Published online Aug 27, 2025. doi: 10.4254/wjh.v17.i8.108063
Revised: May 26, 2025
Accepted: July 9, 2025
Published online: August 27, 2025
Processing time: 139 Days and 16.7 Hours
There is increasing incidence of alcohol-associated liver disease in females. Despite this recent increased incidence, there is a paucity of research on the clinical course and outcomes of alcohol-associated hepatitis (AH) in females compared to males.
To assess if there may be sex differences in severity, outcomes, and healthcare utilization for patients hospitalized for AH.
This study used ICD-9-CM and ICD-10-CM codes to retrospectively identify inpatient encounters for AH at the University of Kentucky from 2012-2021 and obtained data on patient demographics and clinical outcomes. Encounters were cohorted by patient sex and differences in patient demographics and clinical outcomes were assessed. Multivariate logistic regression models were constructed to assess risk of mortality, sepsis, and mechanical ventilation during the encounter.
Of 1386 subjects, 511 (36.9%) were female and 875 (63.1%) were male. Both sexes had similar baseline characteristics of race/ethnicity, discriminant function score, model of end-stage liver disease score, and length of hospital stay. However, the incidence of urinary tract infection, sepsis, and norepinephrine administration was significantly higher for females. Males had a significantly higher incidence of esophageal variceal bleed. On multivariate logistic regression analysis, females had higher odds of encounter sepsis (OR 1.41; 95%CI: 1.064-1.869) and mechanical ventilation (OR 1.352; 95%CI: 1.006-1.816). Odds of encounter mortality were significantly increased in encounters with sepsis (OR 2.309; 95%CI: 1.419-3.757) and mechanical ventilation (OR 9.301; 95%CI: 5.724-15.114).
This study shows sex-based differences in AH outcomes at the University of Kentucky. Future studies are warranted to assess whether tailoring treatments will improve clinical outcomes in females with AH.
Core Tip: Females with alcohol-associated hepatitis had higher incidence of urinary tract infection, sepsis, and norepinephrine administration on univariate analysis. On multivariate logistic regression analysis, females had higher odds of sepsis and mechanical ventilation, which were both found to be independent predictors of mortality.
- Citation: Harris E, Rhudy C, Roy L, Cloud A, Leyson CD. Sex differences in severity, outcomes, and healthcare utilization in alcohol-associated hepatitis. World J Hepatol 2025; 17(8): 108063
- URL: https://www.wjgnet.com/1948-5182/full/v17/i8/108063.htm
- DOI: https://dx.doi.org/10.4254/wjh.v17.i8.108063
Females develop alcohol-associated liver disease (ALD) with less exposure to alcohol than males due to a variety of proposed mechanisms[1,2]. The increased sensitivity of women to alcohol-related liver damage is well-established in the literature and studies have investigated potential mechanisms for this finding. One proposed mechanism is that the decreased body water content of females compared to males leads to a higher effective concentration of alcohol[3]. A study from the early 1990s also found that females have decreased gastric alcohol dehydrogenase activity and reduced first-pass metabolism of alcohol compared to males[4]. Studies have also found sex differences in the expression of cytochrome p450 2E1 enzyme-one of the enzymes responsible for hepatic metabolism of alcohol-with lower expression in females due to differences in growth hormone regulation[5].
There is also evidence that differences in estrogen levels sensitize women to more liver damage from alcohol. In males (both human and rat models) chronic alcohol ingestion is associated with increased estrogen receptor expression that is then associated with increased hepatic proliferation[6]. However, in females, hepatic estrogen receptor expression does not appear to be affected by chronic alcohol consumption, so that hepatocytes are more likely to undergo apoptosis rather than proliferation[6]. Another study of female rats chronically fed alcohol along with an anti-estrogen medication-toremifene-found that while toremifene had no effect on steatosis, it did reduce the observed hepatic inflammation and necrosis[7]. The mechanism appeared potentially related to regulation of enzymes involved in oxidation, with decreased activity of the pro-oxidant enzyme CYP2E1 and increased activity of the oxidant-protective enzyme Se-glutathione peroxidase. These protective effects of an estrogen receptor antagonist in reducing liver inflammation and necrosis in female rat models further support the finding that the baseline higher levels of estrogen in females plays a role in their increased sensitivity to alcohol-related liver damage.
There may be a role for anti-estrogens in preventing alcohol-related liver damage in females. A study on female rodents who were chronically fed alcohol along with an anti-estrogen agent, Toremifene, found that while toremifene had no effect on steatosis, it did reduce the observed hepatic inflammation and necrosis[7]. The mechanism appeared potentially related to regulation of enzymes involved in oxidation, with decreased activity of the pro-oxidant enzyme CYP2E1 and increased activity of the oxidant-protective enzyme Se-glutathione peroxidase[7]. The use of anti-estrogens in patients with alcohol-associated hepatitis (AH) could be explored in future randomized clinical studies, given the limited treatment options for AH.
Whether there are sex differences in the clinical course and severity of ALD is less clear. Recent studies show an alarming increase in the incidence of alcohol use and ALD in females[8,9]. While alcohol use disorder (AUD) has historically had a strong male predominance, several studies show that this sex gap has been narrowing over the past thirty years[10,11]. The recent coronavirus disease 2019 pandemic appears to have further accelerated the rising incidence of AUD and ALD in females[9]. Despite this recent increased incidence of AUD among females, there is a paucity of research on the clinical course and outcomes of (AH) in females compared to males. The few studies comparing mortality between male and female patients are conflicting, with one study reporting male sex as an independent predictor of mortality, while other studies report female sex as an independent predictor of mortality[11-13]. Studies have shown that female gender was associated with higher hospital readmission rates and increased acute on chronic liver failure risk[14,15]. This study aimed to assess if there are sex differences in severity, outcomes, and healthcare utilization for patients hospitalized for AH.
This retrospective cohort study examined inpatient healthcare encounters at the University of Kentucky between 1/1/2012 and 4/1/2021. Encounters were included if they were associated with ICD-9-CM or ICD-10-CM codes for terms indicating AH (ICD-CM-9 5711- acute alcoholic hepatitis; ICD-CM-10 K70.10-alcoholic hepatitis; K70.11-alcoholic hepatitis with ascites). Chart review was done to exclude patients whose laboratory data did not meet criteria for AH. This study used the laboratory criteria as established by The National Institute on Alcohol Abuse and Alcoholic Hepatitis Consortia of serum bilirubin greater than 3 mg/dL, elevated aspartate aminotransferase (AST) in range of 50 U/L to 400 U/L, and AST: Alanine aminotransferase (ALT) ratio of more than 1.5 to define AH[16]. Patients who had AST or ALT > 1000 U/L were also excluded (Supplementary Figure 1). Encounters were classified into cohorts based upon patient sex and clinical outcomes were compared between male and female cohorts. Sex was self-reported. This study was approved by the Institutional Review Board of the University of Kentucky. All research was conducted in accordance with both the Declarations of Helsinki and Istanbul.
Data extraction was performed using services of the University of Kentucky Center for Clinical and Translational Sciences. Chart review was performed by the investigators on the included encounters to collect relevant data on patient demographics, acuity at presentation, as well as clinical outcomes of the encounter. Demographic factors including age, ethnicity, and body mass index (BMI) were collected for all encounters where available. Acuity of the patient was determined through measures of patient comorbidity such as the Charlson Comorbidity Index (CCI) as well as calculating disease-specific prognostic measures based upon laboratory values at admission, including the Model for End-Stage Liver Disease with serum sodium concentration (MELD-Na) score and Maddrey’s Discriminant Function Score for Alcoholic Hepatitis (DF)[17-19].
Encounter clinical outcomes included relevant complicating diagnoses, medication utilization during the encounter, procedures performed, and post-discharge disposition. Relevant complicating diagnoses collected included ascites, esophageal variceal bleeding, hepatic encephalopathy (HE), hepatorenal syndrome (HRS), infections [bacteremia, clostridium difficile, pneumonia, peritonitis, sepsis, and urinary tract infection (UTI)], portal vein thrombosis, and substance/AUD. Manual chart review was subsequently done to obtain sepsis etiology. Etiologies of sepsis identified through chart review included UTI, spontaneous bacterial peritonitis (SBP), bacteremia, clostridium difficile colitis, cellulitis, cholangitis, cholecystitis, colitis, diverticulitis, endocarditis, fungemia, pancreatitis, peritoneal dialysis catheter infection, secondary peritonitis following bowel perforation, pneumonia, septic arthritis, and cases in which source of sepsis could not be determined. Medication utilization outcomes included any use of corticosteroids, N-acetylcysteine (NAC), or vasopressors (norepinephrine, epinephrine, vasopressin) during the encounter. Procedure outcomes collected were the use of hemodialysis, mechanical ventilation, and renal replacement therapy at any time during the encounter. Disposition outcomes included total length of stay (LOS) for the encounter, and discharge disposition to hospice, inpatient death, and death at discharge occurring greater than 48 hours post-admission.
Univariate analysis was performed on all collected variables for males and females. We compared groups using Mann Whitney U test for continuous variables, and Pearson’s χ2 test for categorical variables.
After review of the univariate analysis, investigators constructed multivariable logistic regression models in order to determine relevant predictors of risk of severe adverse outcomes (mechanical ventilation, sepsis, and mortality). A stepwise selection algorithm was used to identify relevant variables for inclusion within the model. All collected variables were included in the stepwise selection algorithm for the encounter mortality model as all variables were hypothesized as potentially contributing to the risk of mortality. Covariates in the mortality model were sex, age class, BMI class, DF class, MELD-Na class, ascites, bacteremia, clostridium difficile, esophageal variceal bleeding, HE, pneumonia, portal vein thrombosis, SBP, substance use disorder, AUD, UTI, pressor use, steroid use, renal replacement therapy, hemodialysis, NAC use, mechanical ventilation. Covariates in the sepsis and mechanical ventilation models were sex, age class, BMI class, CCI, DF class, and model for end-stage liver disease (MELD) class. As mechanical ventilation and sepsis outcomes occur during the course of the inpatient encounter, only demographic and patient acuity variables were considered by the stepwise algorithm for the sepsis and mechanical ventilation models. This was done to prevent confounding from reverse causality in the model (i.e., odds of sepsis predicted by vasopressor use, when in actuality vasopressor use was precipitated by the sepsis diagnosis).
Of 1386 encounters included in this analysis, 511 (36.9%) were for female patients and 875 (63.1%) were male patients. The female cohort were a younger median age (female 45 years old; male 47 years old; P < 0.01) and lower median BMI than males (female 26.6 kg/m2; male 27.6 kg/m2; P = 0.01) (Table 1). No significant difference was observed in ethnicity or in the median CCI between males and females (both cohorts 4; P = 0.92). Similarly, there was also no significant difference in the median MELD-Na (female 26; male 26; P = 0.30) or DF scores (female 42.5; male 39.9; P = 0.27).
| Attribute | Overall (n = 1386) | Male (n = 875) | Female (n = 511) | P value |
| Age at encounter (years) | 0.0031b | |||
| Median | 46 | 47 | 45 | |
| Range | 24-79 | 25-79 | 24-75 | |
| Age group at encounter | 0.0088b | |||
| ≤ 34 | 197 (14.2) | 112 (12.8) | 85 (16.6) | |
| 35-44 | 423 (30.5) | 260 (29.7) | 163 (31.9) | |
| 45-54 | 449 (32.4) | 277 (31.7) | 172 (33.7) | |
| 55-64 | 266 (19.2) | 188 (21.5) | 78 (15.3) | |
| ≥ 65 | 51 (3.7) | 38 (4.3) | 13 (2.5) | |
| Ethnicity | 0.3009 | |||
| Hispanic/Latino | 38 (2.8) | 21 (2.4) | 17 (3.3) | |
| Non-Hispanic/Latino | 1231 (89.8) | 772 (89.5) | 459 (90.4) | |
| Unknown | 102 (7.4) | 70 (8.1) | 32 (6.3) | |
| BMI (kg/m2) | 0.0127a | |||
| Median | 27.2 | 27.6 | 26.6 | |
| Range | 15.8-69.7 | 17.3-69.7 | 15.8-51.3 | |
| BMI (kg/m2) | 0.2583 | |||
| ≤ 18.5 | 50 (3.6) | 26 (3) | 24 (4.7) | |
| 18.6-24.9 | 450 (32.5) | 272 (31.1) | 178 (34.8) | |
| 25-29.9 | 409 (29.5) | 265 (30.3) | 144 (28.2) | |
| 30-34.9 | 254 (18.3) | 171 (19.5) | 83 (16.2) | |
| 35-39.9 | 133 (9.6) | 85 (9.7) | 48 (9.4) | |
| ≥ 40 | 90 (6.5) | 56 (6.4) | 34 (6.7) | |
| CCI | 0.9201 | |||
| Median | 4 | 4 | 4 | |
| Range | 0-16 | 0-16 | 0-13 | |
| CCI group | 0.1693 | |||
| Mild (≤ 2) | 161 (11.6) | 91 (10.4) | 70 (13.7) | |
| Moderate (3-4) | 628 (45.3) | 405 (46.3) | 223 (43.6) | |
| Severe (≥ 5) | 597 (43.1) | 379 (43.3) | 218 (42.7) | |
| Discriminant function score | 0.2262 | |||
| Median | 41.2 | 39.9 | 42.5 | |
| Range | 0-470 | 0-470 | 2-374 | |
| Discriminant function score | 0.2739 | |||
| < 32 | 503 (36.3) | 327 (37.4) | 176 (34.4) | |
| ≥ 32 | 883 (63.7) | 548 (62.6) | 335 (65.6) | |
| MELD-Na score | 0.3012 | |||
| Median | 41.2 | 39.9 | 42.5 | |
| Range | 11-40 | 11-40 | 11-40 | |
| MELD-Na score | 0.8612 | |||
| < 15 | 89 (6) | 54 (6.2) | 29 (5.7) | |
| 16-20 | 247 (17.8) | 151 (17.3) | 96 (18.8) | |
| 21-22 | 126 (9.1) | 77 (8.8) | 49 (9.6) | |
| 23-26 | 292 (21.1) | 184 (21) | 108 (21.1) | |
| 27-21 | 299 (21.6) | 186 (21.3) | 113 (22.1) | |
| ≥ 32 | 339 (24.5) | 223 (25.5) | 116 (22.7) |
A significantly higher percentage of females developed sepsis (female n = 124, 24.3%; male n = 167, 19.1%; P = 0.02) and UTI (female n = 149, 29.2%; male n = 122, 13.9%; P < 0.01) (Table 2). UTI was identified in a higher percentage of female sepsis patients compared to male sepsis patients (female n = 124, 16.9%; male n = 167, 9.0%; P < 0.0001). The three most prevalent causes of sepsis in males were pneumonia, unknown source, and SBP (male n = 167; 25.7%; 19.8%; 17.4% respectively; P < 0.0001). The three most prevalent causes of sepsis in females were pneumonia, UTI, and unknown source (female n = 124; 18.5%; 16.9%; 14.5% respectively; P < 0.0001). Male encounters had increased incidence of esophageal variceal bleed (female n = 25, 4.9%; male n = 96, 11%; P < 0.01). There were no significant differences in utilization of vasopressors, steroids, or NAC, however female encounters had a higher incidence of norepinephrine administration than males (female n = 97, 19%; male n = 130, 14.9%; P = 0.05). No statistically significant difference was observed in mechanical ventilation (female n = 103, 20.2%; male n = 149, 17%; P = 0.15), intermittent hemodialysis (female n = 2, 0.4%; male n = 8, 0.9%; P = 0.27), or continuous renal replacement therapy (female n = 18, 3.5%; male n = 18, 2.1%; P = 0.10) between the two cohorts. The median encounter LOS was the same in both cohorts (female 5.7 days, male 5.5 days; P = 0.50). Similar proportions of encounters in the male and female cohorts resulted in discharge to hospice (female n = 15, 2.9%; male n = 41, 4.7%; P = 0.11) and mortality (female n = 57, 11.2%; male 93, 10.6%; P = 0.76).
| Attribute | Overall (n = 1386) | Male (n = 875) | Female (n = 511) | P value |
| Diagnoses | ||||
| Ascites | 1009 (72.8) | 365 (71.4) | 644 (73.6) | 0.3808 |
| Bacteremia | 89 (6.4) | 62 (7.1) | 27 (5.3) | 0.1867 |
| Clostridium difficile infection | 61 (4.4) | 35 (4) | 26 (5.1) | 0.3407 |
| Esophageal variceal bleed | 121 (8.7) | 96 (11) | 25 (4.9) | 0.0001b |
| Hepatic encephalopathy | 575 (41.5) | 376 (43) | 199 (38.9) | 0.1420 |
| Hepatorenal syndrome | 231 (16.7) | 158 (18.1) | 73 (14.3) | 0.0691 |
| Pneumonia | 234 (16.9) | 138 (15.8) | 96 (18.8) | 0.1483 |
| Portal vein thrombosis | 55 (4) | 40 (4.6) | 15 (2.9) | 0.1323 |
| Spontaneous bacterial peritonitis | 1 (0.1) | 0 | 1 (0.1) | 0.4447 |
| Sepsis | 291 (21) | 167 (19.1) | 124 (24.3) | 0.0223a |
| Substance/alcohol use disorder | 1075 (77.6) | 687 (78.5) | 388 (75.9) | 0.2658 |
| Urinary tract infection | 271 (19.6) | 122 (13.9) | 149 (29.2) | < 0.0001b |
| Medications: | ||||
| Use of any vasopressors | 235 (17) | 137 (15.7) | 98 (19.2) | 0.0919 |
| Norepinephrine | 227 (16.4) | 130 (14.9) | 97 (19) | 0.0453a |
| Epinephrine | 58 (4.2) | 39 (4.5) | 19 (3.7) | 0.5074 |
| Vasopressin | 118 (8.5) | 65 (7.4) | 53 (10.4) | 0.0582 |
| Use of steroids | 503 (36.3) | 328 (37.5) | 175 (34.3) | 0.2263 |
| Use of N-acetylcysteine | 137 (9.9) | 82 (9.4) | 55 (10.8) | 0.4023 |
| Procedures: | ||||
| Renal replacement therapy | 36 (2.6) | 18 (2.1) | 18 (3.5) | 0.0980 |
| Hemodialysis | 10 (0.7) | 8 (0.9) | 2 (0.4) | 0.2671 |
| Mechanical ventilation | 252 (18.2) | 149 (17) | 103 (10.8) | 0.1452 |
| Length of stay (days) | ||||
| Median | 5.6 | 5.5 | 5.7 | 0.4989 |
| Range | 0.1-92.7 | 0.1-92.7 | 0.1-75.7 | |
| Disposition | ||||
| Hospice | 56 (4) | 41 (4.7) | 15 (2.9) | 0.1104 |
| Inpatient death | 150 (10.8) | 93 (10.6) | 57 (11.2) | 0.7610 |
| Inpatient death > 48 hours after admission | 122 (8.8) | 74 (8.5) | 48 (9.4) | 0.5529 |
| Inpatient death or hospice | 206 (14.9) | 134 (15.3) | 72 (14.1) | 0.5365 |
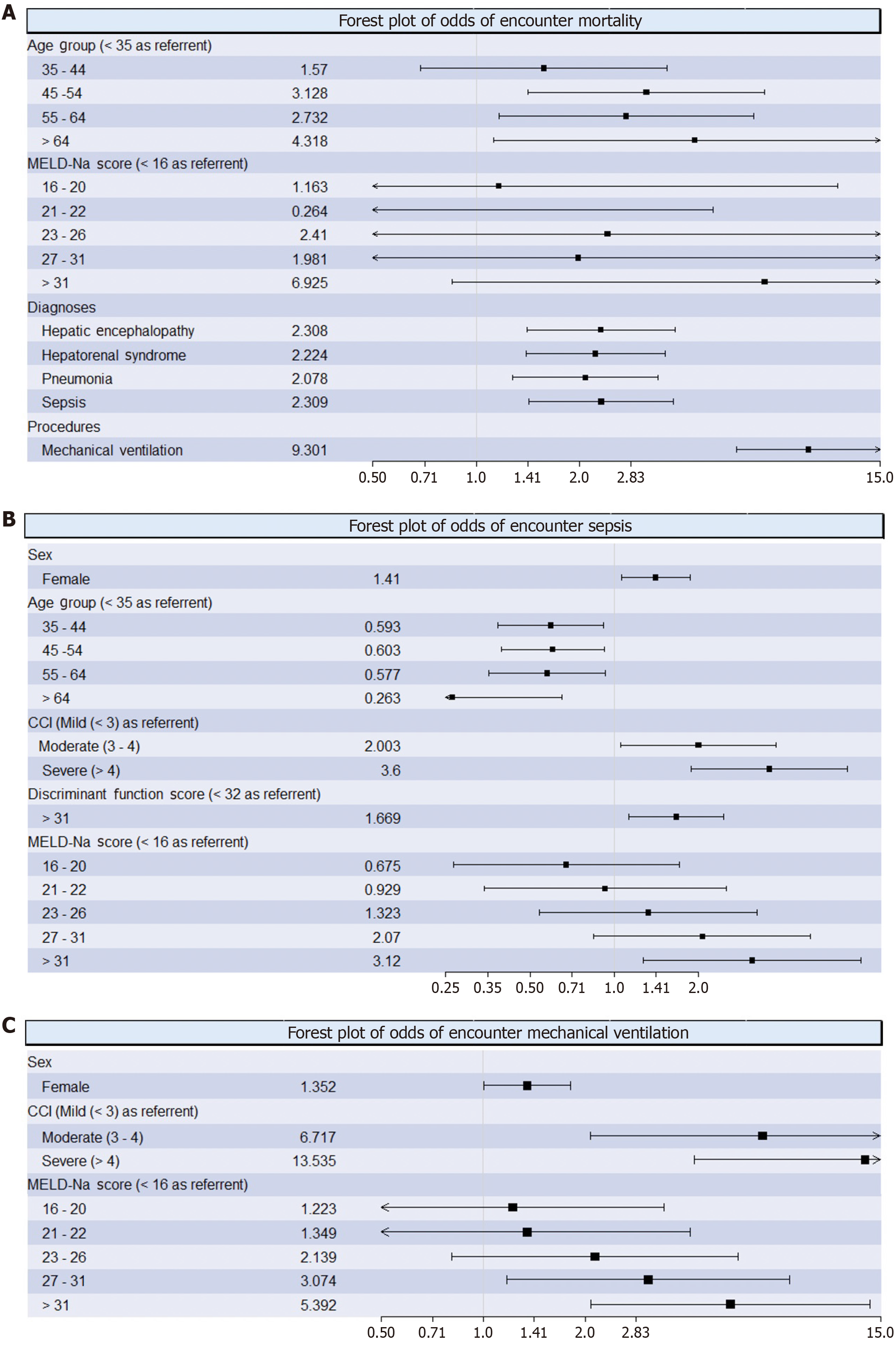
After stepwise variable selection, age (referent ≤ 34), MELD-Na (< 16 as referent), the diagnoses of HE, HRS, pneumonia, sepsis, and mechanical ventilation were included within the optimal logistic regression model (Table 3). Sex was not selected for inclusion by the algorithm within this model. Covariates in this model were sex, age class, BMI class, DF class, MELD-Na class, ascites, bacteremia, clostridium difficile, esophageal variceal bleeding, HE, pneumonia, portal vein thrombosis, SBP, substance use disorder, AUD, UTI, pressor use, steroid use, renal replacement therapy, hemodialysis, NAC use, mechanical ventilation. Age > 65 years significantly increased odds of encounter mortality (OR 4.318; 95%CI: 1.124-16.589) compared to referent age of ≤ 34 (Figure 1A). The presence of HE (OR 2.308; 95%CI: 1.403-3.797), HRS (OR 2.224; 95%CI: 1.394-3.551), pneumonia (OR 2.078; 95%CI: 1.276-3.382), and sepsis (OR 2.309; 95%CI: 1.419-3.757) all significantly increased the odds of a mortality event (Figure 1A). Mechanical ventilation was associated with the greatest increase in odds of a mortality event (OR 9.301; 95%CI: 5.724-15.114).
| Attribute | Odds ratio | 95%CI | P value |
| Age group (≤ 34 as referent) | |||
| 35-44 | 1.57 | 0.686-3.592 | 0.2855 |
| 45-54 | 3.128 | 1.410-6.939 | 0.005b |
| 55-64 | 2.732 | 1.163-6.418 | 0.0211a |
| ≥ 65 | 4.318 | 1.124-16.589 | 0.0331a |
| MELD-Na score (≤ 15 as referent) | |||
| 16-20 | 1.163 | 0.119-11.311 | 0.8967 |
| 21-22 | 0.264 | 0.014-4.908 | 0.3717 |
| 23-26 | 2.41 | 0.282-20.596 | 0.4217 |
| 27-31 | 1.981 | 0.235-16.722 | 0.5300 |
| ≥ 32 | 6.925 | 0.851-56.355 | 0.0704 |
| Diagnoses | |||
| Hepatic encephalopathy | 2.308 | 1.403-3.797 | 0.0010b |
| Hepatorenal syndrome | 2.224 | 1.394-3.551 | 0.0008b |
| Pneumonia | 2.078 | 1.276-3.382 | 0.0033b |
| Sepsis | 2.309 | 1.419-3.757 | 0.0008b |
| Procedures | |||
| Mechanical ventilation | 9.301 | 5.724-15.114 | < 0.0001b |
After stepwise variable selection, gender, age, CCI, DF, and MELD-Na were included within the optimal model for odds of sepsis (Table 4). Covariates in this model were sex, age class, BMI class, CCI, DF class, and MELD class. Female sex was associated with increased odds of sepsis as compared to male sex (OR 1.41; 95%CI: 1.064-1.869) (Figure 1B). Higher CCI (≥ 5) (OR 3.6; 95%CI: 1.886-6.874), DF score (≥ 32) (OR 1.669; 95%CI: 1.130-2.467), and MELD-Na score (> 31) (OR 3.12; 95%CI: 1.272-7.653) were all associated with increased odds of sepsis (Figure 1B). Advanced age was associated with decreased odds of sepsis as compared to the referent group (≤ 34).
| Attribute | Odds ratio | 95%CI | P value |
| Sex | |||
| Female | 1.41 | 1.064-1.869 | 0.0168a |
| Age group (≤ 34 as referent) | |||
| 35-44 | 0.593 | 0.383-0.917 | 0.0189a |
| 45-54 | 0.603 | 0.394-0.921 | 0.0192a |
| 55-64 | 0.577 | 0.357-0.93 | 0.0248a |
| ≥ 65 | 0.263 | 0.106-0.652 | 0.0039b |
| CCI [Mild (≤ 2) as referent] | |||
| Moderate (3-4) | 2.003 | 1.054-3.809 | 0.0340a |
| Severe (≥ 5) | 3.6 | 1.886-6.874 | 0.0001b |
| Discriminant function score (< 32 as referent) | |||
| ≥ 32 | 1.669 | 1.130-2.467 | 0.0101a |
| MELD-Na score (≤ 15 as referent) | |||
| 16-20 | 0.675 | 0.266-1.717 | 0.4097 |
| 21-22 | 0.929 | 0.342-2.525 | 0.8854 |
| 23-26 | 1.323 | 0.539-3.249 | 0.5409 |
| 27-31 | 2.07 | 0.846-5.065 | 0.1111 |
| ≥ 32 | 3.12 | 1.272-7.653 | 0.0129a |
In the optimal model for odds of mechanical ventilation, sex, CCI (mild defined as ≤ 2 as referent), and MELD-Na (< 16 as referent) were included by the stepwise variable selection algorithm (Table 5). Covariates in this model were sex, age class, BMI class, CCI, DF class, and MELD class. The odds of mechanical ventilation significantly increased in female encounters (OR 1.352; 95%CI: 1.006-1.816) (Figure 1C). Higher CCI (≥ 5) (OR 13.535; 95%CI: 4.221-43.405) and MELD-Na score (> 31) (OR 5.392; 95%CI: 2.087-13.934) were also associated with increased odds of mechanical ventilation (Figure 1C).
| Attribute | Odds ratio | 95%CI | P value |
| Sex | |||
| Female | 1.352 | 1.006-1.816 | 0.0168a |
| CCI [Mild (≤ 2) as referent] | |||
| Moderate (3-4) | 6.717 | 2.081-21.680 | 0.0014b |
| Severe (≥ 5) | 13.535 | 4.221-43.405 | < 0.0001b |
| MELD-Na score (≤ 15 as referent) | |||
| 16-20 | 1.223 | 0.437-3.425 | 0.7018 |
| 21-22 | 1.349 | 0.443-4.109 | 0.5986 |
| 23-26 | 2.139 | 0.804-5.690 | 0.1275 |
| 27-31 | 3.074 | 1.173-8.06 | 0.0224a |
| ≥ 32 | 5.392 | 2.087-13.934 | 0.0005a |
This study shows that females admitted for AH at the University of Kentucky have a higher incidence of sepsis, UTI, and norepinephrine administration. Males had an increased incidence of esophageal variceal bleed. On multivariate logistic regression analysis, females had higher odds of encounter sepsis and mechanical ventilation, outcomes which were found to be independent predictors of mortality. These differences in clinical outcomes occurred despite similar baseline DF and MELD-Na scores – current prognostic indicators for patients with AH. While females trended to have higher odds of mortality on logistic regression, confidence interval did not support statistical significance, likely due to sample size.
Based on our study, it appears that females have a higher incidence of complications from infections, notably UTIs and sepsis. UTIs are at baseline more prevalent in females than males due to the shorter female urethral length, so the increased incidence of UTIs in female AH patients is expected[20]. However, it is significant that our study found that female AH patients were more likely to develop sepsis. UTIs did account for a higher percentage of sepsis in females than males. While pneumonia was the most prevalent cause of sepsis in both males and females and males had a higher prevalence of SBP, females had an overall higher incidence of sepsis. This finding suggests that the propensity of females to develop UTIs contributes to the increased incidence of sepsis in female AH patients and their poorer clinical outcomes. In addition to being more prone to UTIs, certain animal studies suggest that hormonal levels may predispose females to developing infections. Rodent studies have shown higher levels of endotoxin, increased gut permeability to endotoxin in females and demonstrated that estrogen sensitizes Kupffer cells to endotoxin-mediated liver injury[21,22]. Whether this translates to a higher pro-inflammatory milieu and infection risk in females with AH needs further exploration.
The increased incidence of sepsis and UTIs observed in females with AH suggests that these patients may benefit from more aggressive infection screening protocols as part of their management. It may also be beneficial to have a lower threshold to treat female AH patients with a urinalysis suggestive of UTI even if minimal symptoms are present. In this study, UTI was identified based on ICD code placed in the chart by the provider. This means that the diagnosis of UTI was at the discretion of the provider who placed the ICD code. Given the increased incidence and odds of sepsis in female AH patients, earlier intervention in the form of treating potential UTI may improve outcomes. Further studies are needed to investigate whether a lower threshold to treat female AH patients for UTI improves clinical outcomes and mortality rates for these patients.
Additionally, the role of corticosteroids in managing alcoholic hepatitis in females warrants closer study. New therapeutic agents to treat AH are in various stages of clinical trials and have different targets including preventing the development of steatosis, limiting innate immune response mediated inflammation, modification of the microbiome, and stimulation of liver regeneration[23]. Females especially may benefit from agents with less broadly immunosuppressive effects, as they appear more likely to develop infectious complications in the setting of AH.
Clinical outcome differences in AH might influence the receipt of liver transplantation in females. A recent retrospective study has demonstrated that among ALD patients evaluated for liver transplantation, females had lower likelihood to be transplanted than males (10% vs 19%; P < 0.05)[24]. Patients who are “too sick for transplant” are less likely to be transplanted or usually inactivated due to infection and requirement of pressor and mechanical ventilation. In our current study, females admitted for AH had a higher incidence of sepsis, UTI, and norepinephrine compared to males and on multivariate analysis, females had higher odds of developing sepsis and requiring mechanical ventilation. As transplantation for AH increases nationwide, these are some factors that may affect decisions that institutions face in deciding which AH patients to transplant and may in turn continue to impact access to transplant.
Efforts to address sex disparity in access to liver transplantation over the last several years culminated in the recent implementation of MELD 3.0 in June 2023. MELD 3.0, which incorporates serum albumin and gender and lowers the creatinine ceiling from 4 to 3, was shown to improve the ability to reflect disease severity and reduced liver tran
In addition to the increased incidence of alcohol use and ALD in females observed in recent years, there is evidence that females are less likely to get treatment for alcohol use disease and have a higher alcohol recidivism rate than males[27,28]. Clinical management of females with ALD therefore also appears to be different than for their male counterparts. This study adds to the growing evidence that females with AH experience a different clinical course than males in the acute inpatient setting. While there is substantial evidence for pathophysiological mechanisms for this difference, it is also possible that there are disparities in care provided. Additionally, the long-term outpatient clinical outcomes for female AH patients may be improved by improving access to treatment for their underlying AUD.
The main limitation of this study is that it is a single center retrospective cohort study. It is also possible that unknown confounders could have influenced the clinical courses and outcomes observed. However, the subjects had similar baseline characteristics, when controlling for age, race, ethnicity, and BMI. Another limitation is the use of ICD codes to identify patients with diagnoses of interest. ICD codes do not always accurately reflect the true clinical condition of a patient. However, additional manual chart review of the subjects was done after they were identified by ICD code.
This study assessed differences in clinical outcomes between males and females hospitalized with AH. Significantly, females had a higher risk of mortality, incidence of norepinephrine administration, UTI, and sepsis. Females also had higher odds of encounter sepsis and mechanical ventilation, outcomes which were found to be independent predictors of mortality. Future studies are warranted to assess whether tailoring treatments and public health interventions based on sex differences will improve outcomes for this worsening healthcare problem.
| 1. | Becker U, Deis A, Sørensen TI, Grønbaek M, Borch-Johnsen K, Müller CF, Schnohr P, Jensen G. Prediction of risk of liver disease by alcohol intake, sex, and age: a prospective population study. Hepatology. 1996;23:1025-1029. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 551] [Cited by in RCA: 526] [Article Influence: 18.1] [Reference Citation Analysis (1)] |
| 2. | Guy J, Peters MG. Liver disease in women: the influence of gender on epidemiology, natural history, and patient outcomes. Gastroenterol Hepatol (N Y). 2013;9:633-639. [PubMed] |
| 3. | Maddur H, Shah VH. Alcohol and Liver Function in Women. Alcohol Res. 2020;40:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 4. | Frezza M, di Padova C, Pozzato G, Terpin M, Baraona E, Lieber CS. High blood alcohol levels in women. The role of decreased gastric alcohol dehydrogenase activity and first-pass metabolism. N Engl J Med. 1990;322:95-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 934] [Cited by in RCA: 804] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 5. | Agrawal AK, Shapiro BH. Intrinsic signals in the sexually dimorphic circulating growth hormone profiles of the rat. Mol Cell Endocrinol. 2001;173:167-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 45] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 6. | Colantoni A, Emanuele MA, Kovacs EJ, Villa E, Van Thiel DH. Hepatic estrogen receptors and alcohol intake. Mol Cell Endocrinol. 2002;193:101-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 7. | Järveläinen HA, Lukkari TA, Heinaro S, Sippel H, Lindros KO. The antiestrogen toremifene protects against alcoholic liver injury in female rats. J Hepatol. 2001;35:46-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 39] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 8. | Mellinger JL. Epidemiology of Alcohol Use and Alcoholic Liver Disease. Clin Liver Dis (Hoboken). 2019;13:136-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 60] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 9. | Sohal A, Khalid S, Green V, Gulati A, Roytman M. The Pandemic Within the Pandemic: Unprecedented Rise in Alcohol-related Hepatitis During the COVID-19 Pandemic. J Clin Gastroenterol. 2022;56:e171-e175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 35] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 10. | Karaye IM, Maleki N, Hassan N, Yunusa I. Trends in Alcohol-Related Deaths by Sex in the US, 1999-2020. JAMA Netw Open. 2023;6:e2326346. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 36] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 11. | White A, Castle IJ, Chen CM, Shirley M, Roach D, Hingson R. Converging Patterns of Alcohol Use and Related Outcomes Among Females and Males in the United States, 2002 to 2012. Alcohol Clin Exp Res. 2015;39:1712-1726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 263] [Article Influence: 29.2] [Reference Citation Analysis (0)] |
| 12. | Gratacós-Gines J, Rodríguez M, Giráldez-Gallego A, Cabezas J, Vázquez IF, Cots MV, Horta D, Sánchez-Delgado J, Acosta-López S, Varasa TA, Marti-Aguado D, Monterde VB, Martin-Mateos R, Clemente A, Tejedor-Tejada J, Bandera JP, Sala M, Badia-Aranda E, Sancho VA, Tomé S, Fernández-Rodríguez C, Caballería J, Pose E. Impact of sex and recurrence in the prognosis of alcoholic hepatitis. J Hepatol. 2022;77:S127. [DOI] [Full Text] |
| 13. | Pang JX, Ross E, Borman MA, Zimmer S, Kaplan GG, Heitman SJ, Swain MG, Burak K, Quan H, Myers RP. Risk factors for mortality in patients with alcoholic hepatitis and assessment of prognostic models: A population-based study. Can J Gastroenterol Hepatol. 2015;29:131-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 14. | Adejumo AC, Cholankeril G, Iqbal U, Yoo ER, Boursiquot BC, Concepcion WC, Kim D, Ahmed A. Readmission Rates and Associated Outcomes for Alcoholic Hepatitis: A Nationwide Cohort Study. Dig Dis Sci. 2020;65:990-1002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 15. | Singal AK, Arsalan A, Dunn W, Arab JP, Wong RJ, Kuo YF, Kamath PS, Shah VH. Alcohol-associated liver disease in the United States is associated with severe forms of disease among young, females and Hispanics. Aliment Pharmacol Ther. 2021;54:451-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 63] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 16. | Crabb DW, Bataller R, Chalasani NP, Kamath PS, Lucey M, Mathurin P, McClain C, McCullough A, Mitchell MC, Morgan TR, Nagy L, Radaeva S, Sanyal A, Shah V, Szabo G; NIAAA Alcoholic Hepatitis Consortia. Standard Definitions and Common Data Elements for Clinical Trials in Patients With Alcoholic Hepatitis: Recommendation From the NIAAA Alcoholic Hepatitis Consortia. Gastroenterology. 2016;150:785-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 273] [Cited by in RCA: 410] [Article Influence: 45.6] [Reference Citation Analysis (0)] |
| 17. | Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373-383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32099] [Cited by in RCA: 38316] [Article Influence: 1008.3] [Reference Citation Analysis (0)] |
| 18. | Kim WR, Biggins SW, Kremers WK, Wiesner RH, Kamath PS, Benson JT, Edwards E, Therneau TM. Hyponatremia and mortality among patients on the liver-transplant waiting list. N Engl J Med. 2008;359:1018-1026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 935] [Cited by in RCA: 1057] [Article Influence: 62.2] [Reference Citation Analysis (0)] |
| 19. | Maddrey WC, Boitnott JK, Bedine MS, Weber FL Jr, Mezey E, White RI Jr. Corticosteroid therapy of alcoholic hepatitis. Gastroenterology. 1978;75:193-199. [PubMed] [DOI] [Full Text] |
| 20. | Fakhraddin Raheem T, Babani SA. Prevalence and Multi-Drug Resistance Patterns of Uropathogenic E.coli isolated from Women Patients in Kirkuk city, Iraq. Iran J Med Microbiol. 2022;16:609-614. [RCA] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 21. | Ikejima K, Enomoto N, Iimuro Y, Ikejima A, Fang D, Xu J, Forman DT, Brenner DA, Thurman RG. Estrogen increases sensitivity of hepatic Kupffer cells to endotoxin. Am J Physiol. 1998;274:G669-G676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 54] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 22. | Bala S, Marcos M, Gattu A, Catalano D, Szabo G. Acute binge drinking increases serum endotoxin and bacterial DNA levels in healthy individuals. PLoS One. 2014;9:e96864. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 200] [Cited by in RCA: 244] [Article Influence: 22.2] [Reference Citation Analysis (0)] |
| 23. | Mitchell MC, Kerr T, Herlong HF. Current Management and Future Treatment of Alcoholic Hepatitis. Gastroenterol Hepatol (N Y). 2020;16:178-189. [PubMed] |
| 24. | McElroy LM, Likhitsup A, Scott Winder G, Saeed N, Hassan A, Sonnenday CJ, Fontana RJ, Mellinger J. Gender Disparities in Patients With Alcoholic Liver Disease Evaluated for Liver Transplantation. Transplantation. 2020;104:293-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 25. | Kim WR, Mannalithara A, Heimbach JK, Kamath PS, Asrani SK, Biggins SW, Wood NL, Gentry SE, Kwong AJ. MELD 3.0: The Model for End-Stage Liver Disease Updated for the Modern Era. Gastroenterology. 2021;161:1887-1895.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 342] [Article Influence: 85.5] [Reference Citation Analysis (0)] |
| 26. | Lai JC, Terrault NA, Vittinghoff E, Biggins SW. Height contributes to the gender difference in wait-list mortality under the MELD-based liver allocation system. Am J Transplant. 2010;10:2658-2664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 101] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 27. | Mellinger JL, Fernandez A, Shedden K, Winder GS, Fontana RJ, Volk ML, Blow FC, Lok ASF. Gender Disparities in Alcohol Use Disorder Treatment Among Privately Insured Patients with Alcohol-Associated Cirrhosis. Alcohol Clin Exp Res. 2019;43:334-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 84] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 28. | Potts JR, Howard MR, Verma S. Recurrent severe alcoholic hepatitis: clinical characteristics and outcomes. Eur J Gastroenterol Hepatol. 2013;25:659-664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |