Published online Jun 28, 2014. doi: 10.4329/wjr.v6.i6.274
Revised: April 7, 2014
Accepted: May 14, 2014
Published online: June 28, 2014
Processing time: 182 Days and 9.4 Hours
Evaluation of rotator cuff is a common indication for magnetic resonance imaging (MRI) scanning of the shoulder. Conventional MRI is the most commonly used technique, while magnetic resonance (MR) arthrography is reserved for certain cases. Rotator cuff disorders are thought to be caused by a combination of internal and external mechanisms. A well-structured MRI report should comment on the relevant anatomic structures including the acromial type and orientation, the presence of os acromiale, acromio-clavicular degenerative spurs and fluid in the subacromial subdeltoid bursa. In addition, specific injuries of the rotator cuff tendons and the condition of the long head of biceps should be accurately reported. The size and extent of tendon tears, tendon retraction and fatty degeneration or atrophy of the muscles are all essential components of a surgically relevant MRI report.
Core tip: This review discusses the relevant anatomy of rotator cuff, mechanisms of rotator cuff injury, techniques of magnetic resonance imaging (MRI) used as well as all relevant MRI findings in an easy and ordered manner with illustrative figures and examples.
- Citation: Tawfik AM, El-Morsy A, Badran MA. Rotator cuff disorders: How to write a surgically relevant magnetic resonance imaging report? World J Radiol 2014; 6(6): 274-283
- URL: https://www.wjgnet.com/1949-8470/full/v6/i6/274.htm
- DOI: https://dx.doi.org/10.4329/wjr.v6.i6.274
Rotator cuff disorders are common in the middle and old age population. They are a major cause of chronic shoulder pain. In addition, rotator cuff disorders result in loss of strength and stability of the shoulder[1,2].
The balanced combination between accurate clinical examination, clear view of the patient’s needs and disabilities and precise radiological diagnostic modalities is invaluable in the correct formulation of the treatment plan of the patient whether surgically or conservatively. Magnetic resonance imaging (MRI), which is considered by many authors the modality of choice for diagnosing rotator cuff disorders[3,4], has a very important role in achieving this balance.
In this article, we discuss the techniques of MRI of the shoulder; the relevant rotator cuff anatomy, mechanisms of injury, and the specific imaging findings of rotator cuff disorders that are important for formulating a well-structured and complete MRI report.
MR imaging of the shoulder for evaluation of rotator cuff disorders may be carried out either conventionally (non-arthrographic), or with direct contrast distension of the joint (MR arthrography). Magnetic resonance arthrography was reported to be the most accurate imaging technique for diagnosis of both partial and full-thickness rotator cuff tears[5,6], but is limited by invasiveness and necessity of fluoroscopic guidance for injection, and therefore not routinely used for rotator cuff disorders[7,8]. An alternative technique, indirect MR arthrography, is a non-invasive technique using intravenous rather than intra-articular contrast to provide the arthrographic effect[9,10]. Continuous advances in MRI field strength, gradients, and coil technology have allowed even more accuracy for conventional MRI than that reported in the early literature, and conventional MRI is still the most commonly used technique for diagnosis of rotator cuff tears[3,11,12].
MR imaging is typically performed in the supine position with the arm by the side of the patient, in the neutral position. Images must be obtained in axial, coronal oblique and sagittal oblique planes[13]. The sequences used may vary but typically include T1-, proton density- and T2-weighted sequences and it is recommended to have both fat-suppressed and non-fat-suppressed sequences. Additional imaging in the abduction external rotation (ABER) position of the shoulder was reported to improve sensitivity and increase diagnostic confidence for partial-thickness tears of the supraspinatus tendon[10,14]. However, this technique is not widely spread due to difficult positioning and prolonged scan time.
The rotator cuff consists of four muscles; the supraspinatus, infraspinatus, subscapularis and teres minor muscles. The supraspinatus muscle originates from the supraspinous fossa of the scapula and passes on the superior aspect of the humeral head to be inserted in the greater tuberosity. The infraspinatus muscle originates from the infraspinous fossa and shares a common tendinous insertion with the supraspinatus tendon on the greater tuberosity; together with the tendon of teres minor muscle[15]. On the other hand, the subscapularis muscle originates from the subscapular fossa of the scapula and its wide tendon inserts in the lesser tuberosity, separated from the insertion of the other rotator muscles by the rotator interval.
The long head of biceps tendon is anatomically and functionally related to the rotator cuff. The tendon arises from the supraglenoid tubercle and its proximal segment (2-3 cm) is intra-articular. It exits the gleno-humeral joint and passes through the rotator interval between the subscapularis and supraspinatus tendons into the bicipital (intertubercular) groove of the proximal humerus[16].
The exact pathophysiology of rotator cuff injury, after exclusion of acute trauma, is still not fully understood. The current understanding is that rotator cuff degeneration is caused by a multifactorial pathogenesis, including both internal and external mechanisms[17,18].
Those are mechanisms that originate from within the tendons in the form of degenerative processes, possibly age-related, that result in alterations in the tendon biology, morphology, vascularity and mechanical properties[18,19].
External mechanisms include the well-known impingement syndromes, of which the subacromial impingement is the commonest. Subacromial impingement is defined as entrapment of the subacromial subdeltoid bursa and the supraspinatus tendon between the coraco-acromial arch and the greater tuberosity of the humerus. The main causes of this type of impingement include abnormal acromion configuration[20], osteoarthritis of the acromio-clavicular (AC) joint and narrowed subacromial space[21].
Another type of impingement syndromes is subcoracoid impingement; defined as entrapment of the subscapularis tendon in the coracohumeral interval (the space between the coracoid process and the anterior humerus). It is usually caused by abnormal coracoid configuration that may be congenital, traumatic or iatrogenic[17,21].
Less common types of impingement include secondary external impingement due to glenohumeral instability in the absence of outlet stenosis of the rotator cuff tendons[22], as well as internal impingement (intra-articular) due to compression of the articular surface of the tendons between the humeral head and glenoid[23].
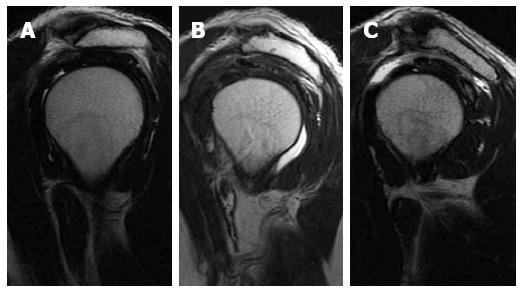
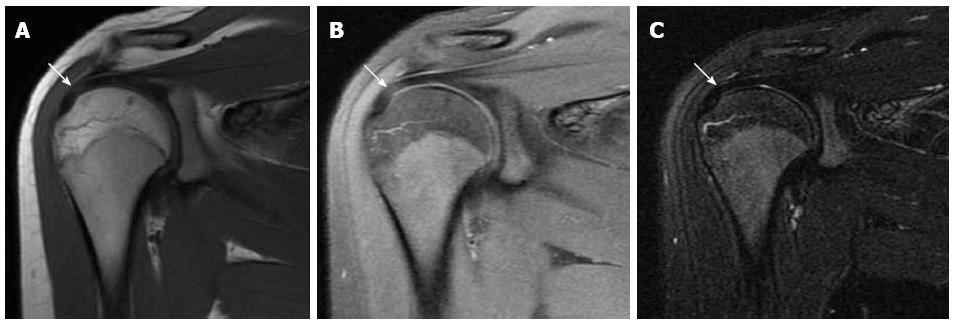
The acromion was classified according to its shape into three types (Figure 1). Type I has a flat under surface, type II a concave under surface and type three a concave under surface with anterior hook. Some authors added a further type IV to the original classification describing a convex under surface[24,25]. The shape of acromion is best depicted on sagittal oblique MR images lateral to the AC joint plane. The most common type is type II, while type III is the one most commonly associated with rotator cuff tears where its anterior hook causes injury of the anterior fibres of the supraspinatus tendon[26].
Normally, the acromion has no slope either anteriorly, laterally or inferiorly. A lateral or anterior down-sloping acromion and a low-lying acromion are thought to play a role in the development of subacromial impingement[21].
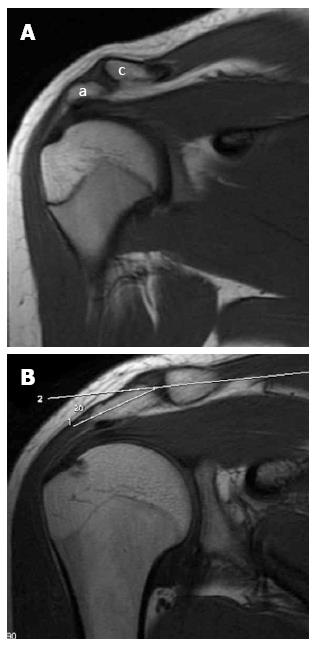
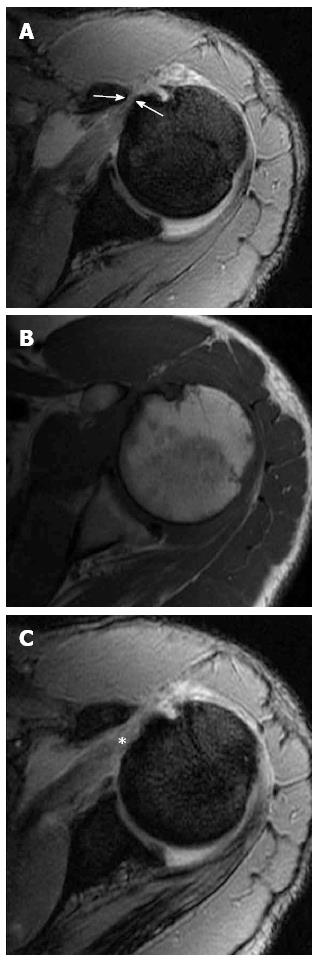
A low-lying acromion is diagnosed when the lower acromial surface is below the lower surface of the clavicle at the AC joint level on the anterior coronal oblique MR image (Figure 2A). On the same image showing the AC joint, an infero-lateral slope is detected by measuring the angle between the acromion axis and the clavicle (Figure 2B); an angle more than 10 degrees is abnormal[27]. An anterior slope is diagnosed on sagittal oblique images when the anterior part of the acromion is closer to the related part of the humeral convexity than its posterior part.
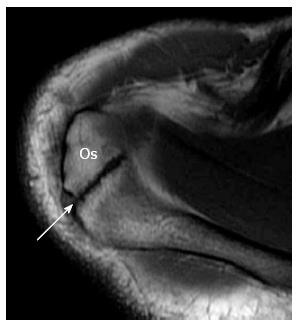
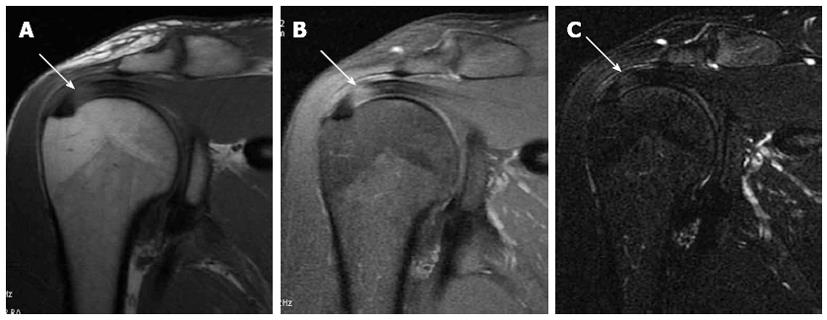
The acromion is formed from multiple ossification centres with complete fusion between the ages of 22 and 25 years. If one of the ossification centres fails to fuse, an accessory ossicle is formed; the os acromiale[28]. The os acromiale is mobile and is thought to contribute to impingement, but the reported incidence in normal population is between 1% and 15%[28]. It is best assessed in the upper axial images (Figure 3) where a low signal space is detected between the high signal marrow of the distal acromion and the non-fused ossicle[25]. On coronal oblique images it can be suspected by the presence of the so called “double AC joint”; the normal AC joint appears on the anterior image and the pseudo-articulation of the os acromiale appears on the posterior one[27].
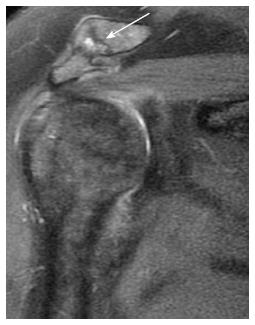
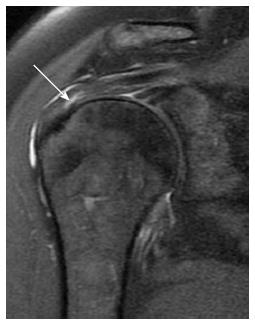
Osteoarthritis of the AC joint (Figure 4) is a common finding in patients with rotator cuff disorders. However, its role in impingement remains unclear and could either be a risk factor for impingement or a result of rotator cuff injury and disturbed shoulder biomechanics. A large osteophyte arising from the inferior surface of the joint is thought to be a cause of tendon tear[29]. MRI is more sensitive than radiography for detection of AC joint degenerative disease. MRI can differentiate joint enlargement due to capsular hypertrophy (intermediate signal intensity) form joint effusion (bright signal on T2-weighted images). Osteophytes appear in late stages of the disease[30].
The subacromial subdeltoid bursa is a synovial lined structure between the acromion and deltoid muscle externally and the rotator cuff tendons internally. The subacromial and subdeltoid components are connected in 95% of individuals. Normally it is not connected with the joint space but communication occurs in association with full thickness supraspinatus tears[31,32].
The normal bursa usually does not exceed 2 mm in thickness and is usually located posteriorly. On coronal MRI, features suggesting abnormal amount of fluid include thickness of fluid signal more than 3 mm, fluid signal medial to the level of the AC joint, and fluid in the anterior part of the bursa[33].
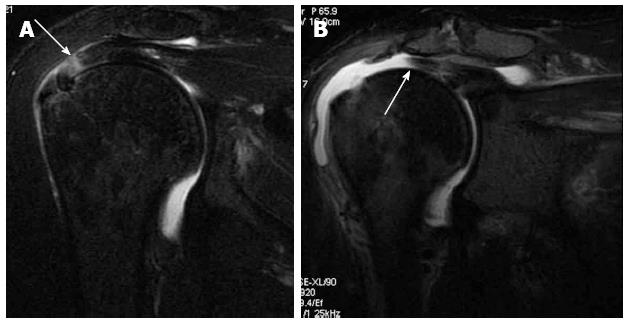
Tendinitis and tendinosis: Tendinitis and tendinosis typically appear as focal areas of increased signal intensity on proton density weighted images, that is less than that of fluid on T2-weighted images (Figure 5). Chronic forms are associated with thickening (tendinosis)[34].
Partial thickness tear: A partial thickness tear appears on fat-suppressed T2-weighted MR images as fluid signal intensity with thinning, or an incomplete gap, in the tendon[3]. The supraspinatus tendon is usually about 12 mm in average cranio-caudal thickness. Partial thickness tears are classified according to their depth into either grade I, in which less than one fourth of the fibres is torn; grade II, when more than one fourth and less than half of the tendon thickness is torn and grade III, when more than half of the tendon thickness is torn[24,35].
According to the tear site, partial thickness tears of the supraspinatus tendon are classified into bursal; articular surface or intra-tendinous tears (Figure 6). Articular surface tears are more common[35].
Full thickness tear: A full-thickness tendon tear appears as a focal, well-defined area of increased signal intensity on both T1- and T2-weighted images (Figure 7), that traverses the whole thickness of the tendon from the bursal to the articular surface[7,36].
Full-thickness tendon tears are classified according to the tear dimensions as small (less than 1 cm), medium (between 1 and 3 cm), large (between 3 and 5 cm) or massive (exceeding 5 cm)[24]. The dimensions are measured on coronal and sagittal T2 fat-suppressed images[37].
The degree of retraction of the torn supraspinatus tendon is typically assessed on coronal oblique images. When the tendon stump is still close to the insertion site, it is classified as stage 1. A stump retracted to the level of the humeral head is classified as stage 2, while stage 3 denotes retraction of the stump to the level of the glenoid[38,39].
Supraspinatus tendon: The most common site for rotator cuff tears is the supraspinatus tendon, especially at its distal part 1 to 2 cm from its insertion, the so called “critical zone”, where the vascularity is low and the effect of subacromial space narrowing or subacromial impingement is maximized (Figure 8)[26].
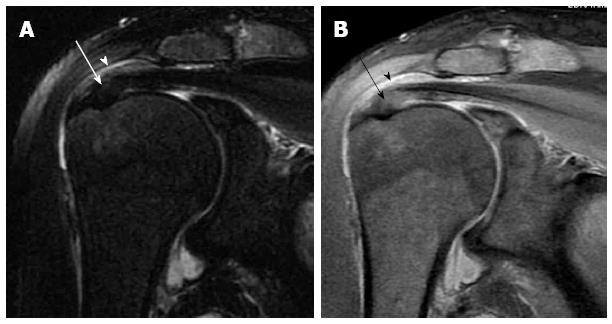
The supraspinatus tendon is best evaluated on the coronal oblique images. A potential pitfall is the magic angle artefact (Figure 9); that may occur whenever parallel collagen fibres are oriented at 55 degrees relative to the magnetic field. This effect is common at the distal end of the supraspinatus tendon and appears as high signal mimicking tendinitis or partial tear on short time of echo (TE) MR sequences. However, the high signal intensity disappears on using long TE sequences; for example T2 fat-suppressed sequence[25].
Infraspinatus tendon: The infraspinatus tendon is most commonly torn as an extension from supraspinatus tear (postero-superior cuff tear)[39]. The infraspinatus tendon is best evaluated on posterior coronal oblique fat-suppressed T2-weighted or STIR images (Figure 10). A second look may also be carried out on the upper axial images as well as on sagittal images[13].
Subscapularis tendon: Subscapularis tendon tears may occur as a component of massive rotator cuff tears[40,41]. The tendinous part of the subscapularis tendon is the broadest tendon among the rotator cuff and therefore commonly affected by tears and tendinitis (Figure 11).
The subscapularis tendon may be affected in isolation in traumatic injury[41]. Subcoracoid impingement, which is a cause of subscapularis degenerative tears (Figure 12), is suspected when the distance between the coracoid process and the lesser tuberosity of the humerus is less than 6 mm on axial MR images[21,42]. Other reported associated MRI signs include subcortical bone marrow edema of the coracoid process and lesser tuberosity of the huemrus.
Teres minor tendon: The teres minor tendon is the least injured among rotator cuff tendons[43]. The teres minor tendon is best evaluated on posterior coronal oblique and axial MR images and to less extent on sagittal images.
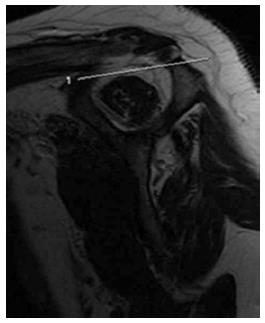
Atrophy of the supraspinatus muscle could be assessed by calculating the occupation ratio of the supraspinatus fossa. On the most lateral oblique sagittal image, atrophy of the supraspinatus muscle is diagnosed if the supraspinatus muscle occupies less than half the area of the fossa[44,45].
The tangent sign (Figure 13) describes an additional straight line drawn from the top of the coracoid process to the top of the spine of the scapula on the same oblique sagittal image as above. Atrophy is diagnosed when the superior border of the muscle lies below the tangent line[24,45,46].
Fatty degeneration of the supraspinatus muscles could be assessed on the T1-weighted oblique sagittal image in which the spine and body of the scapula appear. Fatty degeneration could be classified as either low (grade 0; no fat or grade 1; some fatty streaks) or moderate (grade 2; muscle > fat) or advanced (grade 3; muscle = fat and grade 4; muscle < fat)[47-49].
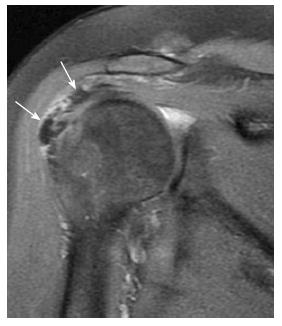
MR evaluation of the intra-articular segment of the long head biceps tendon (LHBT) is best carried out on both oblique sagittal and oblique coronal images, while the extra-articular segment is best evaluated on axial images. The LHBT tendon is covered with a synovial sheath connected with the joint space along its course within the bicipital grove[30,50]. Therefore, fluid signal around the tendon may be seen in cases of joint effusion, nevertheless in proportionate amount, and should not be mistaken for tenosynovitis. Tenosynovitis of the LHBT is diagnosed if fluid is detected around the tendon only or if the amount of fluid around the tendon is clearly out of proportion to that in the glenohumeral joint[51]. Tendinosis of the LHBT is suspected when focal thickening and high signal (but less than that of fluid) of the tendon or part of it is noted, usually associated with fluid signal within the synovial covering[52]. The most commonly affected part is the supra-humeral portion or the horizontal part and it may be a result of impingement. Tears of the LHBT vary from partial to complete tear to tendon avulsion. Tears appear as focal areas of high signal intensity, similar to that of fluid on T2-weighted images. Avulsion is diagnosed by noting the absence of the intra-articular segment of the tendon with no signs of dislocation. A dislocated LHBT (Figure 14) is often medially displaced, and is commonly associated with subscapularis tendon tear[50-52].
Bony changes: Erosions and degenerative changes of the lateral aspect of the greater tuberosity of the humerus (Figure 15) are common associated findings in rotator cuff disorders[30]. This finding is important for surgical planning as it significantly decreases the hold of anchors used during repair of torn rotator cuff tendons.
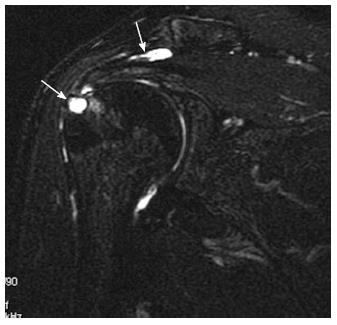
Calcific tendinopathy: Calcific tendinopathy most commonly affects the supraspinatus tendon, less commonly the infraspinatus and other tendons. On MRI, calcifications typically appear as focal areas of signal void on all spin echo sequences (Figure 16), and is usually difficult to detect within the natively low signal tendon[7,53]. The area of signal void increases on gradient-recalled echo sequence due to susceptibility effects[30].
Evaluation of rotator cuff disorders is one of the commonest indications for magnetic resonance (MR) imaging of the shoulder. Complete and accurate interpretation of MR images is essential to provide the treating clinician with adequate information for choosing the best therapy and avoiding unnecessary interventions. Radiologists should be familiar with the technique of MRI of the shoulder, the anatomy of the rotator cuff and the mechanisms of rotator cuff injury. A well-structured MRI report should include full comment on the rotator cuff tendons and all other relevant structures.
P- Reviewer: Iagulli ND S- Editor: Ji FF L- Editor: A E- Editor: Zhang DN
| 1. | Meislin RJ, Sperling JW, Stitik TP. Persistent shoulder pain: epidemiology, pathophysiology, and diagnosis. Am J Orthop (Belle Mead NJ). 2005;34:5-9. [PubMed] [Cited in This Article: ] |
| 2. | Burbank KM, Stevenson JH, Czarnecki GR, Dorfman J. Chronic shoulder pain: part I. Evaluation and diagnosis. Am Fam Physician. 2008;77:453-460. [PubMed] [Cited in This Article: ] |
| 3. | Magee T, Williams D. 3.0-T MRI of the supraspinatus tendon. AJR Am J Roentgenol. 2006;187:881-886. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 59] [Cited by in F6Publishing: 61] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 4. | Tuite MJ. Magnetic resonance imaging of rotator cuff disease and external impingement. Magn Reson Imaging Clin N Am. 2012;20:187-200, ix. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 17] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 5. | Waldt S, Bruegel M, Mueller D, Holzapfel K, Imhoff AB, Rummeny EJ, Woertler K. Rotator cuff tears: assessment with MR arthrography in 275 patients with arthroscopic correlation. Eur Radiol. 2007;17:491-498. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 110] [Cited by in F6Publishing: 84] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 6. | de Jesus JO, Parker L, Frangos AJ, Nazarian LN. Accuracy of MRI, MR arthrography, and ultrasound in the diagnosis of rotator cuff tears: a meta-analysis. AJR Am J Roentgenol. 2009;192:1701-1707. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 410] [Cited by in F6Publishing: 430] [Article Influence: 28.7] [Reference Citation Analysis (0)] |
| 7. | Teh J. Imaging of shoulder pathology. Orthopaedics and Trauma. 2011;25:19-29. [DOI] [Cited in This Article: ] [Cited by in Crossref: 2] [Cited by in F6Publishing: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 8. | Jana M, Gamanagatti S. Magnetic resonance imaging in glenohumeral instability. World J Radiol. 2011;3:224-232. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 22] [Cited by in F6Publishing: 18] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 9. | Yagci B, Manisali M, Yilmaz E, Ozkan M, Ekin A, Ozaksoy D, Kovanlikaya I. Indirect MR arthrography of the shoulder in detection of rotator cuff ruptures. Eur Radiol. 2001;11:258-262. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 37] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 10. | Herold T, Bachthaler M, Hamer OW, Hente R, Feuerbach S, Fellner C, Strotzer M, Lenhart M, Paetzel C. Indirect MR arthrography of the shoulder: use of abduction and external rotation to detect full- and partial-thickness tears of the supraspinatus tendon. Radiology. 2006;240:152-160. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 51] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 11. | Dinnes J, Loveman E, McIntyre L, Waugh N. The effectiveness of diagnostic tests for the assessment of shoulder pain due to soft tissue disorders: a systematic review. Health Technol Assess. 2003;7:iii, 1-166. [PubMed] [Cited in This Article: ] |
| 12. | Foad A, Wijdicks CA. The accuracy of magnetic resonance imaging and magnetic resonance arthrogram versus arthroscopy in the diagnosis of subscapularis tendon injury. Arthroscopy. 2012;28:636-641. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 1] [Reference Citation Analysis (0)] |
| 13. | Farshad-Amacker NA, Jain Palrecha S, Farshad M. The primer for sports medicine professionals on imaging: the shoulder. Sports Health. 2013;5:50-77. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 7] [Cited by in F6Publishing: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 14. | Schreinemachers SA, van der Hulst VP, Willems WJ, Bipat S, van der Woude HJ. Detection of partial-thickness supraspinatus tendon tears: is a single direct MR arthrography series in ABER position as accurate as conventional MR arthrography? Skeletal Radiol. 2009;38:967-975. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 30] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 15. | Mochizuki T, Sugaya H, Uomizu M, Maeda K, Matsuki K, Sekiya I, Muneta T, Akita K. Humeral insertion of the supraspinatus and infraspinatus. New anatomical findings regarding the footprint of the rotator cuff. J Bone Joint Surg Am. 2008;90:962-969. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 226] [Cited by in F6Publishing: 206] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 16. | Denard PJ, Dai X, Hanypsiak BT, Burkhart SS. Anatomy of the biceps tendon: implications for restoring physiological length-tension relation during biceps tenodesis with interference screw fixation. Arthroscopy. 2012;28:1352-1358. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 99] [Cited by in F6Publishing: 91] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 17. | Seagger RM, Wallace AL. Degenerative rotator cuff disease and impingement. Orthopaedics and Trauma. 2011;25:1-10. [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 18. | Seitz AL, McClure PW, Finucane S, Boardman ND, Michener LA. Mechanisms of rotator cuff tendinopathy: intrinsic, extrinsic, or both? Clin Biomech (Bristol, Avon). 2011;26:1-12. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 277] [Cited by in F6Publishing: 269] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 19. | Via AG, De Cupis M, Spoliti M, Oliva F. Clinical and biological aspects of rotator cuff tears. Muscles Ligaments Tendons J. 2013;3:70-79. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 49] [Article Influence: 4.5] [Reference Citation Analysis (2)] |
| 20. | Balke M, Schmidt C, Dedy N, Banerjee M, Bouillon B, Liem D. Correlation of acromial morphology with impingement syndrome and rotator cuff tears. Acta Orthop. 2013;84:178-183. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 148] [Cited by in F6Publishing: 147] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 21. | Mulyadi E, Harish S, O’Neill J, Rebello R. MRI of impingement syndromes of the shoulder. Clin Radiol. 2009;64:307-318. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 21] [Cited by in F6Publishing: 22] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 22. | Cowderoy GA, Lisle DA, O’Connell PT. Overuse and impingement syndromes of the shoulder in the athlete. Magn Reson Imaging Clin N Am. 2009;17:577-93, v. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 27] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 23. | Giaroli EL, Major NM, Higgins LD. MRI of internal impingement of the shoulder. AJR Am J Roentgenol. 2005;185:925-929. [PubMed] [Cited in This Article: ] |
| 24. | Morag Y, Jacobson JA, Miller B, De Maeseneer M, Girish G, Jamadar D. MR imaging of rotator cuff injury: what the clinician needs to know. Radiographics. 2006;26:1045-1065. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 108] [Cited by in F6Publishing: 87] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 25. | Fitzpatrick D, Walz DM. Shoulder MR imaging normal variants and imaging artifacts. Magn Reson Imaging Clin N Am. 2010;18:615-632. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 26. | Khan Y, Nagy MT, Malal J, Waseem M. The painful shoulder: shoulder impingement syndrome. Open Orthop J. 2013;7:347-351. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 27. | Stabler A. Impingement and Rotator Cuff Disease. Imaging of the Shoulder: Techniques and Applications. 1st ed. Berlin Heidelberg: Springer-Verlag 2004; 121-159. [Cited in This Article: ] |
| 28. | Boehm TD, Rolf O, Martetschlaeger F, Kenn W, Gohlke F. Rotator cuff tears associated with os acromiale. Acta Orthop. 2005;76:241-244. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 29. | Roidis NT, Motamed S, Vaishnav S, Ebramzadeh E, Karachalios TS, Itamura JM. The influence of the acromioclavicular joint degeneration on supraspinatus outlet impingement and the acromion shape. J Orthop Surg (Hong Kong). 2009;17:331-334. [PubMed] [Cited in This Article: ] |
| 30. | Vahlensieck M. MRI of the shoulder. Eur Radiol. 2000;10:242-249. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 20] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 31. | van Holsbeeck M, Strouse PJ. Sonography of the shoulder: evaluation of the subacromial-subdeltoid bursa. AJR Am J Roentgenol. 1993;160:561-564. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 108] [Cited by in F6Publishing: 85] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 32. | Precerutti M, Garioni E, Madonia L, Draghi F. US anatomy of the shoulder: Pictorial essay. J Ultrasound. 2010;13:179-187. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 33. | White EA, Schweitzer ME, Haims AH. Range of normal and abnormal subacromial/subdeltoid bursa fluid. J Comput Assist Tomogr. 2006;30:316-320. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 31] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 34. | Sein ML, Walton J, Linklater J, Harris C, Dugal T, Appleyard R, Kirkbride B, Kuah D, Murrell GA. Reliability of MRI assessment of supraspinatus tendinopathy. Br J Sports Med. 2007;41:e9. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 45] [Cited by in F6Publishing: 46] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 35. | Finnan RP, Crosby LA. Partial-thickness rotator cuff tears. J Shoulder Elbow Surg. 2010;19:609-616. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 60] [Cited by in F6Publishing: 55] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 36. | Reinus WR, Shady KL, Mirowitz SA, Totty WG. MR diagnosis of rotator cuff tears of the shoulder: value of using T2-weighted fat-saturated images. AJR Am J Roentgenol. 1995;164:1451-1455. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 95] [Cited by in F6Publishing: 97] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 37. | Dwyer T, Razmjou H, Henry P, Gosselin-Fournier S, Holtby R. Association between pre-operative magnetic resonance imaging and reparability of large and massive rotator cuff tears. Knee Surg Sports Traumatol Arthrosc. 2013;Epub ahead of print. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 60] [Cited by in F6Publishing: 55] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 38. | Patte D. Classification of rotator cuff lesions. Clin Orthop Relat Res. 1990;81-86. [PubMed] [Cited in This Article: ] |
| 39. | Cooper A, Ali A. Rotator cuff tears. Surgery. 2013;31:168-171. [DOI] [Cited in This Article: ] [Cited by in Crossref: 2] [Cited by in F6Publishing: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 40. | Bergin D, Parker L, Zoga A, Morrison W. Abnormalities on MRI of the subscapularis tendon in the presence of a full-thickness supraspinatus tendon tear. AJR Am J Roentgenol. 2006;186:454-459. [PubMed] [Cited in This Article: ] |
| 41. | Lyons RP, Green A. Subscapularis tendon tears. J Am Acad Orthop Surg. 2005;13:353-363. [PubMed] [Cited in This Article: ] |
| 42. | Richards DP, Burkhart SS, Campbell SE. Relation between narrowed coracohumeral distance and subscapularis tears. Arthroscopy. 2005;21:1223-1228. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 118] [Cited by in F6Publishing: 109] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 43. | Melis B, DeFranco MJ, Lädermann A, Barthelemy R, Walch G. The teres minor muscle in rotator cuff tendon tears. Skeletal Radiol. 2011;40:1335-1344. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 56] [Cited by in F6Publishing: 59] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 44. | Thomazeau H, Rolland Y, Lucas C, Duval JM, Langlais F. Atrophy of the supraspinatus belly. Assessment by MRI in 55 patients with rotator cuff pathology. Acta Orthop Scand. 1996;67:264-268. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 347] [Cited by in F6Publishing: 330] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 45. | Moosmayer S, Tariq R, Stiris MG, Smith HJ. MRI of symptomatic and asymptomatic full-thickness rotator cuff tears. A comparison of findings in 100 subjects. Acta Orthop. 2010;81:361-366. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 45] [Cited by in F6Publishing: 45] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 46. | Zanetti M, Gerber C, Hodler J. Quantitative assessment of the muscles of the rotator cuff with magnetic resonance imaging. Invest Radiol. 1998;33:163-170. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 358] [Cited by in F6Publishing: 339] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 47. | Goutallier D, Postel JM, Bernageau J, Lavau L, Voisin MC. Fatty muscle degeneration in cuff ruptures. Pre- and postoperative evaluation by CT scan. Clin Orthop Relat Res. 1994;78-83. [PubMed] [Cited in This Article: ] |
| 48. | Fuchs B, Weishaupt D, Zanetti M, Hodler J, Gerber C. Fatty degeneration of the muscles of the rotator cuff: assessment by computed tomography versus magnetic resonance imaging. J Shoulder Elbow Surg. 1999;8:599-605. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 968] [Cited by in F6Publishing: 981] [Article Influence: 39.2] [Reference Citation Analysis (0)] |
| 49. | Oh JH, Kim SH, Choi JA, Kim Y, Oh CH. Reliability of the grading system for fatty degeneration of rotator cuff muscles. Clin Orthop Relat Res. 2010;468:1558-1564. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 134] [Cited by in F6Publishing: 137] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 50. | Nakata W, Katou S, Fujita A, Nakata M, Lefor AT, Sugimoto H. Biceps pulley: normal anatomy and associated lesions at MR arthrography. Radiographics. 2011;31:791-810. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 71] [Cited by in F6Publishing: 77] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 51. | Beall DP, Williamson EE, Ly JQ, Adkins MC, Emery RL, Jones TP, Rowland CM. Association of biceps tendon tears with rotator cuff abnormalities: degree of correlation with tears of the anterior and superior portions of the rotator cuff. AJR Am J Roentgenol. 2003;180:633-639. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 118] [Cited by in F6Publishing: 116] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 52. | Vinson EN, Wittstein J, Garrigues GE, Taylor DC. MRI of selected abnormalities at the anterior superior aspect of the shoulder: potential pitfalls and subtle diagnoses. AJR Am J Roentgenol. 2012;199:534-545. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 53. | Zubler C, Mengiardi B, Schmid MR, Hodler J, Jost B, Pfirrmann CW. MR arthrography in calcific tendinitis of the shoulder: diagnostic performance and pitfalls. Eur Radiol. 2007;17:1603-1610. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 40] [Cited by in F6Publishing: 36] [Article Influence: 2.0] [Reference Citation Analysis (0)] |