Published online Oct 15, 2023. doi: 10.4239/wjd.v14.i10.1502
Peer-review started: June 9, 2023
First decision: July 18, 2023
Revised: July 31, 2023
Accepted: August 28, 2023
Article in press: August 28, 2023
Published online: October 15, 2023
Processing time: 122 Days and 0.1 Hours
The gut microbiome is defined as an ecological community of commensal symbiotic and pathogenic microorganisms that exist in our body. Gut microbiome dysbiosis is a condition of dysregulated and disrupted intestinal bacterial homeostasis, and recent evidence has shown that dysbiosis is related to chronic inflammation, insulin resistance, cardiovascular diseases (CVD), type 2 diabetes mellitus (T2DM), and obesity. It is well known that obesity, T2DM and CVD are caused or worsened by multiple factors like genetic predisposition, environmental factors, unhealthy high calorie diets, and sedentary lifestyle. However, recent evidence from human and mouse models suggest that the gut microbiome is also an active player in the modulation of metabolic syndrome, a set of risk factors including obesity, hyperglycemia, and dyslipidemia that increase the risk for CVD, T2DM, and other diseases. Current research aims to identify treatments to increase the number of beneficial microbiota in the gut microbiome in order to modulate metabolic syndrome by reducing chronic inflammation and insulin resistance. There is increasing interest in supplements, classified as prebiotics, probiotics, synbiotics, or postbiotics, and their effect on the gut microbiome and metabolic syndrome. In this review article, we have summarized current research on these supplements that are available to improve the abundance of beneficial gut microbiota and to reduce the harmful ones in patients with metabolic syndrome.
Core Tip: Gut microbiome dysbiosis is related to chronic inflammation, insulin resistance, metabolic syndrome, cardiovascular diseases (CVD), and obesity. It is well known that obesity, type 2 diabetes mellitus and CVD are caused or worsened by multiple factors like genetic predisposition, environmental factors, unhealthy high calorie diets, and sedentary lifestyle. However, recent evidence from human and mouse models suggest that the gut microbiome is also an active player in modulation of these metabolic diseases. Hence it is important to review the role of microbiome supplementation that has been shown to improve the gut microbiome in patients with metabolic syndrome.
- Citation: Antony MA, Chowdhury A, Edem D, Raj R, Nain P, Joglekar M, Verma V, Kant R. Gut microbiome supplementation as therapy for metabolic syndrome. World J Diabetes 2023; 14(10): 1502-1513
- URL: https://www.wjgnet.com/1948-9358/full/v14/i10/1502.htm
- DOI: https://dx.doi.org/10.4239/wjd.v14.i10.1502
The gut microbiome is defined as an ecological community of commensal symbiotic and pathogenic microorganisms that exist in the body[1,2]. Gut microbiome dysbiosis is defined as dysregulated and disrupted intestinal bacterial homeostasis[2,3]. Recent evidence has shown that dysbiosis is related to chronic inflammation, insulin resistance, type 2 diabetes mellitus (T2DM), cardiovascular diseases (CVD), and obesity[2]. It is known that obesity, T2DM and CVD are caused or worsened by multiple factors like genetic predisposition, environmental factors, unhealthy high calorie diet, and sedentary lifestyle[4-6]. However, recent evidence from human and mouse models suggests that the gut microbiome is also an active player in the modulation of these diseases[7]. Host and gut microbiome dysbiosis can influence local or systemic immunity and inflammation by regulating intestinal barrier permeability or by triggering the innate immune system as seen in obesity and T2DM[7]. Akkermansia muciniphila and Faecalibacterium prausnitzii are among the protective bacteria that play a significant role in maintaining this intestinal barrier[7]. Hyperglycemia in T2DM can disrupt this intestinal barrier, which causes gram negative bacterial products like lipopolysaccharides (LPS) to enter the systemic circulation, leading to endotoxemia, and further local and systemic inflammation[3,7].
Metabolism is the process used by the body to create energy from the food we eat, and metabolic diseases, such as type 2 diabetes and obesity, occur due to metabolic dysregulation. Metabolic syndrome refers to a set of risk factors including hyperglycemia, dyslipidemia, and obesity that increases the risk for CVD, T2DM, non-alcoholic fatty liver, and other diseases[8]. Animal studies have shown a causal link between the gut microbiome profile and metabolic syndrome[8]. A study in mice fed a 30-d high fat diet showed significant increase of bacteria of the phylum Firmicutes and Proteobacteria with reduction of Bacteroidetes and Verrucomicrobia[9]. Another study with high-glucose or high-fructose fed mice showed the gut microbiome of these mice to be significantly altered with an increase in Proteobacteria and decrease in Bacteroidetes[10].
The gut microbiota consume the host’s diet and produces certain metabolites which act on the host receptors and exert their endocrine effects, leading to hormone secretion, inflammation, and insulin resistance[11]. Studies have shown that these microbiota are sensitive to the host’s diet composition and the microbiome diversity changes with animal vs. plant-based diets[1]. The gut microbiome produces certain beneficial metabolites like short chain fatty acids (SCFA)[2], such as butyrate which promotes colonic health and is protective against T2DM and CVD[1,3,7,11]; propionate which promotes the release of glucagon like peptide-1 and peptide YY, which improves insulin sensitivity and weight[2]. Secondary bile acids are converted from primary bile acids by the gut microbes, which activate takeda G protein coupled receptor 5, increasing cyclic adenosine monophosphate production, improving insulin sensitivity, interacting with the farnesoid X receptor, pregnane X receptor and vitamin D receptor to regulate lipid metabolism and glucose metabolism, subsequently slowing the progression of CVD and T2DM[7,11]. Other favorable metabolites are esculin, anthocyanin, urolithin A, and enterolactone[3].
Research has also revealed some unfavorable metabolites produced by the gut microbiome, of which trimethylamine is the most prominent. Trimethylamine is oxidized to trimethylamine N-oxide which works by increasing low-density lipoprotein uptake in cells, reducing cholesterol excretion, and promoting recruitment of activated leukocytes and platelet aggregation, and thus, trimethylamine promotes atherosclerosis, thrombosis, CVD and diabetes[3,12]. In patients with chronic kidney disease (CKD), studies have shown that high indoxyl sulfate levels predict major adverse cardiac events, high P-cresyl sulfate levels correlate with CVD and all-cause mortality, and phenylacetylglutamine is associated with overall mortality and CVD[13,14]. Since all of the above mentioned metabolites are uremic toxins, CKD can increase the buildup and worsen the effects of these metabolites resulting in CVD progression[3].
Current research is directed towards finding treatment options for improving the number of beneficial gut microbes and to reduce the harmful ones with the use of probiotics and prebiotics[7]. Efforts are also underway to identify novel gut microbiome-host interactions, their associations and mechanisms leading to T2DM, CVD and to design new therapies to modulate these disease processes[11]. The purpose of this article is to summarize the use of microbiome supplementation to improve the abundance of the beneficial gut microbes and to reduce the harmful ones in patients with diabetes mellitus and metabolic syndrome.
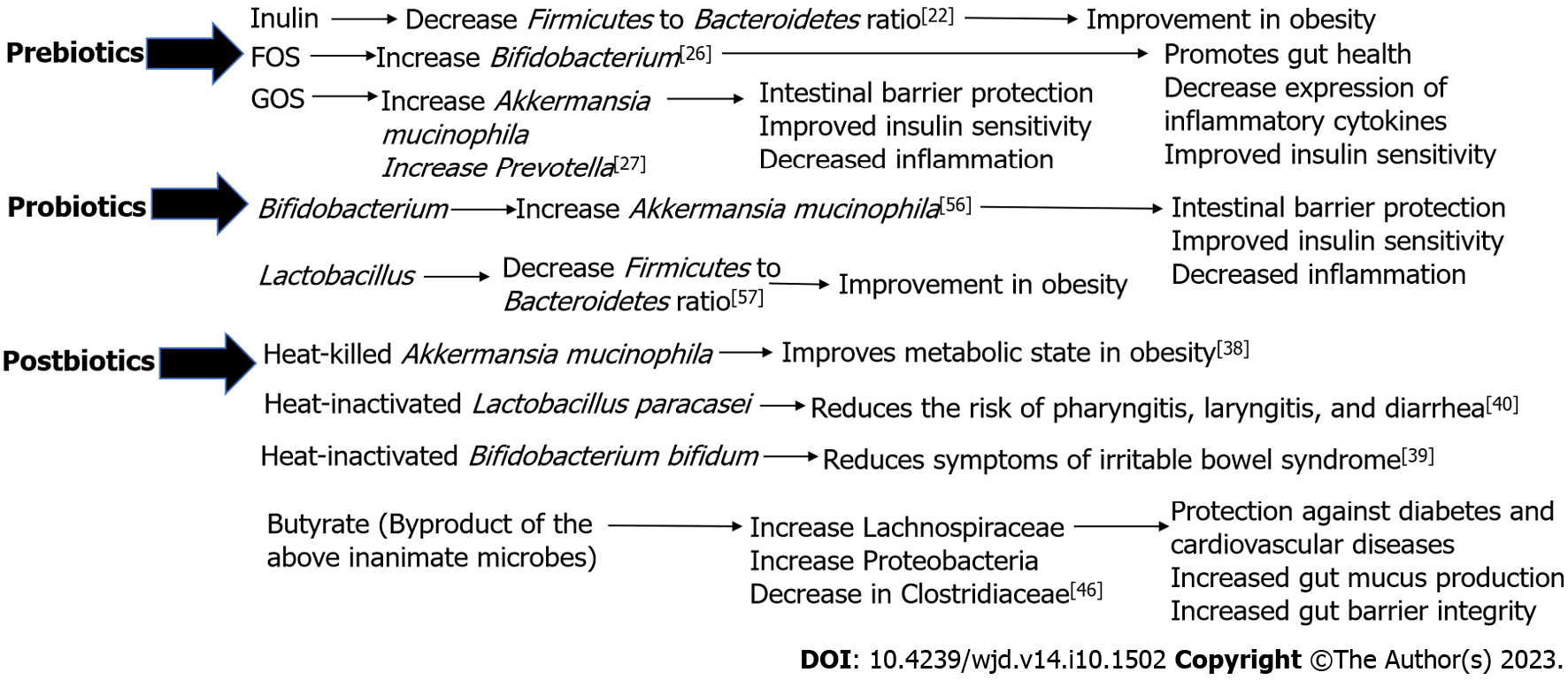
Microbiome supplementation has been found to alter the composition of the gut microbiome and possibly have effects on human health and diseases[15]. There are four types of supplements that are studied. In 2016, the International Scientific Association for Probiotics and Prebiotics (ISAPP) defined prebiotics as a substrate utilized selectively by microorganisms in the host and conferring a benefit to the host’s health[16]. The widely accepted scientific definition of probiotics as defined by an expert panel convened by the ISAPP in 2013 is “Live microorganisms that, when administered in adequate amounts, confer a health benefit on the host”[17]. In 2019, the ISAPP convened and defined synbiotics as, “a mixture comprising live microorganisms and substrate(s) selectively utilized by host microorganisms that confers a health benefit on the host”; thus, synbiotics are a combination of prebiotics and probiotics[18]. Lastly, in 2019, ISAPP defined postbiotics to be inanimate microorganisms with or without their components that confer a health benefit to the host[19]. This section will review the current data for each of these supplements and directions for future research. Table 1 summarizes the definitions and examples of the each of these supplements and we have summarized the similarities and differences between each supplements’ influence on metabolic syndrome in Table 2 and presented the same in a figure format in Figure 1.
| Category | Definition | Examples |
| Prebiotic | Non-digestible substances utilized by microbiota and confer a benefit to the host | Inulin |
| Lactulose | ||
| Fructooligosaccharides | ||
| Galactooligosaccharides | ||
| Probiotic | Live microorganisms that provide a benefit to the host | Bifidobacterium |
| Lactobacillus | ||
| Synbiotic | Prebiotics and probiotics taken together | |
| Postbiotic | Inanimate strains with or without their byproducts that provide a benefit to the host | Heat killed Akkermansia Mucinophila |
| Heat inactivated Lactobacillus paracasei | ||
| Heat-inactivated Bifidobacterium bifidum | ||
| Byproduct of the above inanimate strains: Butyrate and Proprionate |
| Category | Influence | ||
| Prebiotic | Inulin | Decrease Firmicutes to Bacteroidetes ratio[22] | Improvement in Obesity |
| Fructooligosaccharides | Increase Bifidobacterium[26] | Promotes gut health | |
| Decrease expression of inflammatory cytokines | |||
| Improved Insulin sensitivity | |||
| Galactooligosaccharides | Increase Akkermansia mucinophila. Increase in Prevotella[27] | Intestinal barrier protection | |
| Improved Insulin sensitivity | |||
| Decrease inflammation | |||
| Probiotic | Bifidobacterium | Increasing Akkermansia mucinophila[56] | Intestinal barrier protection |
| Improved Insulin sensitivity | |||
| Decrease inflammation | |||
| Lactobacillus | Decreasing Firmicutes to Bacteroidetes ratio[57] | Improvement in Obesity | |
| Postbiotic | Heat-killed Akkermansia mucinophila | Improves metabolic state in Obesity | |
| Heat-inactivated Lactobacillus paracasei | Reduces the risk of pharyngitis, laryngitis, and diarrhea | ||
| Heat-inactivated Bifidobacterium bifidum | Reduces symptoms of irritable bowel syndrome | ||
| Butyrate (SCFA produced by the inactive microbe) | Increasing Lachnospiraceae[46] | Protection against diabetes and cardio-vascular diseases | |
| Increasing Proteobacteria[46] | Increased gut mucus production | ||
| Decreasing Clostridiaceae[46] | Increased gut barrier integrity | ||
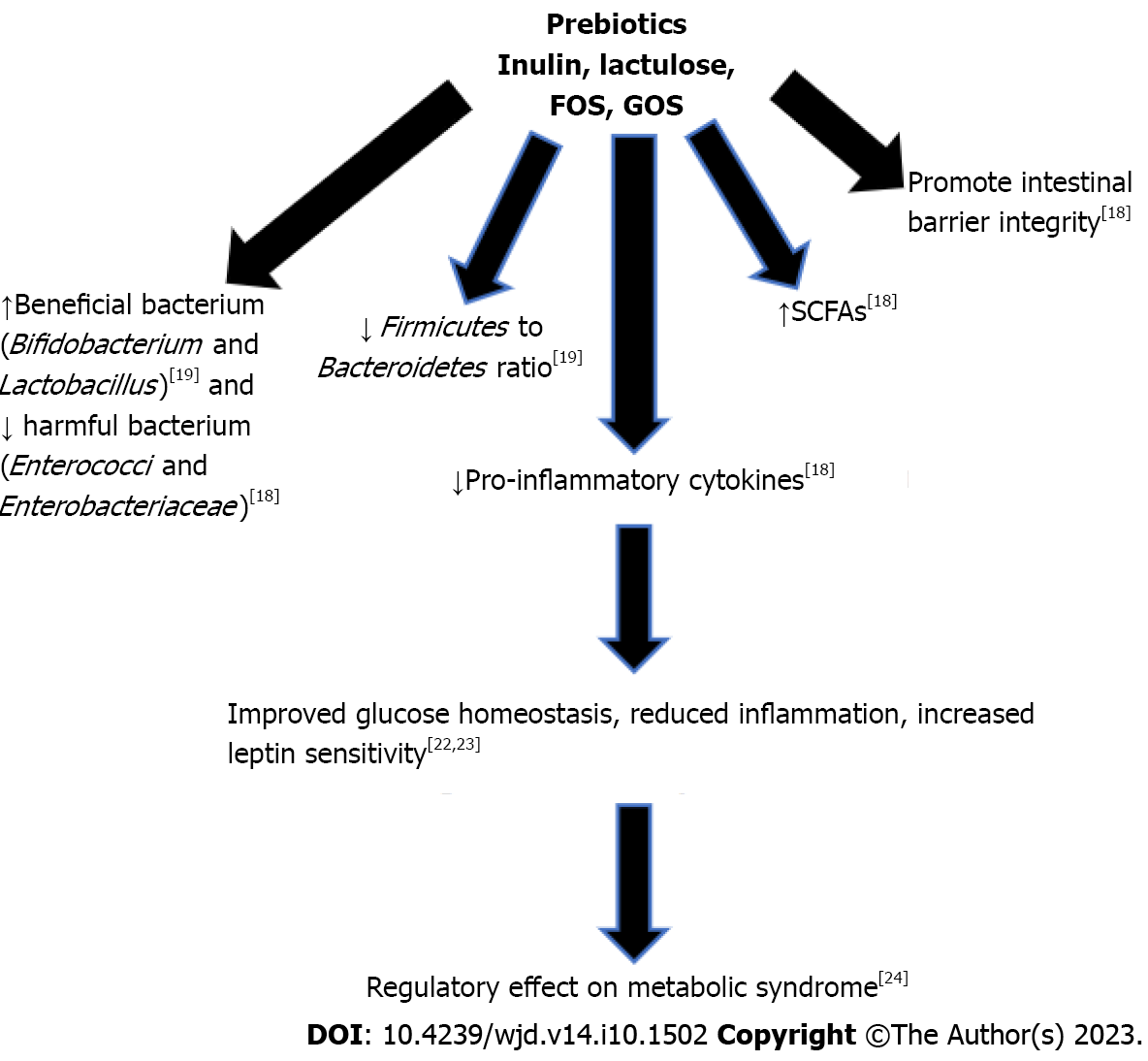
Prebiotics are consumable substances selectively utilized by microorganisms in the host and confer a benefit to the host[16]. Prebiotic effects have been studied in several metabolic diseases with animal studies, but few studies have been done on humans. Inulin, lactulose, fructooligosaccharides (FOS), and galactooligosaccharides (GOS) are the most widely known prebiotics[20]. Other prebiotics include human milk oligosaccharides, polydextrose, pectic oligosaccharides, arabinoxylans, and xylooligosaccharides[21]. Prebiotics confer a wide range of health benefits, including immune modulation through increased production of interleukins and immunoglobulins with reduction of pro-inflammatory interleukins and production of SCFAs, such as butyrate and acetate[21]. SCFAs indicate bacterial fermentation in the gastrointestinal tract and improve the health of the gut through mucus production, protecting against inflammation, and promoting the intestinal barrier integrity[21]. Production of SCFAs also results in a reduction on intestinal pH, inhibiting the growth of pathogenic bacteria[21].
Bacteria that promote gut health, such as Bifidobacterium and Lactobacillus, proliferate upon consumption of prebiotics[11]. Inulin oligofructose supplementation in mice fed a high fat diet showed a reduction in the Firmicutes to Bacteroidetes ratio[22], and reduction in the Firmicutes to Bacteroidetes ratio has been the hallmark of obesity[23,24]. A study done with a group of 10 elderly women over 19 days of inulin supplementation showed an increase in Bifidobacteria and decrease in Enterococci and Enterobacteriaceae[21], which is associated with a decreased risk of inflammatory bowel disease[25]. FOS supplementation in a rat model improved the gut microbiome by increasing Bifidobacterium[26]. Additional, fermentation of FOS generates SCFAs, decreasing the luminal pH and increasing the bioavailability of minerals in the gut[21]. GOS supplementation for prolonged periods in mice fed with a western diet led to increased abundance in Akkermansia mucinophila and Prevotella[27]. In another study, the activity of GOS was analyzed in sequencing fecal samples from humans after GOS administration, and the data showed an increased in Facecalibacterium prausnitzii and Bifidobacteria with a decrease in Bacteroides[21]. Facecalibacterium prausnitzii produces the SCFA butyrate and is associated with reduced inflammation[28]. Bifidobacteria promotes gut health, decreases expression of inflammatory cytokines, and improves insulin sensitivity[29]. Decrease in Bacteroides is beneficial to humans, as it is a pathogen common in anaerobic infections with significant antibiotic resistance[30].
Additional studies found that treatment with prebiotics improved glucose homeostasis and increased leptin sensitivity[31]. It also decreased inflammation by improving gut barrier integrity, thus decreasing the number of endotoxins able to leak from the gut lumen into the bloodstream[32]. They have also been shown to have a regulatory effect on metabolic disorders, especially those associated with obesity such as dyslipidemia, hypertension, diabetes, and liver steatosis[33]. Thus, several in vivo and in vitro studies have shown that prebiotics have beneficial impact on diabetes and obesity. Many prebiotics cause an increase in the growth of Lactobacillus and Bifidobacterium, but it is not fully understood how prebiotics cause these changes in the gut microbiome[21]. It is well-known that prebiotics are fermented by gut microbiota, leading the production of SCFAs, lowering the pH of the colon[21]. Figure 2 summarizes the beneficial effects of prebiotics leading to improved glucose homeostasis and reduced inflammation. Further research is necessary to elucidate the impact of prebiotics on the gut microbiome and the molecular signaling mechanisms of SCFAs.
Synbiotics are combinations of prebiotics and probiotics, consisting of a combination of live microorganisms and substances that are selectively utilized by the host microbiota to confer a benefit to the host[18]. The benefits of synbiotics are thought to come from the initial selection of beneficial commensal microbiome species and aiding these species in subsequent food processing and fermentation[34]. These include reduced oxidative stress on intestinal cells and overall decreased inflammation, thus maintaining the gut barrier[35]. Studies show that supplementation with synbiotics or probiotics may lead to beneficial reduction in fasting blood glucose (FBG), although the impact on FBG was more pronounced when multispecies probiotics were used instead of single species probiotics[36]. Current synbiotics include the most well-studied probiotics, including Bifidobacterium and Lactobacillus, which ferment indigestible sugars, such as FOS[34]. Thus, synbiotic administration of probiotics with FOS aims to increase the abundance of FOS fermentation products in the gut, such as lactic acid[34].
Many species rely on products from other species for survival, for example some species require lactic acid for substrate production and thus rely on lactic acid-producing species. This suggests that synbiotics will need to become more complex and involve multiple strains rather than just one, in addition to the prebiotics required for survival[34]. The effects of these supplements may also be altered by the individual’s characteristics[37]. Person-to-person variation in gene expression was shown in one study to be the main determinant for differences in transcriptomes created post-supplementation[37]. This may be a species-specific phenomenon or even location specific, i.e., in the duodenum but not in the jejunum; thus, further trials are required.
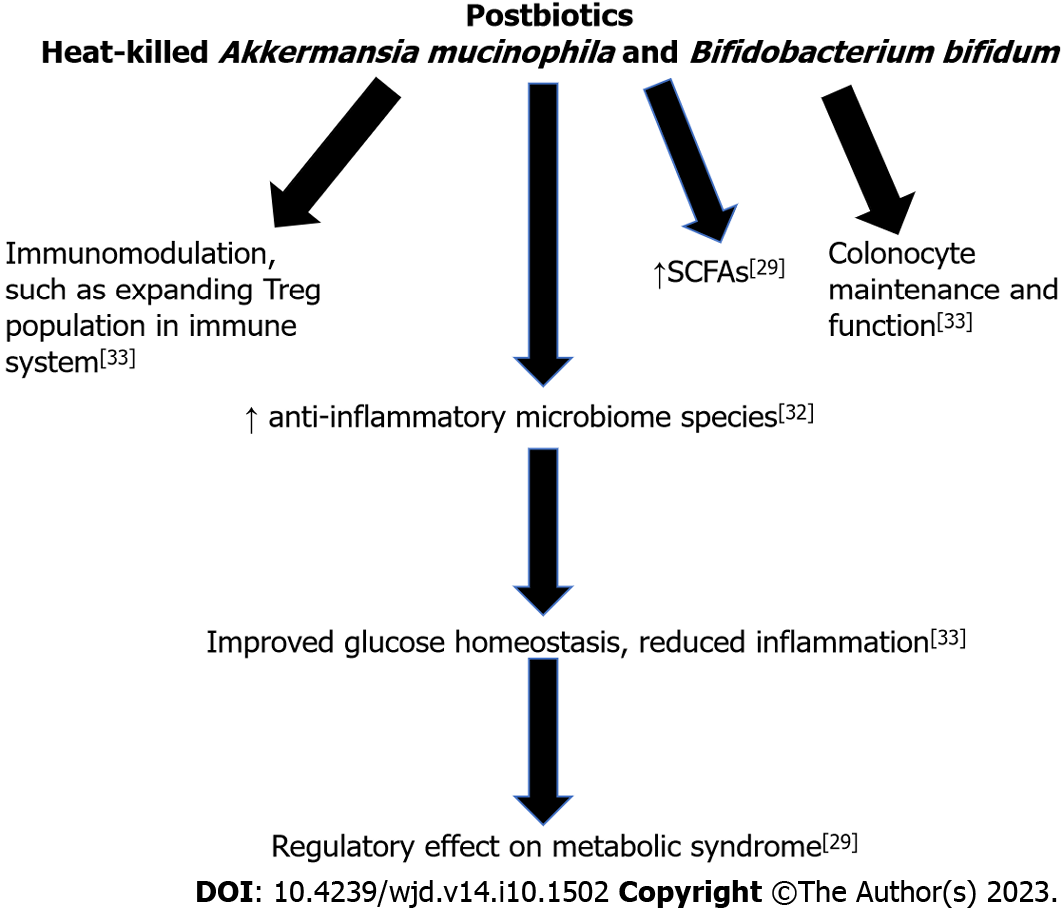
Postbiotics consist of inanimate microorganisms with or without their components and metabolites that confer a health benefit to the host[38]. Contrasting with probiotics, which consist of live microorganisms, postbiotics consist of microorganisms that are no longer alive, such as heat-killed Akkermansia mucinophila[38]. There are several challenges to the survival of probiotics during production and storage of food, such as reactions with chemical compounds, acidity, and storage temperature[38]. It has long been known that non-viable microbes in addition to their components and metabolites can have significant impact on health[38]. In one clinical trial, heat-inactivated Bifidobacterium bifidum was found significantly alleviate the symptoms of irritable bowel syndrome[39]. In a similar study, Akkermansia muciniphila was found to improve the metabolic state of obese and overweight participants in both its living and inactive forms[38]. In another systematic review, postbiotics were studied for the prevention and treatment of infectious diseases in children under five years of age, revealing treatment with heat-killed Lactobacillus acidophilus reduced the duration of diarrhea and heat-inactivated Lactobacillus paracasei reduced the risk of pharyngitis, laryngitis, and diarrhea[40].
Postbiotics are promising for the development of food supplements with longer stability in comparison to prebiotic supplements[38]. Additionally, postbiotics have the potential to broaden the spectrum of microbes used for supplementation, as microbes that could not be administered live due to safety concerns can be administered in the inanimate form. The mechanism of action of postbiotics is due to both the components of the inactivated microbes and the metabolites produced by the microbes, such as SCFAs[38].
An inverse relationship between decline in anti-inflammatory microbiome species and abnormal SCFA production has been demonstrated[41]. SCFAs, such as butyrate and propionate, are among the metabolites produced from inactive microbes. Gut microbiome produced SCFA have been shown to have strong effects on metabolic and cardiovascular health through a variety of tissue-specific mechanisms including appetite regulation, glucose homeostasis and metabolism, proper gut barrier and colonocyte maintenance and function, and immunomodulation[41]. For example, butyrate is integral in colonocytes for energy production and expanding the regulatory T cell population in the immune system, while propionate is suggested to have a role in gluconeogenesis[42]. However, increase in acetate production has been shown to activate glucose-stimulated insulin secretion, increase ghrelin secretion and hyperphagia, leading to obesity and related diseases[43]. This suggests that to develop a treatment protocol, the specific proportion of postbiotics in a patient will need to be examined to allow for appropriate adjustment. Increased production of acetate has been found in obesity and decreased production of butyrate and propionate is seen in T2DM[44,45]. In mice studies, butyrate was shown to be associated with increased production of Lachnospiraceae and Proteobacteria and decreased production of Clostridiaceae[46].
However, the mechanism directly responsible remains elusive, and some results from animal models conflict with results from human trials. Many metabolic diseases are associated with a chronic state of low-grade inflammation. The maintenance of the gut barrier is also critical for reducing the amount of pro-inflammatory bacterial byproducts that can cross into the bloodstream, thus potentially decreasing the level of inflammation in the body. Specifically, a high fat diet reduces expression of tight junction genes, thus leading to a leaky barrier[47]. This allows inflammatory bacterial byproducts, such as LPS, to circulate in the body leading to an inflammatory response[48]. Hyperglycemia can also increase this leakiness and cause hyperpermeability, leading to a similar inflammatory phenomenon[49]. However, these findings are mainly in animal studies and further studies in humans are required. Additionally, there is a need for studies on the impact of inanimate microbes on the host without associated metabolites to determine the extent that health benefits are conferred. There is also need for additional research on the mechanisms that are driving the benefits of postbiotics. Figure 3 summarizes the beneficial effects of postbiotics, leading to improved glucose homeostasis and reduced inflammation.
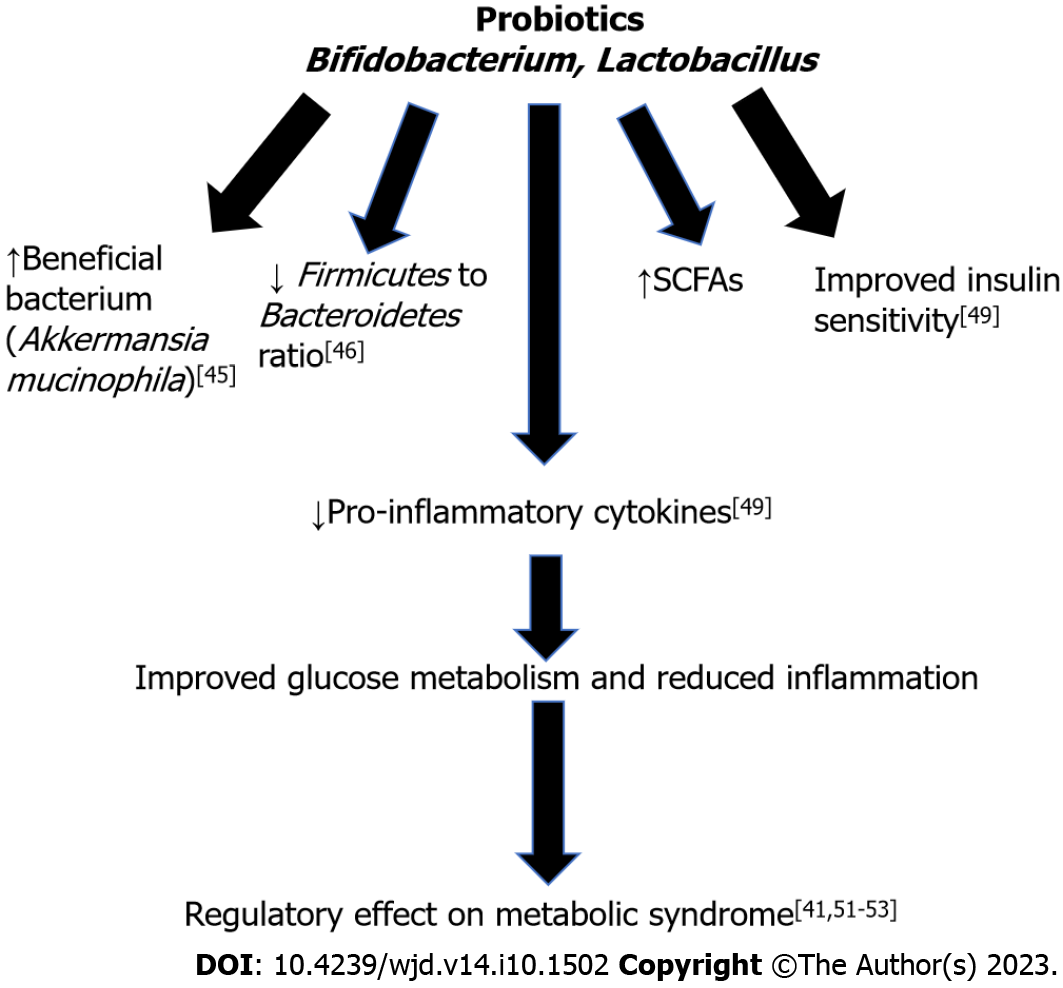
Probiotics are live microorganisms that confer a benefit to the host when administered[17]. Bifidobacterium and Lactobacillus are the two most widely known probiotics. Various studies in animals have shown their benefits in improving gut microbiome composition[50,51]. Probiotic administration has been shown to be potential therapeutic target for metabolic syndrome prevention and treatment[52]. There is a fine balance between the host’s immune system and gut microbiome, and imbalance can lead to systemic inflammation through passage of bacteria and bacterial fragments, such as LPS, through the gut barrier and into systemic circulation[52]. Chronic systemic inflammation can lead to the development of insulin resistance and obesity[52].
Additionally, many diseases have been found to have microbial dysbiosis either from an overgrowth of pathogenic species or a loss of microbiome diversity[53]. However, it should be noted that there is not a clear definition of a healthy gut microbiome composition, so the term “dysbiosis” is inherently vague[45]. This change in microbial composition is found to be associated with increased inflammation, specifically in obese patients since low-grade inflammation is a common finding in many metabolic disorders[45]. This suggests that microbiome composition affects the inflammatory state of people. An increase of pro-inflammatory bacterial species has also been found in patients with T2DM, especially with a decrease of anti-inflammatory species[41].
The microbiome present in obese individuals has been found to be different from that of lean individuals though specific differences are difficult to qualify[54]. In a study with obese and lean adolescents, it was found that a lower amount of Bacteroidetes and higher proportion of Firmicutes was associated with obesity[55]. The microbiome of obese individuals has been shown in animal models to extract more energy from the diet, and this phenomenon still occurs when the microbiome from obese mice is transplanted into lean mice[54]. Bifidobacterium supplementation in diet-induced obese and insulin resistant mice showed an increase in Akkermansia mucinophila[56]. In another study, Lactobacillus supplementation in high-fat diet induced hypertensive mice showed a reduction in the ratio of Firmicutes to Bacteroidetes[57].
Probiotics have been found to have an influence on the expression of inflammation-related genes and proteins[58]. Many animal studies have shown interactions between the gut microbiome and the immune system[45]. These studies reduced the gene expression of immune system components with known or theorized links to metabolic dysfunction. Researchers then studied the effects or interactions of these mutation with the gut microbiome. Knock out of toll-like receptor 5 (TLR5) in mice was found to cause the hallmark features of metabolic syndrome in correlation with changes to the gut microbiome and findings of colitis[59]. Upon transfer of the microbiome from the knock-out mice to wild-type germ-free mice, metabolic dysfunction was also transferred, leading to hyperlipidemia, hypertension, and insulin resistance. Food restriction was able to prevent obesity but had no effect on insulin resistance, suggesting that the TLR5 and subsequent microbiome changes have a metabolic effect. Additionally, deletion of myeloid differentiation factor 88 and a high fat diet induced hyperglycemia, leading to metabolic syndrome in knock-out mice along with an increase in bacterial translocation across the intestinal barrier[29]. The mice were then given Bifidobacterium animalis subsp. lactis 420 (B420) as a probiotic. The result was a general normalization of gut microbiome composition, a decrease in the expression of major inflammatory cytokines, and a complete normalization of insulin sensitivity and levels, although glucose metabolism was only moderately affected[29]. However, data on specific benefits conflict from study to study. Improvements in glucose metabolism is more significant in patients with T2DM, and some studies report little effect on cholesterol and lipid levels[45]. Supplementation of Akkermansia muciniphila has been found in rodents and humans to improve insulin sensitivity and decrease inflammation[60]. The findings are less prominent in humans but indicate the supplement’s clinical potential. Another study with 40 participants with insulin resistance were placed in a double-blind trial and given either Akkermansia muciniphila or a placebo, and the study showed reduction in inflammatory markers and improved insulin sensitivity in the Akkermansia muciniphila group[61].
Only a small number of studies have been conducted to analyze the effect of probiotic administration on weight and glycemic control in humans. In one study, 87 subjects with higher body mass index (BMI) (24.2-30.7 kg/m2) were randomly assigned to a group received fermented milk containing Lactobacillus (LG2055) or fermented milk without Lactobacillus for 12 wk. It was found that the group receiving the milk containing LG2055 experienced a significant reduction in abdominal visceral and subcutaneous fat and BMI[62]. Another study showed probiotic yogurt consumption reduced FBG and HgbA1c in patients with type 2 diabetes[63]. A double-blind trial with 21 participants showed administration of Lactobacillus reuteri improved insulin and incretin secretion[64]. Thus, the results from clinical experiments are encouraging, but larger trials are needed to confirm the effect of probiotics on improving insulin sensitivity and weight loss[65]. Figure 4 summarizes the beneficial effects of probiotics, leading to improved glucose homeostasis and reduced inflammation.
Future research is needed to confirm the clinical efficacy of microbiome supplementation. Several times, studies have reached opposing or ambiguous conclusions, even though the research was of high methodological quality[58]. This may be due to several factors including data collection methods, different analysis parameters and metrics, and varied methods of interpretation[58]. Some studies used self-reporting surveys to measure patient quality of life and psychosocial effects. Others looked at lab values that may not necessarily have significance clinically, such as inflammatory markers or glucose metabolism protein levels in generally healthy individuals. Some studies had animal models while others involved human subjects and may have been observational or randomized with placebo controls. There is also the question of funding and conflicts of interest, as some studies are funded or linked to probiotic companies. While that does not necessarily bias the project, independent research should be a focus in the future.
Additionally, research on the effects of specific strains is lacking and may even vary from stain to strain, thus weakening the argument for the use of specific strains in a project[58]. One study found that both mice and humans had colonization resistance to probiotics based on the current composition of their gut microbiome, and that in humans this resistance varies from person to person because microbiome composition is individualized based on person-specific needs, geographic region, and diet among other factors[66]. In fact, many of the live probiotic strains were found to still be viable in stool samples after passage through the GI tract[66], and it remains unclear if the colonization that does occur persists after supplementation ceases. This contradicts in vitro studies in which probiotics were able to adhere to human GI mucosal cells[67], indicating that lab-based work may be a poor predictor of efficacy in human subjects[58]. In vitro studies in general may be poor models for this topic of research since it does not include in vivo signaling and factors that may play an important role in colonization and efficacy[58]. This could also contribute to conflicting results and would require further in vivo studies.
Additional studies have also looked at whether the effects of probiotics change depending on a person’s specific microbiome composition and have found that it does make a difference. Song et al[68] classified fifty obese but otherwise healthy subjects based on the ratio of two bacterial species, Prevotella and Bacteroidetes, two of the major enterotypes[68]. The administration of probiotics improved obesity-related markers, but the efficacy was greater in the Prevotella-dominant enterotype. This, along with colonization resistance, could explain why previous studies have found such varied results and accounting for these differences could help reconcile conflicting data[66]. This highlights the need for a patient-centered protocol rather than general supplementation.
Postbiotics is the newest area of research and thus will require the most work in future studies. There is the potential to alter bacteria to produce new biological compounds. In one study using a mouse model of alcoholic liver disease, Lactobacillus reuteri was engineered to produce interleukin-22 (IL-22), an anti-inflammatory cytokine, after it was determined that chronic alcohol use reduces intestinal production of IL-22[69]. IL-22 has been found in previous studies to protect against atherosclerosis and CVD[70] as well as protect against beta cell stress and normalize hyperglycemia and insulin levels[71]. The increased levels of IL-22 allowed for increased expression of the regenerating islet-derived genes (REG3-gamma gene), which creates a protein that prevents bacterial translocation across the gut barrier. This reduced ethanol-induced steatohepatitis, a direct hepato-protective effect made possible by genetically altered probiotic supplementation. Through this, we have found that it is possible to manipulate commensal bacteria to fit the roles needed in the patient and can treat an enormous variety of metabolic diseases.
Meta analyses may help resolve some of the ambiguity but are not impervious to biases[58]. They may include studies that involve different strains of bacteria and thus are difficult to compare. They may also include outlier studies that skew the data and conclusions or be diluted by papers without significant findings. Therefore, efforts should be placed in developing randomized, large-scale, and high-quality experiments and clinical trials to assess the use of prebiotics, probiotics, and postbiotics to modify the gut microbiome and affect various metabolic syndromes.
The relationship between human health and the microbiome has piqued researchers' curiosity in the last decade. Our knowledge of the gut microbiome's composition and functions has considerably improved over the past several years due to rapid advancements in metagenomic sequencing techniques. As a result, it is evident that almost no area of host physiology is fully immune to the effects of gut microbes and their products. Indeed, the gut microbiota's influence extends beyond the gastrointestinal tract's traditional digestion function to include altering the physiology of other organ systems such as the liver, adipose tissue, lung, and brain. With better insight into the interactions between the host and microbiota, human gut microbiome supplementation has emerged as a promising novel therapeutic target. Current research is directed towards finding treatment options for improving the number of beneficial gut microbiota and to reduce the harmful ones with the use of prebiotics, probiotics, synbiotics, and postbiotics. Efforts are also underway to identify novel gut microbiota-host interactions, their mechanisms, and associations with T2DM, CVD and to design new therapies to modulate these disease processes.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Endocrinology and metabolism
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Ma JH, China; Sun H, China; Horowitz M, Australia S-Editor: Fan JR L-Editor: A P-Editor: Fan JR
| 1. | Dabke K, Hendrick G, Devkota S. The gut microbiome and metabolic syndrome. J Clin Invest. 2019;129:4050-4057. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 450] [Article Influence: 90.0] [Reference Citation Analysis (0)] |
| 2. | Kant R, Chandra L, Verma V, Nain P, Bello D, Patel S, Ala S, Chandra R, Antony MA. Gut microbiota interactions with anti-diabetic medications and pathogenesis of type 2 diabetes mellitus. World J Methodol. 2022;12:246-257. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 11] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 3. | Zhao Y, Wang Z. Gut microbiome and cardiovascular disease. Curr Opin Cardiol. 2020;35:207-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 4. | Groop L. Genetics of the metabolic syndrome. Br J Nutr. 2000;83 Suppl 1:S39-S48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 138] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 5. | Gluckman PD, Hanson MA. The developmental origins of the metabolic syndrome. Trends Endocrinol Metab. 2004;15:183-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 457] [Cited by in RCA: 423] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 6. | Edwardson CL, Gorely T, Davies MJ, Gray LJ, Khunti K, Wilmot EG, Yates T, Biddle SJ. Association of sedentary behaviour with metabolic syndrome: a meta-analysis. PLoS One. 2012;7:e34916. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 315] [Cited by in RCA: 354] [Article Influence: 27.2] [Reference Citation Analysis (0)] |
| 7. | Zhou Z, Sun B, Yu D, Zhu C. Gut Microbiota: An Important Player in Type 2 Diabetes Mellitus. Front Cell Infect Microbiol. 2022;12:834485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 139] [Article Influence: 46.3] [Reference Citation Analysis (0)] |
| 8. | Wang PX, Deng XR, Zhang CH, Yuan HJ. Gut microbiota and metabolic syndrome. Chin Med J (Engl). 2020;133:808-816. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 130] [Article Influence: 32.5] [Reference Citation Analysis (0)] |
| 9. | Tomas J, Mulet C, Saffarian A, Cavin JB, Ducroc R, Regnault B, Kun Tan C, Duszka K, Burcelin R, Wahli W, Sansonetti PJ, Pédron T. High-fat diet modifies the PPAR-γ pathway leading to disruption of microbial and physiological ecosystem in murine small intestine. Proc Natl Acad Sci U S A. 2016;113:E5934-E5943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 185] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 10. | Do MH, Lee E, Oh MJ, Kim Y, Park HY. High-Glucose or -Fructose Diet Cause Changes of the Gut Microbiota and Metabolic Disorders in Mice without Body Weight Change. Nutrients. 2018;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 248] [Cited by in RCA: 302] [Article Influence: 43.1] [Reference Citation Analysis (0)] |
| 11. | Massey W, Brown JM. The Gut Microbial Endocrine Organ in Type 2 Diabetes. Endocrinology. 2021;162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 12. | Wang Z, Klipfell E, Bennett BJ, Koeth R, Levison BS, Dugar B, Feldstein AE, Britt EB, Fu X, Chung YM, Wu Y, Schauer P, Smith JD, Allayee H, Tang WH, DiDonato JA, Lusis AJ, Hazen SL. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature. 2011;472:57-63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4469] [Cited by in RCA: 4049] [Article Influence: 289.2] [Reference Citation Analysis (0)] |
| 13. | Wang CH, Cheng ML, Liu MH, Shiao MS, Hsu KH, Huang YY, Lin CC, Lin JF. Increased p-cresyl sulfate level is independently associated with poor outcomes in patients with heart failure. Heart Vessels. 2016;31:1100-1108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 14. | Poesen R, Claes K, Evenepoel P, de Loor H, Augustijns P, Kuypers D, Meijers B. Microbiota-Derived Phenylacetylglutamine Associates with Overall Mortality and Cardiovascular Disease in Patients with CKD. J Am Soc Nephrol. 2016;27:3479-3487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 154] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 15. | Li HY, Zhou DD, Gan RY, Huang SY, Zhao CN, Shang A, Xu XY, Li HB. Effects and Mechanisms of Probiotics, Prebiotics, Synbiotics, and Postbiotics on Metabolic Diseases Targeting Gut Microbiota: A Narrative Review. Nutrients. 2021;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 211] [Article Influence: 52.8] [Reference Citation Analysis (0)] |
| 16. | Gibson GR, Hutkins R, Sanders ME, Prescott SL, Reimer RA, Salminen SJ, Scott K, Stanton C, Swanson KS, Cani PD, Verbeke K, Reid G. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol. 2017;14:491-502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2256] [Cited by in RCA: 3189] [Article Influence: 398.6] [Reference Citation Analysis (0)] |
| 17. | Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, Morelli L, Canani RB, Flint HJ, Salminen S, Calder PC, Sanders ME. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11:506-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4055] [Cited by in RCA: 5549] [Article Influence: 504.5] [Reference Citation Analysis (2)] |
| 18. | Swanson KS, Gibson GR, Hutkins R, Reimer RA, Reid G, Verbeke K, Scott KP, Holscher HD, Azad MB, Delzenne NM, Sanders ME. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of synbiotics. Nat Rev Gastroenterol Hepatol. 2020;17:687-701. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 326] [Cited by in RCA: 732] [Article Influence: 146.4] [Reference Citation Analysis (0)] |
| 19. | Salminen S, Collado MC, Endo A, Hill C, Lebeer S, Quigley EMM, Sanders ME, Shamir R, Swann JR, Szajewska H, Vinderola G. The International Scientific Association of Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat Rev Gastroenterol Hepatol. 2021;18:649-667. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 304] [Cited by in RCA: 990] [Article Influence: 247.5] [Reference Citation Analysis (1)] |
| 20. | Umu ÖCO, Rudi K, Diep DB. Modulation of the gut microbiota by prebiotic fibres and bacteriocins. Microb Ecol Health Dis. 2017;28:1348886. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 70] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 21. | Megur A, Daliri EB, Baltriukienė D, Burokas A. Prebiotics as a Tool for the Prevention and Treatment of Obesity and Diabetes: Classification and Ability to Modulate the Gut Microbiota. Int J Mol Sci. 2022;23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 56] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 22. | Kumar SA, Ward LC, Brown L. Inulin oligofructose attenuates metabolic syndrome in high-carbohydrate, high-fat diet-fed rats. Br J Nutr. 2016;116:1502-1511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 47] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 23. | Ley RE, Turnbaugh PJ, Klein S, Gordon JI. Microbial ecology: human gut microbes associated with obesity. Nature. 2006;444:1022-1023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7222] [Cited by in RCA: 6407] [Article Influence: 337.2] [Reference Citation Analysis (0)] |
| 24. | De Filippo C, Cavalieri D, Di Paola M, Ramazzotti M, Poullet JB, Massart S, Collini S, Pieraccini G, Lionetti P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci U S A. 2010;107:14691-14696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3584] [Cited by in RCA: 4027] [Article Influence: 268.5] [Reference Citation Analysis (0)] |
| 25. | Baldelli V, Scaldaferri F, Putignani L, Del Chierico F. The Role of Enterobacteriaceae in Gut Microbiota Dysbiosis in Inflammatory Bowel Diseases. Microorganisms. 2021;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 185] [Article Influence: 46.3] [Reference Citation Analysis (0)] |
| 26. | Klancic T, Laforest-Lapointe I, Choo A, Nettleton JE, Chleilat F, Noye Tuplin EW, Alukic E, Cho NA, Nicolucci AC, Arrieta MC, Reimer RA. Prebiotic Oligofructose Prevents Antibiotic-Induced Obesity Risk and Improves Metabolic and Gut Microbiota Profiles in Rat Dams and Offspring. Mol Nutr Food Res. 2020;64:e2000288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 27. | Mistry RH, Liu F, Borewicz K, Lohuis MAM, Smidt H, Verkade HJ, Tietge UJF. Long-Term β-galacto-oligosaccharides Supplementation Decreases the Development of Obesity and Insulin Resistance in Mice Fed a Western-Type Diet. Mol Nutr Food Res. 2020;64:e1900922. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 28. | Maioli TU, Borras-Nogues E, Torres L, Barbosa SC, Martins VD, Langella P, Azevedo VA, Chatel JM. Possible Benefits of Faecalibacterium prausnitzii for Obesity-Associated Gut Disorders. Front Pharmacol. 2021;12:740636. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 85] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 29. | Amar J, Chabo C, Waget A, Klopp P, Vachoux C, Bermúdez-Humarán LG, Smirnova N, Bergé M, Sulpice T, Lahtinen S, Ouwehand A, Langella P, Rautonen N, Sansonetti PJ, Burcelin R. Intestinal mucosal adherence and translocation of commensal bacteria at the early onset of type 2 diabetes: molecular mechanisms and probiotic treatment. EMBO Mol Med. 2011;3:559-572. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 560] [Cited by in RCA: 625] [Article Influence: 44.6] [Reference Citation Analysis (0)] |
| 30. | Wexler HM. Bacteroides: the good, the bad, and the nitty-gritty. Clin Microbiol Rev. 2007;20:593-621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1141] [Cited by in RCA: 1506] [Article Influence: 83.7] [Reference Citation Analysis (0)] |
| 31. | Everard A, Lazarevic V, Derrien M, Girard M, Muccioli GG, Neyrinck AM, Possemiers S, Van Holle A, François P, de Vos WM, Delzenne NM, Schrenzel J, Cani PD. Responses of gut microbiota and glucose and lipid metabolism to prebiotics in genetic obese and diet-induced leptin-resistant mice. Diabetes. 2011;60:2775-2786. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 755] [Cited by in RCA: 822] [Article Influence: 58.7] [Reference Citation Analysis (0)] |
| 32. | Cani PD, Bibiloni R, Knauf C, Waget A, Neyrinck AM, Delzenne NM, Burcelin R. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes. 2008;57:1470-1481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3224] [Cited by in RCA: 3542] [Article Influence: 208.4] [Reference Citation Analysis (0)] |
| 33. | Cani PD, Delzenne NM. The role of the gut microbiota in energy metabolism and metabolic disease. Curr Pharm Des. 2009;15:1546-1558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 625] [Cited by in RCA: 650] [Article Influence: 40.6] [Reference Citation Analysis (0)] |
| 34. | Gurry T. Synbiotic approaches to human health and well-being. Microb Biotechnol. 2017;10:1070-1073. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 35. | Kahlert S, Junnikkala S, Renner L, Hynönen U, Hartig R, Nossol C, Barta-Böszörményi A, Dänicke S, Souffrant WB, Palva A, Rothkötter HJ, Kluess J. Physiological Concentration of Exogenous Lactate Reduces Antimycin A Triggered Oxidative Stress in Intestinal Epithelial Cell Line IPEC-1 and IPEC-J2 In Vitro. PLoS One. 2016;11:e0153135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 36. | Nikbakht E, Khalesi S, Singh I, Williams LT, West NP, Colson N. Effect of probiotics and synbiotics on blood glucose: a systematic review and meta-analysis of controlled trials. Eur J Nutr. 2018;57:95-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 89] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 37. | van Baarlen P, Troost F, van der Meer C, Hooiveld G, Boekschoten M, Brummer RJ, Kleerebezem M. Human mucosal in vivo transcriptome responses to three lactobacilli indicate how probiotics may modulate human cellular pathways. Proc Natl Acad Sci U S A. 2011;108 Suppl 1:4562-4569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 231] [Cited by in RCA: 246] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 38. | Vinderola G, Sanders ME, Salminen S. The Concept of Postbiotics. Foods. 2022;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 134] [Article Influence: 44.7] [Reference Citation Analysis (0)] |
| 39. | Andresen V, Gschossmann J, Layer P. Heat-inactivated Bifidobacterium bifidum MIMBb75 (SYN-HI-001) in the treatment of irritable bowel syndrome: a multicentre, randomised, double-blind, placebo-controlled clinical trial. Lancet Gastroenterol Hepatol. 2020;5:658-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 111] [Article Influence: 22.2] [Reference Citation Analysis (0)] |
| 40. | Malagón-Rojas JN, Mantziari A, Salminen S, Szajewska H. Postbiotics for Preventing and Treating Common Infectious Diseases in Children: A Systematic Review. Nutrients. 2020;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 77] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 41. | Qin J, Li Y, Cai Z, Li S, Zhu J, Zhang F, Liang S, Zhang W, Guan Y, Shen D, Peng Y, Zhang D, Jie Z, Wu W, Qin Y, Xue W, Li J, Han L, Lu D, Wu P, Dai Y, Sun X, Li Z, Tang A, Zhong S, Li X, Chen W, Xu R, Wang M, Feng Q, Gong M, Yu J, Zhang Y, Zhang M, Hansen T, Sanchez G, Raes J, Falony G, Okuda S, Almeida M, LeChatelier E, Renault P, Pons N, Batto JM, Zhang Z, Chen H, Yang R, Zheng W, Yang H, Wang J, Ehrlich SD, Nielsen R, Pedersen O, Kristiansen K. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature. 2012;490:55-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3971] [Cited by in RCA: 4828] [Article Influence: 371.4] [Reference Citation Analysis (1)] |
| 42. | Chambers ES, Preston T, Frost G, Morrison DJ. Role of Gut Microbiota-Generated Short-Chain Fatty Acids in Metabolic and Cardiovascular Health. Curr Nutr Rep. 2018;7:198-206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 289] [Cited by in RCA: 439] [Article Influence: 73.2] [Reference Citation Analysis (0)] |
| 43. | Perry RJ, Peng L, Barry NA, Cline GW, Zhang D, Cardone RL, Petersen KF, Kibbey RG, Goodman AL, Shulman GI. Acetate mediates a microbiome-brain-β-cell axis to promote metabolic syndrome. Nature. 2016;534:213-217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 724] [Cited by in RCA: 950] [Article Influence: 105.6] [Reference Citation Analysis (1)] |
| 44. | Sanna S, van Zuydam NR, Mahajan A, Kurilshikov A, Vich Vila A, Võsa U, Mujagic Z, Masclee AAM, Jonkers DMAE, Oosting M, Joosten LAB, Netea MG, Franke L, Zhernakova A, Fu J, Wijmenga C, McCarthy MI. Causal relationships among the gut microbiome, short-chain fatty acids and metabolic diseases. Nat Genet. 2019;51:600-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 784] [Cited by in RCA: 1008] [Article Influence: 168.0] [Reference Citation Analysis (0)] |
| 45. | Scheithauer TPM, Rampanelli E, Nieuwdorp M, Vallance BA, Verchere CB, van Raalte DH, Herrema H. Gut Microbiota as a Trigger for Metabolic Inflammation in Obesity and Type 2 Diabetes. Front Immunol. 2020;11:571731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 378] [Article Influence: 75.6] [Reference Citation Analysis (0)] |
| 46. | Jiminez JA, Uwiera TC, Abbott DW, Uwiera RRE, Inglis GD. Butyrate Supplementation at High Concentrations Alters Enteric Bacterial Communities and Reduces Intestinal Inflammation in Mice Infected with Citrobacter rodentium. mSphere. 2017;2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 87] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 47. | Rohr MW, Narasimhulu CA, Rudeski-Rohr TA, Parthasarathy S. Negative Effects of a High-Fat Diet on Intestinal Permeability: A Review. Adv Nutr. 2020;11:77-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 329] [Cited by in RCA: 395] [Article Influence: 79.0] [Reference Citation Analysis (0)] |
| 48. | Brenchley JM, Douek DC. Microbial translocation across the GI tract. Annu Rev Immunol. 2012;30:149-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 276] [Cited by in RCA: 342] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 49. | Thaiss CA, Levy M, Grosheva I, Zheng D, Soffer E, Blacher E, Braverman S, Tengeler AC, Barak O, Elazar M, Ben-Zeev R, Lehavi-Regev D, Katz MN, Pevsner-Fischer M, Gertler A, Halpern Z, Harmelin A, Aamar S, Serradas P, Grosfeld A, Shapiro H, Geiger B, Elinav E. Hyperglycemia drives intestinal barrier dysfunction and risk for enteric infection. Science. 2018;359:1376-1383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 416] [Cited by in RCA: 619] [Article Influence: 88.4] [Reference Citation Analysis (0)] |
| 50. | Cao L, Yang XJ, Li ZJ, Sun FF, Wu XH, Yao JH. Reduced lesions in chickens with Clostridium perfringens-induced necrotic enteritis by Lactobacillus fermentum 1.20291. Poult Sci. 2012;91:3065-3071. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 67] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 51. | Chaves BD, Brashears MM, Nightingale KK. Applications and safety considerations of Lactobacillus salivarius as a probiotic in animal and human health. J Appl Microbiol. 2017;123:18-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 60] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 52. | He M, Shi B. Gut microbiota as a potential target of metabolic syndrome: the role of probiotics and prebiotics. Cell Biosci. 2017;7:54. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 200] [Article Influence: 25.0] [Reference Citation Analysis (1)] |
| 53. | Santos VM, Brito AKP, Amorim AT, Souza IR, Santos MB, Campos GB, Dos Santos DC, Júnior ACRB, Santana JM, Santos DB, Mancini MC, Timenetsky J, Marques LM. Evaluation of fecal microbiota and its correlation with inflammatory, hormonal, and nutritional profiles in women. Braz J Microbiol. 2022;53:1001-1009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 54. | Ridaura VK, Faith JJ, Rey FE, Cheng J, Duncan AE, Kau AL, Griffin NW, Lombard V, Henrissat B, Bain JR, Muehlbauer MJ, Ilkayeva O, Semenkovich CF, Funai K, Hayashi DK, Lyle BJ, Martini MC, Ursell LK, Clemente JC, Van Treuren W, Walters WA, Knight R, Newgard CB, Heath AC, Gordon JI. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science. 2013;341:1241214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2415] [Cited by in RCA: 2722] [Article Influence: 226.8] [Reference Citation Analysis (0)] |
| 55. | Ferrer M, Ruiz A, Lanza F, Haange SB, Oberbach A, Till H, Bargiela R, Campoy C, Segura MT, Richter M, von Bergen M, Seifert J, Suarez A. Microbiota from the distal guts of lean and obese adolescents exhibit partial functional redundancy besides clear differences in community structure. Environ Microbiol. 2013;15:211-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 171] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 56. | Alard J, Lehrter V, Rhimi M, Mangin I, Peucelle V, Abraham AL, Mariadassou M, Maguin E, Waligora-Dupriet AJ, Pot B, Wolowczuk I, Grangette C. Beneficial metabolic effects of selected probiotics on diet-induced obesity and insulin resistance in mice are associated with improvement of dysbiotic gut microbiota. Environ Microbiol. 2016;18:1484-1497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 125] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 57. | Hsu CN, Hou CY, Chan JYH, Lee CT, Tain YL. Hypertension Programmed by Perinatal High-Fat Diet: Effect of Maternal Gut Microbiota-Targeted Therapy. Nutrients. 2019;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 74] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 58. | Suez J, Zmora N, Segal E, Elinav E. The pros, cons, and many unknowns of probiotics. Nat Med. 2019;25:716-729. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 405] [Cited by in RCA: 837] [Article Influence: 139.5] [Reference Citation Analysis (0)] |
| 59. | Vijay-Kumar M, Aitken JD, Carvalho FA, Cullender TC, Mwangi S, Srinivasan S, Sitaraman SV, Knight R, Ley RE, Gewirtz AT. Metabolic syndrome and altered gut microbiota in mice lacking Toll-like receptor 5. Science. 2010;328:228-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1496] [Cited by in RCA: 1588] [Article Influence: 105.9] [Reference Citation Analysis (0)] |
| 60. | Everard A, Belzer C, Geurts L, Ouwerkerk JP, Druart C, Bindels LB, Guiot Y, Derrien M, Muccioli GG, Delzenne NM, de Vos WM, Cani PD. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc Natl Acad Sci U S A. 2013;110:9066-9071. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2639] [Cited by in RCA: 3309] [Article Influence: 275.8] [Reference Citation Analysis (0)] |
| 61. | Depommier C, Everard A, Druart C, Plovier H, Van Hul M, Vieira-Silva S, Falony G, Raes J, Maiter D, Delzenne NM, de Barsy M, Loumaye A, Hermans MP, Thissen JP, de Vos WM, Cani PD. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat Med. 2019;25:1096-1103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 774] [Cited by in RCA: 1425] [Article Influence: 237.5] [Reference Citation Analysis (0)] |
| 62. | Kadooka Y, Sato M, Imaizumi K, Ogawa A, Ikuyama K, Akai Y, Okano M, Kagoshima M, Tsuchida T. Regulation of abdominal adiposity by probiotics (Lactobacillus gasseri SBT2055) in adults with obese tendencies in a randomized controlled trial. Eur J Clin Nutr. 2010;64:636-643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 462] [Cited by in RCA: 469] [Article Influence: 31.3] [Reference Citation Analysis (0)] |
| 63. | Ejtahed HS, Mohtadi-Nia J, Homayouni-Rad A, Niafar M, Asghari-Jafarabadi M, Mofid V. Probiotic yogurt improves antioxidant status in type 2 diabetic patients. Nutrition. 2012;28:539-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 380] [Cited by in RCA: 442] [Article Influence: 31.6] [Reference Citation Analysis (0)] |
| 64. | Simon MC, Strassburger K, Nowotny B, Kolb H, Nowotny P, Burkart V, Zivehe F, Hwang JH, Stehle P, Pacini G, Hartmann B, Holst JJ, MacKenzie C, Bindels LB, Martinez I, Walter J, Henrich B, Schloot NC, Roden M. Intake of Lactobacillus reuteri improves incretin and insulin secretion in glucose-tolerant humans: a proof of concept. Diabetes Care. 2015;38:1827-1834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 259] [Article Influence: 25.9] [Reference Citation Analysis (0)] |
| 65. | Li WZ, Stirling K, Yang JJ, Zhang L. Gut microbiota and diabetes: From correlation to causality and mechanism. World J Diabetes. 2020;11:293-308. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 64] [Cited by in RCA: 91] [Article Influence: 18.2] [Reference Citation Analysis (5)] |
| 66. | Zmora N, Zilberman-Schapira G, Suez J, Mor U, Dori-Bachash M, Bashiardes S, Kotler E, Zur M, Regev-Lehavi D, Brik RB, Federici S, Cohen Y, Linevsky R, Rothschild D, Moor AE, Ben-Moshe S, Harmelin A, Itzkovitz S, Maharshak N, Shibolet O, Shapiro H, Pevsner-Fischer M, Sharon I, Halpern Z, Segal E, Elinav E. Personalized Gut Mucosal Colonization Resistance to Empiric Probiotics Is Associated with Unique Host and Microbiome Features. Cell. 2018;174:1388-1405.e21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 702] [Cited by in RCA: 971] [Article Influence: 138.7] [Reference Citation Analysis (0)] |
| 67. | Van Tassell ML, Miller MJ. Lactobacillus adhesion to mucus. Nutrients. 2011;3:613-636. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 192] [Cited by in RCA: 214] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 68. | Song EJ, Han K, Lim TJ, Lim S, Chung MJ, Nam MH, Kim H, Nam YD. Effect of probiotics on obesity-related markers per enterotype: a double-blind, placebo-controlled, randomized clinical trial. EPMA J. 2020;11:31-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 69. | Hendrikx T, Duan Y, Wang Y, Oh JH, Alexander LM, Huang W, Stärkel P, Ho SB, Gao B, Fiehn O, Emond P, Sokol H, van Pijkeren JP, Schnabl B. Bacteria engineered to produce IL-22 in intestine induce expression of REG3G to reduce ethanol-induced liver disease in mice. Gut. 2019;68:1504-1515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 261] [Article Influence: 43.5] [Reference Citation Analysis (0)] |
| 70. | Fatkhullina AR, Peshkova IO, Dzutsev A, Aghayev T, McCulloch JA, Thovarai V, Badger JH, Vats R, Sundd P, Tang HY, Kossenkov AV, Hazen SL, Trinchieri G, Grivennikov SI, Koltsova EK. An Interleukin-23-Interleukin-22 Axis Regulates Intestinal Microbial Homeostasis to Protect from Diet-Induced Atherosclerosis. Immunity. 2018;49:943-957.e9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 123] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 71. | Hasnain SZ, Borg DJ, Harcourt BE, Tong H, Sheng YH, Ng CP, Das I, Wang R, Chen AC, Loudovaris T, Kay TW, Thomas HE, Whitehead JP, Forbes JM, Prins JB, McGuckin MA. Glycemic control in diabetes is restored by therapeutic manipulation of cytokines that regulate beta cell stress. Nat Med. 2014;20:1417-1426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 201] [Article Influence: 18.3] [Reference Citation Analysis (0)] |