Published online May 6, 2021. doi: 10.12998/wjcc.v9.i13.3056
Peer-review started: November 3, 2020
First decision: February 12, 2021
Revised: February 22, 2021
Accepted: March 11, 2021
Article in press: March 11, 2021
Published online: May 6, 2021
Processing time: 169 Days and 21.6 Hours
Crumbs homolog 2 (CRB2) is a recently discovered gene that is closely related to the maintenance of normal polarity in podocytes; mutations can directly lead to steroid-resistant nephrotic syndrome (SRNS). However, the characteristics of nephrotic syndrome (NS) caused by CRB2 mutations have not been described.
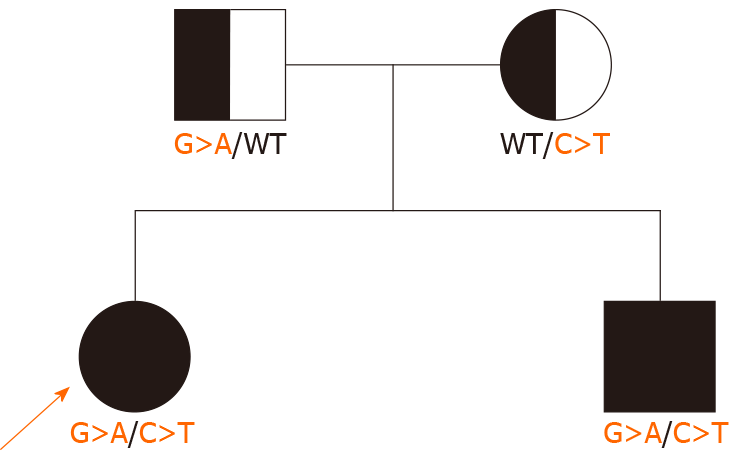
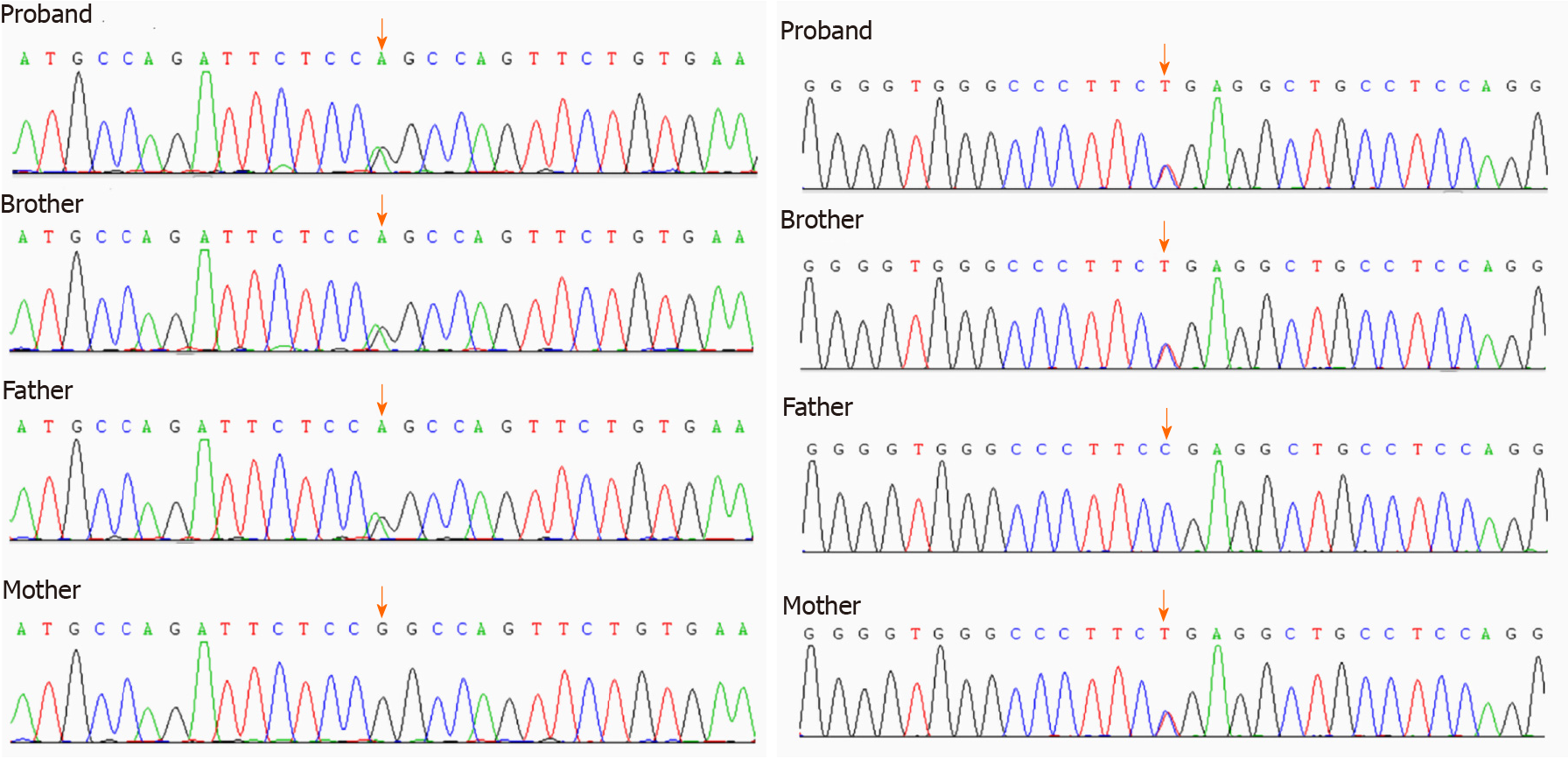
We report a novel compound heterozygous mutation of the CRB2 gene in two siblings with SRNS. The two siblings had edema, proteinuria, hypoproteinemia and hyperlipidemia. Both their father and mother had normal phenotypes (no history of NS). Whole exon sequencing (WES) of the family showed a novel compound heterozygous mutation, c.2290 (exon 8) C > T and c.3613 (exon 12) G > A. Glucocorticoid therapy (methylprednisolone pulse therapy or oral prednisone) and immunosuppressive agents (tacrolimus) had no effect. During a 3-year follow-up after genetic diagnosis by WES, proteinuria persisted, but the patient was healthy.
CRB2 mutations related to SRNS often occur in exons 7, 10, and 12. Clinical manifestations of SRNS caused by CRB2 mutations are often less severe than in other forms of SRNS.
Core Tip: Crumbs homolog 2 (CRB2) mutation is related to early-onset steroid-resistant nephrotic syndrome (SRNS). We here report two siblings with a novel compound heterozygous CRB2 mutation. We also reviewed the literature and found that CRB2 mutations related to SRNS often occur in exons 7, 10 and 12. Clinical manifestations of SRNS caused by CRB2 mutations, such as edema, are often less severe than in other forms of SRNS.
- Citation: Lu J, Guo YN, Dong LQ. Crumbs homolog 2 mutation in two siblings with steroid-resistant nephrotic syndrome: Two case reports. World J Clin Cases 2021; 9(13): 3056-3062
- URL: https://www.wjgnet.com/2307-8960/full/v9/i13/3056.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i13.3056
Primary nephrotic syndrome (NS) is a common urinary system disease in children. According to its sensitivity to corticosteroids, it can be divided into steroid-sensitive and steroid-resistant NS (SRNS). The majority of children with NS are sensitive to steroids and have a good prognosis, but 10%–20% of them have SRNS, and 30%-40% of children with SRNS progress to end-stage renal disease (ESRD) within 10 years[1]. Studies of primary NS confirm the importance of genetic testing to find mutations. A number of studies have shown that some cases of SRNS are caused by mutations of genes encoding glomerular podocyte cytoskeletal proteins. NS with gene mutations is now called hereditary NS. So far, more than 20 different single-gene mutations such as WT1, NPHS1, NPHS2 and PLCE1[2-5] have been reported to cause SRNS, and those mutations are the cause of most hereditary NS.
Podocytes are highly polarized and specialized cells that play an important role in renal glomerular filtration. Many gene mutations affecting the kidneys are related to podocyte proteins, such as mutation of the Crumbs homolog 2 (CRB2) gene. CRB2 belongs to the Crumbs family, which includes CRB1, CRB2 and CRB3. The latter two are found in mammals[6]. The CRB2 gene is located on chromosome 9q33.3, consists of 13 exons, and encodes a 1285 amino acid transmembrane protein. It is a recently discovered gene that is closely associated with SRNS, prenatal onset ventriculomegaly, and retinitis pigmentosa. By characterization of a loss-of-function mutation in zebrafish crb2b, Ebarasi et al[7] demonstrated that the CRB2 gene is important for correct podocyte foot process arborization, slit diaphragm formation, and proper nephrin trafficking[7]. It may be that different manifestations are related to the location of the mutation.
This report describes two siblings diagnosed with SRNS and carrying a novel compound heterozygous mutation of CRB2 gene that was inherited from their parents. We reviewed the literature on CRB2 gene mutations reported in recent years, summarize their clinical manifestations, pathology and management; and suggest corresponding exon mutations related to NS.
Case 1: An 11-year-old girl presented with repeated edema of the eyelids and lower extremities for > 9 years. She had been taking oral prednisone, tripterygium, and dipyridamole without regular follow-up.
Case 2: A 9-year-old boy, who is the younger brother of case 1, was admitted to hospital with the chief complaint of repeated proteinuria for > 5 years. Eyelid edema appeared at the onset of the disease. He was diagnosed with NS 1 year before and was started on long-term oral steroid therapy, but there was no significant improvement in his proteinuria.
SRNS.
Both cases had no history of significant past illnesses.
Their parents had normal phenotypes and had no history of NS. Their father and grandfather had histories of renal calculi.
Case 1: The patient had a Cushing-like appearance, edema of both eyelids, and mild edema of both lower extremities.
Case 2: The patient presented with slight swelling of the eyelids.
Case 1: Routine blood tests showed a white blood cell (WBC) count of 9.68 × 109/L, neutrophils 69.7%, hemoglobulin (Hb) 145 g/L, a platelet (PLT) count of 286 × 109/L, and C-reactive protein 6.45 mg/L. Routine urinalysis revealed protein 3+, 24 h urinary protein 1.46 g, serum albumin 19.8 g/L, and cholesterol 9.78 mmol/L. Serum creatinine, urea nitrogen, and complement C3 and C4 were all normal. Doppler ultrasound showed slightly enhanced echogenicity of the bilateral renal parenchyma. Renal biopsy revealed mild mesangial proliferative glomerulonephritis.
Case 2: Routine blood tests showed a WBC count of 6.16 × 109/L, neutrophils 36.7%, Hb 130 g/L, PLT count of 362 × 109/L, 24 h urinary protein 1.73 g, albumin 17.3 g/L, triglyceride 2.76 mmol/L, and cholesterol 10.07 mmol/L. Serum creatinine, urea nitrogen, complement C3 and C4 and chest radiography were all normal.
The two siblings and their parents all underwent whole exon sequencing, which showed that both siblings had a novel compound heterozygous mutation on exon 8, c.2290C>T and exon 12, c.3613G>A of CRB2. The mother had a heterozygous mutation on exon 8, c.2290C>T, and the father had a heterozygous mutation on exon 12, c.3613G>A (Figures 1 and 2).
SRNS with CRB2 mutation
The two siblings were treated with methylprednisolone pulse therapy (400 mg × 3 d and 300 mg × 3 d, respectively) followed by oral prednisone (25 mg bid), and tacrolimus. Other therapies included low molecular-weight heparin calcium, captopril, multivitamins, and dipyridamole.
During a 3-year follow-up, their proteinuria and mild edema persisted; the girl underwent surgery for hormone-related glaucoma.
NS is a common kidney disease in childhood. Although most cases are sensitive to steroids, 10%-20% of them are resistant, and 30%-40% of children with SRNS will develop ESRD within the next 10 years. Genetic diagnosis is important in SRNS and can avoid excessive use of steroids and immunosuppressants, thereby preventing adverse effects such as infection and cytotoxicity and improving quality of life. SRNS with a monogenic cause is known as hereditary NS[8], which can result in an isolated glomerular disease or syndrome with renal and extrarenal manifestations. Genes encoding podocyte proteins play an important role, such as WT1, NPHS1, PLCE1, CRB2, LAMB2, EXT1 andCOL4A5[9].
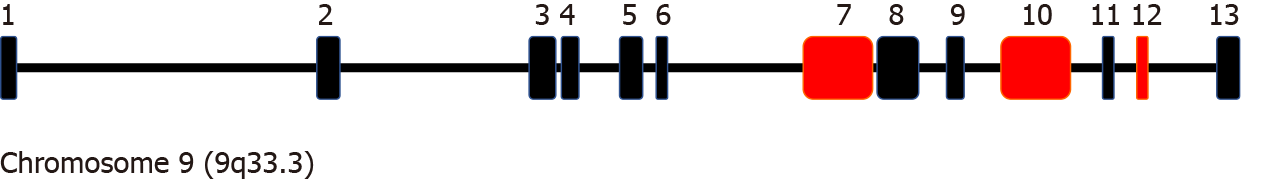
There are a few clinical reports of NS caused by CRB2 gene mutation. CRB2 is a recently discovered gene that is closely related to the maintenance of normal podocyte polarity, and its mutation can directly lead to SRNS[10]. The family of Crumbs proteins including CRB1, CRB2 and CRB3, includes human homologs of drosophila Crb and plays a key role in the establishment and maintenance of epithelial polarity[11]. Katoh and Kotah[13] first identified CRB2, localized on 9q33.3, consisting of 13 exons and encoding 1285 amino acid transmembrane protein, containing large extracellular domains with epidermal growth factor[13,14]. Ebarasi et al[7] suggested that in zebrafish, crb2b has important effects on the correct podocyte foot process arborization, slit diaphragm formation, and proper renin trafficking[7]. The CRB2-null zebrafish presents with pronephric cysts, pericardial edema and smaller eyes, which demonstrates that the CRB2 gene mutation can cause the corresponding loss-of-function of podocytes, and that CRB2 mutations in humans can lead to NS. Jaron et al[15] presented two cases with ventriculomegaly and hydronephrosis and concluded that CRB2-associated disease is a new ciliopathy[15]. CRB2 is a member of the Crumbs family of proteins implicated in planar cell polarity and is highly expressed in brain, retina and kidney[14]. CRB2 missense mutation of different domains can lead to different clinical manifestations, and exons related to SRNS include 7, 10 and 13[7,16]. The CRB2 gene has 13 exons in total, and mutation of this gene can cause NS, hydrocephalus, and retinitis pigmentosa. Whether a patient with CRB2 gene mutation presents with NS, hydrocephalus or retinitis pigmentosa depends on the mutation sites. In the literature review, we found that CRB2 mutations related to SRNS often occur in exons 7, 10 and 12 (Figure 3).
We found that the two siblings had a novel compound heterozygous mutation that led to SRNS. Their parents were carriers of CRB2 gene mutation, but the mutation sites were different. The mother’s mutation site was chromosome 9, CRB2 gene exon 8 c.2290C>T, while the father’s mutation site was chromosome 9, CRB2 gene exon 12 c.3613G>A. The siblings presented clinical phenotypes as SRNS. The two mutation sites are reported here for the first time.
As CRB2 is mainly expressed in the brain, retina and kidney, the main manifestations include ventriculomegaly, hydronephrosis, and SRNS (Table 1). Jaron et al[15] reported two Ashkenazi Jewish siblings clinically diagnosed with severe congenital hydrocephalus and mild urinary tract anomalies[15]. Lamont et al[16] reported five individuals with biallelic variants in CRB2 presenting with lateral ventriculomegaly and bilateral echogenic kidneys, loss of corticomedullary differentiation, and multiple small cysts in both kidneys[16]. Ebarasi et al[7] identified recessive mutations of CRB2 in four different families affected by SRNS[7]. Watanabe et al[10] reported the long-term clinicopathological observation of a Japanese female patient with SRNS caused by a compound heterozygous mutation of CRB2[10]. Udagawa et al[17] analyzed the expression of CRB2 and slit diaphragm molecules in a 3-year-old girl with SRNS and concluded that CRB2 mutations were the mechanism of SRNS[17]. Fan et al[18] reported a 7-year-old boy with SRNS who had heterozygous mutations of CRB2[18]. Chen et al[19] reported the identification of CRB2 as a novel retinitis pigmentosa causative gene in a Chinese consanguineous family[19]. Slavotinek et al[20] reported five fetuses and a child from three families who shared a phenotype comprising cerebral ventriculomegaly and echogenic kidneys with histopathological findings of congenital nephrosis[20].
| Reference | Age at diagnosis | Gender | Main clinical manifestations | Nucleotide change | Exon |
| Jaron et al[15] | 7 yr | Male | Ventriculomegaly and hydronephrosis | c.2400C>G; c.3089_3104dup | 8; 10 |
| 5.5 yr | Male | Ventriculomegaly and hydronephrosis | c.2400C>G; c.3089_3104dup | 8; 10 | |
| Lamont et al[16] | 8 mo | Female | Ventriculomegaly and nephromegaly with multiple macrocysts | c.(3291_3292delCT); c.(3343C>T) | 10; 10 |
| 2 mo | Male | Ventriculomegaly and increased echogenicity of kidney | c.(3385T>C); c.3108_3109insCCGGCGCGGCCCCGGC | 10; 10 | |
| 8 d | Female | persistent echogenicity of the kidneys and ventriculomegaly | c.(1494G>T); c.(3227A>C) | 7; 10 | |
| 8 d | Female | persistent echogenicity of the kidneys and ventriculomegaly | c.(1494G>T); c.(3227A>C) | 7; 10 | |
| Ebarasi et al[7] | 6 yr | ND | SRNS | c.1859G>C | 7 |
| 4 yr | ND | SRNS | c.1859G>C | 7 | |
| 9 mo | ND | SRNS | c.1882C>T; (c.3089_3104dup) | 7; 10 | |
| 3 yr | ND | SRNS | c.1886G>C | 7 | |
| NR | ND | SRNS | c.3746G>A | 13 | |
| Watanabe et al[10] | 3.5 yr | Female | SRNS | c.1882C>T; c.2515G>T | 7; 9 |
| Udagawa et al[17] | 3 yr | Female | SRNS | c.3256_3273del18insCGGCCCGGGGTGG; c.3551A>C | 10; 12 |
| Fan et al[18] | 7 yr | Male | SRNS | c.3190C>T; c.2705C>T | 10; 10 |
| Slavotinek et al[20] | 1 mo | Male | Cerebral ventriculomegaly and congenital nephrosis | c.1897C>T | 7 |
| Chen et al[19] | 51 yr | Female | Retinitis pigmentosa | c.3745C>G | 13 |
| 46 yr | Female | Retinitis pigmentosa | c.3745C>G | 13 |
Renal biopsy findings from SRNS patients with CRB2 mutations range from minor glomerular abnormalities to advanced focal segmental glomerulosclerosis (FSGS). Ebarasi et al[7] reported four individuals with renal histology of FSGS. In a 3-year-old girl, renal pathology showed FSGS with effaced podocyte foot processes in a small area. Fan reported renal histology of FSGS in a 7-year-old boy[7]. In a case report by Watanabe, six sequential renal biopsy specimens revealed histological alteration ranging from minor glomerular abnormalities to FSGS[10]. In our case, renal biopsy showed mild mesangial proliferative glomerulonephritis. It seems that renal biopsy in SRNS patients caused by CRB2 mutation is related to the stage of the disease course.
For children with CRB2 mutations, treatment is difficult. A Japanese girl reported by Udagawa et al[17] was unresponsive to corticosteroids, immunosuppressants, and rituximab[17]. Fan et al[18] suggested that high-dose methylprednisolone pulses and cyclosporine were effective[18]. Watanabe suggested presumptive podocyte-protective therapy including low-dose tacrolimus and angiotensin receptor blocker[10]. The two siblings in our report presented with SRNS, and immunosuppressive agents had no effect. In a 2-year follow-up, proteinuria remained, along with slight edema. Judging from the SRNS cases reported above as well as the two siblings reported here, although proteinuria is difficult to control in NS caused by CRB2 mutation, the patients looked healthy and their renal function remained normal during 2–16.5 years of follow-up. Thus, it may be that in SRNS patients with CRB2 mutations, regular follow-up is more important than treatment with corticosteroids and immunosuppressant agents, which may induce adverse effects such as infection, short stature, and glaucoma.
CRB2 gene is related to early-onset SRNS. We report two siblings with a novel compound heterozygous CRB2 mutation. In the literature review, we found that CRB2 mutations related to SRNS often occur in exons 7, 10 and 12. Unlike other forms of SRNS caused by genetic mutations, clinical manifestations of SRNS caused by CRB2 mutations, such as edema, are often less severe, but proteinuria is difficult to control. Regular follow-up is more important than drug therapy (e.g., corticosteroids and immunosuppressant agents). As more cases are reported, more information about the disease will be gathered and more appropriate treatments will be found.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, research and experimental
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Ali FEM, Wagner KD S-Editor: Fan JR L-Editor: Filipodia P-Editor: Ma YJ
| 1. | Mekahli D, Liutkus A, Ranchin B, Yu A, Bessenay L, Girardin E, Van Damme-Lombaerts R, Palcoux JB, Cachat F, Lavocat MP, Bourdat-Michel G, Nobili F, Cochat P. Long-term outcome of idiopathic steroid-resistant nephrotic syndrome: a multicenter study. Pediatr Nephrol. 2009;24:1525-1532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 132] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 2. | Wagner N, Wagner KD, Afanetti M, Nevo F, Antignac C, Michiels JF, Schedl A, Berard E. A novel Wilms' tumor 1 gene mutation in a child with severe renal dysfunction and persistent renal blastema. Pediatr Nephrol. 2008;23:1445-1453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 3. | Aya K, Shimizu J, Ohtomo Y, Satomura K, Suzuki H, Yan K, Sado Y, Morishima T, Tanaka H. NPHS1 gene mutation in Japanese patients with congenital nephrotic syndrome. Nephrol Dial Transplant. 2009;24:2411-2414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 4. | Sun H, Zhou W, Wang J, Yin L, Lu Y, Fu Q. A novel mutation in NPHS2 gene identified in a Chinese pedigree with autosomal recessive steroid-resistant nephrotic syndrome. Pathology. 2009;41:661-665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 5. | Shimizu M, Irabu H, Kaneda H, Ohta K, Nozu K. Familial focal segmental glomerulosclerosis with PLCE1 mutation in siblings. Pediatr Int. 2019;61:726-727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 6. | Alves CH, Boon N, Mulder AA, Koster AJ, Jost CR, Wijnholds J. CRB2 Loss in Rod Photoreceptors Is Associated with Progressive Loss of Retinal Contrast Sensitivity. Int J Mol Sci. 2019;20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 7. | Ebarasi L, Ashraf S, Bierzynska A, Gee HY, McCarthy HJ, Lovric S, Sadowski CE, Pabst W, Vega-Warner V, Fang H, Koziell A, Simpson MA, Dursun I, Serdaroglu E, Levy S, Saleem MA, Hildebrandt F, Majumdar A. Defects of CRB2 cause steroid-resistant nephrotic syndrome. Am J Hum Genet. 2015;96:153-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 84] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 8. | Ha TS. Genetics of hereditary nephrotic syndrome: a clinical review. Korean J Pediatr. 2017;60:55-63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 9. | Saleem MA. New developments in steroid-resistant nephrotic syndrome. Pediatr Nephrol. 2013;28:699-709. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 10. | Watanabe S, Aizawa T, Tsukaguchi H, Tsugawa K, Tsuruga K, Shono A, Nozu K, Iijima K, Joh K, Tanaka H. Long-term clinicopathologic observation in a case of steroid-resistant nephrotic syndrome caused by a novel Crumbs homolog 2 mutation. Nephrology (Carlton). 2018;23:697-702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 11. | Slavotinek AM. The Family of Crumbs Genes and Human Disease. Mol Syndromol. 2016;7:274-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 12. | Tepass U, Knust E. Crumbs and stardust act in a genetic pathway that controls the organization of epithelia in Drosophila melanogaster. Dev Biol. 1993;159:311-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 170] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 13. | Katoh M, Katoh M. Identification and characterization of Crumbs homolog 2 gene at human chromosome 9q33.3. Int J Oncol. 2004;24:743-749. [PubMed] |
| 14. | van den Hurk JA, Rashbass P, Roepman R, Davis J, Voesenek KE, Arends ML, Zonneveld MN, van Roekel MH, Cameron K, Rohrschneider K, Heckenlively JR, Koenekoop RK, Hoyng CB, Cremers FP, den Hollander AI. Characterization of the Crumbs homolog 2 (CRB2) gene and analysis of its role in retinitis pigmentosa and Leber congenital amaurosis. Mol Vis. 2005;11:263-273. [PubMed] |
| 15. | Jaron R, Rosenfeld N, Zahdeh F, Carmi S, Beni-Adani L, Doviner V, Picard E, Segel R, Zeligson S, Carmel L, Renbaum P, Levy-Lahad E. Expanding the phenotype of CRB2 mutations - A new ciliopathy syndrome? Clin Genet. 2016;90:540-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 16. | Lamont RE, Tan WH, Innes AM, Parboosingh JS, Schneidman-Duhovny D, Rajkovic A, Pappas J, Altschwager P, DeWard S, Fulton A, Gray KJ, Krall M, Mehta L, Rodan LH, Saller DN Jr, Steele D, Stein D, Yatsenko SA, Bernier FP, Slavotinek AM. Expansion of phenotype and genotypic data in CRB2-related syndrome. Eur J Hum Genet. 2016;24:1436-1444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 38] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 17. | Udagawa T, Jo T, Yanagihara T, Shimizu A, Mitsui J, Tsuji S, Morishita S, Onai R, Miura K, Kanda S, Kajiho Y, Tsurumi H, Oka A, Hattori M, Harita Y. Altered expression of Crb2 in podocytes expands a variation of CRB2 mutations in steroid-resistant nephrotic syndrome. Pediatr Nephrol. 2017;32:801-809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 18. | Fan J, Fu R, Ren F, He J, Wang S, Gou M. A case report of CRB2 mutation identified in a Chinese boy with focal segmental glomerulosclerosis. Medicine (Baltimore). 2018;97:e12362. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 19. | Chen X, Jiang C, Yang D, Sun R, Wang M, Sun H, Xu M, Zhou L, Chen M, Xie P, Yan B, Liu Q, Zhao C. CRB2 mutation causes autosomal recessive retinitis pigmentosa. Exp Eye Res. 2019;180:164-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 20. | Slavotinek A, Kaylor J, Pierce H, Cahr M, DeWard SJ, Schneidman-Duhovny D, Alsadah A, Salem F, Schmajuk G, Mehta L. CRB2 mutations produce a phenotype resembling congenital nephrosis, Finnish type, with cerebral ventriculomegaly and raised alpha-fetoprotein. Am J Hum Genet. 2015;96:162-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 69] [Article Influence: 6.9] [Reference Citation Analysis (0)] |