Published online Jul 7, 2011. doi: 10.3748/wjg.v17.i25.3035
Revised: January 2, 2011
Accepted: January 9, 2011
Published online: July 7, 2011
AIM: To determine the incidence of peripheral fractures in patients with celiac disease (CD) and the effect of treatment on fracture risk.
METHODS: We compared the incidence and risk of peripheral fractures before and after diagnosis between a cohort of 265 patients who had been diagnosed with CD at least 5 years before study entry and a cohort of 530 age- and sex-matched controls who had been diagnosed with functional gastrointestinal disorders. Data were collected through in-person interviews with an investigator. The overall assessment window for patients was 9843 patient-years (2815 patient-years after diagnosis).
RESULTS: Compared with the control group, the CD cohort showed significantly higher incidence rate and risk of first peripheral fracture before diagnosis [adjusted hazard ratio (HR): 1.78, 95% CI: 1.23-2.56, P < 0.002] and in men (HR: 2.67, 95% CI: 1.37-5.22, P < 0.004). Fracture risk was significantly associated with the classic CD presentation with gastrointestinal symptoms (P < 0.003). In the time period after diagnosis, the risk of fractures was comparable between the CD cohort and controls in both sexes (HR: 1.08, 95% CI: 0.55-2.10 for women; HR: 1.57, 95% CI: 0.57-4.26 for men).
CONCLUSION: CD patients have higher prevalence of fractures in the peripheral skeleton before diagnosis. This is associated with male sex and classic clinical presentation. The fracture risk was reduced after the treatment.
- Citation: Sánchez MIP, Mohaidle A, Baistrocchi A, Matoso D, Vázquez H, González A, Mazure R, Maffei E, Ferrari G, Smecuol E, Crivelli A, Paula JA, Gómez JC, Pedreira S, Mauriño E, Bai JC. Risk of fracture in celiac disease: Gender, dietary compliance, or both? World J Gastroenterol 2011; 17(25): 3035-3042
- URL: https://www.wjgnet.com/1007-9327/full/v17/i25/3035.htm
- DOI: https://dx.doi.org/10.3748/wjg.v17.i25.3035
In the past two decades, the effect of celiac disease (CD) on bone health has been extensively studied[1]. Osteopenia or osteoporosis detected by bone mineral density measurements has been seen in > 50% of patients at the time of their diagnosis of CD[2-5]. Data have accumulated to show that low bone mineral density is more common in adults and is present even if patients have atypical or asymptomatic CD at the time of diagnosis[1,6-8]. The impact of CD treatment on bone density has received some attention but remains under-explored[6,9-13]. Few studies have evaluated the risk of fractures, a more relevant clinical endpoint than bone mineral density, in CD patients[1,14-21]. A recent systematic review with a meta-analysis that pooled 20 995 CD patients and 97 777 controls, from eight studies published between 2000 and 2007, concluded that CD patients have a 43% higher risk of fractures compared with people without CD [pooled odds ratio: 1.43, 95% CI: 1.15-1.78][22]. A more recent study, published after the systematic review, confirmed the significant association between CD and increased fracture risk[23]. It should be noted that available studies are limited by heterogeneity in study methodology, patient population, and potential biases; thus, results have varied widely[22]. Nevertheless, evidence suggests that physicians should carefully examine the bone health of patients with CD.
Current evidence is mixed on fracture risk in patients who are maintaining a gluten-free diet (GFD) to treat CD[24]. In a seminal study that we have conducted previously[14], we found a lower rate of any type of fractures among treated CD patients compared with untreated CD patients. However, three subsequent studies with different study designs have reported different findings[18,20,21,23]. All of these studies showed that the risk of fractures in diagnosed and treated CD patients was significantly higher than in healthy controls. A fifth study did not show any significant difference between patients, before and after diagnosis[16]. Moreover, a Swedish population-based study[21] recently has reported that the elevated risk of fractures among CD patients remains unchanged 20 years after diagnosis. These studies employed different methodologies, which makes it difficult to extrapolate their findings to the general CD patient population.
Theoretically, dietary treatment can improve patients’ bone health and reduce the risk of falls, which in turn, may reduce the risk of fractures[1]. Given the equivocal evidence, a better understanding of the effect of GFD on patients’ fracture risk is of clinical importance to physicians and patients. The present study aimed to assess the risk of fractures in a large cohort of CD patients and the effect of GFD on this risk.
A cohort of 265 adult patients (> 18 years old) with a diagnosis of CD and a cohort of 530 age- and sex-matched controls with functional gastrointestinal disorders were recruited at the gastroenterology units in four medical centers in Buenos Aires, Argentina from March 2007 to November 2009. The CD diagnosis was based on a combination of positive clinical findings (presence of symptoms or risk factors such as family history), characteristic CD enteropathy in duodenal biopsy at the time of diagnosis, positive CD-specific serology, and a positive clinical and/or histological response to a GFD. The presence of positive CD-related serological tests at diagnosis (antigliadin antibodies, anti-tissue transglutaminase antibodies and/or antiendomysium antibodies) was considered sufficient for a diagnosis of CD without follow-up assessments. Patients were enrolled in the study if their diagnosis of CD had been established at least 5 years prior to their entry to the study. Confirmation of the CD diagnosis was required at the time of enrollment irrespective of the patient’s compliance with the GFD. We excluded 163 patients who were diagnosed with other disorders that could independently reduce bone health (e.g. uncontrolled thyroid dysfunction, rheumatoid arthritis, inflammatory bowel disease, diabetes), who took medications that may affect bone metabolism (e.g. steroids, calcium, vitamin D, alendronate, anticonvulsants, thyroid hormones, estrogen or androgen replacement), and who had complicated CD. Two controls subjects attending the same gastroenterology unit were enrolled for each CD patient in the study. These control subjects were selected if a definitive diagnosis of functional gastrointestinal disorder based on Rome III criteria was confirmed by their medical records, and if they had the same age and sex as the enrolled CD patient.
Medical history related to CD and fractures was taken from the CD patient and control cohorts using a standard questionnaire through in-person interviews conducted by the investigators, who were experienced with CD. The interview included demographic information; age at which the patient began to experience CD-like symptoms such as diarrhea, weight loss and anemia; age at diagnosis of CD; gynecological and obstetric history; and fracture history, including the type and severity of trauma that produced the fracture and the site of the fracture. All study participants were further questioned about their smoking habits, long-term medications, and hormone replacement therapy. Participants were asked whether they had ever broken a bone and which bone they had fractured. All data reported at the time of the interview were checked with those reported in patient records. If any discrepancy was detected, patients were contacted by telephone to confirm observations. If the discrepancy still persisted and no documentation of the event was available, the patient was excluded from the study. Trauma was considered as: (1) severe, if it involved a traffic accident, was sports-related, or caused by falling from a height; (2) moderate, if the fracture resulted from slipping or stumbling, or from a fall on level ground; and (3) mild, if minimal trauma was involved. Body weight was determined for all enrolled patients, and body mass index (BMI) was calculated.
CD patient adherence to GFD was estimated based on multiple assessments: (1) opinion of the patient’s primary treating physician; (2) patient’s self-report; and (3) a validated questionnaire[25]. The degree of adherence was characterized by one investigator as one of the following categories: (1) strict (adherence for > 90% of the time); (2) partial (50%-90% of the time); or (3) poor (< 50% of the time).
Each study unit tabulated data in a centralized Excel spread sheet. The data were periodically verified via comparison with patients’ medical records and, if necessary, corrected by three investigators who were not involved in data collection. If discrepancies were noted for a study subject, the subject was contacted by the data reviewer and the most accurate information available was accepted as valid. Data on each year of diagnosis and clinical presentation of CD were confirmed by the patient’s medical records. Based on the clinical presentation at the time of CD diagnosis, a patient was categorized as presenting with classic (predominantly gastrointestinal symptoms), atypical (extra-intestinal symptoms), or silent (asymptomatic cases detected through screening) CD. The periods before and after diagnosis for control subjects were categorized according to the index CD case.
Results are reported as median and range, mean and 95% CI, or mean and standard error of the mean ± SE as appropriate for the data distribution. In the statistical analysis, the time period “before diagnosis” for both populations was defined as the period from a patient’s date of birth to 1 year after the date of diagnosis of CD in the index case. Conversely, the time period “after diagnosis” was defined as the period between 1 year after the diagnosis and the time of study enrollment. We included the first year after diagnosis as part of the “before diagnosis” period to minimize the potential residual effect produced by a long-term disease and slow recovery on GFD. It has previously been observed that the risk of complications may be elevated in the immediate period before and after CD diagnosis[26]. Time at risk of fractures for patients and controls was defined as the period between birth (before diagnosis) or diagnosis of CD (after diagnosis) and the time (age) of the first fracture or the enrollment in the study, whichever came first. The rate of factures was compared between the CD and control cohorts.
Comparisons between cohorts were performed using Student’s t test or Mann-Whitney test. Multivariate linear regression analyses were performed. Data were also reported as incidence rate (IR), which represents the number of events/1000 subject-years at risk, and as the excess number of events (IR of CD patients minus IR of controls). Cox regression analysis was conducted to estimate and compare the risk of fractures between cohorts. Results were reported as hazard ratio (HR) and 95% CI. Separate analysis of fractures was performed by the before/after CD diagnosis period and by sex. The risk of fractures before diagnosis was also analyzed by clinical presentation (classic CD vs atypical/silent forms). The HR was adjusted for potential confounders, including age, age at diagnosis, BMI, smoking, and gynecological and obstetric history. The effect of GFD treatment on fracture risk was analyzed by the degree of compliance with the GFD. Statistical significance was defined as 95% CI not including 1.0.
Table 1 summarizes the demographic and clinical characteristics of the CD and control cohorts. The sex and age distributions were well matched between the cohorts. Most subjects were female (84%). Among the CD patients, the median age at CD diagnosis was 30 years, and 65% of the cases were diagnosed at ≤ 16 years of age.
| CD patients | Control population | P value | |
| No. of patients (F/M) | 265 (223/42) | 530 (446/84) | |
| Median age (yr) (range) | 42 (18-85) | 43 (16-87) | |
| Age at diagnosis (yr) median (range) | 30 (1-80) | - | |
| BMI (kg/m2, mean ± SE) | 22.5 ± 0.2 | 24.3 ± 0.2 | 0.001 |
| Age at menarche (yr) median (range) | 13 (9-17) | 12 (9-20) | |
| Age at menopause (yr) median (range) | 48 (30-54) | 49 (36-59) | |
| Person-years before diagnosis | 7028 | 14 532 | |
| Person-years after diagnosis | 2815 | 5628 | |
| Total No. of fractures | 89 | 93 | 0.0001 |
| Total No. of cases with at least one fracture | 61 | 82 | 0.02 |
| No. of patients with at least one fracture before diagnosis | 40 | 45 | 0.006 |
| No. of patients with at least one fracture after diagnosis | 21 | 37 | |
| Age at first fracture before diagnosis (yr), median (range) | 10 (2-61) | 15 (1-74) | |
| Age at first fracture after diagnosis (yr), median (range) | 21 (5-75) | 37 (6-71) | |
| Type of trauma producing fracture (No. of cases) | |||
| Mild | 27 | 24 | |
| Moderate | 24 | 34 | |
| Severe/sports | 10 | 24 |
CD patients had significantly lower BMI at study enrollment compared with controls (P < 0.001). Female CD patients and controls were comparable in age at menarche or menopause. The overall assessment period was 9843 patient-years for the CD cohort and 20 160 person-years for the control cohort.
Table 2 presents CD patients’ clinical characteristics and fracture history according to gender. Female patients were on average older at study entry and at CD diagnosis than male patients (P < 0.04 and P < 0.003, respectively). Male CD patients had significantly higher BMI at the time of enrolment (P < 0.05) and a greater proportion of time at risk after diagnosis (61% vs 37%). According to our assessment of patient adherence to GFD, 85 (38%), 48 (22%) and 90 (40%) female patients and 19 (45%), 7 (17%) and 16 (38%) of male patients were deemed as poor, partial, and strict adherents, respectively.
| Female | Male | P value | |
| No. of patients | 223 | 42 | |
| Median age (yr), range | 42 (18-62) | 35 (18-66) | 0.04 |
| Age at diagnosis (yr), median (range) | 31 (1-80) | 19 (1-52) | 0.003 |
| BMI (kg/m2, mean ± SD) | 22.5 ± 0.5 | 23.7 ± 0.6 | 0.01 |
| Person-years before diagnosis | 6380 | 647 | |
| Person-years after diagnosis | 2371 | 444 | |
| Total no. of fractures | 57 | 32 | 0.0001 |
| Total no. of cases with at least one fracture | 42 | 19 | 0.0005 |
| No. of patients with at least one fracture before diagnosis | 29 | 11 | 0.05 |
| No. of patients with at least one fracture after diagnosis | 13 | 8 | 0.01 |
| Age at first fracture before diagnosis (yr), median (range) | 14 (2-61) | 10 (6-32) | 0.04 |
| Age at first fracture after diagnosis (yr), median (range) | 54 (5-75) | 13 (5-60) | |
| Type of trauma producing first fracture (No. of cases) | |||
| Mild | 20 | 7 | |
| Moderate | 18 | 6 | |
| Severe/sportive | 4 | 6 |
Overall, CD patients reported a significantly higher rate of having experienced at least one fracture (23%) compared with controls (15%) (Table 1). Twenty-eight percent of the CD patients with a history of fractures had more than one fracture, compared with only 12% of controls (P < 0.04). The mean number of fractures was 1.46 per CD patient and 1.13 per control subject (P < 0.0001). Multiple fractures appeared to be limited to a subset of cases. Ten of the 11 control subjects with multiple fractures had two; however, 17 CD patients with multiple fractures reported up to four different fractures in the peripheral skeleton. Compared with controls, CD patients had a lower median age at the time of the first fracture (P < 0.05). Cole’s fracture was the most common site in the peripheral skeleton for CD patients, as well as controls (54% vs 42%, respectively); possibly because most cases and controls were < 50 years old. One CD patient and no controls reported hip fracture. Finally, compared with controls, more CD patients with fractures reported that the event was caused by mild trauma (29% vs 44%, respectively, P < 0.05). No differences were observed between cohorts in terms of moderate and severe/sport-related traumas.
Among CD patients, the rate of fractures was higher in male (59%) than female (26%) population (P < 0.0001) (Table 2). Male patients had the first fracture at an earlier age than females (P < 0.04). Mild trauma was the most common cause of first fracture in women (48% of cases with at least one fracture vs 37% in men) and a severe/sports injury was more common in men (32% vs 9.5% in women).
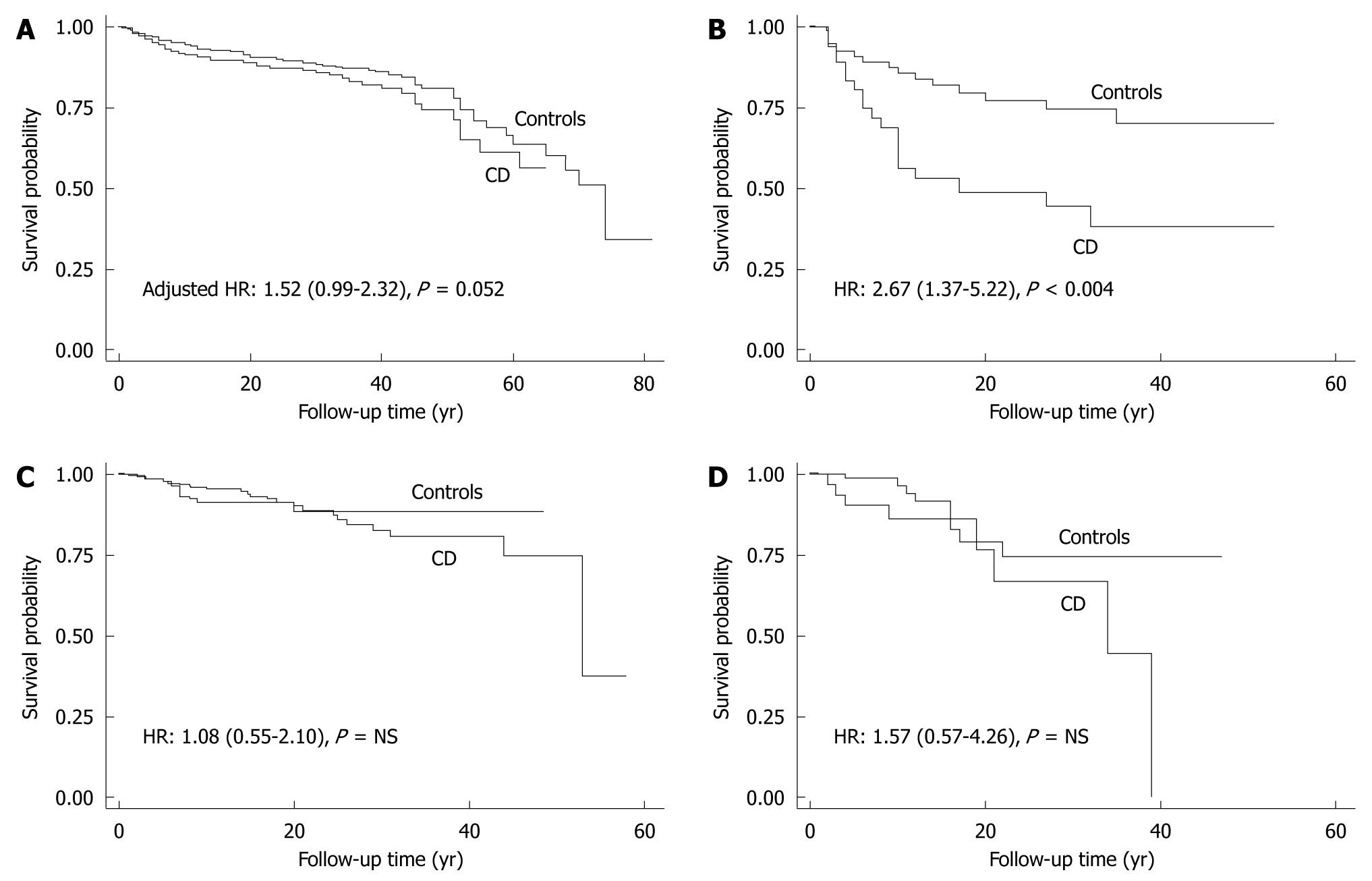
As shown in Table 3, the risk of fractures in the peripheral skeleton before the diagnosis of CD was higher in the CD than in the control cohort. Compared with controls, the excess number of fractures estimated in the CD cohort was 3.03 per 1000 patients/year. Although the excess of fractures (1.49 events) in female CD patients was marginally higher than in the matched female controls, the excess number of fractures was significantly higher in male CD patients (19.15 events) than in male controls. The risk of fractures before diagnosis was linked to confounders such as age at study entry, age at CD diagnosis, smoking, menopause and BMI only in female patients (Table 3). However, none of these confounders individually modified the estimated risk above 10%. Among CD patients, fractures before CD diagnosis occurred at younger age in male than in female patients (P < 0.04) (Table 2).
| CD patients | Controls | HR (95% CI) | P | |
| Before diagnosis | ||||
| Overall population | ||||
| IR | 8.67 | 5.64 | 1.53 (1.05-2.14) | 0.01 |
| Adjusted HR | 1.78 (1.23-2.56) | 0.002 | ||
| Females | ||||
| IR | 6.58 | 5.09 | 1.28 (0.87-1.88) | NS |
| Adjusted HR | 1.52 (0.99-2.32) | 0.052 | ||
| Males | ||||
| IR | 29.35 | 10.20 | 2.67 (1.37-5.22) | 0.004 |
| Adjusted HR | 2.63 (1.24-5.59) | 0.01 | ||
| After diagnosis | ||||
| Overall population | ||||
| IR | 7.45 | 6.04 | 1.28 (0.74-2.21) | NS |
| Adjusted HR | No significant change | |||
| Females | ||||
| IR | 5.48 | 5.30 | 1.08 (0.55-2.10) | NS |
| Adjusted HR | No significant change | |||
| Males | ||||
| IR | 18.02 | 9.83 | 1.57 (0.57-4.26) | NS |
| Adjusted HR | No significant change | |||
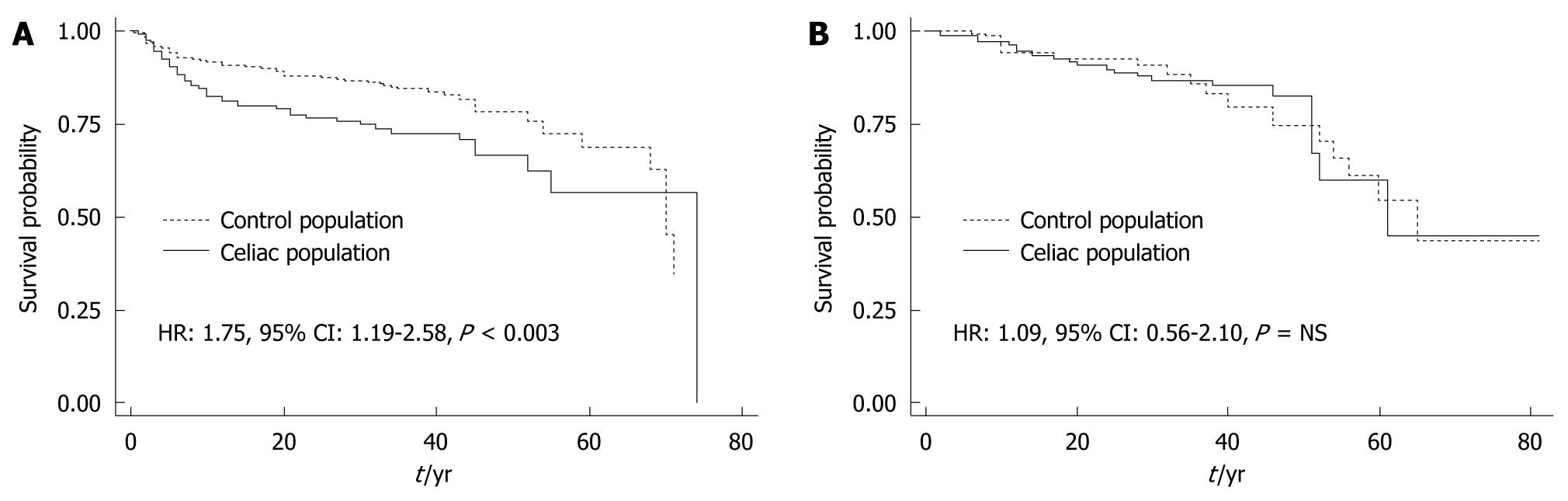
Figure 1 depicts the survival curves of time to first fracture in CD patients by their clinical presentation at the time of CD diagnosis as compared with controls. The IR of fractures in the classic CD patients was almost twice that of their matched controls (10.14 vs 5.73 per 1000, respectively; HR 1.75, 95% CI: 1.19-2.58, P < 0.003). In contrast, the incidence of fractures in atypical/silent CD patients did not differ significantly from that of their matched controls (5.44 vs 5.84, respectively; HR: 1.09, 95% CI: 0.57-2.10, P = NS).
Compared with the time period before diagnosis, the IR for the first peripheral fracture after CD diagnosis was comparable between the control and CD cohorts. After CD diagnosis, the IR of fractures for the CD cohort decreased from the pre-diagnosis period (-1.22 events per 1000 patients/year). Furthermore, compared with matched controls, all CD cases had an excess of fractures of 1.41 events (HR: 1.28, 95% CI: 0.74-2.21, P = NS) in the after-diagnosis period. Compared with the before-diagnosis period, female CD patients had a lower IR (-1.10 events) after diagnosis, and their risk of fractures was comparable to that of controls (excess of fractures in cases: 0.18 events) (P = NS) (Table 3). Male patients had a significant decrease in fractures from before to after CD diagnosis (-11.33 events/1000 patients per year). However, in the post-diagnosis period, male CD patients continued to have an excess number of fractures (8.19 events) compared with controls, which was not statistically significant (HR: 1.57, 95% CI: 0.57-4.26, P = NS). Female patients categorized as poorly adherent with the GFD (n = 90) had an IR very similar to the before-diagnosis IR (6.41 events/1000 patients per year). Although only one of 16 (7.2%) strictly adherent male patients had at least one fracture after diagnosis, seven of 19 (36.8%) poorly adherent male patients had a fracture. The small number of male patients prevented us from estimating their IR and fracture risk. Figure 2 shows the survival curve of time for first fracture for patients and matched control population before and after the diagnosis of CD according to gender.
Previous studies have demonstrated that CD patients have an increased risk of fractures in the peripheral skeleton. Whether this risk can be modified by a GFD is still unclear. Our seminal study has suggested that the prevalence of fractures decreases after initiation of a GFD[14]. However, this conclusion has been challenged by other studies[18,20,21,23]. Some authors have suggested that an early diagnosis and therapeutic intervention for CD before bone damage occurs is the only way to significantly lower the risk of fractures in CD patients.
The present study confirmed the increased risk of fractures overall among CD patients compared to controls with functional gastrointestinal disorders, and this increased risk was most prominent before their CD diagnosis. Additional supportive findings included the increased incidence of fractures produced by mild trauma events (for female cases) and a history of multiple fractures (up to four different events) seen in a subset of CD patients. The increased risk for female CD patients was more pronounced and statistically borderline when data were adjusted for potential confounders such as age at study entry, age at CD diagnosis, smoking, menopause, and BMI. Thus, older age, later diagnosis, cigarette smoking, and lower BMI were factors that contributed to the higher incidence of fractures in the peripheral skeleton. The effect of these confounders was not significant in male patients before CD diagnosis and in the overall patient cohort after CD diagnosis. Our study also confirmed our previous observation that the increased risk of peripheral fractures before CD diagnosis was associated with the classic clinical presentation but not with atypical/silent forms.
Of note, the present study is believed to be the first to identify sex as a relevant risk factor for fracture risk in CD patients; especially before they are diagnosed. The fact that the IR in male controls was more than twice that in female controls indicates that males have a higher exposure to trauma, regardless of whether they have CD. Furthermore, the IR in male CD patients was more than fourfold higher than that in female CD patients, and almost threefold higher than that in matched male controls. In addition, male CD patients had their first fractures before diagnosis at a younger age than female CD patients. Our findings do not support the hypothesis that the increased IR in male CD patients is due to osteoporotic fractures (i.e. events caused by mild trauma).
Our present study also provides original evidence for the profound impact of treatment with GFD on the risk of fractures in the peripheral skeleton. The improvement in bone health was seen in both sexes. These findings are in line with previous evidence that has shown that gluten restriction can reverse the systemic and local physiological mechanisms in bone deterioration of CD patients[3,11-13,27]. Although normalization of bone mass is unlikely in adult CD patients, significant re-mineralization of axial and peripheral skeleton has been shown in several studies[3,6,10-13]. It should be noted that reducing the risk of fracture does not solely depend on increasing bone mass and mineral density[27-32]. Other risk factors, such as structural alteration of bones with impairment of the mechanical quality (stiffness of cortical bones), deterioration of protective factors from trauma (body mass, fat and muscle compartments), and neuromuscular dysfunction, also contribute to bone weakness in CD patients[28,30]. In this context, improving body mass and fat/muscle composition, nutritional status, and bone architecture through long-term GFD treatment may reduce the overall risk of fractures in CD patients. Our study provides further support to the clinical benefits of GFD. Although the conclusion is limited by sample size, our data suggest that greater adherence to a GFD may be beneficial in male as well as female patients.
The sex differences observed in the risk of fractures in CD patients have not been reported before and deserve further comment. A previous study on bone structure and strength in CD patients detected some sex differences in mineral and bone metabolism, localization of bone damage (predominantly cortical/subcortical bone mass of the radius), mechanical quality of bones, and changes induced by 1-year treatment with a GFD[27], which may be related to differences in the development of the male and female mammalian skeleton. At 1-year follow-up, gluten-free treatment appeared to correct only the metabolically induced disturbances, which were predominant in women. However, the current results suggest that long-term adherence to a GFD may significantly reduce fracture risk in male patients as well.
Although intriguing, the current study results were limited by a relatively small number of male patients; larger studies are needed to confirm these findings. Another limitation was that fractures were based on self-report and may have been subject to recall errors; however, the risk of failed recall is expected to be similar between patients and controls. Misclassification of the type of trauma may have biased the results toward a positive association between bone disorders and osteoporotic fractures in CD patients. However, this association is well-established in female patients and not corroborated in male patients; therefore, the conclusions are not likely to have been altered. The assessment of GFD adherence is difficult, particularly in retrospective analyses. Our assessment relied on patients’ self-reports and detailed interviews conducted by expert physicians, and was characterized by an independent researcher unaware of other clinical information.
In conclusion, this cohort study confirms the increased risk of fractures in the peripheral skeleton in undiagnosed CD patients and an association of bone damage with the classic, but not the atypical/silent clinical presentation of CD. In addition, this study is believed to be the first to demonstrate a higher excess risk of fracture in male patients compared with female patients before CD diagnosis. Sex differences in the pathogenesis of bone weakness should be further explored. Finally, the study is also believed to be the first to recognize a beneficial effect of a GFD in reversing the elevated risk of fractures, and patients who adhere to long-term GFD can achieve a similar risk of fracture to those without CD, which provides a further argument for strict adherence to the diet to prevent complications of CD.
Reduced bone health is seen in > 50% of celiac disease (CD) patients at the time of diagnosis. Very few studies have evaluated the risk of fractures in CD patients. A recent systematic review and meta-analysis of eight studies, published between 2000 and 2007, concluded that CD patients had a 43% higher risk of fractures compared with people without the disorder.
Although several studies have shown a positive effect of a gluten-free diet (GFD) on bone density and other bone-protective factors, the impact of treatment on the risk of fractures remains controversial. Some studies have suggested that the risk of fractures detected before diagnosis of CD remains elevated several years after diagnosis. In this study, the authors explored the incidence of fractures in the peripheral skeleton of CD patients before diagnosis and the effect of CD treatment on fracture risk.
This study confirms that, before diagnosis, CD patients have a significantly higher rate of fractures in the peripheral skeleton compared with controls with functional gastrointestinal disorders. In addition, the risk is associated with the classic presentation of CD (predominantly gastrointestinal symptoms). This study is believed to be the first to demonstrate that the increased incidence of fractures in CD patients is associated with male sex and that, with treatment GFD, the fracture risk becomes comparable to controls.
The study further supports the importance of adherence to a GFD to reduce the risk of bone complications in CD patients.
The paper provides relevant and novel information, but some issues deserve discussion. I would strongly suggest to engage in a much more in depth discussion and speculation on their opposite findings in CD patients.
Peer reviewer: Dr. Giuseppe Chiarioni, Gastroenterological Rehabilitation Division of the University of Verona, Valeggio sul Mincio Hospital, Azienda Ospedale di Valeggio s/M, Valeggio s/M 37067, Italy
S- Editor Tian L L- Editor Kerr C E- Editor Zheng XM
| 1. | Corazza GR, Di Stefano M, Mauriño E, Bai JC. Bones in coeliac disease: diagnosis and treatment. Best Pract Res Clin Gastroenterol. 2005;19:453-465. |
| 2. | Caraceni MP, Molteni N, Bardella MT, Ortolani S, Nogara A, Bianchi PA. Bone and mineral metabolism in adult celiac disease. Am J Gastroenterol. 1988;83:274-277. |
| 3. | González D, Mazure R, Mautalen C, Vazquez H, Bai J. Body composition and bone mineral density in untreated and treated patients with celiac disease. Bone. 1995;16:231-234. |
| 4. | McFarlane XA, Bhalla AK, Reeves DE, Morgan LM, Robertson DA. Osteoporosis in treated adult coeliac disease. Gut. 1995;36:710-714. |
| 5. | Corazza GR, Di Sario A, Cecchetti L, Tarozzi C, Corrao G, Bernardi M, Gasbarrini G. Bone mass and metabolism in patients with celiac disease. Gastroenterology. 1995;109:122-128. |
| 6. | Mora S, Weber G, Barera G, Bellini A, Pasolini D, Prinster C, Bianchi C, Chiumello G. Effect of gluten-free diet on bone mineral content in growing patients with celiac disease. Am J Clin Nutr. 1993;57:224-228. |
| 7. | Mazure R, Vazquez H, Gonzalez D, Mautalen C, Pedreira S, Boerr L, Bai JC. Bone mineral affection in asymptomatic adult patients with celiac disease. Am J Gastroenterol. 1994;89:2130-2134. |
| 8. | Mustalahti K, Collin P, Sievänen H, Salmi J, Mäki M. Osteopenia in patients with clinically silent coeliac disease warrants screening. Lancet. 1999;354:744-745. |
| 9. | Bodé S, Hassager C, Gudmand-Høyer E, Christiansen C. Body composition and calcium metabolism in adult treated coeliac disease. Gut. 1991;32:1342-1345. |
| 10. | McFarlane XA, Bhalla AK, Robertson DA. Effect of a gluten free diet on osteopenia in adults with newly diagnosed coeliac disease. Gut. 1996;39:180-184. |
| 11. | Valdimarsson T, Löfman O, Toss G, Ström M. Reversal of osteopenia with diet in adult coeliac disease. Gut. 1996;38:322-327. |
| 12. | Mautalen C, González D, Mazure R, Vázquez H, Lorenzetti MP, Maurino E, Niveloni S, Pedreira S, Smecuol E, Boerr LA. Effect of treatment on bone mass, mineral metabolism, and body composition in untreated celiac disease patients. Am J Gastroenterol. 1997;92:313-318. |
| 13. | Bai JC, Gonzalez D, Mautalen C, Mazure R, Pedreira S, Vazquez H, Smecuol E, Siccardi A, Cataldi M, Niveloni S. Long-term effect of gluten restriction on bone mineral density of patients with coeliac disease. Aliment Pharmacol Ther. 1997;11:157-164. |
| 14. | Vasquez H, Mazure R, Gonzalez D, Flores D, Pedreira S, Niveloni S, Smecuol E, Mauriño E, Bai JC. Risk of fractures in celiac disease patients: a cross-sectional, case-control study. Am J Gastroenterol. 2000;95:183-189. |
| 15. | Fickling WE, McFarlane XA, Bhalla AK, Robertson DA. The clinical impact of metabolic bone disease in coeliac disease. Postgrad Med J. 2001;77:33-36. |
| 16. | Vestergaard P, Mosekilde L. Fracture risk in patients with celiac Disease, Crohn’s disease, and ulcerative colitis: a nationwide follow-up study of 16,416 patients in Denmark. Am J Epidemiol. 2002;156:1-10. |
| 17. | Thomason K, West J, Logan RF, Coupland C, Holmes GK. Fracture experience of patients with coeliac disease: a population based survey. Gut. 2003;52:518-522. |
| 18. | West J, Logan RF, Card TR, Smith C, Hubbard R. Fracture risk in people with celiac disease: a population-based cohort study. Gastroenterology. 2003;125:429-436. |
| 19. | Moreno ML, Vazquez H, Mazure R, Smecuol E, Niveloni S, Pedreira S, Sugai E, Mauriño E, Gomez JC, Bai JC. Stratification of bone fracture risk in patients with celiac disease. Clin Gastroenterol Hepatol. 2004;2:127-134. |
| 20. | Davie MW, Gaywood I, George E, Jones PW, Masud T, Price T, Summers GD. Excess non-spine fractures in women over 50 years with celiac disease: a cross-sectional, questionnaire-based study. Osteoporos Int. 2005;16:1150-1155. |
| 21. | Ludvigsson JF, Michaelsson K, Ekbom A, Montgomery SM. Coeliac disease and the risk of fractures - a general population-based cohort study. Aliment Pharmacol Ther. 2007;25:273-285. |
| 22. | Olmos M, Antelo M, Vazquez H, Smecuol E, Mauriño E, Bai JC. Systematic review and meta-analysis of observational studies on the prevalence of fractures in coeliac disease. Dig Liver Dis. 2008;40:46-53. |
| 23. | Jafri MR, Nordstrom CW, Murray JA, Van Dyke CT, Dierkhising RA, Zinsmeister AR, Melton LJ. Long-term fracture risk in patients with celiac disease: a population-based study in Olmsted County, Minnesota. Dig Dis Sci. 2008;53:964-971. |
| 25. | Nachman F, del Campo MP, González A, Corzo L, Vázquez H, Sfoggia C, Smecuol E, Sánchez MI, Niveloni S, Sugai E. Long-term deterioration of quality of life in adult patients with celiac disease is associated with treatment noncompliance. Dig Liver Dis. 2010;42:685-691. |
| 26. | Ludvigsson JF, Montgomery SM, Ekbom A. Celiac disease and risk of adverse fetal outcome: a population-based cohort study. Gastroenterology. 2005;129:454-463. |
| 27. | Ferretti J, Mazure R, Tanoue P, Marino A, Cointry G, Vazquez H, Niveloni S, Pedreira S, Mauriño E, Zanchetta J. Analysis of the structure and strength of bones in celiac disease patients. Am J Gastroenterol. 2003;98:382-390. |
| 28. | Ferretti JL. Biomechanical properties of bone. Bone densitometry and osteoporosis. Berlin: Springer 1998; 143-161. |
| 29. | Marshall D, Johnell O, Wedel H. Meta-analysis of how well measures of bone mineral density predict occurrence of osteoporotic fractures. BMJ. 1996;312:1254-1259. |
| 31. | Villani P, Brondino-Riquier R, Bouvenot G. [Fragility of scientifically acquired data. The example of fluoride salts in osteoporosis]. Presse Med. 1998;27:361-362. |
| 32. | Rho JY, Kuhn-Spearing L, Zioupos P. Mechanical properties and the hierarchical structure of bone. Med Eng Phys. 1998;20:92-102. |