Published online Jul 28, 2012. doi: 10.4329/wjr.v4.i7.291
Revised: June 5, 2012
Accepted: June 12, 2012
Published online: July 28, 2012
AIM: To evaluate the sources of variation influencing the microvascularization parameters measured by dynamic contrast-enhanced ultrasonography (DCE-US).
METHODS: Firstly, we evaluated, in vitro, the impact of the manual repositioning of the ultrasound probe and the variations in flow rates. Experiments were conducted using a custom-made phantom setup simulating a tumor and its associated arterial input. Secondly, we evaluated, in vivo, the impact of multiple contrast agent injections and of examination day, as well as the influence of the size of region of interest (ROI) associated with the arterial input function (AIF). Experiments were conducted on xenografted B16F10 female nude mice. For all of the experiments, an ultrasound scanner along with a linear transducer was used to perform pulse inversion imaging based on linear raw data throughout the experiments. Semi-quantitative and quantitative analyses were performed using two signal-processing methods.
RESULTS: In vitro, no microvascularization parameters, whether semi-quantitative or quantitative, were significantly correlated (P values from 0.059 to 0.860) with the repositioning of the probe. In addition, all semi-quantitative microvascularization parameters were correlated with the flow variation while only one quantitative parameter, the tumor blood flow, exhibited P value lower than 0.05 (P = 0.004). In vivo, multiple contrast agent injections had no significant impact (P values from 0.060 to 0.885) on microvascularization parameters. In addition, it was demonstrated that semi-quantitative microvascularization parameters were correlated with the tumor growth while among the quantitative parameters, only the tissue blood flow exhibited P value lower than 0.05 (P = 0.015). Based on these results, it was demonstrated that the ROI size of the AIF had significant influence on microvascularization parameters: in the context of larger arterial ROI (from 1.17 ± 0.6 mm3 to 3.65 ± 0.3 mm3), tumor blood flow and tumor blood volume were correlated with the tumor growth, exhibiting P values lower than 0.001.
CONCLUSION: AIF selection is an essential aspect of the deconvolution process to validate the quantitative DCE-US method.
- Citation: Gauthier M, Pitre-Champagnat S, Tabarout F, Leguerney I, Polrot M, Lassau N. Impact of the arterial input function on microvascularization parameter measurements using dynamic contrast-enhanced ultrasonography. World J Radiol 2012; 4(7): 291-301
- URL: https://www.wjgnet.com/1949-8470/full/v4/i7/291.htm
- DOI: https://dx.doi.org/10.4329/wjr.v4.i7.291
Tumor angiogenesis is a process characterized by the development of new blood vessels supplying tumors with nutrients and oxygen[1,2]. This process is an essential step for tumor growth as well as the initiation of metastasis. In this context, research is currently focused on developing anti-angiogenic treatments resulting in the destruction of neoblood vessels, which often occurs initially without any morphological changes[3-5].
Since February 2000, Response Evaluation Criteria in Solid Tumors has been used by the entire cancer clinical research community as an essential tool in evaluating responses to anti-tumor treatments[6]. As this involves analyzing morphological information[7], it is commonly recognized that these criteria are no longer optimal in the early assessment of anti-angiogenic therapies, which primarily target microvasculature. Consequently, functional imaging is recognized as the best modality for evaluating such treatments.
However, the current microvascularization parameters on which therapy evaluations are based do not properly correlate with the vascular physiology, which might interfere with the final interpretation. Such parameters are defined as semi-quantitative since they only provide a relative access to the physiological parameters through the current ultrasound imaging mode. These limits may be overcome by working with the arterial input function (AIF), which takes into account physiological variations such as the cardiac flow rate, the arterial blood pressure, the vascular structure, and the way the contrast agent is injected[8]. This is done through a deconvolution process which allows access to quantitatively-defined microvascularization parameters, since it provides absolute evaluation of microvasculature parameters. Deconvolution has been already performed using other imaging modalities[9-11]. Previous studies have been performed to develop a deconvolution technique based on Tikhonov regularization[12,13]: preliminary results demonstrated a diminution in microvascularization parameter coefficients of variation from 30% to 13%, respectively measured without and with the deconvolution process.
Considering these results, the aim of this study was to determine and analyze sources of variation that impact microvascularization parameters measured using dynamic contrast-enhanced ultrasonography (DCE-US) combined with a deconvolution process, both in vitro and in vivo, following bolus injections of SonoVue® (Bracco, Milan, Italy).
DCE-US data were acquired following bolus injections of SonoVue®, a second generation echo contrast agent made of microbubbles filled with sulphur hexafluoride (SF6) and stabilized by a shell of amphiphilic phospholipids[14,15]. The diameter of the microbubbles, ranging from 1 to 10 μm[15], allowed a purely intravascular circulation through the whole blood volume, which made it ideal for the evaluation of microvascularization[15]. SF6 is an innocuous, stable and inert gas that does not interact with any other molecules found in the body. In addition, SonoVue® is highly resistant to pressure changes and exhibits a non-linear response of the microbubbles when insonated at low acoustic power, thus provides continuous real-time ultrasonographic (US) imaging without any bubble destruction[14,16,17]. SonoVue® is available as a kit which includes one vial of SF6, a powder combining phospholipids and pharmaceutical grade polyethyleneglycol, and one syringe pre-filled with 5 mL of 0.9% sodium chloride. Before any US exam, reconstruction of the contrast agent was performed by introducing the content of the syringe into the vial followed by manual shaking for at least 20 s. After a few minutes of no use, the microbubbles tend to accumulate at the upper surface because of buoyancy. Thus, to inject a homogeneous bolus of contrast agent, the vial was manually checked before each injection. SonoVue® is stable for 6 h, and all experiments were conducted within this limit[14].
The time-intensity method is based on the dye-dilution theory, which provides a mathematical method for estimating microvascularization parameters based on monitoring the tracer concentration as a function of time generating a time intensity curve (TIC). Microvascularization parameters to be analyzed are extracted from the TIC[18,19]. To be valid, a series of assumptions must be verified[20]: (1) flow must be constant to ensure the amount of microbubbles injected has no effect on the flux; (2) blood and contrast agent must be mixed homogeneously; (3) recirculation should not interfere with the first pass; and (4) the mixing of the contrast agent must exhibit linearity and stability[21]. In this context, linearity refers to the linear relationship between the injected concentration of contrast agents and the measured US signal. This was confirmed for low doses[15] and in the context of bolus injections of contrast agent[22].
In this study, these conditions were assumed to be satisfied, allowing the direct extraction of the microvascularization parameters from the TIC.
Images were acquired using a Toshiba Aplio® XG ultrasound scanner (Toshiba, Japan) connected to a 7-14 MHz linear transducer (PLT 1202S probe). (1) B-mode and Doppler imaging were performed at 14 MHz; and (2) Harmonic imaging was performed at 10 MHz using the pulse inversion mode in which two pulses were sent into the tissue, the second pulse being an inverted copy of the first one. Thus, the associated echoes were detected and summed[23]. Linear scattering from the two pulses produced echoes that were inverted copies of each other, resulting in a null signal. Linear scattering dominates in tissue. Conversely, non-linear scattering of the two pulses, which dominates in microbubbles, produced echoes that were no longer inverted, resulting in a non-null signal[24].
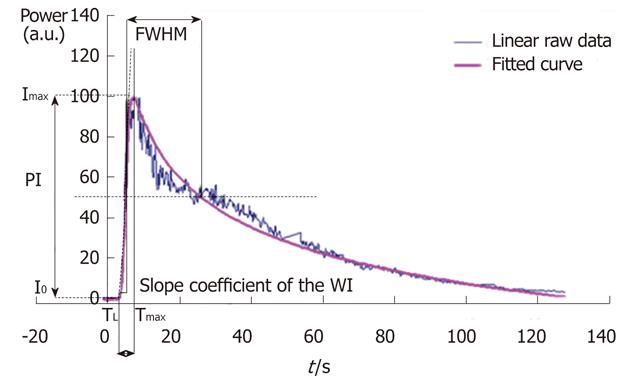
Semi-quantitative microvascularization parameters (the current method): Acquisitions and analyses were based on linear raw data (uncompressed data before standard video visualization). Such data exhibit the advantage of a linear dynamic range, which is the essential aspect of TIC analysis[25,26]. Following bolus injections of SonoVue®, several harmonic images were acquired. Image analysis followed a protocol described in previous studies[27,28] using dedicated software called CHIQ® (Toshiba). Thus, the current method allowed one to graphically extract semi-quantitative microvascularization parameters following the fitting of the tumor TIC based on minimizing the differences between the parameters of the raw curve and the coefficients of the IGR equation (Patent: WO/2008/053268 entitled “Method and system for quantification of tumoral vascularization”):
I(t) describes the variation in the intensity of contrast uptake as a function of time. a0 is the intensity before the arrival of the contrast agent. a1 is linked to the maximum value of contrast uptake. a2 is linked to the rise time to the peak intensity (PI). p is a coefficient related to the increase in intensity. q is a coefficient related to the decrease in intensity. A and B are arbitrary parameters.
Semi-quantitative microvascularization parameters have already been described in previous studies[27,28]. These are PI, the time to peak intensity (TPI), the area under the curve (AUC), the wash-in (AUWI), the wash-out (AUWO) and the full width at half maximum (FWHM) (Figure 1[12]).
Quantitative microvascularization parameters (the deconvolution process): Based on the dye-dilution theory, the influence of the arterial input manifests itself through the following fundamental convolution equation:
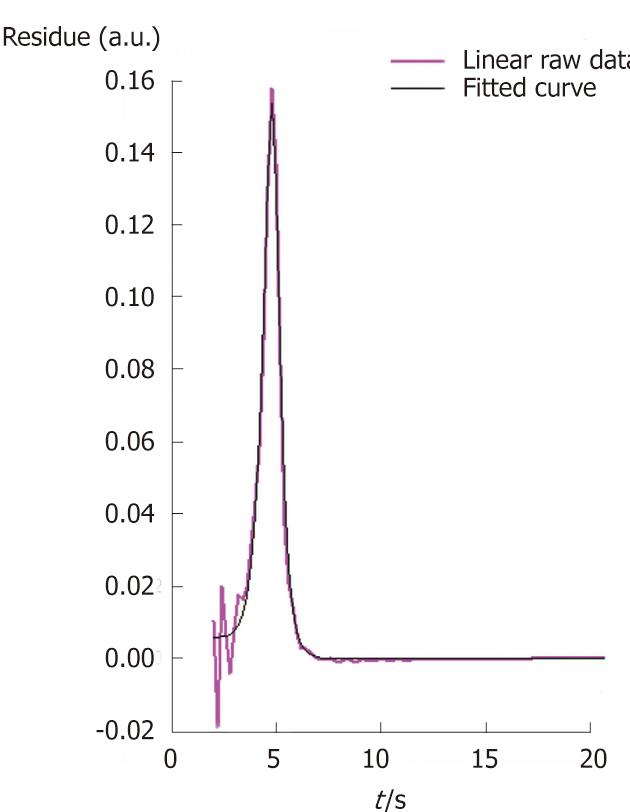
where Ct(t) is the concentration of the contrast agent measured in the tumor as a function of time, Ca(t) is the AIF, BF is the tissue blood flow (unit: mL/min per 100 g of tissue), ρ is the tissue density (unit: g/mL) and kH represents the difference in hematocrit between capillaries and arteries: ρ and kH are commonly approximated by 1[29,30]. R(t) is the residue function and is defined as the relative amount of tracer in the region of interest (ROI) under the hypothesis of an instantaneous unit bolus injection [R(0) = 1 ; R(∞) = 0]. The shape of the R(t) function reflects the properties of both the vasculature and the tissue[31].
In order to derive quantitative parameters from this equation, the residue function R(t) can be determined using a process called deconvolution. The deconvolution method developed in our lab and dedicated to the DCE-US imaging is based on the Tikhonov regularization and was previously described by Gauthier et al[12].
Three quantitative microvascularization parameters are thus graphically extracted from the residue function: the BF, the tissue blood volume (BV) and the mean transit time (MTT) (Figure 2[27]).
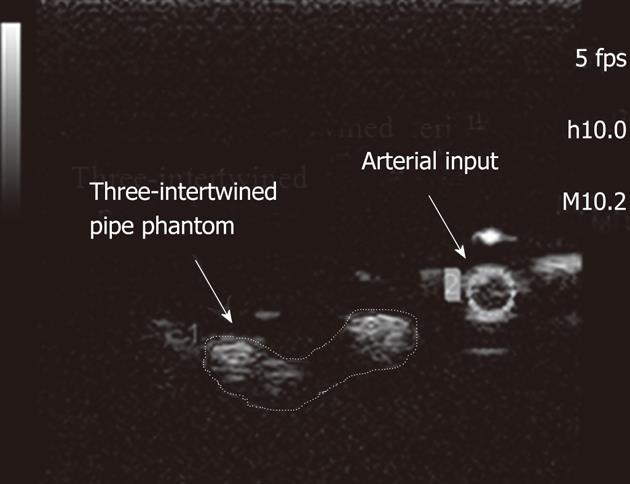
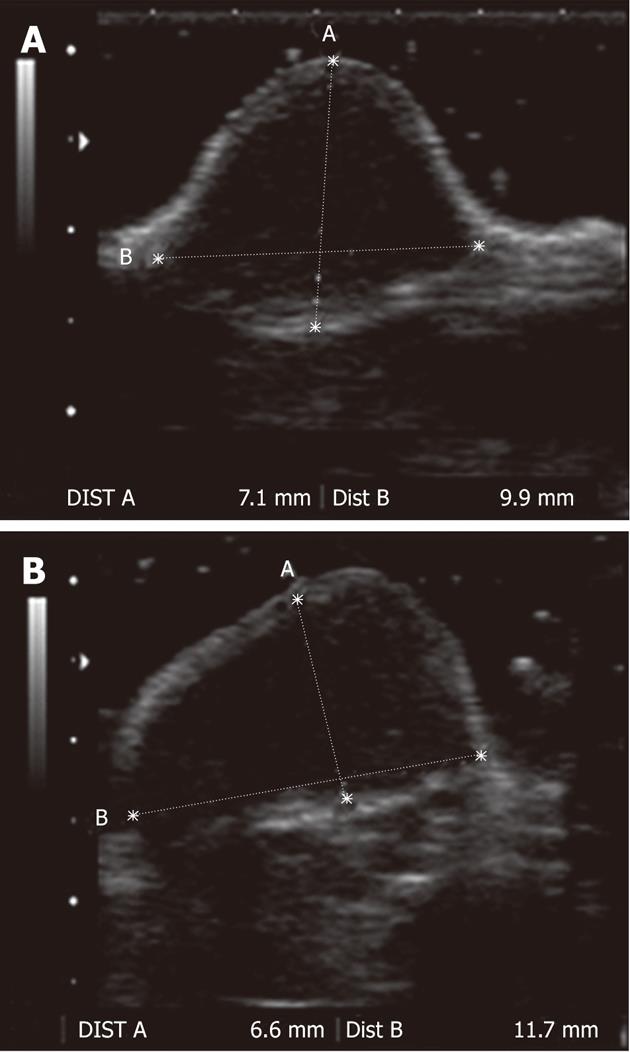
Three-intertwined pipe phantom: The phantom used in the in vitro experiments consisted of three-intertwined silicone pipes simulating a heterogeneous structure akin to that of vessels in tumor and feeding pipes which simulated the arterial input (Figure 3).
The phantom was immersed in a custom-made water tank connected to a peristaltic pump (SP vario/ PD 5101, Heidolph®, Germany) which provided a water flow rate set as required for the experiments. The three silicone pipes had an internal diameter of 2 mm with a 1 mm thick wall. Both the input and the output of the phantom were composed of three-way taps (Discofix®, B. Braun, Melsungen, Germany) allowing linkage between the three pipes. A new ROI, containing both pipes, feeding pipe and water spaces, was drawn for each injection and the associated TIC was obtained.
The total amount of water in the circuit was set at 60 mL. The amount of injected contrast agent was 0.06 mL. This volume corresponded to the ratio granted by French marketing approval (“Autorisation de Mise sur le Marché”: AMM) (2.4 mL of SonoVue® for 5 L of blood): this was defined as the AMM ratio. The second volume was associated with the ratio routinely used for clinical exams (4.8 mL of SonoVue® for 5 L of blood). In particular, this ratio was used in four studies led by the IGR involving 117 patients and 800 DCE-US exams[32,33] as well as in a French national project supported by the “Institut National du Cancer” (French National Cancer Institute)[34]. This ratio was defined as the internal growth rate ratio.
Bolus injections of SonoVue® were performed using a 1 mL syringe (Terumo®, Belgium). To minimize potential errors due to SonoVue® residues, all of the injection materials were changed before each injection: the circuit was entirely emptied, rinsed and reset with water. Thus no contrast agent residues were present in the circuit, allowing the initial conditions to be exactly the same throughout all of the experiments[27].
Before the series of acquisitions, a Doppler image was acquired to help the operator image the same setup through all the experiments.
Acquisitions were performed at a low mechanical index (MI = 0.21) and at a rate of 5 frames per second (fps). Two ROIs were selected to be analyzed (Figure 4[12]): the first one was associated with the three-intertwined pipe phantom while the second was associated with its feeding pipe. Deconvolution was performed between the two TICs extracted from these ROIs through the CHIQ® software.
Experimental protocol: Sources of DCE-US variability include the repositioning of the US probe before each examination: this has to be the same for each acquisition of the therapeutic evaluation process which can last several months or years. Indeed, evaluation of therapy is based on analyzing evolution of microvascularization parameters linked to a specific target along the whole duration of the treatment. In this context, the aim of this study was to evaluate the impact of the repositioning of the US probe on microvascularization parameters. The experiments consisted of: (1) 10 consecutive contrast agent injections with the US probe unchanged; and (2) 10 consecutive contrast agent injections with the US probe manually replaced before each exam.
Experimental protocol: The absolute evaluation of the microvascularization parameters is not possible through the methodology which does not take into account the arterial input[8]. Until now, different studies have investigated and used the link between the microvascularization parameters directly extracted from the tumor TIC and the physiological parameters of interest[35-38].
Thus, the BF is linked to the slope of the WI and the TPI, which are currently the two parameters mentioned in the literature. None of the semi-quantitative microvascularization parameters are specific to the BF. On the contrary, as expected by definition, only one quantitative microvascularization parameter should be specific to the blood flow: it is BF.
To perform the study, 4 injections per flow rate were performed and 3 flow rates were tested: 21.2 mL/min, 42.4 mL/min and 63.6 mL/min. Variations in flow rates were controlled using the peristaltic pump.
Animals and tumor model: Animal experiments were conducted with five nude female mice aged from six to eight weeks with the approval of the European Convention for the Protection of Vertebrate Animals used for experimental and other scientific purposes (Strasbourg, 18.III.1986; text amended according to the provisions of protocol ETS No. 170 as of its entry into force on 2nd December 2005). The tumor model B16F10 (CRL-6475, ATCC, American Type Culture Collection) melanoma cell line, a murine skin cancer, was selected to perform the in vivo experiments. Tumor cells were prepared and cultured in Dulbeco Minimum Essential Medium (Gibco Life Technologies, France) combined with 10% fetal bovine serum, 1% penicillin/streptomycin and glutamate (Invitrogen Life Technologies, Inc., France) to avoid bacterial contamination of the solution. While growing, cells were maintained in an incubator at 37 °C. Tumors were xenografted onto the right flank (Figure 5[12,27]) of five mice through a subcutaneous injection of 2 × 106 melanoma cells in 0.2 mL of phosphate buffered saline. DCE-US exams were performed following three 0.1 mL injections of SonoVue® according to the methodology used in our lab.
Mice received chemical anesthesia based upon their weight. Product was injected intraperitoneally using the 1 mL syringe. The solution consisted of ketamine (10 mg/mL, Ketalar®, Parapharm, France) and xylazine (2%, Rompum®, Bayer, France). To ensure the mice remained asleep throughout the experiment, 150 μL/g per mouse was systematically injected.
In vivo images were acquired using the same ultrasound machine and probe as the in vitro experiments. Images were acquired using a mechanical index set at 0.21 and a rate of 5 fps. In addition, preliminary fundamental B-mode images were acquired to evaluate the tumor volume prior to the SonoVue® injection (Figure 6[12]). The three perpendicular tumor diameters were determined and the tumor volume was derived according to the following formula:
In the following study, two new ROIs were selected for each acquisition. The first ROI included only the tumor while the second ROI was associated with an arterial input. We gave preference to the location of the arterial ROI instead of its size to avoid any delay or dispersion effects that can impact the quantitative microvascularization parameters[30]. Two TICs were extracted from these ROIs. To ensure the complete elimination of the contrast agent between each acquisition, a break of 15 min and 3 min of insonation at high MI (MI = 1.04) were observed. Mice were kept asleep no more than 2 h[39]. This duration included the time required for the mice to obtain a stable heart rate after the administration of anesthesia, acquisition time and the time of the break between each injection.
Multiple injections/day of examination: The first evaluated factor was the order of injection. This is a parameter of interest especially in the context of DCE-US exams that sometimes require the re-injection of the contrast agent. This study aimed to confirm that multiple injections had no significant impact on semi-quantitative and quantitative microvascularization parameters.
The second factor included different parameters. It was defined as the examination day to ensure no restriction of its meaning. Because melanoma cell lines exhibit a high doubling time of about 20 h[40-42], the examination day may be a source of variation. Thus, to analyze this second factor amounts to evaluating whether any morphological or functional modification of the tumor significantly interferes with the microvascularization parameters.
For logistic reasons, a total of 3 mice were evaluated over 4 d following a maximum of 4 contrast agent injections performed during the study (Table 1).
| Mouse number | Number of days | Number of injections |
| Multiple injections and day of examination | ||
| 1 | 4 | 3 |
| 4 | ||
| 3 | ||
| 4 | ||
| 2 | 2 | 3 |
| 2 | ||
| 3 | 2 | 2 |
| 4 | ||
| Influence of the size of the arterial region of interest | ||
| 1 | 3 | 3 |
| 3 | ||
| 2 | ||
| 2 | 3 | 2 |
| 3 | ||
| 3 | ||
| 3 | 2 | 4 |
| 3 | ||
| 4 | 2 | 4 |
| 3 | ||
Until now, no consensus has existed concerning the ideal location to select the AIF. In the literature, the need to find a compromise between the size of the arterial input and its location with regards to the tumor is often highlighted. Indeed, to avoid any partial volume effects which affect the ROI, the AIF must be extracted from an artery exhibiting a diameter larger than the intrinsic resolution of the functional imaging technique used[43,44]. On the other hand, an AIF measured too far from the tissue of interest causes additional delay and dispersion involving wrong estimation of quantitative parameters, in particular, an underestimation of the BF and an overestimation of MTT[8,44,45].
In the previous in vivo study, the arterial ROI was selected as close to the tumor as possible, whatever its size. In the second in vivo study (Table 1), the impact of the examination day on microvascularization parameters, giving preference in the selection of the arterial input to its size rather than its location, was evaluated.
The aim of the first in vitro study was to determine whether there is an impact on the microvascularization parameters while repositioning the US probe during experiments. Statistical analysis was based on comparing the variances of both the series of 10 acquisitions with the US probe unchanged and replaced. To do so, a bilateral Fisher test was performed. It consisted of calculating:
where s1 and s2 are the variances of each of the series. The determined value of F was compared to the F-value with (n1 - 1)(n2 - 1) degrees of freedom where n1 and n2 are the number of experiments performed in the two series of acquisitions (n1 = n2 = 10).
Results were significant for a P-value lower than 0.05.
The aim of the second in vitro study was to determine whether there was significant modification of the microvascularization parameters while the flow rate was modified. Thus, statistical analysis was based on comparing the means of microvascularization parameters for the three series of acquisitions. To do so, an analysis of variance (ANOVA) was performed between the 3 series of experimentations. Results were significant for a P-value lower than 0.05.
In vivo, statistical analyses were performed using the same multivariate ANOVA as the second in vitro study: the two parameters included in the process were the order of injection and the day of examination. Results were significant for P-value lower than 0.05.
Repositioning of the US probe:In vitro, no microvascularization parameters, whether semi-quantitative or quantitative, were significantly correlated with the repositioning of the probe. Table 2 provides P-values associated with each of the microvascularization parameters.
| In vitro results | In vivo results | |||
| Repositioning of the probe: Fisher’s test P values | Variation of the flow rate: ANOVA P values | Multiple injections: ANOVA P values | Day of examination: ANOVA P values | |
| Semi-quantitative microvascularization parameters | ||||
| PI | 0.077 | < 0.001 | 0.080 | < 0.001 |
| TPI | 0.302 | 0.004 | 0.595 | 0.641 |
| Slope of the WI | 0.059 | 0.006 | 0.141 | < 0.001 |
| MTT | 0.562 | < 0.001 | 0.885 | 0.016 |
| AUC | 0.731 | < 0.001 | 0.073 | 0.01 |
| AUWI | 0.276 | 0.024 | 0.060 | 0.001 |
| AUWO | 0.860 | 0.006 | 0.078 | 0.016 |
| Quantitative microvascularization parameters | ||||
| BF | 0.055 | 0.004 | 0.071 | 0.116 |
| BV | 0.063 | 0.053 | 0.429 | 0.015 |
| MTT | 0.131 | 0.649 | 0.126 | 0.645 |
Variation of the flow rate: No semi-quantitative microvascularization parameter was specific to the flow rate: none of the semi-quantitative parameters were correlated with the factor of interest. In addition, among the quantitative microvascularization parameter, only BF varied significantly with the flow rate: BF exhibited a P-value of 0.004. All the P-values are summarized in Table 2.
Multiple injections/day of examination: Multiple injections had no significant impact on the microvascularization parameters. Indeed, no parameter exhibited P-value lower than 0.05. All the P-values are summarized in Table 2.
On the other hand, to perform therapy evaluation, it is expected to obtain microvascularization parameters varying significantly with the tumor growth: any modification in microvascularization would be detected through the analysis of the microvascularization parameters. In the study, it was demonstrated that semi-quantitative microvascularization parameters were correlated with the tumor growth while among the quantitative microvascularization parameter, only the BV was.
The impact of the size of the arterial ROI on microvascularization parameters was evaluated according to the same protocol used in the previous in vivo study.
It was demonstrated that for larger arterial ROIs (from 1.17 ± 0.6 mm3 to 3.65 ± 0.3 mm3) (Table 3), all quantitative microvascularization parameters were correlated with the tumor growth. Table 4 summarizes all of the P-values associated with this study.
| Mean (mm2) | SD | Min | Max | |
| First in vivo study | 1.17 | 0.6 | 0.4 | 2.3 |
| Second in vivo study | 3.65 | 0.3 | 3.5 | 4.1 |
| Day of examination : P-ANOVA values | |
| Semi-quantitative microvascularization parameters | |
| PI | 0.027 |
| TPI | 0.010 |
| Slope of the WI | 0.070 |
| MTT | < 0.001 |
| AUC | 0.808 |
| AUWI | 0.562 |
| AUWO | 0.832 |
| Quantitative microvascularization parameters | |
| BF | < 0.001 |
| BV | < 0.001 |
| MTT | 0.033 |
In vitro, it was demonstrated that semi-quantitative and quantitative microvascularization parameters were not significantly correlated with the repositioning of the US probe before each acquisition. This is a promising result as in the context of therapy evaluation, the same target must be imaged during the entire treatment as evaluation is based on analyzing the microvascularization parameter evolution of a specific target through these exams. Results associated with the variation of the flow rates study were in agreement with the remarks mentioned before the experiments: all of the semi-quantitative microvascularization parameters were correlated with the flow rate while among the quantitative microvascularization parameters, only BF was specific to the flow rate.
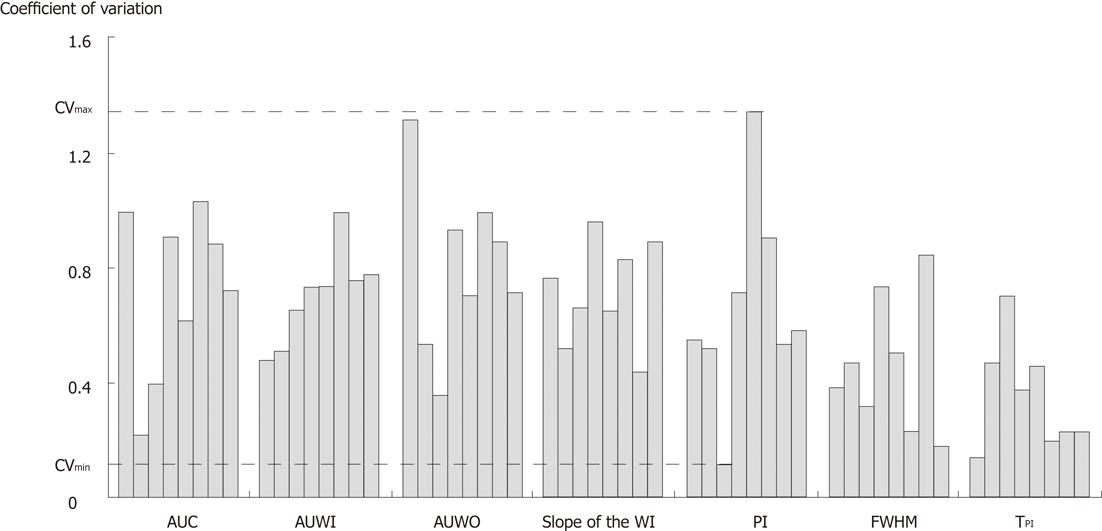
In vivo, the importance of the size of the arterial ROI was shown through the first study: the arterial ROI must be large enough to allow the deconvolution process to be relevant in the context of therapy evaluation. This result is consistent with previous studies evaluating the importance of the arterial ROI selection[9]. In particular, the partial volume effect on the AIF and its consequences on analyses have been reported in the literature[46]. Here, the first results demonstrated significant correlation between semi-quantitative and quantitative microvascularization parameters except for TPI, BF and MTT. In the context of experiments performed on mice, the TPI and MTT were extremely short (in the order of a few seconds). Consequently, such low values make it difficult to distinguish modification in times linked to modification in microvascularization because of high statistical fluctuations. Previous studies showed the existing link between semi-quantitative microvascularization parameters and microvascularization quantitative ones. In particular, Lassen et al[47] pointed out that the BF was related to the time to peak parameter. Thus, the same explanation may justify the absence of significance concerning this parameter. Further investigations have been performed to better interpret these first results. Coefficients of variation associated with the semi-quantitative microvascularization parameters extracted from the arterial ROI were evaluated (Figure 7). This evaluation provided a mean CV value of 0.63% ± 0.28% which is high compared to previous intra-operator findings (< 30%)[27,48-53]. Such a result may be due to the very small arterial ROI prone to partial volume effects which induce noise in the deconvolution process, leading to the loss of correlation between quantitative microvascularization parameters and the tumor growth. Finally, the last experiments performed in vivo demonstrated significant correlation between quantitative microvascularization parameters and tumor growth for larger arterial ROIs. Identical results were obtained in the case of the semi-quantitative microvascularization parameters except for the areas where P > 0.05. One explanation may involve the high inter-subject variability noticed in the study. Thus, this high variability implies fluctuations in semi-quantitative microvascularization mean values that hide modifications in BV. On the other hand, as deconvolution allows freeing oneself from inter-subject variability, results associated with the quantitative microvascularization parameters were significant.
In vitro, both the fluid and the pipes used did not exhibit the same ultrasound properties as blood and vessels, respectively[54-56]. In vivo, quantification may have been difficult to perform because of the mouse’s respiratory and cardiac movements which lead to instability in ROI locations. Another limitation involved the stability of the ultrasound contrast agent. Indeed, even if each experiment duration was less than 2 h on account of the stability of SonoVue® (6 h after its reconstitution as described by Schneider[14]), recent studies have reported a significant incidence of spontaneous gas diffusion phenomena on temporal evolution of contrast microbubble size[57-59]. This study did not take into account gas diffusion phenomena occurring for 2 h from initial formation of contrast agent. This assumption might have impacted the final results.
The last in vivo study presented in the article was a preliminary study investigating the best selection of the arterial input. Further analysis must be performed to improve such a difficult selection. Indeed, Calamante et al[60] have demonstrated that an arterial ROI not selected directly at the entrance of the tumor may lead to an underestimation of the BF and an overestimation of the MTT. Such wrong estimations were due to the delay and dispersion occurring between the site of injection and the arterial ROI. On the other hand, it may sometimes be difficult to access an arterial ROI large enough and in the close vicinity of the tumor leading to partial volume effect influencing the results[16]. Thus, a trade-off between size and location of the arterial ROI must be determined. Additional corrections to the deconvolution process may be required, as mentioned by van Osch et al[61], to ensure microvascularization parameters are accurately evaluated. Modeling of the AIF might also be necessary for the deconvolution process to be applicable in all cases [62]. Finally, a proof of concept may be interesting in evaluating the robustness of the deconvolution method in a clinical context: additional studies may be performed to definitely validate the deconvolution process.
As already mentioned, the current DCE-US method does not take into account the arterial input, which greatly depends on patient physiological conditions as well as on which contrast agent is injected. Parameters determined according to the method are graphically extracted from the tumor TIC and if studies demonstrated correlation between some of them and the physiological parameters, absolute quantification is not possible through that method. On the other hand, the deconvolution process allows absolute quantification providing clinically meaningful microvascularization parameters.
In addition, results presented in the article support the possible use of the deconvolution method in a similar context to the current DCE-US, as the last in vivo study exhibited identical results whatever the quantification process applied.
In the study, sources of variation that impact the microvascularization parameters measured using DCE-US were evaluated, both taking and not taking into account the arterial input.
Throughout all of the experiments, two main conclusions can be drawn. First of all, it is necessary to pay attention to the selection of the arterial ROI as a small ROI may be at the origin of noise in the deconvolution method leading to its impossible use as a quantification tool. Finally, when the arterial ROI is large enough, deconvolution exhibits similar results as the current DCE-US. Such results suggest that the deconvolution method may be useful in a similar context with the advantage of providing absolute quantification of the microvascularization.
The early and functional evaluation of new treatments in oncology, such as anti-angiogenic therapies, is a main goal. Today, dynamic contrast-enhanced ultrasonography (DCE-US) is commonly recognized as a functional imaging technique. It is a highly available and sensitive modality which allows early predicting of the tumor response to treatments involving changes in microvascularity before any morphological ones occur.
Microbubble contrast agents for DCE-US have developed over the past 10 years and are currently approved in Europe, Asia and Canada. As it provides real time imaging, is widely available, non-ionizing, and low cost, ultrasound imaging is recognized as an ideal modality for angiogenesis. Recently, a deconvolution method dedicated to DCE-US imaging was developed to take into account the arterial input function which provides an absolute evaluation of microvasculature parameters. In this study, the authors aim to evaluate the sources of variation influencing the microvascularization parameters measured using the routinely used DCE-US imaging method and the deconvolution method.
DCE-US is supported by the French National Cancer Institute which is currently studying the technique in several pathologies to establish the optimal microvascularization parameters and timing for quantitative anticancer efficacy assessments. This study firstly suggests the selection of the arterial region of interest (ROI) is important when using the deconvolution process, and secondly, when using the appropriate arterial ROI, analyses using deconvolution DCE-US and routine DCE-US exhibit similar results.
By comparing the deconvolution DCE-US and the routine DCE-US methods, this study may confirm the use of the deconvolution method in a similar context to the current routinely used DCE-US method. The deconvolution method exhibits the additional advantage of allowing absolute microvascularization quantification.
DCE-US imaging involves the use of microbubble contrast agents and specialized imaging techniques to evaluate semi-quantitative microvascularization parameters. The deconvolution method is based on the Tikhonov regularization and aims to perform an absolute quantification of the microvascularization.
This is part of an enthusiastic series of works, and is well organized. Other articles of the same authors are open. The end point of this study is clear: To evaluate the sources of variation influencing the microvascularization parameters measured by DCE-US. They performed an intensive experimental study using an in vitro and an in vivo model, and the results were clearly described. Even though a clinical situation may be further complicated, their study showed that their semi-quantitative microvascularization parameters might reflect reliable information of the nature of each parameter.
Peer reviewers: Zhonghua Sun, MD, PhD, Associate Professor, Discipline of Medical Imaging, Department of Imaging and Applied Physics, Curtin University of Technology, GPO Box U 1987, Perth, Western Australia 6845, Australia; Dr. Kazushi Kishi, Department of Radiology, Wakayama Medical University, Kimiidera 811-1, Wakayama City 641-8510, Japan
S- Editor Cheng JX L- Editor Logan S E- Editor Xiong L
| 1. | Lanza GM, Caruthers SD, Winter PM, Hughes MS, Schmieder AH, Hu G, Wickline SA. Angiogenesis imaging with vascular-constrained particles: the why and how. Eur J Nucl Med Mol Imaging. 2010;37 Suppl 1:S114-S126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 2. | Eisenbrey JR, Forsberg F. Contrast-enhanced ultrasound for molecular imaging of angiogenesis. Eur J Nucl Med Mol Imaging. 2010;37 Suppl 1:S138-S146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 45] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 3. | Kalva SP, Namasivayam S, Vasuedo Sahani D. Imaging Angiogenesis. Cancer Drug Discovery and Development: Antiangiogenic Agents in Cancer Therapy. Totowa, NJ: Humana Press 2008; 189-203. |
| 4. | Lassau N, Chami L, Pronneau P. Imagerie de contraste ultrasonore pour l'valuation prcoce des thrapeutiques cibles. Echographie de contraste. Paris: Springer 2007; 81-86. |
| 5. | Miller JC, Pien HH, Sahani D, Sorensen AG, Thrall JH. Imaging angiogenesis: applications and potential for drug development. J Natl Cancer Inst. 2005;97:172-187. [PubMed] |
| 6. | Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000;92:205-216. [PubMed] |
| 7. | Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228-247. [PubMed] |
| 8. | de Marco G, Dassonvalle P, Henry-Feugeas MC, Onen F, Idy-Peretti I. Cerebral perfusion: dynamic suceptibility contrast MR imaging. Part 2: vascular models and data extraction. Mdecine nuclaire. 2004;28:35-48. |
| 9. | Lythgoe DJ, Ostergaard L, William SC, Cluckie A, Buxton-Thomas M, Simmons A, Markus HS. Quantitative perfusion imaging in carotid artery stenosis using dynamic susceptibility contrast-enhanced magnetic resonance imaging. Magn Reson Imaging. 2000;18:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 76] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 10. | Rumboldt Z, Al-Okaili R, Deveikis JP. Perfusion CT for head and neck tumors: pilot study. AJNR Am J Neuroradiol. 2005;26:1178-1185. [PubMed] |
| 11. | Li PC, Yeh CK, Wang SW. Time-intensity-based volumetric flow measurements: an in vitro study. Ultrasound Med Biol. 2002;28:349-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Gauthier M, Tabarout F, Leguerney I, Polrot M, Pitre S, Peronneau P, Lassau N. Assessment of quantitative perfusion parameters by dynamic contrast-enhanced sonography using a deconvolution method: an in vitro and in vivo study. J Ultrasound Med. 2012;31:595-608. [PubMed] |
| 13. | Gauthier M, Tabarout F, Leguerney I, Thalmensi J, Roche A, Lassau N. Assessment of quantitative tumor perfusion parameters by dynamic contrast-enhanced ultrasonography using a deconvolution method: an in vitro and in vivo study. J Ultrasound Med. 2012;31:595-608. [PubMed] |
| 14. | Schneider M. Characteristics of SonoVuetrade mark. Echocardiography. 1999;16:743-746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 183] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 15. | Greis C. Technology overview: SonoVue (Bracco, Milan). Eur Radiol. 2004;14 Suppl 8:P11-P15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 100] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 16. | Quaia E, Calliada F, Bertolotto M, Rossi S, Garioni L, Rosa L, Pozzi-Mucelli R. Characterization of focal liver lesions with contrast-specific US modes and a sulfur hexafluoride-filled microbubble contrast agent: diagnostic performance and confidence. Radiology. 2004;232:420-430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 355] [Cited by in RCA: 338] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 17. | von Herbay A, Vogt C, Willers R, Häussinger D. Real-time imaging with the sonographic contrast agent SonoVue: differentiation between benign and malignant hepatic lesions. J Ultrasound Med. 2004;23:1557-1568. [PubMed] [DOI] [Full Text] |
| 18. | Hamilton WF, Moore JW Kinsman MJ. Studies on the circulation: IV. Further analysis of the injection method, and of changes in hemodynamics under physiological and pathological conditions. Am J Physiol. 1929;99:534-551. |
| 19. | Kinsman MJ, Moore JW Hamilton WF. Studies on the circulation: I. Injection methods: physical and mathematical considerations. Am J Physiol. 1929;89:322-330. |
| 20. | Mischi M, Del Prete Z, Korsten HHM. Indicator dilution techniques in cardiovascular quantification. Biomechanical Systems Technology: Cardiovascular Systems. Singapore: World Scientific Publishing Company 2007; 89-156. |
| 21. | Li PC, Yang MJ. Transfer function analysis of ultrasonic time-intensity measurements. Ultrasound Med Biol. 2003;29:1493-1500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Lampaskis M, Averkiou M. Investigation of the relationship of nonlinear backscattered ultrasound intensity with microbubble concentration at low MI. Ultrasound Med Biol. 2010;36:306-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 63] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 23. | Burns PN, Wilson SR, Simpson DH. Pulse inversion imaging of liver blood flow: improved method for characterizing focal masses with microbubble contrast. Invest Radiol. 2000;35:58-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 236] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 24. | Whittingham TA. Contrast-specific imaging techniques: technical perspective. Contrast media in ultrasonography: basic principles and clinical applications. Berlin, Heidelberg, New York: Springer 2005; 43-70. |
| 25. | Verbeek XA, Willigers JM, Prinzen FW, Peschar M, Ledoux LA, Hoeks AP. High-resolution functional imaging with ultrasound contrast agents based on RF processing in an in vivo kidney experiment. Ultrasound Med Biol. 2001;27:223-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 26. | Peronneau P, Lassau N, Leguerney I, Roche A, Cosgrove D. Contrast ultrasonography: necessity of linear data processing for the quantification of tumor vascularization. Ultraschall Med. 2010;31:370-378. [PubMed] |
| 27. | Gauthier M, Leguerney I, Thalmensi J, Chebil M, Parisot S, Peronneau P, Roche A, Lassau N. Estimation of intra-operator variability in perfusion parameter measurements using DCE-US. World J Radiol. 2011;3:70-81. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 29] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 28. | Lavisse S, Lejeune P, Rouffiac V, Elie N, Bribes E, Demers B, Vrignaud P, Bissery MC, Brulé A, Koscielny S. Early quantitative evaluation of a tumor vasculature disruptive agent AVE8062 using dynamic contrast-enhanced ultrasonography. Invest Radiol. 2008;43:100-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 67] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 29. | Xiong Z, Cheng Z, Zhang X, Patel M, Wu JC, Gambhir SS, Chen X. Imaging chemically modified adenovirus for targeting tumors expressing integrin alphavbeta3 in living mice with mutant herpes simplex virus type 1 thymidine kinase PET reporter gene. J Nucl Med. 2006;47:130-139. [PubMed] |
| 30. | Calamante F, Gadian DG, Connelly A. Delay and dispersion effects in dynamic susceptibility contrast MRI: simulations using singular value decomposition. Magn Reson Med. 2000;44:466-473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 31. | Willats L, Connelly A, Calamante F. Improved deconvolution of perfusion MRI data in the presence of bolus delay and dispersion. Magn Reson Med. 2006;56:146-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 42] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 32. | Lassau N, Koscielny S, Albiges L, Chami L, Benatsou B, Chebil M, Roche A, Escudier BJ. Metastatic renal cell carcinoma treated with sunitinib: early evaluation of treatment response using dynamic contrast-enhanced ultrasonography. Clin Cancer Res. 2010;16:1216-1225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 137] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 33. | Lassau N, Koscielny S, Chebil M, Chami L, Bendjilali R, Roche A, Escudier B, Le Cesne A Soria J. Functional imaging using DCE-US: Which parameter for the early evaluation of antiangiogenic therapies [abstract]. J Clin Oncol. 2009;27:3524. |
| 34. | Lassau N, Lacroix J, Aziza R, Vilgrain V, Taeb S Koscielny S. French Multicentric Prospective Evaluation of Dynamic Contrast-enhanced Ultrasound (DCE-US) for the Evaluation of Antiangiogenic Treatments. Chicago: Radiological Society of North America 2009; . |
| 35. | Ugolini P, Delouche A, Herment A, Diebold B. In vitro flow quantification with contrast power Doppler imaging. Ultrasound Med Biol. 2000;26:113-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 36. | Thijssen JM, de Korte CL. Modeling ultrasound contrast measurement of blood flow and perfusion in biological tissue. Ultrasound Med Biol. 2005;31:279-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 37. | Krix M, Kiessling F, Farhan N, Schmidt K, Hoffend J, Delorme S. A multivessel model describing replenishment kinetics of ultrasound contrast agent for quantification of tissue perfusion. Ultrasound Med Biol. 2003;29:1421-1430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 67] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 38. | Vicenzini E, Delfini R, Magri F, Puccinelli F, Altieri M, Santoro A, Giannoni MF, Bozzao L, Di Piero V, Lenzi GL. Semiquantitative human cerebral perfusion assessment with ultrasound in brain space-occupying lesions: preliminary data. J Ultrasound Med. 2008;27:685-692. [PubMed] |
| 39. | Workman P, Aboagye EO, Balkwill F, Balmain A, Bruder G, Chaplin DJ, Double JA, Everitt J, Farningham DA, Glennie MJ. Guidelines for the welfare and use of animals in cancer research. Br J Cancer. 2010;102:1555-1577. [PubMed] |
| 40. | Kwee JK, Mitidieri E, Affonso OR. Lowered superoxide dismutase in highly metastatic B16 melanoma cells. Cancer Lett. 1991;57:199-202. [PubMed] |
| 41. | Ohira T, Ohe Y, Heike Y, Podack ER, Olsen KJ, Nishio K, Nishio M, Miyahara Y, Funayama Y, Ogasawara H. In vitro and in vivo growth of B16F10 melanoma cells transfected with interleukin-4 cDNA and gene therapy with the transfectant. J Cancer Res Clin Oncol. 1994;120:631-635. [PubMed] |
| 42. | Yerlikaya A, Erin N. Differential sensitivity of breast cancer and melanoma cells to proteasome inhibitor Velcade. Int J Mol Med. 2008;22:817-823. [PubMed] |
| 43. | Lüdemann L, Wust P, Gellermann J. Perfusion measurement using DCE-MRI: implications for hyperthermia. Int J Hyperthermia. 2008;24:91-96. [PubMed] |
| 44. | Calamante F, Gadian DG, Connelly A. Quantification of perfusion using bolus tracking magnetic resonance imaging in stroke: assumptions, limitations, and potential implications for clinical use. Stroke. 2002;33:1146-1151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 206] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 45. | Duhamel G, Schlaug G, Alsop DC. Measurement of arterial input functions for dynamic susceptibility contrast magnetic resonance imaging using echoplanar images: comparison of physical simulations with in vivo results. Magn Reson Med. 2006;55:514-523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 46. | Hansen AE, Pedersen H, Rostrup E, Larsson HB. Partial volume effect (PVE) on the arterial input function (AIF) in T1-weighted perfusion imaging and limitations of the multiplicative rescaling approach. Magn Reson Med. 2009;62:1055-1059. [PubMed] |
| 47. | Lassen N, Perl W. Tracer Kinetic Methods in Medical Physiology. New York: Raven; 1979; . |
| 48. | Cenic A, Nabavi DG, Craen RA, Gelb AW, Lee TY. Dynamic CT measurement of cerebral blood flow: a validation study. AJNR Am J Neuroradiol. 1999;20:63-73. [PubMed] |
| 49. | Evelhoch JL, LoRusso PM, He Z, DelProposto Z, Polin L, Corbett TH, Langmuir P, Wheeler C, Stone A, Leadbetter J. Magnetic resonance imaging measurements of the response of murine and human tumors to the vascular-targeting agent ZD6126. Clin Cancer Res. 2004;10:3650-3657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 128] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 50. | Groves AM, Goh V, Rajasekharan S, Kayani I, Endozo R, Dickson JC, Menezes LJ, Shastry M, Habib SB, Ell PJ. CT coronary angiography: quantitative assessment of myocardial perfusion using test bolus data-initial experience. Eur Radiol. 2008;18:2155-2163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 51. | Marcus CD, Ladam-Marcus V, Cucu C, Bouché O, Lucas L, Hoeffel C. Imaging techniques to evaluate the response to treatment in oncology: current standards and perspectives. Crit Rev Oncol Hematol. 2009;72:217-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 104] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 52. | Morgan B, Utting JF, Higginson A, Thomas AL, Steward WP, Horsfield MA. A simple, reproducible method for monitoring the treatment of tumours using dynamic contrast-enhanced MR imaging. Br J Cancer. 2006;94:1420-1427. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 52] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 53. | Ng CS, Raunig DL, Jackson EF, Ashton EA, Kelcz F, Kim KB, Kurzrock R, McShane TM. Reproducibility of perfusion parameters in dynamic contrast-enhanced MRI of lung and liver tumors: effect on estimates of patient sample size in clinical trials and on individual patient responses. AJR Am J Roentgenol. 2010;194:W134-W140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 56] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 54. | Gupta LC, Sahu UC. Diagnostic Ultrasound. New Delhi: Jay- pee Brothers Publishers; 2007; . |
| 55. | Hedrick W, Hykes D Starchman D. Ultrasound physics and instrumentation. Saint-Louis, MO: Mosby 1995; . |
| 56. | Goldstein A, Powis RL. Medical Ultrasonic Diagnostics. Ultrasonic Instruments and Devices: Reference for Modern Instrumentation, Techniques, and Technology. San Diego, CA: Academic Press 1999; 46-193. |
| 57. | Sarkar K, Katiyar A, Jain P. Growth and dissolution of an encapsulated contrast microbubble: effects of encapsulation permeability. Ultrasound Med Biol. 2009;35:1385-1396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 117] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 58. | Kwan JJ, Borden MA. Microbubble dissolution in a multigas environment. Langmuir. 2010;26:6542-6548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 109] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 59. | Casciaro S, Palmizio Errico R, Conversano F, Demitri C, Distante A. Experimental investigations of nonlinearities and destruction mechanisms of an experimental phospholipid-based ultrasound contrast agent. Invest Radiol. 2007;42:95-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 30] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 60. | Calamante F. Bolus dispersion issues related to the quantification of perfusion MRI data. J Magn Reson Imaging. 2005;22:718-722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 63] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 61. | van Osch MJ, Vonken EJ, Bakker CJ, Viergever MA. Correcting partial volume artifacts of the arterial input function in quantitative cerebral perfusion MRI. Magn Reson Med. 2001;45:477-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 62. | Maxwell RJ, Wilson J, Prise VE, Vojnovic B, Rustin GJ, Lodge MA, Tozer GM. Evaluation of the anti-vascular effects of combretastatin in rodent tumours by dynamic contrast enhanced MRI. NMR Biomed. 2002;15:89-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 84] [Article Influence: 3.7] [Reference Citation Analysis (0)] |