Published online Dec 7, 2015. doi: 10.3748/wjg.v21.i45.12787
Peer-review started: May 11, 2015
First decision: June 2, 2015
Revised: July 24, 2015
Accepted: October 13, 2015
Article in press: October 13, 2015
Published online: December 7, 2015
Processing time: 213 Days and 0.6 Hours
AIM: To evaluate the inflammasome activation and the effect of peroxisome proliferator-activated receptors (PPAR)-δ agonist treatment in nonalcoholic fatty liver disease (NAFLD) models.
METHODS: Male C57BL/6J mice were classified according to control or high fat diet (HFD) with or without PPAR-δ agonist (GW) over period of 12 wk [control, HFD, HFD + lipopolysaccharide (LPS), HFD + LPS + GW group]. HepG2 cells were exposed to palmitic acid (PA) and/or LPS in the absence or presence of GW.
RESULTS: HFD caused glucose intolerance and hepatic steatosis. In mice fed an HFD with LPS, caspase-1 and interleukin (IL)-1β in the liver were significantly increased. Treatment with GW ameliorated the steatosis and inhibited overexpression of pro-inflammatory cytokines. In HepG2 cells, PA and LPS treatment markedly increased mRNA of several nucleotide-binding and oligomerization domain-like receptor family members (NLRP3, NLRP6, and NLRP10), caspase-1 and IL-1β. PA and LPS also exaggerated reactive oxygen species production. All of the above effects of PA and LPS were reduced by GW. GW also enhanced the phosphorylation of AMPK-α.
CONCLUSION: PPAR-δ agonist reduces fatty acid-induced inflammation and steatosis by suppressing inflammasome activation. Targeting the inflammasome by the PPAR-δ agonist may have therapeutic implication for NAFLD.
Core tip: Until now, the underlying mechanisms of disease progression and therapeutic targets were uncertain in nonalcoholic fatty liver disease (NAFLD). Our study were to evaluate the inflammasome activation and the effect of peroxisome proliferator-activated receptors (PPAR)-δ agonist treatment in NAFLD models. In our NAFLD models, mRNA of several NOD-like receptor family members, caspase-1 and interleukin-1β were markedly increased. All of those effects were reduced by PPAR-δ agonist treatment. It also ameliorated the steatosis and inhibited overexpression of pro-inflammatory cytokines. In conclusion, PPAR-δ agonist reduces fatty acid-induced inflammation and steatosis by suppressing inflammasome activation.
- Citation: Lee HJ, Yeon JE, Ko EJ, Yoon EL, Suh SJ, Kang K, Kim HR, Kang SH, Yoo YJ, Je J, Lee BJ, Kim JH, Seo YS, Yim HJ, Byun KS. Peroxisome proliferator-activated receptor-delta agonist ameliorated inflammasome activation in nonalcoholic fatty liver disease. World J Gastroenterol 2015; 21(45): 12787-12799
- URL: https://www.wjgnet.com/1007-9327/full/v21/i45/12787.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i45.12787
Nonalcoholic fatty liver disease (NAFLD) is one of the most common liver diseases, occurring in 10%-35% of the general population, and its prevalence has increased in parallel with the worldwide epidemic of obesity and its related insulin-resistant state[1,2]. The spectrum of NAFLD ranges from simple steatosis to nonalcoholic steatohepatitis (NASH) and fibrosis. Certain portion of NAFLD eventually progressed to liver cirrhosis and hepatocellular carcinoma[1,3]. Recently, the two-hit hypothesis was been reported in the literature[4], but the precise mechanism involved in the development and progression of NAFLD is not entirely understood.
Regarding the pathogenesis of NAFLD[5,6], steatosis sensitizes the liver and makes it susceptible to additional insults. Factors such as increased oxidative stress, pro-inflammatory cytokines and impaired adenosine triphosphate (ATP) production[7,8] could trigger necroinflammation and lead to the progression of steatohepatitis. Although many kind of drugs such as thiazolidinediones, vitamin E, losartan, and silybin have been evaluated in several studies, few pharmacological treatments can be recommended at present[9-11].
Inflammasome is a large, intracellular multi-protein complex that is a sensor of the exogenous and endogenous danger signals, such as pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs), that promotes the cleavage and maturation of pro-inflammatory cytokines such as pro-interleukin (IL)-1β and pro-IL-18[12,13]. Most DAMPs induce the production of reactive oxygen species (ROS), which results in nucleotide-binding and oligomerization domain (NOD)-like receptor family pyrin domain-containing 3 (NLRP3) inflammasome activation[14]. The NLRP3 inflammasome is known to be related to the pathogenesis of obesity, insulin resistance, and development of diabetes[15-18]. But, the role of inflammasome in the pathogenesis of NAFLD/NASH has not been elucidated.
Peroxisome proliferator-activated receptors (PPARs) are a group of nuclear receptor proteins, and three types have been identified in mammals: PPAR-α, PPAR-γ, and PPAR-δ. Pharmacological targets of PPAR-α and PPAR-γ are relatively well-known in the treatment of dyslipidemia and diabetes[19]. It has become increasingly apparent that PPAR-δ also play important roles in the regulation of metabolism, as its activation increases fatty acid (FA) oxidation, ameliorates glucose homeostasis, and attenuates macrophage inflammatory responses[19,20]. In the mouse model of NASH, PPAR-δ improves hepatic steatosis and inflammation by regulation of lipid metabolism and inhibition of inflammatory response[21].
In this study, we determined whether treatment with the PPAR-δ agonist could ameliorate high fat-induced inflammasome activation and the inflammatory response using in vivo and in vitro NAFLD models.
PPAR-δ agonist, GW501516 (GW), was obtained from Enzo Life Sciences (Farmingdale, NY, United States). Lipopolysaccharide (LPS) (Escherichia coli 0111:B4) and palmitic acid (PA) were purchased from Sigma-Aldrich (St Louis, MO, United States). Solutions and reagents used for cell culture were obtained from Invitrogen (Carlsbad, CA, United States) unless otherwise noted. Antibodies were purchased from Cell Signaling Technology Inc. (Beverly, MA, United States).
The animal protocol was designed to minimize pain or discomfort to the animals. Male 4-5-wk-old C57BL/6J mice were obtained from Japan SLC Inc. (Shizuoka, Japan). They were acclimatized to laboratory conditions (22-24 °C and 37%-64% humidity, with a 12-h dark-light cycle, free access to food and water) for one week prior to experimentation. Reflecting endotoxemia in NAFLD, we were decided to inject non-lethal very low dosage of LPS. Mice were randomly divided into four groups, which were treated for 12 wk as follows: standard diet (control, n = 5); HFD (HFD, 60% kcal from fat; D12492, Research Diets; New Brunswick, NJ, United States, n = 5); HFD plus one daily oral gavage of vehicle (0.5% carboxymethyl cellulose solution) with one weekly intraperitoneal (IP) injection of LPS (1 mg/kg per week) (HFD + LPS, n = 5); and HFD plus one daily oral dose of 3 mg/kg per day of GW501516, which was dissolved in the vehicle, with IP injection of LPS (HFD + LPS + GW, n = 6). Vehicle and GW501516 were administered for the last 3 wk. Body weight and food intake were recorded weekly. Body length (from nose to anus) was measured before sacrifice. Body mass index (BMI) was calculated by dividing body weight by the square of the body length (g/cm2)[22].
The study was reviewed and approved by the Institutional Review Board of Korea University and it was approved by the institutional animal review board of Korea University, Seoul, Korea, KUIACUC-2013-66 and conducted in compliance with the Guide for the Care and Use of Laboratory Animals.
The glucose tolerance test was conducted in all animals at 11 wk after dietary manipulation. After overnight fasting, 2 g/kg glucose was injected intraperitoneally, and blood samples were taken from the tail vein at 0, 15, 30, 60, 90, and 120 min. Blood glucose was measured using an Accu-Check Compact kit (Roche Diagnostics GmbH; Mannheim, Germany).
At 12 wk, the mice were intraperitoneally anesthetized with a mixture of tiletamine/zolazepam (30 mg/kg, Zoletil; Yuhan Corp.; Seoul, Korea) and xylazine (10 mg/kg, Rompun; Bayer, Inc.; Frankfurt, Germany), and sacrificed by exsanguination. Blood samples were extracted and the serum was isolated. The livers were rapidly excised and weighed. Serum alanine transaminase (ALT), aspartate transaminase (AST), triglyceride (TG), and total cholesterol (TC) levels were measured using common biochemical kits (Mindray Medical International Ltd.; Shenzhen, China).
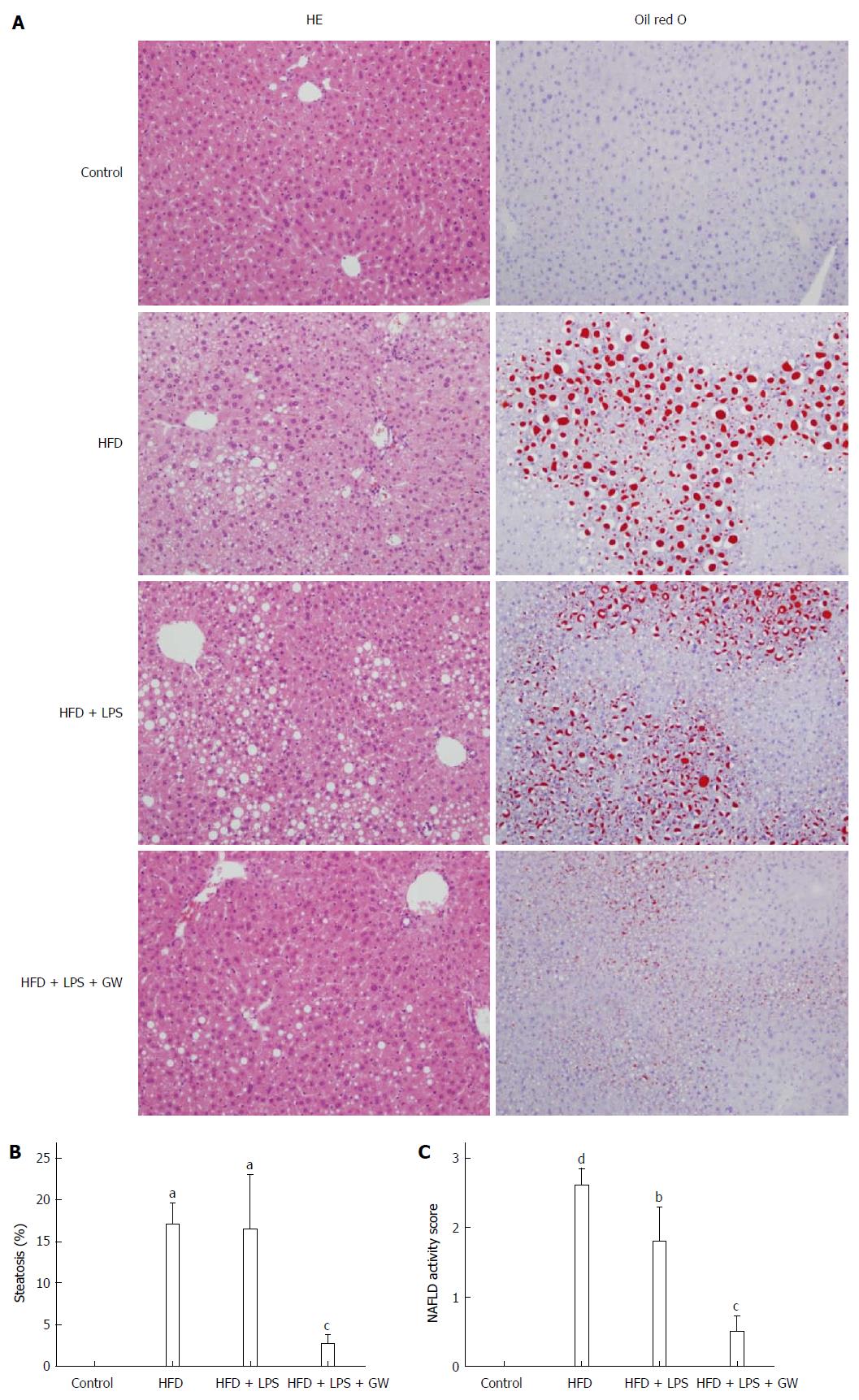
The right liver lobe was stored at -80 °C until analysis of mRNA and protein. The left liver lobe was immediately fixed in 10% neutral-buffered formalin, paraffin-embedded, sectioned, and sections were stained with hematoxylin and eosin (HE). To visualize the neutral lipids, some of the frozen sections of fresh liver were stained using Oil Red O reagent. The liver samples were examined histologically in a blind manner by an experienced pathologist using the histological scoring system for NAFLD[23].
The human hepatoma HepG2 cell line (ATCC; Manassas, VA, United States) cells were cultured as manufacture’s instruction. In all experiments, PA concentration of 0.2 mmol/L which had no influence on cell viability was selected. The cells were stimulated with PA-BSA (0.2 μmol/L), LPS (1 μg/mL), or both, with or without GW501516 (1 or 10 μmol/L).
Total RNA was extracted from mouse liver tissues and HepG2 cells with TRIzol reagent according to the manufacturer’s instructions. RNA was reverse-transcribed into cDNA using the High Capacity cDNA Reverse Transcription kit (Applied Biosystems; Foster City, CA, United States). Gene expressions of NLRP1, NLRP3, NLRP6, NLRP10, NLRC4, caspase-1, and IL-1β were quantified by real-time polymerase chain reaction (PCR) with a 7000 Real-time PCR system (Applied Biosystems). PCR reactions were prepared with the Power SYBR Green PCR Master mix (Applied Biosystems) in 20 μL. The cycling parameters were 10 min at 95 °C followed by 40 cycles of 95 °C (15 s) and 30 s at 60 °C, followed by melting curve analysis. Gene expression by real-time PCR was presented relative to glyceraldehyde 3-phosphate dehydrogenase. The PCR primers used are listed in the Table 1.
| mRNA | Forward (5'-, -3') | Reverse (5'-, -3') |
| Mouse | ||
| Caspase-1 | CACAGCTCTGGAGATGGTGA | GGTCCCACATATTCCCTCC |
| IL-1β | GCTGCTTCCAAACCTTTGAC | AGCTTCTCCACAGCCACAAT |
| GAPDH | CAGCCTCAAGATCATCAGCA | GTCTTCTGGGTGGCAGTGAT |
| Human | ||
| NLRP1 | TCTCAAGGGGACCTGCATAC | AGCCAGCTACAGGGAAGTGA |
| NLRP3 | AAGGAAGTGGACTGCGAGA | AACGTTCGTCCTTCCTTCCT |
| NLRP6 | TTCATCACCAGCGTTCTGAG | GTTGCTCCAGTTCCTTCTCG |
| NLRP10 | GGATGAGAAGCAAGCTGACC | CTTTGCCTCTCTCCATCTGC |
| NLRC4 | GCAAGGCTCTGACCAAGTTC | TGTCTGCTTCCTGATTGTGC |
| Caspase-1 | CCACAATGGGCTCTGTTTTT | CATCTGGCTGCTCAAATGAA |
| IL-1β | GGACAAGCTGAGGAAGATGC | TCGTTATCCCATGTGTCGAA |
| GAPDH | CAGCCTCAAGATCATCAGCA | GTCTTCTGGGTGGCAGTGAT |
Mouse liver tissues and HepG2 cell were lysed using M-PER Mammalian Protein Extraction Reagent (Pierce Chemical; Rockford, IL, United States). The protein concentration was determined using the bicinchoninic acid (BCA) method. Equal amounts (20 μg) of total proteins were boiled for 5 min and subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), followed by electrotransfer to polyvinylidene difluoride (PVDF) membranes. The membranes were blocked with 5% BSA in Tris-buffered saline (TBS) for 1 h at room temperature, followed by incubation with antibodies: anti-capase-1 (D7F10), anti-IL-1β (3A6), anti-p-AMPK-α (Thr172;40H9), anti-AMPK-α (23A3), and anti-β-actin (AC-15). The bands were visualized with an enhanced chemiluminescent direct labeling (ECL) system. The Gel Pro Analyzer 4.5, 2000 software (Media Cybernetics; Silver Spring, MD, United States) was used to determine the band density.
Liver was perfused with saline and homogenized in Tris-HCl buffer (20 mmol/L, pH 7.4). The homogenates were centrifuged at 2500 ×g for 10 min at 4 °C. Two hundred microliters of homogenates was analyzed for malondialdehyde (MDA) levels using a kit (Cell Biolabs, Inc.; San Diego, CA, United States), and the value was read at 532 nm.
Intracellular ROS levels in HepG2 cells were determined using 2′,7′-dichlorodihydrofluorescein diacetate (DCFDA). Cells were seeded on a 96-well bottom dishes. After the treatment described above in Cell culture and treatment, the cells were collected, and incubated with 10 μmol/L DCFDA (Molecular Probes; CA, United States) for 30 min at 37 °C. DCFDA fluorescence intensity was measured at 530 nm with an excitation wavelength of 488 nm using flow cytometry (Becton Dickinson; Heidelberg, Germany). The percentage of ROS-producing cells was determined by counting only those cells that produced high levels of ROS.
Data are presented as mean ± SD. Statistical significance was determined with one-way analysis of variance (ANOVA) followed by Tukey’s test for multiple comparisons using the GraphPad Instant program (GraphPad Software version 5.00; GraphPad Software Inc.; San Diego, CA). P < 0.05 was considered statistically significant. The statistical methods of this study were reviewed by a biomedical statistician.
The body weight and BMI of the HFD group were significantly higher compared with those of control mice (all P < 0.05) (Table 2). The food intake was greater in the HFD group compared with the HFD + LPS and HFD + LPS + GW501516 groups. The proportion of liver weight to body weight or body length was similar among the four groups. Serum AST and ALT levels were significantly increased in the HFD group compared with the control group (AST, 123.8 ± 30.54 IU/L vs 52.6 ± 10.33 IU/L; ALT, 136.6 ± 69.43 IU/L vs 23.4 ± 4.04 IU/L, all P < 0.05). GW treatment in HFD+LPS group significantly reduced serum AST and ALT compared with HFD (AST, 45.67 ± 11.11 IU/L vs 123.8 ± 30.54 IU/L; ALT, 20.5 ± 6.12 IU/L vs 136.6 ± 69.43 IU/L, all P < 0.05). HFD and/or LPS injection had no significant effect on TG or TC levels.
| Control | HFD | HFD + LPS | HFD + LPS + GW | |
| (n = 5) | (n = 5) | (n = 5) | (n = 6) | |
| Food/wk (g) | 20.78 ± 1.41 | 17.33 ± 0.82a | 14.08 ± 1.28ac | 13.92 ± 1.23ac |
| BW (g) | 26.70 ± 0.69 | 41.04 ± 4.69a | 30.98 ± 3.86c | 29.87 ± 3.31c |
| BMI (g/cm2) | 0.33 ± 0.02 | 0.41 ± 0.04a | 0.36 ± 0.02c | 0.35 ± 0.02c |
| Liver/BW (%) | 3.50 ± 0.30 | 3.04 ± 0.46 | 2.77 ± 0.27 | 3.08 ± 0.94 |
| Liver/BL (g/cm) | 0.10 ± 0.01 | 0.13 ± 0.03 | 0.09 ± 0.01 | 0.10 ± 0.03 |
| AST (IU/L) | 52.6 ± 10.33 | 123.8 ± 30.54a | 62.0 ± 13.71c | 45.67 ± 11.11c |
| ALT (IU/L) | 23.4 ± 4.04 | 136.6 ± 69.43a | 49.6 ± 31.09c | 20.5 ± 6.12c |
| TG (mg/dL) | 97.8 ± 19.33 | 69.6 ± 21.55 | 65.2 ± 13.16 | 79.2 ± 23.82 |
| TC (mg/dL) | 86.6 ± 10.92 | 118.4 ± 37.16 | 116.0 ± 9.43 | 135.3 ± 34.09a |
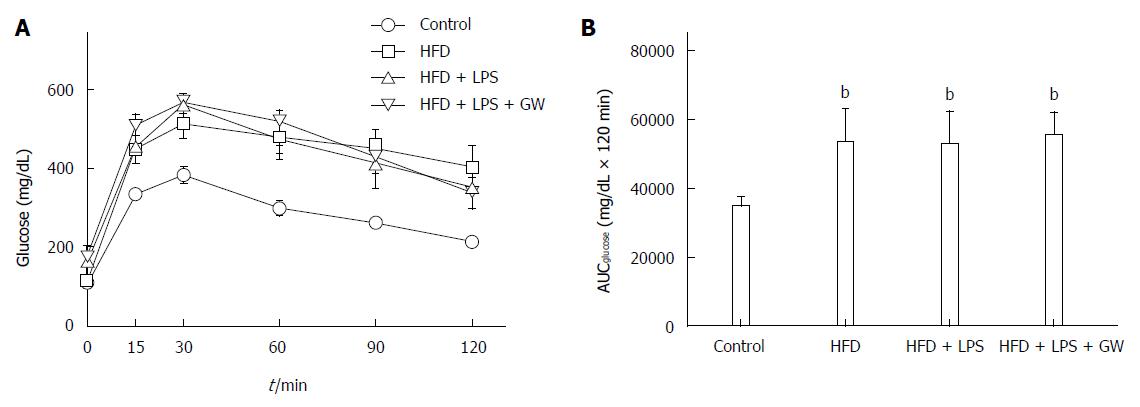
When subjected to a glucose tolerance test, glucose levels of control mice peaked at 30 min and returned to the basal level (Figure 1A). As expected, mice fed the HFD with or without LPS infection were more glucose-intolerant compared with controls, as demonstrated by the significant increase in the area under the curve (AUC) (53108 ± 9750 mg/dL and 52653 ± 9425 mg/dL vs 34545 ± 2776 mg/dL × 120 min, all P < 0.01) (Figure 1B). However, this glucose intolerance was not abolished by GW501516 treatment.
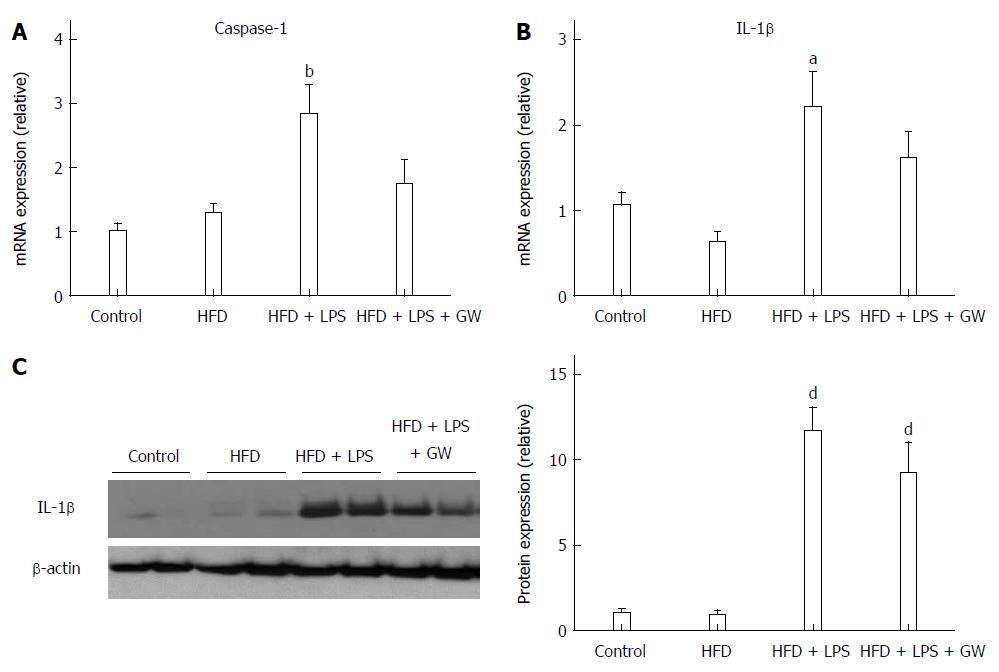
In addition to steatosis, chronic inflammation is an important contributing factor in NASH pathogenesis[5]. To investigate the inflammatory response in this model, we measured hepatic caspase-1 and IL-1β levels. The gene expression level of caspase-1, which is known to be activated by the NLRP3 inflammasome complex and to cleave pro-IL-1β to the active form, was increased in the HFD + LPS group relative to the control group (P < 0.01) (Figure 2A). The hepatic gene expression and protein levels of IL-1β were also increased in the HFD + LPS group relative to the control group (P < 0.05 for mRNA expression and P < 0.001 for protein-level expression) (Figure 2B and C). When compared with HFD+LPS, PPAR-delta agonist GW treatment reduced hepatic caspase-1 mRNA (2.8-fold vs 1.7-fold) and IL-1β mRNA (2.2-fold vs 1.6-fold).
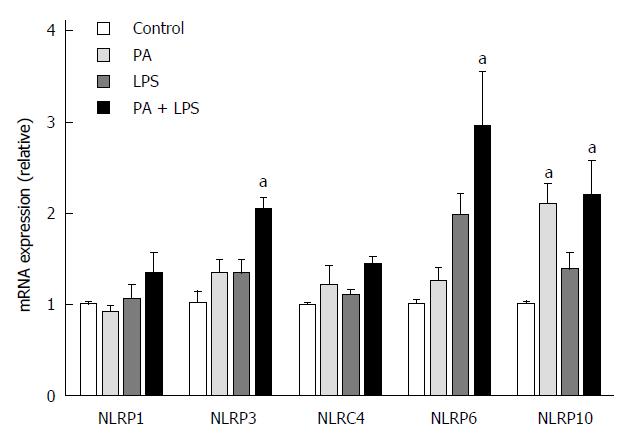
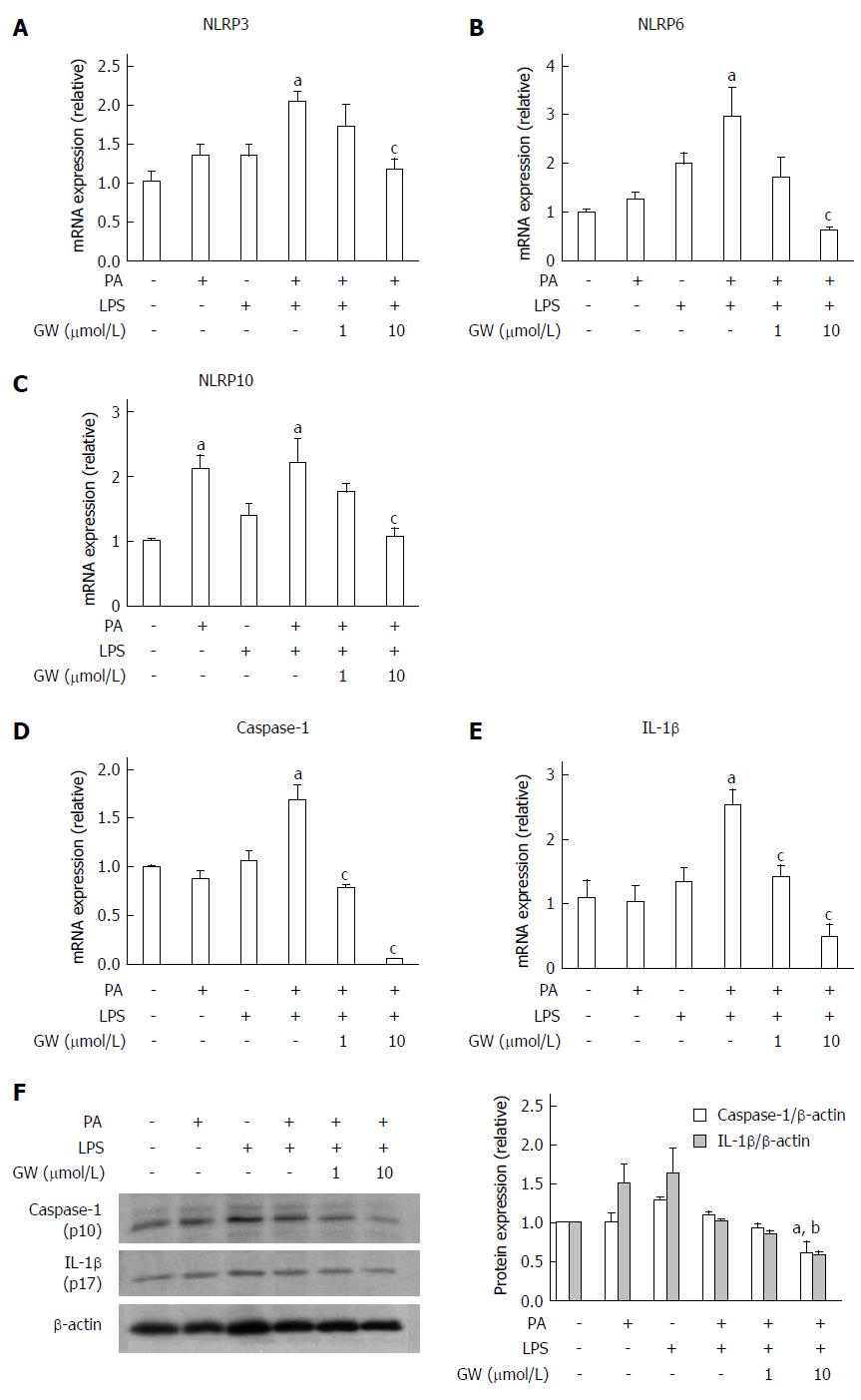
We also investigated the effect of PPAR-δ activator on inflammasome induced by PA and LPS in HepG2 cells. First of all, we analyzed the PPAR-δ protein expression itself in various stimuli. In vitro exposure with- or without stimuli such as LPS only, PA only or GW only, or combined with it, PPAR-delta protein expressions were not different. After then, we analyzed the expression of several inflammasome-related mRNAs upon activation with PA and/or LPS. PA and LPS together elicited the mRNA expression of NLRP3, NRLP6, and NLRP10 (all P < 0.05) (Figure 3). We next tested whether GW501516 could prevent the overexpression of such inflammasome components and the downstream pro-inflammatory signals. As shown in Figure 4, treatment with 10 μmol/L GW501516 reduced the PA and LPS-induced mRNA expression of NLRP3, NRLP6, and NLRP10 (all P < 0.05). GW501516 also reduced the expression of caspase-1 and IL-1β mRNA in a dose-dependent manner (all P < 0.05) (Figure 4D and E). In concordance with mRNA expression, the presence of GW501516 reduced the PA and LPS-induced production of caspase-1 and IL-1β protein expression (Figure 4F).
Histological images of liver pathology were obtained. Liver sections were stained with HE and Oil Red O. In mice fed the standard diet, there was no detectable fatty change in the microscopic image. By contrast, relative to the control group, mice on a 12-wk HFD with or without LPS injection developed extensive macrovesicular steatosis (0.0 vs 17.0% ± 5.7% and 16.4% ± 14.7%, all P < 0.05) and inflammation around the perisinusoidal area. Ballooned hepatocytes and fibrosis were not observed in any study group. In mice that were administrated GW501516, the intensity of fat accumulation (2.7% ± 2.6%) and NAS (0.5) were significantly decreased (P < 0.05) compared to those of the HFD + LPS group (16.4% ± 14.7% and 1.8%, respectively) (Figure 5A, B and C).
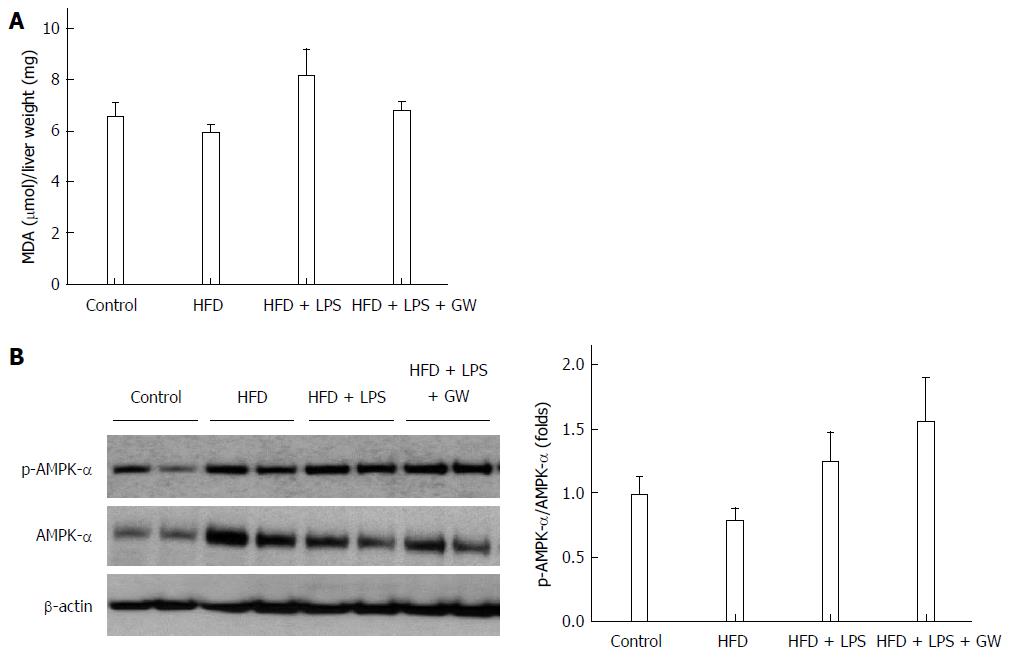
Oxidative stress is thought to play a role in the pathogenesis of NASH, and ROS are essential for inflammasome activation[7,14]. A large body of evidence indicates that AMP-activated protein kinase (AMPK) is an essential regulator of fatty acid metabolism[21], and suppresses ROS generation by regulating intracellular nicotinamide adenine dinucleotide phosphate (NADPH) production[18,24]. In the in vivo study, levels of MDA, a product of lipid peroxidation, in liver homogenates, there was no statistically significant difference in four groups (Figure 6A). We also examined the effect of GW501516 on AMPK activation. As the activity of AMPK correlates with phosphorylation at Thr-172, the activation of AMPK was assessed by determining phosphorylation of AMPKα. As shown in Figure 6B, treatment with GW501516 increased the levels of AMPKα phosphorylation in mice fed the HFD with LPS injection by 1.6-fold compared to the control group.
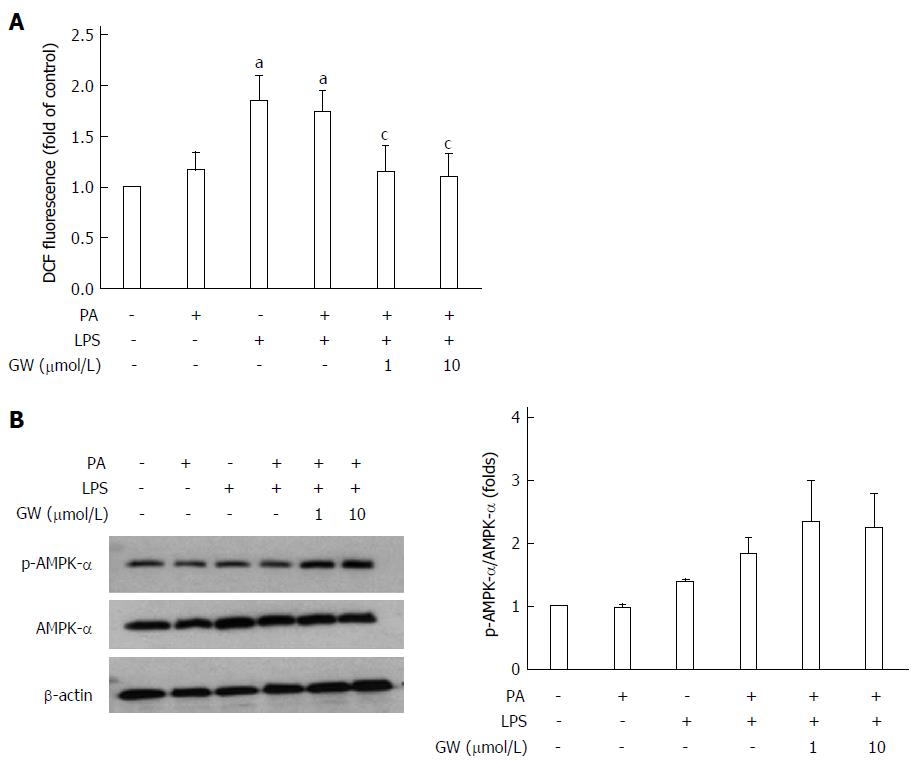
In the in vitro study, the effect of GW501516 on PA and LPS-induced ROS production was investigated using DCFDA to detect cellular ROS levels. As illustrated in Figure 7A, incubating HepG2 cells with LPS and LPS + PA induced a significant increase in the cellular ROS level at 16 h (all P < 0.05 vs control group). Treatment with GW501516 (1 μmol/L and 10 μmol/L) attenuated the ROS generation in the PA- and LPS-stimulated HepG2 cells (all P < 0.05 vs PA + LPS group). In addition, we assessed AMPK activation with PA and LPS. PA or LPS alone had little effect on phosphorylation of the AMPKα subunit. However, adding GW501516 (1 μmol/L and 10 μmol/L) substantially increased phosphorylation of the AMPKα subunit in HepG2 cells (2.3- and 2.2-fold vs control group) (Figure 7B). These in vivo and in vitro results demonstrated that GW501516 could suppress hepatic oxidative stress and enhance phosphorylation of the AMPKα under high fat conditions.
Although inflammasome is known to have an important role in metabolic syndrome such as obesity and diabetes, there is few study to assess its role in the pathogenesis of NAFLD/NASH and the therapeutic implication of the PPAR-δ activator on inflammasome activation in NAFLD model.
The results of our study suggested that IL-1β, caspase, NLRP6 and NLRP10, as well as NLRP3, were activated in NAFLD models and PPAR-δ activator GW501516 has the ability to reduce hepatic steatosis, inflammation, and oxidative stress in mice fed with HFD. Also, these results were confirmed in the in vitro models.
A recent study demonstrated that NLRP6 is important in the self-renewal and integrity of the intestinal epithelium[25]. NLRP6 seemed to negatively regulate the progression from NAFLD to NASH by preventing the increase in colitogenic bacteria, indicating a role for NLRP6 in the pathogenesis of NAFLD[17,26]. On the other hand, the function of NLRP10 remains largely uncharacterized. With the exception of the present study, few studies have described the association between NLRP10 inflammasome and NAFLD. Further research is required into the exact mechanisms of how NLRP6 or NLRP10 functions in NAFLD.
To date, NLRP3 is the most widely studied inflammasome linked to obesity, insulin resistance, and atherosclerosis[15-18]. Excessive formation of ROS, which results in oxidative stress, is the central and common element for activating the NLRP3 inflammasome[14]. In addition, IL-1β, which is stimulated by islet amyloid polypeptide, promotes β-cell dysfunction and cell death, linking NLPR3 activation to insulin resistance[27]. Therefore, the role of inflammasome in the pathogenesis of NAFLD, the hepatic manifestation of metabolic syndrome, has been receiving increased attention.
In the present study, we showed that several inflammasomes, including NLRP3, NRLP6, and NLRP10, were activated in parallel with the production of ROS and the overexpression of caspase-1 and IL-1β in HepG2 cells after stimulation with PA and LPS. Overproduction of these pro-inflammatory cytokines was also confirmed in mice fed the HFD with LPS injection. These results are consistent with those from previous diet-induced NASH models including the methionine-choline-deficient (MCD) diet[28], the HFD [18], and the choline-deficient amino acid-defined (CDAA) diet[29].
PPAR-δ is the only subtype in the PPAR subfamily of nuclear receptors that is not a target for currently used drugs. Several studies have revealed that PPAR-δ activation exerts many metabolic effects, including reducing hepatic glucose production, increasing fatty acid catabolism in adipose tissue and muscle, and lowering the inflammatory status[19-21,30,31]. Until now, studies on the relationship between PPAR-δ activation and inflammasome have been scarce. In a recent study, the PPAR-δ agonist GW0742 was shown to attenuate the renal dysfunction and inflammation caused by chronic high-fructose corn syrup (HFCS-55) exposure by preventing activation of the NLRP3 inflammasome in the kidney[32]. In the present study, the PPAR-δ agonist GW501516 was found to reduce the activation of inflammasome as well as the overproduction of pro-inflammatory cytokines in HepG2 cells and the livers of mice. Moreover, GW501516 alleviated hepatic steatosis in vivo.
AMPK is a key metabolic regulator in cellular and organismal survival owing to its ability to maintain metabolic homeostasis; it is an essential mediator for fatty acid metabolism[18,21,33]. Several studies have demonstrated that PPAR-δ prevents the downregulation of AMPK[21,30]. In this study, although GW501516 treatment did not have an influence on the lipid profile in mice, it did increase phosphorylation of the AMPK-α subunit, both in vivo and in vitro. AMPK also has an anti-inflammatory effect and is associated with oxidative stress via suppression of ROS production[18,24]. The current study also demonstrated, for the first time, that the PPAR-δ agonist suppressed the ROS production induced by PA and LPS in the hepatocyte cell line. In immune cells such as macrophages, the AMPK-ROS signaling pathway is known to be associated with the activation of the NLRP3 inflammasome[18]. The current study may provide a clue into the complex role of PPAR-δ in metabolism, and suggests that the negative effect of PPAR-δ agonist on inflammasome activation in hepatocytes may be associated with the AMPK-ROS signaling pathway.
In the present study, we could not demonstrate statistical significant positive effect of PPAR-δ activator on insulin resistance and hyperlipidemia. Consistent with these findings, PPAR-δ protein expression were not different in controls and PA treatment with or without GW501516. The lipid-lowering and anti-diabetic effects of PPAR-δ, as well as its liver-protective effect, are already well documented[21,34-36]. In recent human clinical trials, 8 wk of therapy with MBX-8025 and GFT505, which are a novel PPAR-δ agonist and a dual PPAR-α/PPAR-δ agonist, respectively, significantly improved the plasma lipid profile, insulin resistance, and, interestingly, also decreased liver enzyme levels[34,37]. In our study, the treatment duration of GW501516 in mice was relatively short (3 wk), which may explain the insufficient effect observed on hyperlipidemia and glucose intolerance. Also, increasing oxidative capacity of muscle cells without provoking insulin sensitivity might supports our findings because PPAR-δ is highly expressed in the skeletal muscle, heart, and pancreatic β-cells, as well as in the liver[38]. Future challenges in determining the complex mechanism and cross-talk of PPAR-δ in different tissues and cell types are expected.
Recently, accumulating evidence has shown that bacterial endotoxins play a key role in the pathogenesis of NASH[28,39,40]. LPS, as an exogenous ligand for Toll-like receptor 4 (TLR4), may be capable of stimulating inflammasome expression, cytokine production, and the accumulation of inflammatory cells[28,39]. Therefore, we expected that the chronic exposure of low-dose LPS would lead to liver inflammation and fibrosis in mice fed an HFD. As for our study, we were decided to inject non-lethal dose of LPS which can reflect low level of endotoxemia of NAFLD. In our animal model, mice fed the HFD with LPS injection developed hepatic steatosis, although it failed to produce the severe form of NASH characterized by balloon degeneration and severe inflammation. However, unlike the MCD diet model[41], HFD with LPS injection resulted in weight gain and glucose intolerance, and therefore might more closely reflect the characteristics of human NAFLD. Future studies using endotoxins as HFD-enhancing factors in the murine NAFLD model are required to support the results of this study.
In conclusion, the results from our in vivo and in vitro studies demonstrate that the PPAR-δ agonist GW501516 suppresses the activation of inflammasome and reduces IL-1β levels, possibly by modulation of AMPK phosphorylation and decreased production of ROS. This anti-inflammatory effect might be associated with improvement of hepatic steatosis in mice. The targeting of inflammasome by the PPAR-δ agonist may have therapeutic implications for the treatment of NAFLD.
Nonalcoholic fatty liver disease (NAFLD) is one of the most common disease of the liver. Although thiazolidinediones and antioxidants such as vitamin E have been evaluated in several clinical trials, few pharmacological treatments can be recommended at present. Inflammasome is a large, intracellular multi-protein complex that is a sensor of pathogen-associated molecular patterns (PAMPs) or damage-associated molecular patterns (DAMPs) that govern the cleavage of pro-inflammatory cytokines such as pro-interleukin (IL)-1β and pro-IL-18. Previous studies have shown that NLRP3 inflammasome is involved in the pathogenesis of obesity-induced inflammation, insulin resistance, and development of type 2 diabetes. However, the role of inflammasome in the pathogenesis of NAFLD/NASH has not been elucidated.
Peroxisome proliferator-activated receptors (PPARs) are members of the nuclear receptor superfamily, and three isotypes exist in mammals. It has become increasingly evident that PPAR-δ is also an important metabolic regulator, as its activation enhances fatty acid oxidation, improves glucose homeostasis, and attenuates macrophage inflammatory responses. Current research hot spot is to whether the treatment with the PPAR-δ agonist could ameliorate high fat-induced inflammasome activation and the inflammatory response using in vivo and in vitro NAFLD models.
Until now, studies on the relationship between PPAR-δ activation and inflammasome have been scarce. In a recent study, the PPAR-δ agonist GW0742 was shown to attenuate the renal dysfunction and inflammation caused by chronic high-fructose corn syrup-55 exposure by preventing activation of the NLRP3 inflammasome in the kidney. To investigate whether the treatment with the PPAR-δ agonist could ameliorate high fat-induced inflammasome activation and the inflammatory response in the liver, the authors were using in vivo and in vitro NAFLD models. This study showed that PPAR-δ agonist GW501516 was found to reduce the activation of inflammasome as well as the overproduction of pro-inflammatory cytokines in HepG2 cells and the livers of mice. Moreover, GW501516 alleviated hepatic steatosis in vivo.
The study results suggested that the PPAR-δ agonist GW501516 suppresses the activation of inflammasome and reduces IL-1β levels in the liver, possibly by modulation of AMPK phosphorylation and decreased production of ROS. This anti-inflammatory effect might be associated with improvement of hepatic steatosis in mice. The targeting of inflammasome by the PPAR-δ agonist may have therapeutic implications for the treatment of NAFLD.
NAFLD is a spectrum of disease entity including simple steatosis, steatosis with inflammation, fibrosis and cirrhosis. Inflammasome is a large, intracellular multi-protein complex that is a sensor of PAMPs or DAMPs that govern the cleavage of pro-inflammatory cytokines such as pro-IL-1β and pro-IL-18.
The authors studied effects of PPAR-δ activator; GW501516 on inflamasome pathway in mouse model of NASH (high fat diet, LPS/PA) and HepG2 cells culture. They show that the beneficial role in mouse liver was through increased NLRP3-10 in HepG2 cells.
P- Reviewer: Abenavoli L, Balaban YH, Laguna JC, Pirola CJ S- Editor: Qi Y L- Editor: A E- Editor: Liu XM
| 1. | Vernon G, Baranova A, Younossi ZM. Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther. 2011;34:274-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2405] [Cited by in RCA: 2292] [Article Influence: 163.7] [Reference Citation Analysis (0)] |
| 2. | Vuppalanchi R, Chalasani N. Nonalcoholic fatty liver disease and nonalcoholic steatohepatitis: Selected practical issues in their evaluation and management. Hepatology. 2009;49:306-317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 428] [Cited by in RCA: 417] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 3. | Musso G, Gambino R, Cassader M, Pagano G. Meta-analysis: natural history of non-alcoholic fatty liver disease (NAFLD) and diagnostic accuracy of non-invasive tests for liver disease severity. Ann Med. 2011;43:617-649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 886] [Cited by in RCA: 919] [Article Influence: 65.6] [Reference Citation Analysis (0)] |
| 4. | Abenavoli L, Milic N, Peta V, Alfieri F, De Lorenzo A, Bellentani S. Alimentary regimen in non-alcoholic fatty liver disease: Mediterranean diet. World J Gastroenterol. 2014;20:16831-16840. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 72] [Cited by in RCA: 77] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 5. | Farrell GC, van Rooyen D, Gan L, Chitturi S. NASH is an Inflammatory Disorder: Pathogenic, Prognostic and Therapeutic Implications. Gut Liver. 2012;6:149-171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 281] [Cited by in RCA: 304] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 6. | Neuschwander-Tetri BA. Hepatic lipotoxicity and the pathogenesis of nonalcoholic steatohepatitis: the central role of nontriglyceride fatty acid metabolites. Hepatology. 2010;52:774-788. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 726] [Cited by in RCA: 841] [Article Influence: 56.1] [Reference Citation Analysis (0)] |
| 7. | Rolo AP, Teodoro JS, Palmeira CM. Role of oxidative stress in the pathogenesis of nonalcoholic steatohepatitis. Free Radic Biol Med. 2012;52:59-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 636] [Cited by in RCA: 719] [Article Influence: 55.3] [Reference Citation Analysis (1)] |
| 8. | Carabelli J, Burgueño AL, Rosselli MS, Gianotti TF, Lago NR, Pirola CJ, Sookoian S. High fat diet-induced liver steatosis promotes an increase in liver mitochondrial biogenesis in response to hypoxia. J Cell Mol Med. 2011;15:1329-1338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 93] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 9. | Musso G, Gambino R, Cassader M, Pagano G. A meta-analysis of randomized trials for the treatment of nonalcoholic fatty liver disease. Hepatology. 2010;52:79-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 416] [Cited by in RCA: 437] [Article Influence: 29.1] [Reference Citation Analysis (0)] |
| 10. | Abenavoli L, Greco M, Nazionale I, Peta V, Milic N, Accattato F, Foti D, Gulletta E, Luzza F. Effects of Mediterranean diet supplemented with silybin-vitamin E-phospholipid complex in overweight patients with non-alcoholic fatty liver disease. Expert Rev Gastroenterol Hepatol. 2015;9:519-527. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 66] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 11. | Rosselli MS, Burgueño AL, Carabelli J, Schuman M, Pirola CJ, Sookoian S. Losartan reduces liver expression of plasminogen activator inhibitor-1 (PAI-1) in a high fat-induced rat nonalcoholic fatty liver disease model. Atherosclerosis. 2009;206:119-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 42] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 12. | Szabo G, Csak T. Inflammasomes in liver diseases. J Hepatol. 2012;57:642-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 339] [Cited by in RCA: 404] [Article Influence: 31.1] [Reference Citation Analysis (0)] |
| 13. | Kubes P, Mehal WZ. Sterile inflammation in the liver. Gastroenterology. 2012;143:1158-1172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 458] [Cited by in RCA: 517] [Article Influence: 39.8] [Reference Citation Analysis (0)] |
| 14. | Martinon F. Signaling by ROS drives inflammasome activation. Eur J Immunol. 2010;40:616-619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 439] [Cited by in RCA: 469] [Article Influence: 31.3] [Reference Citation Analysis (0)] |
| 15. | Stienstra R, Tack CJ, Kanneganti TD, Joosten LA, Netea MG. The inflammasome puts obesity in the danger zone. Cell Metab. 2012;15:10-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 212] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 16. | Vandanmagsar B, Youm YH, Ravussin A, Galgani JE, Stadler K, Mynatt RL, Ravussin E, Stephens JM, Dixit VD. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat Med. 2011;17:179-188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2148] [Cited by in RCA: 2051] [Article Influence: 146.5] [Reference Citation Analysis (0)] |
| 17. | Henao-Mejia J, Elinav E, Jin C, Hao L, Mehal WZ, Strowig T, Thaiss CA, Kau AL, Eisenbarth SC, Jurczak MJ. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature. 2012;482:179-185. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1620] [Cited by in RCA: 1880] [Article Influence: 144.6] [Reference Citation Analysis (0)] |
| 18. | Wen H, Gris D, Lei Y, Jha S, Zhang L, Huang MT, Brickey WJ, Ting JP. Fatty acid-induced NLRP3-ASC inflammasome activation interferes with insulin signaling. Nat Immunol. 2011;12:408-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1214] [Cited by in RCA: 1413] [Article Influence: 100.9] [Reference Citation Analysis (0)] |
| 19. | Poulsen Ll, Siersbæk M, Mandrup S. PPARs: fatty acid sensors controlling metabolism. Semin Cell Dev Biol. 2012;23:631-639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 300] [Cited by in RCA: 364] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 20. | Tailleux A, Wouters K, Staels B. Roles of PPARs in NAFLD: potential therapeutic targets. Biochim Biophys Acta. 2012;1821:809-818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 211] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 21. | Barroso E, Rodríguez-Calvo R, Serrano-Marco L, Astudillo AM, Balsinde J, Palomer X, Vázquez-Carrera M. The PPARβ/δ activator GW501516 prevents the down-regulation of AMPK caused by a high-fat diet in liver and amplifies the PGC-1α-Lipin 1-PPARα pathway leading to increased fatty acid oxidation. Endocrinology. 2011;152:1848-1859. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 131] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 22. | Buchner DA, Burrage LC, Hill AE, Yazbek SN, O’Brien WE, Croniger CM, Nadeau JH. Resistance to diet-induced obesity in mice with a single substituted chromosome. Physiol Genomics. 2008;35:116-122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 26] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 23. | Kleiner DE, Brunt EM, Van Natta M, Behling C, Contos MJ, Cummings OW, Ferrell LD, Liu YC, Torbenson MS, Unalp-Arida A. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313-1321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6807] [Cited by in RCA: 8234] [Article Influence: 411.7] [Reference Citation Analysis (5)] |
| 24. | Wang S, Zhang M, Liang B, Xu J, Xie Z, Liu C, Viollet B, Yan D, Zou MH. AMPKalpha2 deletion causes aberrant expression and activation of NAD(P)H oxidase and consequent endothelial dysfunction in vivo: role of 26S proteasomes. Circ Res. 2010;106:1117-1128. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 270] [Cited by in RCA: 271] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 25. | Normand S, Delanoye-Crespin A, Bressenot A, Huot L, Grandjean T, Peyrin-Biroulet L, Lemoine Y, Hot D, Chamaillard M. Nod-like receptor pyrin domain-containing protein 6 (NLRP6) controls epithelial self-renewal and colorectal carcinogenesis upon injury. Proc Natl Acad Sci USA. 2011;108:9601-9606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 251] [Cited by in RCA: 296] [Article Influence: 21.1] [Reference Citation Analysis (0)] |
| 26. | Kempster SL, Belteki G, Forhead AJ, Fowden AL, Catalano RD, Lam BY, McFarlane I, Charnock-Jones DS, Smith GC. Developmental control of the Nlrp6 inflammasome and a substrate, IL-18, in mammalian intestine. Am J Physiol Gastrointest Liver Physiol. 2011;300:G253-G263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 73] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 27. | Masters SL, Dunne A, Subramanian SL, Hull RL, Tannahill GM, Sharp FA, Becker C, Franchi L, Yoshihara E, Chen Z. Activation of the NLRP3 inflammasome by islet amyloid polypeptide provides a mechanism for enhanced IL-1β in type 2 diabetes. Nat Immunol. 2010;11:897-904. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1102] [Cited by in RCA: 1034] [Article Influence: 68.9] [Reference Citation Analysis (0)] |
| 28. | Csak T, Ganz M, Pespisa J, Kodys K, Dolganiuc A, Szabo G. Fatty acid and endotoxin activate inflammasomes in mouse hepatocytes that release danger signals to stimulate immune cells. Hepatology. 2011;54:133-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 442] [Cited by in RCA: 533] [Article Influence: 38.1] [Reference Citation Analysis (0)] |
| 29. | Miura K, Yang L, van Rooijen N, Brenner DA, Ohnishi H, Seki E. Toll-like receptor 2 and palmitic acid cooperatively contribute to the development of nonalcoholic steatohepatitis through inflammasome activation in mice. Hepatology. 2013;57:577-589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 230] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 30. | Coll T, Alvarez-Guardia D, Barroso E, Gómez-Foix AM, Palomer X, Laguna JC, Vázquez-Carrera M. Activation of peroxisome proliferator-activated receptor-{delta} by GW501516 prevents fatty acid-induced nuclear factor-{kappa}B activation and insulin resistance in skeletal muscle cells. Endocrinology. 2010;151:1560-1569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 74] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 31. | Lee CH, Olson P, Hevener A, Mehl I, Chong LW, Olefsky JM, Gonzalez FJ, Ham J, Kang H, Peters JM. PPARdelta regulates glucose metabolism and insulin sensitivity. Proc Natl Acad Sci USA. 2006;103:3444-3449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 378] [Cited by in RCA: 399] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 32. | Collino M, Benetti E, Rogazzo M, Mastrocola R, Yaqoob MM, Aragno M, Thiemermann C, Fantozzi R. Reversal of the deleterious effects of chronic dietary HFCS-55 intake by PPAR-δ agonism correlates with impaired NLRP3 inflammasome activation. Biochem Pharmacol. 2013;85:257-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 41] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 33. | Sid B, Verrax J, Calderon PB. Role of AMPK activation in oxidative cell damage: Implications for alcohol-induced liver disease. Biochem Pharmacol. 2013;86:200-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 117] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 34. | Bays HE, Schwartz S, Littlejohn T, Kerzner B, Krauss RM, Karpf DB, Choi YJ, Wang X, Naim S, Roberts BK. MBX-8025, a novel peroxisome proliferator receptor-delta agonist: lipid and other metabolic effects in dyslipidemic overweight patients treated with and without atorvastatin. J Clin Endocrinol Metab. 2011;96:2889-2897. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 131] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 35. | Cariou B, Zaïr Y, Staels B, Bruckert E. Effects of the new dual PPAR α/δ agonist GFT505 on lipid and glucose homeostasis in abdominally obese patients with combined dyslipidemia or impaired glucose metabolism. Diabetes Care. 2011;34:2008-2014. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 136] [Cited by in RCA: 137] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 36. | Nagasawa T, Inada Y, Nakano S, Tamura T, Takahashi T, Maruyama K, Yamazaki Y, Kuroda J, Shibata N. Effects of bezafibrate, PPAR pan-agonist, and GW501516, PPARdelta agonist, on development of steatohepatitis in mice fed a methionine- and choline-deficient diet. Eur J Pharmacol. 2006;536:182-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 179] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 37. | Cariou B, Hanf R, Lambert-Porcheron S, Zaïr Y, Sauvinet V, Noël B, Flet L, Vidal H, Staels B, Laville M. Dual peroxisome proliferator-activated receptor α/δ agonist GFT505 improves hepatic and peripheral insulin sensitivity in abdominally obese subjects. Diabetes Care. 2013;36:2923-2930. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 167] [Cited by in RCA: 181] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 38. | de Lange P, Lombardi A, Silvestri E, Goglia F, Lanni A, Moreno M. Peroxisome Proliferator-Activated Receptor Delta: A Conserved Director of Lipid Homeostasis through Regulation of the Oxidative Capacity of Muscle. PPAR Res. 2008;2008:172676. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 39. | Kudo H, Takahara T, Yata Y, Kawai K, Zhang W, Sugiyama T. Lipopolysaccharide triggered TNF-alpha-induced hepatocyte apoptosis in a murine non-alcoholic steatohepatitis model. J Hepatol. 2009;51:168-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 136] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 40. | Shanab AA, Scully P, Crosbie O, Buckley M, O’Mahony L, Shanahan F, Gazareen S, Murphy E, Quigley EM. Small intestinal bacterial overgrowth in nonalcoholic steatohepatitis: association with toll-like receptor 4 expression and plasma levels of interleukin 8. Dig Dis Sci. 2011;56:1524-1534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 155] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 41. | Takahashi Y, Soejima Y, Fukusato T. Animal models of nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. World J Gastroenterol. 2012;18:2300-2308. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 379] [Cited by in RCA: 414] [Article Influence: 31.8] [Reference Citation Analysis (2)] |