Published online Aug 28, 2025. doi: 10.4329/wjr.v17.i8.111065
Revised: July 4, 2025
Accepted: August 5, 2025
Published online: August 28, 2025
Processing time: 67 Days and 6.8 Hours
Anoxic brain injury is a potentially lethal condition characterized by cerebral hy
To explore the relationship between regional ASL perfusion patterns and clinical outcomes following cardiac arrest.
We performed a retrospective review to identify patients with clinical suspicion of anoxic brain injury who underwent MRI within 15 days of cardiac arrest. Receiver operator characteristic (ROC) analysis and univariate logistic regression were used to evaluate associations between ASL perfusion scores, DWI signal intensity, and the following clinical features: (1) Myoclonus status epilepticus (MSE) within 24 hours; (2) Absent extensor or motor reflexes (EMR) at day 3 post-arrest; and (3) Absent brainstem reflexes (BSR) within 15 days.
Twenty-eight patients met inclusion criteria. Increased ASL signal in the left occipital lobe was significantly associated with MSE (P = 0.038), while a trend was observed between right frontal ASL signal and EMR (P = 0.078). ROC analysis showed that ASL scores ≥ 7 were associated with higher odds of absent BSR (OR 2.14, P = 0.53), though this did not reach statistical significance. DWI signal intensity did not show significant associations with clinical outcomes. The overall discriminatory performance of ASL for predicting outcomes was limited (AUC ≈ 0.52).
This exploratory study suggests that regional ASL hyperperfusion, particularly in the left occipital and right frontal lobes, may be associated with adverse clinical signs following cardiac arrest. However, most findings did not reach statistical significance, and the study was underpowered to detect small-to-moderate effects. These preliminary results should be interpreted with caution and considered hypothesis-generating. Larger, prospective studies are warranted to clarify the prognostic value of ASL perfusion imaging in anoxic brain injury.
Core Tip: Anoxic brain injury is a devastating clinical entity characterized by severe cerebral hypoperfusion with resultant neuronal cell death. Arterial spin-labeling (ASL) perfusion and diffusion-weighted imaging magnetic resonance imaging has recently been used to detect global and focal cerebral ischemic changes that may play a role in the assessment of anoxic brain injury. In this retrospective cohort study, we found that elevated ASL signal involving the left occipital lobe and right frontal lobe is predictive of poor outcomes, suggesting that ASL signal may have important prognostic implications in the setting of anoxic brain injury.
- Citation: Beutler BD, Antwi-Amoabeng D, Weinert D, Shah I, Ulanja MB, Moody AE, Lei X, Lerner A, Shiroishi MS, Assadsangabi R. Prognostic value of arterial spin-labeling perfusion in anoxic brain injury: A retrospective cohort study. World J Radiol 2025; 17(8): 111065
- URL: https://www.wjgnet.com/1949-8470/full/v17/i8/111065.htm
- DOI: https://dx.doi.org/10.4329/wjr.v17.i8.111065
Anoxic brain injury is a dynamic process characterized by cell swelling and oncotic cell death, which manifests as diffusion restriction on magnetic resonance imaging (MRI), and a later period of reperfusion injury and disruption of the blood-brain barrier, which may present as superphysiologic cerebral blood flow on arterial spin-labeling (ASL) perfusion sequences. The current prognostic tools and clinical prediction models rely primarily on correlation of patient history, neurological examination, and electroencephalography. However, as of this writing, imaging plays a limited role in predicting outcomes and likelihood of functional recovery.
There is emerging evidence that diffusion-weighted imaging (DWI) and ASL sequences can depict underlying pathophysiological processes that have important prognostic implications in the setting of anoxic brain injury. A retrospective study by Pollock et al[1] established that the presence of increased signal on both DWI and ASL sequences was predictive of unfavorable patient outcomes. In addition, although the sample size was relatively small, some patients who were found to have superphysiologic hyperperfusion on ASL sequences without corresponding diffusion restriction demonstrated relatively favorable outcomes with at least partial neurological recovery[1]. A subsequent study by Li et al[2] showed a positive relationship between ASL and DWI signal intensity. Authors hypothesized that anoxia manifests with regional diffusion restriction, likely related to cell swelling, and corresponding regional hyperperfusion, reflecting disruption of the blood-brain barrier and loss of autoregulation.
We aimed to evaluate the relationship between the pattern of diffusion restriction, cerebral hyperperfusion, and patient outcomes in the setting of anoxic brain injury. The identification of imaging findings predictive of patient outcomes could help improve the accuracy and reliability of prognostication and ultimately inform management.
We conducted a retrospective review of the electronic health record at our institution to identify participants with clinical suspicion of anoxic brain injury who underwent at least one MRI study within 15 days of cardiac arrest and subsequent return of spontaneous circulation (ROSC). The study period spanned from September 1, 2020 to July 1, 2022.
The study was approved by the Institutional Review Board of the University of Southern California, Keck School of Medicine.
All MRI studies were performed using a 1.5T General Electric scanner. The imaging protocols included ASL and DWI sequences. The ASL imaging parameters were as follows: Flip angle (FA) of 155 degrees, repetition time of 4885 ms, and echo time of 10.47 ms. For DWI, the parameters included a FA of 90 degrees and a b-value of 1000 s/mm².
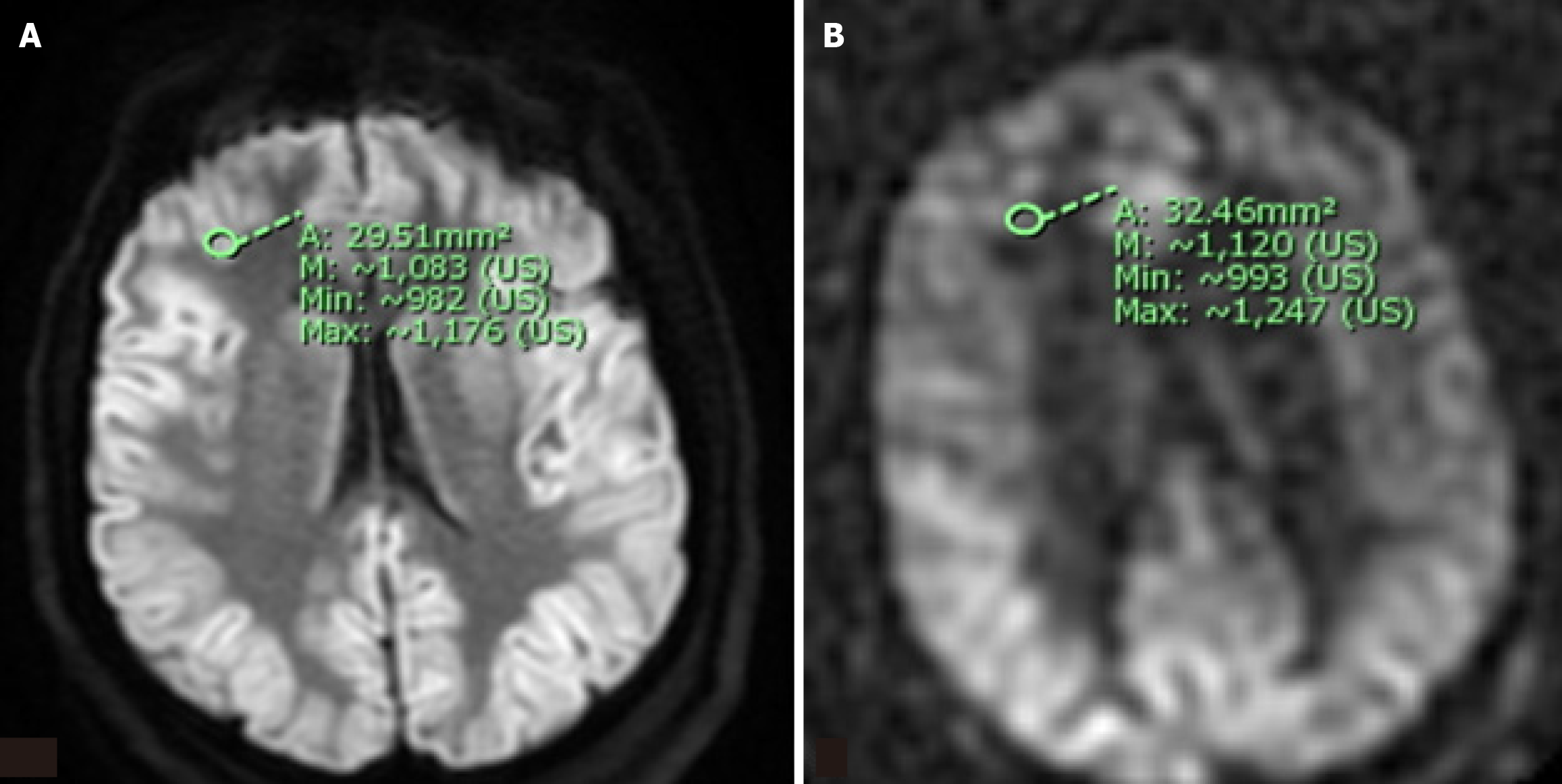
ASL perfusion and DWI signal intensity was measured in twelve brain regions (bilateral frontal, parietal, temporal, and occipital lobes; thalami; and basal ganglia) by two independent observers. The observers used a region of interest (ROI) approach, selecting approximately 30 mm² areas for analysis (Figure 1). The mean and maximum signal intensity within each ROI was recorded and normalized using the signal intensity within the ipsilateral cerebellar hemisphere as an internal reference value.
Relevant clinical data were recorded for each patient, including the time from ROSC to the MRI, discharge disposition (e.g., discharged home, transferred to a rehabilitation facility, or deceased), and changes in Glasgow Coma Scale (GCS) score from ROSC to disposition.
In addition, we assessed the presence of three established clinical parameters associated with unfavorable outcomes in anoxic brain injury.
Absent brainstem reflexes: Evaluated at any point within the 15 days following cardiac arrest.
Myoclonus status epilepticus: Assessed within the first day of cardiac arrest.
Absent pupil or corneal reflexes or absent extensor or motor reflexes: Assessed on day 3 post-arrest.
The mean and maximum ASL perfusion and DWI signal intensity was quantitatively assessed at 12 different brain regions using a 30 mm² ROI. A normalized scale was then constructed using the signal intensity within the ipsilateral cerebellar hemisphere as an internal reference value. Scores ranged from 0-12 based on the signal intensity within the selected brain region relative to the cerebellar hemisphere; a score of 0 represented ASL perfusion or DWI signal intensity equal to or lower than that of the ipsilateral cerebellar hemisphere while a score of 12 indicated ASL perfusion or DWI signal intensity markedly greater than that of the cerebellar hemisphere. Receiver operator characteristics (ROC) analysis was employed to evaluate the performance of ASL perfusion scores in predicting clinical outcomes. The optimal cutoff point was determined based on sensitivity and specificity analyses.
Univariate logistic regression was performed to assess the odds of the selected clinical parameters for scores above the optimal cutoff point. Confidence intervals (CI) and P values were calculated to provide statistical significance of the findings. Inter-rater reliability for ASL perfusion and DWI measurements was assessed using the intraclass correlation coefficient (ICC). Due to the small sample size and limited outcome events, multivariable modeling was not performed to adjust for potential confounders such as time from arrest to MRI or patient comorbidities. Univariate logistic regression was used to explore associations, with the recognition that confounding effects may influence results.
ROC curve analysis was used to evaluate the discriminative ability of ASL perfusion scores and DWI signal intensity for predicting unfavorable clinical outcomes, including absent brainstem reflexes (BSR) and myoclonus status epilepticus (MSE). Optimal cutoff values were determined by maximizing Youden’s index.
Univariate logistic regression was employed to estimate odds ratios for clinical outcomes above or below the ASL cutoff scores. Given the small sample size and limited number of outcome events, multivariable models were not constructed to avoid model overfitting. As such, no adjustment for potential confounders [e.g., age, time to MRI, targeted temperature management (TTM)] was performed. This approach was intended to preserve statistical stability and should be interpreted as exploratory.
All analyses were performed using complete-case data; no imputation methods were applied. Clinical and imaging variables were recorded prospectively and reviewed retrospectively to minimize missingness, and data were complete for all primary variables used in ROC and regression analyses.
A total of 28 patients (19 males, 9 females; mean age 58, range 26-84) were included in the analysis. The time between ROSC and MRI acquisition ranged from 3 to 16 days (mean time to acquisition: 6.2 days; median time to acquisition: 5 days). Electroencephalographic (EEG) findings included generalized burst suppression, triphasic waves, focal epilepitform discharges, generalized polyspike and slow wave epileptiform discharges, and most commonly, nonspecific diffuse slowing.
The ROC analysis showed that ASL scores ≥ 7 were associated with a non-significant increase in the odds of absent BSR (OR 2.14, P = 0.53), with high specificity (100%) and negative predictive value (85.7%). However, this association did not reach statistical significance and should be interpreted as exploratory. In addition, ASL scores ≥ 7 showed a non-significant trend toward increased odds of MSE (OR 1.19, P = 0.82), with a sensitivity of 100% and a positive predictive value of 57.1%. This trend did not achieve statistical significance and is considered hypothesis-generating. The area under the ROC curve was 0.52 for ASL perfusion in predicting absent BSR and 0.52 for predicting MSE-values indicating no meaningful discriminatory power. These results suggest that the ASL score cutoff of ≥ 7 Lacks prognostic utility in this sample (Table 1).
| Variable/region | Finding | P value |
| Right occipital lobe | ↑ ASL in late subacute scans | < 0.05 |
| Left parietal lobe | ↑ ASL in those with absent BSR | < 0.05 |
| Occipital/temporal lobes | ↓ ASL in those with absent EMR | < 0.05 |
| ASL-DWI correlation (frontal, occipital, etc.) | Positive correlation | < 0.05 |
The ICC for raw ASL was 0.63, indicating moderate reliability between observers, while normalized ASL showed poor reliability (ICC 0.21). For DWI, the ICC was 0.86 for raw data and 0.74 for normalized data, reflecting high inter-observer agreement.
The low inter-rater reliability for normalized ASL signal intensity is likely attributable to the variability introduced by the normalization strategy. We selected the ipsilateral cerebellar hemisphere as an internal reference region in an effort to control for patient-level and scanner-level differences in perfusion signal intensity, a method previously described in cerebral perfusion imaging literature. However, this reference region may itself be affected in global hypoperfusion states, such as post-anoxic injury, introducing inconsistency across measurements. Additionally, while ROI placement was guided by consistent anatomical landmarks and involved approximately 30 mm² areas, subtle differences in manual placement can disproportionately impact normalized values-especially in cortical regions with low perfusion or heterogeneous signal.
The Pearson correlation between ASL and DWI revealed a moderate positive relationship in the frontal lobe (0.53-0.68,
ASL signal intensity analysis related to clinical variables revealed higher signal intensity in the right occipital lobe for late subacute scans compared to early subacute scans (P < 0.05). There were no significant differences in ASL signal intensity between patients with and without changes in GCS (n = 10 vs 18), between those transferred and those who died in the hospital (n = 9 vs 19), or between those with and without MSE within the first day of arrest (n = 16 vs 12). However, patients with absent BSRs at any point within the 15 days following survived cardiac arrest (no vs yes, n = 24 vs 4) had higher mean intensity in the left parietal lobe (P < 0.05).
Patients with absent pupil or corneal reflexes or absent extensor or motor reflexes (EMR) at day 3 post-arrest (no vs yes, n = 9 vs 18) had lower intensity in occipital lobes and mean intensity in temporal lobes (P < 0.05).
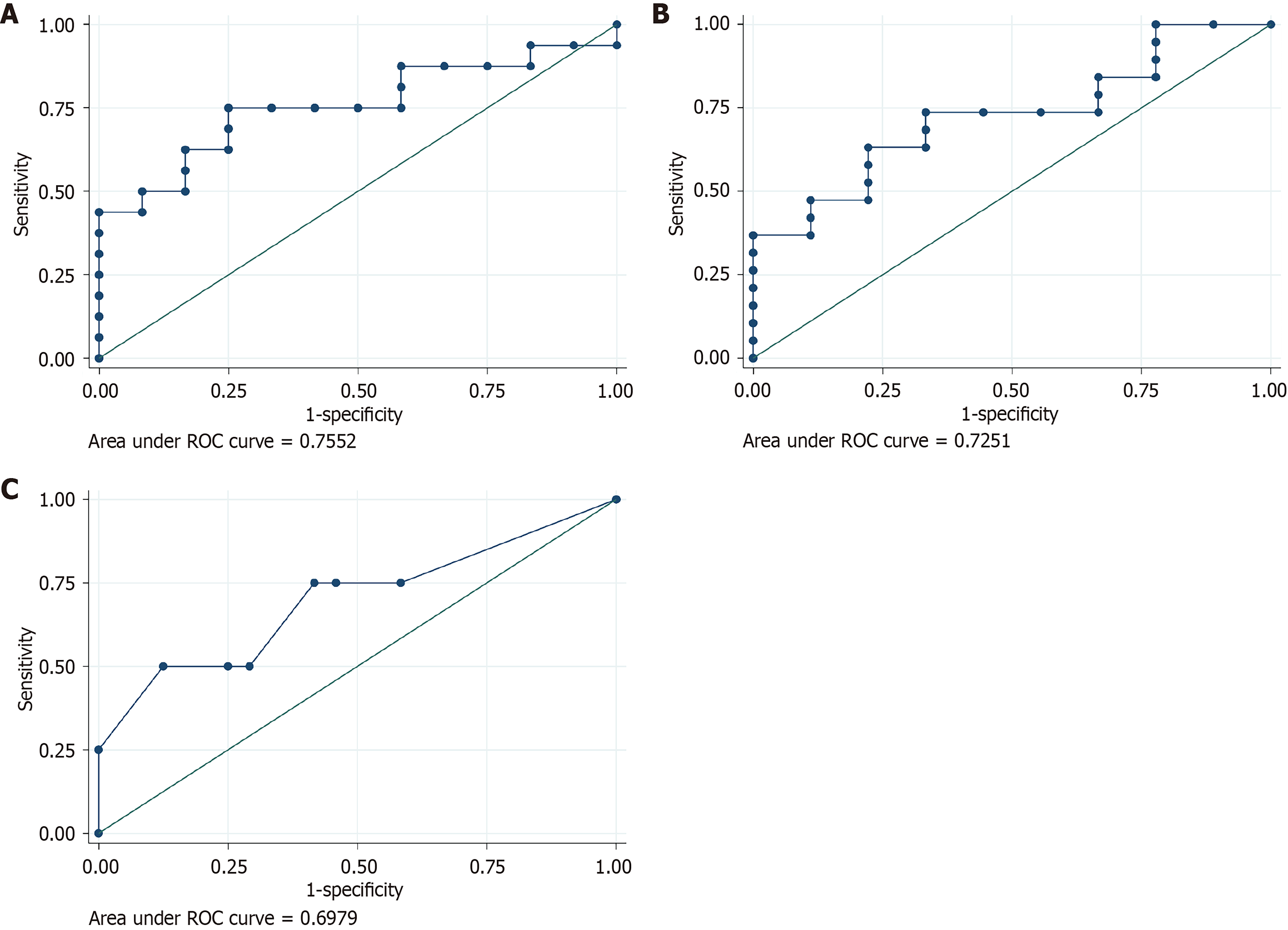
The mean ASL perfusion signal intensity in the left frontal lobe showed good performance for predicting MSE (AUC = 0.7552). In addition, the mean ASL perfusion signal intensity in the right frontal lobe demonstrated good performance for predicting absent BSR (AUC = 0.7251) (Figure 3A and B). The relationship between ASL perfusion signal intensity and outcomes is summarized in Table 1.
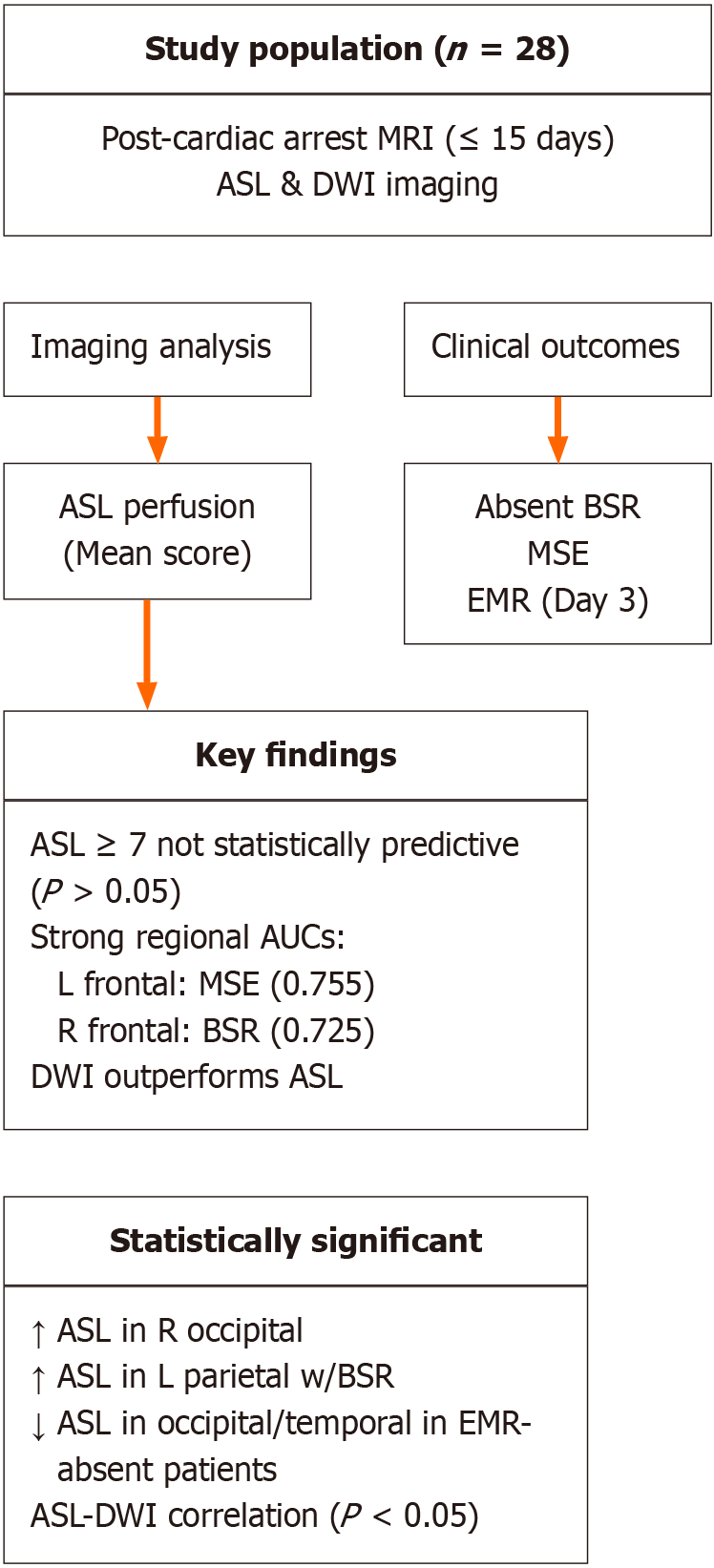
The mean DWI signal intensity averaged across all 12 brain regions showed better performance for predicting absent BSR as compared to ASL perfusion signal intensity (Figure 3C). A detailed visual summary of the study population, methodology, and statistically significant findings is shown in Figure 4.
A post hoc power analysis was conducted to evaluate the statistical power of the study given the sample size of 28 patients. For the ROC analysis evaluating the predictive performance of ASL perfusion in the left frontal lobe for MSE (AUC = 0.76), the estimated power was approximately 70%-75% at a significance level of 0.05. In contrast, the analysis predicting absent BSRs using right frontal ASL perfusion (AUC = 0.73) had lower power (approximately 45%-55%) due to the small number of patients with this outcome (n = 4). These findings suggest moderate power to detect associations in some comparisons, but limited power in others, particularly those with low event rates.
Although the criteria for MSE, EMR, and BSR were defined using established prognostic standards, we acknowledge that clinical heterogeneity across patients may influence outcome interpretation. Factors such as targeted TTM, sedative use, and EEG patterns can affect the timing and reliability of these prognostic signs.
To explore these effects, subgroup analyses were performed. Among the 28 patients, 14 (50%) received TTM following cardiac arrest. Within this subgroup, no statistically significant differences in ASL perfusion scores were observed compared to non-TTM patients, though the small sample size limits interpretability. Similarly, EEG patterns were available in 24 patients. Of these, 8 exhibited generalized burst suppression, 5 had epileptiform discharges (polyspike or focal), and 11 showed diffuse slowing. ASL perfusion scores were descriptively higher in patients with epileptiform activity, but these differences did not reach statistical significance. These findings highlight the complexity of prognosticating anoxic brain injury and underscore the need to account for clinical modifiers such as TTM and EEG background. The lack of significant differences may reflect limited power rather than true absence of effect. Larger, prospective studies with stratified enrollment are warranted to better evaluate the interaction between perfusion imaging and these clinical variables.
Anoxic brain injury, defined as irreversible neuronal damage secondary to prolonged hypoxia or anoxia, is a leading cause of death and disability in the United States and worldwide, affecting hundreds of thousands of individuals annually. The prognosis is dire. Survivors face poor functional outcomes while family members often confront profound economic hardship associated with prolonged inpatient rehabilitation and round-the-clock care[3,4].
Cardiac arrest represents the most common underlying cause of anoxic brain injury, accounting for the vast majority of cases[5]. However, any pathophysiologic process that results in diminished cerebral blood flow-including drug overdose, stroke, toxic exposure, asphyxiation, and trauma-can lead to anoxic brain injury[6]. A diagnosis is established based on correlation of clinical history, neurologic examination, electroencephalography, and laboratory studies[7].
The prognosis of patients with anoxic brain injury varies based on age, the severity of insult, and the duration of hypoxia or anoxia, among other factors. However, as of this writing, there are no validated criteria that can reliably predict patient outcomes. Clinical manifestations of anoxic brain injury that are highly suggestive of poor outcomes include loss of BSRs, status myoclonus, and a GCS motor score of two or lower[8,9]. Somatosensory evoked potentials may also have prognostic value; limited data suggest that an absent electrical response within the bilateral median nerves is highly specific for poor neurological outcomes[10]. High serum neuron-specific enolase and S-100B values may portend a poor prognosis, but the data are inconsistent, and specific cutoff values remain to be established[8,11].
There are two distinct mechanisms by which anoxia causes reversible or irreversible brain injury in the setting of cardiac arrest or any other medical event that reduces cerebral perfusion. In the first phase of anoxic brain injury, interruption of cellular aerobic metabolism results in adenosine triphosphate depletion and failure of the transmembrane sodium-potassium pump, which in turn leads to an overwhelming influx of sodium and water and consequent cell swelling. The increased cell volume reduces the extracellular space by an order of magnitude, preventing the free diffusion of water molecules; increased signal on diffusion weighted MRI sequences represents the imaging manifestation of the oncotic cell death pathway[12]. In the central nervous system, astrocytic swelling is significantly more pronounced relative to neuronal swelling, and thus the presence of diffusion restriction within the brain does not necessarily indicate irreversible neuronal loss[13]. The second phase of anoxic brain injury is characterized by the activation of homeostatic physiologic mechanisms, often aided by cardiopulmonary resuscitation, resulting in postischemic hyperperfusion. Increased blood flow within the cerebrovascular bed produces a neuroinflammatory cascade, mediated by microglia and circulating leukocytes, and activation of proteases and phospholipases that cause further neuronal damage[14,15].
Neuroimaging, including computed tomography (CT), MRI, and positron emission tomography, plays a central role in the assessment of patients with suspected anoxic brain injury. A CT scan is typically acquired shortly after presentation to exclude intracranial hemorrhage, mass effect, large territory infarct, of any other potential precipitating cause of anoxia or hypoxia. However, CT scan is of limited value for assessing patient prognosis. Other neuroimaging modalities, such as MRI, are significantly more sensitive and specific to evaluate the extent of anoxic brain injury.
DWI and fluid-attenuated inversion recovery (FLAIR) MRI sequences have emerged as powerful predictive tools for individuals who have suffered anoxic brain injury. In a 2010 prospective study by Choi et al[16], authors found that a pattern of diffusion restriction involving the cortex and deep gray nuclei was associated with poor outcomes, whereas the absence of diffusion abnormalities within these regions indicated a high probability of at least partial neurological recovery. Quantitative analysis of diffusion abnormalities using apparent diffusion coefficient (ADC) sequences has also be used to predict patient outcomes. In a prospective study by Wijman et al[17], investigators assessed the value of ADC signal intensity to predict survival and functional outcomes using the validated Glasgow Outcome Scale (GOS). Patients were divided into three groups: Death at six months; GOS score of 3 at six months, indicating severe disability; or a GOS score of 4 or 5 at six months, indicating some degree of neurological recovery with only mild to moderate disability. Authors concluded that the volume of brain parenchyma with an ADC value below 650 × 10-6 mm2/sec correlated with the risk of death; patients with a high percentage of brain tissue below the ADC threshold were significantly less likely to survive than those with values above the ADC threshold. Authors also found that survivors with ADC values greater than 450 × 10-6 mm2/sec were significantly more likely than those with lower ADC values to experience a favorable functional outcome[17]. Notably, the Wijman group acquired imaging between two and five days after the initial event that precipitated anoxic brain injury, as previous studies have shown that DWI and ADC values evolve during the first 48 hours of anoxia and may not depict the full extent of injury[18,19].
A qualitative scoring system developed by Hirsch et al[20] that incorporates DWI and FLAIR signal characteristics was recently shown to predict outcomes in patients with anoxic brain injury. In a 2015 study that included 68 patients who presented after cardiac arrest, investigators assessed the prognostic value of a scoring system based on lesion location and degree of DWI and FLAIR signal abnormality. A high total score based on the qualitative signal intensity within the cortex was up to 63% sensitive and 100% specific for prediction of unfavorable outcomes. The sensitivity increased to 84% when correlated with known predictors of poor outcomes, including absent motor reflexes and somatosensory evoked potentials. Authors concluded that integration of the imaging-based scoring system with established clinical parameters could help reliably predict patient outcomes in the setting of anoxic brain injury.
It has recently been hypothesized that postischemic hyperperfusion can be detected and quantified using ASL perfusion sequences. In a 2008 retrospective study by Pollock et al[1], authors showed that pulsed ASL perfusion sequences reliably demonstrated global cerebral hyperperfusion in the setting of anoxic brain injury. Diffusion restriction was present in most patients and was often diffuse, involving the cortices, basal ganglia, or watershed territories. Notably, of the four patients who survived in the Pollock study, two were found to have global hyperperfusion on pulsed ASL perfusion sequences without evidence of concomitant diffusion restriction, suggesting that there may be some prognostic significance to the pattern of superphysiologic cerebral perfusion relative to diffusion restriction on imaging. In a subsequent study by Li et al[2], authors described a homogeneously positive relationship between ASL and DWI signal intensity in patients with anoxic brain injury, with discrete regional ASL perfusion abnormalities corresponding to areas of diffusion restriction. Authors proposed that coexistent regional hyperperfusion and diffusion restriction may reflect disruption of the blood-brain barrier and aberrant autoregulation in areas of high demand.
The results of our analysis are consistent with those described by Pollock et al[1] and suggest that DWI and ASL perfusion MRI sequences may play a role in predicting patient outcomes in anoxic brain injury. Although our study was underpowered to establish statistical significance, there were several observations that may be clinically meaningful and warrant further examination. First, there was a direct correlation between mean DWI signal intensity and the likelihood of absent BSR following the anoxic event, suggesting that global elevation in DWI signal intensity may predict poor outcomes. Second, patients who presented with MSE within 24 hours of anoxic brain injury were found to have elevated ASL perfusion signal intensity within the left frontal lobe, which indicates that ASL perfusion sequences may be assessed alongside physical examination and electroencephalography to confirm suspected MSE. Third, there was a direct correlation between ASL perfusion signal intensity within the right frontal lobe and the likelihood of absent BSR on day three post-anoxic brain injury; these findings suggest that a frontal lobe ASL perfusion signal abnormality may be related to outcomes, although a larger sample size would again be required to establish statistical significance.
One of the novel contributions of this study is the observation that increased ASL perfusion signal intensity in the frontal lobes-particularly the left frontal lobe-was associated with the presence of MSE following cardiac arrest. While prior work has described global hypoperfusion or posterior-predominant changes in anoxic brain injury, few studies have investigated focal hyperperfusion patterns and their clinical significance. Our findings suggest that frontal ASL hyperintensity may reflect selective regional vulnerability or excitotoxic cortical hyperactivity related to seizure activity, which may not be evident on conventional structural imaging. Although the ROC performance was moderate (AUC = 0.76), this association warrants further investigation, especially in the context of perfusion-based prognostication strategies. These results, if validated, could help identify a subset of patients with poor neurologic prognosis and support earlier risk stratification.
We propose that qualitative or semiquantitative assessment of ASL signal intensity may have prognostic significance if there is concurrent diffusion restriction, with higher ASL signal intensity relative to an internal control indicating a higher likelihood of an unfavorable outcome or death. While clinical examination and EEG remain central to prognostication following cardiac arrest, these modalities can be limited by sedation, metabolic derangements, or technical variability. ASL perfusion imaging offers a noninvasive, quantitative method to assess regional cerebral blood flow, which may provide supplementary information-particularly in patients with unclear or suppressed neurological findings. In our cohort, increased frontal lobe ASL signal was associated with MSE, a clinical marker of poor prognosis, suggesting that ASL may help identify cortical hyperexcitability or early neurovascular uncoupling. Although these findings are preliminary, the integration of ASL into a multimodal prognostic algorithm could enhance confidence in early decision-making, especially when clinical signs are ambiguous or discordant. However, in the absence of diffusion restriction, ASL signal intensity may be of limited value for prognostication. Indeed, ASL signal characteristics should be interpreted with caution and correlated with the clinical history and other imaging findings. Global or regional ASL hyperintense signal can be seen in hepatic encephalopathy, posterior reversible encephalopathy syndrome, and migraines, among other etiologies, and the presence of superphysiologic cerebral blood flow does not necessarily indicate anoxic injury[21-24].
There are several limitations of our study. The sample size was small and few patients experienced functional or neurological recovery. A larger sample size would allow for a more precise ASL signal value threshold to predict unfavorable outcomes. Although an ASL score ≥ 7 was associated with higher odds of absent BSR and MSE, the ROC analysis demonstrated poor discriminatory performance for these outcomes (AUCs of 0.52 and 0.52, respectively), and the results were not statistically significant. These findings suggest that the ASL cutoff score in this cohort lacked sufficient sensitivity or specificity for reliable clinical use. Given the small sample size and low event rates, the apparent trends may reflect statistical noise rather than a true association. Accordingly, these results should be interpreted with caution and considered hypothesis-generating rather than confirmatory. Larger studies are needed to validate any potential perfusion-based thresholds in post-anoxic injury prognostication. In addition, other causes of elevated ASL signal intensity, such as luxury reperfusion related to hypoperfusion-related infarct, could not be definitively excluded. Image quality was acceptable but somewhat degraded by motion artifact in some patients and it is possible that ASL perfusion signal intensity within the predefined structures was over- or underestimated in some cases. All imaging in this study was conducted on a 1.5T General Electric MRI scanner, which may limit the generalizability of our findings to studies performed at 3.0T or on scanners from other manufacturers. While 3.0T systems typically offer higher signal-to-noise ratios and improved spatial resolution, they may also introduce increased susceptibility effects and variability in ASL quantification parameters. These technical differences can affect both perfusion signal intensity and normalization metrics. As such, validation of our results across different field strengths and platforms will be important to confirm the clinical applicability of ASL-based prognostication in broader settings.
One additional limitation of this study was the low inter-rater reliability for normalized ASL signal intensity. This may be due in part to variability introduced by the normalization process, which used the ipsilateral cerebellar hemisphere as the internal reference region. While cerebellar normalization is a commonly used method to account for inter-individual and scanner-related signal differences in ASL studies[25], it can be problematic in post-arrest patients where diffuse hypoperfusion or cerebellar involvement may compromise its stability as a reference. Additionally, manual ROI placement-although anatomically guided-may have introduced variability, especially in regions with low or heterogeneous perfusion. Small discrepancies in ROI positioning could disproportionately affect normalized values due to signal scaling. In future studies, adopting a consistent and structurally preserved reference region, such as the pons or occipital cortex, may help reduce variability and improve reproducibility[26]. Automated or atlas-based segmentation may also offer a more objective approach to ROI definition. These methodological refinements may strengthen the reliability of normalized ASL measures and should be considered in subsequent validation studies.
Our analysis provides important insight into the value of ASL perfusion and DWI sequences for the prediction of patient outcomes in anoxic brain injury. In addition, our findings provide a framework for future analyses and suggest that careful examination of ASL perfusion signal intensity within the frontal lobes may have prognostic significance. Given the limited sample size and the exploratory nature of this study, the observed trends should be interpreted with caution. Most associations between ASL or DWI signal intensity and clinical outcomes did not reach statistical significance, and the analyses were underpowered to detect modest effects. While some regional ASL patterns-such as frontal hyperperfusion associated with MSE-appear promising, these findings require confirmation in larger studies. Our results should be considered hypothesis-generating and may help guide the design of future prospective trials investigating perfusion imaging as a prognostic tool in post-anoxic brain injury.
| 1. | Pollock JM, Whitlow CT, Deibler AR, Tan H, Burdette JH, Kraft RA, Maldjian JA. Anoxic injury-associated cerebral hyperperfusion identified with arterial spin-labeled MR imaging. AJNR Am J Neuroradiol. 2008;29:1302-1307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 37] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 2. | Li N, Wingfield MA, Nickerson JP, Pettersson DR, Pollock JM. Anoxic Brain Injury Detection with the Normalized Diffusion to ASL Perfusion Ratio: Implications for Blood-Brain Barrier Injury and Permeability. AJNR Am J Neuroradiol. 2020;41:598-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 3. | Nichol G, Stiell IG, Hebert P, Wells GA, Vandemheen K, Laupacis A. What is the quality of life for survivors of cardiac arrest? A prospective study. Acad Emerg Med. 1999;6:95-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 109] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 4. | Burke DT, Shah MK, Dorvlo AS, Al-Adawi S. Rehabilitation outcomes of cardiac and non-cardiac anoxic brain injury: a single institution experience. Brain Inj. 2005;19:675-680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 5. | Laver S, Farrow C, Turner D, Nolan J. Mode of death after admission to an intensive care unit following cardiac arrest. Intensive Care Med. 2004;30:2126-2128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 665] [Cited by in RCA: 738] [Article Influence: 35.1] [Reference Citation Analysis (0)] |
| 6. | Messina Z, Hays Shapshak A, Mills R. Anoxic Encephalopathy. 2023 Jan 28. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. [PubMed] |
| 7. | Ramiro JI, Kumar A. Updates on management of anoxic brain injury after cardiac arrest. Mo Med. 2015;112:136-141. [PubMed] |
| 8. | Horn J, Cronberg T, Taccone FS. Prognostication after cardiac arrest. Curr Opin Crit Care. 2014;20:280-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 9. | Wijdicks EF, Hijdra A, Young GB, Bassetti CL, Wiebe S; Quality Standards Subcommittee of the American Academy of Neurology. Practice parameter: prediction of outcome in comatose survivors after cardiopulmonary resuscitation (an evidence-based review) [RETIRED]: report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 2006;67:203-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1034] [Cited by in RCA: 859] [Article Influence: 45.2] [Reference Citation Analysis (0)] |
| 10. | Cronberg T, Brizzi M, Liedholm LJ, Rosén I, Rubertsson S, Rylander C, Friberg H. Neurological prognostication after cardiac arrest--recommendations from the Swedish Resuscitation Council. Resuscitation. 2013;84:867-872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 98] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 11. | Sandroni C, Cavallaro F, Callaway CW, D'Arrigo S, Sanna T, Kuiper MA, Biancone M, Della Marca G, Farcomeni A, Nolan JP. Predictors of poor neurological outcome in adult comatose survivors of cardiac arrest: a systematic review and meta-analysis. Part 2: Patients treated with therapeutic hypothermia. Resuscitation. 2013;84:1324-1338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 240] [Cited by in RCA: 220] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 12. | Liang D, Bhatta S, Gerzanich V, Simard JM. Cytotoxic edema: mechanisms of pathological cell swelling. Neurosurg Focus. 2007;22:E2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 311] [Cited by in RCA: 286] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 13. | Lafrenaye AD, Simard JM. Bursting at the Seams: Molecular Mechanisms Mediating Astrocyte Swelling. Int J Mol Sci. 2019;20:330. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 14. | Bhalala US, Koehler RC, Kannan S. Neuroinflammation and neuroimmune dysregulation after acute hypoxic-ischemic injury of developing brain. Front Pediatr. 2014;2:144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 75] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 15. | Sandroni C, Cronberg T, Sekhon M. Brain injury after cardiac arrest: pathophysiology, treatment, and prognosis. Intensive Care Med. 2021;47:1393-1414. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 262] [Article Influence: 65.5] [Reference Citation Analysis (0)] |
| 16. | Choi SP, Park KN, Park HK, Kim JY, Youn CS, Ahn KJ, Yim HW. Diffusion-weighted magnetic resonance imaging for predicting the clinical outcome of comatose survivors after cardiac arrest: a cohort study. Crit Care. 2010;14:R17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 88] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 17. | Wijman CA, Mlynash M, Caulfield AF, Hsia AW, Eyngorn I, Bammer R, Fischbein N, Albers GW, Moseley M. Prognostic value of brain diffusion-weighted imaging after cardiac arrest. Ann Neurol. 2009;65:394-402. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 231] [Cited by in RCA: 199] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 18. | Hald JK, Brunberg JA, Dublin AB, Wootton-Gorges SL. Delayed diffusion-weighted MR abnormality in a patient with an extensive acute cerebral hypoxic injury. Acta Radiol. 2003;44:343-346. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 19. | Greer DM. MRI in anoxic brain injury. Neurocrit Care. 2004;1:213-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 20. | Hirsch KG, Mlynash M, Jansen S, Persoon S, Eyngorn I, Krasnokutsky MV, Wijman CA, Fischbein NJ. Prognostic value of a qualitative brain MRI scoring system after cardiac arrest. J Neuroimaging. 2015;25:430-437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 64] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 21. | Li Y, Liu H, Yang J, Tian X, Yang H, Geng Z. Combining arterial-spin labeling with functional magnetic resonance imaging measurement for characterizing patients with minimal hepatic encephalopathy. Hepatol Res. 2017;47:862-871. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Weinstein JD, Hamam O, Urrutia VC, Lu H, Luna LP, Tekes-Brady A, Bahouth M, Yedavalli V. Added Value of Arterial Spin Labeling in Detecting Posterior Reversible Encephalopathy Syndrome as a Stroke Mimic on Baseline Neuroimaging: A Single Center Experience. Front Neurol. 2022;13:831218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 23. | Pollock JM, Deibler AR, Burdette JH, Kraft RA, Tan H, Evans AB, Maldjian JA. Migraine associated cerebral hyperperfusion with arterial spin-labeled MR imaging. AJNR Am J Neuroradiol. 2008;29:1494-1497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 65] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 24. | Zhang XD, Zhang LJ, Wu SY, Lu GM. Multimodality magnetic resonance imaging in hepatic encephalopathy: an update. World J Gastroenterol. 2014;20:11262-11272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 20] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 25. | Alsop DC, Detre JA, Golay X, Günther M, Hendrikse J, Hernandez-Garcia L, Lu H, MacIntosh BJ, Parkes LM, Smits M, van Osch MJ, Wang DJ, Wong EC, Zaharchuk G. Recommended implementation of arterial spin-labeled perfusion MRI for clinical applications: A consensus of the ISMRM perfusion study group and the European consortium for ASL in dementia. Magn Reson Med. 2015;73:102-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1554] [Cited by in RCA: 1663] [Article Influence: 166.3] [Reference Citation Analysis (0)] |
| 26. | Mutsaerts HJ, Steketee RM, Heijtel DF, Kuijer JP, van Osch MJ, Majoie CB, Smits M, Nederveen AJ. Inter-vendor reproducibility of pseudo-continuous arterial spin labeling at 3 Tesla. PLoS One. 2014;9:e104108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 62] [Article Influence: 5.6] [Reference Citation Analysis (0)] |