Published online Apr 15, 2017. doi: 10.4239/wjd.v8.i4.154
Peer-review started: November 26, 2016
First decision: January 16, 2017
Revised: February 11, 2017
Accepted: February 28, 2017
Article in press: March 2, 2017
Published online: April 15, 2017
Processing time: 139 Days and 17.9 Hours
To determine the short-term biochemical effects and clinical tolerability of intermittent fasting (IF) in adults with type 2 diabetes mellitus (T2DM).
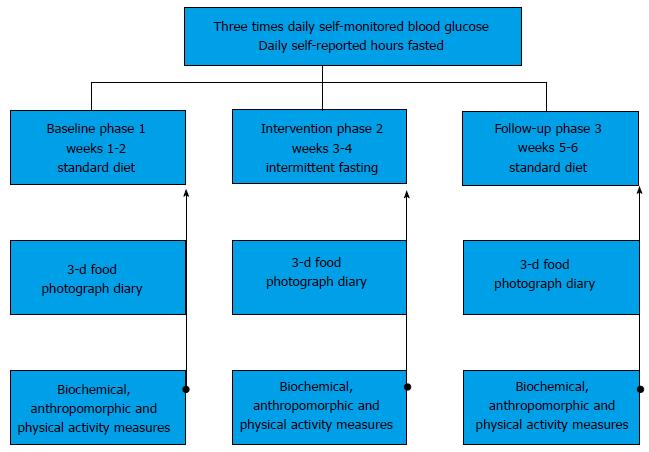
We describe a three-phase observational study (baseline 2 wk, intervention 2 wk, follow-up 2 wk) designed to determine the clinical, biochemical, and tolerability of IF in community-dwelling volunteer adults with T2DM. Biochemical, anthropometric, and physical activity measurements (using the Yale Physical Activity Survey) were taken at the end of each phase. Participants reported morning, afternoon and evening self-monitored blood glucose (SMBG) and fasting duration on a daily basis throughout all study stages, in addition to completing a remote food photography diary three times within each study phase. Fasting blood samples were collected on the final days of each study phase.
At baseline, the ten participants had a confirmed diagnosis of T2DM and were all taking metformin, and on average were obese [mean body mass index (BMI) 36.90 kg/m2]. We report here that a short-term period of IF in a small group of individuals with T2DM led to significant group decreases in weight (-1.395 kg, P = 0.009), BMI (-0.517, P = 0.013), and at-target morning glucose (SMBG). Although not a study requirement, all participants preferentially chose eating hours starting in the midafternoon. There was a significant increase (P < 0.001) in daily hours fasted in the IF phase (+5.22 h), although few attained the 18-20 h fasting goal (mean 16.82 ± 1.18). The increased fasting duration improved at-goal (< 7.0 mmol/L) morning SMBG to 34.1%, from a baseline of 13.8%. Ordinal Logistic Regression models revealed a positive relationship between the increase in hours fasted and fasting glucose reaching target values (χ2 likelihood ratio = 8.36, P = 0.004) but not for afternoon or evening SMBG (all P > 0.1). Postprandial SMBGs were also improved during the IF phase, with 60.5% readings below 9.05 mmol/L, compared to 52.6% at baseline, and with less glucose variation. Neither insulin resistance (HOMA-IR), nor inflammatory markers (C-reactive protein) normalized during the IF phase. IF led to an overall spontaneous decrease in caloric intake as measured by food photography (Remote Food Photography Method). The data demonstrated discernable trends during IF for lower energy, carbohydrate, and fat intake when compared to baseline. Physical activity, collected by a standardized measurement tool (Yale Physical Activity Survey), increased during the intervention phase and subsequently decreased in the follow-up phase. IF was well tolerated in the majority of individuals with 6/10 participants stating they would continue with the IF regimen after the completion of the study, in a full or modified capacity (i.e., every other day or reduced fasting hours).
The results from this pilot study indicate that short-term daily IF may be a safe, tolerable, dietary intervention in T2DM patients that may improve key outcomes including body weight, fasting glucose and postprandial variability. These findings should be viewed as exploratory, and a larger, longer study is necessary to corroborate these findings.
Core tip: Intermittent fasting (IF) involves limiting food intake into a single 4 to 8 h period, daily. We observed the tolerability, safety and health benefits of IF in 10 type 2 diabetes mellitus (T2DM) patients during a 2-wk IF intervention. Outcomes were measured after the three study phases; baseline, intervention, and follow-up. Although short, the IF phase significantly improved weight loss and fasting glucose levels, was well tolerated, and hypoglycemia was not observed. During follow-up, glucose levels reverted. This simple, outpatient-directed dietary manipulation may prove valuable in T2DM individuals with exercise intolerance, who are resistant to complex diet regimes, or are not at glycemic goals.
- Citation: Arnason TG, Bowen MW, Mansell KD. Effects of intermittent fasting on health markers in those with type 2 diabetes: A pilot study. World J Diabetes 2017; 8(4): 154-164
- URL: https://www.wjgnet.com/1948-9358/full/v8/i4/154.htm
- DOI: https://dx.doi.org/10.4239/wjd.v8.i4.154
The incidence of type 2 diabetes mellitus (T2DM) is reaching epidemic levels worldwide, and correlates with rising obesity rates and sedentary lifestyles. In fact, it is predicted that 439 million adults will have diabetes by 2030[1]. This is significant, as diabetes is closely associated with cardiovascular disease, retinopathy, neuropathy, and kidney disease. In turn, this places increasing stress on the health care system, and these patients utilize medical resources three to four times the amount of those without diabetes[2].
Modest weight loss and exercise regimes can both prevent the onset of T2DM and improve metabolic control[3]. According to most national diabetes associations and clinical practice, dietary interventions are considered essential in the treatment and prevention of diabetes-related complications[4]. There are many types of dietary interventions people may use; one of which is intermittent fasting (IF). This is a dietary intervention that time-restricts feeding to 4-6 h and extends the overnight fast from 12 towards 18 or 20 h, may be a beneficial additional dietary strategy used in T2DM management.
After diagnosis, most individuals (depending on individual circumstances) with T2DM are given management goals of an HbA1c below 7.0%, FPG 4.0-7.0 mmol/L, and post-prandial levels of 5.0-10.0 mmol/L[3,5,6]. Self-monitoring of blood glucose (SMBG) using glucometers can be critical for patient feedback and recognition of glucose control, as symptoms poorly predict glucose levels alone. Glucose goals are typically reached through an individualized treatment regime including lifestyle (diet and exercise) and medication use, yet a great number fail to reach, or maintain, these diabetic goals. As a result, a plethora of medication combinations are tried, as are weight loss approaches including consideration for bariatric surgery for morbidly obese individuals. Given the intense focus on weight and dietary measures, IF has the potential to be included in the armament in the fight to improve SMBG levels while contributing to weight loss in those in whom it would be beneficial.
In addition to glucose levels and body weight, there are other important aspects of T2DM worth considering. First is the degree of insulin resistance, which theoretically contributes to the difficulty of maintaining euglycemia[7]. Outside of the research clinic, insulin resistance can be measured in healthy patients using surrogate index measures derived from glucose and insulin measures in the fasting state. These surrogate index measures include glucose/insulin ratio, log fasting insulin, Homeostasis Model Assessment (HOMA-IR), log HOMA-IR, and Quantitative Insulin Sensitivity Check Index (QUICK1). A decrease in insulin resistance can improve glucose control, and exercise and weight loss both favorably decrease resistance states. Hence, we measured HOMA-IR, which has the most agreement with the clamp technique in assessing insulin resistance in T2DM patients[8], to help determine if IF may be beneficial.
Although the causal role is unknown, an association between chronic low-grade inflammation and diabetes has been shown to occur[9]. C-reactive protein (CRP) is a biomarker used to determine if inflammation is present. Elevated levels of CRP have been associated with insulin resistance, nephropathy progression and elevated fasting glucose in diabetes patients[10,11]. It is also known that CRP can be decreased with dietary therapy[12]. Hence, we measured CRP to see whether IF favorably decreased this inflammatory marker.
It can be challenging to measure dietary and exercise habits in clinical studies, and there is no defined intervention that is shown to be better than one another[13]. Monitoring devices worn daily are acceptable methods of tracking physical activity, with accelerometers being the current gold standard[14]. An alternative method to use in free-living adults is a questionnaire. Although not the gold standard, they have been shown to be a reliable method. The Yale Physical Activity Survey (YPAS) is a particular questionnaire that has shown to be reliable for capturing physical activity[14,15]. The YPAS has considerable test-retest reliability which makes it a useful tool for a repeated measures design in free living-adults[16,17]. Hence, the YPAS questionnaire was employed to track physical activity in this study.
The gold standard for measuring energy intake is the Doubly-Labelled Water method (DLW)[18]. However, this is difficult to use in those who are not experienced. One alternative to the DLW is the remote food photography method (RFPM). The RFPM has been shown to be an efficient and accurate method of capturing dietary intake in free-living adults[19,20]. Hence, the RFPM for 3 d in each of the study phases was utilized in this study to capture estimates of energy intake.
The Canadian Diabetes Association (CDA) publishes Clinical Practice Guidelines that currently recommends that individuals with T2DM follow the Canada Food Guide for nutritional needs[21]. Further, the CDA promotes various other nutritional strategies, such as portion control, carbohydrate counting, and grouping foods according to their glycemic index[4]. While these are all appropriate recommendations, some people have difficulty grasping these suggestions and fitting them into their diet[22]. Even an activity as simple as calorie-counting has been reported to lead to an increase in self-perceived psychological stress and cortisol levels, effects not seen when participants restricted calories unintentionally[23].
Another valuable resource for helping attain nutritional and diabetic goals is for patients to see a dietitian and/or attend a self-management program. However, many barriers exist for patients to access these programs or adhering to dietary advice[24]. Some of the barriers listed by people with diabetes range from feelings of helplessness and frustration, to not attaining desired glycemic control, to not being able to accommodate suggestions regarding food restrictions[22,25].
To overcome some of the barriers that some diabetes patients face with dietary interventions, alternative solutions should be explored. An example of an alternative dietary solution is IF. There are many variations of IF, but it is essentially restricting caloric intake to a specified period of time. One method of IF is to have people restrict their caloric intake for 18 to 20 h per day and eat ad libidum during those other 4 to 6 h. The feeding period will usually occur midday to early evening, and an increased protein intake may or may not be recommended to help increase satiety. During the fasting period, people are allowed to consume water, coffee, or tea. With this method of IF, caloric intake occurs when there is a diurnal peak in insulin sensitivity, and, similarly, a diurnal peal in cortisol levels during the fasting period. This may theoretically benefit glucose control. Similar protocols have been shown to have beneficial effects in non-diabetic populations with varying effects on glucose uptake, lipid levels, cortisol levels, and body fat[26-29]. Previous studies have shown that an IF intervention whereby all of one’s daily calorie consumption are consumed in a four-hour window led to participants feeling too full and some weight loss, despite recommendations to consume more calories[26,27]. Hence, it is possible that IF can lead to a spontaneous caloric deficit in adult patients in their homes[30]. However, another study found that an IF intervention led to worsening of glycemic control[27], hence there are conflicting results as to its overall effects in a healthy individuals.
IF has received increasing attention for its potential therapeutic role in the treatment and prevention of cardiovascular disease and T2DM. However, we could only identify 2 studies in the literature evaluating the effects of IF in T2DM, although other trials are ongoing. The first evaluated a Mediterranean-style diet, with and without breakfast, on postprandial glucose, insulin, triglycerides and gastric inhibitory polypeptide in individuals with T2DM over a single day[31]. The Mediterranean lunch without breakfast revealed no increase in glucose, insulin, or triglycerides during the breakfast period (despite coffee intake), which did significantly increase above baseline during the comparator arms of low-fat and low-carbohydrate breakfasts. Another 12-wk had people undergo an IF fast whereby they only consumed caloric content in the morning and early afternoon, and in this study, a decrease in FBG, HbA1c, and an improved response to an OGTT were observed[32]. Given these potential benefits, we designed a short-term observational pilot study to assess tolerability, safety, and specific anthropomorphic, biochemical and blood glucose health benefits of daily IF in patients with T2DM, and measured their persistence upon return to an unstructured diet.
Participants were recruited from within the Saskatoon Health Region, which serves a population catchment of approximately 325000 people. Posters were delivered to general practitioners’ offices in Saskatoon, as well as the 3 hospitals within the city of Saskatoon. Advertisements to the general public were placed in local newspapers and via internet outlets (i.e., Kijiji) alerting them of the study.
Individuals with a diagnosis of T2DM (confirmed by fasting glucose > 7.0 mmol, HbA1c > 6.5%, or OGTT > 11.0 mmol) and between the ages of 18-65 were eligible to enroll in this study. Certain medical conditions were excluded from enrollment, such as the presence of ischemic heart disease or heart failure, chronic inflammatory diseases, chronic infections, moderate to severe renal disease (GFR < 45), uncontrolled hypertension and hypoglycemic unawareness. Lastly, participants were excluded if they currently managed their diabetes with either insulin or glyburide due to their increased risk of hypoglycemia.
Interested participants that met inclusion criteria provided written consent and were educated on study procedures, and the risk, detection and management of hypoglycemia. The study was divided into 3 phases, baseline (run-in), intervention, and follow-up (Figure 1). During the baseline period, participants engaged in their normal dietary patterns (breakfast, lunch and dinner) for a period of 2 wk. During the intervention phase (weeks 3-4), participants were instructed to follow the IF meal timing pattern. This consisted of a fasting goal for a period of 18-20 h per day, with ad libidum zero-calorie coffee, tea, and water intake during fasting hours being permitted. During feeding time, participants were allowed to eat whatever they chose, but were encouraged to include at least 1/3 plate of protein to promote satiety (visual representations provided during training). The intervention phase was followed by a 2 wk follow-up phase with normal dietary patterns. There were no embedded criteria for weight loss, calorie restriction, or changes to exercise habits for the participants.
Self-reporting: Hours fasted, SMBG, caloric intake and exercise were all self-reported. Throughout all study phases, participants reported SMBG 3 times daily (fasting morning, random afternoon, and postprandial evening) with the use of a glucometer and logbook that was provided to them free of charge. In the same logbook, they also kept a diary of total consecutive hours fasted each day. In each of the three time periods (baseline, intervention, follow-up) participants completed a random 3-d food diary using the RFPM. During these days, participants received customized text message prompts by study staff to ensure compliance with RFPM. Participants responded to all text prompts to confirm that they had adhered to RFPM, as well as sent images of their food (before and after consumption to capture food waste). Physical activity/spontaneous energy expenditures were captured using the YPAS tool at the end of each of the study phases.
Biochemical and anthropometric measurements: Participants underwent fasted blood draws on the last day of the baseline phase, intervention phase, and follow-up phase. Fasting insulin, fasting plasma glucose (FPG) (with subsequent calculation for HOMA-IR) and CRP were measured. On each of these days, participants also underwent anthropometric measurements (height, weight, blood pressure, waist circumference). These measurements were all performed by the same individual.
All statistical procedures were performed on SPSS v. 22 and STATA v. 13. Data preparation was done using Excel 2011 and STATA v. 13. Significance was set at alpha = 0.05 (95%CI) for all tests. Repeat measures ANOVA were performed for measuring changes in anthropometric and biochemical changes.
The group means and standard deviations of days 1 through 42 (6 wk total study time) were calculated individually for three daily measurements: fasted morning (M), random afternoon (A), and postprandial evening (E) SMBG measurements. Linear and quadratic regression of group means and standard deviations were used to explore the relationship between study phase and SMBG.
OLR was used to explore the impact of relative daily fasting duration on SMBG. Cut-offs for OLR were created using standards for the diabetic fasting goal (< 7.0 mmol/L) and frank hyperglycemia (> 11.1 mmol/L), with an additional midpoint (9.05 mmol/L). The variable created for OLR was the hours fasted difference (HFD), the difference between actual hours fasted and the average hours fasted during the baseline phase (Table 1).
| SMBG value cut-off | Morning fasted | Evening postprandial |
| < 7.0 mmol/L | Normal/goal level | Normal/goal level |
| 7.0-9.05 mmol/L | Above goal | Normal/goal level |
| 9.05-11.1 mmol/L | Above goal | Above goal |
| > 11.1 mmol/L | Above goal | Above goal |
No category was created to represent hypoglycemic events, as none were recorded throughout the duration of the study.
Summarized in Table 2 are the anthropomorphic and biochemical measurements (with standard deviation) of the ten participants at study entry. Normal values for clinical and biochemical parameters are referenced. Subjects had a mean age of 53.8 years old (ranging 44-62) and BMI of 36.9 (range of 28-45 kg/m2), the latter corresponding on average to Class II obesity, with very high disease risk compared to normal weight individuals. Insulin resistance was confirmed, with a mean HOMA-IR of 6.91, and baseline fasting blood glucose levels were above the goal of < 7.0 mmol/L (mean 7.45 mmol/L). A nonspecific biochemical marker of inflammation (CRP) was also elevated at baseline in this cohort. All participants took daily metformin as part of their diabetic management, eight as monotherapy. One participant was also taking gliclazide while another was also using liraglutide.
| Measurement | Mean ± SD |
| Anthropomorphic | |
| Age (yr) | 53.8 ± 9.11 |
| Weight | 100.6 ± 21.75 kg |
| BMI | 36.9 ± 8.29 kg/m2 |
| Waist circumference (reference < 88 female, < 102 male) | 109.6 ± 11.1 cm |
| Daily hours fasted | 11.6 ± 1.9 h/d |
| Systolic BP (mmHg) T2DM goal < 130 | 130.00 ± 17.80 |
| Diastolic BP (mmHg) T2DM goal < 80 | 80.50 ± 13.20 |
| Biochemical | |
| C-reactive protein (mg/L) (reference < 1.0 mg/L) | 4.31 ± 3.80 |
| HOMA-IR calculated (normal < 2.5) | 6.91 ± 3.00 |
| Fasting glucose (normal < 7.0 mmol/L) | 7.45 ± 1.52 mmol/L |
| Medications present during study period | |
| Metformin | 10 (10) |
| Sulfonylureas | 1 (10) |
| Other diabetic medications | 1 (10) |
| Other non-diabetic medications | 8 (10) |
The clinical and biochemical changes resulting from a 2 wk dietary intervention of IF are shown in Table 3. The means of the measured biochemical and anthropomorphic outcomes of all subjects were calculated for each variable within the three phases of the study, with standard deviations shown: Phase 1 (baseline), Phase 2 (intervention) and Phase 3 (follow-up). Repeat measures ANOVA comparisons between study phases were done to reveal significant differences (P < 0.05). Clinical measures revealed that IF decreased mean body weight, BMI, blood pressure, and waist circumference as compared to baseline with significant changes only for body weight (-1.4 kg; P = 0.009) and BMI (-0.52; P = 0.01). The beneficial changes observed were not sustained once the IF was complete. After a return to normal diet (Phase 3, follow-up), there was an inflection back toward baseline values for all parameters except a further non-significant decrease in waist circumference (-0.3, P = 1.0). All participants increased their fasting time during the intervention phase. The daily hours fasting increased from a baseline of 11.6 to 16.8 h during the intervention phase (+5.2 h; P < 0.001), and essentially returned to baseline (11.5 h) after the follow-up period.
| Measurement | Mean ± SD Phase 1 | Mean ± SD Phase 2 | Mean ± SD Phase 3 | Mean difference Phase 1 to 2 | Mean difference Phase 2 to 3 | Mean difference Phase 1 to 3 |
| Clinical outcome | ||||||
| Weight (kg) | 100.6 ± 21.7 | 99.2 ± 21.3 | 99.5 ± 21.5 | -1.4 (P = 0.009) | +0.28 (P = 1.0) | -1.12 (P = 0.08) |
| BMI (kg/m2) | 36.9 ± 8.3 | 36.4 ± 8.1 | 36.5 ± 8.1 | -0.52 (P = 0.01) | +0.1 (P = 1.0) | -0.42 (P = 0.09) |
| Waist circumference (cm) | 109.6 ± 11.1 | 107.8 ± 11.1 | 107.5 ± 10.9 | -1.75 (P = 0.086) | -0.30 (P = 1.0) | -2.05 (P = 0.24) |
| Systolic BP (mmHg) | 130.0 ± 17.8 | 127.0 ± 21.4 | 128.5 ± 14.3 | -3 (P = 0.83) | +1.5 (P = 1.0) | -1.5 (P = 1.0) |
| Diastolic BP (mmHg) | 80.5 ± 13.2 | 79.8 ± 15.7 | 81.7 ± 12.2 | -0.72 (P = 1.0) | +1.9 (P = 0.76) | +1.2 (P = 1.0) |
| Daily hours fasted | 11.6 ± 1.9 | 16.8 ± 1.2 | 11.5 ± 2.0 | +5.2 (P < 0.005) | -5.3 (P < 0.005) | -0.09 (P = 1.0) |
| Biochemical outcome | ||||||
| C-reactive protein (mg/L) | 4.3 ± 3.8 | 4.0 ± 3.7 | 4.1 ± 3.5 | -0.3 (P = 0.9) | +0.09 (P = 1.0) | -0.25 (P = 1.0) |
| HOMA-IR | 6.9 ± 3.0 | 6.5 ± 2.4 | 6.6 ± 3.0 | -0.46 (P = 1.0) | +0.11 (P = 1.0) | -0.35 (P = 1.0) |
The averages of SMBG reported daily from home glucometers decreased during the intervention phase for fasting morning, afternoon and postprandial time points (Table 4). Pooled averages of SMBG taken three times daily throughout the study are presented by study phase, indicating the % change from baseline to IF (Phase 1 to 2), and IF to follow-up (Phase 2 to 3).
| 14 d averaged SMBG pooled | Mean ± SD Phase 1 | Mean ± SD Phase 2 | Mean ± SD Phase 3 | % change from Phase 1 to 2 | % change from Phase 2 to 3 |
| μfasting SMBG | 8.2 ± 1.3 | 7.7 ± 1.8 | 8.1 ± 1.4 | -6.10% | +5.20% |
| μafternoon SMBG | 7.5 ± 1.0 | 7.2 ± 1.2 | 7.0 ± 0.9 | -4.00% | -2.80% |
| μpost prandial SMBG | 8.7 ± 1.9 | 8.6 ± 1.9 | 8.8 ± 1.7 | -1.10% | +2.30% |
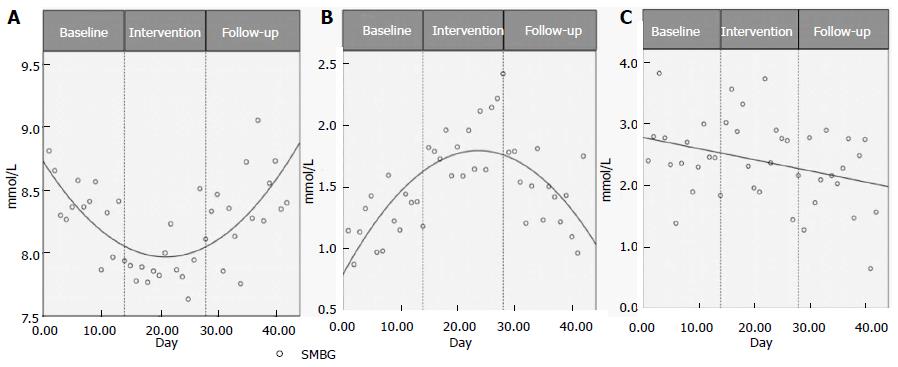
IF improves morning fasted glucose levels and decreases postprandial variability. To further investigate potential benefits of IF on glucose levels, daily (rather than grouped by study phase) SMBGs were plotted over the 42 d study (Figure 2A). To investigate the glucose variances from the mean for fasting morning glucose levels, the Kolmogorov-Smirnov (KS) test was used. Figure 2B indicates that the daily distribution of morning SMBG was greatest during the intervention as compared to baseline (P = 0.002), or follow-up (P = 0.003) phases. Lastly, a similar assessment of the daily variation from the mean for evening postprandial values (Figure 2C) demonstrates wide scatter throughout all phases, with decreases in glucose variability from baseline to intervention, as well as from intervention to follow-up phase. A significant difference was found for evening SMBG distributions (P = 0.044) between the intervention and follow-up phases only (all other phases P > 0.1). There were no statistically significant differences observed between any phases for the afternoon SMBG distributions (P > 0.1).
IF enhances the proportion of fasting SMBG at goal and decreases postprandial glucose excursions. Raw SMBG counts and percentages of SMBG residing within defined glucose categories were tabulated for each study phase. Table 5 reflects morning fasted and evening post-prandial SMBG. SMBG results were placed within four discrete glucose ranges including normal/target fasting (< 7 mmol/L), frank hyperglycemia (> 11.1 mmol/L), and an equal cut-off bridging the two (cut off at 9.05 mmol/L) to determine the cause of the increased variation noted for morning SMBG during IF. The distributed results for fasting SMBG readings Table 5 highlight that the incidence of at-goal SMBG < 7.0 mmol/L increased 2.5-fold during the IF intervention (13.8% baseline increased to 34.1% during IF). Also noted during IF was the overall decrease in fasting hyperglycemia (> 7.0 mmol/L) that was offset by a greater incidence of elevated fasting levels (> 11.1 mmol/L noted at 0.8% baseline, increasing to 7.1% during IF). The improvement of readings at target glucose levels was lost during follow up, upon return to normal eating patterns.
| Measured SMBG (mmol/L) | Baseline | Intervention | Follow-up |
| Morning SMBG by phase | |||
| < 7.0 | 13.8% | 34.1% | 15.1% |
| 7.0-9.05 | 52.0% | 40.7% | 49.6% |
| 9.05-11.1 | 33.3% | 18.0% | 32.8% |
| > 11.1 | 0.8% | 7.1% | 2.5% |
| Evening SMBG by phase | |||
| < 7.0 | 24.5% | 27.7% | 12.9% |
| 7.0-9.05 | 28.1% | 32.9% | 41.6% |
| 9.05-11.1 | 27.4% | 19.7% | 28.7% |
| > 11.1 | 20.0% | 19.7% | 16.8% |
Considering the pooled incidence of evening postprandial SMBG up to and including 9.05 mmol/L as “at-goal” for postprandial blood glucose, IF resulted in a higher proportion in the desirable range (Table 5). Specifically, 65.7% of SMBG were < 9.05 mmol/L during the IF phase of the study, compared to 52.6% at baseline. Similar levels (54.1%) were found at goal during the follow-up phase, as at baseline. The proportion of SMBG > 11.1 mmol/L changed little with IF. The greatest change was found in the decrease in SMBG between 9.05-11.1 mmol/L, specific to the IF phase.
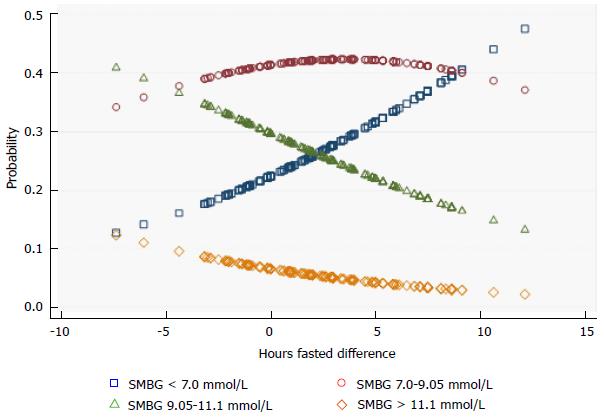
The increase from baseline, rather than the absolute number, of hours fasted improves the probability of optimal glucose control. HFD was calculated for baseline and intervention phases to explore the relationship between the increase in hours fasted from baseline and SMBG improvements. In order to determine the relationship between glucose levels and time spent fasting, OLR was performed on the non-equalized data sets from the 8 participants who had completed both their daily-hours-fasted and full SMBG logs. As presented in Figure 3, these results support the previous SMBG regression findings; mean morning SMBG readings decreased, and improvements in these readings were primarily a result of increased fasting hours from baseline. The HFD and OLR models showed a statistically significant association between change in HFD with decreased SMBG morning readings (χ2 likelihood ratio = 8.36, P = 0.004) but not for afternoon or evening SMBG readings (P > 0.1, Table 6).
| SMBG | Overall model | ||
| Odds ratio | P value | 95%CI | |
| Morning | 0.91 | 0.004 | 0.85-0.97 |
| Afternoon | 0.95 | 0.181 | 0.88-1.02 |
| Evening | 1.00 | 0.900 | 0.94-1.07 |
The IF phase was associated with spontaneous decreases in caloric intake and increases in physical activity. To calculate energy intake during the study, five (baseline and intervention) and four (follow-up) participants recorded their food intake via the RFPM. This was performed for 3 d of each study phase. From this, estimates of the total kcal/day, and the proportionate contribution from protein, carbohydrate and fat was determined as shown in Table 7. Total kcal/day decreased 18.6% with the IF intervention, and further decreased 6% into the follow-up phase. Carbohydrate and fat intake decreased 33% and 36% respectively during the IF period, whereas protein intake remained constant. To estimate physical activity during the study, all participants completed the YPAS once during each of the three phases. From this survey, an estimate of physical activity (kcal/wk) during the three study phases was determined. During the intervention phase, physical activity increased (+1856.3 kcal/wk), but then decreased at study end (in the follow-up phase; -2449.6 kcal/wk).
| Measure | Baseline (mean ± SD) | Intervention (mean ± SD) | Follow-up (mean ± SD) |
| Extrapolated energy intake (kcal/d) | 1904.3 ± 404.1 | 1605.7 ± 375.5 | 1510.5 ± 755.41 |
| Protein intake (g/d) | 94.2 ± 26.6 | 93.2 ± 26.1 | 79.4 ± 30.71 |
| Carbohydrate intake (g/d) | 190.6 ± 58.5 | 142.7 ± 62.1 | 164.2 ± 93.91 |
| Fat intake (g/d) | 86.9 ± 16.6 | 63.6 ± 25.2 | 60.9 ± 35.51 |
| Physical activity (kcal/wk) | 4922.3 ± 3774.4 | 6778.56 ± 4329.5 | 4329.0 ± 3440.8 |
Many individuals with T2DM would benefit from a simple and accessible nutrition intervention that is simple to implement and teach, and improves their glycemic control. Teaching diabetes patients about IF only required between 15-30 min of the study coordinator’s time, hence it was easy to teach. Although short term (2 wk IF intervention), and without oversight (self-reported and self-controlled eating hours), the intervention resulted in significant improvements in diabetic glucose control. The IF phase yielded a significant increase in the incidence of fasting blood sugars at target (34.1% vs 13.4% baseline), and favorable decreases in postprandial hyperglycemia (39.4% vs 47.4% baseline). There was also a spontaneous 18% decrease in caloric intake and increase in energy expenditure (+1856 kcal/wk), coinciding with a significant decrease in weight (P < 0.009) and BMI (P = 0.01). A strong association between the increase in hours fasted from baseline, and the probability of attaining a normal fasting glucose level was found (+LR 8.36), despite few individuals reaching the 18-20 h fasting goal. We did not find statistically significant changes to blood pressure, insulin resistance or inflammatory markers after 2 wk of IF, although all trended towards normal during this phase. Importantly, the diet was found to be tolerable and safe, with zero incidences of hypoglycemia.
Upon a return to normal eating habits, the improvements to fasting glucose levels reversed to baseline, a trend also seen for the non-significant improvements in CRP, HOMA-IR and BP. The sample size of this study is too small to determine if the sustained decreases in waist circumference, postprandial glucose variability, and energy intake into the follow-up period are meaningful.
The most similar study to ours that we could identify in the literature evaluated IF was a 3 mo crossover study in T2DM patients[32]. These studies differed with respect to when participants were instructed to undergo the prolonged fasting period (participants in our study were allowed to self-select their fasting period, and chose late afternoon to early evening). Similar trends were observed with respect to weight loss and decreased waist circumference, whereas the study by Kahleova et al[32] also showed significant improvements in fasting insulin (and presumably insulin resistance), which our did not. In contrast, other studies in non-diabetic, normal-weight participants, have shown that IF resulted in increased insulin resistance, fasting glucose, and lipids, despite weight loss occurring[26,27]. These different observed effects between diabetic and non-diabetic individuals to similar dietary changes may be related to the differences in body weight and hyperglycemia between the studies, or due to as yet unknown factors.
This work demonstrates that there is a correlation between hours fasted and improvements in fasted glucose levels, leading us to question if simply skipping breakfast (and thereby prolonging the fast) would improve T2DM control. A study by Thomas et al[33] tested the effects of skipping breakfast in obese women (without diabetes). They found that when regular breakfast eaters skipped breakfast, they had greater insulin and free fatty acid excursions in response to lunch as opposed to days when both breakfast and lunch were eaten[33]. In contrast, those who regularly skipped breakfast did not experience irregular responses at lunch, regardless of whether breakfast was eaten or not[33]. Clearly, extended fasting will not benefit all and has the potential to worsen measures of health if applied to patients with T2DM.
We also query if the observed benefits in this study arise from altering the normal eating rhythm, rather than inducing a net negative energy state (such as during fasting, or exercise). Although some evidence from animal studies suggest that biological clocks may be altered as a result of changes in feeding habits[34,35], evidence of the effects of feeding entrainment on glycemic excursions in humans are lacking.
Our study involved ten individuals (9 female, one male) who present with the typical characteristics of the majority of the worlds’ T2DM population; middle age, overweight, with insulin resistance and average fasting blood sugars above goal. There are several unique aspects to this study that provide useful information, not the least of which was the patient-controlled meal timing and content, all without caloric restriction. It is the first study designed with patients directly transitioning from their normal dietary habits, to an IF intervention, to a follow-up period to see if any of the effects were sustained. Also, the capturing of SMBG readings along with the actual total hours fasted allowed us to make inferences on this relationship, which has not been done before. Another strength is our use of the RFPM, which has been shown to have good validity[19,20]. Adherence with the study protocol, as reflected by SMBG and the recorded hours fasted, was high. Also, as a surrogate for tolerability of the IF intervention, 7/10 participants viewed the intervention as tolerable when asked, and 6/10 stating they would consider continuing a modified form of IF.
The greatest weakness was the lack of power from low recruitment, which was the limiting factor in determining clear effects of IF on markers of health in T2DM, or the detection of sustained effects during follow-up. An uneven distribution between females (n = 9) and males (n = 1) is also a limiting factor for generalizability. Also of importance to note was the failure to reach the goal of 18-20 h of fasting per day, as these longer fasting times may have accentuated health benefits more clearly. Ultimately, the 16.8 ± 1.2 h fasting during Phase 2 of the study is perhaps a true reflection of the feasibility of fasting times for this diet in free-living adults. Further a longer duration of the study phases would have allowed for comparative HbA1c measures to be done, a widely accepted marker of average changes to overall glucose control reflecting the previous three months. Lastly, it is possible that all of the SMBG testing that patients did may have in itself led to behavior changes that affected the overall recorded glucose levels. Any information learned from this observational pilot study can be used to inform a larger, longer, observational or interventional trial.
Our understanding of the association between feeding entrainment and diurnal rhythms is limited, and requires further study. Also, our study took blood draws from patients at specific time points, for study ease. Ideally, patients would have blood draws performed regular over a 24 h period so that fluctuations could be noted that may be due to diurnal variations, as the consumption of calories changes over the IF period. Since our participants only had blood draws performed in the morning, some biochemical changes that may have occurred during other time periods would have been missed. It would also be ideal for future studies to have a longer study period which would allow for significant weight loss to occur, and then see if any biochemical changes are sustained during an extended follow-up period. Of course, a larger sample size would allow for a more robust conclusion as to the association between SMBG and IF and would shed som light on the true effects on morning, afternoon and evening SMBG results.
The authors would like to acknowledge the help and support that Dr. Hasitha Welihinda provided for this study. Further, we would like to thank the Department of Medicine as well as the College of Pharmacy and Nutrition, University of Saskatchewan, for support of this project. The authors would also like to acknowledge Sanofi Canada for the provision of glucometers and test strips.
Intermittent fasting (IF) is an increasingly popularized dietary method amongst varying population groups. The reasons for its increasing popularity are multi-factorial, but one of which is for weight loss. Although research has been performed evaluating an IF intervention in animals and healthy subjects, there is very little known about its effects, beneficial or otherwise, in those with type 2 diabetes.
As the prevalence of diabetes continues to increase, new dietary measures are necessary to pursue, as one size does not fit all. It is possible that IF may be a good option for people with diabetes alongside other popularized diets such as the Mediterranean diet, the DASH diet, and the Newcastle diet. With type 2 diabetes affecting so many people worldwide, any new potential treatment options are welcome.
To our knowledge, there is only one other study examining IF in type 2 diabetes (Kahleova et al). The study varied in that patients were allowed to self-select their meal times and meal options. Further, the study showed that when practiced with self-selected meal times and meal options, there is a spontaneous reduction in caloric intake (whereas the study by Kahleova provided meals and all patients were assigned to a caloric deficit). Further, the participants chose to fast mostly in the mornings and leave their meals to nighttime, whereas the study by Kahleova had their participants eat only in the morning and afternoon, and fasted in the evenings). All told, there is very little literature examining IF in diabetes patients, and so all information can be seen as important and innovative in their approach to the treatment of type 2 diabetes.
Managing the dietary aspect of diabetes can be very difficult and tiresome for some people. It can often create unnecessary stress and sense of failure in those who are unable or unwilling to adhere to dietary recommendations to help control their blood glucose and body weight. IF is a potential alternative for certain people who wish to consume their caloric intake for the day in a manner which is not considered “recommended” for people with diabetes. It also may lead to some weight loss through less caloric consumption, which is often welcome for many people with type 2 diabetes.
IF: An eating pattern that time-restricts feeding to 4-6 h and extends the overnight fast from 12 h towards 18 or 20 h.
The study is valuable and of interest as intermittent fasting is a hot topic regarding weight loss and related benefits including treatment of diabetes.
Manuscript source: Invited manuscript
Specialty type: Endocrinology and metabolism
Country of origin: Canada
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B, B
Grade C (Good): 0
Grade D (Fair): D
Grade E (Poor): 0
P- Reviewer: Masaki T, Saleh JS, Tamemoto H, Zhao J S- Editor: Ji FF L- Editor: A E- Editor: Wu HL
| 1. | Shaw JE, Sicree RA, Zimmet PZ. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract. 2010;87:4-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4438] [Cited by in RCA: 4370] [Article Influence: 291.3] [Reference Citation Analysis (4)] |
| 2. | Pelletier C, Dai S, Roberts KC, Bienek A, Onysko J, Pelletier L. Report summary. Diabetes in Canada: facts and figures from a public health perspective. Chronic Dis Inj Can. 2012;33:53-54. [PubMed] |
| 3. | Turner RC, Cull CA, Frighi V, Holman RR. Glycemic control with diet, sulfonylurea, metformin, or insulin in patients with type 2 diabetes mellitus: progressive requirement for multiple therapies (UKPDS 49). UK Prospective Diabetes Study (UKPDS) Group. JAMA. 1999;281:2005-2012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1765] [Cited by in RCA: 1682] [Article Influence: 64.7] [Reference Citation Analysis (0)] |
| 4. | Booth G, Cheng AY. Canadian Diabetes Association 2013 clinical practice guidelines for the prevention and management of diabetes in Canada. Methods. Can J Diabetes. 2013;37 Suppl 1:S4-S7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 43] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 5. | Imran SA, Rabasa-Lhoret R, Ross S. Targets for glycemic control. Can J Diabetes. 2013;37 Suppl 1:S31-S34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 44] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 6. | Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359:1577-1589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5314] [Cited by in RCA: 5273] [Article Influence: 310.2] [Reference Citation Analysis (0)] |
| 7. | Gordon BA, Fraser SF, Bird SR, Benson AC. Insulin sensitivity in response to a single resistance exercise session in apparently healthy individuals. J Endocrinol Invest. 2012;35:665-669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 8. | Sarafidis PA, Lasaridis AN, Nilsson PM, Pikilidou MI, Stafilas PC, Kanaki A, Kazakos K, Yovos J, Bakris GL. Validity and reproducibility of HOMA-IR, 1/HOMA-IR, QUICKI and McAuley’s indices in patients with hypertension and type II diabetes. J Hum Hypertens. 2007;21:709-716. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 137] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 9. | Pickup JC. Inflammation and activated innate immunity in the pathogenesis of type 2 diabetes. Diabetes Care. 2004;27:813-823. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 926] [Cited by in RCA: 932] [Article Influence: 44.4] [Reference Citation Analysis (0)] |
| 10. | Navarro JF, Mora C, Maca M, Garca J. Inflammatory parameters are independently associated with urinary albumin in type 2 diabetes mellitus. Am J Kidney Dis. 2003;42:53-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 176] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 11. | Natali A, Toschi E, Baldeweg S, Ciociaro D, Favilla S, Saccà L, Ferrannini E. Clustering of insulin resistance with vascular dysfunction and low-grade inflammation in type 2 diabetes. Diabetes. 2006;55:1133-1140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 144] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 12. | Dworatzek PD, Arcudi K, Gougeon R, Husein N, Sievenpiper JL, Williams SL. Nutrition therapy. Can J Diabetes. 2013;37 Suppl 1:S45-S55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 100] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 13. | Prince SA, Adamo KB, Hamel ME, Hardt J, Connor Gorber S, Tremblay M. A comparison of direct versus self-report measures for assessing physical activity in adults: a systematic review. Int J Behav Nutr Phys Act. 2008;5:56. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1662] [Cited by in RCA: 2016] [Article Influence: 118.6] [Reference Citation Analysis (0)] |
| 14. | Colbert LH, Matthews CE, Havighurst TC, Kim K, Schoeller DA. Comparative validity of physical activity measures in older adults. Med Sci Sports Exerc. 2011;43:867-876. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 173] [Cited by in RCA: 168] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 15. | Starling RD, Matthews DE, Ades PA, Poehlman ET. Assessment of physical activity in older individuals: a doubly labeled water study. J Appl Physiol (1985). 1999;86:2090-2096. [PubMed] |
| 16. | Pennathur A, Magham R, Contreras LR, Dowling W. Test-retest reliability of Yale Physical Activity Survey among older Mexican American adults: a pilot investigation. Exp Aging Res. 2004;30:291-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 17. | De Abajo S, Larriba R, Marquez S. Validity and reliability of the Yale Physical Activity Survey in Spanish elderly. J Sports Med Phys Fitness. 2001;41:479-485. [PubMed] |
| 18. | Hill RJ, Davies PS. The validity of self-reported energy intake as determined using the doubly labelled water technique. Br J Nutr. 2001;85:415-430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 398] [Cited by in RCA: 405] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 19. | Martin CK, Correa JB, Han H, Allen HR, Rood JC, Champagne CM, Gunturk BK, Bray GA. Validity of the Remote Food Photography Method (RFPM) for estimating energy and nutrient intake in near real-time. Obesity (Silver Spring). 2012;20:891-899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 197] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 20. | Martin CK, Nicklas T, Gunturk B, Correa JB, Allen HR, Champagne C. Measuring food intake with digital photography. J Hum Nutr Diet. 2014;27 Suppl 1:72-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 155] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 21. | Health Canada. Eating Well with Canada’s Food Guide 2016 (August 23, 2016). Available from: http://www.hc-sc.gc.ca/fn-an/alt_formats/hpfb-dgpsa/pdf/food-guide-aliment/view_eatwell_vue_bienmang-eng.pdf. |
| 22. | Nagelkerk J, Reick K, Meengs L. Perceived barriers and effective strategies to diabetes self-management. J Adv Nurs. 2006;54:151-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 233] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 23. | Tomiyama AJ, Mann T, Vinas D, Hunger JM, Dejager J, Taylor SE. Low calorie dieting increases cortisol. Psychosom Med. 2010;72:357-364. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 131] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 24. | Gucciardi E, Chan V, Fortugno M. Primary Care Physician Referral Patterns to Diabetes Education Programs in Southern Ontario, Canada. Can J Diab. 2011;35:262-268. [RCA] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 25. | Shultz JA, Sprague MA, Branen LJ, Lambeth S. A comparison of views of individuals with type 2 diabetes mellitus and diabetes educators about barriers to diet and exercise. J Health Commun. 2001;6:99-115. [PubMed] [DOI] [Full Text] |
| 26. | Stote KS, Baer DJ, Spears K, Paul DR, Harris GK, Rumpler WV, Strycula P, Najjar SS, Ferrucci L, Ingram DK. A controlled trial of reduced meal frequency without caloric restriction in healthy, normal-weight, middle-aged adults. Am J Clin Nutr. 2007;85:981-988. [PubMed] |
| 27. | Carlson O, Martin B, Stote KS, Golden E, Maudsley S, Najjar SS, Ferrucci L, Ingram DK, Longo DL, Rumpler WV. Impact of reduced meal frequency without caloric restriction on glucose regulation in healthy, normal-weight middle-aged men and women. Metabolism. 2007;56:1729-1734. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 203] [Cited by in RCA: 191] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 28. | Halberg N, Henriksen M, Söderhamn N, Stallknecht B, Ploug T, Schjerling P, Dela F. Effect of intermittent fasting and refeeding on insulin action in healthy men. J Appl Physiol (1985). 2005;99:2128-2136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 178] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 29. | Soeters MR, Lammers NM, Dubbelhuis PF, Ackermans M, Jonkers-Schuitema CF, Fliers E, Sauerwein HP, Aerts JM, Serlie MJ. Intermittent fasting does not affect whole-body glucose, lipid, or protein metabolism. Am J Clin Nutr. 2009;90:1244-1251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 75] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 30. | Taylor R. Type 2 diabetes: etiology and reversibility. Diabetes Care. 2013;36:1047-1055. [PubMed] |
| 31. | Fernemark H, Jaredsson C, Bunjaku B, Rosenqvist U, Nystrom FH, Guldbrand H. A randomized cross-over trial of the postprandial effects of three different diets in patients with type 2 diabetes. PLoS One. 2013;8:e79324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 32. | Kahleova H, Belinova L, Malinska H, Oliyarnyk O, Trnovska J, Skop V, Kazdova L, Dezortova M, Hajek M, Tura A. Eating two larger meals a day (breakfast and lunch) is more effective than six smaller meals in a reduced-energy regimen for patients with type 2 diabetes: a randomised crossover study. Diabetologia. 2014;57:1552-1560. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 132] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 33. | Thomas EA, Higgins J, Bessesen DH, McNair B, Cornier MA. Usual breakfast eating habits affect response to breakfast skipping in overweight women. Obesity (Silver Spring). 2015;23:750-759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 40] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 34. | Schibler U, Ripperger J, Brown SA. Peripheral circadian oscillators in mammals: time and food. J Biol Rhythms. 2003;18:250-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 35. | Stephan FK. The “other” circadian system: food as a Zeitgeber. J Biol Rhythms. 2002;17:284-292. [PubMed] |