Published online Apr 6, 2012. doi: 10.4292/wjgpt.v3.i2.7
Revised: September 21, 2011
Accepted: September 28, 2011
Published online: April 6, 2012
Cholesterol gallstone disease is a common clinical condition influenced by genetic factors, increasing age, female gender, and metabolic factors. Although laparoscopic cholecystectomy is currently considered the gold standard in treating patients with symptomatic gallstones, new perspectives regarding medical therapy of cholelithiasis are currently under discussion, also taking into account the pathogenesis of gallstones, the natural history of the disease and the analysis of the overall costs of therapy. A careful selection of patients may lead to successful non-surgical therapy in symptomatic subjects with a functioning gallbladder harboring small radiolucent stones. The classical oral litholysis by ursodeoxycholic acid has been recently paralleled by new experimental observations, suggesting that cholesterol-lowering agents which inhibit cholesterol synthesis (statins) or intestinal cholesterol absorption (ezetimibe), or drugs acting on specific nuclear receptors involved in cholesterol and bile acid homeostasis, might be proposed as additional approaches for treating cholesterol gallstones. In this review we discuss old, recent and future perspectives on medical treatment of cholesterol cholelithiasis.
- Citation: Portincasa P, Ciaula AD, Bonfrate L, Wang DQ. Therapy of gallstone disease: What it was, what it is, what it will be. World J Gastrointest Pharmacol Ther 2012; 3(2): 7-20
- URL: https://www.wjgnet.com/2150-5349/full/v3/i2/7.htm
- DOI: https://dx.doi.org/10.4292/wjgpt.v3.i2.7
The prevalence of gallstones increases with age, and is associated with a number of major risk factors (Table 1)[1-3]. In westernized countries, well known risk factors are: obesity, type 2 diabetes, dyslipidaemia, and hyperinsulinaemia, which are often components of the metabolic syndrome[4-8]. Although the majority of stones in the gallbladder remain “silent” and do not require medical or surgical treatment, gallstone disease is still one of the most common digestive diseases requiring hospital admission and financial resources, since its prevalence ranges from 10% to 15% in adults and medical expenses for gallstone treatment exceeded $6 billion in the year 2000 in the United States[1,9-11].
| Age |
| Female gender |
| High-calorie, low-fiber diet |
| High-carbohydrate diet, dietary glycemic load |
| Obesity |
| Physical inactivity |
| Rapid weight loss/surgery for obesity |
| Total gastrectomy with lymph node dissection |
| Spinal cord injury |
| Infections: enterohepatic Helicobacter species, malaria |
| Biliary strictures |
| Drugs: estrogens, calcineurin inhibitors, fibrates, octreotide, ceftriaxone |
| Total parenteral nutrition |
| Duodenal diverticulum |
| Extended ileal resection (black pigment stones) |
| Vitamin B12/folic acid deficient diet (black pigment stones) |
| Pancreatic insufficiency |
| Cholangitis (brown pigment bile duct stones) |
To know exactly the composition of gallstones is an essential step to select patients responsive to oral litholysis with bile acids (see below). In principle, the only gallstones amenable to litholysis are cholesterol-enriched, calcium-free stones. Cholesterol gallstones represent about 75% of the gallstones in westernized countries[12-14] and can be dissolved when no calcium has deposited in the stones[1,15]. Historically, the Renaissance physician, botanist, alchemist and astrologer Paracelsus (Philippus Aureolus Theophrastus Bombastus von Hohenheim) was the first one to hypothesize that gallbladder concrements were originating from the precipitation of solid material made of tartaric acid[16,17]. To date, we know that specific pathogenetic factors contributing to the formation of cholesterol gallstones must include: hepatic hypersecretion of cholesterol into bile leading to a supersaturated bile, accelerated nucleation/crystallization of cholesterol, defective gallbladder motility (a form of leiomyopathy) leading to gallbladder stasis, increased absorption of intestinal cholesterol, and influence of LITH genes[1,18-24]. The remaining gallstones are pigment stones that contain less than 30% cholesterol, i.e., black pigment stones which are about 20% of all gallstones found in the gallbladder and/or bile duct (containing mainly insoluble bilirubin pigment polymer mixed with calcium phosphate and carbonate, and cholesterol) and brown pigment stones which are about 5% of all gallstones, found in bile ducts (containing calcium bilirubinate, calcium palmitate, stearate and cholesterol)[25].
Patients presenting with a typical colicky pain (“symptomatic”) do need treatment because of the high rates of complications (e.g., acute cholecystitis, acute biliary pancreatitis or cholangitis), and early recurrence of symptoms. The high costs of both surgical and medical therapeutic interventions and the natural history of the disease indicate restricting the treatment to a subgroup of symptomatic patients with specific symptoms[1,23,26].
The first cholecystectomy was performed in 1882 by Carl Langenbuch in Berlin[27,28], which was the first milestone in the treatment of gallstones. Initial experiments on the dissolution of gallstones were already happening at the end of the 19th century[29,30] and in the first half of the 20th century[31]. However, it was Danzinger et al[32] in 1972 who reported that the primary bile acid chenodeoxycholic acid (CDCA) could dissolve cholesterol gallstones in humans when given orally for 6 mo. These days, oral litholysis by ursodeoxycholic acid (UDCA) plays a limited role in cholesterol gallstone treatment. However, some novel and interesting therapeutic options have been suggested by data from pathogenetic and pharmacological studies[1], in particular in subjects permanently or temporarily at risk for gallstone disease (Table 1). Experimental data on the capacity of the Niemann-Pick C1-like 1 (NPC1L1) protein inhibitor ezetimibe to reduce intestinal absorption of cholesterol[33], the effects of statins to inhibit cholesterol synthesis[34], or drugs acting on specific nuclear receptors (NRs) involved in cholesterol and bile acid homeostasis[35] may offer an integrate, potent and innovative strategy for the medical treatment of cholesterol gallstones[36]. Major updated therapeutic aspects in patients with gallstones will be reviewed in this paper.
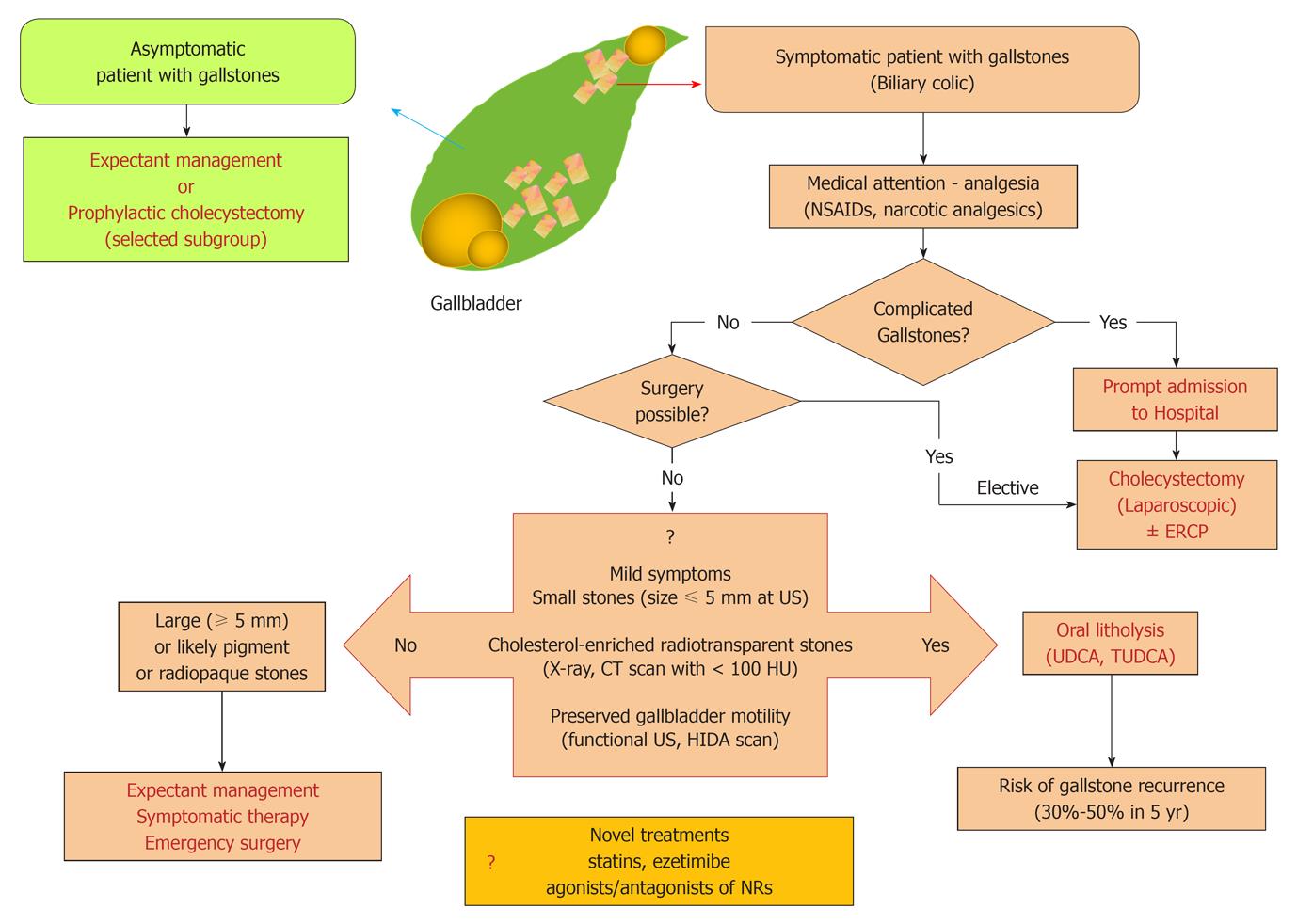
The therapeutic option of gallstone disease is based on few crucial steps, i.e., presence/absence of typical symptoms (i.e., colicky pain), presence of complications, and gallbladder function, as well as composition and size of gallstones (Figure 1).
| Pathogenesis | Visceral pain caused by the impaction of the stone in the cystic duct or the ampulla of Vater, followed by distension of the gallbladder and/or biliary tract with activation of visceral sensory neurons[161] |
| Onset | Not exclusively postprandial, typically intermittent |
| Intensity | Mean visual analogue scale of 9 cm on a 0-10 cm scale |
| Localization | Most frequently right upper quadrant of the abdomen and/or the epigastrium (representative dermatomes T8/9) |
| Duration | Generally longer than 15-30 min. Can last several hours and be associated non-specific symptoms of indigestion |
| Radiation | Angle of the right scapula and/or shoulder (about 60% of cases), retrosternal area (less than 10% of cases) |
| Associated features | Urgency to walk[162] (two-third of patients), nausea or vomit[42,161,162] |
| Relief | If the stone returns into the gallbladder lumen, passes through the ampulla of Vater into the duodenum or migrates back to the common bile duct[26] |
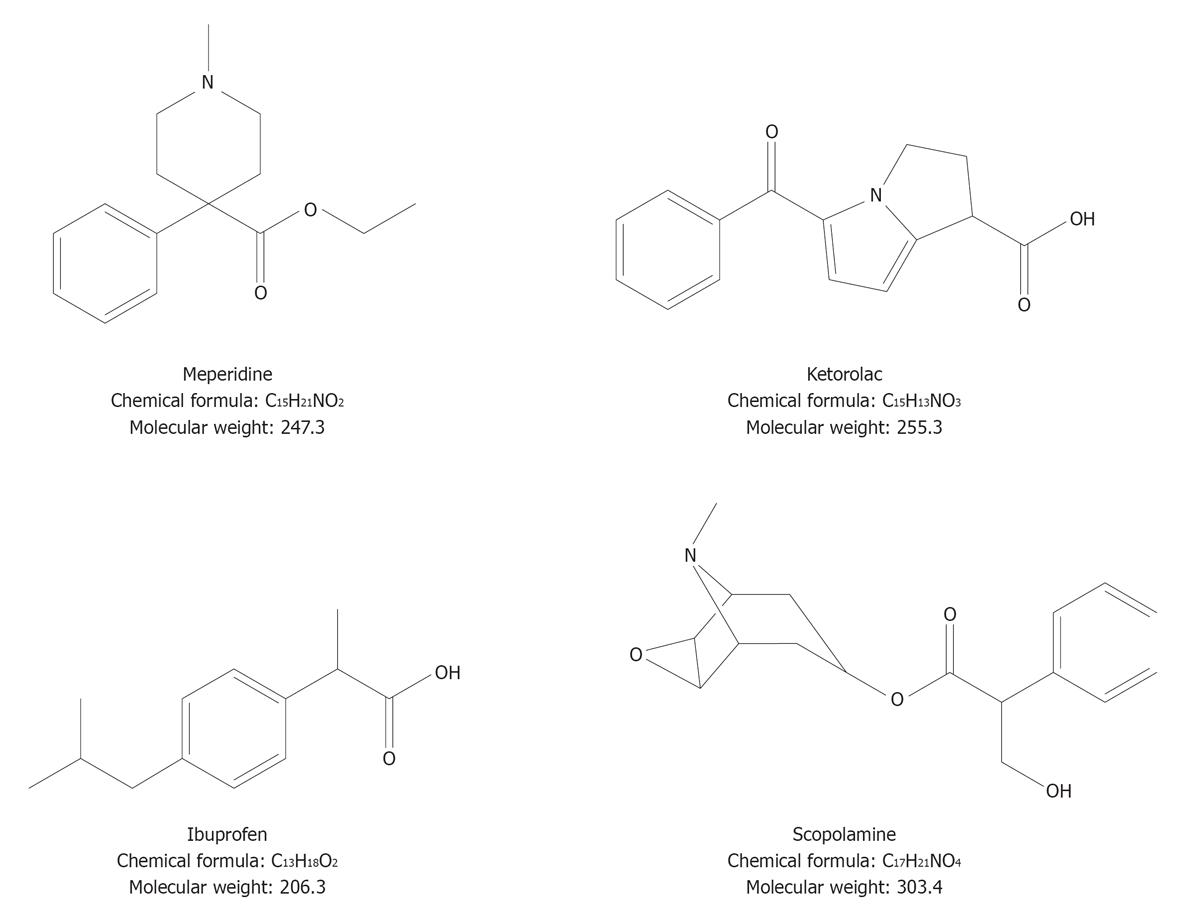
| First-line therapy | Fast-acting narcotic analgesics (meperidine[163]) or non-steroidal anti-inflammatory drugs (NSAIDs) (im or iv ketorolac or ibuprofen po) which could also reduce the risk of evolution towards acute cholecystitis[164-167] |
| Second-line therapy | Antispasmodic (anticholinergic) agents like hyoscine (scopolamine). Less effective than NSAIDs[164] |
| Recommendations | Fasting, to avoid release of endogenous cholecystokinin and further gallbladder contraction |
| Children (because they are exposed to the long-term physical presence of stones[58]) |
| Morbid obese patients undergoing bariatric surgery (high risk to become symptomatic during rapid weight loss[62]) |
| Increased risk for gallbladder cancer[63] |
| Patients with large gallstones (greater than 3 cm)[64,65] |
| A “porcelain” gallbladder[66] or gallbladder polyps rapidly growing or larger than 1 cm |
| Native Americans with gallstones (risk of gallbladder cancer 3 to 5 percent)[67] |
| Gallstone patients with sickle cell anemia (formation of calcium bilirubinate gallstones due to chronic hemolysis. Patients may become symptomatic with recurrent episodes of abdominal pain[68]) |
| Coexistence of small gallstones and gallbladder dysmotility (increased risk of pancreatitis[47]) |
| Additional findings compared to uncomplicated biliary pain | Leukocytosis, nausea, jaundice, vomiting, fever |
| Underlying potential complications | Acute pancreatitis, acute cholecystitis, biliary obstruction and cholangitis, gallbladder perforation, abscess formation, mucocele of the gallbladder |
| Decision | Quick admission to the hospital |
| Therapies | Antibiotics or invasive procedures with or without surgical procedures (Figure 1) |
| Early laparoscopic cholecystectomy recommended between 2 and 4[168] in mild and moderate acute cholecystitis |
Bearing in mind data on epidemiology and overall costs of both medical and surgical therapies, it is not routinely recommended to treat asymptomatic gallstone patients[37-39]. Thus, an expectant management (medical attention) is currently considered the most appropriate choice in patients with gallstones of any type without specific symptoms (i.e., biliary colic). Indeed, approximately 60%-80% of patients with gallstones are completely asymptomatic[40-42] and stones are frequently found during routine abdominal ultrasonography[40-42]. In general, the risk of developing typical biliary pain is low (2.0%-2.6% per year[43-46]) although microlithiasis or biliary sludge in the gallbladder lumen puts patients at risk for colicky pain or acute pancreatitis[47,48]. Nevertheless, the overall risk rate for complications (yearly incidence 0.3%) and gallbladder cancer (0.02%) are very low[49,50]. If biliary pain and/or complications are present, cholecystectomy represents the gold standard (see below), as oral litholysis with hydrophilic bile acids have a limited role, and are reserved to symptomatic patients with small radiolucent gallstones in a well functioning gallbladder with a patent cystic duct[1,23]. Before cholecystectomy, however, careful medical attention and analgesia are often required. Major features of the uncomplicated biliary colic are depicted in Table 2, concerning pathogenesis, onset, intensity, localization, duration, radiation, associated features, relief of pain, and therapeutic aspects. The chemical formula of drugs currently used to induce analgesia in patients with colicky pain is depicted in Figure 2.
Cholecystectomy can be performed by laparoscopy, by a small-incision (< 8 cm in length), or by open operation, and several meta-analyses indicate surgical procedures as the gold standard for the treatment of symptomatic gallstones[51-53]. Laparoscopic cholecystectomy, or alternatively, small incision cholecystectomy[53], are both safe with a similar mortality rate ranging from 0.1% to 0.7%[52,54]. Both these procedures are cost-effective compared with open cholecystectomy[52]. Hospital stay and convalescence are shorter, as is the total cost lower for laparoscopic cholecystectomy compared with open cholecystectomy[54]. The overall incidence of bile duct injuries requiring corrective surgery varies between 0.1% and 0.3%[55-57] and both laparoscopic and open cholecystectomies yield similar complication rates[52,54].
Principally due to the low rate of complications, it is currently under discussion if cholecystectomy may be suggested also for patients with asymptomatic gallstones, but it is generally conceived that surgical procedures are not recommended routinely in symptom-free patients (Figure 1). Few indications for prophylactic cholecystectomy in asymptomatic patients with gallstones are reported in Table 3. For example, cholecystectomy should be considered in children with asymptomatic gallstones[58] (in particular with sickle cell disease[59,60], spherocytosis, and elliptocytosis[60]) who are exposed to the risk of pain and complications. In this group the natural history of gallstones is not well known[61], although a recent study suggests that clinically silent gallstones in children and infants are associated with low rates of complications and can be therefore managed conservatively[60], as in adults. Other groups in which prophylactic cholecystectomy must be considered are the morbidly obese undergoing bariatric surgery, patients at high risk for gallbladder cancer, patients with sickle cell anemia, and coexistence of small gallstones and gallbladder dysmotility[47,62-68]. A totally different approach is necessary in the case of complicated biliary colic, as also shown in Table 4.
There is no established medical therapy for dissolution of pigment stones or calcified stones of any type. For cholesterol gallstones, current medical treatment includes oral litholysis with bile acids (see below). Medical therapies alternative to oral bile acids have been proposed in the past, including direct stone dissolution with methyl tert-butyl ether (MTBE), a potent organic cholesterol solvent[69], extracorporeal shock-wave lithotripsy (ESWL)[70], or in combination[71], followed by oral litholysis with bile acids. The interest in such options, however, has vanished due to their invasiveness, potential toxicity (MTBE) or traumatic (ESWL) side effects and, for both, the high post-dissolution recurrence rate[1,72,73]. Novel treatments to be discussed include statins, ezetimibe, and agonists/antagonists of NRs.
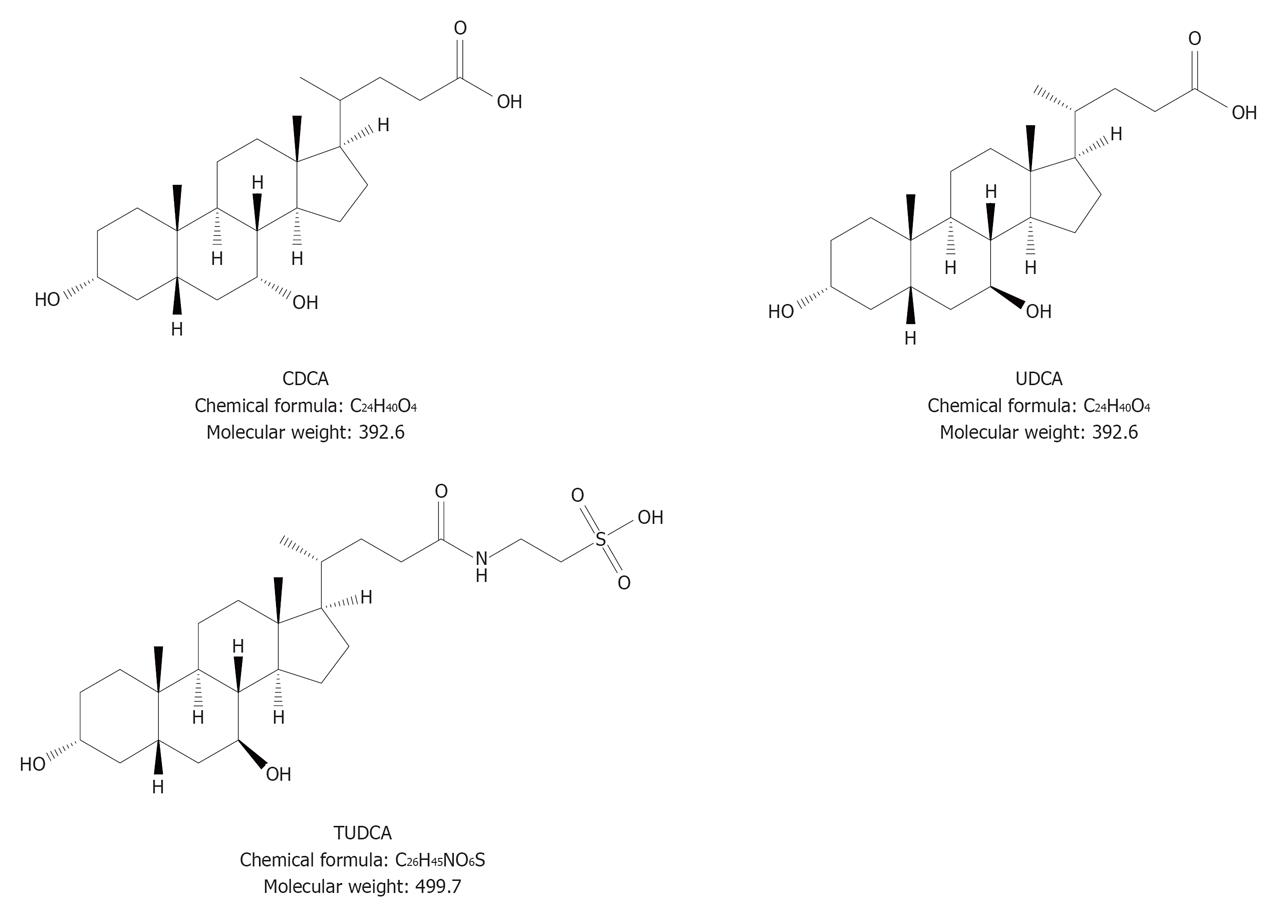
The first successful dissolution of cholesterol gallstones was achieved in 1972 by oral administration of the natural primary tri-hydroxy bile acid CDCA[32] (Figure 3). The use of CDCA was abandoned because side effects were noticed, including a dose-dependent increase in serum liver enzymes, an increase in serum low-density lipoprotein (LDL) cholesterol, and diarrhea.
A further step was to use the more hydrophilic tri-hydroxy bile acid UDCA[74]. UDCA is more hydrophilic and less toxic than CDCA, and is currently employed for oral litholysis of small cholesterol gallstones in patients with a functioning gallbladder (Figure 3). This bile acid, in a dose of 10-14 mg/kg per day, increases its proportion in the bile acid pool (it originally accounts for less than 8%-10% of the biliary bile acid pool in healthy subjects), inducing a decreased hepatic secretion of biliary cholesterol and the formation of unsaturated gallbladder bile (cholesterol saturation index of less than 1)[75-77], the key factor which promotes the dissolution of cholesterol crystals and gallstones.
The fine mechanisms involved in UDCA-induced dissolution of cholesterol stones are rather complex. The so-called ternary phase diagram is used to explain the molecular effects of UDCA on bile composition and cholesterol solubility[78]. A group of the equilibrium phase diagram of cholesterol-lecithin-taurine-conjugated bile acid systems (37 °C, 0.15 M NaCl, pH 7.0, total lipid concentration 7.5 g/dL) are drawn to display varied positions and configuration of crystallization regions due to decreasing bile acid hydrophobicity, with the lipid components being expressed in moles percent. At the bottom, the one-phase micellar zone exists (i.e., high bile acid-lecithin moles percent), while above this zone two-phase zones exist on both sides from a central three-phase zone. The study of solid and liquid crystallization sequences present in bile shows that different regions exist within each zone, namely A, B in the left two-phase, C, D in the central three-phase regions, and E in the right 2-phase zone. The number of phases given represents the equilibrium state and develop as cholesterol monohydrate crystals and saturated micelles for crystallization regions A and B; cholesterol monohydrate crystals, saturated micelles and liquid crystals for regions C and D; and liquid crystals of variable compositions and saturated micelles for region E[78]. As the bile acid hydrophobicity decreases, the maximum micellar cholesterol solubility is reduced and crystallization pathways A-E move to the left. This change results in an enlarged region E that extends to the left and overlaps pathophysiological compositions as exemplified in the tauroursodeoxycholate (TUDC)-lecithin-cholesterol system. This event induces a greatly reduced chance for the formation of solid plate-like cholesterol monohydrate crystals in bile.
A bedtime administration of UDCA or TUDCA, is recommended since it maintains hepatic bile acid secretion rate overnight, thus reducing secretion of supersaturated bile and increasing the dissolution rate[79,80]. The hydrophilic bile acid UDCA is also able to act as a litholytic agent through the reduction of intestinal cholesterol absorption[81-83] and as a possible “prokinetic” agent capable of ameliorating postprandial gallbladder emptying as suggested by observations in vitro on isolated gallbladder smooth muscle strips from both animals and gallstone patients[84,85]. The improvement of gallbladder smooth muscle contractility probably also results from the prevention of the impairment of smooth muscle contractility induced by the more hydrophobic and toxic deoxycholate[86,87].
However, although the majority of gallstones (about two-thirds) in westernized countries are mainly composed of cholesterol, only a minority of patients (less than 10% of total) with cholesterol-enriched gallstones is amenable to oral dissolution therapy with UDCA or with its taurine-conjugates TUDCA[1,26]. In fact, dissolution therapy with oral bile acids can be only suggested to symptomatic gallstone patients who are unfit for surgery and have small (equal to or less than 5 mm in size), uncalcified (radiolucent), and cholesterol-enriched (i.e., more than 80%) stones in a functioning gallbladder with a patent cystic duct[88]. A number of diagnostic techniques provide essential information for appropriate selection of patients.
Gallbladder ultrasonography allows the accurate visualization of gallstone number, size, burden, biliary sludge[39,89,90] and explores the morphology and contractile property of the gallbladder, the features of the gallbladder wall with respect to the (acute-chronic) inflammatory status, and the patency of the cystic duct[91-97]. An abdominal plain radiography or a computed tomography (CT) scan[98,99] are needed to exclude the presence of calcified stones[25]. By CT scan, in particular, values of < 100 Hounsfield Units predict radiolucent cholesterol rich, dissolvable stones[100] (see also Figure 1 for explanation).
An accurate selection of gallstone patients with the characteristics described above offers a higher chance of successful oral litholysis alone or after ESWL inducing stone fragmentation[93-96,101], with an expected dissolution rate of about 1 mm decrement in stone diameter per month[102].
The complete disappearance of stones with a diameter of less than 5 mm has been described after 6 mo of UDCA administration in about 90% of cases[103]. The chance of dissolution is significantly lower (less than 40%-50% after 1 year of the treatment) in patients with larger or multiple stones[49,104].
Main limits of the dissolution therapy by oral bile acids are the possibility of gallstone recurrence (about 10% per year up to 5 years[105,106]) and the risk of appearance of a surface calcification on cholesterol gallstones during bile acid therapy in about 10% of cases[107]. A recurrence rate of 30%-50% at 5 years is seen after bile acid therapy or lithotripsy[94,108-110], particularly in patients with multiple gallstones[109]. After gallstone disappearance, the persistence of the same pathogenetic factors inducing gallstone formation is principally responsible for their recurrence[1]. It has to be underlined, however, that recurrent gallstones respond well to a re-treatment[99,111].
Although limited to a relatively small subgroup of patients, the dissolution therapy with UDCA or TUDCA still remains at present an interesting tool in patients who form gallstones as a consequence of transient and non-genetic risk factors (i.e., pregnancy, convalescence from abdominal surgery, obese patients during rapid weight loss[1,112-114], Table 1) and, thus, have a minimum risk of recurrence. Early non-randomized or placebo-controlled studies[115-117] suggested that UDCA might also reduce the risk of biliary colic. A large randomized, double-blind, placebo-controlled trial on the effects of UDCA in highly symptomatic gallstone patients scheduled for cholecystectomy, however, found that UDCA was ineffective on biliary colic. In fact, the likelihood of remaining colic-free is comparable in patients with strong or weak baseline gallbladder contraction as determined by ultrasonography after a standard mixed meal[118].
Bile supersaturation with cholesterol is a key factor for cholesterol gallstone formation, and it is principally related to a sustained hepatic hypersecretion of cholesterol depending on the source; from hepatic cholesterol biosynthesis, intestinal cholesterol absorption and HDL-derived cholesterol[18]. As a consequence, all drugs targeting these steps are potentially able to influence both cholesterol gallstone formation and dissolution. Statins and ezetimibe have interesting effects.
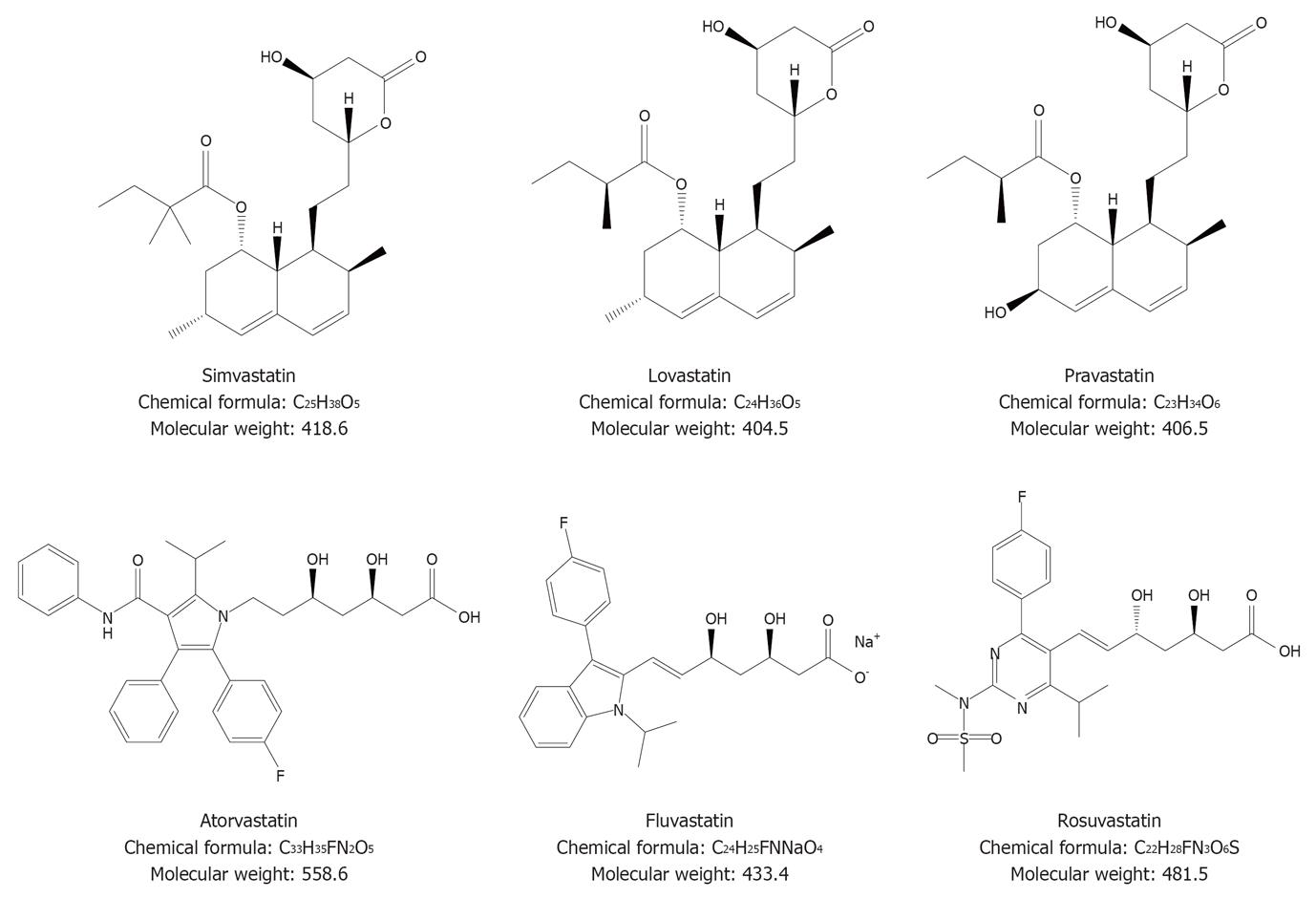
Statins are competitive inhibitors of 3-hydroxy-3-methyl-glutaryl-CoA (HMG-CoA) reductase, a rate-limiting enzyme for cholesterol biosynthesis, and they are able to reduce biliary cholesterol independently of their ability to suppress hepatic cholesterol synthesis[119-122]. Several statins are being used (Figure 4) and their pharmacological properties modulate cholesterol homeostasis both in bile and in the liver, potentially leading to a reduction of cholesterol gallstone formation[123-125], as clearly demonstrated by animal studies[126,127]. In humans, by contrast, the potential beneficial effects of statins on cholesterol gallstones are not so clear.
The risk of cholecystectomy decreased slightly in a cohort of US women self-reporting long-term use of statins[128]. Similar results were suggested by a case-control analysis using the UK-based General Practice Research Database and evaluating incident patients between 1994 and 2004. In this study the long-term use of statins (1 to 1.5 years) was associated with a decreased risk of gallstones followed by cholecystectomy, compared with patients without statin use[129]. Furthermore, a recent population-based control study using medical databases from northern Denmark showed a decreased odds ratio for gallstone disease in current statin users (1-2 years of statin use), as compared with nonusers[130]. However, experimental studies show controversial results, since a decreased biliary cholesterol concentration, a reduced gallstone formation, or gallstone dissolution has been found by some[131-134] but not all studies[124,135-138].
Recent advances underscore the role of intestinal factors as a key factor for cholesterol absorption, biliary secretion and cholesterol gallstones[1,139]. In fact, it has been found experimentally that if dietary cholesterol is absent, all biliary cholesterol derives mainly from a limited de novo synthesis (less than 15%). Thus, the small intestine must be seen as a unique organ providing dietary and re-absorbed biliary cholesterol to the body[139]. This step plays a crucial role in cholesterol gallstone pathogenesis, since animal studies demonstrate that there is a significant positive correlation between the efficiency of intestinal cholesterol absorption and the prevalence of cholesterol gallstone formation[21].
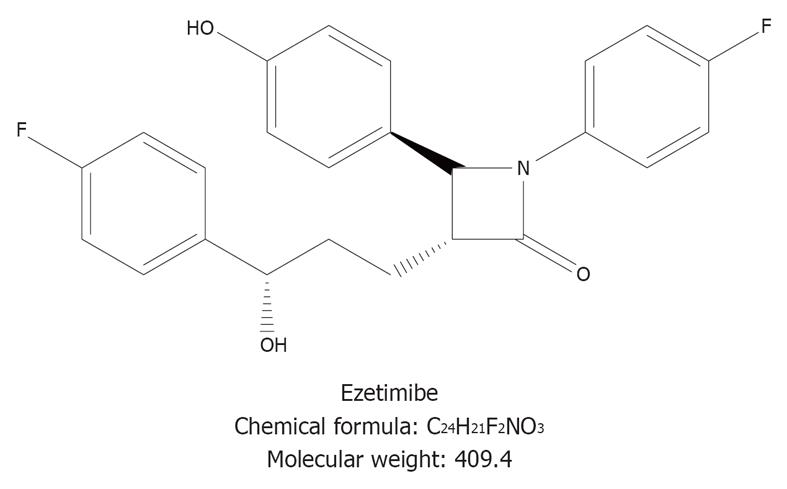
Ezetimibe, in this respect, is an interesting drug since it has novel hypocholesterolemic effect[140] (Figure 5). Ezetimibe has a strong inhibitory effect on intestinal cholesterol absorption; cholesterol is indeed the most effective substrate of the NPC1L1 protein, the protein that governs intestinal absorption of cholesterol by recycling between the endocytic recycling compartment and plasma membrane[141]. NPC1L1 is highly expressed in the small intestine and localized along the brush border in both humans and mice[142,143], but also present in the human liver[143,144]. In mice, ezetimibe largely reduces cholesterol, and to some extent phospholipid content, but not the bile acid content in gallbladder bile. However, all crystallization pathways and phase boundaries on the bile phase diagram are essentially similar, regardless of whether animals are treated with or without ezetimibe[36]. By inhibiting both the cholesterol absorption in the intestine and the hepatic uptake of chylomicron remnants, ezetimibe might lower biliary cholesterol secretion and saturation[145]. Furthermore, it has been also demonstrated that increasing doses of ezetimibe lead the relative lipid composition of gallbladder bile to a progressive shift down and to the left of the phase diagram, which goes into the one-phase (protective) micellar zone, with an abundance of unsaturated micelles but never solid cholesterol crystals or liquid crystals. As a consequence, in gallbladder bile the micellar cholesterol solubility is increased, with more cholesterol molecules transferred from the cholesterol monohydrate surface into unsaturated micelles. In this environment, gallstones are reduced in size and can be completely dissolved[36,146]. Ezetimibe might therefore act as a new tool in treating/preventing cholesterol gallstones[147] but also induce amelioration of gallbladder motility, as a consequence of bile desaturation[36]. Ezetimibe is also effective in humans, since it has been demonstrated in a Mexican population that this drug in a dosage of 20 mg po/d for 1 mo, is able to significantly reduce cholesterol saturation and cholesterol saturation index and to retard cholesterol crystallization in gallstone patients[36].
In the near future, well designed experimental studies might confirm the efficacy of statins and ezetimibe, alone and/or in association with hydrophilic bile acids, in symptomatic patients without genetic risk of gallstone formation but in the presence of several predisposing conditions (Table 1). Obesity, in particular, is associated with an increased cholesterol biosynthesis in the liver, mostly due to higher levels of HMG-CoA reductase activity. Thus, in obese patients, the administration of statin might be potentially useful to prevent gallstone formation[148]. It may be also useful in patients with rapid weight loss, a condition characterized by an increased hepatic secretion of biliary cholesterol, an increase in mucin production by the gallbladder epithelium, and a significant impairment of gallbladder motility[149].
Multiple physiological, developmental, and toxicological processes in the body are regulated by sets of genes, which are coordinated and activated by ligand-activated transcription factors, the NRs[150]. Lipid sensing NRs drive lipid homeostasis in the hepatobiliary and gastrointestinal systems. A key function is exerted by the oxysterol receptor liver X receptor (LXR) and by the bile acid receptor farnesoid X receptor (FXR); both are involved in the molecular regulation of hepatic and biliary lipid metabolism, and modulate bile flow and cholesterol gallstone formation. LXR acts as the intracellular “sensor” of cholesterol[151], while FXR is the intracellular sensor of bile acids[152,153]. To maintain lipid homeostasis, cells synthesize oxysterols under conditions of cholesterol overload, and oxysterols, in turn, bind and activate LXR, which acts to reduce the systemic cholesterol burden[154]. In the enterohepatic system, FXR highly determines expression levels of genes involved in the maintenance of cholesterol, bile acid and triglyceride homeostasis[155].
FXR also up-regulates hepatic expression of bile acid and lipid transporters on the canalicular membrane of hepatocytes and increases activity of regulatory enzymes responsible for bile acid detoxification. These biochemical properties characterize FXR as a potential suitable target for drugs to be employed in the treatment of both cholestasis and cholelithiasis[156]. Animal studies confirmed a direct role of LXR and FXR in the processes leading to cholesterol precipitation in bile. FXR-null mice are prone to cholesterol gallstone formation, while the activation of FXR via specific synthetic ligands such as GW4064 restores a normal homeostasis between cholesterol, bile acids and phospholipids in bile[157]. This mechanism depends on FXR-induced activity of the energy-dependent ATP-Binding Cassette (ABC) transporters ABCB11 for bile acids and ABCB4 for phospholipid[158] and it is linked to a better cholesterol solubilization in bile, thus preventing the formation of cholesterol crystals and gallstones. The activation of FXR promotes an increase in cholesterol secretion by a direct up-regulation of the main hepatocyte canalicular transporters (ABCG5 and ABCG8) leading to increased biliary cholesterol saturation and precipitation of cholesterol crystals, gallstone formation and growth[159]. Such innovative and intriguing results from animal studies have not been confirmed in humans, so far. Future studies are required to assess the usefulness and safety of synthetic, liver-specific FXR agonists and LXR antagonists in humans, not only targeting gallstone disease but also type II diabetes, dyslipidaemia and several cancers[35].
The gold standard for treating symptomatic gallstones remains laparoscopic cholecystectomy. Oral litholysis (basically restricted to few oral hydrophilic bile acids) has a limited role in a scant subgroup of selected patients with symptomatic cholesterol gallstones, but is complicated by the high rate of gallstone recurrence after dissolution treatment and a negative cost-benefit balance. As a consequence of novel and recent animal and human studies, the research agenda in the field of non-surgical therapy of cholesterol cholelithiasis is filled with several possibilities. Drugs affecting cholesterol synthesis and intestinal absorption (i.e., statins, ezetimibe) and agonists/antagonists of the NRs FXR/LXR involved in biliary lipid secretion may offer, in the near future, promising agents to treat cholesterol gallstones or to prevent their formation in populations at risk.
The authors are indebted Paola De Benedictis, Rosa De Venuto, Michele Persichella and Ornella de Bari, for their skillful technical assistance.
Peer reviewer: Rakesh K Tandon, Professor, Head, Department of Gastroenterology, Pushpawati Singhania Research Institute for Liver, Renal and Digestive Diseases, Sheikh Sarai – Phase II, New Delhi 110017, India
S- Editor Wang JL L- Editor Hughes D E- Editor Zheng XM
| 1. | Portincasa P, Moschetta A, Palasciano G. Cholesterol gallstone disease. Lancet. 2006;368:230-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 554] [Cited by in RCA: 489] [Article Influence: 25.7] [Reference Citation Analysis (2)] |
| 2. | Katsika D, Grjibovski A, Einarsson C, Lammert F, Lichtenstein P, Marschall HU. Genetic and environmental influences on symptomatic gallstone disease: a Swedish study of 43,141 twin pairs. Hepatology. 2005;41:1138-1143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 175] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 3. | Kratzer W, Mason RA, Kächele V. Prevalence of gallstones in sonographic surveys worldwide. J Clin Ultrasound. 1999;27:1-7. [PubMed] [DOI] [Full Text] |
| 4. | Grundy SM, Barnett JP. Metabolic and health complications of obesity. Dis Mon. 1990;36:641-731. [PubMed] |
| 5. | Grundy SM. Metabolic syndrome scientific statement by the American Heart Association and the National Heart, Lung, and Blood Institute. Arterioscler Thromb Vasc Biol. 2005;25:2243-2244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 158] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 6. | Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin BA, Gordon DJ, Krauss RM, Savage PJ, Smith SC. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation. 2005;112:2735-2752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7515] [Cited by in RCA: 8282] [Article Influence: 414.1] [Reference Citation Analysis (0)] |
| 7. | Eckel RH, Grundy SM, Zimmet PZ. The metabolic syndrome. Lancet. 2005;365:1415-1428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4273] [Cited by in RCA: 4495] [Article Influence: 224.8] [Reference Citation Analysis (0)] |
| 8. | Tsai CJ, Leitzmann MF, Willett WC, Giovannucci EL. Prospective study of abdominal adiposity and gallstone disease in US men. Am J Clin Nutr. 2004;80:38-44. [PubMed] |
| 9. | Wang DQ, Afdhal NH. Genetic analysis of cholesterol gallstone formation: searching for Lith (gallstone) genes. Curr Gastroenterol Rep. 2004;6:140-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 52] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 10. | Everhart JE, Khare M, Hill M, Maurer KR. Prevalence and ethnic differences in gallbladder disease in the United States. Gastroenterology. 1999;117:632-639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 549] [Cited by in RCA: 536] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 11. | Sandler RS, Everhart JE, Donowitz M, Adams E, Cronin K, Goodman C, Gemmen E, Shah S, Avdic A, Rubin R. The burden of selected digestive diseases in the United States. Gastroenterology. 2002;122:1500-1511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1062] [Cited by in RCA: 1037] [Article Influence: 45.1] [Reference Citation Analysis (0)] |
| 12. | Diehl AK. Epidemiology and natural history of gallstone disease. Gastroenterol Clin North Am. 1991;20:1-19. [PubMed] |
| 13. | Attili AF, Carulli N, Roda E, Barbara B, Capocaccia L, Menotti A, Okoliksanyi L, Ricci G, Capocaccia R, Festi D. Epidemiology of gallstone disease in Italy: prevalence data of the Multicenter Italian Study on Cholelithiasis (M.I.COL.). Am J Epidemiol. 1995;141:158-165. [PubMed] |
| 14. | Attili AF, Capocaccia R, Carulli N, Festi D, Roda E, Barbara L, Capocaccia L, Menotti A, Okolicsanyi L, Ricci G. Factors associated with gallstone disease in the MICOL experience. Multicenter Italian Study on Epidemiology of Cholelithiasis. Hepatology. 1997;26:809-818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 139] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 15. | Portincasa P, Di Ciaula A, Wang HH, Moschetta A, Wang DQ. Medicinal treatments of cholesterol gallstones: old, current and new perspectives. Curr Med Chem. 2009;16:1531-1542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 22] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 16. | Theophrastus Bombastus von Hohenheim; Das Buch von den tartarischen Krankheiten nach dem alten Namen vom Stein/sand und Grieβ. In: Brisgoius IH, editor. Ander Theil der Bucher und Schrifften des Philippi Theophrasti Bombast von Hohenheim/Paracelsi. Basel: Conrad Waldkirk, 1589: 246-250. . |
| 17. | Portincasa P. Gallbladder and bile in health and gallstone disease: The role of motility, gallstones and bile lipid composition. Utrecht: PhD Thesis Utrecht University 1995; . |
| 18. | Wang HH, Portincasa P, Wang DQ. Molecular pathophysiology and physical chemistry of cholesterol gallstones. Front Biosci. 2008;13:401-423. [PubMed] |
| 19. | Portincasa P, Di Ciaula A, Wang HH, Palasciano G, van Erpecum KJ, Moschetta A, Wang DQ. Coordinate regulation of gallbladder motor function in the gut-liver axis. Hepatology. 2008;47:2112-2126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 91] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 20. | Wittenburg H, Lammert F. Genetic predisposition to gallbladder stones. Semin Liver Dis. 2007;27:109-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 39] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 21. | Wang DQ, Zhang L, Wang HH. High cholesterol absorption efficiency and rapid biliary secretion of chylomicron remnant cholesterol enhance cholelithogenesis in gallstone-susceptible mice. Biochim Biophys Acta. 2005;1733:90-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 44] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 22. | Lammert F, Sauerbruch T. Mechanisms of disease: the genetic epidemiology of gallbladder stones. Nat Clin Pract Gastroenterol Hepatol. 2005;2:423-433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 94] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 23. | Portincasa P, Moschetta A, Puglisi F, Wang DQH. Medical treatment of gallstone disease. Biliary lithiasis. Basic Science, Current Diagnosis and Management. Milano: Springer Italia S.r.l 2008; 149-157. |
| 24. | Lammert F, Miquel JF. Gallstone disease: from genes to evidence-based therapy. J Hepatol. 2008;48 Suppl 1:S124-S135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 70] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 25. | Sherlock S, Dooley J. Diseases of the liver and biliary system. Oxford: Blackwell Science 2002; . |
| 26. | Portincasa P, Moschetta A, Petruzzelli M, Palasciano G, Di Ciaula A, Pezzolla A. Gallstone disease: Symptoms and diagnosis of gallbladder stones. Best Pract Res Clin Gastroenterol. 2006;20:1017-1029. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 69] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 27. | Ammon HV, Hofmann AF. The Langenbuch paper. I. An historical perspective and comments of the translators. Gastroenterology. 1983;85:1426-1433. [PubMed] |
| 28. | Langenbuch C. Ein Fall von Extirpation der Gallenblase wegen chronischer. Cholelithiasis: Heilung. Berlin Klin Wochenschr. 1882;19:725-727. |
| 29. |
Schiff M; Il coleinato di soda nella cura dei calcoli biliari. |
| 30. |
Dabney WM; The use of choleate of soda to prevent the formation of gall-stones. |
| 31. | Rewbridge AG. The disappearance of gallstone shadows following the prolonged admnistration of bile acids. Surgery. Oxford: Blackwell Science 1937; 395-400. |
| 32. | Danzinger RG, Hofmann AF, Schoenfield LJ, Thistle JL. Dissolution of cholesterol gallstones by chenodeoxycholic acid. N Engl J Med. 1972;286:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 512] [Cited by in RCA: 405] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 33. | Hou R, Goldberg AC. Lowering low-density lipoprotein cholesterol: statins, ezetimibe, bile acid sequestrants, and combinations: comparative efficacy and safety. Endocrinol Metab Clin North Am. 2009;38:79-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 71] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 34. | Istvan ES, Deisenhofer J. Structural mechanism for statin inhibition of HMG-CoA reductase. Science. 2001;292:1160-1164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 980] [Cited by in RCA: 1099] [Article Influence: 45.8] [Reference Citation Analysis (0)] |
| 35. | Zhu Y, Li F, Guo GL. Tissue-specific function of farnesoid X receptor in liver and intestine. Pharmacol Res. 2011;63:259-265. [PubMed] |
| 36. | Wang HH, Portincasa P, Mendez-Sanchez N, Uribe M, Wang DQ. Effect of ezetimibe on the prevention and dissolution of cholesterol gallstones. Gastroenterology. 2008;134:2101-2110. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 123] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 38. | Hofmann AF, Amelsberg A, vanSonnenberg E. Pathogenesis and treatment of gallstones. N Engl J Med. 1993;328:1854-1855. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 39. | Johnston DE, Kaplan MM. Pathogenesis and treatment of gallstones. N Engl J Med. 1993;328:412-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 125] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 40. | Gibney EJ. Asymptomatic gallstones. Br J Surg. 1990;77:368-372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 79] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 41. | Jørgensen T. Abdominal symptoms and gallstone disease: an epidemiological investigation. Hepatology. 1989;9:856-860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 84] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 42. | Festi D, Sottili S, Colecchia A, Attili A, Mazzella G, Roda E, Romano F. Clinical manifestations of gallstone disease: evidence from the multicenter Italian study on cholelithiasis (MICOL). Hepatology. 1999;30:839-846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 40] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 43. | Thistle JL, Cleary PA, Lachin JM, Tyor MP, Hersh T. The natural history of cholelithiasis: the National Cooperative Gallstone Study. Ann Intern Med. 1984;101:171-175. [PubMed] |
| 44. | Friedman GD, Raviola CA, Fireman B. Prognosis of gallstones with mild or no symptoms: 25 years of follow-up in a health maintenance organization. J Clin Epidemiol. 1989;42:127-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 134] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 45. | Gracie WA, Ransohoff DF. The natural history of silent gallstones: the innocent gallstone is not a myth. N Engl J Med. 1982;307:798-800. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 430] [Cited by in RCA: 339] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 46. | Friedman GD. Natural history of asymptomatic and symptomatic gallstones. Am J Surg. 1993;165:399-404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 250] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 47. | Venneman NG, Renooij W, Rehfeld JF, VanBerge-Henegouwen GP, Go PM, Broeders IA, van Erpecum KJ. Small gallstones, preserved gallbladder motility, and fast crystallization are associated with pancreatitis. Hepatology. 2005;41:738-746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 58] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 48. | Venneman NG, Buskens E, Besselink MG, Stads S, Go PM, Bosscha K, van Berge-Henegouwen GP, van Erpecum KJ. Small gallstones are associated with increased risk of acute pancreatitis: potential benefits of prophylactic cholecystectomy. Am J Gastroenterol. 2005;100:2540-2550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 78] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 49. | Paumgartner G, Carr-Locke DL, Dubois F, Roda E, Thistle JL. Strategies in the treatment of gallstone disease. Working team report. Gastroenterol Int. 1993;6:65-75. |
| 50. | Brugge WR. The silent gallstone. Gallbladder and biliary tract diseases. New York, NY: Marcel Dekker Inc 2000; 447-453. |
| 51. | Keus F, de Jong JA, Gooszen HG, van Laarhoven CJ. Small-incision versus open cholecystectomy for patients with symptomatic cholecystolithiasis. Cochrane Database Syst Rev. 2006;CD004788. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 52. | Keus F, de Jong JA, Gooszen HG, van Laarhoven CJ. Laparoscopic versus open cholecystectomy for patients with symptomatic cholecystolithiasis. Cochrane Database Syst Rev. 2006;CD006231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 252] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 53. | Keus F, de Jong JA, Gooszen HG, van Laarhoven CJ. Laparoscopic versus small-incision cholecystectomy for patients with symptomatic cholecystolithiasis. Cochrane Database Syst Rev. 2006;CD006229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 32] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 54. | Lammert F, Neubrand MW, Bittner R, Feussner H, Greiner L, Hagenmüller F, Kiehne KH, Ludwig K, Neuhaus H, Paumgartner G. [S3-guidelines for diagnosis and treatment of gallstones. German Society for Digestive and Metabolic Diseases and German Society for Surgery of the Alimentary Tract]. Z Gastroenterol. 2007;45:971-1001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 56] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 55. | Giger UF, Michel JM, Opitz I, Th Inderbitzin D, Kocher T, Krähenbühl L. Risk factors for perioperative complications in patients undergoing laparoscopic cholecystectomy: analysis of 22,953 consecutive cases from the Swiss Association of Laparoscopic and Thoracoscopic Surgery database. J Am Coll Surg. 2006;203:723-728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 230] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 56. | Tuveri M, Tuveri A. Laparoscopic cholecystectomy: complications and conversions with the 3-trocar technique: a 10-year review. Surg Laparosc Endosc Percutan Tech. 2007;17:380-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 29] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 57. | Duca S, Bãlã O, Al-Hajjar N, Lancu C, Puia IC, Munteanu D, Graur F. Laparoscopic cholecystectomy: incidents and complications. A retrospective analysis of 9542 consecutive laparoscopic operations. HPB (Oxford). 2003;5:152-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 79] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 58. | Pokorny WJ, Saleem M, O'Gorman RB, McGill CW, Harberg FJ. Cholelithiasis and cholecystitis in childhood. Am J Surg. 1984;148:742-744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 28] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 59. | Gumiero AP, Bellomo-Brandão MA, Costa-Pinto EA. Gallstones in children with sickle cell disease followed up at a Brazilian hematology center. Arq Gastroenterol. 2008;45:313-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 33] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 60. | Bogue CO, Murphy AJ, Gerstle JT, Moineddin R, Daneman A. Risk factors, complications, and outcomes of gallstones in children: a single-center review. J Pediatr Gastroenterol Nutr. 2010;50:303-308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 63] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 61. | Chan S, Currie J, Malik AI, Mahomed AA. Paediatric cholecystectomy: Shifting goalposts in the laparoscopic era. Surg Endosc. 2008;22:1392-1395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 62. | Amaral JF, Thompson WR. Gallbladder disease in the morbidly obese. Am J Surg. 1985;149:551-557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 124] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 63. | Sleisenger MH, Fordtran JS. Gastrointestinal disease: pathophysiology, diagnosis, management. 8th ed. Philadelphia: W.B. Saunders 2006; . |
| 64. | Lowenfels AB, Walker AM, Althaus DP, Townsend G, Domellöf L. Gallstone growth, size, and risk of gallbladder cancer: an interracial study. Int J Epidemiol. 1989;18:50-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 98] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 65. | Randi G, Franceschi S, La Vecchia C. Gallbladder cancer worldwide: geographical distribution and risk factors. Int J Cancer. 2006;118:1591-1602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 523] [Cited by in RCA: 573] [Article Influence: 30.2] [Reference Citation Analysis (0)] |
| 66. | Ashur H, Siegal B, Oland Y, Adam YG. Calcified ballbladder (porcelain gallbladder). Arch Surg. 1978;113:594-596. [PubMed] |
| 67. | Lowenfels AB, Lindström CG, Conway MJ, Hastings PR. Gallstones and risk of gallbladder cancer. J Natl Cancer Inst. 1985;75:77-80. [PubMed] |
| 68. | Bonatsos G, Birbas K, Toutouzas K, Durakis N. Laparoscopic cholecystectomy in adults with sickle cell disease. Surg Endosc. 2001;15:816-819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 23] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 69. | Thistle JL, May GR, Bender CE, Williams HJ, LeRoy AJ, Nelson PE, Peine CJ, Petersen BT, McCullough JE. Dissolution of cholesterol gallbladder stones by methyl tert-butyl ether administered by percutaneous transhepatic catheter. N Engl J Med. 1989;320:633-639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 147] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 70. | Sauerbruch T, Delius M, Paumgartner G, Holl J, Wess O, Weber W, Hepp W, Brendel W. Fragmentation of gallstones by extracorporeal shock waves. N Engl J Med. 1986;314:818-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 440] [Cited by in RCA: 344] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 71. | Vergunst H, Brakel K, Nijs HG, Laméris JS, Ten Kate FJ, Schröder FH, Terpstra OT. Methyl tert-butyl ether improves the efficacy of extracorporeal shock wave lithotripsy of human gallstones implanted in pigs. Eur J Surg. 1994;160:619-625. [PubMed] |
| 72. | Sackmann M, Niller H, Klueppelberg U, von Ritter C, Pauletzki J, Holl J, Berr F, Neubrand M, Sauerbruch T, Paumgartner G. Gallstone recurrence after shock-wave therapy. Gastroenterology. 1994;106:225-230. [PubMed] |
| 73. | Pauletzki J, Holl J, Sackmann M, Neubrand M, Klueppelberg U, Sauerbruch T, Paumgartner G. Gallstone recurrence after direct contact dissolution with methyl tert-butyl ether. Dig Dis Sci. 1995;40:1775-1781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 74. | Makino I, Shinozaki K, Yoshino K, Nakagawa S. [Dissolution of cholesterol gallstones by long-term administration of ursodeoxycholic acid]. Nihon Shokakibyo Gakkai Zasshi. 1975;72:690-702. [PubMed] |
| 75. | van Erpecum KJ, Portincasa P, Stolk MF, van de Heijning BJ, van der Zaag ES, van den Broek AM, van Berge Henegouwen GP, Renooij W. Effects of bile salt and phospholipid hydrophobicity on lithogenicity of human gallbladder bile. Eur J Clin Invest. 1994;24:744-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 76. | Van Erpecum KJ, Portincasa P, Gadellaa M, Van de Heijning BJ, Van Berge Henegouwen GP, Renooij W. Effects of bile salt hydrophobicity on crystallization of cholesterol in model bile. Eur J Clin Invest. 1996;26:602-608. [PubMed] |
| 77. | Portincasa P, van Erpecum KJ, Jansen A, Renooij W, Gadellaa M, vanBerge-Henegouwen GP. Behavior of various cholesterol crystals in bile from patients with gallstones. Hepatology. 1996;23:738-748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 29] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 78. | Wang DQ, Carey MC. Complete mapping of crystallization pathways during cholesterol precipitation from model bile: influence of physical-chemical variables of pathophysiologic relevance and identification of a stable liquid crystalline state in cold, dilute and hydrophilic bile salt-containing systems. J Lipid Res. 1996;37:606-630. [PubMed] |
| 79. | Kupfer RM, Maudgal DP, Northfield TC. Gallstone dissolution rate during chenic acid therapy. Effect of bedtime administration plus low cholesterol diet. Dig Dis Sci. 1982;27:1025-1029. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 19] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 80. | Lanzini A, Facchinetti D, Northfield TC. Maintenance of hepatic bile acid secretion rate during overnight fasting by bedtime bile acid administration. Gastroenterology. 1988;95:1029-1035. [PubMed] |
| 81. | Hardison WG, Grundy SM. Effect of ursodeoxycholate and its taurine conjugate on bile acid synthesis and cholesterol absorption. Gastroenterology. 1984;87:130-135. [PubMed] |
| 82. | Uchida K, Akiyoshi T, Igimi H, Takase H, Nomura Y, Ishihara S. Differential effects of ursodeoxycholic acid and ursocholic acid on the formation of biliary cholesterol crystals in mice. Lipids. 1991;26:526-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 83. | Wang DQ, Tazuma S, Cohen DE, Carey MC. Feeding natural hydrophilic bile acids inhibits intestinal cholesterol absorption: studies in the gallstone-susceptible mouse. Am J Physiol Gastrointest Liver Physiol. 2003;285:G494-G502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 158] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 84. | van de Heijning BJ, van de Meeberg PC, Portincasa P, Doornewaard H, Hoebers FJ, van Erpecum KJ, Vanberge-Henegouwen GP. Effects of ursodeoxycholic acid therapy on in vitro gallbladder contractility in patients with cholesterol gallstones. Dig Dis Sci. 1999;44:190-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 85. | Guarino MP, Cong P, Cicala M, Alloni R, Carotti S, Behar J. Ursodeoxycholic acid improves muscle contractility and inflammation in symptomatic gallbladders with cholesterol gallstones. Gut. 2007;56:815-820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 44] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 86. | Xiao ZL, Rho AK, Biancani P, Behar J. Effects of bile acids on the muscle functions of guinea pig gallbladder. Am J Physiol Gastrointest Liver Physiol. 2002;283:G87-G94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 36] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 87. | Stolk MF, Van de Heijning BJ, Van Erpecum KJ, Verheem A, Akkermans LM, Van Berge-Henegouwen GP. Effect of bile salts on in vitro gallbladder motility: preliminary study. Ital J Gastroenterol. 1996;28:105-110. [PubMed] |
| 88. | Paumgartner G, Pauletzki J, Sackmann M. Ursodeoxycholic acid treatment of cholesterol gallstone disease. Scand J Gastroenterol Suppl. 1994;204:27-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 89. | Leopold GR, Amberg J, Gosink BB, Mittelstaedt C. Gray scale ultrasonic cholecystography: a comparison with conventional radiographic techniques. Radiology. 1976;121:445-448. [PubMed] |
| 90. | Portincasa P, Di Ciaula A, Palmieri V, Vendemiale G, Altomare E, Palasciano G. Sonographic evaluation of gallstone burden in humans. Ital J Gastroenterol. 1994;26:141-144. [PubMed] |
| 91. | Everson GT, Braverman DZ, Johnson ML, Kern F. A critical evaluation of real-time ultrasonography for the study of gallbladder volume and contraction. Gastroenterology. 1980;79:40-46. [PubMed] |
| 92. | Portincasa P, Moschetta A, Colecchia A, Festi D, Palasciano G. Measurements of gallbladder motor function by ultrasonography: towards standardization. Dig Liver Dis. 2003;35 Suppl 3:S56-S61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 93. | Pauletzki J, Althaus R, Holl J, Sackmann M, Paumgartner G. Gallbladder emptying and gallstone formation: a prospective study on gallstone recurrence. Gastroenterology. 1996;111:765-771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 66] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 94. | Portincasa P, van Erpecum KJ, van De Meeberg PC, Dallinga-Thie GM, de Bruin TW, van Berge-Henegouwen GP. Apolipoprotein E4 genotype and gallbladder motility influence speed of gallstone clearance and risk of recurrence after extracorporeal shock-wave lithotripsy. Hepatology. 1996;24:580-587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 42] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 95. | Venneman NG, vanBerge-Henegouwen GP, Portincasa P, Stolk MF, Vos A, Plaisier PW, van Erpecum KJ. Absence of apolipoprotein E4 genotype, good gallbladder motility and presence of solitary stones delay rather than prevent gallstone recurrence after extracorporeal shock wave lithotripsy. J Hepatol. 2001;35:10-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 96. | Festi D, Frabboni R, Bazzoli F, Sangermano A, Ronchi M, Rossi L, Parini P, Orsini M, Primerano AM, Mazzella G. Gallbladder motility in cholesterol gallstone disease. Effect of ursodeoxycholic acid administration and gallstone dissolution. Gastroenterology. 1990;99:1779-1785. [PubMed] |
| 97. | Portincasa P, Di Ciaula A, Baldassarre G, Palmieri V, Gentile A, Cimmino A, Palasciano G. Gallbladder motor function in gallstone patients: sonographic and in vitro studies on the role of gallstones, smooth muscle function and gallbladder wall inflammation. J Hepatol. 1994;21:430-440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 63] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 98. | Pereira SP, Veysey MJ, Kennedy C, Hussaini SH, Murphy GM, Dowling RH. Gallstone dissolution with oral bile acid therapy. Importance of pretreatment CT scanning and reasons for nonresponse. Dig Dis Sci. 1997;42:1775-1782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 99. | Pereira SP, Hussaini SH, Kennedy C, Dowling RH. Gallbladder stone recurrence after medical treatment. Do gallstones recur true to type. Dig Dis Sci. 1995;40:2568-2575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 100. | Hussaini SH, Pereira SP, Murphy GM, Kennedy C, Wass JA, Besser GM, Dowling RH. Composition of gall bladder stones associated with octreotide: response to oral ursodeoxycholic acid. Gut. 1995;36:126-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 101. | Sackmann M, Eder H, Spengler U, Pauletzki J, Holl J, Paumgartner G, Sauerbruch T. Gallbladder emptying is an important factor in fragment disappearance after shock wave lithotripsy. J Hepatol. 1993;17:62-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 102. | Senior JR, Johnson MF, DeTurck DM, Bazzoli F, Roda E. In vivo kinetics of radiolucent gallstone dissolution by oral dihydroxy bile acids. Gastroenterology. 1990;99:243-251. [PubMed] |
| 103. | Jazrawi RP, Pigozzi MG, Galatola G, Lanzini A, Northfield TC. Optimum bile acid treatment for rapid gall stone dissolution. Gut. 1992;33:381-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 21] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 104. | Paumgartner G. Therapeutic options and choice of appropriate treatment. Advances in basic and clinical bile acid research. Dordrecht: Kluwer Academic Publishers 1996; 205-210. |
| 105. | Lanzini A, Jazrawi RP, Kupfer RM, Maudgal DP, Joseph AE, Northfield TC. Gallstone recurrence after medical dissolution. An overestimated threat. J Hepatol. 1986;3:241-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 49] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 106. | Rabenstein T, Radespiel-Tröger M, Höpfner L, Benninger J, Farnbacher M, Greess H, Lenz M, Hahn EG, Schneider HT. Ten years experience with piezoelectric extracorporeal shockwave lithotripsy of gallbladder stones. Eur J Gastroenterol Hepatol. 2005;17:629-639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 25] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 107. | Bateson MC, Bouchier IA, Trash DB, Maudgal DP, Northfield TC. Calcification of radiolucent gall stone during treatment with ursodeoxycholic acid. Br Med J (Clin Res Ed). 1981;283:645-646. [PubMed] |
| 108. | Sackmann M, Ippisch E, Sauerbruch T, Holl J, Brendel W, Paumgartner G. Early gallstone recurrence rate after successful shock-wave therapy. Gastroenterology. 1990;98:392-396. [PubMed] |
| 109. | Villanova N, Bazzoli F, Taroni F, Frabboni R, Mazzella G, Festi D, Barbara L, Roda E. Gallstone recurrence after successful oral bile acid treatment. A 12-year follow-up study and evaluation of long-term postdissolution treatment. Gastroenterology. 1989;97:726-731. [PubMed] |
| 110. | Petroni ML, Jazrawi RP, Pazzi P, Zuin M, Lanzini A, Fracchia M, Facchinetti D, Alvisi V, Ferraris R, Bland JM. Risk factors for the development of gallstone recurrence following medical dissolution. The British-Italian Gallstone Study Group. Eur J Gastroenterol Hepatol. 2000;12:695-700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 14] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 111. | Petroni ML, Jazrawi RP, Lanzini A, Zuin M, Pazzi P, Fracchia M, Boga E, Facchinetti D, Alvisi V, Galatola G. Repeated bile acid therapy for the long-term management of cholesterol gallstones. J Hepatol. 1996;25:719-724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 112. | O'Leary DP, Johnson AG. Future directions for conservative treatment of gallbladder calculi. Br J Surg. 1993;80:143-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 113. | Gilat T, Konikoff F. Pregnancy and the biliary tract. Can J Gastroenterol. 2000;14 Suppl D:55D-59D. [PubMed] |
| 114. | Sugerman HJ, Brewer WH, Shiffman ML, Brolin RE, Fobi MA, Linner JH, MacDonald KG, MacGregor AM, Martin LF, Oram-Smith JC. A multicenter, placebo-controlled, randomized, double-blind, prospective trial of prophylactic ursodiol for the prevention of gallstone formation following gastric-bypass-induced rapid weight loss. Am J Surg. 1995;169:91-96; discussion 96-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 299] [Cited by in RCA: 225] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 115. | Tint GS, Salen G, Colalillo A, Graber D, Verga D, Speck J, Shefer S. Ursodeoxycholic acid: a safe and effective agent for dissolving cholesterol gallstones. Ann Intern Med. 1982;97:351-356. [PubMed] |
| 116. | Meredith TJ, Williams GV, Maton PN, Murphy GM, Saxton HM, Dowling RH. Retrospective comparison of 'Cheno' and 'Urso' in the medical treatment of gallstones. Gut. 1982;23:382-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 117. | Tomida S, Abei M, Yamaguchi T, Matsuzaki Y, Shoda J, Tanaka N, Osuga T. Long-term ursodeoxycholic acid therapy is associated with reduced risk of biliary pain and acute cholecystitis in patients with gallbladder stones: a cohort analysis. Hepatology. 1999;30:6-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 64] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 118. | Venneman NG, Besselink MG, Keulemans YC, Vanberge-Henegouwen GP, Boermeester MA, Broeders IA, Go PM, van Erpecum KJ. Ursodeoxycholic acid exerts no beneficial effect in patients with symptomatic gallstones awaiting cholecystectomy. Hepatology. 2006;43:1276-1283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 45] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 119. | Kallien G, Lange K, Stange EF, Scheibner J. The pravastatin-induced decrease of biliary cholesterol secretion is not directly related to an inhibition of cholesterol synthesis in humans. Hepatology. 1999;30:14-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 120. | Duane WC, Hunninghake DB, Freeman ML, Pooler PA, Schlasner LA, Gebhard RL. Simvastatin, a competitive inhibitor of HMG-CoA reductase, lowers cholesterol saturation index of gallbladder bile. Hepatology. 1988;8:1147-1150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 86] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 121. | Loria P, Bertolotti M, Cassinadri MT, Dilengite MA, Bozzoli M, Carubbi F, Concari M, Guicciardi ME, Carulli N. Short-term effects of simvastatin on bile acid synthesis and bile lipid secretion in human subjects. Hepatology. 1994;19:882-888. [PubMed] |
| 122. | Hanson DS, Duane WC. Effects of lovastatin and chenodiol on bile acid synthesis, bile lipid composition, and biliary lipid secretion in healthy human subjects. J Lipid Res. 1994;35:1462-1468. [PubMed] |
| 123. | Saunders KD, Cates JA, Abedin MZ, Roslyn JJ. Lovastatin and gallstone dissolution: a preliminary study. Surgery. 1993;113:28-35. [PubMed] |
| 124. | Smit JW, van Erpecum KJ, Renooij W, Stolk MF, Edgar P, Doornewaard H, Vanberge-Henegouwen GP. The effects of the 3-hydroxy, 3-methylglutaryl coenzyme A reductase inhibitor pravastatin on bile composition and nucleation of cholesterol crystals in cholesterol gallstone disease. Hepatology. 1995;21:1523-1529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 125. | Tazuma S, Hatsushika S, Aihara N, Sagawa H, Yamashita G, Sasaki M, Sasaki H, Mizuno S, Tao S, Kajiyama G. Inhibitory effects of pravastatin, a competitive inhibitor of hydroxymethylglutaryl coenzyme A reductase, on cholesterol gallstone formation in prairie dogs. Digestion. 1992;51:179-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 126. | Abedin MZ, Narins SC, Park EH, Smith PR, Kirkwood KS. Lovastatin alters biliary lipid composition and dissolves gallstones: a long-term study in prairie dogs. Dig Dis Sci. 2002;47:2192-2210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 127. | Davis KG, Wertin TM, Schriver JP. The use of simvastatin for the prevention of gallstones in the lithogenic prairie dog model. Obes Surg. 2003;13:865-868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 128. | Tsai CJ, Leitzmann MF, Willett WC, Giovannucci EL. Statin use and the risk of cholecystectomy in women. Gastroenterology. 2009;136:1593-1600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 45] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 129. | Bodmer M, Brauchli YB, Krähenbühl S, Jick SS, Meier CR. Statin use and risk of gallstone disease followed by cholecystectomy. JAMA. 2009;302:2001-2007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 86] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 130. | Erichsen R, Frøslev T, Lash TL, Pedersen L, Sørensen HT. Long-term statin use and the risk of gallstone disease: A population-based case-control study. Am J Epidemiol. 2011;173:162-170. [PubMed] |
| 131. | Chapman BA, Burt MJ, Chisholm RJ, Allan RB, Yeo KH, Ross AG. Dissolution of gallstones with simvastatin, an HMG CoA reductase inhibitor. Dig Dis Sci. 1998;43:349-353. [PubMed] |
| 132. | Porsch-Ozçürümez M, Hardt PD, Schnell-Kretschmer H, von Bergmann K, Darui C, Nonhoff J, Abletshauser C, Klör HU. Effects of fluvastatin on biliary lipids in subjects with an elevated cholesterol saturation index. Eur J Clin Pharmacol. 2001;56:873-879. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 133. | Smith JL, Roach PD, Wittenberg LN, Riottot M, Pillay SP, Nestel PJ, Nathanson LK. Effects of simvastatin on hepatic cholesterol metabolism, bile lithogenicity and bile acid hydrophobicity in patients with gallstones. J Gastroenterol Hepatol. 2000;15:871-879. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 134. | Wilson IR, Hurrell MA, Pattinson NR, Chapman BA. The effect of simvastatin and bezafibrate on bile composition and gall-bladder emptying in female non-insulin-dependent diabetics. J Gastroenterol Hepatol. 1994;9:447-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 135. | Miettinen TE, Kiviluoto T, Taavitsainen M, Vuoristo M, Miettinen TA. Cholesterol metabolism and serum and biliary noncholesterol sterols in gallstone patients during simvastatin and ursodeoxycholic acid treatments. Hepatology. 1998;27:649-655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 136. | Sharma BC, Agarwal DK, Baijal SS, Saraswat VA. Pravastatin has no effect on bile lipid composition, nucleation time, and gallbladder motility in persons with normal levels of cholesterol. J Clin Gastroenterol. 1997;25:433-436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 137. | Caroli-Bosc FX, Le Gall P, Pugliese P, Delabre B, Caroli-Bosc C, Demarquay JF, Delmont JP, Rampal P, Montet JC. Role of fibrates and HMG-CoA reductase inhibitors in gallstone formation: epidemiological study in an unselected population. Dig Dis Sci. 2001;46:540-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 138. | González-Pérez A, García Rodríguez LA. Gallbladder disease in the general population: association with cardiovascular morbidity and therapy. Pharmacoepidemiol Drug Saf. 2007;16:524-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 139. | Wang DQ. Regulation of intestinal cholesterol absorption. Annu Rev Physiol. 2007;69:221-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 205] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 140. | Davis HR, Veltri EP. Zetia: inhibition of Niemann-Pick C1 Like 1 (NPC1L1) to reduce intestinal cholesterol absorption and treat hyperlipidemia. J Atheroscler Thromb. 2007;14:99-108. [PubMed] |
| 141. | Ge L, Wang J, Qi W, Miao HH, Cao J, Qu YX, Li BL, Song BL. The cholesterol absorption inhibitor ezetimibe acts by blocking the sterol-induced internalization of NPC1L1. Cell Metab. 2008;7:508-519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 255] [Cited by in RCA: 269] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 142. | Davis HR, Zhu LJ, Hoos LM, Tetzloff G, Maguire M, Liu J, Yao X, Iyer SP, Lam MH, Lund EG. Niemann-Pick C1 Like 1 (NPC1L1) is the intestinal phytosterol and cholesterol transporter and a key modulator of whole-body cholesterol homeostasis. J Biol Chem. 2004;279:33586-33592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 530] [Cited by in RCA: 526] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 143. | Altmann SW, Davis HR, Zhu LJ, Yao X, Hoos LM, Tetzloff G, Iyer SP, Maguire M, Golovko A, Zeng M. Niemann-Pick C1 Like 1 protein is critical for intestinal cholesterol absorption. Science. 2004;303:1201-1204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1297] [Cited by in RCA: 1295] [Article Influence: 61.7] [Reference Citation Analysis (0)] |
| 144. | Davies JP, Scott C, Oishi K, Liapis A, Ioannou YA. Inactivation of NPC1L1 causes multiple lipid transport defects and protects against diet-induced hypercholesterolemia. J Biol Chem. 2005;280:12710-12720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 217] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 145. | Wang HH, Wang DQ. Reduced susceptibility to cholesterol gallstone formation in mice that do not produce apolipoprotein B48 in the intestine. Hepatology. 2005;42:894-904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 146. | Zúñiga S, Molina H, Azocar L, Amigo L, Nervi F, Pimentel F, Jarufe N, Arrese M, Lammert F, Miquel JF. Ezetimibe prevents cholesterol gallstone formation in mice. Liver Int. 2008;28:935-947. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 147. | Ahmed MH, Byrne CD. Potential therapeutic uses for ezetimibe beyond lowering LDL-c to decrease cardiovascular events. Diabetes Obes Metab. 2010;12:958-966. [PubMed] |
| 148. | Di Ciaula A, Wang DQ, Wang HH, Bonfrate L, Portincasa P. Targets for current pharmacologic therapy in cholesterol gallstone disease. Gastroenterol Clin North Am. 2010;39:245-264, viii-ix. [PubMed] |
| 149. | Shiffman ML, Sugerman HJ, Kellum JM, Brewer WH, Moore EW. Gallstone formation after rapid weight loss: a prospective study in patients undergoing gastric bypass surgery for treatment of morbid obesity. Am J Gastroenterol. 1991;86:1000-1005. [PubMed] |
| 150. | Mangelsdorf DJ, Thummel C, Beato M, Herrlich P, Schütz G, Umesono K, Blumberg B, Kastner P, Mark M, Chambon P. The nuclear receptor superfamily: the second decade. Cell. 1995;83:835-839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5272] [Cited by in RCA: 5177] [Article Influence: 172.6] [Reference Citation Analysis (0)] |
| 151. | Janowski BA, Willy PJ, Devi TR, Falck JR, Mangelsdorf DJ. An oxysterol signalling pathway mediated by the nuclear receptor LXR alpha. Nature. 1996;383:728-731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1374] [Cited by in RCA: 1386] [Article Influence: 47.8] [Reference Citation Analysis (0)] |
| 152. | Parks DJ, Blanchard SG, Bledsoe RK, Chandra G, Consler TG, Kliewer SA, Stimmel JB, Willson TM, Zavacki AM, Moore DD. Bile acids: natural ligands for an orphan nuclear receptor. Science. 1999;284:1365-1368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1717] [Cited by in RCA: 1753] [Article Influence: 67.4] [Reference Citation Analysis (0)] |
| 153. | Makishima M, Okamoto AY, Repa JJ, Tu H, Learned RM, Luk A, Hull MV, Lustig KD, Mangelsdorf DJ, Shan B. Identification of a nuclear receptor for bile acids. Science. 1999;284:1362-1365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2012] [Cited by in RCA: 2090] [Article Influence: 80.4] [Reference Citation Analysis (0)] |
| 154. | Repa JJ, Mangelsdorf DJ. The liver X receptor gene team: potential new players in atherosclerosis. Nat Med. 2002;8:1243-1248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 310] [Cited by in RCA: 312] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 155. | Kalaany NY, Mangelsdorf DJ. LXRS and FXR: the yin and yang of cholesterol and fat metabolism. Annu Rev Physiol. 2006;68:159-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 449] [Cited by in RCA: 470] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 156. | Modica S, Moschetta A. Nuclear bile acid receptor FXR as pharmacological target: are we there yet. FEBS Lett. 2006;580:5492-5499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 67] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 157. | Moschetta A, Bookout AL, Mangelsdorf DJ. Prevention of cholesterol gallstone disease by FXR agonists in a mouse model. Nat Med. 2004;10:1352-1358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 248] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 158. | Liu Y, Binz J, Numerick MJ, Dennis S, Luo G, Desai B, MacKenzie KI, Mansfield TA, Kliewer SA, Goodwin B. Hepatoprotection by the farnesoid X receptor agonist GW4064 in rat models of intra- and extrahepatic cholestasis. J Clin Invest. 2003;112:1678-1687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 292] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 159. | Uppal H, Zhai Y, Gangopadhyay A, Khadem S, Ren S, Moser JA, Xie W. Activation of liver X receptor sensitizes mice to gallbladder cholesterol crystallization. Hepatology. 2008;47:1331-1342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 59] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 160. | Grünhage F, Lammert F. Gallstone disease. Pathogenesis of gallstones: A genetic perspective. Best Pract Res Clin Gastroenterol. 2006;20:997-1015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 161. | Diehl AK, Sugarek NJ, Todd KH. Clinical evaluation for gallstone disease: usefulness of symptoms and signs in diagnosis. Am J Med. 1990;89:29-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 56] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 162. | Berhane T, Vetrhus M, Hausken T, Olafsson S, Søndenaa K. Pain attacks in non-complicated and complicated gallstone disease have a characteristic pattern and are accompanied by dyspepsia in most patients: the results of a prospective study. Scand J Gastroenterol. 2006;41:93-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 36] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 163. | Elta GH, Barnett JL. Meperidine need not be proscribed during sphincter of Oddi manometry. Gastrointest Endosc. 1994;40:7-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 30] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 164. | Kumar A, Deed JS, Bhasin B, Kumar A, Thomas S. Comparison of the effect of diclofenac with hyoscine-N-butylbromide in the symptomatic treatment of acute biliary colic. ANZ J Surg. 2004;74:573-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 165. | Al-Waili N, Saloom KY. The analgesic effect of intravenous tenoxicam in symptomatic treatment of biliary colic: a comparison with hyoscine N-butylbromide. Eur J Med Res. 1998;3:475-479. [PubMed] |
| 166. | Akriviadis EA, Hatzigavriel M, Kapnias D, Kirimlidis J, Markantas A, Garyfallos A. Treatment of biliary colic with diclofenac: a randomized, double-blind, placebo-controlled study. Gastroenterology. 1997;113:225-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 52] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 167. | Goldman G, Kahn PJ, Alon R, Wiznitzer T. Biliary colic treatment and acute cholecystitis prevention by prostaglandin inhibitor. Dig Dis Sci. 1989;34:809-811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 38] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 168. | Gurusamy KS, Samraj K. Early versus delayed laparoscopic cholecystectomy for acute cholecystitis. Cochrane Database Syst Rev. 2006;CD005440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 80] [Article Influence: 4.2] [Reference Citation Analysis (0)] |