Published online Nov 21, 2014. doi: 10.3748/wjg.v20.i43.16167
Revised: May 5, 2014
Accepted: June 21, 2014
Published online: November 21, 2014
Processing time: 262 Days and 3.8 Hours
Liver regeneration after major surgery may activate occult micrometastases and facilitate tumor growth, leading to liver tumor recurrence. Molecular changes during liver regeneration can provide a microenvironment that stimulates intrahepatic tumor propagation through alterations in cellular signaling pathways, where activation and proliferation of mature hepatocytes, hepatic progenitor cells, non-parenchymal liver cells might favor both liver regeneration and tumor growth. This review highlights recent advances of tumor growth and development in the regenerating liver, possible mechanisms and clinical implications.
Core tip: The liver has a unique capacity for regeneration after cellular damage or partial removal of tissue, and modern liver surgery relies on this property. Liver regeneration is a complex process involving a large array of growth factors, cytokines and cells to restore hepatic mass and function. Experimental and clinical data indicate that the regeneration signals facilitate the growth of both primary and secondary liver tumors and can alter their malignant potential. This is an important mechanism underlying recurrence after liver surgery. New therapeutic strategies founded on better insight into the relationship between liver regeneration and tumor biology are needed.
- Citation: Shi JH, Line PD. Effect of liver regeneration on malignant hepatic tumors. World J Gastroenterol 2014; 20(43): 16167-16177
- URL: https://www.wjgnet.com/1007-9327/full/v20/i43/16167.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i43.16167
Malignant liver tumors can be classified into primary cancers and secondary (metastatic) tumors. Hepatocellular carcinoma (HCC) is the most common primary malignancy of the liver, accounting for 70%-85% of the total liver cancer cases, is the leading cause of death amongst cirrhotic patients and the third leading cause of cancer mortality worldwide[1-4]. The prognosis of HCC without specific treatment is poor with median survival of early and advanced HCC being 6-9 mo and 1-2 mo, respectively[5]. In developed countries 30%-40% of HCC patients are now being diagnosed at earlier stages when curative treatments can be optimally applied[6-8]. Liver surgery, including liver resection and liver transplantation (LTx), are the primary curative options from an intention-to-treat perspective for HCC with an overall 5-year survival rate of 40%-70% after resection and a superior 5-year overall survival after liver transplantation above 70% in well-selected candidates[7-12]. Although liver surgery can achieve long-term control in some patients with early HCC, tumor recurrence rates are high at approximately 50% at 3-years observation after hepatic resection[13].
The majority of malignant liver tumors in western countries are metastatic tumors. Colorectal cancer (CRC) is the third most common cancer[1] and the fourth leading cause of cancer deaths worldwide[14,15]. javascript:newshowcontent (“active”, “references”); The liver is the most frequent and often unique site of metastasis (colorectal liver metastases - CLM) and are present at diagnosis of CRC in about 30% of patients (synchronous) and in about 40% of patients after aggressive surgery on the primary tumor (metachronous)[16,17]. Modern surgical techniques and chemotherapy can improve the five-year survival rate for selected patients with CLM, but intrahepatic recurrence after liver resection occurs in 40%-60% of patients, usually within 12-18 mo postoperatively[18,19].
Although some patients may benefit from more aggressive approaches when recurrence is still observed in the liver, this is generally associated with a poor outcome in most of cases owing to multi-centricity or inadequate functional liver reserve if a resection were to be attempted[20-23]. The high frequency of tumor recurrence following liver surgery is still a significant clinical problem impairing the long-term survival.
Accumulating clinical and experimental studies have suggested that liver regeneration following surgical resection may activate occult micrometastases, facilitate tumor growth and thereby contribute to tumor recurrence[16,24-27]. Similarly, an enhanced tumor growth rate has been reported following portal vein embolization when performed as a preparation to liver resection[28]. The association between the development of micrometastases, neoplastic progression and the factors involved in liver regeneration that particularly promote neoplastic progression are only partly understood. It is therefore necessary to investigate mechanisms of recurrent liver tumor in the regenerating liver, in order to find strategies for suppressing tumor recurrence after surgery.
The liver accounts for 30%-91% of recurrent HCC and 40% of CRC metastases after surgery[18,29-31]. The presence of satellite lesions, intrahepatic microvascular invasion and poor histological differentiation are markers for recurrence and a bad prognosis[11,32]. There are two distinct categories of recurrences: undetected micrometastasis due to multicentric tumor or “de novo” cancer arising as a result of an underlying carcinogenic liver disease in the liver remnant[11,30,31,33].
This distinction may have significant prognostic and therapeutic implications, but it can be difficult to make a differentiation between the two types in individual patients. In general, recurrence due to residual tumors missed at initial workup or incomplete resection margins usually occur earlier, well within the first 24 postoperative months, and the shorter the time frame from primary operation to diagnosis of relapse, the worse the prognosis[34]. Patients with multicentric primary liver cancer presenting with a late solitary recurrence and good liver function, can after thorough evaluation appear to be good candidates for re-resection, as opposed to patients with intrahepatic metastases, which are often multifocal[31]. Further studies based on a systematic analysis of both recurrent and primary tumors may help to clarify this issue[35,36].
Liver surgery, including liver resection and in selected cases LTx, as well as radiofrequency ablation (RFA), are the first-line options for early HCC and limited CLM. Liver resection may be performed when there is a sufficient functional reserve in the liver remnant, no extrahepatic metastases and no severe portal hypertension. Since 1996, orthotopic LTx[37] has been the preferred treatment for small HCC developed in cirrhosis as it cures both the tumor and the cirrhosis[38,39]. Living-donor transplantation is an alternative, which enables patients to avoid the long waiting time before LTx and is a more effective approach to reduce the dropout rate from the waiting list[40]. Despite that CLM is still considered an absolute contraindication for liver transplantation, recent single center experience have shown that patients with CLM can achieve long-term survivals of 95%, 68% and 60% at 1, 3 and 5 years respectively[41]. RFA has become the most frequently used form of ablative therapy[42]. The main advantage of RFA is the ability to treat a variable number of liver lesions, and to increase resectability rate by combining RFA with resection if several lesions throughout the parenchyma have to be removed simultaneously[43]. RFA achieves a local complete response rate of more than 80%[44] , improves the overall survival compared with other modalities such as chemotherapy or percutaneous ethanol injection[45,46], and has in selected series demonstrated a similar 5-year survival rate as a primary liver resection[47].
Liver regeneration is a reparative process following parenchymal damage or surgical resection of the liver[48]. The compensatory process of hepatic regeneration involves numerous cells and molecules (cytokines and growth factors) that mediate their role through multiple molecular pathways[48-52] (Table 1).
| Regeneration Phases | Molecular factors | Origin | Targets and Effects | Involved Signalling pathways |
| Priming stage | Nitric oxide, prostaglandins, tumor necrosis factor, interleukin 6 | NPLCs | Trigger of liver regeneration, sensitizing hepatocytes to growth factors for replication | NF-κB, JAK/STAT3 and MAPK signaling pathway |
| Proliferation stage | HGF | HSCs and other NPLCs, hepatocytes; mesenchymal cells | Cooperative effects allow the hepatocytes to overcome cell-cycle | HGF/cMet signaling pathway |
| TGF-α | Hepatocytes, HSCs | Checkpoint-controls and transition of hepatocytes from G0, through G1, to the S phase of the cell cycle, leading to DNA synthesis and cell proliferation | EGFR signaling pathway IGF-1R and insulin receptor | |
| EGF | Salivary glands in intestine and pancreas | |||
| IGF | Hepatocytes, NPLCs | |||
| PDGF | Hepatocytes | Mitosis in NPLCs, remodelling of ECM | PDGFR in HSCs | |
| VEGF | VEGF receptor in endothelial cells | |||
| Termination stage | TGF-β | HSCs, other NPLCs, mesenchymal cells | Inhibition of hepatocyte DNA synthesis, remodeling of ECM, restoration at the end of regeneration | TGF-β signaling pathway |
| Others | Wnt/β-catenin plays a supportive role in liver regeneration; metalloproteinase play a pivotal role in ECM degradation, generation and degradation of active growth factor and signaling molecules in the ECM | |||
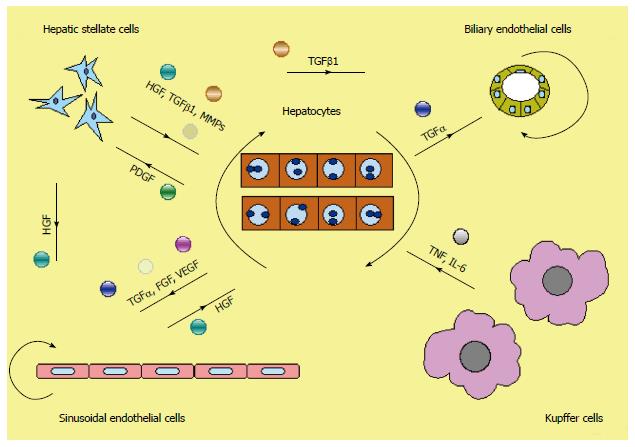
The progression of liver regeneration is segmented roughly in three phases: priming (G0 to G1 phase), proliferation (G1 to M phase) and termination. Liver regeneration progression is highly coordinated by the signal communication between hepatocytes and non-parenchymal cells, and is also influenced by endocrine factors, sympathetic intervention, and blood perfusion of the liver. The process of restoration of liver volume is initiated by the replication of various types of intrahepatic cells. Replication of hepatocytes generally starts within 1 d after major resection in both rodents and humans. This is predominantly due to that the hepatocytes go from the quiescent G0 phase to the G1 phase, undergo mitosis and continue during sufficient rounds of mitosis to restore the original mass of the liver trough the stimulation of growth factors and other mitogens. Nonparenchymal cells, such as sinusoidal endothelial cells, Kupffer cells, and biliary duct cells replicate in a delayed fashion[48,50,53,54], but demonstrate a similar synchronicity in DNA synthesis and mitosis as seen in hepatocytes[48]. Accompanying the cellular replication, there is also a breakdown and remodeling of the extracellular matrix (ECM), promoted by cell-ECM interactions, which is also of vital importance in regulating liver regeneration[55].
The original size and weight of the liver is re-established within one week after hepatectomy in rodents and about 3 mo in humans[48]. The parenchyma that have been damaged or removed does not regrow and the growth of the remnant liver or graft is a restoration of function by induced hyperplasia. Thereafter, the hepatocytes enter their non-replicative and quiescent, functional state. The lobes are slowly reorganized and hepatic histology is completely restored after 2-3 wk in rodents and 3-6 mo in humans[48,56]. Clinical data shows that liver regeneration is significantly impaired in damaged livers due to viral hepatitis[57] and this is true also for a variety of other chronic liver diseases.
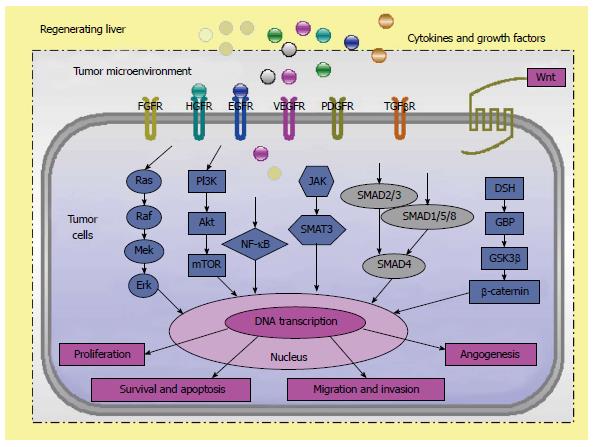
Development and progression of liver cancer and liver metastases is a multifaceted molecular process, which is linked to alterations in some prominent cellular signalling pathways, including RAS/RAF-MAPK pathway, phosphatidylinositol-3 kinase (PI3K)/AKT pathway, WNT/β-catenin pathway, insulin-like growth factor pathway (IGF), hepatocyte growth factor (HGF)/c-MET pathway and growth factor-regulated angiogenic signaling[16,19,58-61].
It has been demonstrated that the cellular and molecular changes following hepatectomy, including surgical stress responses, ischemia reperfusion injury can alter the kinetics of tumor growth and thereby contribute to recurrence[16,62-64]. The factors related to the surgical stress itself represent transient changes, whereas the process of liver regeneration can act as a stronger and more sustained stimulus that promotes the growth of occult tumors and neoplastic development. Liver regeneration after surgery provides a microenvironment with many growth factors and cytokines that are favorable for tumorgenesis and tumor propagation through cell activation, proliferation, migration and angiogenesis[16,19,24-27]. The majority of studies in HCC indicate that factors linked to tumor invasiveness and vascular involvement are the most important[20]. In addition, the mechanisms involving endocrine, autocrine and paracrine occurring during liver regeneration can influence dormant micrometastases and tumorgenesis in the remnant liver[16,50].
Major surgery results in a rapid influx of cytokines, such as tumour necrosis factor α (TNF-α) and interleukin 6 (IL-6), and angiogenic factors that alter the microenvironment of distant dormant tumor deposits. Growth factors such as HGF, epidermal growth factor, transforming growth factor alpha (TGF-α) and TGF-β, which play an essential role in liver regeneration, have been shown to be associated with increased tumor aggressiveness and metastasis[58-61,65] (Figure 1, Figure 2 and Table 2). Tumor growth requires the balance of growth factors and cytokines in the microenvironment to favor angiogenesis[66]. Angiogenic inhibitors, ECM proteins and ECM protein fragments are determining for maintaining the dormant state[67,68]. ECM breakdown and rebuilding during liver regeneration are required for tumor metastasis into within the liver, or through the blood and lymphatic circulation. Well documented factors common to liver ECM changes during liver regeneration include matrix metallopeptidases (MMPs), fibroblast growth factor (FGF), hypoxia-induced factor-1a, vascular endothelial growth factor (VEGF) and platelet-derived growth factor (PDGF)[50].
| Signaling pathways | Cell survival | Cell proliferation | Cell differentiation | Cell migration | Angiogenesis | |
| Receptor tyrosine kinase signaling pathways | HGF/c-MET | √ | √ | √ | √ | |
| EGF(TGF-α)/EGFR | √ | √ | ||||
| IGF/IGFR | √ | √ | √ | |||
| VEGF/VEGFR | √ | |||||
| PDGF/PDGFR | √ | |||||
| TGF-β1/SMAD | √ | √ | ||||
| Intercellular kinase signaling pathways | RAF/MEK/ERK | √ | √ | √ | √ | |
| PI3K/AKT/mTOR | √ | √ | √ | |||
| Wnt/β-catenin | √ | √ | √ | √ | ||
| Hedgehog | √ | √ | √ | |||
| NK-κB, JAK/STAT3 | √ | √ | √ | √ | √ | |
The epithelial cells from the primary or recurrent liver tumor, may in addition themselves undergo an epithelial to mesenchymal transition (EMT) and thereby increase their invasive potential as a response to various stimuli within the tumor microenvironment[69]. The growth factors that bind to tyrosine-kinase receptors, such as EGF, HGF, TGF-β, FGF and MMPs, can promote EMT in carcinoma cells[70]. These factors are upregulated by liver surgery and in ECM remodeling during liver regeneration and can thereby account for an increased growth and metastatic potential in the case of remnant tumor cells.
In the normal adult liver after injury or surgery, loss of liver parenchyma is repaired by replication of mature hepatocytes[71-73]. However, pre-existing liver diseases such as chronic HBV and HCV infection, alcoholic fatty liver disease, hemochromatosis and other conditions severely impair the ability of hepatocytes to replicate. This can actuate a second regenerative mechanism by activation of hepatic progenitor cells (HPCs)[48,74]. HPCs (also called oval cells in rodents) are quiescent, existing in low numbers around the periportal region (the canals of Hering) in a normal liver. Following severe and prolonged liver trauma, these cells are capable of proliferation and differentiation into both hepatocytes and cholangiocytes, and typically form the hepatoblast, comprising small ductules and strings of cholangiocytes, termed the ductular reaction[75]. Previous reports have shown that the presence of HPCs might contribute to liver regeneration following transplantation[76,77]. Activation of HPCs under diverse situations, like liver diseases or major liver surgery can be of significance to maintain and restore liver homeostasis after stress.
No single marker offers complete specificity for the identification of HPC[73]. Multiple combinations of different phenotypes and molecular markers have been used to differentiate between HPCs and other hepatic cells. The stem compartment of the liver is assumed to consist of cells with various phenotypes and multiple molecular markers, including markers of adult hepatocytes (albumin, cytokeratins 8 and 18), cholangiocytic markers (cytokeratin 7 and 19, OV-6 and Glutathione-S-transpeptidase), fetal hepatoblasts (αFP, delta-like protein), and hematopoietic stem cell (Thy -1, Sca-1, c-kit, CD34, CD133)[71,78].
Previous studies have shown that HPCs can be hematopoietic derived or liver-resident cells. Hematopoietic stem cells (HSCs) may be recruited from the bone marrow by chemo-attractants that cause them to migrate and infiltrate into the liver lobules via the canals of Hering. This effect seems to be related to the extent of resection and the presence of concomitant liver disease[79]. Liver resident stem cells may also be recruited and activated by hepatocellular damage. This initiates an immune response in the liver, which leads to the secretion of a complex mixture of cytokines and growth factors. TNF-α and IL-6 released by Kupffer cells stimulate proliferation of HPCs, while interferon (IFN) can prime HPCs to respond to mitogenic stimuli. Growth factors of HPC released by hepatic stellate cells include EGF, TGF-α, HGF and TGF-β. Thus, the cytokines released by Kupffer cells, hepatic stellate cells and HPCs themselves may act in concert to control HPC proliferation and remodeling of the liver parenchyma[71,80,81].
Both hematopoietic and hepatic derived HPCs require the essential inflammatory cytokines and regenerative growth factors to proliferate and transdifferentiate. These include TNF, IL-6, IFN-α, TGF-α, FGF, TGF-β and HGF[81,82], indicating that the activation and differentiation of HPCs is dependent on a delicate regulatory interplay between liver stress and regenerative capability and response. Several studies have reported that liver surgery and acute liver injury can lead to mobilization of progenitor cells, both after partial hepatectomy in general and in HCC patients[75,83-86]. The exact mechanism is still not fully clarified. Recently Langenberg et al[85] showed that major liver surgery could mobilize HSCs into the liver where they differentiated into HPCs. This effect could last up to 24 h in response to elevated levels of granulocyte colony-stimulating factor (G-CSF), indicating that G-CSF may have a significant role in the activation of progenitor cells of hematopoietic origin[86].
Activation of progenitor cells in liver regeneration might directly facilitate hepatic carcinogenesis[87-89]. This hypothesis has been tested and confirmed in many rodent and human experiments. In human chronic liver diseases, particularly chronic HBV or HCV infection with accompanying liver cirrhosis, proliferation of HPCs are directly related to disease severity, suggesting that activation of this cell compartment is associated with an increased risk of HCC development[90]. Hepatocellular cancers that derive from progenitor cells have been indicated by the fact that many human HCC tumors contain a mixture of mature hepatocytes and an intermediate phenotypes between HPCs and mature hepatocytes, similar to progenitor cells[87,91,92]. In addition, these tumors exhibit gene expression profiles identical to hepatoblasts derived from HPCs. HCC tumors that are derived from HPCs, display a significantly poorer prognosis and a higher recurrence rate after surgical resection and liver transplantation in retrospective reviews[87,91,92]. Furthermore, recruitment of progenitor cells has been shown to contribute to liver cancer formation in a preclinical model, and probably give rise to HCC as well as intrahepatic cholangiocarcinoma, supporting the view that progenitor cells in some cases have a critical role in the initiation and progression of liver cancers[48,88,89,93,94]. These findings might also support the hypothesis of genuine liver cancer stem cells that might be derived from HPCs[94-96].
The progenitor cells can also affect HCC progression through indirect contribution of growth factors. In experimental rodent models with intrahepatic tumor implantation and concomitant liver resection, the presence of tumor seems to stimulate liver regeneration in major resected animals (> 70%) compared with resected animals without tumor, and this could possibly partly be attributed to the activation of progenitor like cells[25,95]. Similarly, in vitro and in vivo models testing the influence of progenitor cells on rodent HCC cells and experimental tumors indicate that the progenitor cells causes a “stem-like” expression profile in the HCC cells making them more resistant to chemotherapy and creating tumors that grow faster with greater invasive and metastatic potential. These changes seems to be partly related to activation of the of MAPK, AKT, TGF-β1 and Wnt/β-catenin pathways[93].
Recurrence following surgical treatment for liver tumors is a common and significant problem. The best strategy to minimize recurrence due to occult tumors is achieved by measures aimed at improvement in preoperative radiologic imaging and staging. Vast improvements have been made the last decade in imaging methodology. Modern computed tomography (CT) and magnetic resonance imaging (MRI) with liver specific contrast media and diffusion-weighted analysis have greatly improved the sensitivity and specificity for detection of small malignant lesions[97]. The cirrhotic liver represents a particular challenge, since small tumors can be hard to distinguish from regeneration nodules. Contrast enhanced ultrasound has been shown to be a very useful method in this respect, yielding similar diagnostic utility as MRI and CT[98]. In difficult cases, a combination of the different methods is often needed. Ultrasound has the advantage that it can also be used intraoperatively to increase the probability of achieving a negative margin (R0) during resection. Some studies have advocated the use of indocyanine green fluorescence imaging and demonstrate that this furthers increases diagnostic accuracy[99].
A relapse of malignant disease in the liver is a bad prognostic sign for the patient, and the therapeutic options might, depending on anatomical location, liver function and general performance status is limited. All the treatment modalities in use for primary treatment of liver tumors, i.e., surgical resection, liver transplantation, local ablation, and transcatheter arterial chemoembolization (TACE) http://en.wikipedia.org/wiki/Transcatheter_arterial_chemoembolization have been tried in setting of recurrent disease[20,100-102]. Repeat hepatectomy appears to be the best treatment with a reported resectability rate ranging from 10% to 77%[29]. Patients treated by repeat hepatectomy have better survival rates than those treated by other palliative methods, but selection bias and lack of randomized trials makes it difficult to ascertain the magnitude of this improvement on an intention to treat basis[29,103-105]. A longer interval from hepatectomy to recurrence is associated with improved survival[101,103]. TACE appears to be the best option in recurrent HCC tumors that are unresectable due to multimodularity or inadequate functional liver reserve[20]. TACE might in a few cases downstage the tumor size making it resectable[106]. In general, multimodal therapy for recurrent liver tumors (TACE, local ablation and re-resection) can result in a prolonged 5-year survival rate of up to 20%[20-23].
New techniques have been suggested to target malignant liver tumors without retarding the capability of liver regeneration.
The most important new trends of therapy against liver tumor are advances in molecular targeted therapy and employment of immunotherapy. Improvements in the understanding of the molecular basis of cancer have led to the development of targeted agents tailored to inhibit specific protein kinases involved in intracellular signal transduction pathways, which drive tumor progression and recurrence, including RAS/RAF-MAPK pathway, PI3K/AKT pathway, WNT/β-catenin pathway, IGF, HGF/c-MET pathway and growth factor-regulated angiogenic signaling. Sorafenib was firstly approved by the Food and Drug Administration in 2007 for the treatment of advanced HCC based on the demonstrated improved overall survival benefits, and several targeted agents have demonstrated early evidence suggesting significant antitumor activity in phase I/II trials. However, few studies have looked at the effects of these new molecular agents on both liver regeneration and regression of liver tumors. Kinoshita et al[107] reported that angiogenesis inhibitor TNP-470 prevented metastases of VX2 carcinoma without retarding liver regeneration after hepatectomy. Despite these results, the angiogenesis mechanism that they discussed is controversial[108]. Liver regeneration is a complex and partly angiogenesis-dependent process. Hepatocellular production of VEGF leads to maximal levels 48-72 h after partial hepatectomy, and this can increase the dilatation and permeability of blood vessels. Inhibition of angiogenesis by angiostatin impairs both angiogenesis and liver regeneration[109-111]. Another approach to inhibit tumor proliferation and angiogenesis has been suggested through blockade of the renin-angiotensin system (RAS)[112]. Interestingly, intervention on RAS utilizing angiotensin converting enzyme inhibitors and angiotensin II type 1 blockers improves liver regeneration and inhibits tumor growth in animal models[113]. To our knowledge, no human trials are available testing the validity of these experimental observations in clinical practice.
In order to combine the diverging need for both liver regeneration and an anti tumor cell microenviroment in the liver remnant following surgery, more efforts are needed comparing gene and protein expressions in normal and regenerating hepatocytes and malignant cells by microarray and proteomic technology in order to identify signaling pathways that are unique to the hepatoma cells. This is probably critical in order to identify novel targets and to better understand the biology of the cancer cell population that escapes the current treatment strategies and thereby cause recurrences.
Immunotherapy is an evolving and promising treatment option for liver tumors, which might have the potential to lower recurrence rates and improve overall survival after surgery for liver tumors. In general, immunotherapy is based on the premise that tumor-related self antigens (e.g., AFP) can be selectively targeted, thereby eliciting a tumor-specific response. Moreover, new insights into immunogenic cancer cell death indicate that certain types of chemotherapy and radiotherapy can be associated with immune responses that are of clinical significance[114,115]. New techniques are being investigated in human subjects and animal models, including the adoptive transfer of viral antigen-specific T cells, cytokines injections, autologous tumor pulsed dendritic cells, the use of AFP- derived peptides and adenoviruses, and immunization with tumor cells fused with antigen-presenting cells[116-118]. The mechanisms that enable liver cancer to escape attack by the immune system still remain unclear. Further knowledge into how immunogenic cell death of cancer cells might be evoked can aid in the development of novel therapeutic principles.
Liver-directed therapy for primary and metastatic disease could be promising for small-sized tumor or “oligometastatic” tumors. Low-power laser irradiation (LPLI) is suggested to be an effective minimally invasive palliative treatment option for patients with small liver tumors[119,120]. Interestingly in this context was that LPLI was shown to improve liver regenerative capacity of a small liver remnant after hepatectomy through activation of the HGF-MAPK/PI3K pathway[121].
Surgical therapy for liver tumors implies and often is dependent on a postoperative liver regeneration processes. Regeneration following surgery provides a microenvironment with many growth factors and cytokines that are necessary for proliferation of mature hepatocytes but concomitantly favorable for liver tumor progression. Progenitor cells can under certain conditions be activated after major surgery, and may have a stimulating and nourishing effect on liver tumors. A better understanding of the relationship between liver regeneration and liver tumor propagation is of significance for the clinical management of patient presenting with primary and secondary liver tumors and will provide a basis for novel therapeutic strategies in order to minimize cancer recurrence after liver surgery.
P- Reviewer: Song GB, Yang H S- Editor: Qi Y L- Editor: A E- Editor: Liu XM
| 1. | Parkin DM, Bray F, Ferlay J, Pisani P. Estimating the world cancer burden: Globocan 2000. Int J Cancer. 2001;94:153-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2651] [Cited by in RCA: 2599] [Article Influence: 108.3] [Reference Citation Analysis (0)] |
| 2. | Edge S, Byrd D, Compton C, Fritz A, Greene F, Trotti III A. AJCC Cancer Staging Manual, 7th ed. American Joint Committee on Cancer. New York: Springer 2009; . |
| 3. | Gospodarowicz M, Wittekind C, Sobin L. TNM Classification of Malignant Tumours. Publisher: Wiley-Blackwell 2009; . |
| 4. | Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23762] [Cited by in RCA: 25541] [Article Influence: 1824.4] [Reference Citation Analysis (7)] |
| 5. | Bosch FX, Ribes J, Díaz M, Cléries R. Primary liver cancer: worldwide incidence and trends. Gastroenterology. 2004;127:S5-S16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1799] [Cited by in RCA: 1816] [Article Influence: 86.5] [Reference Citation Analysis (0)] |
| 6. | Bruix J, Llovet JM. Prognostic prediction and treatment strategy in hepatocellular carcinoma. Hepatology. 2002;35:519-524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 837] [Cited by in RCA: 851] [Article Influence: 37.0] [Reference Citation Analysis (0)] |
| 7. | Wayne JD, Lauwers GY, Ikai I, Doherty DA, Belghiti J, Yamaoka Y, Regimbeau JM, Nagorney DM, Do KA, Ellis LM. Preoperative predictors of survival after resection of small hepatocellular carcinomas. Ann Surg. 2002;235:722-730; discussion 730-731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 141] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 8. | Takayama T, Makuuchi M, Hirohashi S, Sakamoto M, Yamamoto J, Shimada K, Kosuge T, Okada S, Takayasu K, Yamasaki S. Early hepatocellular carcinoma as an entity with a high rate of surgical cure. Hepatology. 1998;28:1241-1246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 310] [Cited by in RCA: 292] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 10. | Mazzaferro V, Llovet JM, Miceli R, Bhoori S, Schiavo M, Mariani L, Camerini T, Roayaie S, Schwartz ME, Grazi GL. Predicting survival after liver transplantation in patients with hepatocellular carcinoma beyond the Milan criteria: a retrospective, exploratory analysis. Lancet Oncol. 2009;10:35-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1267] [Cited by in RCA: 1572] [Article Influence: 92.5] [Reference Citation Analysis (1)] |
| 11. | Llovet JM, Fuster J, Bruix J. Intention-to-treat analysis of surgical treatment for early hepatocellular carcinoma: resection versus transplantation. Hepatology. 1999;30:1434-1440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1331] [Cited by in RCA: 1271] [Article Influence: 48.9] [Reference Citation Analysis (0)] |
| 12. | Fong Y, Sun RL, Jarnagin W, Blumgart LH. An analysis of 412 cases of hepatocellular carcinoma at a Western center. Ann Surg. 1999;229:790-799; discussion 799-800. [PubMed] |
| 13. | Mulcahy MF. Management of hepatocellular cancer. Curr Treat Options Oncol. 2005;6:423-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 14. | Seitz K. Contrast-enhanced ultrasound in the diagnosis of hepatocellular carcinoma and liver metastases. Ultraschall Med. 2005;26:267-269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 15. | Yan FH. [Imaging diagnosis of hepatocellular carcinoma]. Zhonghua Yixue Zazhi. 2005;85:297-299. [PubMed] |
| 16. | de Jong KP, Slooff MJ, de Vries EG, Brouwers MA, Terpstra OT. Effect of partial liver resection on tumour growth. J Hepatol. 1996;25:109-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 17. | Lee WJ. [Imaging diagnosis of hepatocellular carcinoma]. Korean J Gastroenterol. 2005;45:234-246. [PubMed] |
| 18. | Murakami T, Takamura M, Kim T, Hori M, Federle MP, Onishi H, Tomoda K, Nakamura H. Double phase CT during hepatic arteriography for diagnosis of hepatocellular carcinoma. Eur J Radiol. 2005;54:246-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 19. | Paschos KA, Bird NC. Liver regeneration and its impact on post-hepatectomy metastatic tumour recurrence. Anticancer Res. 2010;30:2161-2170. [PubMed] |
| 20. | Marín-Hargreaves G, Azoulay D, Bismuth H. Hepatocellular carcinoma: surgical indications and results. Crit Rev Oncol Hematol. 2003;47:13-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 58] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 21. | Ikeda K, Saitoh S, Tsubota A, Arase Y, Chayama K, Kumada H, Watanabe G, Tsurumaru M. Risk factors for tumor recurrence and prognosis after curative resection of hepatocellular carcinoma. Cancer. 1993;71:19-25. [PubMed] |
| 22. | Poon RT, Fan ST, O’Suilleabhain CB, Wong J. Aggressive management of patients with extrahepatic and intrahepatic recurrences of hepatocellular carcinoma by combined resection and locoregional therapy. J Am Coll Surg. 2002;195:311-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 90] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 23. | Minagawa M, Makuuchi M, Takayama T, Kokudo N. Selection criteria for repeat hepatectomy in patients with recurrent hepatocellular carcinoma. Ann Surg. 2003;238:703-710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 327] [Cited by in RCA: 357] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 24. | Heinrich S, Jochum W, Graf R, Clavien PA. Portal vein ligation and partial hepatectomy differentially influence growth of intrahepatic metastasis and liver regeneration in mice. J Hepatol. 2006;45:35-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 58] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 25. | Shi JH, Huitfeldt HS, Suo ZH, Line PD. Growth of hepatocellular carcinoma in the regenerating liver. Liver Transpl. 2011;17:866-874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 49] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 26. | Yoon SS, Kim SH, Gonen M, Heffernan NM, Detwiller KY, Jarnagin WR, D’Angelica M, Blumgart LH, Tanabe KK, Dematteo RP. Profile of plasma angiogenic factors before and after hepatectomy for colorectal cancer liver metastases. Ann Surg Oncol. 2006;13:353-362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 50] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 27. | Harun N, Nikfarjam M, Muralidharan V, Christophi C. Liver regeneration stimulates tumor metastases. J Surg Res. 2007;138:284-290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 58] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 28. | Hoekstra LT, van Lienden KP, Verheij J, van der Loos CM, Heger M, van Gulik TM. Enhanced tumor growth after portal vein embolization in a rabbit tumor model. J Surg Res. 2013;180:89-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 29. | Neeleman N, Andersson R. Repeated liver resection for recurrent liver cancer. Br J Surg. 1996;83:893-901. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 65] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 30. | Shimada M, Matsumata T, Taketomi A, Yamamoto K, Itasaka H, Sugimachi K. Repeat hepatectomy for recurrent hepatocellular carcinoma. Surgery. 1994;115:703-706. [PubMed] |
| 31. | Poon RT, Fan ST, Lo CM, Liu CL, Wong J. Intrahepatic recurrence after curative resection of hepatocellular carcinoma: long-term results of treatment and prognostic factors. Ann Surg. 1999;229:216-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 437] [Cited by in RCA: 462] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 32. | Nagasue N, Uchida M, Makino Y, Takemoto Y, Yamanoi A, Hayashi T, Chang YC, Kohno H, Nakamura T, Yukaya H. Incidence and factors associated with intrahepatic recurrence following resection of hepatocellular carcinoma. Gastroenterology. 1993;105:488-494. [PubMed] |
| 33. | Bismuth H, Samuel D, Engerran L. Treatment of unresectable hepatocellular carcinoma. N Engl J Med. 1995;333:878. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 34. | Del Gaudio M, Ercolani G, Ravaioli M, Cescon M, Lauro A, Vivarelli M, Zanello M, Cucchetti A, Vetrone G, Tuci F. Liver transplantation for recurrent hepatocellular carcinoma on cirrhosis after liver resection: University of Bologna experience. Am J Transplant. 2008;8:1177-1185. [RCA] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 125] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 35. | Chen PJ, Chen DS, Lai MY, Chang MH, Huang GT, Yang PM, Sheu JC, Lee SC, Hsu HC, Sung JL. Clonal origin of recurrent hepatocellular carcinomas. Gastroenterology. 1989;96:527-529. [PubMed] |
| 36. | Yamamoto T, Kajino K, Kudo M, Sasaki Y, Arakawa Y, Hino O. Determination of the clonal origin of multiple human hepatocellular carcinomas by cloning and polymerase chain reaction of the integrated hepatitis B virus DNA. Hepatology. 1999;29:1446-1452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 63] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 37. | Moolten FL, Bucher NL. Regeneration of rat liver: transfer of humoral agent by cross circulation. Science. 1967;158:272-274. [PubMed] |
| 38. | Mazzaferro V, Regalia E, Doci R, Andreola S, Pulvirenti A, Bozzetti F, Montalto F, Ammatuna M, Morabito A, Gennari L. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med. 1996;334:693-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5110] [Cited by in RCA: 5311] [Article Influence: 183.1] [Reference Citation Analysis (0)] |
| 39. | Wanless IR. Liver biopsy in the diagnosis of hepatocellular carcinoma. Clin Liver Dis. 2005;9:281-285, vii. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 40. | Kassahun WT, Fangmann J, Harms J, Hauss J, Bartels M. Liver resection and transplantation in the management of hepatocellular carcinoma: a review. Exp Clin Transplant. 2006;4:549-558. [PubMed] |
| 41. | Hagness M, Foss A, Line PD, Scholz T, Jørgensen PF, Fosby B, Boberg KM, Mathisen O, Gladhaug IP, Egge TS. Liver transplantation for nonresectable liver metastases from colorectal cancer. Ann Surg. 2013;257:800-806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 284] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 42. | Frezza EE. Therapeutic management algorithm in cirrhotic and noncirrhotic patients in primary or secondary liver masses. Dig Dis Sci. 2004;49:866-871. [PubMed] |
| 43. | Rhim H, Dodd GD. Radiofrequency thermal ablation of liver tumors. J Clin Ultrasound. 1999;27:221-229. [PubMed] |
| 44. | Scudamore CH, Lee SI, Patterson EJ, Buczkowski AK, July LV, Chung SW, Buckley AR, Ho SG, Owen DA. Radiofrequency ablation followed by resection of malignant liver tumors. Am J Surg. 1999;177:411-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 104] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 45. | Hoshida Y, Villanueva A, Kobayashi M, Peix J, Chiang DY, Camargo A, Gupta S, Moore J, Wrobel MJ, Lerner J. Gene expression in fixed tissues and outcome in hepatocellular carcinoma. N Engl J Med. 2008;359:1995-2004. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1044] [Cited by in RCA: 1006] [Article Influence: 59.2] [Reference Citation Analysis (0)] |
| 46. | Bruix J, Sherman M. Management of hepatocellular carcinoma. Hepatology. 2005;42:1208-1236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4333] [Cited by in RCA: 4507] [Article Influence: 225.4] [Reference Citation Analysis (0)] |
| 47. | Ellis LM, Chase J, Patt Y, Curley SA. Hepatic arterial infusion chemotherapy for colorectal cancer metastasis to the liver. Liver Cancer New York, NY: Springer 1998; 150-172. |
| 48. | Michalopoulos GK. Liver regeneration. J Cell Physiol. 2007;213:286-300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1246] [Cited by in RCA: 1158] [Article Influence: 64.3] [Reference Citation Analysis (0)] |
| 49. | Taub R. Liver regeneration: from myth to mechanism. Nat Rev Mol Cell Biol. 2004;5:836-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1091] [Cited by in RCA: 1175] [Article Influence: 56.0] [Reference Citation Analysis (0)] |
| 50. | Christophi C, Harun N, Fifis T. Liver regeneration and tumor stimulation--a review of cytokine and angiogenic factors. J Gastrointest Surg. 2008;12:966-980. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 35] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 51. | Jia C. Advances in the regulation of liver regeneration. Expert Rev Gastroenterol Hepatol. 2011;5:105-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 65] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 52. | Schoen Smith JM, Lautt WW. Nitric oxide and prostaglandins potentiate the liver regeneration cascade. Can J Gastroenterol. 2006;20:329-334. [PubMed] |
| 53. | Kountouras J, Boura P, Lygidakis NJ. Liver regeneration after hepatectomy. Hepatogastroenterology. 2001;48:556-562. [PubMed] |
| 54. | Mangnall D, Bird NC, Majeed AW. The molecular physiology of liver regeneration following partial hepatectomy. Liver Int. 2003;23:124-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 109] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 55. | Kim TH, Mars WM, Stolz DB, Petersen BE, Michalopoulos GK. Extracellular matrix remodeling at the early stages of liver regeneration in the rat. Hepatology. 1997;26:896-904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 140] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 56. | Wagenaar GT, Chamuleau RA, Pool CW, de Haan JG, Maas MA, Korfage HA, Lamers WH. Distribution and activity of glutamine synthase and carbamoylphosphate synthase upon enlargement of the liver lobule by repeated partial hepatectomies. J Hepatol. 1993;17:397-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 57. | Umeda Y, Iwagaki H, Ozaki M, Ogino T, Iwamoto T, Yoshida R, Shinoura S, Matsuda H, Sadamori H, Tanaka N. Refractory response to growth factors impairs liver regeneration after hepatectomy in patients with viral hepatitis. Hepatogastroenterology. 2009;56:971-977. [PubMed] |
| 58. | Cornellà H, Alsinet C, Villanueva A. Molecular pathogenesis of hepatocellular carcinoma. Alcohol Clin Exp Res. 2011;35:821-825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 59. | Kudo M. Hepatocellular carcinoma in 2011 and beyond: from the pathogenesis to molecular targeted therapy. Oncology. 2011;81 Suppl 1:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 60. | Whittaker S, Marais R, Zhu AX. The role of signaling pathways in the development and treatment of hepatocellular carcinoma. Oncogene. 2010;29:4989-5005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 608] [Cited by in RCA: 680] [Article Influence: 45.3] [Reference Citation Analysis (0)] |
| 61. | Nalesnik MA, Michalopoulos GK. Growth factor pathways in development and progression of hepatocellular carcinoma. Front Biosci (Schol Ed). 2012;4:1487-1515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 62. | Tsuchiya Y, Sawada S, Yoshioka I, Ohashi Y, Matsuo M, Harimaya Y, Tsukada K, Saiki I. Increased surgical stress promotes tumor metastasis. Surgery. 2003;133:547-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 151] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 63. | Man K, Ng KT, Lo CM, Ho JW, Sun BS, Sun CK, Lee TK, Poon RT, Fan ST. Ischemia-reperfusion of small liver remnant promotes liver tumor growth and metastases--activation of cell invasion and migration pathways. Liver Transpl. 2007;13:1669-1677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 110] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 64. | Man K, Lo CM, Xiao JW, Ng KT, Sun BS, Ng IO, Cheng Q, Sun CK, Fan ST. The significance of acute phase small-for-size graft injury on tumor growth and invasiveness after liver transplantation. Ann Surg. 2008;247:1049-1057. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 70] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 65. | He G, Karin M. NF-κB and STAT3 - key players in liver inflammation and cancer. Cell Res. 2011;21:159-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 879] [Cited by in RCA: 951] [Article Influence: 67.9] [Reference Citation Analysis (0)] |
| 66. | Hanahan D, Folkman J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell. 1996;86:353-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4848] [Cited by in RCA: 4796] [Article Influence: 165.4] [Reference Citation Analysis (0)] |
| 67. | Weinstat-Saslow DL, Zabrenetzky VS, VanHoutte K, Frazier WA, Roberts DD, Steeg PS. Transfection of thrombospondin 1 complementary DNA into a human breast carcinoma cell line reduces primary tumor growth, metastatic potential, and angiogenesis. Cancer Res. 1994;54:6504-6511. [PubMed] |
| 68. | O’Reilly MS, Boehm T, Shing Y, Fukai N, Vasios G, Lane WS, Flynn E, Birkhead JR, Olsen BR, Folkman J. Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell. 1997;88:277-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3249] [Cited by in RCA: 3111] [Article Influence: 111.1] [Reference Citation Analysis (0)] |
| 69. | Thiery JP. Epithelial-mesenchymal transitions in development and pathologies. Curr Opin Cell Biol. 2003;15:740-746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1279] [Cited by in RCA: 1322] [Article Influence: 60.1] [Reference Citation Analysis (0)] |
| 70. | Kalluri R, Zeisberg M. Fibroblasts in cancer. Nat Rev Cancer. 2006;6:392-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3347] [Cited by in RCA: 3529] [Article Influence: 185.7] [Reference Citation Analysis (1)] |
| 71. | Bird TG, Lorenzini S, Forbes SJ. Activation of stem cells in hepatic diseases. Cell Tissue Res. 2008;331:283-300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 124] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 72. | Santoni-Rugiu E, Jelnes P, Thorgeirsson SS, Bisgaard HC. Progenitor cells in liver regeneration: molecular responses controlling their activation and expansion. APMIS. 2005;113:876-902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 81] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 73. | Fausto N, Campbell JS. The role of hepatocytes and oval cells in liver regeneration and repopulation. Mech Dev. 2003;120:117-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 504] [Cited by in RCA: 491] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 74. | Fausto N, Campbell JS, Riehle KJ. Liver regeneration. Hepatology. 2006;43:S45-S53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1126] [Cited by in RCA: 1205] [Article Influence: 63.4] [Reference Citation Analysis (0)] |
| 75. | Gaudio E, Carpino G, Cardinale V, Franchitto A, Onori P, Alvaro D. New insights into liver stem cells. Dig Liver Dis. 2009;41:455-462. [PubMed] |
| 76. | Conzelmann LO, Hines IN, Kremer M, Perry AW, Lemasters JJ, Wheeler MD. Extrahepatic cells contribute to the progenitor/stem cell response following reduced-size liver transplantation in mice. Exp Biol Med (Maywood). 2007;232:571-580. [PubMed] |
| 77. | Sun Z, Zhang X, Locke JE, Zheng Q, Tachibana S, Diehl AM, Williams GM. Recruitment of host progenitor cells in rat liver transplants. Hepatology. 2009;49:587-597. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 78. | Cantz T, Manns MP, Ott M. Stem cells in liver regeneration and therapy. Cell Tissue Res. 2008;331:271-282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 76] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 79. | Zocco MA, Piscaglia AC, Giuliante F, Arena V, Novi M, Rinninella E, Tortora A, Rumi C, Nuzzo G, Vecchio FM. CD133+ stem cell mobilization after partial hepatectomy depends on resection extent and underlying disease. Dig Liver Dis. 2011;43:147-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 80. | Oertel M, Shafritz DA. Stem cells, cell transplantation and liver repopulation. Biochim Biophys Acta. 2008;1782:61-74. [PubMed] |
| 81. | Lowes KN, Croager EJ, Olynyk JK, Abraham LJ, Yeoh GC. Oval cell-mediated liver regeneration: Role of cytokines and growth factors. J Gastroenterol Hepatol. 2003;18:4-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 119] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 82. | Sigal SH, Brill S, Fiorino AS, Reid LM. The liver as a stem cell and lineage system. Am J Physiol. 1992;263:G139-G148. [PubMed] |
| 83. | Gehling UM, Willems M, Dandri M, Petersen J, Berna M, Thill M, Wulf T, Müller L, Pollok JM, Schlagner K. Partial hepatectomy induces mobilization of a unique population of haematopoietic progenitor cells in human healthy liver donors. J Hepatol. 2005;43:845-853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 62] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 84. | Lemoli RM, Catani L, Talarico S, Loggi E, Gramenzi A, Baccarani U, Fogli M, Grazi GL, Aluigi M, Marzocchi G. Mobilization of bone marrow-derived hematopoietic and endothelial stem cells after orthotopic liver transplantation and liver resection. Stem Cells. 2006;24:2817-2825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 67] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 85. | Langenberg MH, Nijkamp MW, Roodhart JM, Snoeren N, Tang T, Shaked Y, van Hillegersberg R, Witteveen PO, Vermaat JS, Kranenburg O. Liver surgery induces an immediate mobilization of progenitor cells in liver cancer patients: A potential role for G-CSF. Cancer Biol Ther. 2010;9:743-748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 86. | De Silvestro G, Vicarioto M, Donadel C, Menegazzo M, Marson P, Corsini A. Mobilization of peripheral blood hematopoietic stem cells following liver resection surgery. Hepatogastroenterology. 2004;51:805-810. [PubMed] |
| 87. | Roskams T. Liver stem cells and their implication in hepatocellular and cholangiocarcinoma. Oncogene. 2006;25:3818-3822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 318] [Cited by in RCA: 313] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 88. | Fang CH, Gong JQ, Zhang W. Function of oval cells in hepatocellular carcinoma in rats. World J Gastroenterol. 2004;10:2482-2487. [PubMed] |
| 89. | Dumble ML, Croager EJ, Yeoh GC, Quail EA. Generation and characterization of p53 null transformed hepatic progenitor cells: oval cells give rise to hepatocellular carcinoma. Carcinogenesis. 2002;23:435-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 172] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 90. | Lowes KN, Brennan BA, Yeoh GC, Olynyk JK. Oval cell numbers in human chronic liver diseases are directly related to disease severity. Am J Pathol. 1999;154:537-541. [PubMed] |
| 91. | Durnez A, Verslype C, Nevens F, Fevery J, Aerts R, Pirenne J, Lesaffre E, Libbrecht L, Desmet V, Roskams T. The clinicopathological and prognostic relevance of cytokeratin 7 and 19 expression in hepatocellular carcinoma. A possible progenitor cell origin. Histopathology. 2006;49:138-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 289] [Cited by in RCA: 303] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 92. | Lee JS, Heo J, Libbrecht L, Chu IS, Kaposi-Novak P, Calvisi DF, Mikaelyan A, Roberts LR, Demetris AJ, Sun Z. A novel prognostic subtype of human hepatocellular carcinoma derived from hepatic progenitor cells. Nat Med. 2006;12:410-416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 729] [Cited by in RCA: 748] [Article Influence: 39.4] [Reference Citation Analysis (0)] |
| 93. | Theise ND, Yao JL, Harada K, Hytiroglou P, Portmann B, Thung SN, Tsui W, Ohta H, Nakanuma Y. Hepatic ‘stem cell’ malignancies in adults: four cases. Histopathology. 2003;43:263-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 154] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 94. | Yao Z, Mishra L. Cancer stem cells and hepatocellular carcinoma. Cancer Biol Ther. 2009;8:1691-1698. [PubMed] |
| 95. | Wang B, Jacob ST. Role of cancer stem cells in hepatocarcinogenesis. Genome Med. 2011;3:11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 96. | Yamashita T, Wang XW. Cancer stem cells in the development of liver cancer. J Clin Invest. 2013;123:1911-1918. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 350] [Cited by in RCA: 415] [Article Influence: 34.6] [Reference Citation Analysis (0)] |
| 97. | Murakami T, Okada M, Hyodo T. CT versus MR imaging of hepatocellular carcinoma: toward improved treatment decisions. Magnetic resonance in medical sciences. J Jpn Soci Magne Reson Med. 2011;11:75-81. [RCA] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 98. | Guang Y, Xie L, Ding H, Cai A, Huang Y. Diagnosis value of focal liver lesions with SonoVue®-enhanced ultrasound compared with contrast-enhanced computed tomography and contrast-enhanced MRI: A meta-analysis. J Cancer Res Clin Oncol. 2011;137:1595-1605. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 54] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 99. | Peloso A, Franchi E, Canepa MC, Barbieri L, Briani L, Ferrario J, Bianco C, Quaretti P, Brugnatelli S, Dionigi P. Combined use of intraoperative ultrasound and indocyanine green fluorescence imaging to detect liver metastases from colorectal cancer. HPB. 2013;15:928-934. [RCA] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 86] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 100. | Rampone B, Schiavone B, Martino A, Viviano C, Confuorto G. Current management strategy of hepatocellular carcinoma. World J Gastroenterol. 2009;15:3210-3216. [PubMed] |
| 101. | Lee PH, Lin WJ, Tsang YM, Hu RH, Sheu JC, Lai MY, Hsu HC, May W, Lee CS. Clinical management of recurrent hepatocellular carcinoma. Ann Surg. 1995;222:670-676. [PubMed] |
| 102. | Hargreaves GM, Adam R, Bismuth H. Results after nonsurgical local treatment of primary liver malignancies. Langenbecks Arch Surg. 2000;385:185-193. [PubMed] |
| 103. | Farges O, Regimbeau JM, Belghiti J. Aggressive management of recurrence following surgical resection of hepatocellular carcinoma. Hepatogastroenterology. 1998;45 Suppl 3:1275-1280. [PubMed] |
| 104. | Hu RH, Lee PH, Yu SC, Dai HC, Sheu JC, Lai MY, Hsu HC, Chen DS. Surgical resection for recurrent hepatocellular carcinoma: prognosis and analysis of risk factors. Surgery. 1996;120:23-29. [PubMed] |
| 105. | Shuto T, Kinoshita H, Hirohashi K, Kubo S, Tanaka H, Tsukamoto T, Okuda T. Indications for, and effectiveness of, a second hepatic resection for recurrent hepatocellular carcinoma. Hepatogastroenterology. 1996;43:932-937. [PubMed] |
| 106. | Majno PE, Adam R, Bismuth H, Castaing D, Ariche A, Krissat J, Perrin H, Azoulay D. Influence of preoperative transarterial lipiodol chemoembolization on resection and transplantation for hepatocellular carcinoma in patients with cirrhosis. Ann Surg. 1997;226:688-701; discussion 701-703. [PubMed] |
| 107. | Kinoshita S, Hirai R, Yamano T, Yuasa I, Tsukuda K, Shimizu N. Angiogenesis inhibitor TNP-470 can suppress hepatocellular carcinoma growth without retarding liver regeneration after partial hepatectomy. Surg Tod. 2004;34:40-46. |
| 108. | Tanaka H, Taniguchi H, Mugitani T, Koishi Y, Masuyama M, Koyama H, Hoshima M, Takahashi T. Angiogenesis inhibitor TNP-470 prevents implanted liver metastases after partial hepatectomy in an experimental model without impairing wound healing. British J Surg. 1996;83:1444-1447. |
| 109. | Drixler TA, Vogten MJ, Ritchie ED, van Vroonhoven TJ, Gebbink MF, Voest EE, Borel Rinkes IH. Liver regeneration is an angiogenesis- associated phenomenon. Ann Surg. 2002;236:703-711; discussion 711-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 110. | Ronco MT, Francés D, de Luján Alvarez M, Quiroga A, Monti J, Parody JP, Pisani G, Carrillo MC, Carnovale CE. Vascular endothelial growth factor and nitric oxide in rat liver regeneration. Life Sci. 2007;81:750-755. [PubMed] |
| 111. | Taniguchi E, Sakisaka S, Matsuo K, Tanikawa K, Sata M. Expression and role of vascular endothelial growth factor in liver regeneration after partial hepatectomy in rats. J Histochem Cytochem. 2001;49:121-130. [PubMed] |
| 112. | Ager EI, Neo J, Christophi C. The renin-angiotensin system and malignancy. Carcinogenesis. 2008;29:1675-1684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 234] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 113. | Koh SL, Ager EI, Christophi C. Liver regeneration and tumour stimulation: implications of the renin–angiotensin system. Liver Int. 2010;30:1414-1426. |
| 114. | Kroemer G, Galluzzi L, Zitvogel L. Immunological effects of chemotherapy in spontaneous breast cancers. Oncoimmunology. 2013;2:e27158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 115. | Kono K, Mimura K, Kiessling R. Immunogenic tumor cell death induced by chemoradiotherapy: molecular mechanisms and a clinical translation. Cell Death Dis. 2013;4:e688. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 77] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 116. | Schmidt N, Neumann-Haefelin C, Thimme R. Cellular immune responses to hepatocellular carcinoma: lessons for immunotherapy. Dig Dis. 2012;30:483-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 117. | Breous E, Thimme R. Potential of immunotherapy for hepatocellular carcinoma. J Hepatol. 2011;54:830-834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 98] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 118. | Matar P, Alaniz L, Rozados V, Aquino JB, Malvicini M, Atorrasagasti C, Gidekel M, Silva M, Scharovsky OG, Mazzolini G. Immunotherapy for liver tumors: present status and future prospects. J Biomed Sci. 2009;16:30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 119. | Bremer C, Allkemper T, Menzel J, Sulkowski U, Rummeny E, Reimer P. Preliminary clinical experience with laser-induced interstitial thermotherapy in patients with hepatocellular carcinoma. J Mag Reson Imag. 1998;8:235-239. |
| 120. | Vogl T, Mack M, Straub R, Eichler K, Engelmann K, Roggan A, Zangos S. Percutaneous interstitial thermotherapy of malignant liver tumors]. RöFo. 2000;172:12. |
| 121. | Araújo TG, de Oliveira AG, Tobar N, Saad MJ, Moreira LR, Reis ER, Nicola EM, de Jorge GL, dos Tártaro RR, Boin IF. Liver regeneration following partial hepatectomy is improved by enhancing the HGF/Met axis and Akt and Erk pathways after low-power laser irradiation in rats. Lasers Med Sci. 2013;28:1511-1517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 20] [Article Influence: 1.7] [Reference Citation Analysis (0)] |