prediction of LN metastases[7,8]. While computed tomography (CT), MRI, positron emission tomography-CT (PET-CT), and endoscopic ultrasonography (EUS) each exhibit variable sensitivity (SEN)/specificity (SPE) for EC staging and LN detection[9-11], contrast-enhanced CT remains the clinical mainstay despite controversial LN detection accuracy (SEN: 37.3%-67.2%; SPE: 63.9%-96.4%)[12]. Notably, the new Chinese EC guidelines has now recognized MRI’s diagnostic parity with CT for LN assessment[13].
At present, radiomics research on EC based on MRI mainly focuses on predicting LN metastases and evaluating the efficacy of neoadjuvant therapy[14-16], However, there has been little research on predicting the early and local progression stages of EC based on MRI radiomics, which has significant clinical significance for personalized treatment of EC patients. Our study addresses this by extracting radiomic features from T2-weighted imaging (T2WI) and T1-weighted imaging (T1WI)-Gd sequences to build a predictive model for distinguishing early (I-II) from locally advanced (III-IV) EC, providing a reference for guiding clinical treatment decisions.
The clinical and MRI data of EC patients who underwent LN dissection confirmed by surgery and pathology between March 2017 and November 2020 were retrospectively collected. The inclusion criteria were: (1) Pathologically confirmed EC; (2) Underwent radical resection and LN dissection, with clear postoperative pathological stage and clear pathological LN results; (3) Preoperative MRI scan (T2WI and T1WI-Gd) within two weeks before surgery; (4) No anticancer treatment prior to MRI; and (5) No history of other malignant tumors. The exclusion criteria were: (1) Received preoperative anticancer therapy; (2) Concurred other malignant tumors; and (3) Non-diagnostic MRI image quality, precluding subsequent feature extraction.
This study was approved by the Ethics Committee on Clinical Researches and Novel Technologies of Meizhou People’s Hospital (Grant No. 2023-C-45), and patient informed consent was waived for this retrospective study. All procedures were carried out in accordance with relevant guidelines and regulations. Ultimately, 210 patients with EC (165 males and 45 females, aged 40 to 86 years, mean age 63.1 ± 7.92 years) were included. Among them, there were 207 cases of squamous cell carcinoma, 2 cases of adenosquamous cell carcinoma, and 1 case of mixed neuroendocrine carcinoma. Based on tumor node metastasis staging, 101 patients had stages I-II (74 males, 27 females, mean age 63.2 ± 8.01 years), and 109 had stages III-IV EC (91 males and 18 females, mean age 62.9 ± 7.86 years). The clinicopathologic characteristics of the patients are presented in Table 1.
| Characteristics | Primary cohort | P value | Validation cohort | P value | ||
| Stage I-II | Stage III-IV | Stage I-II | Stage III-IV | |||
| Age (years) | 62.0 | 61.0 | 0.548 | 62.5 | 64.0 | 0.842 |
| Gender | 0.073 | 0.613 | ||||
| Male | 50 | 63 | 24 | 28 | ||
| Female | 21 | 13 | 6 | 5 | ||
| Location | 0.33 | 0.705 | ||||
| Upper | 2 | 6 | 2 | 2 | ||
| Middle | 47 | 51 | 17 | 22 | ||
| Lower | 22 | 19 | 11 | 9 | ||
Imaging was performed with a 3.0 T Siemens (Erlangen, Germany) Skyra MR scanner equipped with 18-channel phased array coil. The patient was placed supine and told to breathe as calmly as possible to reduce motion artifacts. The scanning range was from the level of the supraclavicular fossa to the gastric cardia. The scanning sequence and parameters were: (1) T2WI: Slice thickness 3 mm, TR 3000 ms, TE 91 ms, NEX = 1, matrix 256 × 256, field of view (FOV) 210 mm × 210 mm and scanning time 2 minutes; and (2) T1WI-Gd: Slice thickness 1.5 mm, TR 4.4 ms, TE 2.0 ms, NEX = 1, matrix 160 × 160, FOV 150 mm × 150 mm, and flip angle 12. During enhanced scanning, Gd-DTPA contrast agent was injected intravenously through the elbow using a high-pressure syringe at 0.1 mmol/kg. The scanning time was 3 minutes and 26 seconds.
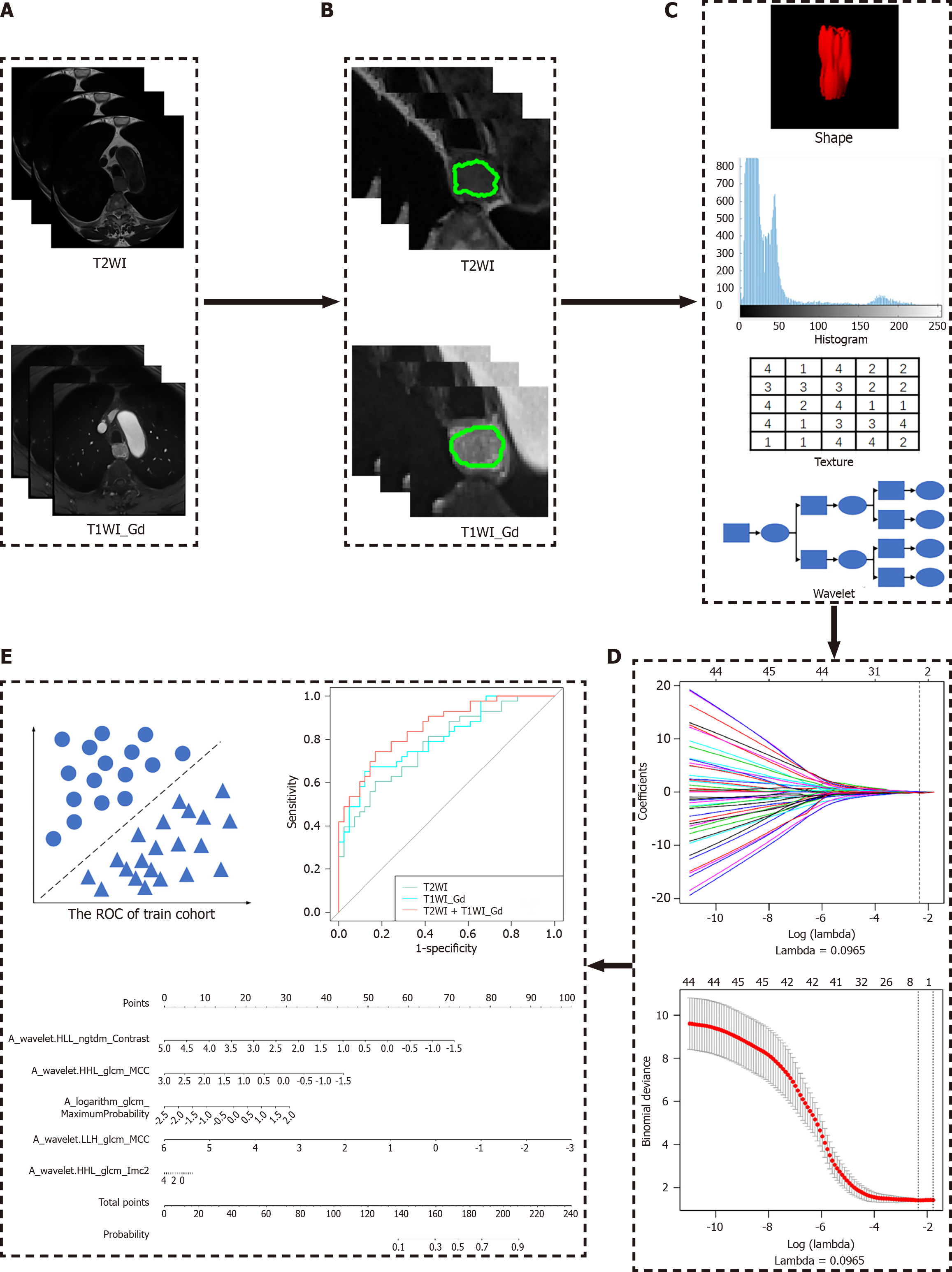
Manual segmentation of the EC was performed on each patient's MR images utilizing an Artificial Intelligent Kit (AK), a new application platform for the overall solution of artificial intelligence in medical imaging radiomics launched by GE Healthcare. Two experienced associate senior radiologists (readers 1 and 2) carefully delineated the tumor on all T2WI and T1WI-Gd images to generate two 3D segmentations of the entire tumor. These regions of interests were applied for subsequent feature extraction and further analysis.
Patients were randomly divided into a primary cohort (n = 147) and a validation cohort (n = 63) at a 7:3 ratio, as according to previous reports[17,18], and the sample size was estimated by using the package of R software (version 4.3.1). Radiomic features were extracted based on the segmentation results from the primary cohort using a homemade program in the AK. Preprocessing included: (1) The image was first resampled using linear interpolation algorithm to a standard voxel spacing of 1 mm × 1 mm × 1 mm; and (2) Z-score for MRI intensity values were standardized. The Python PyRadiomics module was used to extract radiomic features through AK, where the bin width of the histogram was set to 5 to quantify the intensity distribution of the image. The distance parameter for texture features such as gray level co-occurrence matrix (GLCM) was uniformly set to 1. For each MRI sequence, the 214 extracted features covered the major feature pool in recent radiomic studies. The features were categorized into six primary types (Figure 1): (1) Histogram features; (2) Form factor features; (3) GLCM and Harilick features; (4) Gray level run length matrix features; (5) Grey level size zone matrix (GLSZM) features; and (6) Gray level dependence matrix (GLDM) features.
A rigorous feature selection pipeline was implemented to simplify the model development and avoid over-fitting. Minimal redundancy maximal relevance (mRMR) and least absolute shrinkage and selection operator (LASSO) method were used for feature dimensionality reduction. For tuning coefficients λ and α, the respective minimum standard deviation and maximum area under the curve (AUC) criteria were followed. Dimensionality reduction was performed using the variance method (excluding features with zero variance), the mRMR method selecting the top 50 features, and the LASSO method with lambda selection standard set to ‘λ.min’, and selecting the optimal λ through five fold cross validation on the training set. For accurate prediction of the EC stages, 16 features selected from the T2WI with T1WI-Gd images made the greatest contribution. Then, each patient’s two radiomic signature sets were integrated into a multivariate logistic regression framework. Model training generated weighted linear combinations of selected features, which constituted the final radiomic signature scores for individual patients.
The statistical analysis was performed in R (version 3.5.1; http://www.Rproject.org). The Shapiro-Wilk test was used to test whether the quantitative data were normally distributed. The rank-sum test was used to compare the differences in age, between patients with EC stage I-II and stage III-IV. The χ2 test or correction χ2 test was used to compare the differences in sex, tumor location, and pathological type between patients with EC stage I-II and stage III-IV. In addition, the primary cohort was used to train the logistic regression model, and the validation cohort was used to test the model. Finally, the receiver operating characteristic (ROC) curve, SEN, and SPE were used to evaluate the performance of the prediction model. The DeLong test compared the AUC between the single-sequence and mixed-sequence models. A two-sided P value < 0.05 was considered statistically significant.
Patient clinicopathological features are summarized in Table 1. No significant intergroup differences (stages I-II vs III-IV) existed in age, sex, or tumor location (P > 0.05).
After feature selection and dimensionality reduction, 14 and 3 of the 214 radiomic features were retained for EC staging prediction (Table 2). Fourteen and three features distinguished stages I-II from stages III-IV in the T1WI and T2WI models, respectively.
| Task | MRI | Feature name | Coefficient |
| Tumor node metastasis stage prediction | T2WI | ||
| Original_firstorder_Median | 0.921642536 | ||
| Original_shape_Elongation | 0.967002183 | ||
| Original_glcm_Correlation | 0.246781324 | ||
| T1WI-Gd | |||
| Original_shape_LeastAxisLength | 0.875999143 | ||
| Original_shape_Elongation | 0.967002356 | ||
| Original_glcm_DifferenceAverage | 0.109732763 | ||
| Original_firstorder_Kurtosis | 0.605404872 | ||
| Original_firstorder_Median | 0.921642536 | ||
| Original_glszm_LowGrayLevelZoneEmphasis | 0.274812737 | ||
| Original_glcm_Imc1 | 0.638235237 | ||
| Original_gldm_LargeDependenceHighGrayLevelEmphasis | 0.209360630 | ||
| Original_shape_MajorAxisLength | 0.149464274 | ||
| Original_ngtdm_Strength | 0.214134941 | ||
| Original_glcm_Idn | 0.983044251 | ||
| Original_firstorder_10Percentile | 0.303070619 | ||
| Original_glszm_SizeZoneNonUniformity | 0.126542445 | ||
| Original_ngtdm_Complexity | 0.524606933 |
The radiomics signature was constructed using the selected features from the previous section, forming a linear combination based on a logistic regression model. There were four first-order features, four shape features, four GLCM features, two GLSZM features, one GLDM feature, and two neighboring gray-tone difference matrix features. The formula for the combined model was as follows: Radscore = T1WI × 5.27600436 + T2WI × 3.21937331 - 4.36897257.
The discriminative power of the radiomic signature for tumor, node, metastasis staging was assessed using ROC curves in both the primary and validation cohorts (Figure 2). Subsequently, the ROC curves corresponding to different models were compared using the Delong test, and the AUC, SEN, and SPE were calculated (Tables 3 and 4). The radiomics signature combining T2WI with T1WI-Gd exhibited superior discrimination of the stages in the primary (AUC: 0.788, 0.859; SEN: 0.75, 0.829; SPE: 0.746, 0.789) and validation cohorts (AUC: 0.851; SEN: 0.697; SPE: 0.793), outperforming single-sequence models (AUC: 0.779, 0.844; SEN: 0.667, 0.636; SPE: 0.8, 0.8). Radiomic scores (Rad-scores) for EC patients in both the primary and validation cohorts were calculated using the elastic net model.
| Model | Primary cohort | Validation cohort | ||||
| T2WI | T1WI | Combined | T2WI | T1WI | Combined | |
| T2WI | 0 | 0.081 | 0.0035 | 0 | 0.27 | 0.09 |
| T1WI | -1.74 | 0 | 0.19 | -1.09 | 0 | 0.80 |
| Combined | -2.95 | -1.30 | 0 | -1.65 | -0.25 | 0 |
| Model | Primary cohort | Validation cohorts | ||||
| AUC (95%CI) | SEN (95%CI) | SPE (95%CI) | AUC (95%CI) | SEN (95%CI) | SPE (95%CI) | |
| T2WI | 0.788 (0.712, 0.858) | 0.75 (0.642, 0.834) | 0.746 (0.634, 0.833) | 0.779 (0.651, 0.892) | 0.667 (0.496, 0.803) | 0.8 (0.627, 0.905) |
| T1WI | 0.859 (0.799, 0.919) | 0.829 (0.729, 0.897) | 0.789 (0.681, 0.868) | 0.844 (0.728, 0.941) | 0.636 (0.466, 0.778) | 0.8 (0.627, 0.905) |
| Combined | 0.877 (0.819, 0.929) | 0.842 (0.744, 0.907) | 0.761 (0.65, 0.845) | 0.851 (0.739, 0.948) | 0.697 (0.527, 0.826) | 0.8 (0.627, 0.905) |
This study demonstrated that a quantitative approach utilizing multimodal MRI can effectively identify EC stages prior to treatment initiation. Leveraging significant differences in the Rad-score, the radiomics signature successfully distinguished between esophageal squamous cell carcinoma stages I-II and III-IV during the preoperative period. These findings were supported by MRI’s superior soft-tissue contrast and multiparametric capabilities, which provide high-quality imaging foundation for precise tumor delineation and radiomics analysis.
Surgical resection remains the cornerstone of EC management, yet surgical intervention for advanced EC (stages III-IV) is associated with significant challenges and a high postoperative recurrence rate. LN status is a critical independent prognostic factor, and the approach for LN dissection plays a pivotal role in treatment strategies[4]. Several studies have suggested that systematic LN dissection should be performed in EC patients only when the tumor invades the submucosa, as extended LN resection may elevate the risk of postoperative complications[19,20]. In addition, accurate LN staging is essential for guiding the implementation of neoadjuvant therapy[21]. Although CT is widely used for preoperative EC staging, its accuracy remains limited. Radiomics addresses these limitations by decoding tumor heterogeneity through high-throughput feature extraction from medical images, thereby enhancing diagnostic and prognostic precision.
This study confirmed the feasibility of predicting EC stages using MR-based radiomic features. We extracted 214 quantitative features from tumors using T2WI and T1WI-Gd images. The seventeen features that contributed most to the differentiation of EC stages were selected for model construction. Radiomics features derived from T2WI are based on the signal intensity of water molecules, primarily reflecting the water content characteristics of the tumor.
The high-order radiomics features comprised the GLCM, which reflects the gray level difference, including distribution uniformity and texture thickness. Greater heterogeneity of the tumor was associated with an uneven gray level and increased texture complexity. Histological features derived from T1WI-Gd images predominantly indicative of microvascular density and histologic grading[22]. Higher-order texture features, exemplified by GLCM metrics, quantified spatial complexity patterns correlating with biologically aggressive phenotypes[23].
Consistent with prior studies, our results showed that integrating radiomics features from dual-sequence MRI achieved strong discriminatory performance in differentiating stage I-II from stage III-IV EC in both the primary and validation cohorts[16,24,25]. Our MRI-based radiomics model outperformed CT and PET-CT radiomics methods for staging EC[26,27]. This advantage may originate from MRI's superior soft-tissue resolution coupled with synergistic information integration from multiparametric sequences. The improvement may also stem from the complementary information provided by multiparametric MRI, which enables a more comprehensive and objective assessment of tumor heterogeneity. In addition, our results demonstrated performance comparable with prior MRI radiomics studies predicting LN metastasis in EC[16]. This reinforces the potential of MRI-based radiomics for EC staging.
This study had several limitations. First, the single-center design and modest sample size necessitate validation through multicenter studies with larger cohorts. Second, although we focused on tumor staging, the prediction efficiency of LN metastasis, including its extent, in different regions remains unassessed. Future work will be dedicated to developing models focused on predicting LN metastasis. Last, the exclusive use of a single MRI scanner limits generalizability. Future work should evaluate the impact of varying equipment, field strength, and scanning parameters.
This study demonstrates that MRI-based radiomics can effectively differentiate between stage I–II and stage III-IV EC before treatment. We developed a multiparametric radiomics model incorporating features from multiple MRI sequences. MRI-based radiomics could serve as a valuable noninvasive adjunct tool for preoperative staging and personalized therapeutic strategy formulation in EC.
| 1. | Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73:17-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 9949] [Article Influence: 4974.5] [Reference Citation Analysis (2)] |
| 2. | Sheikh M, Roshandel G, McCormack V, Malekzadeh R. Current Status and Future Prospects for Esophageal Cancer. Cancers (Basel). 2023;15:765. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 125] [Article Influence: 62.5] [Reference Citation Analysis (0)] |
| 3. | Liu S, Zheng H, Pan X, Chen L, Shi M, Guan Y, Ge Y, He J, Zhou Z. Texture analysis of CT imaging for assessment of esophageal squamous cancer aggressiveness. J Thorac Dis. 2017;9:4724-4732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 4. | Rice TW, Ishwaran H, Hofstetter WL, Schipper PH, Kesler KA, Law S, Lerut EM, Denlinger CE, Salo JA, Scott WJ, Watson TJ, Allen MS, Chen LQ, Rusch VW, Cerfolio RJ, Luketich JD, Duranceau A, Darling GE, Pera M, Apperson-Hansen C, Blackstone EH. Esophageal Cancer: Associations With (pN+) Lymph Node Metastases. Ann Surg. 2017;265:122-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 93] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 5. | Coroller TP, Agrawal V, Huynh E, Narayan V, Lee SW, Mak RH, Aerts HJWL. Radiomic-Based Pathological Response Prediction from Primary Tumors and Lymph Nodes in NSCLC. J Thorac Oncol. 2017;12:467-476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 168] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 6. | Shi L, Wang X, Li C, Bai Y, Zhang Y, Li H. Radiomics applications in the modern management of esophageal squamous cell carcinoma. Med Oncol. 2025;42:221. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 7. | Ma D, Zhou T, Chen J, Chen J. Radiomics diagnostic performance for predicting lymph node metastasis in esophageal cancer: a systematic review and meta-analysis. BMC Med Imaging. 2024;24:144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 8. | Jannatdoust P, Valizadeh P, Pahlevan-Fallahy MT, Hassankhani A, Amoukhteh M, Behrouzieh S, Ghadimi DJ, Bilgin C, Gholamrezanezhad A. Diagnostic accuracy of CT-based radiomics and deep learning for predicting lymph node metastasis in esophageal cancer. Clin Imaging. 2024;113:110225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 9. | Li X, Wang Y, Kong M, Lin J. Systematic review and meta-analysis of endoscopic ultrasonography in staging diagnosis of esophageal cancer after neoadjuvant radiotherapy and chemotherapy. J Gastrointest Oncol. 2022;13:1525-1540. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 10. | Goense L, Heethuis SE, van Rossum PSN, Voncken FEM, Lagendijk JJW, Lam MGEH, Terhaard CH, van Hillegersberg R, Ruurda JP, Mook S, van Lier ALHMW, Lin SH, Meijer GJ. Correlation between functional imaging markers derived from diffusion-weighted MRI and 18F-FDG PET/CT in esophageal cancer. Nucl Med Commun. 2018;39:60-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 11. | Luo LN, He LJ, Gao XY, Huang XX, Shan HB, Luo GY, Li Y, Lin SY, Wang GB, Zhang R, Xu GL, Li JJ. Evaluation of preoperative staging for esophageal squamous cell carcinoma. World J Gastroenterol. 2016;22:6683-6689. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 27] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 12. | Liu J, Wang Z, Shao H, Qu D, Liu J, Yao L. Improving CT detection sensitivity for nodal metastases in oesophageal cancer with combination of smaller size and lymph node axial ratio. Eur Radiol. 2018;28:188-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 13. | Chinese National Cancer Center; Chinese Association of Thoracic Surgeons; Chinese Society for Thoracic and Cardiovascular Surgery; Chinese Society for Diseases of the Esophagus. [Chinese Guidelines on Perioperative Management of Resectable Esophageal Cancer (2023 edition)]. Zhonghua Yi Xue Za Zhi. 2023;103:2552-2570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 14. | Guo H, Tang HT, Hu WL, Wang JJ, Liu PZ, Yang JJ, Hou SL, Zuo YJ, Deng ZQ, Zheng XY, Yan HJ, Jiang KY, Huang H, Zhou HN, Tian D. The application of radiomics in esophageal cancer: Predicting the response after neoadjuvant therapy. Front Oncol. 2023;13:1082960. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 15. | Lu S, Wang C, Liu Y, Chu F, Jia Z, Zhang H, Wang Z, Lu Y, Wang S, Yang G, Qu J. The MRI radiomics signature can predict the pathologic response to neoadjuvant chemotherapy in locally advanced esophageal squamous cell carcinoma. Eur Radiol. 2024;34:485-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 13] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 16. | Qu J, Shen C, Qin J, Wang Z, Liu Z, Guo J, Zhang H, Gao P, Bei T, Wang Y, Liu H, Kamel IR, Tian J, Li H. The MR radiomic signature can predict preoperative lymph node metastasis in patients with esophageal cancer. Eur Radiol. 2019;29:906-914. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 64] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 17. | Luo T, Wan J, Liu S, Wang X, Zhou P, Xue Q, Hou J, Wang P. Establishment of a scoring model for predicting clinical outcomes in patients with unilateral primary aldosteronism after superselective adrenal artery embolization. Ir J Med Sci. 2024;193:2269-2279. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 18. | Yan X, Fu X, Gui Y, Chen X, Cheng Y, Dai M, Wang W, Xiao M, Tan L, Zhang J, Shao Y, Wang H, Chang X, Lv K. Development and validation of a nomogram model based on pretreatment ultrasound and contrast-enhanced ultrasound to predict the efficacy of neoadjuvant chemotherapy in patients with borderline resectable or locally advanced pancreatic cancer. Cancer Imaging. 2024;24:13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 19. | Visser E, van Rossum PSN, Ruurda JP, van Hillegersberg R. Impact of Lymph Node Yield on Overall Survival in Patients Treated With Neoadjuvant Chemoradiotherapy Followed by Esophagectomy for Cancer: A Population-based Cohort Study in the Netherlands. Ann Surg. 2017;266:863-869. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 71] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 20. | Booka E, Takeuchi H, Nishi T, Matsuda S, Kaburagi T, Fukuda K, Nakamura R, Takahashi T, Wada N, Kawakubo H, Omori T, Kitagawa Y. The Impact of Postoperative Complications on Survivals After Esophagectomy for Esophageal Cancer. Medicine (Baltimore). 2015;94:e1369. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 131] [Cited by in RCA: 177] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 21. | van Rossum PSN, van Hillegersberg R, Lever FM, Lips IM, van Lier AL, Meijer GJ, van Leeuwen MS, van Vulpen M, Ruurda JP. Imaging strategies in the management of oesophageal cancer: what's the role of MRI? Eur Radiol. 2013;23:1753-1765. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 55] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 22. | Dong X, Chunrong Y, Hongjun H, Xuexi Z. Differentiating the lymph node metastasis of breast cancer through dynamic contrast-enhanced magnetic resonance imaging. BJR Open. 2019;1:20180023. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 23. | Brown AM, Nagala S, McLean MA, Lu Y, Scoffings D, Apte A, Gonen M, Stambuk HE, Shaha AR, Tuttle RM, Deasy JO, Priest AN, Jani P, Shukla-Dave A, Griffiths J. Multi-institutional validation of a novel textural analysis tool for preoperative stratification of suspected thyroid tumors on diffusion-weighted MRI. Magn Reson Med. 2016;75:1708-1716. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 24. | Hou Z, Li S, Ren W, Liu J, Yan J, Wan S. Radiomic analysis in T2W and SPAIR T2W MRI: predict treatment response to chemoradiotherapy in esophageal squamous cell carcinoma. J Thorac Dis. 2018;10:2256-2267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 32] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 25. | van Rossum PSN, Xu C, Fried DV, Goense L, Court LE, Lin SH. The emerging field of radiomics in esophageal cancer: current evidence and future potential. Transl Cancer Res. 2016;5:410-423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 26. | Wu L, Wang C, Tan X, Cheng Z, Zhao K, Yan L, Liang Y, Liu Z, Liang C. Radiomics approach for preoperative identification of stages I-II and III-IV of esophageal cancer. Chin J Cancer Res. 2018;30:396-405. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 50] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 27. | Lei X, Cao Z, Wu Y, Lin J, Zhang Z, Jin J, Ai Y, Zhang J, Du D, Tian Z, Xie C, Yin W, Jin X. Preoperative prediction of clinical and pathological stages for patients with esophageal cancer using PET/CT radiomics. Insights Imaging. 2023;14:174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |