Published online Oct 6, 2021. doi: 10.12998/wjcc.v9.i28.8524
Peer-review started: May 18, 2021
First decision: June 15, 2021
Revised: June 29, 2021
Accepted: August 2, 2021
Article in press: August 2, 2021
Published online: October 6, 2021
Processing time: 133 Days and 6.7 Hours
Endometriosis (EMs), an estrogen-dependent disease, refers to the appearance of mucosa-covered endometrial tissues (glandular and interstitial) growing in the uterine cavity outside the uterine myometrium. It is commonly seen in women aged 25 to 45, with an incidence of approximately 10%-15%.
A 35-year-old unmarried female who denied a history of sex with an intact hymen had multiple dysmenorrhea and pain in the left lower abdomen that recurred during menstruation. Ultrasound examination revealed a dark cystic area measuring 4.9 cm × 4.6 cm on the left side with poor light transmittance, which suggested a left endometriotic cyst. The patient was treated with pain medications (four capsules t.i.d., p.o.). After one month, computed tomography of the abdomen and pelvis revealed a low-density focus measuring approximately 38 mm in diameter, a blurred mesentery fat plane in the pelvic cavity, and pelvic effusion. Ultrasound showed a complex echo density measuring 5.2 cm × 3.0 cm × 4.2 cm in the left ovarian area and a fluid sonolucent area with a depth of 2.0 cm in the pelvic cavity. Left ovarian cystectomy, electrocautery for endometriotic lesions, myomectomy, and pelvic adhesion lysis were performed under laparoscopy. The postoperative diagnosis was left ovarian chocolate cyst rupture and EMs (stage III, ovarian type, peritoneal type).
Laparoscopic surgery can safely control the symptoms of EMs and effectively eradicate the disease.
Core Tip: With the advancement of laparoscopic technology, minimally invasive laparoscopic surgery has successfully become the treatment option for ruptured chocolate cysts. We would like to share our experience of laparoscopic treatment of ruptured ovarian endometriotic cysts.
- Citation: Wang L, Jiang YJ. Rupture of ovarian endometriotic cyst complicated with endometriosis: A case report. World J Clin Cases 2021; 9(28): 8524-8530
- URL: https://www.wjgnet.com/2307-8960/full/v9/i28/8524.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i28.8524
Endometriosis (EMs) is a benign disease caused by the presence of endometrium outside the uterine cavity. It is commonly seen in women aged 25 to 45, with an incidence of 10%-15%, and frequent symptoms, including lower abdominal pain, infertility, and dysmenorrhea, adversely affect both health and quality of life[1,2]. There are three types of EMs: Ovarian EMs, peritoneal EMs, and deep invasive EMs, among which ovarian endometriotic cysts (commonly known as chocolate cysts) are a common type, accounting for approximately 80% of EMs, with an incidence of approximately 20%-45% in females with infertility[3,4]. Studies have revealed a relationship between EMs and heredity, immunity, inflammation, and so on[5]. The incidence of both chocolate cyst rupture and EMs are increasing; however, Ems is often misdiagnosed as diseases such as trans pelvic inflammatory disease, ectopic pregnancy, and appendicitis[6].
With the development of laparoscopic technology, laparoscopic surgery has become the primary method for treating chocolate cyst rupture given its accurate diagnosis, symptom alleviation and success in adhesion separation and lesion elimination. This article analyzed the effect of laparoscopic surgery on a patient with ruptured ovarian cysts with the following report.
The patient was a 35-year-old unmarried female who denied a sexual history and presented with an intact hymen. Menophania occurred at 14 years of age, with a menstrual cycle of 30 d and a 7-10 d duration for each cycle. Breast swelling occurred prior to each period with no significant changes in leukorrhea. Positive results for polymenorrhea and dysmenorrhea were obtained, but the symptoms worsened over the prior four months, mostly at the beginning of each period. The previous menstrual period was on May 4, 2020, and the last menstrual period was on June 11, 2020, with the dysmenorrhea slightly improved over that of the previous month.
Beginning in February 2020, the patient started to experience recurrent left lower abdominal pain 2 d prior to and during the first 3 d of the period, which was asso
At 1 pm on June 16, the patient started to experience intermittent upper abdominal pain without inducement, associated with anal distension and fever of 38.3°C. There was no constipation, diarrhea, cold sweat, dizziness, or syncope.
Coronavirus disease 2019 was ruled out at the fever clinic.
Ultrasound examination revealed a dark cystic area measuring 4.9 cm × 4.6 cm on the left side with poor light transmittance, which suggested a left endometriotic cyst. Accordingly, the patient was given pain medications (four capsules t.i.d., p.o.). She returned to our clinic on June 10, 2020, and her period started the next day with sig
Left ovarian chocolate cyst rupture; EMs (stage III, ovarian type, peritoneal type).
With no pain relief and after diagnosis of left ruptured ovarian cyst, the patient was admitted to the hospital on June 17, and left ovarian cystectomy, electrocautery for the endometriotic lesions, myomectomy, and pelvic adhesion lysis were performed laparoscopically.
The patient was placed in a supine position, and the lower abdomen was routinely disinfected after successful anesthesia induction. A 1.0 cm transverse incision was made at the umbilicus, where a 10 mm trocar was placed after placement of a Veress needle followed by pneumoperitoneum with CO2. Under the direct inspection of a laparoscope, two 5 mm trocars were placed at McBurney’s point and its reflection on the left side, and a 10 mm trocar was placed approximately 3 cm from the left side of the umbilicus. The laparoscopic view revealed a normal-sized uterus, while there was a 1 cm myoma-looking lesion at the front wall. The left ovarian cyst had enlarged to approximately 5 cm× 6 cm × 5 cm, had ruptured with some active bleeding, and was closely attached to the posterior portion of the broad ligament, the posterior wall of the uterus and the intestines. Douglas' pouch was partially obstructed, the right ovary was slightly swollen, and there were multiple follicular cysts and blue-purple nodules on its surface. Extensive adhesion of some of the intestines to the posterior portion of the right broad ligament as well as the isthmus of the uterus was observed. The posterior wall of the uterus was covered with flocculent secretions, blue-purple nodules, and flame-shaped lesions. The American Society for Reproductive Medicine score was 36, and there was approximately 200 mL of free chocolate-colored fluid in the pelvic cavity. The bilateral fallopian tubes were soft with free and mildly swollen fimbriated extremities.
The surgical steps were as follows: (1) Pelvic blood was irrigated and evacuated with negative suction; (2) Blunt and sharp dissection were combined during adhesion lysis to rebuild the normal anatomy; an incision was made along ruptured end of the left ovarian cyst to fully remove the cyst wall, and it was closed with interrupted 3-0 absorbable sutures. The uterine fibroma at the front wall was resected, followed by electrocautery to the endometriosis focus. After complete excision of the specimen, it was found to consist of grayish, swirl-like fibroid tissue surrounded by a smooth cyst wall. The specimen was sent for surgical pathology after review with the patient’s family; (3) Thorough irrigation, suction of the abdominopelvic cavity, and satisfactory hemostasis were performed, and 3 pieces of soluble hemostatic gauze and 2 pieces of gel foam were left at the surgical field; and (4) The trocars were removed after desufflation, and the incisions were closed.
The surgery was uneventful, and the patient remained stable during the whole procedure. The total blood loss was approximately 5 mL; the urine output was 300 mL and clear. The patient was sent back to the ward safely after surgery.
Zoladex was provided as supplemental medical therapy after surgery, with one dose every 28 d (one cycle) via subcutaneous injection in the abdomen for 6 cycles.
A chemiluminescence immunoassay (DIX800 Chemiluminescence Immunoassay Ana
| May 9, 2020 | June 10, 2020 | June 16, 2020 | June 17, 2020 | June 18, 2020 | June 22, 2020 | |
| CA-125 | 41.65 U/mL | 31.09 U/mL | 723.29 U/mL | 125 448.32 U/mL | ||
| WBC | 13.88 × 109/L | 9.98 × 109/L | ||||
| hs-CRP | 8.6 mg/L | 3380 μg/L | ||||
| NEU | 88.7% | 79.0% | ||||
| D-D | 3380 μg/L | 2220 μg/L | ||||
| AMH | 3.09 ng/mL | |||||
| FSH | 8.50 IU/L |
June 15, 2020: The uterus was antepositioned, with normal size and shape, uniform echo in the myometrium, and a thickness of 0.97 cm. The right ovary presented with a normal size and shape and no abnormal echo. The left ovary was not clearly demon
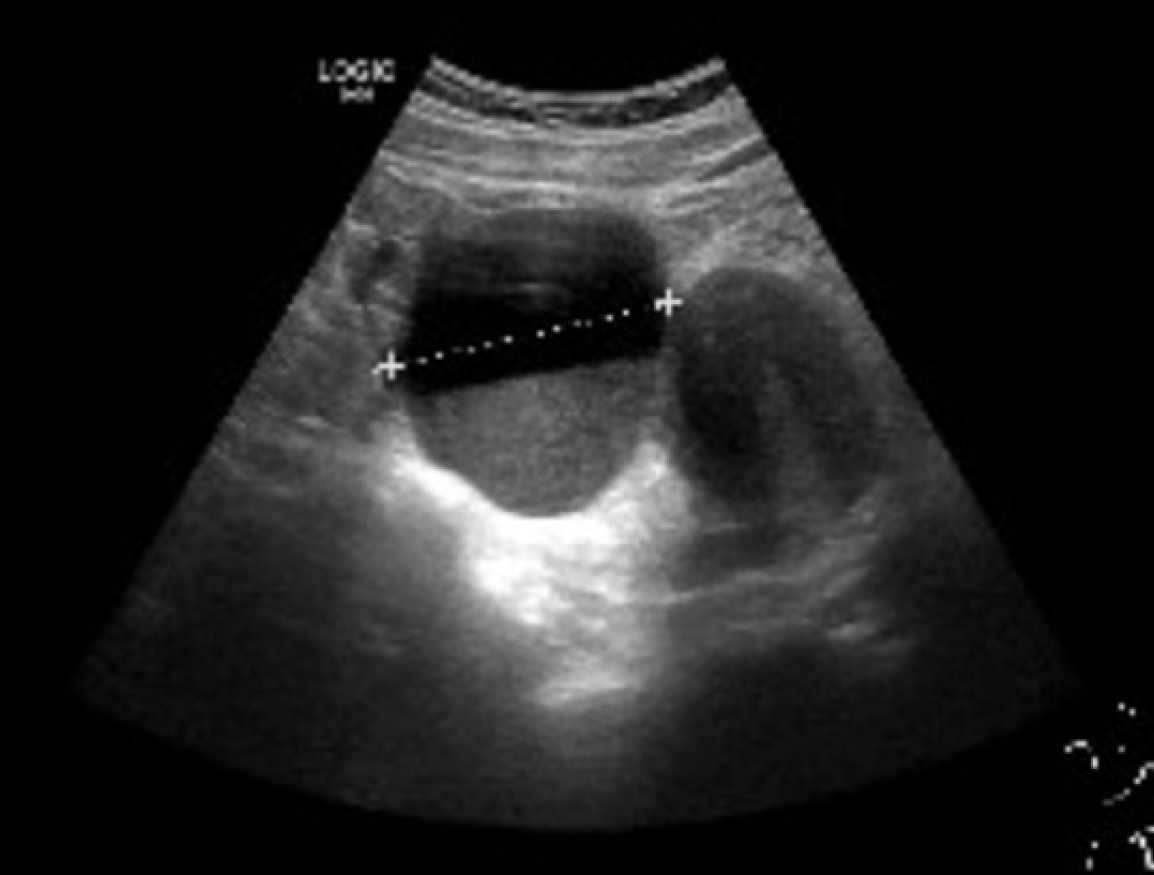
June 16, 2020: The uterus was retropositioned, with normal size, uniform echo in the myometrium, and a thickness of 0.64 cm. A cystic lesion measuring 2.1 cm × 0.9 cm × 1.9 cm was observed in the right ovary. A mixed echoic area measuring5.2 cm × 3.0 cm × 4.2 cm was identified on the left, with clear margins but an irregular shape. Dark fluid shadows were observed within, measuring approximately 4.2 cm × 3.0 cm × 3.8 cm, with poor transmittance. CDFI indicated blood flow in and around the mixed echoic area, and free fluid in the pelvis had a depth of approximately 2.0 cm, poor transmittance, and dense light spots. A 0.7-cm hyperechogenic spot was also observed in the gallbladder.
The uterus was considered in a normal state in terms of shape, size, and density. A low-density focus at the left ovarian area was observed, with a diameter of approximately 38 mm, clear margins, blurred mesentery fat plane, and spots with fluid density.
The mechanism behind the formation of chocolate cysts remains unclear but is currently thought to be secondary to the following process[7,8]. Pieces of endometrium travel back to the pelvic cavity with menses via the fallopian tubes and are seeded onto the ovarian surface; during each cycle and under the effect of estrogen, the pieces of endometrium seeded onto the ovary bleed and gradually form into a cyst when dra
The gold standard diagnostic tool for this condition is laparoscopy, which allows visualization of the chocolate fluid in the cyst under direct observation and resection of the cyst for final pathological diagnosis[10]. In our case, the postoperative diagnosis of EMs and ruptured chocolate cyst agreed with the pathology. Ultrasound allows only a rough evaluation of chocolate cysts, which may consequently result in the misdiagnosis of physiologic cysts that disappear when the period concludes as chocolate cysts. Moreover, small cysts likely shrink after taking birth control pills due to their inhibitory effects on the endometrium[11]. When our patient was found to have a 4.9 cm × 4.6 cm dark cystic area around the left ovary on ultrasound that suggested a potential left endometriotic cyst on May 9, 2020, she was treated with pain medication, which contributed to inhibiting endometrial growth and shrinking the cyst. However, a repeat ultrasound on June 15, 2020, revealed that the dark region had grown to a size measuring 5.66 cm × 6.58 cm× 5.79 cm; the patient refused the recommendation of surgical treatment and returned to the hospital with abdominal pain the next day. Abdominal CT suggested free fluid collection in the pelvis, and ultrasound was remarkable, revealing a 5.2 cm× 3.0 cm × 4.2 cm complex echo density around the left ovarian area as well as a 2.0-cm deep fluid pocket. The patient was admitted on the 17th day of June with a ruptured ovarian cyst, and surgical resection was completed with laparoscopy[12,13]. Once the disease is diagnosed, surgery should be performed promptly. The outflow of the contents of chocolate-like cysts after rupture results in secondary chemical peritonitis by irritation of the peritoneum, which may be fatal for the patient. Differentiation of rupture of ovarian ectopic cysts from other acute abdominal diseases, such as acute appendicitis, is imperative. The incidence of endometriosis comorbid with infertility is as high as 50%. The cyst content flows into the abdominal cavity after rupture, which may result in secondary adhesions and endometrial implantation without timely treatment, leading to further fertility damage. Therefore, timely and accurate diagnosis is paramount. If the cyst content outflow is insufficient to cause acute abdomen due to a small rupture, the patient might be asymptomatic. However, the inflammatory response could lead to the formation of adhesions in the surrounding region, and the cyst could undergo repetitive processes of shrinkage, regrowth, rupture, bleeding, and healing, imposing more complexity on the potential surgical treatment. Since ovarian endometrial cysts are often accompanied by symptoms such as abdominal pain and anal swelling during or near the menstrual period, a detailed medical history and gynecological exami
Chocolate cysts not only manifest with severe abdominal pain but also worsen pelvic adhesions and potential new EMs, eventually triggering infertility. For patients of childbearing age, conservative surgery to preserve reproductive and endocrine function is the treatment of choice. Surgical treatment, its promising therapeutic effect notwithstanding, is undermined by a high recurrence rate, especially conservative surgery and semi radical surgery. It has been reported that the recurrence rate of endometriotic cysts subjected to surgery is higher than that for unruptured patients, which emphasizes the importance of thorough irrigation of the abdominal cavity during surgery to prevent iatrogenic secondary seeding by the extensive contamination of the contents of the cyst in the pelvic cavity. In our case, consolidate treatment with Zoladex for 6 mo after surgery was applied to prevent recurrence. Laparoscopic surgery has now become the first treatment of choice with more ad
The carbohydrate antigen level increases to a lesser degree in the presence of benign tumors and inflammatory disease than in malignant conditions. Studies have also demonstrated that CA-125 Levels are increased in the presence of benign ovarian tumors[18,19]. Koo et al[20] reported lower levels of CA-125 after treatment in cho
We reported a case of a ruptured chocolate cyst. Ultrasound, abdominopelvic CT, and serum D-dimer and CA-125 were useful diagnostic tools, and laparoscopic surgery was confirmed to be safe for symptom control and effective disease eradication.
Manuscript source: Unsolicited manuscript
Specialty type: Obstetrics and gynecology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Darai E S-Editor: Wang JL L-Editor: A P-Editor: Wu RR
| 1. | Barra F, Scala C, Mais V, Guerriero S, Ferrero S. Investigational drugs for the treatment of endometriosis, an update on recent developments. Expert Opin Investig Drugs. 2018;27:445-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 37] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 2. | Tsonis O, Gkrozou F, Harrison E, Stefanidis K, Vrachnis N, Paschopoulos M. Female genital tract microbiota affecting the risk of preterm birth: What do we know so far? Eur J Obstet Gynecol Reprod Biol. 2020;245:168-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 3. | Irwin AL, Smith K, Sargant N. Ovarian cyst haemorrhage as a complication of acute myelomonocytic leukaemia induction therapy. Clin Med (Lond). 2019;19:509-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 4. | Ikeda M, Negishi Y, Akira S, Morita R, Takeshita T. Inflammation related to high-mobility group box-1 in endometrial ovarian cyst. J Reprod Immunol. 2021;145:103292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 5. | Park S, Lim W, Bazer FW, Song G. Apigenin induces ROS-dependent apoptosis and ER stress in human endometriosis cells. J Cell Physiol. 2018;233:3055-3065. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 49] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 6. | Gadot Y, Tsafrir Z, Hazan Y. EP31.23: Tailgut cyst mistaken for endometriosis: a case report. Ultrasound Obstet Gynecol. 2019;54. |
| 7. | Nagira K, Taniguchi F, Nakamura K, Tokita Y, Tsuchiya N, Khine YM, Harada T. Tokishakuyakusan, a Kampo medicine, attenuates endometriosis-like lesions and hyperalgesia in murine with endometriosis-like symptoms. Am J Reprod Immunol. 2019;82:e13182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 8. | Koga K, Osuga Y, Takemura Y, Takamura M, Taketani Y. Recurrence of endometrioma after laparoscopic excision and its prevention by medical management. Front Biosci (Elite Ed). 2013;5:676-683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 9. | Posadzka E, Nocuń A, Jach R, Nessler M, Nessler K, Kiałka M. Assessment of ovarian reserve in patients with ovarian endometriosis following laparoscopic enucleation of a cyst accompanied by CO₂ laser ablation or electroablation. Przegl Lek. 2016;73:6-10. [PubMed] |
| 10. | Kim SM, Hwang KA, Go RE, Sung JH, Choi DW, Choi KC. Exposure to cigarette smoke via respiratory system may induce abnormal alterations of reproductive organs in female diabetic rats. Environ Toxicol. 2019;34:13-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | Shaltout MF, Elsheikhah A, Maged AM, Elsherbini MM, Zaki SS, Dahab S, Elkomy RO. A randomized controlled trial of a new technique for laparoscopic management of ovarian endometriosis preventing recurrence and keeping ovarian reserve. J Ovarian Res. 2019;12:66. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 12. | Nowak-Psiorz I, Ciećwież SM, Brodowska A, Starczewski A. Treatment of ovarian endometrial cysts in the context of recurrence and fertility. Adv Clin Exp Med. 2019;28:407-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 13. | Gonzalo-Carballes M, Ríos-Vives MÁ, Fierro EC, Azogue XG, Herrero SG, Rodríguez AE, Rus MN, Planes-Conangla M, Escudero-Fernandez JM, Coscojuela P. A Pictorial Review of Postpartum Complications. Radiographics. 2020;40:2117-2141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 14. | Hong YL, Tan Y, Yin YY, Zou YJ, Guo YH, Nie XW. [Effect of electro-acupuncture on clinical outcomes and ovarian hyperstimulation syndrome in in vitro fertilization and embryo transplantation]. Zhongguo Zhong Xi Yi Jie He Za Zhi. 2014;34:1292-1296. [PubMed] |
| 15. | Cansu A, Bulut E, Dinc G, Bekircavusoglu S, Eyuboglu I, Guven ES, Ahmetoglu A. Diagnostic Efficacy of T2 Dark Spot, T2 Dark Rim Signs, and T2 Shading on Magnetic Resonance Imaging in Differentiating Endometriomas From Hemorrhagic Cysts. J Comput Assist Tomogr. 2019;43:619-622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 16. | Pankhurst MW, Kelley RL, Sanders RL, Woodcock SR, Oorschot DE, Batchelor NJ. Anti-Müllerian hormone overexpression restricts preantral ovarian follicle survival. J Endocrinol. 2018;237:153-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 17. | Bonci EA, Buiga R, Badan M, Iuliana Maja L, Gata VA, Lisencu IC, Irimie A, Achimas Cadariu P, Piciu D. Follicle-stimulating hormone receptors: A comparison of commercially-available monoclonal and polyclonal antibodies as immunohistochemical markers for cancer research. J BUON. 2018;23:1912-1921. [PubMed] |
| 18. | Monteiro S, Franco F, Costa S, Monteiro P, Vieira H, Coelho L, Oliveira L, Providência LA. Prognostic value of CA125 in advanced heart failure patients. Int J Cardiol. 2010;140:115-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 19. | Markman M. Prolonged symptom-free survival in a patient with persistent primary peritoneal carcinoma and a rising CA-125: a note of caution. Gynecol Oncol. 2001;81:509-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 20. | Koo JH, Lee I, Han K, Seo SK, Kim MD, Lee JK, Kwon JH, Kim GM, Lee J, Won JY. Comparison of the therapeutic efficacy and ovarian reserve between catheter-directed sclerotherapy and surgical excision for ovarian endometrioma. Eur Radiol. 2021;31:543-548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |