Published online Jan 26, 2020. doi: 10.12998/wjcc.v8.i2.415
Peer-review started: September 19, 2019
First decision: December 4, 2019
Revised: December 17, 2019
Accepted: December 22, 2019
Article in press: December 22, 2019
Published online: January 26, 2020
Processing time: 119 Days and 15.9 Hours
Myocarditis refers to a variety of myocardial inflammatory lesions. A variety of factors such as infection and physical and chemical factors can cause myocarditis. Depending on the severity of myocardial damage, myocarditis patients can manifest heart failure, cardiogenic shock, and even sudden death. Here we present a case of viral myocarditis that mimicked acute coronary syndrome.
A middle-aged male patient presented with chest pain and elevated troponin I after a flu-like infection. This patient had a history of hypertension and a habit of alcohol and tobacco use. Electrocardiography showed typical changes in acute myocardial infarction, with the T-wave increasing. Coronary angiogram revealed no stenosis. Cardiac magnetic resonance imaging revealed edema of the middle and apical septal and apical anterior walls on T2-weighted images and the T1 mapping. Late gadolinium enhancement of the middle and apical septal and apical anterior walls could be found. Rubella virus immunoglobulin G and immunoglobulin M antibodies were abnormally elevated. The patient was given antiviral and antibiotic treatments, and serum biomarkers and electrocardiograph returned to normal after 5 d of treatment. After one-year follow-up, the patient showed no symptoms, and cardiac magnetic resonance showed that myocardial thickness was significantly thinner than before, and fibrosis was less than before.
This case illustrates the utility of cardiac magnetic resonance for diagnosis of infarction-like myocarditis when the angiogram is normal.
Core tip: We present a case of myocarditis with typical acute myocardial infarction symptoms. The diagnosis was confirmed by cardiac magnetic resonance. We followed the patients up to 13 mo, and performed three times of cardiac magnetic resonance. We observed the changes of the myocardial edema and the area of enhancement. We also discuss myocarditis that mimics acute myocardial infraction in order to help clinicians improve their diagnosis and treatment skills.
- Citation: Hou YM, Han PX, Wu X, Lin JR, Zheng F, Lin L, Xu R. Myocarditis presenting as typical acute myocardial infarction: A case report and review of the literature. World J Clin Cases 2020; 8(2): 415-424
- URL: https://www.wjgnet.com/2307-8960/full/v8/i2/415.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v8.i2.415
Myocarditis has heterogeneous manifestations with respect to pathological immune processes in the heart. A variety of factors, such as infection and immune diseases, can cause myocarditis, which may manifest as acute heart failure, ventricular arrhythmias, myocardial infarction, or even cardiogenic shock. Serum biomarkers, especially troponin I (TnI) and troponin T (TnT), can be elevated. Abnormalities in electrocardiogram (ECG) can include nonspecific ST-segment changes, T-wave inversions, and ST-segment elevations[1]. The cause of myocarditis is usually a viral infection; the most cited viruses are enteroviruses, adenoviruses, parvovirus B19, human herpesvirus 6, human cytomegalovirus, Epstein-Barr virus, and hepatitis virus[2-4]. Cardiovascular magnetic resonance (CMR) has become the primary tool for non-invasive assessment of myocardial inflammation in patients with suspected myocarditis[5]. Here, we present a case of viral myocarditis that mimicked acute coronary syndrome and performed a literature. Compared to the cases reported in the literature, our patient’s ECG showed typical dynamic changes of acute myocardial infarction. We also used CMR to closely record the long-term dynamic progression of the patient’s prognosis.
A 42-year-old man presented with cough that had started 1 wk earlier and chest pain that had begun 2 d earlier.
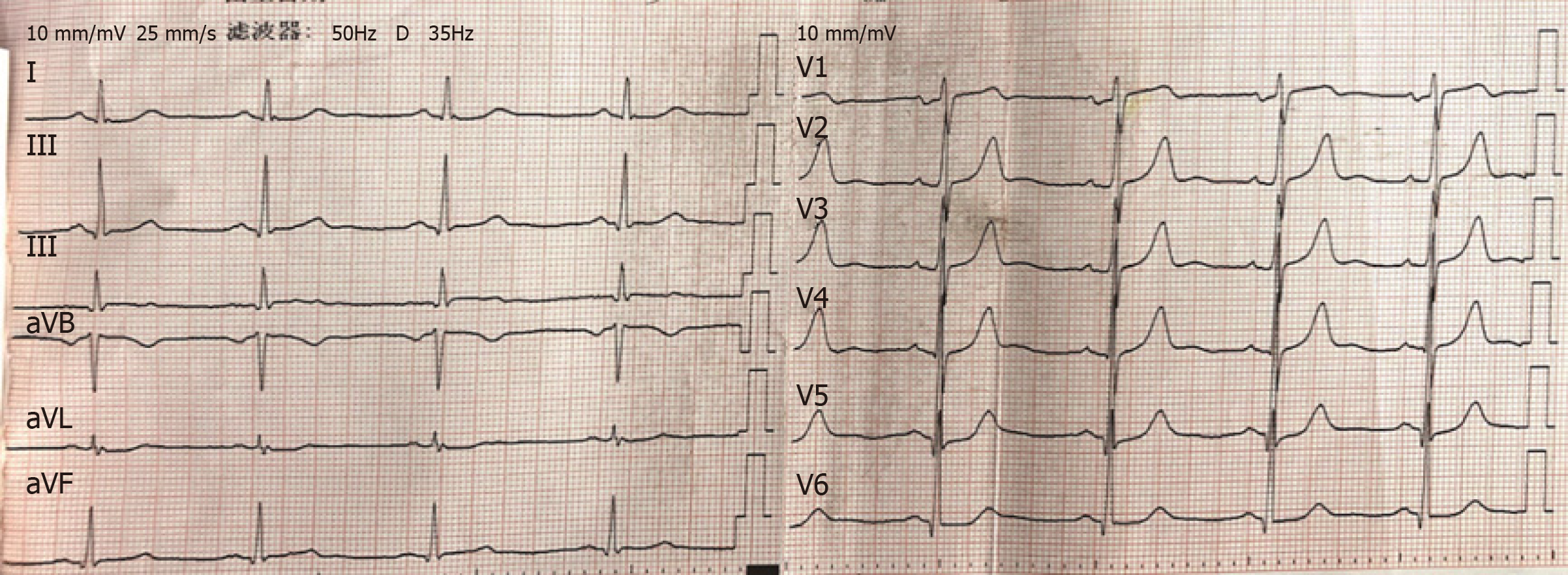
The patient presented with cough that had started 1 wk earlier; he was treated with azithromycin (500 mg, qd, i.v.) for a common cold at a private clinic. His symptoms were alleviated after treatment. However, 2 d later, the patient experienced chest pain radiating to the right arm while walking in a cold wind. He was evaluated at another hospital, where his hypersensitive troponin T level was found to be 34.34 pg/mL (normal range: 3-14 pg/mL). He also exhibited increased white blood cell and platelet counts and an increased neutrophil percentage. His ECG (Figure 1) was normal. Echocardiography showed that the left ventricular ejection fraction (LVEF) was 59%, with mild regional abnormalities in the ventricular and apical septa and a dilated left atrium. The patient’s hypersensitive troponin T was 685.30 pg/mL and the creatine kinase isozyme (CK-MB) level was 11.85 ng/mL (normal range: 0.1-4.94 ng/mL) after 8 h. Based on these findings, the patient was admitted to our hospital.
The patient had a 2-year history of hypertension and a smoking history for 20 years.
No specific personal or family history of disease was recorded.
The patient's temperature was 37.8 °C, his body mass was 78 kg, his blood pressure was 164/114 mmHg, and there were no positive signs on a physical examination.
Laboratory testing showed the following on admission: serum TnI, 2.57 ng/mL (normal range: 0-0.034 ng/mL); hypersensitive C-reactive protein, 4.95 mg/L (normal range: 0-2.87 mg/L); erythrocyte sedimentation rate, 45 mm/h; brain natriuretic peptide, 168 pg/mL; white blood cell count, 13.30 × 109/L; neutrophil percentage, 0.735; and platelet count, 312 × 109/L. His liver function, kidney function, thyroid function, D-dimer, electrolytes, blood lipids, and blood glucose levels showed no significant abnormalities; however, his rubella virus immunoglobulin G (IgG) and immunoglobulin M (IgM) antibody levels were elevated on admission.
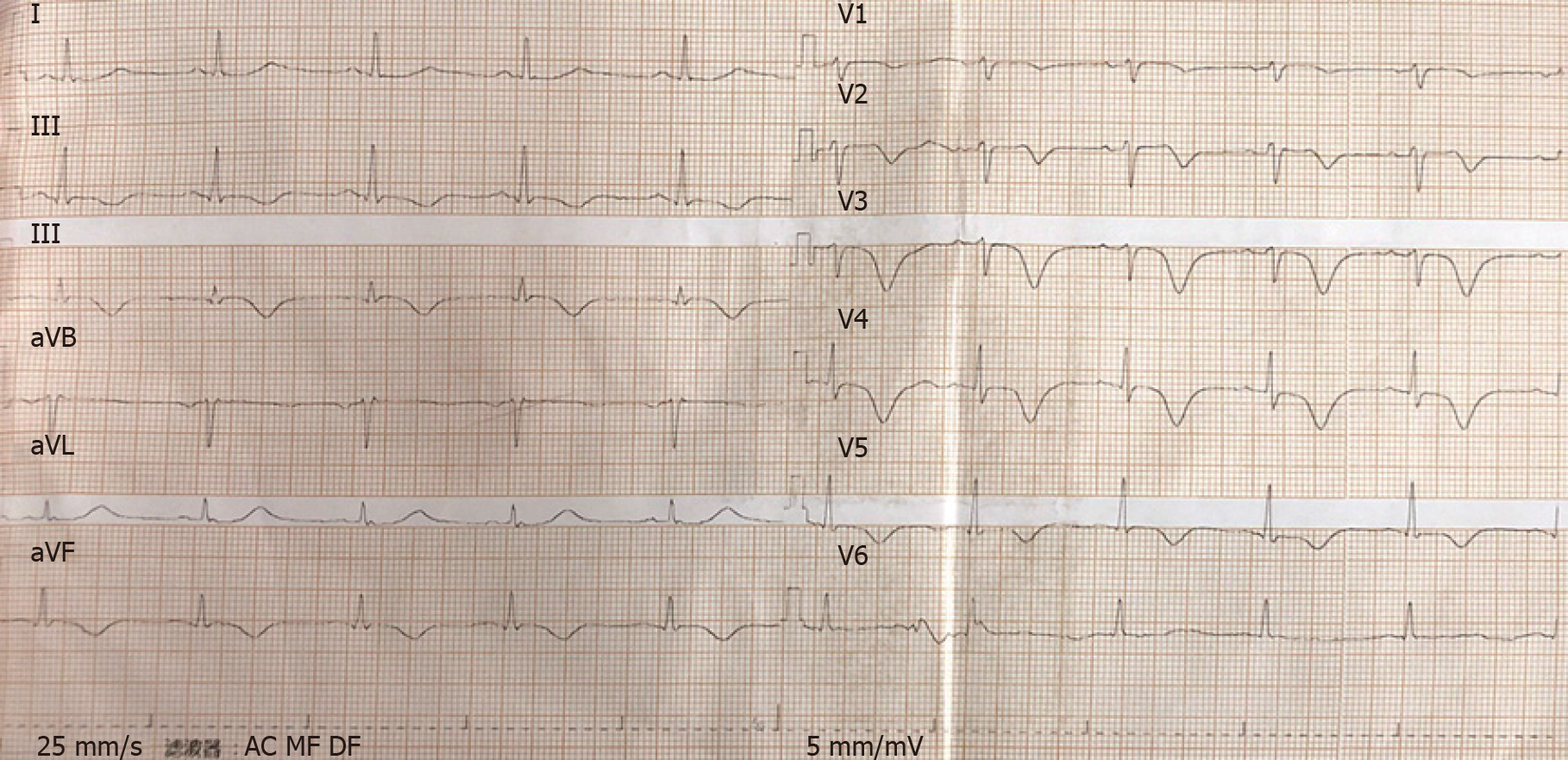
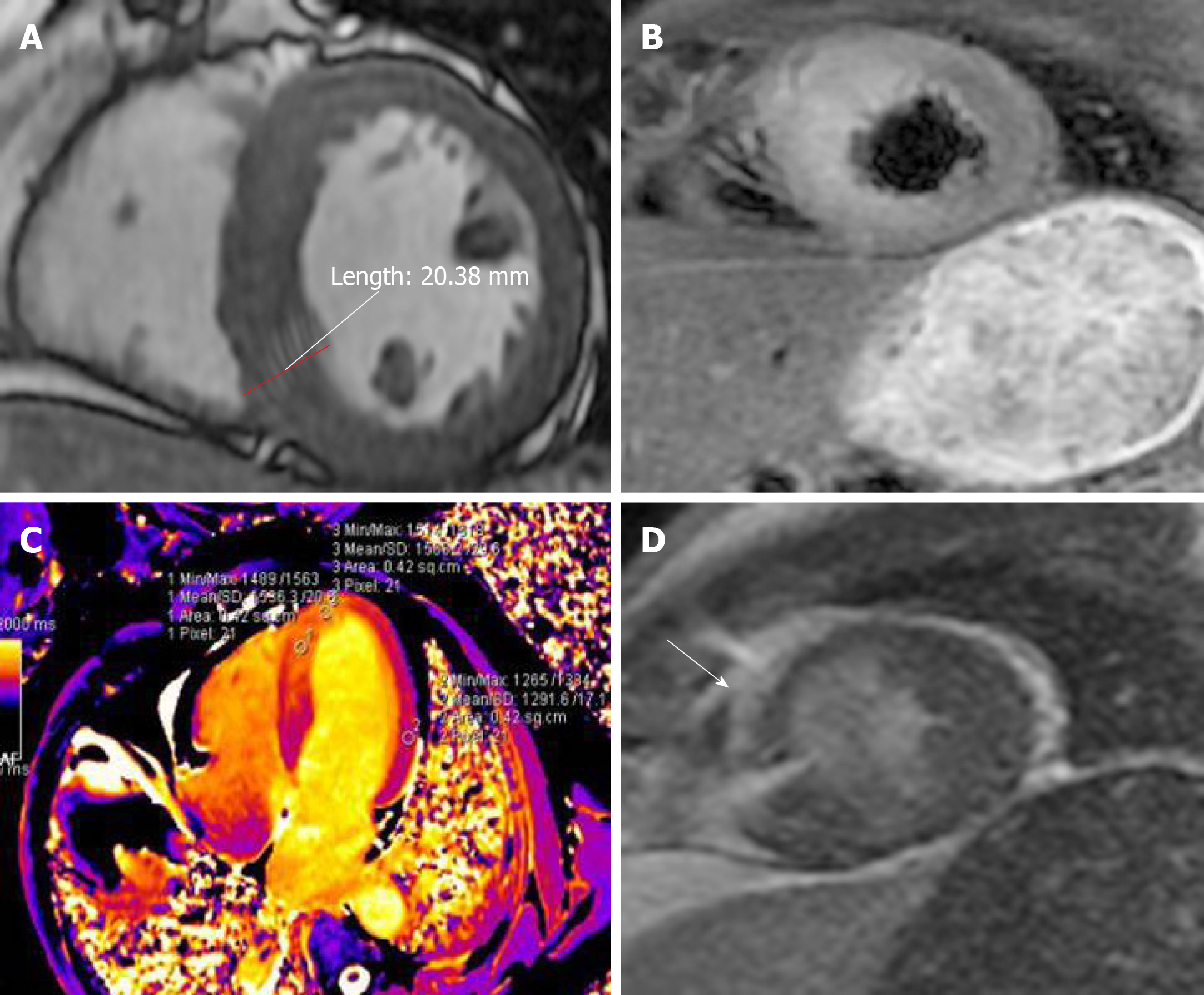
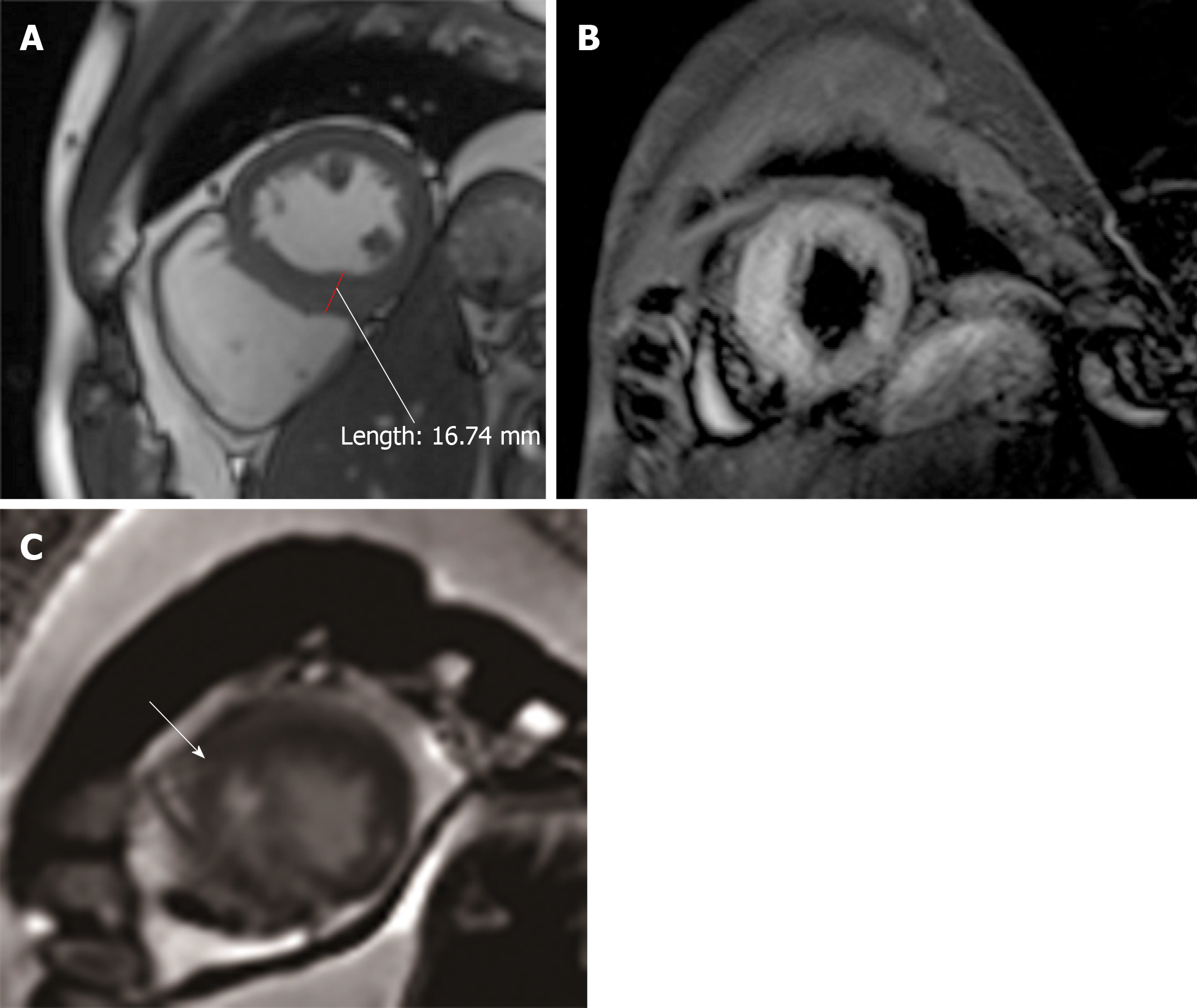
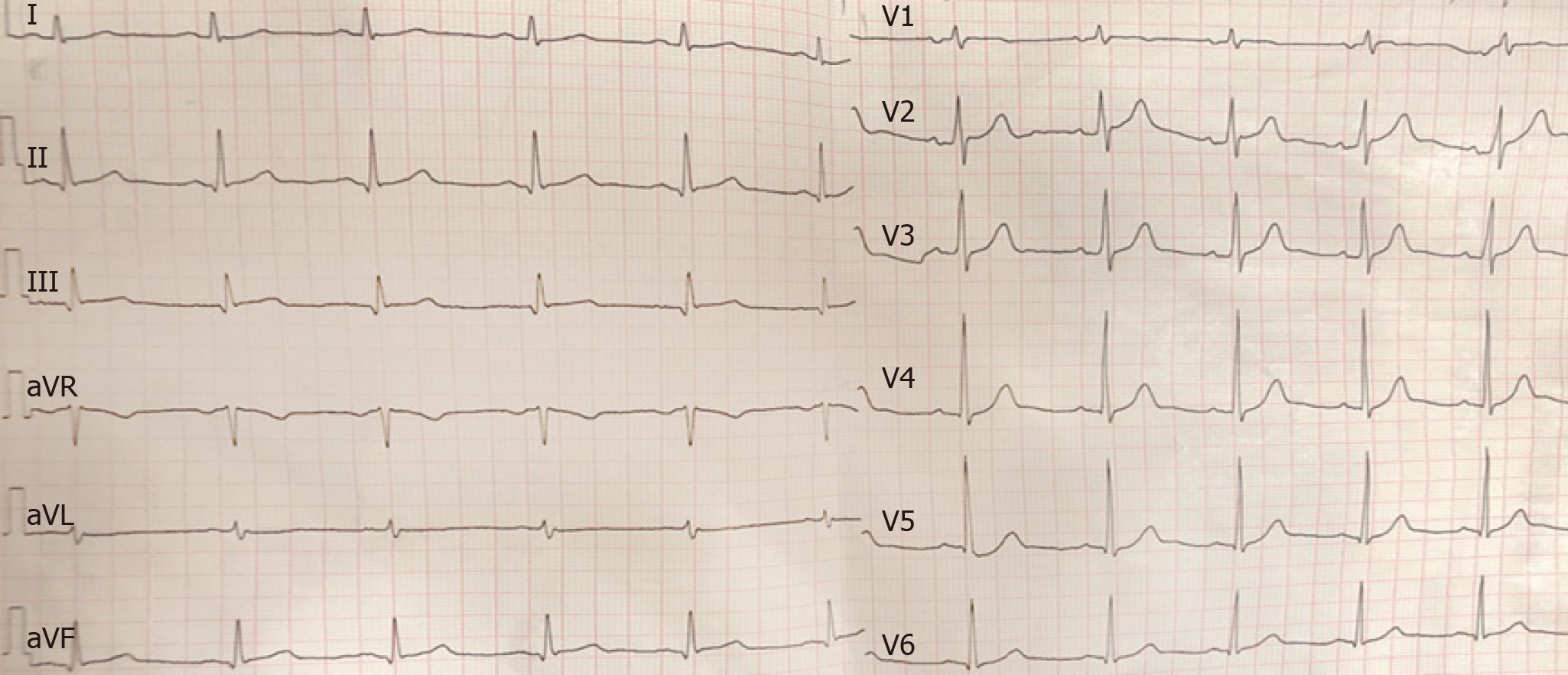
The patient’s ECG (Figure 2) showed II, III, and aVF T-wave inversions, and a wide range of precordial segment T-wave inversions that exhibited dynamic changes compared with the previous ECG. Electrocardiography (Figure 3) revealed the abovementioned segment T-wave inversion after 20 h. Coronary angiography was performed on day 5 after admission. There was no obvious coronary stenosis during the operation. Cine CMR imaging showed wall motion abnormalities (mid septal, apical septal, and apical anterior), and the anterior walls were obviously thicker than the normal walls (Figure 4A), with the thickest part of the ventricular wall being approximately 20 mm. Fat suppressed T2-weighted imaging showed the edema of the mid septal, apical septal, and apical anterior walls (Figure 4B). We observed the myocardial edema more specifically on T1 mapping (Figure 4C). The endocardium and middle myocardium of the mid septal and apical septal walls showed patchy late gadolinium enhancement (LGE) (Figure 4D). The high signal area was smaller than the area of edema.
Taking into accounts the patient’s disease history, symptoms, and examination findings, his suspected diagnosis was viral myocarditis.
He was given aspirin (100 mg, qd, p.o.) and clopidogrel (75 mg, qd, p.o.) for 6 d before the CMR examination. After the CMR examination, the patient was treated using acyclovir (250 mg, q12h, i.v.) combined with levofloxacin (500 mg, qd, i.v.) for 11 d. The patient was treated with trimetazidine and creatine phosphate for 19 d during hospitalization.
The patient had no more chest pain and his temperature returned to normal, as did his C-reactive protein and TnI (0.027) levels; his rubella virus-specific IgG and IgM antibody levels were slightly lower than before. A review of the CMR images showed that the edema observed in the mid septal, apical septal, and apical anterior walls was less extensive than before on T2WI; the thickest part of the ventricular wall was approximately 17 mm (Figure 5A and B) and the anterior and interior enhancement moved to the middle myocardium, confirming the diagnosis of myocarditis (Figure 5C).
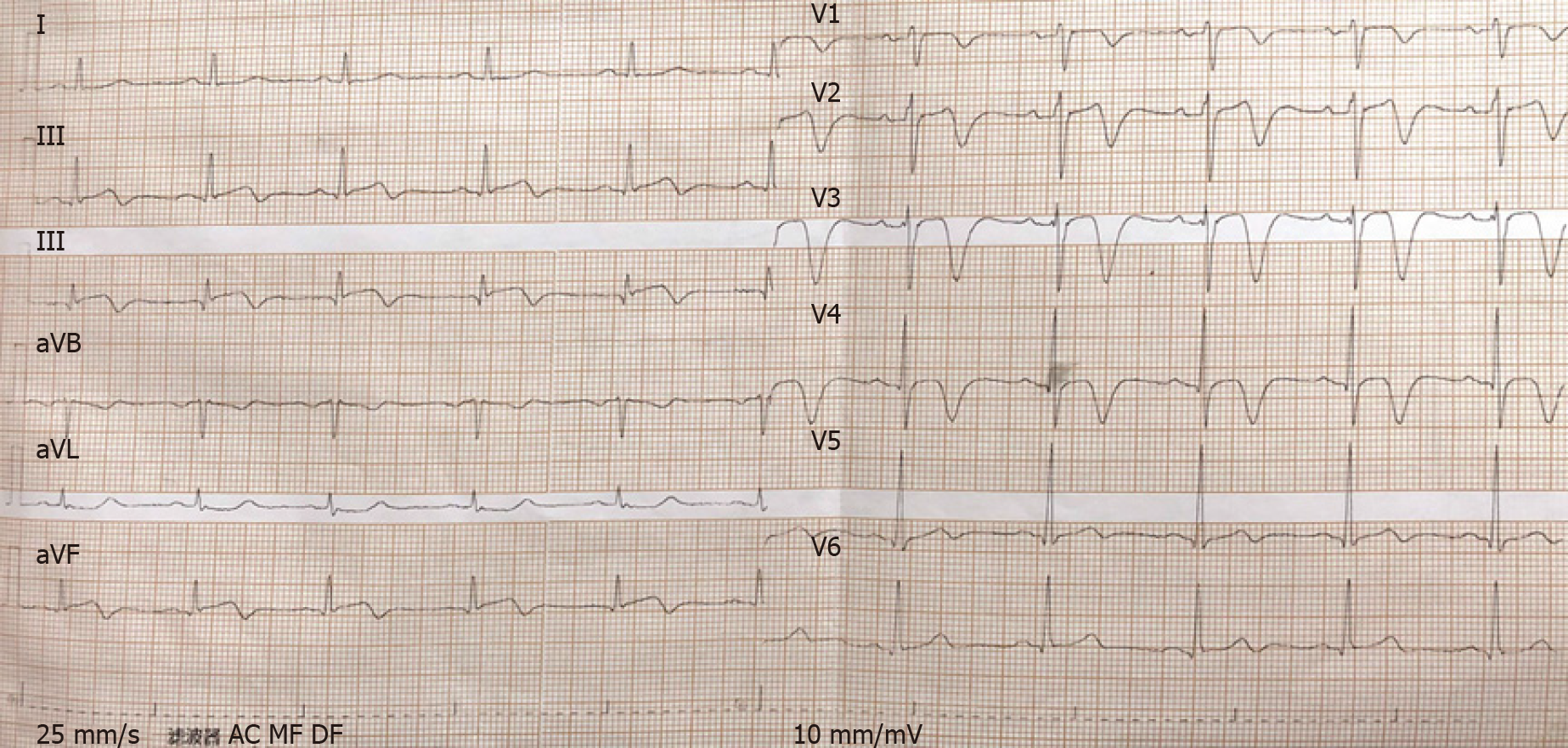
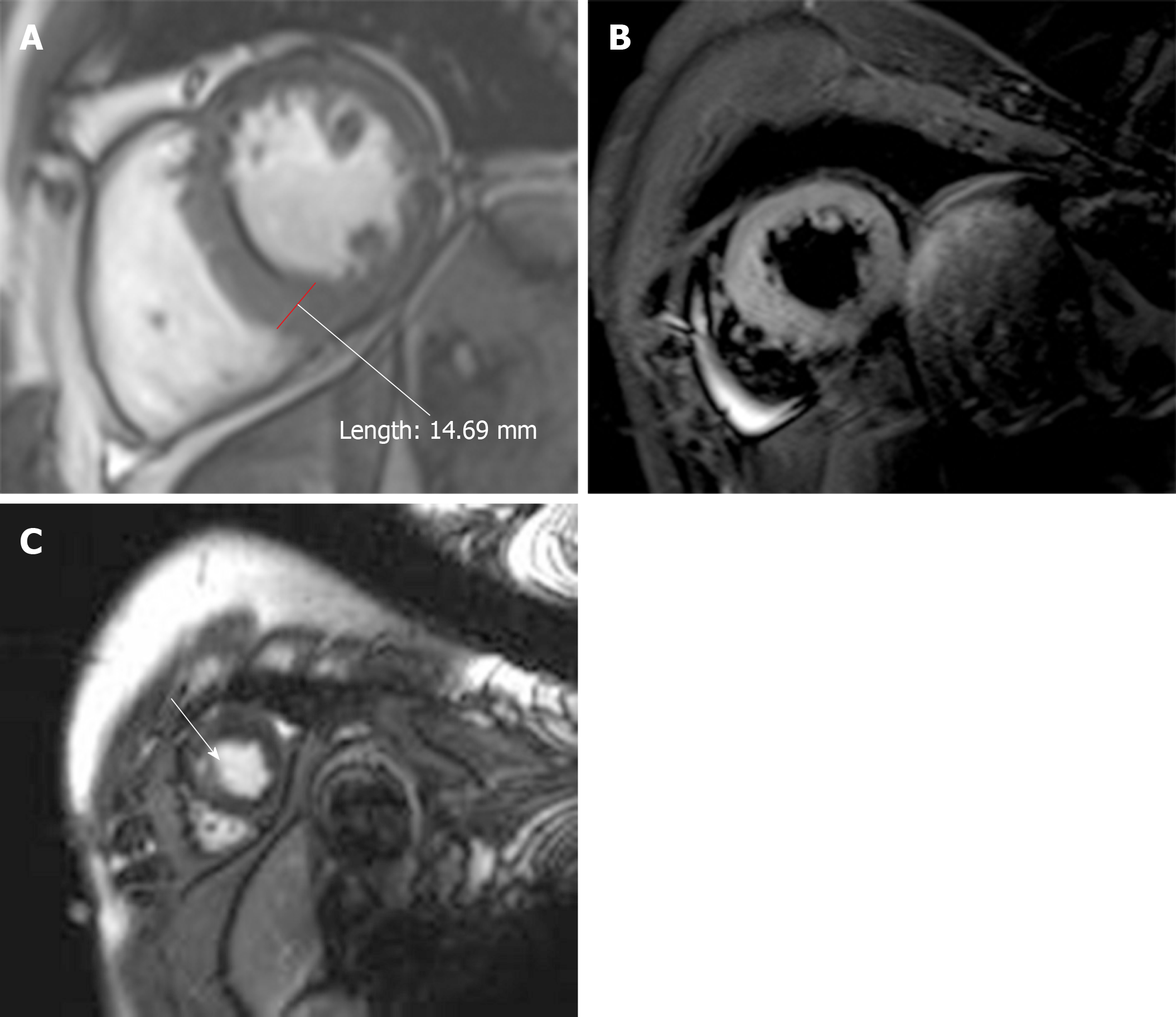
The patient’s condition improved and he was discharged. Ten months later, the patient was asymptomatic and the ECG was normal (Figure 6). Ultrasound echocardiography showed normal wall motion but patchy enhancement of the ventricular septal wall. At his 13-mo follow-up examination, the patient remained asymptomatic; CMR showed that the edema had disappeared and the thickest part of the ventricular wall was approximately 14 mm (Figure 7A and B). Given the patient’s history of hypertension, we consider that the hypertrophy was due to high blood pressure. LGE was distributed in the myocardium, indicating fibrosis (Figure 7C).
Myocarditis has numerous manifestations. This report describes a case of myocarditis that masqueraded as acute coronary syndrome. Myocarditis that mimics myocardial infarction presents with elevated serum TnI, TnT, and myocardial enzyme (e.g., CK-MB) levels, and electrocardiograph may reveal ST-segment elevation or depression, and/or Q- and T-wave inversions. The coronary angiogram is usually normal. In a study that showed the patients with an initial clinical presentation of acute myocardial infarction with normal coronary angiograms, almost 77% were suspected of having myocarditis according to the imaging data[6]. In addition to coronary angiography, myocarditis and myocardial infarction can be identified by CMR. The “Lake Louise Criteria” recommend a combination of CMR techniques, including T1-weighted early gadolinium enhancement, LGE, and T2-weighted edema imaging[5]. Gerbaud et al[7] reported that CMR is able to identify the cardiac etiology of troponin elevation in more than 75% of patients. In a large T1 and T2 mapping trial, T1 mapping provided the best diagnostic parameters in patients who had been diagnosed with acute myocarditis during 2 wk[8]. A 16-mo follow-up study showed that infarction-like myocarditis usually recovers to normal cardiac function, but still has the possibility of becoming enlarged[9]. Although endomyocardial biopsy (EMB) is the gold standard for diagnosing myocarditis, its invasiveness and low sensitivity limit its clinical application. The optimum management of myocarditis remains controversial. Currently, the management focuses largely on supportive care; patients receive standard heart failure treatment and are instructed to refrain from competitive athletics for up to 6 mo[10]. Immunosuppression should be started only after ruling out an active infection on EMB[11]. The utility of intravenous immunoglobulin in viral myocarditis remains to be investigated, but it was shown to improve the LVEF in some clinical trials of viral myocarditis[12].
In the case described herein, the patient’s initial symptom was chest pain; biomarkers such as TnI and CK-MB were elevated and the ECG showed ST-segment elevation and T-wave inversion. The patient’s chest radiograph and D-dimer level were normal, thus excluding pulmonary embolism. There was no difference in blood pressure between the two sides of the limb, thus excluding aortic dissection. Valvular disease was excluded by echocardiography. Pericarditis was also ruled out because there was no pericardial friction or effusion. The patient’s initial presentation suggested acute myocardial infarction. We recommended coronary angiography for several times within 72 h after the patient was admitted to the hospital. The patient felt that the chest pain was alleviated and there was no hemodynamic disorder, so he refused the examination. Upon our repeated recommendation, the patient agreed to undergo coronary angiography 5 d after admission. However, the normal coronary angiography contradicted this diagnosis. CMR revealed diffuse LGE in the endocardium and myocardium, but later it moved to the myocardium. A previous study demonstrated that the presence of a subendocardial or transmural pattern on LGE was strongly associated with acute myocardial infarction and that the presence of midmyocardial and epicardial or patchy LGE indicated acute myocarditis[13]; thus, we excluded the possibility of ischemic cardiomyopathy in making our diagnosis. The chest pain experienced by our patient may have started with a viral infection of endothelial cells in myocardial vessels, leading to endothelial dysfunction and coronary vasospasms; the subsequent migration of inflammatory cells into the myocardial interstitium preceded myocyte damage[14]. In addition, the patient’s rubella virus-specific IgG and IgM antibody levels were abnormally elevated. To sum up, we confirmed the diagnosis of myocarditis. The diagnosis of viral myocarditis was established according to the 2015 ESC myocarditis task force guidelines. Although the patient did not undergo EMB, the 2015 ESC guidelines on CMR for pericardial disease recommend confirmation of myocardial involvement (myocarditis) as a Class I recommendation[15]. Regarding viral infection, without EMB, the pathogen can only be suspected as rubella virus. However, due to the combination of symptoms and signs in this patient, and the history of flu-infection, we still consider this as a case of viral myocarditis.
We also performed a literature review (Table 1)[16-25]. MEDLINE was searched via PubMed for English articles using the terms “myocarditis” AND “chest pain or acute coronary syndrome”; the type of article searched for was “case reports” and 100 such reports were found. Articles were included if (1) the cause of myocarditis was confirmed or suspected as viral infection; (2) there were no concomitant diseases, such as pericarditis or myopercarditis; and (3) the age of the patients was greater than 12 years. The search identified 10 articles.
| Ref. | Age (Yr) | Sex | CK-MB (U/L) | TnI (ng/mL) | CRP (mg/L) | Premonitory symptoms | Medical history | Etiology | ECGZ | UCG | CAG | CMR | Treatment |
| Drighil et al[16] | 17 | F | 39 | 2.99 | 8 | Vesicular rash, flu-like infection | Healthy | Varicella zoster virus | N/A | Apical, inferolateral, inferoseptal hypokinesis, normal LVEF | N/A | Inferoseptal hypokinesis, minimal pericardial effusion | Acyclovir, diclofenac |
| Raj et al[17] | 34 | F | N/A | 3.8 | 2.96 | N/A | N/A | HIV | AnterolateralST elevation | N/A | N/A | epicardialand midwall LGE | Antiretroviral |
| Bigalke et al[18] | 67 | F | 69 | 24.2 | 0.96 | Flu-like infection | Healthy | human herpes virus 6 subtype A | ST elevation | N/A | Normal | N/A | Medication for heart, failure |
| Chen et al[19] | 20 | M | N/A | 2.3 | N/A | Flu-like infection | Healthy | Unk-nown | Inferolateral ST elevation inferolaterally | Inferior and inferolateral wall hypokinesis, LVEF 45% | N/A | N/A | No use |
| Noutsias et al[20] | 39 | F | 45 | N/A | N/A | Flu-like infection | Healthy | Parvovirus B19 | II, III, avF ST elevation | N/A | Normal | Apical thrombi, apical and inferoseptal LGE, inferolateral segment motion abnormality | Heparin, nitroglycerin |
| Gibelli et al[21] | 26 | M | 76 | 5.5 | N/A | Flu-like infection | Healthy | N/A | Inferolateral ST elevation, right bundle branch block | Global hypokinesia; LVEF 48% | Normal | LGE of the subepicardium of the left inferolateral wall | Heparin, nitroglycerin |
| Hakeem et al[22] | 24 | M | 34.6 | 10 | N/A | Flu-like infection | Healthy | Adenovirus | Normal | Inferior and posterior wall hypokinesis | Normal | Subepicardial and intramyocardial LGE | Lisinopril, carvedilol |
| Mancio et al[23] | 49 | M | N/A | 8.94 | 8.45 | Flu-like infection | Hypertension, dyslipidemia, obstructive sleep apnea | Coxsackie A9 virus | Grade 3 atrioventricular block | Right-ventricular wall hyperkinesia | Normal | Subepicardial LGE of the right ventricular wall | Non-steroidal anti-inflammatory drug |
| Deason et al[24] | 23 | M | 36 | 20.5 | N/A | Flu-like infection | Healthy | N/A | Peaked T waves in anterior leads | Normal | N/A | N/A | Antibio-tics |
| Barto[25] | 30 | M | 97.9 | 29.8 | N/A | Flu-like infection | N/A | N/A | II, III, avF ST elevation | and N/A | Normal | N/A | Pain relieving drug |
Except for human immunodeficiency virus, all cases presented with flu-like premonitory symptoms. Four of the ten cases did not have a specific pathogen; however, combined with other evaluations, they were also diagnosed with viral myocarditis. Dr. Chen holds the view that biopsy may be more appropriate for fulminant or giant cell myocarditis[19].
The ECG showed ST-T segment changes in most of the eight patients. Our case showed a change typical of acute coronary syndrome, and this is a major reason why we first suspected acute coronary syndrome.
Six of the ten patients underwent CMR. All subjects showed wall motion abnormalities; five showed LGE. A feature of our case is that we performed the coronary angiography and observed a complete process from severe edema of the ventricular wall to significant reduction in short-term review and dissipation after 13 mo. The area of LGE was significantly smaller than that on T2WI the first time, and the ECG showed ischemia on the inferior wall and over a wide area of the anterior wall. We believe that the main cause of ischemia was not necrosis, but edema. Edema of the myocardium causes microcirculatory disturbances, leading to myocardial ischemia at the corresponding site. After 12 d, a review of the CMR revealed that the edema had subsided compared with the previous scan; the thickest part was reduced from 20 to 17 mm and the enhancement moved to the middle myocardium. Anti-heart failure and antiviral drugs were used for treatment. Although the optimal treatment for myocarditis remains unclear, our patient's edema decreased within 12 d after treatment, proving that the application of anti-heart failure and antiviral drugs is effective. No abnormal heart function was detected at the 13-mo follow-up examination. Thirteen months later, CMR showed that the patient’s edema had disappeared; the thickest wall was only approximately 14 mm. The apical septal LGE suggested fibrosis in the myocardium. A previous study showed that CMR is more likely to detect myocardial damage if the scan is performed within 2 wk after the onset of myocardial infarction[26]. The levels of cardiac and inflammatory markers were also decreased compared to normal; thus, dynamic observation by CMR is essential. A previous study demonstrated that cardiac enzymes and inflammatory parameters do not sufficiently reflect LGE in myocarditis[27]. Therefore, in patients presenting with acute coronary symptoms, abnormal ECG, and raised troponin levels, early myocardial magnetic resonance imaging is essential.
In this case, we conclude that myocarditis may have mimicked myocardial infarction. For a middle-aged man with myocardial infarction, the possibility of acute myocardial infarction should be ruled out first. It is also important to consider myocarditis, such that early CMR has a strategic role in the differential diagnosis. Traditional heart failure medications and antiviral treatments are effective for myocarditis.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, research and experimental
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Nag DS, Sijens PE S-Editor: Zhang L L-Editor: Wang TQ E-Editor: Qi LL
| 1. | Magnani JW, Dec GW. Myocarditis: current trends in diagnosis and treatment. Circulation. 2006;113:876-890. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 424] [Cited by in RCA: 376] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 2. | Andréoletti L, Lévêque N, Boulagnon C, Brasselet C, Fornes P. Viral causes of human myocarditis. Arch Cardiovasc Dis. 2009;102:559-568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 145] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 3. | Schultz JC, Hilliard AA, Cooper LT, Rihal CS. Diagnosis and treatment of viral myocarditis. Mayo Clin Proc. 2009;84:1001-1009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 159] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 4. | Kawai C, Matsumori A. Dilated cardiomyopathy update: infectious-immune theory revisited. Heart Fail Rev. 2013;18:703-714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 5. | Friedrich MG, Sechtem U, Schulz-Menger J, Holmvang G, Alakija P, Cooper LT, White JA, Abdel-Aty H, Gutberlet M, Prasad S, Aletras A, Laissy JP, Paterson I, Filipchuk NG, Kumar A, Pauschinger M, Liu P; International Consensus Group on Cardiovascular Magnetic Resonance in Myocarditis. Cardiovascular magnetic resonance in myocarditis: A JACC White Paper. J Am Coll Cardiol. 2009;53:1475-1487. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1951] [Cited by in RCA: 1740] [Article Influence: 108.8] [Reference Citation Analysis (0)] |
| 6. | Sarda L, Colin P, Boccara F, Daou D, Lebtahi R, Faraggi M, Nguyen C, Cohen A, Slama MS, Steg PG, Le Guludec D. Myocarditis in patients with clinical presentation of myocardial infarction and normal coronary angiograms. J Am Coll Cardiol. 2001;37:786-792. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 112] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 7. | Gerbaud E, Harcaut E, Coste P, Erickson M, Lederlin M, Labèque JN, Perron JM, Cochet H, Dos Santos P, Durrieu-Jaïs C, Laurent F, Montaudon M. Cardiac magnetic resonance imaging for the diagnosis of patients presenting with chest pain, raised troponin, and unobstructed coronary arteries. Int J Cardiovasc Imaging. 2012;28:783-794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 59] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 8. | Lurz P, Luecke C, Eitel I, Föhrenbach F, Frank C, Grothoff M, de Waha S, Rommel KP, Lurz JA, Klingel K, Kandolf R, Schuler G, Thiele H, Gutberlet M. Comprehensive Cardiac Magnetic Resonance Imaging in Patients With Suspected Myocarditis: The MyoRacer-Trial. J Am Coll Cardiol. 2016;67:1800-1811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 288] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 9. | Chopra H, Arangalage D, Bouleti C, Zarka S, Fayard F, Chillon S, Laissy JP, Henry-Feugeas MC, Steg PG, Vahanian A, Ou P. Prognostic value of the infarct- and non-infarct like patterns and cardiovascular magnetic resonance parameters on long-term outcome of patients after acute myocarditis. Int J Cardiol. 2016;212:63-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 36] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 10. | Caforio AL, Pankuweit S, Arbustini E, Basso C, Gimeno-Blanes J, Felix SB, Fu M, Heliö T, Heymans S, Jahns R, Klingel K, Linhart A, Maisch B, McKenna W, Mogensen J, Pinto YM, Ristic A, Schultheiss HP, Seggewiss H, Tavazzi L, Thiene G, Yilmaz A, Charron P, Elliott PM; European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2013;34:2636-2648, 2648a-2648d. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1714] [Cited by in RCA: 2259] [Article Influence: 188.3] [Reference Citation Analysis (0)] |
| 11. | Caforio ALP, Adler Y, Agostini C, Allanore Y, Anastasakis A, Arad M, Böhm M, Charron P, Elliott PM, Eriksson U, Felix SB, Garcia-Pavia P, Hachulla E, Heymans S, Imazio M, Klingel K, Marcolongo R, Matucci Cerinic M, Pantazis A, Plein S, Poli V, Rigopoulos A, Seferovic P, Shoenfeld Y, Zamorano JL, Linhart A. Diagnosis and management of myocardial involvement in systemic immune-mediated diseases: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Disease. Eur Heart J. 2017;38:2649-2662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 111] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 12. | Gullestad L, Aass H, Fjeld JG, Wikeby L, Andreassen AK, Ihlen H, Simonsen S, Kjekshus J, Nitter-Hauge S, Ueland T, Lien E, Frøland SS, Aukrust P. Immunomodulating therapy with intravenous immunoglobulin in patients with chronic heart failure. Circulation. 2001;103:220-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 243] [Cited by in RCA: 242] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 13. | Monney PA, Sekhri N, Burchell T, Knight C, Davies C, Deaner A, Sheaf M, Baithun S, Petersen S, Wragg A, Jain A, Westwood M, Mills P, Mathur A, Mohiddin SA. Acute myocarditis presenting as acute coronary syndrome: role of early cardiac magnetic resonance in its diagnosis. Heart. 2011;97:1312-1318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 94] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 14. | Yilmaz A, Mahrholdt H, Athanasiadis A, Vogelsberg H, Meinhardt G, Voehringer M, Kispert EM, Deluigi C, Baccouche H, Spodarev E, Klingel K, Kandolf R, Sechtem U. Coronary vasospasm as the underlying cause for chest pain in patients with PVB19 myocarditis. Heart. 2008;94:1456-1463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 115] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 15. | Adler Y, Charron P, Imazio M, Badano L, Barón-Esquivias G, Bogaert J, Brucato A, Gueret P, Klingel K, Lionis C, Maisch B, Mayosi B, Pavie A, Ristic AD, Sabaté Tenas M, Seferovic P, Swedberg K, Tomkowski W; ESC Scientific Document Group. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases: The Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC)Endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2015;36:2921-2964. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1457] [Cited by in RCA: 1582] [Article Influence: 158.2] [Reference Citation Analysis (0)] |
| 16. | Drighil A, Hammiri AE, Belmourida F. Chest Pain in a 17-Year-Old Girl with Chickenpox. Pediatr Ann. 2015;44:359-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 17. | Raj V, Joshi S, Pennell DJ. Images in Cardiovascular Medicine. Cardiac magnetic resonance of acute myocarditis in an human immunodeficiency virus patient presenting with acute chest pain syndrome. Circulation. 2010;121:2777-2779. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 18. | Bigalke B, Klingel K, May AE, Kandolf R, Gawaz MG. Human herpesvirus 6 subtype A-associated myocarditis with 'apical ballooning'. Can J Cardiol. 2007;23:393-395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 19. | Chen EF, Paterick TE. 20-year-old man with chest pain and malaise. Mayo Clin Proc. 2007;82:1124-1127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 20. | Noutsias M, Kuehl U, Lassner D, Gross U, Pauschinger M, Schultheiss HP, Gutberlet M. Images in cardiovascular medicine. Parvovirus-B19-associated active myocarditis with biventricular thrombi. Results of endomyocardial biopsy investigations and cardiac magnetic resonance imaging. Circulation. 2007;115:e378-e380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 21. | Gibelli G, Devizzi S, Brioschi P, Rimini A, Biasi S. Sudden ST elevation with angina-like pain in myocarditis. An uncommon course of a common disease: strategic role of cardiac magnetic resonance. J Cardiovasc Med (Hagerstown). 2009;10:264-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Hakeem A, Bhatti S, Fuh A, Mallof M, Stone C, Thornton F, Chang SM. Viral myocarditis masquerading acute coronary syndrome (ACS)--MRI to the rescue. Int J Cardiol. 2007;119:e74-e76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 23. | Mancio J, Bettencourt N, Oliveira M, Pires-Morais G, Ribeiro VG. Acute right ventricular myocarditis presenting with chest pain and syncope. BMJ Case Rep. 2013;2013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 24. | Deason J, Hope B. A 23-year-old man with chest pressure, pallor, tachypnea, and tonsillitis. J Emerg Nurs. 2005;31:199-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 25. | Barto D. A case of mistaken identity: an inflamed heart. Nursing. 2014;44:18-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 26. | Monney PA, Sekhri N, Burchell T, Knight C, Davies C, Deaner A, Sheaf M, Baithun S, Petersen S, Wragg A, Jain A, Westwood M, Mills P, Mathur A, Mohiddin SA. Acute myocarditis presenting as acute coronary syndrome: role of early cardiac magnetic resonance in its diagnosis. Heart. 2011;97:1283. [RCA] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 27. | Berg J, Kottwitz J, Baltensperger N, Kissel CK, Lovrinovic M, Mehra T, Scherff F, Schmied C, Templin C, Lüscher TF, Heidecker B, Manka R. Cardiac Magnetic Resonance Imaging in Myocarditis Reveals Persistent Disease Activity Despite Normalization of Cardiac Enzymes and Inflammatory Parameters at 3-Month Follow-Up. Circ Heart Fail. 2017;10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 60] [Article Influence: 7.5] [Reference Citation Analysis (0)] |