Published online Mar 14, 2015. doi: 10.3748/wjg.v21.i10.3139
Peer-review started: July 24, 2014
First decision: September 27, 2014
Revised: November 7, 2014
Accepted: December 20, 2014
Article in press: December 22, 2014
Published online: March 14, 2015
Processing time: 237 Days and 21.9 Hours
Eosinophilic gastroenteritis (EG) is a rare, distinct clinical entity, and EG associated with eosinophilic cystitis (EC) is extremely rare and has not been well documented. Here, we report two cases of EG and coexistent EC along with findings from computed tomography (CT) and magnetic resonance imaging (MRI). An 18-year-old male with a history of hematuria, urgency and occasional urodynia for two weeks and a 34-year-old male with a history of abdominal distention for one week were admitted to our hospital. Abdominal contrast-enhanced CT in both patients revealed wall thickening in different parts of the gastrointestinal tract with inhomogeneous reinforcement, coexistent with local or diffuse bladder wall thickening with progressive enhancement, and also showed that the bladder mucosal lining was nondestructive. Pelvic MRI showed that the local or diffuse thickened bladder wall was iso-intense on T1-weighted images, hypo-intense on T2-weighted images, and slightly restricted on diffusion weighted imaging (DWI) in one case. After therapy, the thickened wall of the gastrointestinal tract and urinary bladder had improved markedly in the two cases. To the best of our knowledge, this is the first report on the radiological imaging of EG and coexistent EC by both CT and MRI and the first with DWI findings.
Core tip: This article reports two rare cases of eosinophilic gastroenteritis (EG) and coexistent eosinophilic cystitis (EC) with findings from computed tomography (CT) and magnetic resonance imaging (MRI), which have not previously been documented in detail. To the best of our knowledge, this is the first report on EG and coexistent EC with examinations by both CT and MRI, as well as the first one with diffusion-weighted imaging findings.
- Citation: Han SG, Chen Y, Qian ZH, Yang L, Yu RS, Zhu XL, Li QH, Chen Q. Eosinophilic gastroenteritis associated with eosinophilic cystitis: Computed tomography and magnetic resonance imaging findings. World J Gastroenterol 2015; 21(10): 3139-3145
- URL: https://www.wjgnet.com/1007-9327/full/v21/i10/3139.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i10.3139
Eosinophilic gastroenteritis (EG), associated with eosinophilic cystitis (EC), is an extremely rare entity first described in 1974 by Gregg et al[1], only a few cases have been documented in the literature[1-5], of which only five cases were the computed tomography (CT) characteristics of EG and coexistent EC[2,4] described in detail. Further, no case that discusses the imaging features of both CT and magnetic resonance imaging (MRI) has been published. Here, we present two patients presenting EG associated with EC and their examination by CT and MRI. To the best of our knowledge, this is the first report describing both CT and MRI data for patients with EG and associated EC and the first one with data from diffusion-weighted imaging (DWI).
An 18-year-old Chinese male patient presented with hematuria, urinary urgency and occasional urodynia for a span of two weeks. Ten days later, he experienced intermittent bouts of nausea, vomiting and upper abdominal distention. He had no history of allergy, eczema or wheezing. On admission, physical examination showed mild upper abdominal tenderness. Laboratory results showed an increased white blood cell count (15100/μL) with 52.5% eosinophils (normal range 0.5%-5%). Immunological studies showed an elevated IgE (490 IU/mL; normal range < 165 IU/mL). Allergy tests showed that antigens of crabs, shrimp, lobsters and salmon were weakly positive.
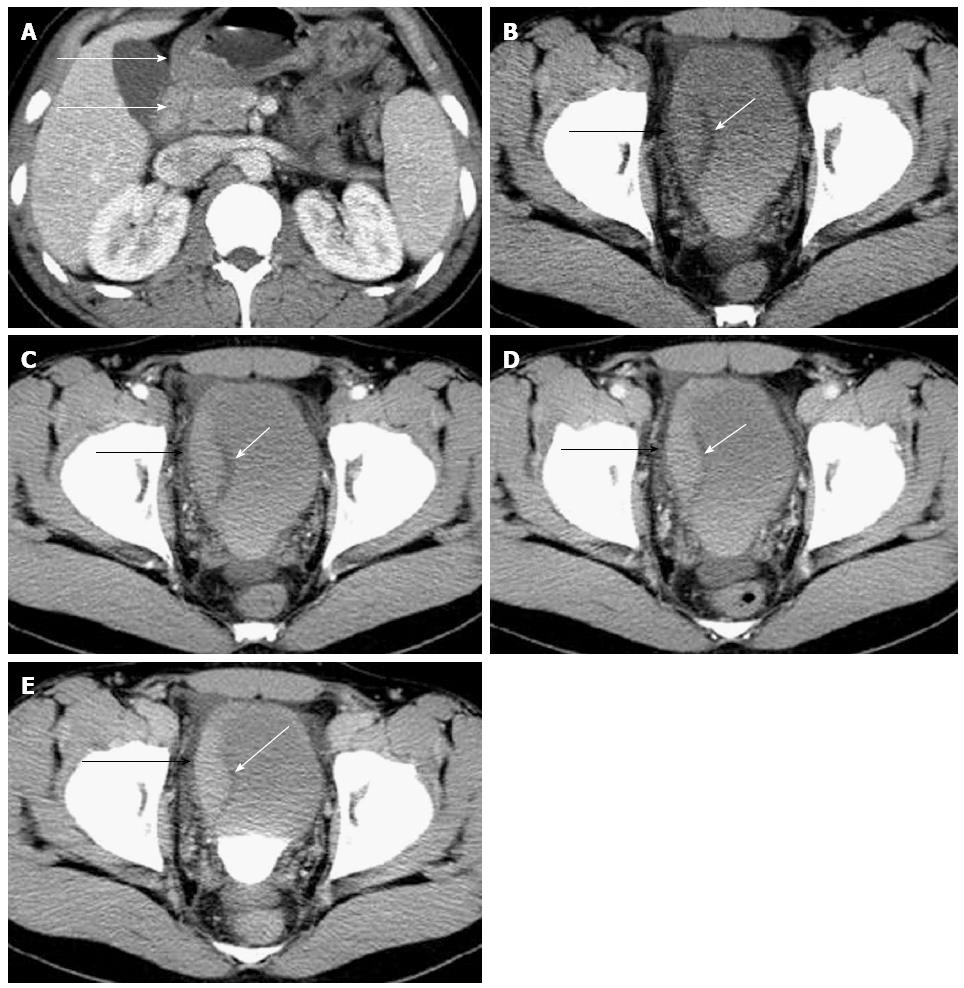
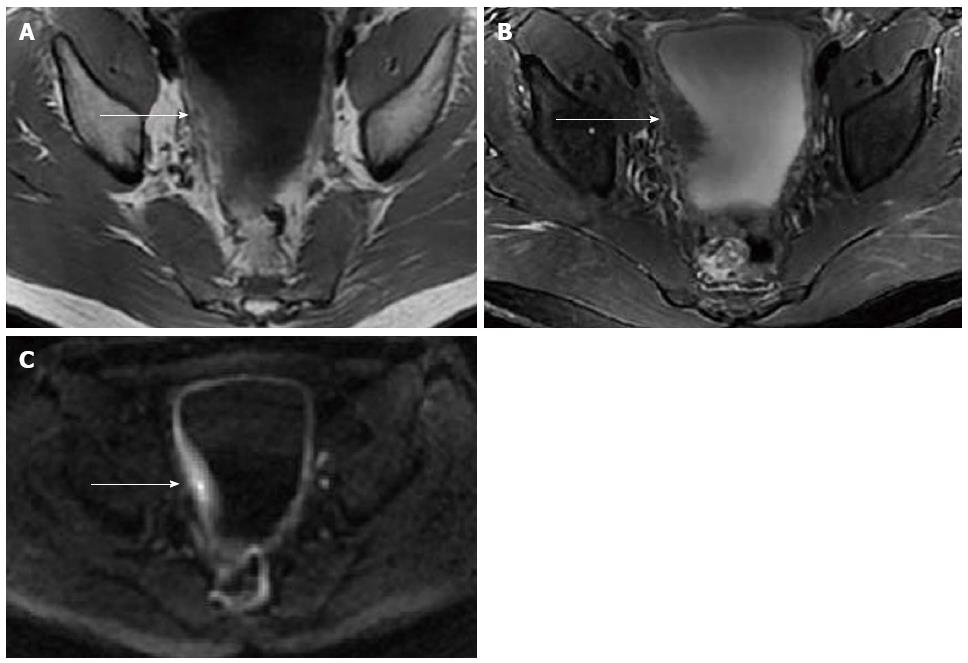
Abdominal ultrasound examination showed bladder wall thickening and moderate ascites. Contrast-enhanced CT showed irregular wall thickening of the gastric antrum and duodenum with inhomogeneous reinforcement (Figure 1A), and the presence of mural stratification and increased enhancement of the mesentery and fat tissue of the small bowel. Contrast-enhanced CT also showed asymmetrical thickening of the right lateral bladder wall with progressive enhancement on the arterial, portal, venous and delayed phase scans (Figure 1B-E). Also present was a low-density mucosal line between the thickened right lateral bladder wall and urinary bladder cavity. Additionally, there were moderate ascites in the abdominopelvic cavity. Pelvic MRI showed that the right part of the bladder wall measured up to 18 mm in thickness. The thickened bladder wall displayed mildly homogeneous hyper-intensity relative to muscle on T1-weighted images (T1WI), hypo-intensity on fat-saturated T2-weighted images (T2WI), and the diffusion was slightly restricted on DWI (b-value = 1000) (Figure 2).
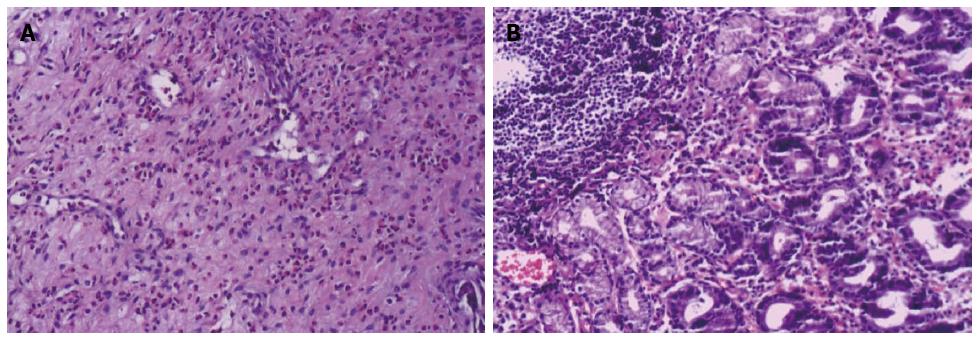
Gastroscopy revealed a gastric antral ulcer 0.6 cm × 0.8 cm in diameter, with mucosal hyperemia and edema and slight secretion in the gastric cavity. Cystoscopy showed a mass in the right lateral bladder wall with edematous and hyperemic mucosa. Biopsy from the patient’s stomach and duodenum demonstrated extensive subserosal eosinophilic infiltration. Histopathological examination of the submitted specimen from cystoscopy further showed abundant eosinophilic infiltrations of the mucosa and submucosa of the bladder wall (Figure 3A and B). Based on the above clinical, imaging and pathological findings, a diagnosis of EG associated with EC was considered.
The patient was treated with oral prednisolone, 60 mg per day (1 mg/kg per day). After one week, the symptoms subsided and the peripheral blood eosinophilia disappeared. Immunological studies returned to normal value, and the thickened wall of the gastric antrum, duodenum and bladder had improved markedly. There had been no evidence of recurrence during the past 32 mo of follow-up.
A 34-year-old Chinese male presented with abdominal distention for one week, acid reflux for three weeks, and loss of appetite for two weeks. The patient had no history of allergy, eczema or wheezing. There were no remarkable urinary symptoms. Physical examination showed right lower quadrant abdominal pain with distention and shifting dullness was positive. Laboratory examination showed an increased white blood cell count (14400/μL) with 21.1% eosinophils (normal range 0.5%-5%). Immunological studies showed elevated IgE (343 IU/mL; normal range < 165 IU/mL), rheumatoid factor (20.7 IU/mL; normal range < 20 IU/mL), and C-Reaction protein (12.9 mg/dL; normal range < 0.8 mg/dL). Allergy test results were unremarkable.
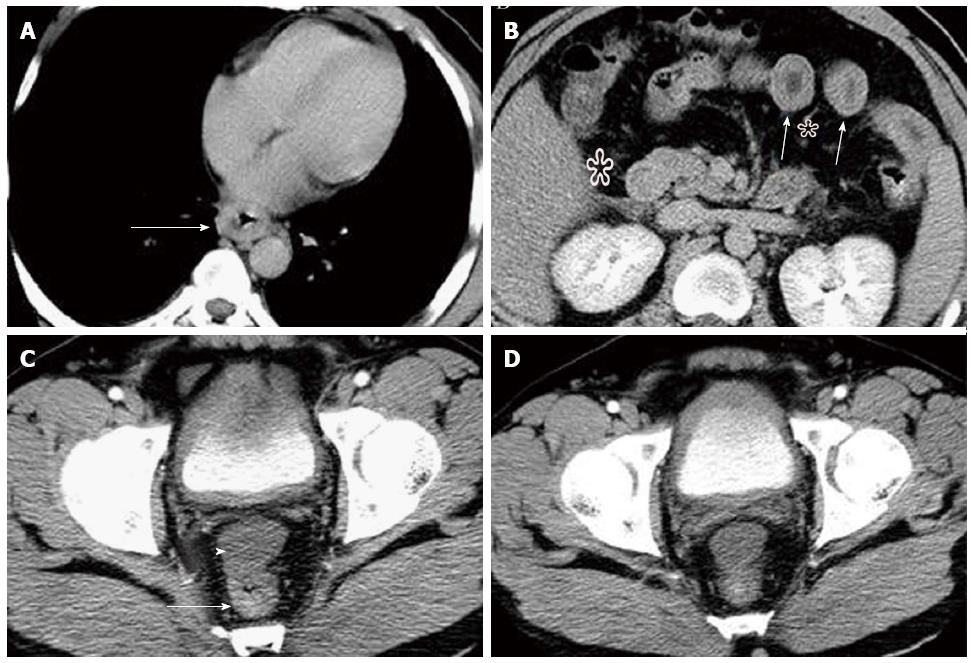
Abdominal ultrasound examination showed severe fatty liver, multiple gallbladder stones, urinary bladder wall thickening and moderate ascites. Contrast-enhanced CT showed multifocal gastrointestinal tract wall thickening (Figure 4A-C) from the lower esophagus to rectum with fat stratification around, and diffuse, marked circumferential bladder wall thickening with clear margins and preservation of the mucosa (Figure 4D). Additionally, there were moderate ascites in the abdominopelvic cavity.
Endoscopy showed edematous and hyperemic mucosa, and mild secretion from the lower esophagus to rectum. Biopsies showed extensive subserosal eosinophilic infiltration from the lower esophagus to rectum. Cystoscopy showed nearly circumferential bladder wall thickening with edematous and hyperemic mucosa. Histopathological examination also showed numerous eosinophilic infiltrations of the mucosa and submucosa. Based on these findings, a diagnosis of EG associated with EC was considered.
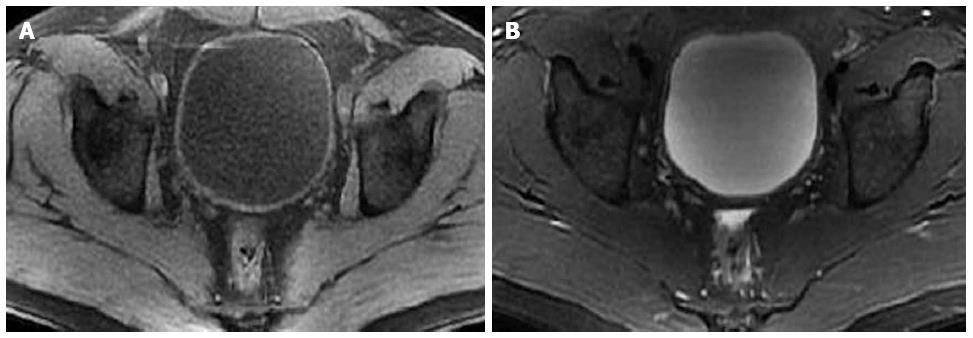
After one week of steroid therapy (oral prednisolone, 1 mg/kg per day), the pelvic MRI scan displayed a smooth bladder wall without evident thickening (Figure 5A and B). The symptoms subsided, the peripheral blood eosinophilia disappeared and immunological studies returned to normal values. The patient was then discharged from the hospital and there has been no evidence of recurrence during the past 24 mo of follow-up.
EG is a rare eosinophil-rich gastrointestinal disorder without identified causes of eosinophilia such as drug reactions, parasitic infections and malignancy. On the other hand, as first reported by Brown[6] and Palubinskas[7] independently in 1960, EC is also a rare clinicopathological condition characterized by transmural eosinophilic inflammation of the bladder. EG is associated with EC disorder in 4.5% of cases[8]. Both of our patients had EG associated with EC disorder. EC affects adult men and women equally[8]. Both of our two cases are males.
Although the definite etiology of EG and EC is unclear, there is evidence that suggests an allergy may cause this disease[9]. Our Case 1 showed allergy tests were weakly positive. Other evidence includes peripheral eosinophilia being present in approximately 43% of patients[8] and in our two cases with elevated IgE, a relief of symptoms was observed after steroid therapy.
The symptoms of EC are most frequently irritative and mimic those of urinary tract infection. The clinical presentation, course and outcome of EG depend on the location and depth of the different layers of the gastrointestinal tract by eosinophilic involvement[10]. According to the reported literature, the primary sites are the stomach and the small intestine. However, the esophagus, the large intestine and the rectum may also be affected[11-13]. EG is classified into mucosal, submucosal or muscular, and serosal types[10]. Mucosal involvement presents with vomiting, blood in the stool, iron deficiency anemia, malabsorption and protein-losing enteropathy. Patients with muscle layer involvement typically present with a pyloric or an intestinal obstruction. Serosal involvement usually presents with ascites, which were also the clinical presentations in our two cases. This disease usually has a relatively benign clinical course and moderate self-limited recurrence[14], though there were no recurrences during follow-up of our two cases after being treated.
The diagnosis of EC and EG is confirmed by pathological biopsy[3,9]. Histologically, there is transmural inflammation, with the most intense eosinophilic inflammatory change found in the lamina propria of the bladder wall, accompanying with edema or muscle necrosis[15]. Both of our two cases demonstrated infiltration in the mucosa and submucosa bladder wall and the different subserosal locations in the gastrointestinal tract of numerous eosinophils. Famularo et al[16] reported that multiple biopsy specimens should be taken to avoid a misdiagnosis of EG.
So far, only a few radiological results of EG with associated EC have been described[2,17,18]. EG may have different CT features owing to the different locations and layers of gut involvement. Intestinal wall thickening is the main CT feature in all types of EG on CT[4,19,20], as well as in our two cases. Mucosal abnormalities of the bowel wall are the earliest radiographic manifestation in EG[19] with layering and fold thickening, which is considered to be a characteristic appearance of the mucosal EG on CT. Intestinal stenosis is the main characteristic of muscular EG on CT. Our two patients presented with luminal narrowing without obstruction, the cause of which is thought to be functional hypertonicity or spasm, which is different from intestinal stenosis. The main CT feature of serosal EG is ascites[4,20], which was the presentation in our two cases. Moreover, both of our two cases demonstrated stratification and increased enhancement of the mesentery and fat tissue of the gastrointestinal tract, which were also the hallmarks of the serosal type of EG, thus highlighting the fact that both of our cases belong to the serosal type of EG. The CT findings of EC show that the bladder wall may be normal or thickened, including single or multiple bladder sessile masses[8,18]. In addition to these, suspected lymph nodes and upper urinary tract dilatation may also be observed[8]. Among them, only five cases described the CT characteristics of EG and coexistent EC[2,4] in detail. Kim et al[2] and Zhou et al[4] described the CT findings from five cases of EC, which showed marked and diffuse bladder wall thickening with intense, progressive enhancement, and preservation of the mucosal line. No case showed a localized mass in the right lateral bladder wall as in Case 1. However, it was compatible with Case 2, which showed diffuse, marked, nearly circumferential bladder wall thickening with clear margins and smooth mucosal lining, based on edema of the inflammation mucosa. Moreover, CT findings from the five cases of EG displayed that generalized edema in the jejunum and ileum, or diffuse wall thickening of the distal ileum or most of the digestive tract with a target sign, were not very comparable with our two cases that showed the wall of the gastric antrum and duodenum irregularly thickened, with inhomogeneous reinforcement in Case 1 and multifocal segmental gastrointestinal tract wall thickening from the lower esophagus to the rectum in Case 2, with surrounding fat stratification.
To the best of our knowledge, there is no case describing the MRI characteristics of EC and coexistent EG. Only a few MRI findings of EC have been described[8,17,18]. Van den Ouden[8] reported that MRI was conducted in only one patient of 135 cases, showing soft tissue at the bladder base causing urinary retention. Wong-You-Cheong et al[18] described a single, sessile mass arising from the posterior bladder wall that was hyperintense with T1WI, isointense with T2WI, and enhanced after administration of contrast material. Case 1 had similar findings with T1WI features, but displayed different hypointense manifestations on T2WI, in contrast to the literature. However, Tamai et al[17] revealed smooth and nearly circumferential thickening of the bladder wall showing a distinct, low signal intensity on T2WI. The signal features on T2WI were consistent with Case 1, which may correlate to massive eosinophilic infiltration and fibrosis determined histologically. Therefore, we think that more studies are needed to prove the signal-intensity features seen by T2WI. Among the existing studies, none have mentioned DWI findings. The DWI features of slight hyperintensity on MRI have not been reported previously according to our literature search, which might also correlate with high cellularity due to massive eosinophilic infiltration and proliferation of fibrous tissue.
For differential diagnosis, the most important imaging features of both bladder and gastrointestinal tract wall thickening should differentiate EC and EG from tuberculosis, Crohn disease, bladder endometriosis, bladder and gastrointestinal neoplasm, and exposure to chemotherapy or irradiation.
In conclusion, we report the CT and MRI findings for two rare cases of EG with associated EC. The characteristic CT and MRI features include multifocal segmental digestive tract wall thickening, luminal narrowing without obstruction, ascites, local or diffuse bladder wall thickening, hypointense or isointense appearance on T2WI, slightly restricted appearance on DWI, and progressive enhancement with the presence of the characteristic mucosal-lining. Moreover, the identification of these features combined with allergy, peripheral eosinophilia, and respond positively to steroid therapy will likely make the correct diagnosis prior to endoscopic biopsies.
The authors thank Mohamed Adil Shah Khoodoruth and Bibi Zaina Ramjaun from Mauritius for their assistance in checking and reviewing this paper.
Two middle-aged male patients presented with dissimilar symptoms; one presented with hematuria, urgency and urodynia, and the other presented with abdominal distention, acid reflux, and loss of appetite with no remarkable urinary symptoms.
The physical signs of the two cases were also dissimilar; upon physical examination, one case had mild upper abdominal tenderness, and the other had right lower quadrant abdominal pain with distention and positive shifting dullness.
Crohn’s disease, bladder endometriosis, bladder and gastrointestinal neoplasm, and the disease caused by exposure to chemotherapy or irradiation.
The first patient had elevated hematological values for white blood cell count (15100/μL), eosinophils (52.5%), IgE (490 IU/mL), rheumatoid factor (20.2 IU/mL), and antistreptolysin O titer (124 IU/mL); while the second patient showed elevated hematological values for white blood cell count (14400/μL), eosinophils (21.1%), IgE (343 IU/mL), rheumatoid factor (20.7 IU/mL), and C-Reaction protein (12.9 mg/dL).
For both cases, the computed tomography and magnetic resonance scans showed multifocal segmental digestive tract wall thickening, luminal narrowing without obstruction, ascites, associated with local or diffuse bladder wall thickening, hypointense or isointense appearance on T2-weighted images, slightly restricted appearance on diffusion-weighted images, and progressive enhancement, with presence of the characteristic mucosal-lining.
For both cases, histological examination showed extensive eosinophilic infiltration of the subserosal digestive tract wall and the mucosa and submucosa of bladder wall.
Both patients were treated with oral prednisolone (1 mg/kg per day) for one week.
Eosinophilic gastroenteritis (EG) with associated eosinophilic cystitis (EC) is extremely rare and only a few radiological findings from EG and EC have been described. Both computed tomography (CT) and magnetic resonance imaging (MRI) images of EG associated with EC have not reported, especially the diffusion weighted imaging findings.
EG is a rare gastrointestinal disorder that selectively affects the gastrointestinal tract with eosinophil-rich inflammation. EC is also a rare clinicopathological condition characterized by transmural inflammation of the bladder, predominantly with eosinophils.
This case report presents the clinical, CT and MRI characteristics of EG with associated eosinophilic cystitis. The authors recommend that for multifocal segmental digestive tract wall thickening, associated with local or diffuse bladder wall thickening combined with allergic history, peripheral eosinophilia and a rapid relief of symptoms by steroid therapy in a patient, a diagnosis of EG associated with EC should be considered before endoscopic biopsies.
The authors have described two rare cases of EG associated with EC that showed rapid relief by steroid therapy. The article highlights the clinical characteristics of this tumor and provides insights into the CT and MRI findings.
P- Reviewer: Chu JP, Sharma S S- Editor: Yu J L- Editor: A E- Editor: Liu XM
| 1. | Gregg JA, Utz DC. Eosinophilic cystitis associated with eosinophilic gastroenteritis. Mayo Clin Proc. 1974;49:185-187. [PubMed] |
| 2. | Kim MS, Park H, Park CS, Lee EJ, Rho MH, Park NH, Joh J. Eosinophilic cystitis associated with eosinophilic enterocolitis: case reports and review of the literature. Br J Radiol. 2010;83:e122-e125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 3. | Peterson NE, Silverman A, Campbell JB. Eosinophilic cystitis and coexistent eosinophilic gastroenteritis in an infant. Pediatr Radiol. 1989;19:484-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 4. | Zhou HC, Lai C, Yang L. Eosinophilic gastroenteritis with involvement of the urinary bladder. Pediatr Radiol. 2014;44:1454-1457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 5. | Ingle SB, Hinge Ingle CR. Eosinophilic gastroenteritis: an unusual type of gastroenteritis. World J Gastroenterol. 2013;19:5061-5066. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 69] [Cited by in RCA: 84] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 7. | Palubinskas AJ. Eosinophilic cystitis. Case report of eosinophilic infiltration of the urinary bladder. Radiology. 1960;75:589-591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 43] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 8. | van den Ouden D. Diagnosis and management of eosinophilic cystitis: a pooled analysis of 135 cases. Eur Urol. 2000;37:386-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 75] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 9. | Hellstrom HR, Davis BK, Shonnard JW. Eosinophilic cystitis. A study of 16 cases. Am J Clin Pathol. 1979;72:777-784. [PubMed] |
| 10. | Khan S, Orenstein SR. Eosinophilic gastroenteritis. Gastroenterol Clin North Am. 2008;37:333-348, v. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 69] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 11. | Talley NJ, Shorter RG, Phillips SF, Zinsmeister AR. Eosinophilic gastroenteritis: a clinicopathological study of patients with disease of the mucosa, muscle layer, and subserosal tissues. Gut. 1990;31:54-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 506] [Cited by in RCA: 513] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 12. | Orenstein SR, Shalaby TM, Di Lorenzo C, Putnam PE, Sigurdsson L, Mousa H, Kocoshis SA. The spectrum of pediatric eosinophilic esophagitis beyond infancy: a clinical series of 30 children. Am J Gastroenterol. 2000;95:1422-1430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 338] [Cited by in RCA: 289] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 13. | Goralski JL, Lercher DM, Davis SD, Dellon ES. Eosinophilic esophagitis in cystic fibrosis: a case series and review of the literature. J Cyst Fibros. 2013;12:9-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 14. | Vitellas KM, Bennett WF, Bova JG, Johnson JC, Greenson JK, Caldwell JH. Radiographic manifestations of eosinophilic gastroenteritis. Abdom Imaging. 1995;20:406-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 42] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 15. | Teegavarapu PS, Sahai A, Chandra A, Dasgupta P, Khan MS. Eosinophilic cystitis and its management. Int J Clin Pract. 2005;59:356-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 16. | Famularo G, Prantera C, Nunnari J, Gasbarrone L. Eosinophilic gastroenteritis in a young man. CMAJ. 2011;183:E65-E67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 17. | Tamai K, Koyama T, Saida S, Nishikomori R, Togashi K. MR imaging findings of eosinophilic cystitis in an 8-year-old girl. Pediatr Radiol. 2007;37:836-839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 18. | Wong-You-Cheong JJ, Woodward PJ, Manning MA, Davis CJ. From the archives of the AFIP: Inflammatory and nonneoplastic bladder masses: radiologic-pathologic correlation. Radiographics. 2006;26:1847-1868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 102] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 19. | Wiesner W, Kocher T, Heim M, Bongartz G. CT findings in eosinophilic enterocolitis with predominantly serosal and muscular bowel wall infiltration. JBR-BTR. 2002;85:4-6. [PubMed] |
| 20. | Zheng X, Cheng J, Pan K, Yang K, Wang H, Wu E. Eosinophilic enteritis: CT features. Abdom Imaging. 2008;33:191-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 1.5] [Reference Citation Analysis (0)] |