Published online May 16, 2021. doi: 10.12998/wjcc.v9.i14.3294
Peer-review started: September 19, 2020
First decision: January 7, 2021
Revised: January 21, 2021
Accepted: March 24, 2021
Article in press: March 24, 2021
Published online: May 16, 2021
Processing time: 221 Days and 20.4 Hours
Cluster headache (CH) is a severe incapacitating headache disorder. By definition, its diagnosis must exclude possible underlying structural conditions.
To review available information on CLH caused by structural lesions and to provide better guides in the distinguishing process and to ensure that there is not a potentially treatable structural lesion.
We conducted a systematic review of 77 published cases of symptomatic CH and cluster-like headache (CLH) in PubMed and Google Scholar databases.
Structural pathologies associated with CH were vascular (37.7%), tumoral (32.5%) and inflammatory (27.2%). Brain mass-like lesions (tumoural and inflammatory) were the most common diseases (28.6%), among which 77.3% lesions were at the suprasellar (pituitary) region. Cases of secondary CH related to sinusitis rose dramatically, occupying 19.5%. The third most common disease was internal carotid artery dissection, accounting for 14.3%. Atypical clinical features raise an early suspicion of a secondary cause: Late age at onset and eye and retroorbital pains were common conditions requiring careful evaluation and were present in at least one-third of cases. Abnormal neurological examination was the most significant red flag for impaired cranial nerves. CLH patients may be responsive to typical CH treatments; therefore, the treatment response is not specific. CLH can be triggered by contralateral structural pathologies. CLH associated with sinusitis and cerebral venous thrombosis required more attention.
Since secondary headache could perfectly mimick primary CH, neuroimaging should be conducted in patients in whom primary and secondary headaches are suspected. Cerebral magnetic resonance imaging scans is the diagnostic management of choice, and further examinations include vessel imaging with contrast agents and dedicated scans focusing on specific cerebral areas (sinuses, ocular and sellar regions). Neuroimaging is as necessary at follow-up visits as at the first observation.
Core Tip: Secondary headache could perfectly mimick primary cluster headache, hence neuroimaging should be conducted in patients in whom primary and secondary headaches are suspected. Cerebral magnetic resonance imaging scans are the diagnostic management of choice, and further examinations include vessel imaging with contrast agents and dedicated scans focusing on specific cerebral areas (sinuses, ocular and sellar regions). Neuroimaging is as necessary during follow-up as at the first observa
- Citation: Long RJ, Zhu YS, Wang AP. Cluster headache due to structural lesions: A systematic review of published cases. World J Clin Cases 2021; 9(14): 3294-3307
- URL: https://www.wjgnet.com/2307-8960/full/v9/i14/3294.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i14.3294
Cluster headache (CH) is classified together with paroxysmal hemicrania, short-lasting neuralgiform headache attacks and hemicrania continua as trigeminal autonomic cephalalgias, which are characterized by unilateral episodes of headache in the trigeminal nerve distribution and ipsilateral cranial sympathetic autonomic featu
CH has a period of active cluster bouts when it tends to occur, lasting from 15 to 180 min, with a frequency of one every other day up to eight per day. In episodic CH, the bouts are followed by remission periods; in contrast, there is no remission or a remission of less than 3 mo in chronic CH[1,2]. Other distinguishing features of CH include male prevalence and circadian periodicity[3].
Despite its relatively low incidence in the population, the excruciating headache intensity of CH makes it a severely incapacitating headache disorder that substantially impairs social activities and quality of life[4,5].
However, a structural lesion may be the cause of the headache, and patients with cluster-like headache (CLH) associated with structural lesions have been reported. Accordingly, these findings raise the question of how to search for an underlying lesion. Indeed, how can we obtain the most benefit for patients, i.e. achieve high diagnostic standards at comparably low cost?
This systematic review seeks to contribute to the knowledge on CLH caused by structural lesions and to provide better guides in the distinguishing process.
A systematic review was conducted to provide an overview of published cases of symptomatic CH and to examine the demographics and clinical characteristics of the patients.
This review was carried out according to the Cochrane Collaboration methodology and is reported according to the PRISMA statement. Relevant articles were identified by searching PubMed and Google Scholar databases using the keywords "secondary cluster headache", "symptomatic cluster headache", "cluster-like headache", "mimicking cluster headache", "causing cluster headache", "presenting cluster headache" and "cluster headache due to". The search of all published English articles of case report was performed in January 2021.
Published articles of case report fulfilling the following criteria were included: (1) CLH presented as the initial main symptom; (2) Headache disappeared instantly after treatment for structural pathologies; and (3) At least a 6-mo follow-up period without attacks. The data obtained included age at onset and diagnosis, sex, headache side and site, attack duration and frequency, accompanying cranial autonomic symptoms, response to typical CH treatment and other associated features.
Exclusion criteria were as follows: (1) CLH did not present as the initial main symptom; (2) Structural pathologies not treated, attacks remained after treating structural pathologies or patient died within 6 mo; (3) Associated with systemic diseases; (4) Associated with head injuries; (5) Associated with surgeries; and (6) Associated with particular triggering factors.
A data abstraction form was devised based on the Cochrane's recommendations. Abstracts identified from searches were screened by two independent authors. Both authors reviewed full-text versions of the articles and articles were retained if they met inclusion criteria. Data extracted from included articles comprised: Authors and date of studies; demographics of patients (age, sex); clinical characteristics (structural pathologies, ages of headache onset, right or left side, headache regions, positional relation of headache and pathologies, headache attack duration and frequency, accompanying symptoms, conditions in intervals, physical and neurological examinations). All data required to answer the study questions were published within the articles, so no contact with authors was necessary. The study did not involve contact with humans, so the ethical approval was not required.
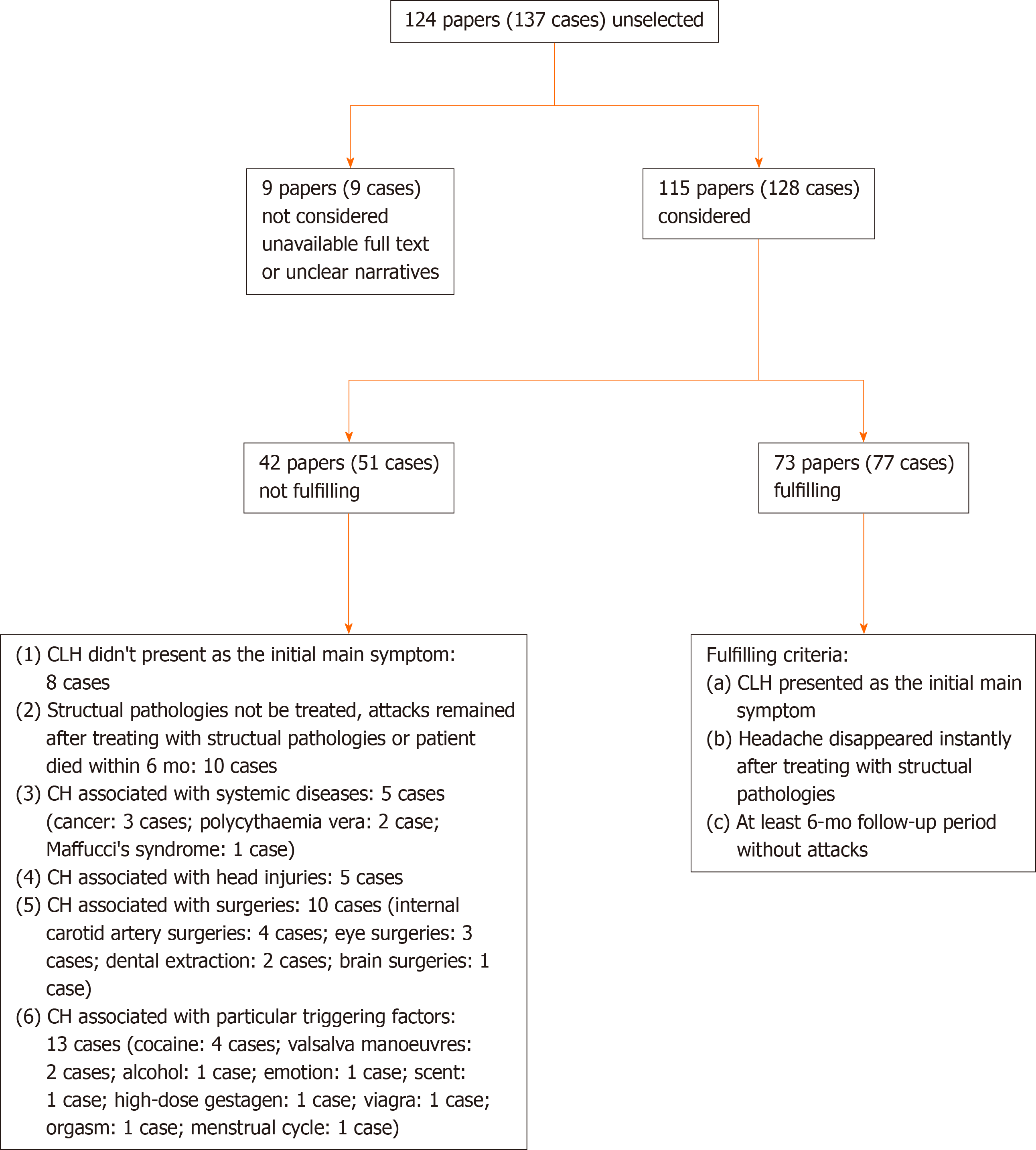
A total of 124 papers (137 cases) were published between 1988 and January 2021. Seventy-seven cases were in accordance with the inclusion criteria. The screening process and results of selection are summarized in a flow chart (Figure 1).
Vascular pathologies (37.7%, n = 29) were the first cause of secondary CH: Internal carotid artery dissection (ICAD) (11 cases), intracranial aneurysm (6 cases), cerebral venous thrombosis (CVT) (5 cases), arteriovenous malformation (3 cases), subclavian artery subclavian steal phenomenon (1 case), vertebral artery dissection (1 case), intra-cavernous internal carotid artery thrombosis (1 case) and dural arteriovenous fistulas (1 case) (Table 1).
| Year | Ref. | Pathologies | Age at onset, interval between age at onset and diagnosis, yr | Sex |
| 1997 | Rosebraugh et al[33] | ICAD | 34 | Male |
| 2003 | Frigerio et al[34] | ICAD | 50 | Female |
| 2005 | Hannerz et al[16] | ICAD | 58 | Male |
| 2006 | Razvi et al[35] | ICAD (in the petrous segment of ICA) | 44 | Male |
| 2007 | Hardmeier et al[36] | ICAD (petrous segment) | 38 | Male |
| 2007 | Straube et al[13] (case 2) | ICAD (cavernous segment) | 35 | Male |
| 2008 | Rigamonti et al[37] (cases 1 and 2) | ICAD (case 1, lacerum segment) | 50 | Male |
| ICAD (case 2, lacerum segment) | 49 | Male | ||
| 2008 | Godeiro-Junior et al[38] | ICAD (petrous segment) | 53 | Male |
| 2008 | Tobin et al[39] | ICAD (cervical segment) | 55 | Male |
| 2013 | Tsivgoulis et al[40] | ICAD (petrous segment) | 32 | Male |
| 1991 | West et al[17] | Aneurysm (posterior inferior cerebellar artery, vertebral artery) | 39 (12) | Male |
| 2000 | McBeath et al[31] | Aneurysm (posterior communicating artery) | 45 (10) | Male |
| 2006 | Gentile et al[11] | Aneurysm (multiple cerebral aneurysms) | 58 | Female |
| 2007 | Valença et al[41] (cases 1 and 2) | Aneurysm (case 1, posterior communicating artery) | 47 | Male |
| Aneurysm (case 2, intracranial ICA) | 57 | Male | ||
| 2009 | Sewell et al[42] | Aneurysm (moyamoya disease) | 18 (16) | Male |
| 2006 | Georgiadis et al[25] | CVT (superior sagittal sinus) | 46 | Male |
| 2006 | Park et al[26] | CVT (right transverse sinus, straight sinus) | 20 (6) | Male |
| 2006 | Peterlin et al[8] | CVT (case 1, left transverse, sigmoid sinus) | 38 (9) | Male |
| CVT (case 2, left transverse sinus, sigmoid sinus, internal jugular vein) | 32 (10) | Male | ||
| 2008 | Rodríguez et al[18] | CVT (superior sagittal sinus and bilateral transversal sinuses) | 51 | Male |
| 1982 | Mani et al[43] | Arteriovenous malformation (occipital lobe) | 22 (14) | Female |
| 1996 | Muñoz et al[19] | Arteriovenous malformation (case 1, temporal lobe) | 52 | Male |
| Arteriovenous malformation (case 2, frontal lobe) | 40 (6) | Male | ||
| 2001 | Piovesan et al[44] | Subclavian steal phenomenon | 51 (9) | Female |
| 2008 | Kim et al[45] | Vertebral artery dissection (posterior inferior cerebellar artery) | 48 | Female |
| 2008 | Ashkenazi et al[20] | Intra-cavernous carotid artery thrombosis | 62 | Male |
| 2017 | Chang et al[46] | Middle meningeal artery dural arteriovenous fistulas | 42 | Male |
Secondary CH cases associated with tumoural pathologies were 32.5% (n = 25), including prolactinoma (8 cases), meningioma (4 cases), epidermoid cyst (3 cases), nonfunctioning pituitary adenoma (2 case), Arnold–Chiari type I malformation (2 cases), arachnoid cyst (1 case), glioblastoma multiforme (1 case), pituitary cyst (1 case), pituitary growth hormone-producing adenoma (1 case), inflammatory myofibroblastic tumour (1 case), and paraganglioma around the internal carotid artery in neck (1 case) (Table 2).
| Year | Ref. | Pathologies | Age at onset (Interval between age at onset and diagnosis, yr) | Sex |
| 1988 | Greve et al[47] (case 3) | Prolactinoma | 57 (1) | Male |
| 2001 | Porta-Etessam et al[48] | Prolactinoma | 28 (8) | Male |
| 2004 | Leone et al[9] | Prolactinoma | 46 (3) | Male |
| 2005 | Negoro et al[27] | Prolactinoma | 14 (3) | Male |
| 2012 | Levy et al[49] | Prolactinoma | 25 | Male |
| 2013 | Edvardsson et al[50] | Prolactinoma | 46 | Male |
| 2017 | Andereggen et al[51] | Prolactinoma | 46 | Male |
| 2017 | Pineyro et al[52] | Prolactinoma | 15 (3) | Female |
| 1991 | Levyman et al[53] | Epidermoid cyst (posterior fossa) | 40 (13) | Female |
| 2006 | Massie et al[10] | Epidermoid cyst (epidermoid clival lesion) | 36 (3) | Male |
| 2007 | Eimil-Ortiz et al[28] | Epidermoid cyst (pontocerebellar angle) | 47 | Male |
| 1989 | Hannerz et al[21] | Parasellar meningioma | 28 (18) | Male |
| 1995 | Taub et al[6] | Meningioma (tentorium cerebelli) | 35-40 (20-25) | Male |
| 2003 | Bigal et al[7] | Inflammatory myofibroblastic tumour (undersurface of the tentorium) | 18.5 (9.5) | Male |
| 2008 | Alty et al[54] | Trigeminal meningioma (cerebellopontine angle) | 30 | Male |
| 2009 | Robbins et al[55] | Meningioma (planum sphenoidale) | 29 (8) | Male |
| 2012 | Edvardsson et al[22] | Parietal glioblastoma multiforme | 41 | Male |
| 2013 | Edvardsson et al[56] | Supra- and intrasellar arachnoid cyst | 43 | Male |
| 1982 | Tfelt-Hansen et al[57] | Pituitary chromophobe adenoma | 21 (31) | Male |
| 2014 | Edvardsson et al[58] | Pituitary chromophobe adenoma | 49 | Male |
| 1996 | Milos et al[59] | Pituitary GH-producing adenoma | 33 (4) | Male |
| 2016 | De Pue et al[32] | Pituitary cyst | 35 (12) | Male |
| 2004 | Seijo-Martinez et al[12] | Arnold–Chiari type I malformation, syringomyelia | 36 | Female |
| 2015 | Kao et al[60] | Arnold–Chiari type I malformation | 26 | Male |
| 2014 | Malissart et al[61] | Carotid paraganglioma | 60 | Female |
Inflammatory pathologies were comparatively less frequent causes, amounting to 27.2% (n = 21): Sinusitis (13 cases), idiopathic orbital myositis (2 cases), pituitary granuloma (2 cases), sinuses mucocele (2 cases), inflammatory orbital pseudotumour (1 case), and posterior scleritis (1 case). In addition, there were 2 cases caused by idiopathic intracranial hypertension but without the discovery of pathological origins (Table 3).
| Year | Ref. | Pathologies | Age at onset (Interval between age at | Sex |
| 1988 | Takeshima et al[62] | Sinusitis (case1, frontal, ethmoidal, maxillary sinusitis) | 31 | Male |
| Sinusitis (case 2, frontal, maxillary sinusitis) | 31 (9) | Male | ||
| 1995 | Zanchin et al[14] | Sinusitis (sphenoid sinus aspergillus infection) | 64 | Female |
| 1997 | Heidegger et al[63] | Sinusitis (sphenoid sinus aspergillus infection) | 68 | Male |
| 2002 | Scorticati et al[23] | Sinusitis (foreign body in the maxillary sinus, maxillary sinusitis) | 26 (8) | Female |
| 2013 | Edvardsson et al[64] | Sisitis (maxillary sinusitis) | 24 | Male |
| 2013 | Edvardsson et al[65] | Sinusitis (maxillary sinusitis) | 21 | Male |
| 2018 | Balgetir et al[66] (cases 1-7) | Sinusitis (frontal, ethmoid, maxillary sinusitis) | 23 | Male |
| Sinusitis (frontal, maxillary sinusitis) | 28 | Male | ||
| Frontoethmoidal mucocele | 33 | Male | ||
| Sinusitis (frontal, ethmoid, maxillary sinusitis) | 27 | Male | ||
| Sinusitis (maxillary, ethmoid sinusitis) | 16 | Male | ||
| Sinusitis (frontal sinusitis) | 31 | Male | ||
| Sinusitis (sphenoid and ethmoid sinusitis) | 39 | Female | ||
| 2018 | Branco et al[67] | Sphenoid sinus mucocele | 62 | Male |
| 2007 | Harley et al[68] | Inflammatory orbital pseudotumour | 33 | Female |
| 2009 | Choi et al[69] | Posterior scleritis | 42 (10) | Female |
| 2017 | Ersoy et al[70] | Idiopathic orbital myositis | 34 | Female |
| 2019 | Douglas et al[71] | Idiopathic orbital myositis | 19 | Female |
| 2007 | Favier et al[15] | Hypothalamus-pituitary granuloma | 26 | Female |
| 2013 | van der Vlist et al[72] | Hypothalamus-pituitary granuloma | 31 | Male |
| 2006 | Volcy et al[73] | Idiopathic intracranial hypertension | 40 | Male |
| 2008 | Testa et al[74] | Idiopathic intracranial hypertension | 28 | Female |
Two cases were ruled out because of an unclear description of age[6,7], and the average age of symptom onset was 38.8 ± 13.0 years. Forty patients (51.9%) were between 20-40-years-old, six (7.8%) were younger than 20-years-old, and 31 (40.3%) were older than 40-years-old. Sixty (77.9%) patients were male and 17 (22.1%) female; the male: Female ratio was 3.53: 1 (60: 17).
CH was strictly unilateral in all patients, with 43 (55.8%) left-sided and 34 (44.2%) right-sided cases. Structural pathologies were ipsilateral to the headache in 61 (79.2%), contralateral in 4 (5.2%) (Case 1)[2,8-10], midline (no visible invasion to the left or right side on neuroimaging) in 10 (13.0%), and unknown in 2[11,12].
Information about attack duration was missing in 4 cases[7,13-15]. For the remaining 73 cases, 56 patients experienced attacks lasting 15–180 min, and duration was unclear for the other 17 (23.3%) cases. Twelve cases lacked accurate information about attack frequency[7,13-23]; sixty of the remaining sixty-five cases had an attack frequency between one every other day and eight per day, and five (7.7%) did not fulfil the frequency criterion.
The headache predominantly affected orbital and periorbital regions in 47 cases (61.0%), temporal regions in 23 (30.0%), and supraorbital regions in 16 (20.1%). Additionally, 26 patients (33.8%) described pain in the eye and retroorbital regions; 14 (18.2%) described pain in other sites. Regarding accompanying autonomic symptoms, 73 patients (94.5%) reported lacrimation, conjunctival injection, and rhinorrhoea; 40 patients (51.2%) reported miosis and ptosis; and 39 patients (50.6%) reported nasal congestion, eyelid oedema, and forehead sweating. In addition to typical CH autonomic symptoms, 13 patients (16.9%) described migraine-like symptoms of nausea, phonophobia and vomiting. Persistent miosis and/or ptosis were observed in 11 patients (14.3%). During the intervals between attacks, 18 patients (23.4%) reported additional headaches of different intensities and characteristics. Altered neurological findings were found in 21 (27.2%) patients (Table 4).
| Number (%)/mean ± range | |
| Age at onset (n = 66) | |
| Average age | 38.3 ± 13.0 |
| 20-40 yr | 40 (51.9%) |
| < 20 yr | 6 (7.8%) |
| > 40 yr | 31 (40.3%) |
| Sex | |
| Male | 60 (77.9%) |
| Female | 17 (22.1%) |
| Side | |
| Left | 43 (55.8%) |
| Right | 34 (44.2%) |
| Sides of headache and structural pathologies (n = 66) | |
| Ipsilateral | 61 (79.5%) |
| Midline | 10 (13.0%) |
| Contralateral | 4 (5.2%) |
| Unknown | 2 (2.6%) |
| Attacks not fulfilling ICHD-3 | |
| Duration (n = 65) | 17 (23.3%) |
| Frequency (n = 57) | 5 (7.7%) |
| Headache sites | |
| Orbital/periorbital | 47 (61.0%) |
| Temporal | 23 (30.0%) |
| Supraorbital | 16 (20.1%) |
| Eye/retroorbital | 26 (33.8%) |
| Other | 14 (18.2%) |
| Associated autonomic symptoms | |
| Lacrimation/conjunctival injection/rhinorrhoea | 73 (94.5%) |
| Miosis/ptosis | 40 (51.2%) |
| Nasal congestion/eyelid oedema/forehead sweating | 39 (50.6%) |
| Nausea/phonophobia/vomiting | 13 (16.9%) |
| Persistent miosis/ptosis | 11 (14.3%) |
| Additional headaches between attacks | 18 (23.4%) |
| Altered neurological examination | 21 (27.2%) |
When establishing a diagnosis of secondary headache, an important causal relationship criterion is to confirm the disappearance or clear relief of headache after the associated condition is removed. In our study, we excluded associated conditions like systemic diseases, injuries and surgeries because the pathologies cannot be removed, in general terms. Associated conditions of specific triggering factors (drug, emotion, movement) were also excluded, for the same reason. Cases of CLH presenting as an accompanying symptom, which means other clear defect symptoms of nervous system lesions occur in the meantime, were also ruled out of the analysis because they were less likely to be confused with primary CH. Therefore, we selected the patients that were both strong "confusing" and in a strong causal link with structural lesions and focused the evaluation on these patients.
CLH-associated conditions: Vascular pathologies (37.7%, n = 29) and tumoural pathologies (32.5%, n = 25) were the most common in CLH patients, followed by inflammatory pathologies (27.2%, n = 21). A previous comprehensive reappraisal of CLH in 2010[24] reported that pathologies associated with CLH were the vascular ones (38.5%), followed by tumours (25.7%) and inflammatory infectious diseases (13.5%). The review also included post-traumatic and other neurological, iatrogenic and dental pathologies in the analysis, which may explain the different figures comparing to our study.
Brain mass-like lesions (tumoural and inflammatory) were the most common diseases (28.6%, n = 22), among which 77.3% (17 in 22) of lesions were at the suprasellar (pituitary) region. The second most common diseases was ICAD, accounting for 14.3% (n = 11) of CLH patients; all the dissections were situated at the extracranial segments of internal carotid artery. An interesting finding is that the figures of CLH related to sinusitis were dramatically higher than a previous report; 15 cases (19.5%) in our study compared to only six cases prior[24], which appears to indicate increased attention related to sinusitis causing CLH. It must be noted that a lack of contributing factors of coagulopathies cannot exclude CVT. In the 3 cases of CVT patients for whom coagulopathies were examined, evidence of coagulopathies was found in none[18,25,26], which may indicate a potential higher incidence of CVT in CLH-associated conditions.
Structural pathologies in CLH were mostly located in the ipsilateral hemisphere; however, under certain circumstances, CLH may be triggered by contralateral structural pathologies (case 1)[2,8,27,28]. Explanations might be as follows: (1) Fos-producing neurons in the dorsal horns of C1 and C2 produce Aδ and C fibres (nociceptive input tracts) that innervate the majority of ipsilateral pain-producing structures and a few contralateral structures[29]. Due to anatomical variation of input fibres or specific anatomical sites, the trigeminocervical complex is sensitive to contralateral nociceptive inputs via the few contralateral Aδ and C fibres; (2) The contralateral mass effect might occur through traction of meningeal structures, producing trigeminal nerve enlargement and trigeminovascular system activation; and (3) Coexistence of contralateral structural pathologies and headache, which is possible but unlikely; the CLH disappears after treatment for structural pathologies is started, strongly indicating causality. CLH with midline structural pathologies may be explained by mild ipsilateral invasions that are not well distinguishable on neuroim
Age at onset and gender: Primary CH usually occurs between the second and fourth decades[1]. The average age at onset of CLH in our study was 38.8 ± 13.0 years; however, 40.3% of the patients were older than 40 years. It has been suggested that the late age at onset requires attention for structural pathological causes in the previous review[24], which is in accordance with our study that older age at onset is the most frequent "red flag".
The sex ratio of secondary CH was calculated to be 3.53:1, which is close to the ratio of primary CH; males experience CH three times more often than females[1]. The prevalence of male sex in secondary CH is unexpected because the structural causes as a whole are not sex-related. Mainardi et al[24] explained the male prevalence by possible anatomical and physiological differences of the hypothalamus in males and females, whereby males possess a greater volume of suprachiasmatic nucleus than do females, which makes males more susceptible[30].
Suspect elements: Atypical headache features can help raise an early suspicion of a secondary cause. The red flags are, in descending order of occurrence, attacks with eye and retroorbital pain (33.8%), altered neurological examinations (27.2%), additional headache between attacks (23.4%), atypical attack duration (23.3%), migraine-like automatic symptoms (16.9%), persistent partial Horner's syndrome (14.3%) and atypical attack frequency (7.7%). An altered neurological examination is the most significant red flag, which highlights impaired cranial nerves and nuclei, among which ophthalmic symptoms and signs are the most frequent, including impaired vision and visual field, diplopia, ocular motility disorders and dysfunctional pupil reflexes.
Attacks with eye and retroorbital pain are described in 33.8% of CLH cases, and they are particularly common in pituitary pathologies (64.3%, 9 in 14), ICAD (63.6%, 7 in 11) and eye diseases (75%, 3 in 4). Another particular "red flag" for ICAD is persistent miosis or ptosis, which is present in 72.7% of cases: 8 in 11. Haematoma in the vessel wall may damage the structure of the sympathetic fibres that travel along the carotid artery, resulting in unresolved sympathetic deficit symptoms between attacks. CH is a strictly paroxysmal cephalalgia in which patients are normal between attacks. Additional headaches during the intervals may suggest a nociceptive input of a different type, representing an element that requires attention. Additional headaches of different characteristics were observed in 23.4% of patients.
Responses to typical CH treatments: Resistance to typical CH treatment should raise suspicion of a secondary origin; however, CLH may be responsive (Table 5), particularly to acute treatment medications, with 46.9% effective responses for acute treatments. In 35.8% of responses, preventive treatments were reported to be effective. Nonetheless, the response to one drug can be completely opposite, even in two cases of the same category of structural origin. This may be due to different formulations, dosages, doses or delivery approaches. There is evidence that a CLH patient with pituitary prolactinoma did not respond to verapamil 80 mg but did respond to verapamil 120 mg three times a day[9]. Therefore, it must be stressed that response to CH treatment is not a sufficiently reliable criterion to discriminate between CH and CLH.
| Effectivenessa (n, %) | |
| Acute treatment | |
| Triptans | 19/28 (67.9%) |
| Oxygen inhalation | 13/24 (54.2%) |
| Ergotamine/ergometrine | 6/20 (30%) |
| NSAIDs | 7/31 (22.6%) |
| Analgesics | 6/8 (75%) |
| Caffeine | 2/2 (100%) |
| Total | 53/113 (46.9%) |
| Preventive treatment | |
| Verapamil | 9/17 (52.9%) |
| Glucocorticoid drugs | 11/15 (73.3%) |
| Propranolol | 1/6 (16.7%) |
| Antipsychotics | 2/11 (18.2%) |
| AEDs | 0/13 (0) |
| Lithium carbonate | 1/1 |
| Other antihypertensive drugs | 0/2 (0) |
| Antihistamine | 0/2 (0) |
| Total | 24/67 (35.8%) |
Research implications: Neuroimaging can confirm the diagnosis of underlying structural lesions, which should be performed in patients in whom both primary and secondary headaches are suspected. Brain magnetic resonance imaging (MRI) scans should be the method choice because they can detect most pathologies.
Vascular pathologies normally cannot be revealed by MRI interpretations, or they might present slight and unspecific signs on MRI scans. Computed tomography angiography and magnetic resonance venography are precise diagnostic tools for vascular pathologies. Other investigations include dedicated MRI scans focusing on particular areas, especially the sinuses and the ocular and sellar regions.
There is a remarkable finding that neuroimaging results may be normal in the headache trajectory before diagnosis[9,17,27,31,32]. Explanations could be minor structural lesions at that time that were not visible or because of low resolutions of neuroimaging. As a result, neuroimaging should be considered during the follow-up period in primary CH patients. Changes in headache characteristics or resistance to treatment may indicate progression of structural lesions and especially indicate repeated neuroimaging.
Our review has limitations. Reporting bias and publication bias cannot be overlooked in our review because we selected the published cases of CLH as samples. Rare causes for secondary CH have a higher chance of being published. Our findings must be interpreted within the context of the limitations and further widely prospective studies need to be conducted to verify the results.
As in all primary headaches, the diagnosis of primary CH must be excluded to rule out underlying causal conditions. This review provides a summary of clinical features of secondary CH cases and highlight imaging examinations in the diagnostic process. There are important implications for the application of these findings into clinical practice to achieve accurate and efficient diagnosis of CH.
Among the primary headaches, cluster headache (CH) presents very particular features allowing a relatively easy diagnosis based on criteria listed in Chapter 3 of the International Classification of Headache Disorders. However, as in all primary headaches, possible underlying causal conditions must be excluded to rule out a secondary cluster-like headache (CLH).
The review aims to contribute to the knowledge on CLH caused by structural lesions and to provide better guides in the distinguishing process.
We analysed the published cases of symptomatic CH and CLH in PubMed and Google Scholar databases.
We conducted a systematic review of published cases of symptomatic CH and CLH and analysed the features of the patients.
Structural pathologies associated with CH were vascular (37.7%), tumoural (32.5%) and inflammatory (27.2%). Patients with atypical clinical features require careful evaluation for structural origins. CLH patients may be responsive to typical CH treatments; the treatment response is not specific. CLH can be triggered by contralateral structural pathologies. CLH-associated sinusitis and cerebral venous thrombosis are worth paying attention to.
Secondary headaches could perfectly mimick primary CH, hence neuroimaging should be conducted in patients in whom primary and secondary headaches are suspected. Cerebral magnetic resonance imaging scans are the diagnostic management of choice, and further examinations include vessel imaging with contrast agents and dedicated scans focusing on specific cerebral areas (sinuses, ocular and sellar regions). Neuroimaging is as necessary at follow-up visits as at the first observation.
We investigated the clinical features of CLH patients in published cases and tried to summarize their distinctive characteristics by comparing to patients with primary CH.
Manuscript source: Unsolicited manuscript
Specialty type: Neurosciences
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Sobanski T S-Editor: Zhang H L-Editor: Filipodia P-Editor: Li JH
| 1. | Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia. 2013;33:629-808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5134] [Cited by in RCA: 5447] [Article Influence: 495.2] [Reference Citation Analysis (0)] |
| 2. | Newman LC. Trigeminal Autonomic Cephalalgias. Continuum (Minneap Minn). 2015;21:1041-1057. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 3. | Hoffmann J, May A. Diagnosis, pathophysiology, and management of cluster headache. Lancet Neurol. 2018;17:75-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 187] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 4. | Wilbrink LA, Louter MA, Teernstra OPM, van Zwet EW, Huygen FJPM, Haan J, Ferrari MD, Terwindt GM. Allodynia in cluster headache. Pain. 2017;158:1113-1117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 5. | Schor LI. Cluster headache: investigating severity of pain, suicidality, personal burden, access to effective treatment, and demographics among a large International survey sample. Cephalalgia. 2017;37:172. [DOI] [Full Text] |
| 6. | Taub E, Argoff CE, Winterkorn JM, Milhorat TH. Resolution of chronic cluster headache after resection of a tentorial meningioma: case report. Neurosurgery. 1995;37:319-21; discussion 321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 7. | Bigal ME, Rapoport AM, Camel M. Cluster headache as a manifestation of intracranial inflammatory myofibroblastic tumour: a case report with pathophysiological considerations. Cephalalgia. 2003;23:124-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 8. | Peterlin BL, Levin M, Cohen JA, Ward TN. Secondary cluster headache: a presentation of cerebral venous thrombosis. Cephalalgia. 2006;26:1022-1024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 9. | Leone M, Curone M, Mea E, Bussone G. Cluster-tic syndrome resolved by removal of pituitary adenoma: the first case. Cephalalgia. 2004;24:1088-1089. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 10. | Massie R, Sirhan D, Andermann F. Chronic cluster-like headache secondary to an epidermoid clival lesion. Can J Neurol Sci. 2006;33:421-422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 11. | Gentile S, Fontanella M, Giudice RL, Rainero I, Rubino E, Pinessi L. Resolution of cluster headache after closure of an anterior communicating artery aneurysm: the role of pericarotid sympathetic fibres. Clin Neurol Neurosurg. 2006;108:195-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 12. | Seijo-Martinez M, Castro del Río M, Conde C, Brasa J, Vila O. Cluster-like headache: association with cervical syringomyelia and Arnold-Chiari malformation. Cephalalgia. 2004;24:140-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 13. | Straube A, Freilinger T, Rüther T, Padovan C. Two cases of symptomatic cluster-like headache suggest the importance of sympathetic/parasympathetic balance. Cephalalgia. 2007;27:1069-1073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 14. | Zanchin G, Rossi P, Licandro AM, Fortunato M, Maggioni F. Clusterlike headache. A case of sphenoidal aspergilloma. Headache. 1995;35:494-497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 15. | Favier I, Haan J, van Duinen SG, Ferrari MD. Typical cluster headache caused by granulomatous pituitary involvement. Cephalalgia. 2007;27:173-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 16. | Hannerz J, Arnardottir S, Bro Skejø HP, Lilja JA, Ericson K. Peripheral postganglionic sympathicoplegia mimicking cluster headache attacks. Headache. 2005;45:84-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 17. | West P, Todman D. Chronic cluster headache associated with a vertebral artery aneurysm. Headache. 1991;31:210-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 33] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 18. | Rodríguez S, Calleja S, Morís G. Cluster-like headache heralding cerebral venous thrombosis. Cephalalgia. 2008;28:906-907. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 19. | Muñoz C, Díez-Tejedor E, Frank A, Barreiro P. Cluster headache syndrome associated with middle cerebral artery arteriovenous malformation. Cephalalgia. 1996;16:202-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 20. | Ashkenazi A, Brown F. Images from headache: cluster-like headache associated with intra-cavernous carotid artery thrombosis. Headache. 2008;48:1214-1215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 21. | Hannerz J. A case of parasellar meningioma mimicking cluster headache. Cephalalgia. 1989;9:265-269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 20] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 22. | Edvardsson B, Persson S. Cluster headache and parietal glioblastoma multiforme. Neurologist. 2012;18:206-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 23. | Scorticati MC, Raina G, Federico M. Cluster-like headache associated to a foreign body in the maxillary sinus. Neurology. 2002;59:643-644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 24] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 24. | Mainardi F, Trucco M, Maggioni F, Palestini C, Dainese F, Zanchin G. Cluster-like headache. A comprehensive reappraisal. Cephalalgia. 2010;30:399-412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 36] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 25. | Georgiadis G, Tsitouridis I, Paspali D, Rudolf J. Cerebral sinus thrombosis presenting with cluster-like headache. Cephalalgia. 2007;27:79-82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 26. | Park KI, Chu K, Park JM, Kim M. Cluster-like Headache Secondary to Cerebral Venous Thrombosis. J Clin Neurol. 2006;2:70-73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 27. | Negoro K, Kawai M, Tada Y, Ogasawara J, Misumi S, Morimatsu M. A case of postprandial cluster-like headache with prolactinoma: dramatic response to cabergoline. Headache. 2005;45:604-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 28. | Eimil-Ortiz M, María-Salgado F, Fontán-Tirado C, González-Santiago R, Villar-Villar ME, Martín E. Pseudo-cluster-like headache secondary to contralateral epidermoid cyst of the pontocerebellar angle. Headache. 2008;48:471-472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 29. | Noseda R, Burstein R. Migraine pathophysiology: anatomy of the trigeminovascular pathway and associated neurological symptoms, CSD, sensitization and modulation of pain. Pain. 2013;154 Suppl 1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 581] [Cited by in RCA: 590] [Article Influence: 49.2] [Reference Citation Analysis (0)] |
| 30. | Swaab DF, Hofman MA, Lucassen PJ, Purba JS, Raadsheer FC, Van de Nes JA. Functional neuroanatomy and neuropathology of the human hypothalamus. Anat Embryol (Berl). 1993;187:317-330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 91] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 31. | McBeath JG, Nanda A. Case reports: sudden worsening of cluster headache: A signal of aneurysmal thrombosis and enlargement. Headache. 2000;40:686-688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 32. | De Pue A, Lutin B, Paemeleire K. Chronic cluster headache and the pituitary gland. J Headache Pain. 2016;17:23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 33. | Rosebraugh CJ, Griebel DJ, DiPette DJ. A case report of carotid artery dissection presenting as cluster headache. Am J Med. 1997;102:418-419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 34. | Frigerio S, Bühler R, Hess CW, Sturzenegger M. Symptomatic cluster headache in internal carotid artery dissection--consider anhidrosis. Headache. 2003;43:896-900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 35. | Razvi SS, Walker L, Teasdale E, Tyagi A, Muir KW. Cluster headache due to internal carotid artery dissection. J Neurol. 2006;253:661-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 36. | Hardmeier M, Gobbi C, Buitrago C, Steck A, Lyrer P, Engelter S. Dissection of the internal carotid artery mimicking episodic cluster headache. J Neurol. 2007;254:253-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 37. | Rigamonti A, Iurlaro S, Reganati P, Zilioli A, Agostoni E. Cluster headache and internal carotid artery dissection: two cases and review of the literature. Headache. 2008;48:467-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 38. | Godeiro-Junior C, Kuster GW, Felício AC, Porto PP Jr, Pieri A, Coelho FM. Internal carotid artery dissection presenting as cluster headache. Arq Neuropsiquiatr. 2008;66:763-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 39. | Tobin J, Flitman S. Cluster-like headaches associated with internal carotid artery dissection responsive to verapamil. Headache. 2008;48:461-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 40. | Tsivgoulis G, Mantatzis M, Vadikolias K, Heliopoulos I, Charalampopoulos K, Mitsoglou A, Georgiadis GS, Giannopoulos S, Piperidou C. Internal carotid artery dissection presenting as new-onset cluster headache. Neurol Sci. 2013;34:1251-1252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 41. | Valença MM, Andrade-Valença LP, Martins C, de Fátima Vasco Aragão M, Batista LL, Peres MF, da Silva WF. Cluster headache and intracranial aneurysm. J Headache Pain. 2007;8:277-282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 42. | Sewell RA, Johnson DJ, Fellows DW. Cluster headache associated with moyamoya. J Headache Pain. 2009;10:65-67. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 43. | Mani S, Deeter J. Arteriovenous malformation of the brain presenting as a cluster headache--a case report. Headache. 1982;22:184-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 42] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 44. | Piovesan EJ, Lange MC, Werneck LC, Kowacs PA, Engelhorn AL. Cluster-like headache. A case secondary to the subclavian steal phenomenon. Cephalalgia. 2001;21:850-851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 45. | Kim JT, Lee SH, Choi SM, Park MS, Kim BC, Kim MK, Cho KH. Spontaneous vertebral artery dissection mimicking cluster headache. Cephalalgia. 2008;28:671-673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 46. | Chang YH, Luo CB, Wang SJ, Chen SP. Cluster headache and middle meningeal artery dural arteriovenous fistulas: A case report. Cephalalgia. 2018;38:1792-1796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 47. | Greve E, Mai J. Cluster headache-like headaches: a symptomatic feature? Cephalalgia. 1988;8:79-82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 39] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 48. | Porta-Etessam J, Ramos-Carrasco A, Berbel-García A, Martínez-Salio A, Benito-León J. Clusterlike headache as first manifestation of a prolactinoma. Headache. 2001;41:723-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 48] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 49. | Levy MJ, Robertson I, Howlett TA. Cluster headache secondary to macroprolactinoma with ipsilateral cavernous sinus invasion. Case Rep Neurol Med. 2012;2012:830469. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 50. | Edvardsson B. Cluster Headache and Pituitary Prolactinoma. J Med Cases. 2013;4:523-525. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 51. | Andereggen L, Mono ML, Kellner-Weldon F, Christ E. Cluster headache and macroprolactinoma: Case report of a rare, but potential important causality. J Clin Neurosci. 2017;40:62-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 52. | Pineyro MM, Sosa G, Finozzi MR, Stecker N, Pisabarro R, Belzarena MC. Chronic cluster-like headache in a patient with a macroprolactinoma presenting with falsely low prolactin levels: bromocriptine versus cabergoline? Clin Case Rep. 2017;5:1868-1873. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 53. | Levyman C, Dagua Filho Ados S, Volpato MM, Settanni FA, de Lima WC. Epidermoid tumour of the posterior fossa causing multiple facial pain--a case report. Cephalalgia. 1991;11:33-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 54. | Alty J, Kempster P, Raghav S. Cluster-like headache secondary to trigeminal meningioma. Neurology. 2008;70:1938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 55. | Robbins MS, Tarshish S, Napchan U, Grosberg BM. Images from headache: atypical cluster headache secondary to giant meningioma. Headache. 2009;49:1052-1053. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 56. | Edvardsson B, Persson S. Cluster headache and arachnoid cyst. Springerplus. 2013;2:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 57. | Tfelt-Hansen P, Paulson OB, Krabbe AA. Invasive adenoma of the pituitary gland and chronic migrainous neuralgia. A rare coincidence or a causal relationship? Cephalalgia. 1982;2:25-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 44] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 58. | Edvardsson B. Cluster headache associated with a clinically non-functioning pituitary adenoma: a case report. J Med Case Rep. 2014;8:451. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 59. | Milos P, Havelius U, Hindfelt B. Clusterlike headache in a patient with a pituitary adenoma. With a review of the literature. Headache. 1996;36:184-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 30] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 60. | Kao YH, Hsu YC. Chiari Malformation Type I Presenting as Cluster-like Headache. Acta Neurol Taiwan. 2015;24:122-124. [PubMed] |
| 61. | Malissart P, Ducros A, Labauge P, De Champfleur NM, Carra-Dalliere C. Carotid paraganglioma mimicking a cluster headache. Cephalalgia. 2014;34:1111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 62. | Takeshima T, Nishikawa S, Takahashi K. Cluster headache like symptoms due to sinusitis: evidence for neuronal pathogenesis of cluster headache syndrome. Headache. 1988;28:207-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 17] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 63. | Heidegger S, Mattfeldt T, Rieber A, Wikstroem M, Kern P, Kern W, Schreiber H. Orbito-sphenoidal Aspergillus infection mimicking cluster headache: a case report. Cephalalgia. 1997;17:676-679. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 64. | Edvardsson B. Cluster headache associated with acute maxillary sinusitis. Springerplus. 2013;2:509. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 65. | Edvardsson B, Persson S. Cluster headache and acute maxillary sinusitis. Acta Neurol Belg. 2013;113:535-536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 66. | Balgetir F, Avcı D, Gönen M, Taşcı İ. Acute Rhinosinusitis as an Infrequent Cause of Symptomatic Cluster Headache: Report of Seven Cases. J Oral Facial Pain Headache. 2019;33:408-412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 67. | Branco M, Rodrigues R, Lopes M, Ruano L. Cluster-Like Headache Secondary to Sphenoid Sinus Mucocele. Case Rep Neurol Med. 2018;2018:5850286. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 68. | Harley JS, Ahmed F. Cluster-like headache heralding inflammatory orbital pseudotumour. Cephalalgia. 2008;28:401-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 69. | Choi JY, Kim YH, Oh K, Yu SW, Jung KY, Kim BJ. Cluster-like headache caused by posterior scleritis. Cephalalgia. 2009;29:906-908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 70. | Ersoy A, Soyturk M. Idiopathic orbital myositis presenting as cluster headache: a case report. Acta Neurol Belg. 2017;117:937-939. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 71. | Douglas VP, Douglas KAA, Rizzo JF 3rd, Chwalisz BK. Case report: Orbital myositis triggering oxygen-responsive cluster headache. Cephalalgia. 2020;40:313-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 72. | van der Vlist SH, Hummelink BJ, Westerga J, Boogerd W. Cluster-like headache and a cystic hypothalamic tumour as first presentation of sarcoidosis. Cephalalgia. 2013;33:421-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 73. | Volcy M, Tepper SJ. Cluster-like headache secondary to idiopathic intracranial hypertension. Cephalalgia. 2006;26:883-886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 74. | Testa L, Mittino D, Terazzi E, Mula M, Monaco F. Cluster-like headache and idiopathic intracranial hypertension: a case report. J Headache Pain. 2008;9:181-183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |