Published online Apr 26, 2019. doi: 10.12998/wjcc.v7.i8.961
Peer-review started: December 3, 2018
First decision: January 26, 2018
Revised: January 31, 2018
Accepted: February 26, 2019
Article in press: February 26, 2019
Published online: April 26, 2019
Processing time: 146 Days and 23.2 Hours
Adrenocorticotropic hormone (ACTH)-independent Cushing’s syndrome (CS) is mostly due to unilateral tumors, with bilateral tumors rarely reported. Its common causes include primary pigmented nodular adrenocortical disease, ACTH-independent macronodular adrenal hyperplasia, and bilateral adrenocortical adenomas (BAAs) or carcinomas. BAAs causing ACTH-independent CS are rare; up to now, fewer than 40 BAA cases have been reported. The accurate diagnosis and evaluation of BAAs are critical for determining optimal treatment options. Adrenal vein sampling (AVS) is a good way to diagnose ACTH-independent CS.
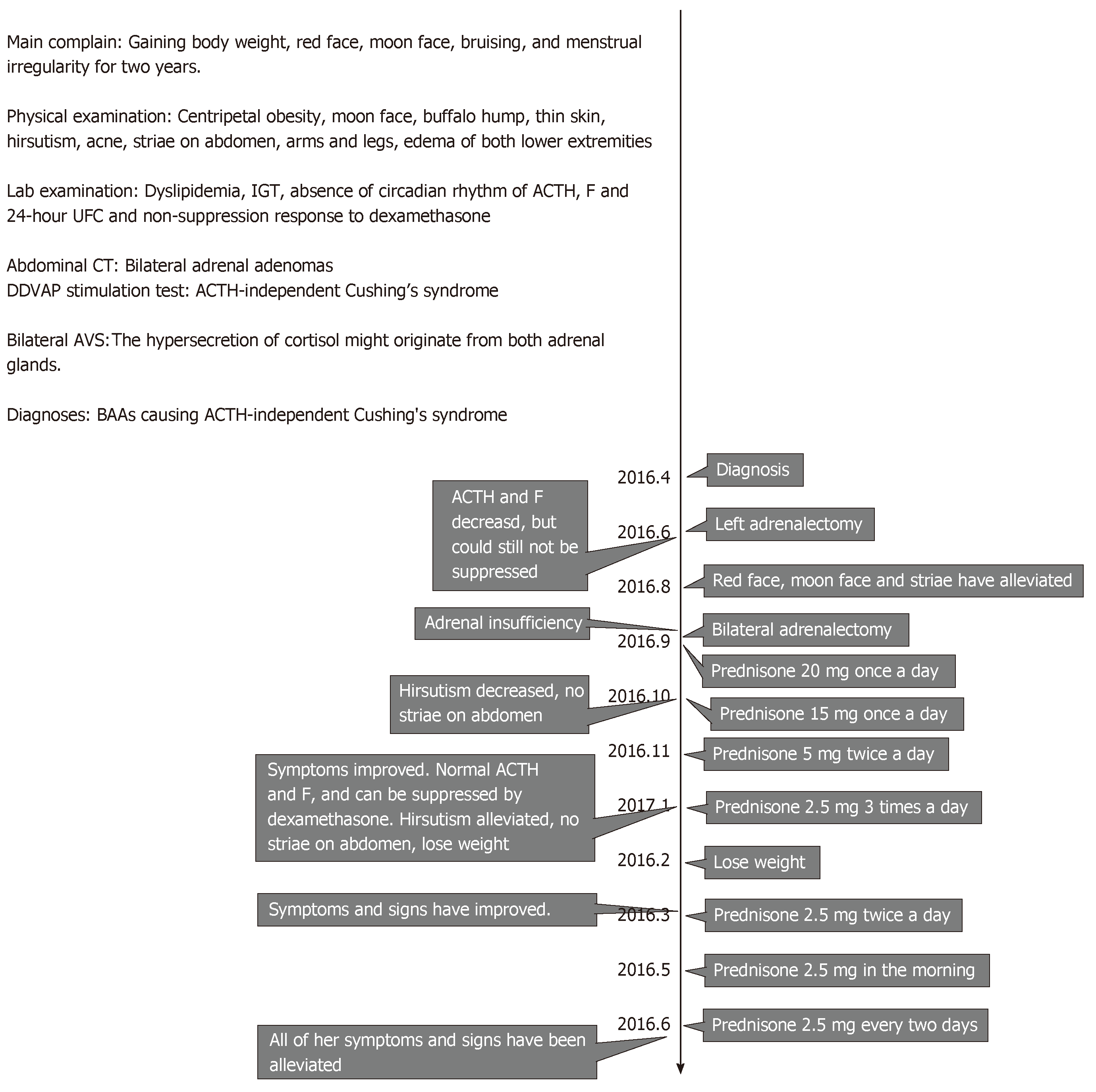
A 31-year-old woman had a typical appearance of CS. The oral glucose tolerance test showed impaired glucose tolerance and obviously increased insulin and C-peptide levels. Her baseline serum cortisol and urine free cortisol were elevated and did not show either a circadian rhythm or suppression with dexamethasone administration. The peripheral 1-deamino-8-D-arginine-vasopressin (DDVAP) stimulation test showed a delay of the peak level, which was 1.05 times as high as the baseline level. Bilateral AVS results suggested the possibility of BAAs. Abdominal computed tomography showed bilateral adrenal adenomas with atrophic adrenal glands (right: 3.1 cm × 2.0 cm × 1.9 cm; left: 2.2 cm × 1.9 cm × 2.1 cm). Magnetic resonance imaging of the pituitary gland demonstrated normal findings. A left adenomectomy by retroperitoneoscopy was performed first, followed by resection of the right-side adrenal mass 3 mo later. Biopsy results of both adenomas showed cortical tumors. Evaluations of ACTH and cortisol showed a significant decrease after left adenomectomy but could still not be suppressed, and the circadian rhythm was absent. Following bilateral adenomectomy, this patient has been administered with prednisone until now, all of her symptoms were alleviated, and she had normal blood pressure without edema in either of her lower extremities.
BAAs causing ACTH-independent CS are rare. AVS is of great significance for obtaining information on the functional state of BAAs before surgery.
Core tip: Bilateral adrenocortical adenomas (BAAs) causing adrenocorticotropic hormone (ACTH)-independent Cushing syndrome (CS) are rare; up to now, fewer than 40 BAA cases have been reported. The accurate diagnosis and evaluation of BAAs are critical for determining optimal treatment options. We report a rare case of BAAs causing ACTH-independent CS, which was diagnosed by adrenal vein sampling (AVS) and computed tomography before surgery. The patient underwent a bilateral retroperitoneoscopy adenomectomy and glucocorticoid replacement therapy. Our case indicates that AVS is of great significance for obtaining information on the functional state of BAAs before surgery.
- Citation: Gu YL, Gu WJ, Dou JT, Lv ZH, Li J, Zhang SC, Yang GQ, Guo QH, Ba JM, Zang L, Jin N, Du J, Pei Y, Mu YM. Bilateral adrenocortical adenomas causing adrenocorticotropic hormone-independent Cushing’s syndrome: A case report and review of the literature. World J Clin Cases 2019; 7(8): 961-971
- URL: https://www.wjgnet.com/2307-8960/full/v7/i8/961.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v7.i8.961
Inappropriate exposure to excessive concentrations of free glucocorticoids results in Cushing’s syndrome (CS)[1], with the clinical features of centripetal obesity, moon face, hirsutism, plethora, and striae, among others. The excess of glucocorticoid comes from endogenous or exogenous sources[2]. Endogenous causes of CS are rare. The incidence rates of CS are estimated to be 2 to 3 cases per million people per year in Europe and 10-15 cases per million people per year in America[3]. Some studies have revealed that subclinical hypercortisolism is significantly more possible in patients with bilateral incidentalomas[4]. Endogenous CS can be divided into adre-nocorticotropic hormone (ACTH)-independent and -dependent forms. ACTH-independent CS is mostly due to unilateral tumors, with bilateral tumors rarely reported. Its main causes include ACTH-independent macronodular adrenal hyperplasia (AIMAH), primary pigmented nodular adrenocortical disease (PPNAD), and bilateral adrenocortical adenomas (BAAs) or carcinomas[5,6]. BAAs causing ACTH-independent CS are rare; up to now, fewer than 40 BAA cases have been reported. The accurate diagnosis and evaluation of BAAs are critical for determining optimal treatment options. Herein, we report a Chinese patient with BAAs, who was diagnosed with ACTH-independent CS by adrenal vein sampling (AVS) and computed tomography (CT).
A 31-year-old woman, who complained of gaining body weight, red face, moon face, bruising, and menstrual irregularity for two years, presented to our outpatient department.
The patient’s symptoms started two years ago with weight gain, red face, moon face, bruising, and menstrual irregularity.
She was diagnosed with hypertension a month ago and was medicated with nifedipine delayed-release tablets (II) 40 mg twice a day. Her family history was negative.
Physical examination revealed centripetal obesity, moon face, buffalo hump, thin skin, hirsutism, acne, striae on her abdomen, arms, and legs, and edema of both lower extremities. The height of this patient was 159 cm, and her body weight was 73.5 kg. Her blood pressure was 130/100 mmHg, with a pulse of 84 beats per minute.
Her blood counts were within the normal limits, and she had no serum electrolytes disorders. The biochemical data showed dyslipidemia. The oral glucose tolerance test showed impaired glucose tolerance (IGT) and obviously increased insulin and C-peptide levels. The endocrinological data showed elevated cortisol (761.0 nmol/L) and 24-h urine free cortisol (UFC) (1650.7 nmol/L) and the absence of a circadian rhythm and a non-suppression response to dexamethasone (dexamethasone 0.5 mg every 6 h for 2 d and 2 mg every 6 h for 2 d) administration (Table 1).
| Baseline | Low-dose dexam test | High-dose dexam test | ||||
| 00:00 h | 08:00 h | 16:00 h | ||||
| Before the operation | ACTH (pmol/L) | 4.26 | 4.13 | 3.62 | 3.51 | 4.1 |
| F (nmol/L) | 606.42 | 761 | 618.08 | 579.34 | 689.18 | |
| UFC (nmol/L) | 1650.7 | - | - | 3073.5 | 2122.6 | |
| After unilateral operation | ACTH (pmol/L) | 7.84 | 7.86 | 7.7 | 9.28 | - |
| F (nmol/L) | 391.78 | 440.08 | 455.65 | 414.36 | - | |
| UFC (nmol/L) | 1024.9 | - | - | 1117 | - | |
| After bilateral operations | ACTH (pmol/L) | 4.66 | 4.86 | 3.66 | 4.93 | - |
| F (nmol/L) | <25.7 | <25.7 | <25.7 | <25.7 | - | |
| UFC (nmol/L) | - | <25.7 | - | - | - | |
Abdominal CT showed bilateral adrenal adenomas with atrophic adrenal glands (right: 3.1 cm × 2.0 cm × 1.9 cm; left: 2.2 cm × 1.9 cm × 2.1 cm), which were enhanced homogeneously with contrast material (Figure 1). A magnetic resonance imaging (MRI) scan of the pituitary gland demonstrated normal findings.
The patient was further evaluated with 1-deamino-8-D-arginine-vasopressin (DDVAP). The peripheral DDVAP stimulation test showed a baseline ACTH level of 6.9 pmol/L, and ACTH reached a peak level (7.26 pmol/L) at 90 min after stimulation, which was 1.05 times as high as the baseline level (Table 2). Then, we performed bilateral AVS to examine if the two adenomas were cortisol-secreting. Bilateral AVS results were as follows: the left-side adrenal vein (LAV)-to-peripheral vein (PV) cortisol gradient was 16.00; the right-side AV (RAV)-to-PV gradient was 8.29; and the ratio of the LAV cortisol-to-left side aldosterone (ALD) gradient to the RAV cortisol-to-right side ALD gradient was 1.23 (Table 3).
| Time | ACTH (pmol/L) | F (nmol/L) |
| -15 min | 6.88 | 640.24 |
| 0 min | 6.97 | 624.03 |
| 15 min | 6.7 | 637.87 |
| 30 min | 6.59 | 590.82 |
| 45 min | 6.59 | 625.47 |
| 60 min | 7.19 | 598.56 |
| 90 min | 7.26 | 524.71 |
| 120 min | 6.68 | 577.39 |
| LAV | RAV | L/R | PV | |
| ACTH (pmol/L) | 8.50 | 8.73 | / | 9.10 |
| F (nmol/L) | 11093.75 | 5746.61 | 14.86 | 693.38 |
| ALD (ng/dL) | 13.95 | 8.90 | / | 5.0 |
| F/ALD | 795 | 645 | 1.23 | / |
It is important to know which side is functioning, which is hard to determine by imaging. We can do bilateral AVS to examine if the two adenomas are cortisol-secreting.
BAAs often: (A) occur in mid-aged females; (B) have varying clinical symptoms; (C) are a kind of ACTH-independent CS, with hypercortisolism, absence of serum cortisol diurnal rhythm, and elevated UFC, though ACTH is in the normal range; (D) are 2.0-3.5 cm in diameter on CT; and (F) have normal finding in MRI of the pituitary. We should determine the source of ACTH, and testing the rhythm of ACTH-F is important.
The patient should undergo surgical treatment twice separately after obtaining the result of bilateral AVS.
The final diagnosis was ACTH-independent CS and BAAs.
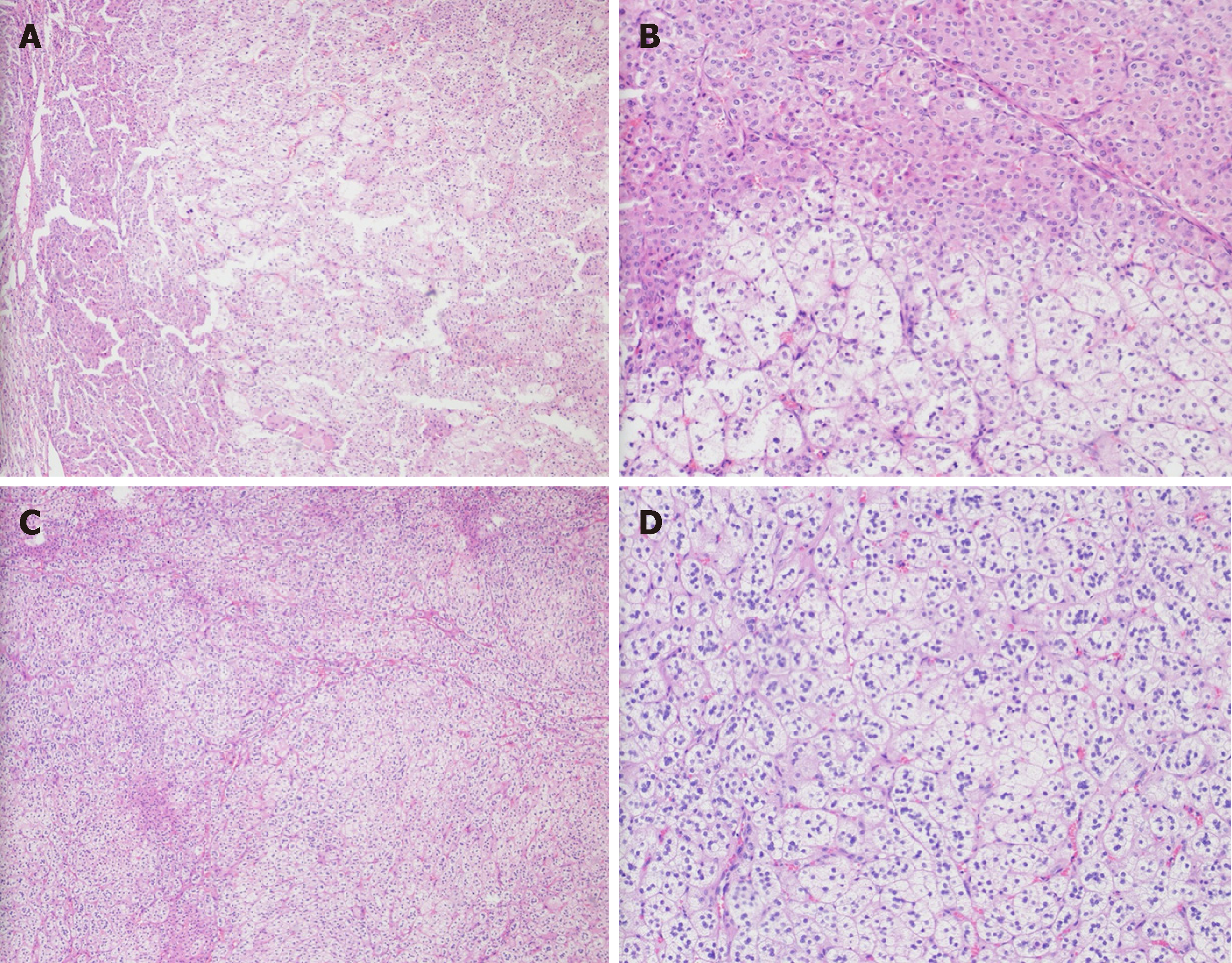
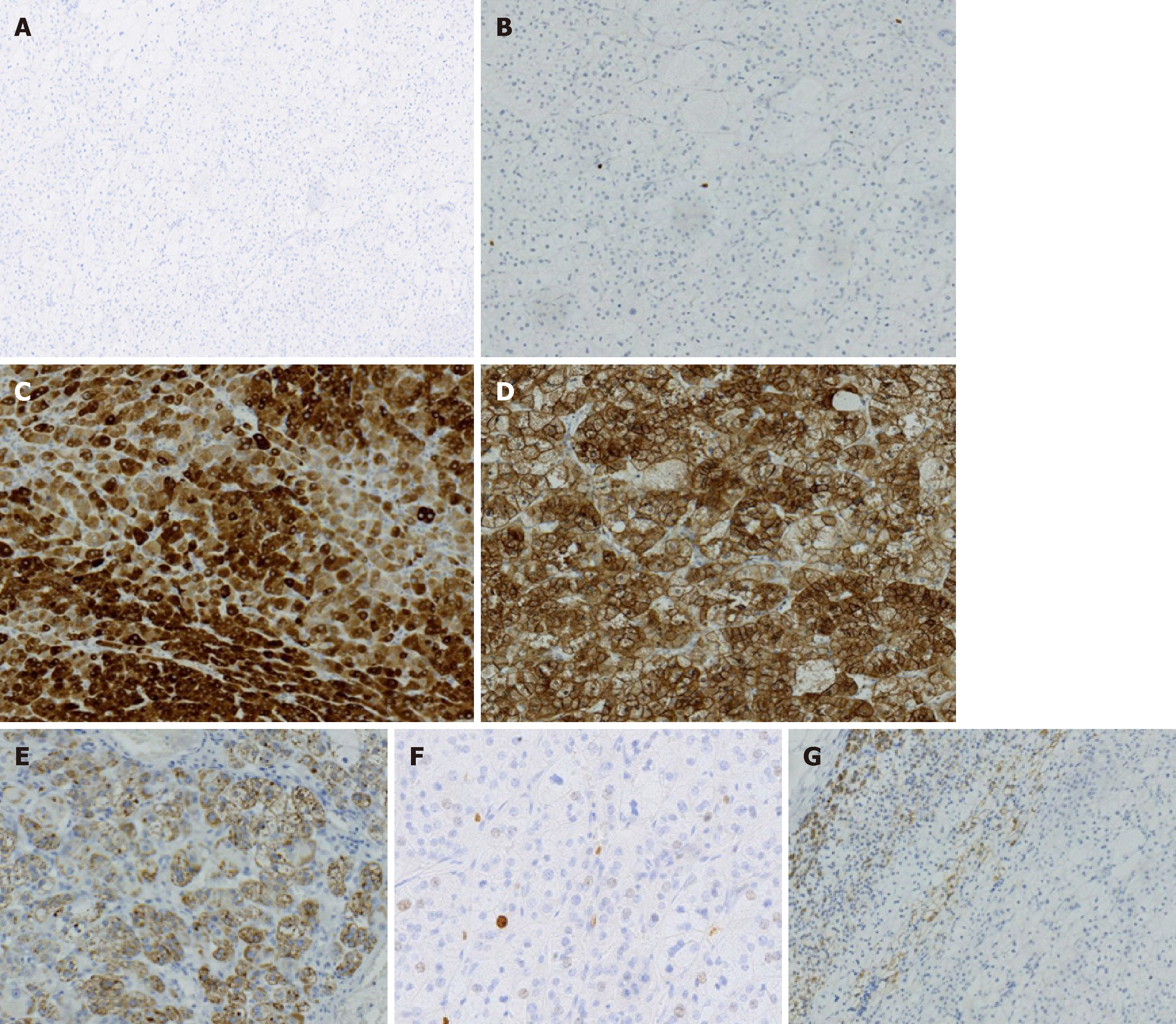
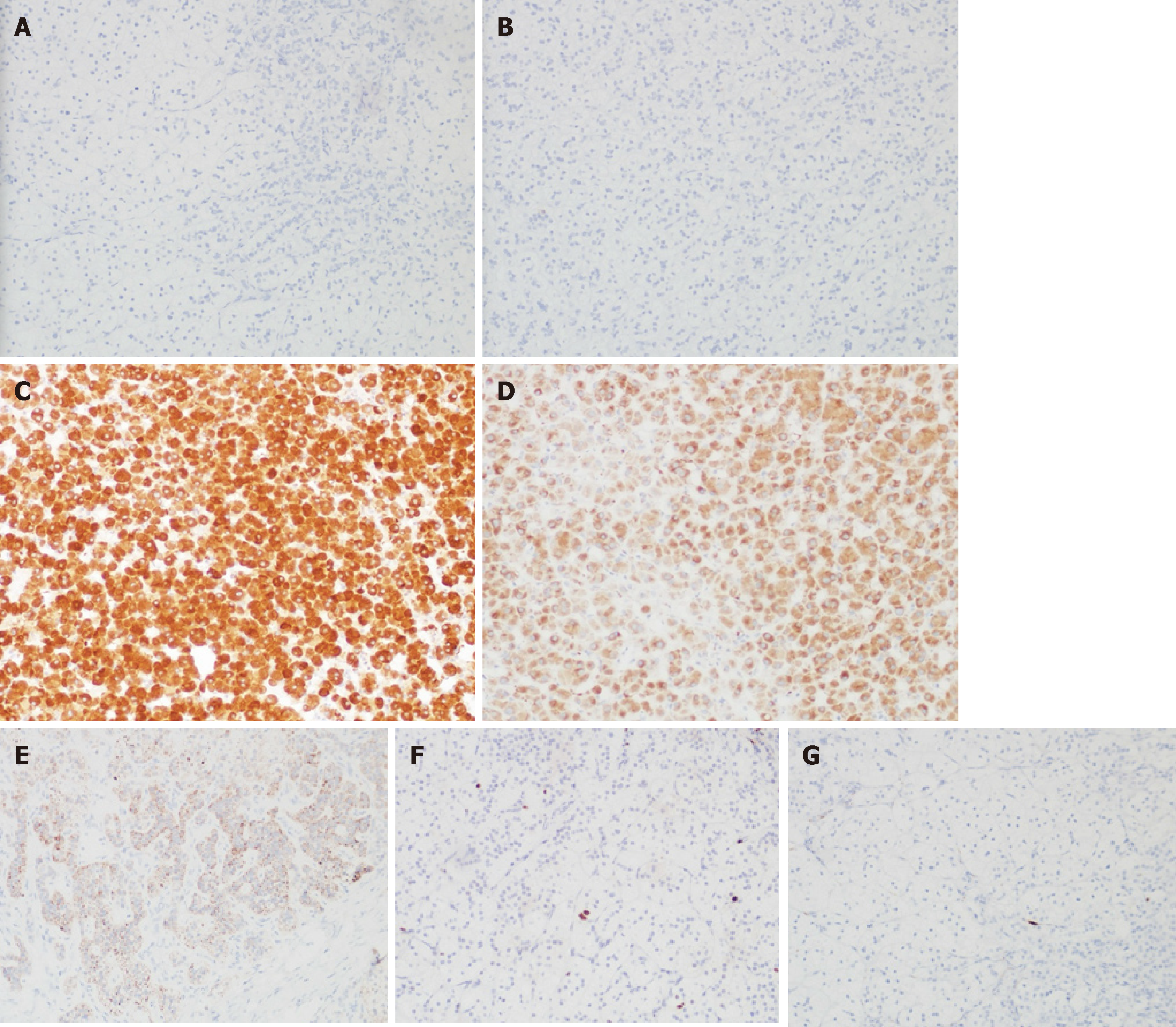
A left adenomectomy by retroperitoneoscopy was performed on June 21, 2016. Irregular gray yellow tissue that measured 3.5 cm × 2.5 cm × 1 cm was observed on the left side. The cut surface was solid and golden yellow in color. Histologically, a single well-encapsulated adrenocortical tumor was observed in the adrenal gland. The Weiss score was 1. The immunohistochemistry results revealed MelanA(+), CgA(-), Syn(+), Ki-67(+2%), Inhibin-a(+), CK(-), and S-100(spotty+) (Figure 2A, 2B and Figure 3). Resection of the right-side adrenal mass was performed 3 mo later. The biopsy revealed nodular tissue on the right side measuring 2 cm × 1.8 cm × 1.5 cm. The cut surface was golden yellow in color. The Weiss score was 1. The immuno-histochemistry results revealed MelanA(+), CgA(-), Syn(+), Ki-67(+2%), Inhibin-a(+), CK(-), S-100(spotty +) (Figure 2C, 2D and Figure 4).
Evaluations of ACTH and cortisol showed a significant decrease after the surgery. All of her symptoms were alleviated, and she had normal blood pressure and no edema in either of her lower extremities. She was administered with prednisone 20 mg once a day for half a month, which was then tapered to 15 mg once a day for a month and 5 mg twice a day, and finally 2.5 mg twice a day. The last follow-up was on June 5, 2017, at which point all of her symptoms and signs have been alleviated (Figure 5).
Our case was confirmed as ACTH independency based on: (1) Very low plasma ACTH with increased serum cortisol level; (2) Unresponsiveness to a high-dose dexamethasone suppression test; (3) Unresponsiveness to a DDVAP stimulation test; and (4) MRI scan of the pituitary gland demonstrated normal findings; however, CT showed bilateral adrenal adenomas with atrophic adrenal glands. According to the tests above, ACTH-independent CS was diagnosed. Cases of ACTH-independent CS associated with adrenocortical adenoma are secondary to unilateral adrenocortical adenomas in approximately 90% of cases[7]. In contrast, bilateral cortisol-secreting tumors are a rare cause of CS[8].
As we know, the possibilities of bilateral adrenal space-occupying lesion are mainly: (1) BAAs; (2) Carcinomas; (3) PPNAD; and (4) AIMAH. Markedly enlarged bilateral hyperplastic adrenal glands with multiple macronodules up to 5 cm in diameter are a characteristic histopathological feature of AIMAH[1]. This disorder may be associated with other receptors besides ACTH, which are expressed in the adrenal cortex ectopically, such as gastric inhibitory polypeptide, luteinizing hormone/ human chorionic gonadotropin and others[9,10]. PPNAD is another bilateral adrenal hyperplastic disease; CT scan shows bilateral small discrete nodules between 2 and 5 mm in size without overall enlargement of the glands. The possible mechanism may be a novel mutation in the type lα regulatory subunit of the protein kinase A (PRKAR1A) gene: p.E17X (c.49G>T), which confirms that the diagnosis of PPNAD could be one component of Carney complex[11]. While carcinoma seems much more aggressive, with a heavier and larger mass, it is always complicated with severe hypertension, masculinization, hypokalemia, or alkalosis. On CT scan, the tumor is generally bigger than 5 cm in diameter, with heterogeneous enhancement with contrast agent[12]. BAAs were first reported by CHAPELL in 1963[13]. To the best of our knowledge, fewer than 40 BAAs cases have been reported. BAAs are the most significant feature of this case.
The mechanism of BAAs is unclear. Some studies have revealed that steroidogenic enzymes, such as P450scc, 3β-HSD, P450c21, P450c17, and P450c11, are intensely expressed in the compact cells of the adenoma, while their expression profiles are negative in the adjacent non-neoplastic cortex. At the same time, the histological features may differ between two adenomas[14,15]. It is difficult to distinguish between functioning or nonfunctioning adrenal cortical masses by the imaging characteristics on CT scanning[1]. AVS is a helpful method to distinguish between these conditions. The compound 6 beta-(131I)-iodomethyl-19-norcholesterol (NP-59) is another possible means for evaluating the function of an adenoma; however, NP-59 scintigraphy is never a common imaging modality in China. AVS provides important information concerning the laterality of excessive ALD, but it also has advantages compared with adrenal scintigraphy and CT in the diagnosis of CS and BAAs[16,17]. However, only a few cases have been reported with the use of AVS in the diagnosis of BAAs before surgery. According to the Mayo Clinic[16], AV-to-PV cortisol gradient greater than 6.5 is more likely to be a cortisol-secreting adenoma. In our case, the gradient of LAV-to-PV cortisol was 16, and the RAV-to-PV cortisol gradient was 8.2. The “ALD-corrected” high-side to low-side AV cortisol gradient was 2.3 or greater, which is consistent with autonomous cortisol secretion from predominantly one adrenal gland; a gradient of 2.0 or less indicates bilateral cortisol hypersecretion. The ratio of L/R of our patient was 1.23, less than 2.0. All of these pieces of evidence indicated that the hypersecretion of cortisol might originate from both adrenal glands. The first case of BAAs in China was reported in the medical literature by our team[18]. However, the AVS technique was not available at that time; therefore, we could not determine which side was functioning before the surgery.
Extirpation of the functioning adenoma is the optimal choice for bilateral CS adenomas[19]. AVS is a useful accessory examination before surgery to design a surgical plan that makes much more sense. Some authors believe that if AVS is unavailable, the larger of the two adenomas should be removed first, and then further management should be determined according to the postoperative hormone tests. However, at least 5 years of follow-up examinations is required, because the con-tralateral adrenal adenoma might have clinically significant endocrine function later[1,8]. Bilateral adenomas generally appear at the same time, but there has been a report of an adenoma appearing 9 years after the contralateral surgery[8]. In our case, the bilateral adenomas were both functioning; we resected the left one first because of its thriving function on AVS. Three months later, we removed the right adenoma. When bilateral adrenal tumors are considered, a unilateral or bilateral adenoma resection is a better choice if functional adrenal tissue can be preserved to avoid acute adrenal insufficiency or lifelong steroid replacement. However, how much residual adrenal tissue is required for adequate adrenal function has not been much known[18]. Brauckhoff estimated that preservation of approximately one-third of the entire adrenal gland could provide sufficient function[20]. Several authors have advocated preserving the adrenal vein, because adequate venous preservation may be important for maintaining the functionality of the remnant tissue[21,22]. But, in most cases, impaired adrenocortical function is likely caused by atrophy of the normal adrenal tissue. Few studies have demonstrated the feasibility of a partial adenomectomy for BAAs. Glucocorticoid therapy has to be employed to maintain the balance of water and electrolyte metabolism after surgery. Some studies have reported that long-term inadequate corticosteroid replacement therapy might provoke pituitary ACTH-producing hyperplasia adenoma[23-26]. Therefore, proper use of corticosteroids is significantly important. It has been suggested that corticosteroid replacement therapy can be divided into several doses (for example, it can be divided equally into three doses a day, taken at 9:00, 14:00, and 20:00 or 2/3 of a total dose in the morning and 1/3 in the early afternoon)[27].
Based on the reported cases, the clinical characteristics of BAAs could be summarized as: (1) Mainly occurring in mid-aged females; (2) Varying clinical symptoms, though most patients visit hospital because of typical symptoms; (3) Being a kind of ACTH-independent CS, with hypercortisolism, absence of serum cortisol diurnal rhythm, elevated UFC, and no suppression of serum cortisol after low-dose or high-dose dexamethasone suppression testing, while ACTH cannot be tested or is in the normal range, resulting in no reaction to the ACTH stimulation test; (4) Probably different pathological features and functioning state; (5) Bilateral well-encapsulated adenomas of 2.0 cm - 3.5 cm in diameter on the CT scan, with the presence of single or numerous nodules[19,28]; and (6) Normal findings of the MRI of the pituitary[19]. It has been reported that except for glucocorticoids, BAAs could also secrete mi-neralocorticoids and androgen[29]. In our patients, the plasma renin activity, the ALD concentration, the ALD-to-renin ratio, and the urine epinephrine and dopamine levels were in the normal range, while norepinephrine was only elevated slightly.
When patients present with bilateral adrenal lesions, it is crucial to make a definitive diagnosis before operation since various treatments are prescribed for different causes. Few cases of BAAs diagnosed using AVS have been reported. Our case indicates that AVS might be useful for obtaining differential diagnoses in such cases. Once considering BAAs, it is not recommended that bilateral adenomas be removed simultaneously. Hormone replacement therapy may be needed for fear of adrenal insufficiency, and the tapered discontinuation of the replacement therapy is necessarily slow.
The authors thank all of the treatment groups of the Chinese People’s Liberation Army General Hospital.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, Research and Experimental
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Lalli E, Aseni P S-Editor: Dou Y L-Editor: Wang TQ E-Editor: Wu YXJ
| 1. | Li Z, Zhu Y, Kong C, Yin L, Gao Z, Zhao W, Gong D. Corticotropin-independent Cushing's syndrome in patients with bilateral adrenal masses. Urology. 2011;77:417-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 2. | Melmed S, Polonsky KS, Larsen PR, Kronenberg H. Williams textbook of endocrinology. 13th ed. Elsevier Health Sciences, Philadelphia. 2015;507. |
| 3. | Guaraldi F, Salvatori R. Cushing syndrome: maybe not so uncommon of an endocrine disease. J Am Board Fam Med. 2012;25:199-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 3.9] [Reference Citation Analysis (2)] |
| 4. | Vassiliadi DA, Ntali G, Vicha E, Tsagarakis S. High prevalence of subclinical hypercortisolism in patients with bilateral adrenal incidentalomas: a challenge to management. Clin Endocrinol (Oxf). 2011;74:438-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 5. | Newell-Price J, Bertagna X, Grossman AB, Nieman LK. Cushing's syndrome. Lancet. 2006;367:1605-1617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 914] [Cited by in RCA: 857] [Article Influence: 45.1] [Reference Citation Analysis (1)] |
| 6. | Nieman LK, Biller BM, Findling JW, Newell-Price J, Savage MO, Stewart PM, Montori VM. The diagnosis of Cushing's syndrome: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2008;93:1526-1540. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1971] [Cited by in RCA: 1648] [Article Influence: 96.9] [Reference Citation Analysis (1)] |
| 7. | Nieman LK. Cushing’s syndrome. Endocrinology. 4th ed. Philadelphia. 2001;1691-1720. |
| 8. | Tung SC, Wang PW, Huang TL, Lee WC, Chen WJ. Bilateral adrenocortical adenomas causing ACTH-independent Cushing's syndrome at different periods: a case report and discussion of corticosteroid replacement therapy following bilateral adrenalectomy. J Endocrinol Invest. 2004;27:375-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 9. | Lacroix A, Bolté E, Tremblay J, Dupré J, Poitras P, Fournier H, Garon J, Garrel D, Bayard F, Taillefer R. Gastric inhibitory polypeptide-dependent cortisol hypersecretion--a new cause of Cushing's syndrome. N Engl J Med. 1992;327:974-980. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 195] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 10. | Reznik Y, Allali-Zerah V, Chayvialle JA, Leroyer R, Leymarie P, Travert G, Lebrethon MC, Budi I, Balliere AM, Mahoudeau J. Food-dependent Cushing's syndrome mediated by aberrant adrenal sensitivity to gastric inhibitory polypeptide. N Engl J Med. 1992;327:981-986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 154] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 11. | Mineo R, Tamba S, Yamada Y, Okita T, Kawachi Y, Mori R, Kyo M, Saisho K, Kuroda Y, Yamamoto K, Furuya A, Mukai T, Maekawa T, Nakamura Y, Sasano H, Matsuzawa Y. A Novel Mutation in the type Iα Regulatory Subunit of Protein Kinase A (PRKAR1A) in a Cushing's Syndrome Patient with Primary Pigmented Nodular Adrenocortical Disease. Intern Med. 2016;55:2433-2438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 12. | Zhang ZP, Liu M, Zhu SC, Wan B. 4 cases report of adrenocortical carcinoma and literature review. Zhongguo Yixue Zazhi. 2016;51:62-65. |
| 13. | Chappell AG. Cushing's Syndrome due to Bilateral Adrenal Adenomata. Proc R Soc Med. 1963;56:165-166. [PubMed] |
| 14. | Nomura K, Saito H, Aiba M, Iihara M, Obara T, Takano K. Cushing's syndrome due to bilateral adrenocortical adenomas with unique histological features. Endocr J. 2003;50:155-162. [PubMed] |
| 15. | Tamura H, Sugihara H, Minami S, Emoto N, Shibasaki T, Shuto Y, Shimizu K, Gomi Y, Sasano H, Wakabayashi I. Cushing's syndrome due to bilateral adrenocortical adenomas with different pathological features. Intern Med. 1997;36:804-809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 16. | Young WF, du Plessis H, Thompson GB, Grant CS, Farley DR, Richards ML, Erickson D, Vella A, Stanson AW, Carney JA, Abboud CF, Carpenter PC. The clinical conundrum of corticotropin-independent autonomous cortisol secretion in patients with bilateral adrenal masses. World J Surg. 2008;32:856-862. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 84] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 17. | Yasuda A, Seki T, Ito K, Takagi A, Watanabe D, Nakamura N, Hanai K, Terachi T, Maekawa T, Sasano H, Fukagawa M. A rare case of Cushing's syndrome due to bilateral adrenocortical adenomas. Tokai J Exp Clin Med. 2014;39:158-165. [PubMed] |
| 18. | Yang GQ, Lu JM. A case report of Cushing’s syndrome due to bilateral adrenocortical adenomas. Zhonghua Neifenmidaixie Zazhi. 2001;17:188-189. |
| 19. | Domino JP, Chionh SB, Lomanto D, Katara AN, Rauff A, Cheah WK. Laparoscopic partial adrenalectomy for bilateral cortisol-secreting adenomas. Asian J Surg. 2007;30:154-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 20. | Brauckhoff M, Gimm O, Thanh PN, Bär A, Ukkat J, Brauckhoff K, Bonsch T, Dralle H. Critical size of residual adrenal tissue and recovery from impaired early postoperative adrenocortical function after sub-total bilateral adrenalectomy. Surg. 2003;134:1020-1027. [RCA] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 65] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 21. | Janetschek G, Finkenstedt G, Gasser R, Waibel UG, Peschel R, Bartsch G, Neumann HP. Laparoscopic surgery for pheochromocytoma: adrenalectomy, partial resection, excision of paragangliomas. J Urol. 1998;160:330-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 162] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 22. | Neumann HP, Reincke M, Bender BU, Elsner R, Janetschek G. Preserved adrenocortical function after laparoscopic bilateral adrenal sparing surgery for hereditary pheochromocytoma. J Clin Endocrinol Metab. 1999;84:2608-2610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 18] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 23. | Clayton R, Burden AC, Schrieber V, Rosenthal FD. Secondary pituitary hyperplasia in Addison's disease. Lancet. 1977;2:954-956. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 24. | Jara-Albarran A, Bayort J, Caballero A, Portillo J, Laborda L, Sampedro M, Cure C, Mateos JM. Probable pituitary adenoma with adrenocorticotropin hypersecretion (corticotropinoma) secondary to Addison's disease. J Clin Endocrinol Metab. 1979;49:236-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 25. | Krautli B, Müller J, Landolt AM, von Schulthess F. ACTH-producing pituitary adenomas in Addison's disease: two cases treated by transsphenoidal microsurgery. Acta Endocrinol (Copenh). 1982;99:357-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 26. | Scheithauer BW, Kovacs K, Randall RV. The pituitary gland in untreated Addison's disease. A histologic and immunocytologic study of 18 adenohypophyses. Arch Pathol Lab Med. 1983;107:484-487. [PubMed] |
| 27. | Groves RW, Toms GC, Houghton BJ, Monson JP. Corticosteroid replacement therapy: twice or thrice daily? J R Soc Med. 1988;81:514-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 63] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 28. | Zheng KW, Li HZ, Zhang XB. Cushing’s syndrome from bilateral adenocortical adenomas (a report of 4 cases and literature review). Xiandai Miniaowaike Zazhi. 2014;19:513-516. |
| 29. | Oki K, Yamane K, Sakashita Y, Kamei N, Watanabe H, Toyota N, Shigeta M, Sasano H, Kohno N. Primary aldosteronism and hypercortisolism due to bilateral functioning adrenocortical adenomas. Clin Exp Nephrol. 2008;12:382-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 30. | Mao JF, Chai XF, Liu LP, Chen S, Wang ZX, Lu L, and Lu ZL. Desmopressin stimulation test for the differential diagnosis of ACTH dependent Cushing syndrome. Zhongguo Shiyongneike Zazhi. 2014;34:1000-1003. [DOI] [Full Text] |