Published online Oct 7, 2009. doi: 10.3748/wjg.15.4627
Revised: September 11, 2009
Accepted: September 18, 2009
Published online: October 7, 2009
Most anemia is related to the digestive system by dietary deficiency, malabsorption, or chronic bleeding. We review the World Health Organization definition of anemia, its morphological classification (microcytic, macrocytic and normocytic) and pathogenic classification (regenerative and hypo regenerative), and integration of these classifications. Interpretation of laboratory tests is included, from the simplest (blood count, routine biochemistry) to the more specific (iron metabolism, vitamin B12, folic acid, reticulocytes, erythropoietin, bone marrow examination and Schilling test). In the text and various algorithms, we propose a hierarchical and logical way to reach a diagnosis as quickly as possible, by properly managing the medical interview, physical examination, appropriate laboratory tests, bone marrow examination, and other complementary tests. The prevalence is emphasized in all sections so that the gastroenterologist can direct the diagnosis to the most common diseases, although the tables also include rare diseases. Digestive diseases potentially causing anemia have been studied in preference, but other causes of anemia have been included in the text and tables. Primitive hematological diseases that cause anemia are only listed, but are not discussed in depth. The last section is dedicated to simplifying all items discussed above, using practical rules to guide diagnosis and medical care with the greatest economy of resources and time.
- Citation: Moreno Chulilla JA, Romero Colás MS, Gutiérrez Martín M. Classification of anemia for gastroenterologists. World J Gastroenterol 2009; 15(37): 4627-4637
- URL: https://www.wjgnet.com/1007-9327/full/v15/i37/4627.htm
- DOI: https://dx.doi.org/10.3748/wjg.15.4627
| Regenerative anemia |
| Acute or chronic bleeding |
| Hemolytic anemia |
| Hereditary (hemoglobinopathy, enzymopathy, membrane-cytoskeletal defects) |
| Acquired (autoimmune, mechanical destruction, toxic-metabolic, drugs, infectious, PNH, hypersplenism) |
| Hypo-regenerative anemia |
| Bone marrow failure caused by stem cell pathology |
| Quantitative disorder |
| Selective: erythroblastopenia (pure red cell aplasia) |
| Global: aplastic anemia |
| Qualitative disorder (dysmyelopoiesis) |
| Inherited: hereditary dyserythropoiesis |
| Acquired: myelodysplasia |
| Bone marrow infiltration |
| Leukemia, lymphoma, multiple myeloma |
| Solid tumors |
| Myelofibrosis |
| Thesaurismosis (Gaucher disease) |
| Inflammatory chronic diseases, microorganisms (Histoplasma, HIV) |
| Drugs, hypothyroidism, uremia |
| Erythropoietic factors deficiency |
| Iron |
| IDA |
| ACD |
| Cobalamin and folate |
| Megaloblastic anemia |
| Hormones: erythropoietin, thyroid hormones, androgens, steroids |
| Microcytic anemia (MCV < 82 fL) |
| IDA |
| Thalassemia |
| Non thalassemic conditions associated with microcytosis |
| ACD (e.g. rheumatoid arthritis, Hodgkin’s lymphoma, chronic infection, neoplasia) |
| Sideroblastic anemia (e.g. hereditary, lead poisoning) |
| Normocytic anemia (MCV = 82-98 fL) |
| Nutritional anemia (iron deficiency, cobalamin y/o folate) |
| Anemia of renal insufficiency |
| Hemolytic anemia |
| Red cell intrinsic causes: membranopathy, enzymopathy, hemoglobinopathy |
| Red cell extrinsic causes: immune-mediated, microangiopathic, associated with infection, chemical agent (spider venoms), metabolic |
| ACD |
| Primary bone marrow disorder |
| Causes that are intrinsic to hematopoietic stem cells: bone marrow aplasia (idiopathic, PNH, Fanconi syndrome), pure red cell aplasia (acquired, congenital, Diamond-Blackfan syndrome), myelodysplastic syndrome |
| Extrinsic causes: drugs, toxins, radiation, viruses, immune-mediated, bone marrow infiltration (metastatic and lymphoma) |
| Macrocytic anemia (MCV > 98 fL) |
| Drugs (hydroxyurea, zidovudine, methotrexate) |
| Nutritional (vitamin B12 and folate deficiency) |
| Drug-induced hemolytic anemia |
| Dyserythropoiesis, myelodysplastic syndrome, clonal hematologic disorder |
| Hereditary hematologic disorders |
| Mild macrocytosis (MCV = 100-110 fL) |
| Reticulocytes |
| Excess alcohol intake, liver disease, smoking |
| Hypothyroidism, Waldenström’s macroglobulinemia |
| Copper deficiency, bone marrow aplasia, erythroblastopenic anemia |
| Down syndrome |
| Chronic obstructive pulmonary disease |
| ↓MCV | Normal MCV | ↑MCV | |
| Normal RDW | β-thalassemia | Normocytic | Bone marrow aplasia |
| α-thalassemia | |||
| Increased RDW | Iron deficiency | Inflammatory anemia Hypothyroidism | Megaloblastic anemia |
| ACD | IDA | Mixed anemia | |
| Iron | ↓ | ↓ | ↓ |
| Transferrin | ↓ or N | ↑ | ↓ |
| Transferrin saturation | ↓ | ↓ | ↓ |
| Ferritin | N or ↑ | ↓ | ↓ or N |
| Serum transferrin receptor | N | ↑ | N or ↑ |
| Ratio: soluble receptor of transferrin/log ferritin | < 1 | > 2 | > 2 |
| Cytokine levels | ↑ | N | ↑ |
| Cobalamin deficiency |
| Poor diet |
| Deficiency of intrinsic factor |
| Pernicious anemia |
| Total or partial Gastrectomy |
| Ingestion of caustic (lye) |
| Functional defect of intrinsic factor |
| Alteration of ileal (microenvironment) |
| Insufficient pancreatic protease activity |
| Inactivation enzyme (Sd. Zollinger-Ellison) |
| Competition for cobalamin |
| Alteration of ileal mucosa |
| Acquired |
| Surgical resection or by-pass |
| Regional enteritis (Crohn’s disease) |
| Tropical esprue |
| Celiac disease, Tuberculosis |
| Lymphomatous infiltration |
| Induced by drugs |
| Congenital |
| Sd Immerslund-Gräsbeck |
| Congenital transcobalamin II deficiency or abnormality |
| Congenital methylmalonic acidemia and aciduria |
| Hemodialysis |
| Urinary losses (congestive heart failure) |
| Folate deficiency |
| Dietary |
| Old age, infancy, poverty, alcoholism, chronic invalids, psychiatrically disturbed, scurvy and kwashiorkor |
| Excess utilization or loss |
| Physiologic: pregnancy and lactation, prematurity |
| Pathologic: Hematologic diseases |
| Malignant diseases |
| Inflammatory disease |
| Metabolic disease |
| Excess urinary loss, congestive heart failure, active liver disease |
| Hemodialysis, peritoneal dialysis |
| Malabsorption |
| Congenital |
| Anti-folate drugs |
| Alcoholism |
| Tropical sprue, gluten-induced enteropathy |
| Extensive jejune resection, Crohn’s disease, partial gastrectomy, systemic bacterial infection, Whipple’s disease |
| Congenital abnormalities of folate metabolism |
| Cyclohydrolase, methionine synthetase |
| Combined deficit of folate and cobalamin |
| Celiac disease |
| Regional enteritis (Crohn’s disease) |
| Congenital disorder of DNA synthesis |
| Disorders of DNA synthesis induced by drugs |
| Anti-folate |
| Purine antagonists |
| Pyrimidine antagonists |
| Alkylating |
| Eritroleucemia |
| Gastrointestinal causes of anemia |
| Microcytic anemia |
| Iron deficiency |
| Decreased iron absorption |
| Frequent: Celiac disease, gastrectomy, H pylori colonization |
| Infrequent: Bowel resection, bacterial overgrowth |
| Occult gastrointestinal blood loss |
| Frequent: aspirin and nonsteroidal anti-inflammatory drug use, colonic carcinoma, gastric ulceration, angiodysplasia, inflammatory bowel diseases |
| Infrequent: esophagitis, esophageal carcinoma, gastric antral vascular carcinoma, small bowel tumors, ampullary carcinoma, Ancylomasta duodenale |
| Non-gastrointestinal blood loss |
| Frequent: menstruation, blood donation, ACD |
| Infrequent: Hematuria, epistaxis |
| Sideroblastic anemia (alcohol, lead, drugs), vitamin B6 |
| ACD |
| Normocytic anemia |
| Frequent: ACD (liver disease, renal insufficiency, malignancy, nutritional deficiency, drug effects, alcoholism, recent trauma or surgery, iron deficiency |
| Infrequent: primary bone marrow disorder |
| Macrocytic anemia |
| Non-megaloblastic |
| Systemic disease: |
| Frequent: liver disease, alcoholism |
| Infrequent: primary bone marrow disease (myelodysplastic syndrome, aplastic anemia), metastatic infiltration, hemolytic anemia, hypothyroidism |
| Megaloblastic anemia |
| Vitamin B12 deficiency: pernicious anemia, gastrectomy, hereditary deficiency of intrinsic factor, inflammatory bowel disease, primary intestinal malabsorptive disorders, parasitic colonization, nutritional deficiencies |
| Folate deficiency: diet poor in folates, regional enteritis, Whipple’s disease, scleroderma, amyloidosis, increase requirements (liver disease, hemolytic anemia) |
| Antifolate drugs: methotrexate |
Erythrocytes and the digestive system are linked closely from the very beginning of life. The yolk sack is the origin of the first generation of erythrocyte precursors. Yolk-sac-derived progenitor cells may seed the developing liver via the circulation and produce mature red blood cells that are required to meet the metabolic needs of the fetus. By week 8, liver-derived red cells are evident and the liver is the only source of erythrocytes until the 18th week of gestation. Afterwards, the spleen and bone marrow take over, but a small percentage of hepatic erythropoiesis remains until the postnatal week 6.
In adult life, the esophagus, stomach, bowel and liver are involved in the pathogenesis of different types of anemia caused by nutritional deficiency, bleeding or chronic inflammation.
Several diseases of the esophagus may produce chronic or acute bleeding: varicose veins, diverticula, diaphragmatic hernia, tumors, ulcers, esophagitis and Mallory-Weiss syndrome. The stomach plays a decisive role in the protection of vitamin B12 until it is absorbed in the final portion of the ileum, and contributes to an optimum absorption of iron reducing Fe3+ to Fe2+. The stomach also may be a source of bleeding in peptic ulcer, gastritis and tumors, and treatment with acetylsalicylic acid and nonsteroidal anti-inflammatory drugs. The absorption of iron is a sophisticated and specialized process located mostly in the duodenum. The integrity of the small-intestinal mucosa is essential for folate absorption. Vitamin B12 absorption takes place chiefly in the ileum. In consequence, several gastrointestinal diseases, hereditary or acquired, including surgical resection, may produce nutritional deficiency anemia. Small-bowel inflammatory disease is a widespread cause of chronic disease anemia. Chronic bleeding is a crucial sign in the diagnosis of malignant and benign lesions (such as diverticula) in the colon and rectum.
Splenomegaly is a relatively common cause of chronic hemolysis, and Zieve’s syndrome is an infrequent cause of acute hemolytic crisis. Last, but not least, the liver plays an essential role in the control of iron metabolism through the hepcidin pathway, and accounts for most vitamin B12, folate and iron stores.
The links between anemia and digestive system are strong and multiple. Because of this, hematologist and the specialist in digestive system should work together to acquire a profound know of common pathogenic processes.
Anemia is defined as a lower than normal hemoglobin concentration. Low hematocrit is a subrogate value for anemia, but it is not measured directly by the hematological analyzer. Instead, it is calculated from hemoglobin and other parameters. The erythrocyte count may be misleading in the evaluation of anemia. In fact, in some cases of microcytic anemia, such as thalassemia, there is usually an elevated erythrocyte count (spurious polycythemia).
In order to make a generalized approach to the diagnosis of anemia, the World Health Organization (WHO) has established a reference range for normal blood hemoglobin concentration, depending on age and sex[1]. According to this criterion, anemia is present if the blood concentration of hemoglobin falls below 130 g/L in men or 120 g/L in women. This rule does not apply to infants, children and pregnant women, who have their own tables of lower limits of hemoglobin concentration. The WHO criterion has been accepted widely for diagnosis and publication, but its universal application has been questioned mainly because of racial differences. Beutler has proposed a lower limit of hemoglobin (1-2 g less) in African Americans than in Caucasians. The reference range of hemoglobin concentration in blood may vary depending on the population analyzed, age, sex, environmental conditions and food habits[2,3].
Anemia causes tissue hypoxia and triggers compensating mechanisms. Both processes together produce the signs and symptoms characteristics of anemic syndrome. Patients with anemia may present with fatigue, dizziness and dyspnea; however, mild anemia shows few clinical signs or symptoms. The sings of anemia include pallor of the conjunctivae, face, nail beds and palmar creases, although the absence of pallor does not rule out anemia[4].
Anemia is one of the most frequent causes of medical visits because of the high incidence in children, young women and elderly people, especially if malnutrition is present. Moreover, anemia is one of the leading sings in many diseases or is the first evidence of disease observed in routine blood cell enumeration. Anemia is unusually prevalent in developing countries because of malnutrition, and genetic, parasitic or infectious diseases[5,6]. The prevalence of anemia varies greatly, from 2.9% to 61%, depending on population, age, sex, and normal limits of hemoglobin used by the author[7].
The most frequent cause of chronic anemia is gastrointestinal pathology, and this explains why patients with anemia are often sent for consultation to a gastroenterological specialist[8].
Anemia is a syndrome, not a disease, and therefore, the etiology must always be investigated and therapy must be directed mainly to the causal disease, and not only to restoring a normal hemoglobin concentration. In a patient with anemia, whose clinical and laboratory data are insufficient to find the cause, examination of the digestive system is a priority for two reasons. The first one is the high frequency of anemia in bowel disease, and the second is the opportunity to diagnose a malignant disease before it is too advanced[9-11].
Anemia can be classified from three points of view: pathogenesis, red cell morphology, and clinical presentation. All are important to guide the diagnosis. Pathogenic mechanisms involved in the production of anemia are very simple: inadequate production and loss of erythrocytes a a result of bleeding or hemolysis. Based on these pathogenic mechanisms, anemia can be divided into two types. (1) Hypo-regenerative: when bone marrow production is decrease as a result of impaired function, decreased number of precursor cells, reduced bone marrow infiltration, or lack of nutrients; (2) Regenerative: when bone marrow responds appropriately to a low erythrocyte mass by increasing production of erythrocytes.
In practice, classification based on basic parameters of red cell morphology such as mean corpuscular volume (MCV), allows for a quicker diagnostic approach.
Anemia also can be classified according to the form of clinical presentation as acute (usually bleeding or hemolysis) or chronic.
Anemia can be classified as microcytic, normocytic or macrocytic, depending on MCV. As stated above, it can be hypo-regenerative or regenerative, which depends on the number of reticulocytes. Using both, the list of possible diagnoses in the individual patient is reduced considerably. Both parameters can be supplied routinely by most of the automatic hematological cell counters.
The reticulocyte count is useful to distinguish anemia in which there is an appropriate bone marrow response from that in which there is a decrease in the production of erythrocytes. The concentration of reticulocytes reports on the bone marrow response to anemia. This approach is especially useful when MCV is normal.
A decrease in hemoglobin stimulates erythropoiesis through an increase in circulating erythropoietin. Therefore, when the bone marrow shows a normal regenerative capacity, there should be an inverse relationship between the decrease in hemoglobin and the increased number of reticulocytes (regenerative anemia). The expected reticulocyte count is much higher than normal to compensate for the anemia. However, when the hemoglobin decreases and the bone marrow does not have regenerative capacity, the expected increase in reticulocytes fails, despite the increase in erythropoietin plasma level (hypo-regenerative anemia)[12].
The reticulocyte count is expressed as a proportion of the number of erythrocytes, which must be corrected for anemia, and for increased lifespan of reticulocytes in peripheral blood. Most hematological analyzers provide directly the number of reticulocytes per mm3, which is a better estimation of erythropoietic activity than a percentage. However, the absolute reticulocyte enumeration per mm3 overestimates the actual activity of erythropoiesis, since reticulocytes are released earlier and remain longer in the circulating blood. To avoid these confounding events, it is recommended to calculate the reticulocyte index.
The next step is the calculation of reticulocyte production index (RPI):
RPI = correcteds reticulocyte count (%) × (haematocrit observed/normal hematocrit) : F (reticulocyte maturation times in vivo)
RPI in a healthy person is 1. An RPI > 3 in a patient with anemia suggests an appropriate bone marrow response, and therefore, regenerative anemia[13,14]. Nevertheless, the best estimate of the actual erythropoietic activity[15], and easiest to calculate, is to divide by two the number of reticulocytes per mm3.
This is characterized by increased generation of erythropoietin in response to decreased hemoglobin concentration, and generally reflects a loss of erythrocytes, due to bleeding or hemolysis (Table 1). In both cases, there is a typical increase in reticulocytes. The bleeding can be intense, with a sharp drop of hematocrit and obvious clinical signs; or of small intensity and chronic, with progressive decrease in hematocrit and MCV, which may go unnoticed. Over time, chronic hemorrhagic anemia becomes hypo-regenerative and microcytic because of depletion of iron stores.
Hemolysis, can be acute (usually intravascular) or chronic (usually extravascular). Acute hemolysis is characterized by sudden episodes with very obvious clinical signs (fever, chills, back pain, dark urine) and typical laboratory data (hemoglobinuria and reduced plasma haptoglobin)[16].
This is caused by alteration of bone marrow progenitor cells, which can be located at different stages of differentiation and maturation. The impairment of pluripotent stem cells usually produces pancytopenia (anemia, leukopenia and thrombocytopenia). Pancytopenia may be caused by intrinsic [bone marrow aplasia, leukemia, myelodysplastic syndrome (MDS) or myelofibrosis] or extrinsic (metastasis, Gaucher disease and other thesaurismosis, tuberculosis, histoplasmosis, viral and parasitic infections). All of them are capable of displacing normal hematopoiesis or changing the microenvironment necessary for regeneration, differentiation and proliferation of stem cells[16,17].
Less frequently, progenitor cells committed to the erythroid line (burst-forming unit-erythroid and colony-forming unit-erythroid) are affected selectively, and the result is pure red cell aplasia. When progenitor red cells are impaired selectively, bone marrow erythroblasts are much reduced or absent, while other hematological cellular lines remain normal. Pure red cell aplasia, hereditary or acquired, is very rare. However, a qualitative alteration of the red cell line (dyserythropoiesis) is often seen in clinical practice. In contrast to pure red cell aplasia, the bone marrow is rich in erythroblasts in patients with dyserythropoiesis. In these cases, the erythropoiesis is abnormal morphologically and functionally. Erythroblastopenia as dyserythropoiesis may have a hereditary or acquired origin. The former is extremely rare. Thymoma is the most common cause of acquired pure red cell aplasia. MDS is the most common cause of acquired dyserythropoiesis. In MDS patients, the lesion is located at the level of very primitive multipotent progenitors, and thus, the morphological and functional alterations can affect all blood cell lines (erythrocyte, granulocyte-monocyte and megakaryocyte)[18-20].
Anemia caused by nutritional deficiencies or decreased production of erythropoietin is much more frequent than that caused by a primitive defect in bone marrow.
Pathogenic classification is very important to understand the mechanisms involved in the genesis of anemia (Table 2). However, in daily clinical practice, it is more useful to start with the analytical parameters of the hemogram. MCV allows us to classify anemia as microcytic (MCV < 82 fL), normocytic (MCV = 82-98 fL) and macrocytic (MCV > 98 fL)[21,22].
MCV has a relationship with mean corpuscular hemoglobin (MCH), which reports on the mean hemoglobin per erythrocyte expressed in picograms (normal range: 27-32 pg). Therefore, MCV and MCH decrease (microcytic, hypochromic anemia) or increase (macrocytic, hyperchromic anemia) jointly. The MCH concentration (MCHC) reports on the average concentration of hemoglobin in each erythrocyte expressed as a percentage (normal range: 32%-36%), and its variations are very small, even in the presence of hypochromia. MCHC increases only in a few rare diseases such as hereditary spherocytosis, and therefore, its practical utility is scarce.
It should always be borne in mind that MCV is an average value and therefore does not provide information about the homogeneity of the erythrocyte population. To resolve this problem, hematological analyzers provide the erythrocyte distribution curve, with an index of dispersion: red blood cell distribution width (RDW) (normal range: 10%-14%). RDW is a rough indicator of anisocytosis and is an essential complement to MCV (Table 3).
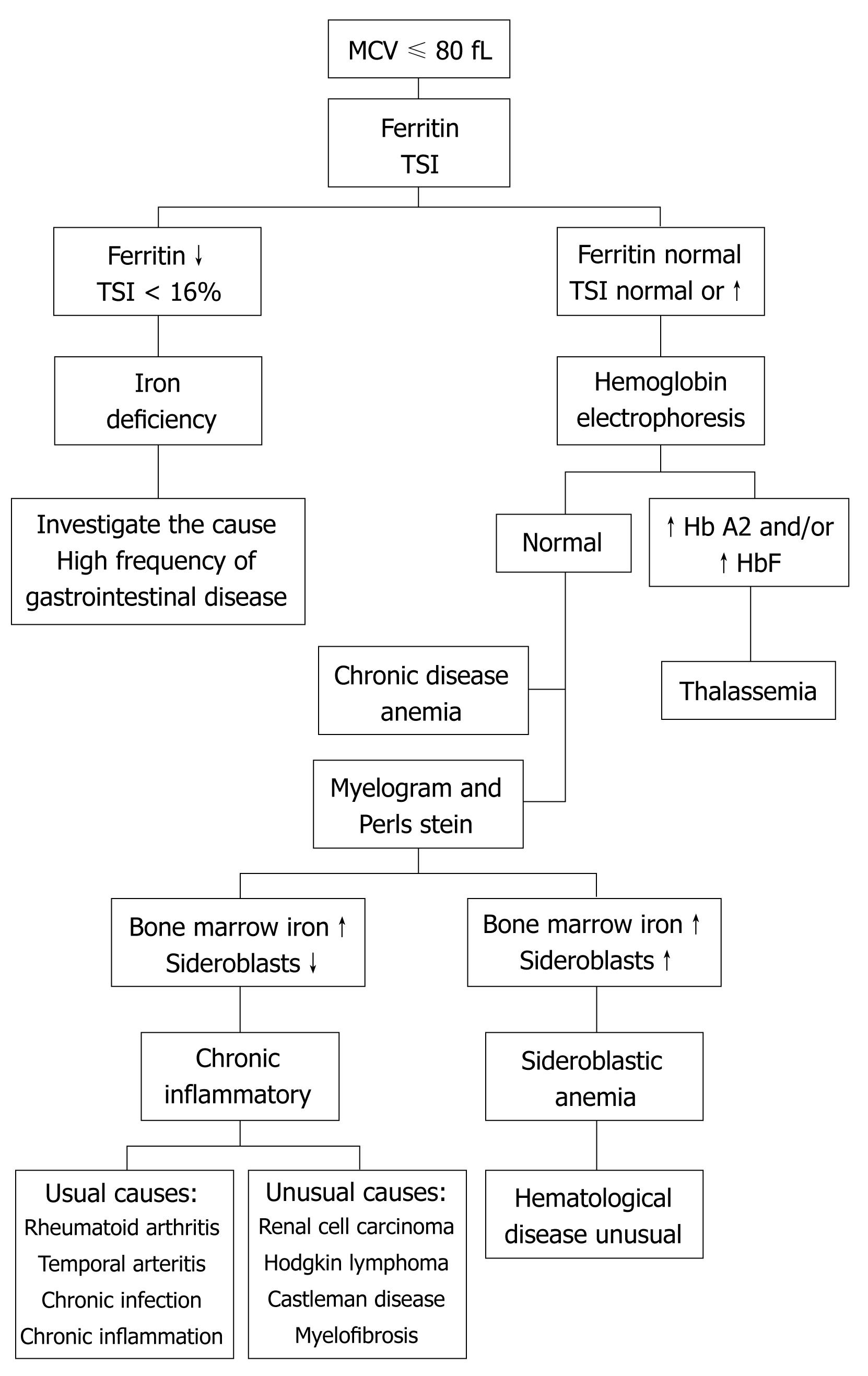
Faced with microcytic anemia, the three main diagnostic possibilities include iron deficiency anemia (IDA), thalassemia, and anemia of chronic disorders (ACD). A fourth possibility, sideroblastic anemia, is so rare that is not considered in the initial diagnosis, unless there is a history of contact with lead (Figure 1).
Iron deficiency is the most common cause of anemia, so the first step in diagnosis should be directed toward confirmation or exclusion of IDA. Serum ferritin allows us to confirm the diagnosis. Despite ferritin being an acute phase reactant, the diagnosis of IDA is unlikely with normal or elevated ferritin levels. Other parameters of iron metabolism (serum iron, total transport capacity of iron, and transferrin) are unable to distinguish with certainty IDA from ACD[23].
IDA occurs in 2%-5% of adult men and postmenopausal women in the developed world[24,25], and is a common cause of referral to a gastroenterology clinic 4%-13%[26].
It is important to note that microcytosis without anemia is characteristic of thalassemia trait, but also of polycythemia associated with iron deficiency[27].
The simple analysis of different parameters provided by the hematological analyzer gives a diagnosis of microcytic anemia. RDW helps to distinguish thalassemia from IDA. RDW is normal in thalassemia; on the contrary, microcytic anemia with RDW > 15 is probably IDA.
In recent years, the importance of serum soluble transferrin receptor has been recognized in differential diagnosis of IDA and ACD. Serum soluble transferrin receptor is increased in IDA, without interference in case ACD is present[28,29]. Intra-erythrocytic ferritin and erythrocyte zinc-protoporphyrin also help to differentiate IDA from ACD. These parameters allow the diagnosis of borderline patients who, at another time, would have undergone unnecessary investigation of iron in the bone marrow[30-32].
In men and postmenopausal women, the bigger concern should be to rule out the presence of occult bleeding. If positive, the first exploration should be a colonoscopy, mainly in men, because of the frequent association of occult blood with adenocarcinoma[33,34]. If the colonoscopy does not reveal the cause of the anemia, gastroscopy should be performed. The study of the small bowel is more controversial, but it is convenient to keep in mind that celiac disease is a cause of IDA[35].
In premenopausal women, genital bleeding is the most frequent cause of anemia. Therefore, gastrointestinal exploration is controversial. An anamnesis directed towards the characteristics of menstrual bleeding, although a subjective criterion, may be useful for distinguishing a subgroup of women without excessive genital bleeding, who require direct assessment of occult bleeding, followed by gastrointestinal exploration if positive. In cases in which occult bleeding is negative, the exploration should be directed to the genital system[36-38].
Microcytosis associated with normal ferritin guides the diagnosis toward hereditary diseases, mainly thalassemia. If there are no family antecedents of microcytosis, it is necessary to investigate acquired causes of non-iron deficiency microcytosis, mainly ACD and sideroblastic anemia. Normal RDW guides the diagnosis toward the former and a high RDW toward the latter.
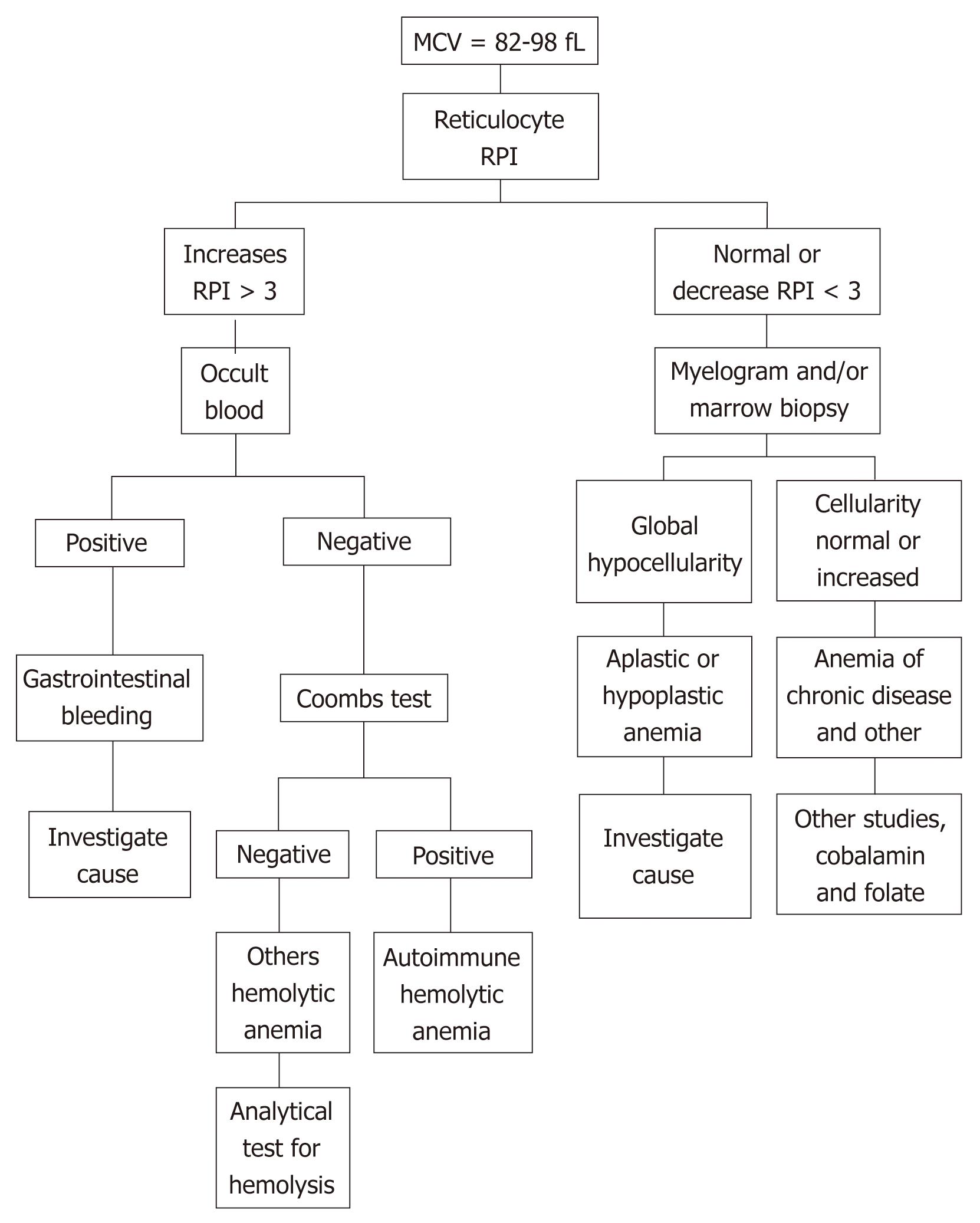
The fundamental question in normocytic anemia is to recognize the causes and susceptibility to treatment as soon as possible. Among the causes are nutritional deficiency, renal failure and hemolytic anemia[21]. Nutritional mixed anemia that combines deficiency of vitamin B12, folic acid and iron is frequent. In consequence, these three parameters should be requested in the initial phase of the diagnosis of normochromic anemia.
In order to differentiate among regenerative (hemolysis and bleeding) or hypo-regenerative anemia (bone marrow aplasia, chronic disease, nutrition deficiency and hemopathy), it is necessary to determine the corrected RPI (Figure 2). In anemia of renal failure, morphological alterations in the blood are scarce and serum erythropoietin may be normal, but inappropriately low for the degree of anemia. The severity of anemia is not evident until the disease is very advanced.
It will always be necessary to rule out hemolysis, by performing easily accessible laboratory tests [lactate dehydrogenase (LDH), indirect bilirubin, haptoglobin and reticulocytes]. These parameters do not inform us about the origin of hemolysis, and it is necessary to evaluate others, such as schistocytes in peripheral blood (intravascular hemolysis), the Coombs test (autoimmune hemolysis), tests of osmotic fragility (extravascular hemolysis), and tests to rule out hemolysis induced by drugs[39]. A detailed anamnesis and Coombs test, with and without the suspected drugs, are very useful in the investigation of drug-induced hemolytic anemia.
When the cause of normocytic anemia is not any of the previously mentioned causes, it is necessary to guide the diagnosis toward a chronic disease or to primitive hematological disease[21,22]. It is difficult to arrive at the definitive diagnosis. In this situation, it is fundamental that a careful clinical evaluation is carried out to rule out other causes of normocytic anemia: alcoholism (more frequently macrocytic), effects of drugs (chemotherapy, immunosuppression), radiotherapy, neoplasia (bone marrow infiltration), infections (mainly in hospitalized patients), surgery, or recent trauma (first phase of bleeding). The association with pathological concomitant processes, elevation of erythrocyte sedimentation rate (ESR) and the absence of morphological alterations in peripheral blood smears supports the suspicion of ACD[40]. ACD is the most frequent cause of anemia after ferropenia[41,42]. It is observed in patients that have immune hyperactivity. Activation of cytokines and the reticuloendothelial system induces changes in iron homeostasis, erythroid precursor proliferation, erythropoietin secretion, and erythrocyte life span[43]. All of these contribute to the pathogenesis of anemia.
ACD can be complicated with chronic bleeding, and in this case, the diagnosis is more difficult because of the presence of microcytosis and ferropenia. The biochemical parameter that better differentiates ACD from IDA is serum ferritin[44] (Table 4).
The hematologist must revise the blood smear if primitive disease of bone marrow is suspected. Depending on the result, a bone marrow study may be necessary to detect hematological diseases or metastasis. The biopsy, bone marrow smear, or both inform us of: the morphology of the hematopoietic cells, on their quantitative distribution, especially the myelo-erythroid ratio (normal 3:1); fibrosis; the presence of non-hematopoietic cell; or possible bone marrow aplasia. Perls’ specific iron stain informs us of the state of iron stores, and it allows quantification of siderocytes and sideroblasts. In a patient with anemia and fever of unknown origin, we must carry out careful microscopic examination of bone marrow, some selective staining, and bone marrow cultures to diagnose any underlying infectious diseases. Some infectious or parasitic diseases that are diagnosed with relative frequency are: tuberculosis, histoplasmosis, kala-azar and malaria[45-47].
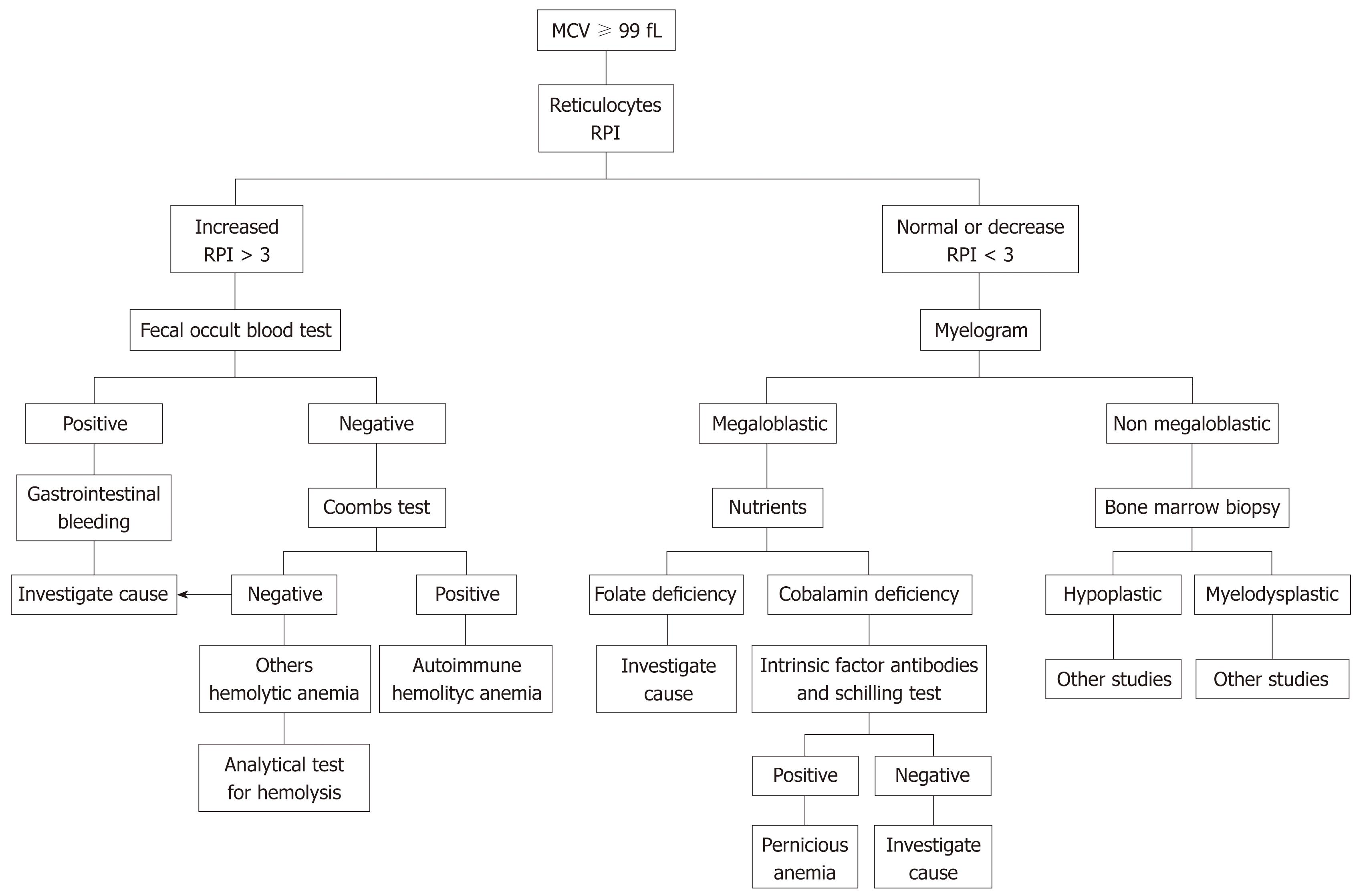
Macrocytosis is observed frequently using blood cell analyzers (Figure 3). Its prevalence is 1.7%-3.9%, but 60% of the patients with macrocytosis do not have anemia[48]. On the other hand, macrocytosis can be physiological in some circumstances (infants, pregnancy, some families). Even keeping in mind the precedent data is convenient to make a careful evaluation of macrocytosis in every patient, to rule out any underlying pathology. In vitamin B12 and folic acid deficiency, as well as in other diseases, macrocytosis (blood) is accompanied by megaloblastosis (bone marrow). In such cases, both terms can be used interchangeably[49].
The starting point in the diagnostic process for macrocytic anemia will be to rule out therapy with drugs that interfere with nucleic acid metabolism, such as.hydroxyurea, methotrexate, trimethoprim, zidovudine or 5-fluorouracil, as well as habitual intake of alcohol[49,50]. Hydroxyurea is the drug that increases most the MCV (> 110 fL); the other drugs and alcohol induce a moderate macrocytosis (100-110 fL).
In the absence of intake of any of the drugs mentioned above or alcohol, the most frequent cause of microcytic anemia is nutritional deficiency[50]. Therefore, serum vitamin B12 and folic acid levels should be evaluated. The last can be modified by the previous day’s folic acid intake. A better alternative is intra-erythrocyte folate, which remains stable during the lifetime of red blood cells and gives a better indication of possible chronic folic acid deficiency. The measurement of erythrocyte folate is a difficult technique that is not available in every laboratory. The determination of homocysteine is an accessible alternative to intra-erythrocyte folate. In the absence of folic acid, homocysteine levels increase rapidly because it cannot be converted into methionine. Normal serum levels of homocysteine are highly unlikely in folate deficiency[21].
In the same way, vitamin B12 deficiency is correlated with low serum levels, although there are clinical situations in which low serum levels are not correlated with vitamin B12 deficiency (pregnancy, elderly patients, and those with low white blood cell counts). In these circumstances, normal serum methyl-malonic acid levels, in the absence of congenital errors of metabolism or renal failure, preclude a deficiency of vitamin B12[51].
To complete the study of vitamin B12 deficiency, it is necessary to look for antibodies to intrinsic factor, which if positive, confirms the diagnosis of pernicious anemia. If not present, it is necessary to investigate malabsorption by performing the Schilling test[52] (Table 5).
The strategy for establishing a diagnosis of anemia by gastroenterologists must follow a classic pattern that includes a clinical interview, a basic physical examination, and careful selection of complementary tests.
The clinical history should emphasize the causes of blood loss, including non-gastrointestinal diseases, recent history of gastrointestinal symptoms, gastrointestinal surgery, use of nonsteroidal anti-inflammatory drugs, acetylsalicylic acid and anticoagulant therapy, family history of hematologic diseases and colorectal neoplasia[53-55]; all symptoms should be recorded, and not only the gastrointestinal ones.
A single blood sample can give information on the concentration of hemoglobin (essential for the diagnosis of anemia according to WHO criteria), the erythrocyte indices MCV and MCH (useful for the morphological classification of anemia), RDW (estimate of anisocytosis) and ESR, which reports on possible ACD.
The hematologist should examine a blood smear if any abnormal erythrocyte indices have been detected. A clotted blood sample (to obtain serum) allows the determination of the rest of the analytical parameters that are useful in diagnosis, which are iron metabolism (mainly ferritin), urea, creatinine, bilirubin and total protein. In cases of suspected IDA, the concentration of soluble transferrin receptor should be included as a key parameter in differentiation between IDA and ACD[56]. The most probable causes of microcytic anemia are IDA, thalassemia or thalassemia trait. RDW and iron metabolism are clues to differentiate between these processes in the initial step, as has been mentioned above. The most likely causes of normocytic anemia are ACD, renal failure and primitive hematological diseases (least frequently). Iron metabolism and routine biochemical tests may help to guide the diagnosis, but in some cases, erythropoietin serum levels and bone marrow examination are necessary.
In cases of macrocytic anemia, serum levels of vitamin B12 and serum and/or erythrocyte levels of folate will guide the diagnosis to deficiency of one of these. Most causes of folic acid deficiency are nutritional, although malabsorption should always be kept in mind. In contrast, vitamin B12 deficiency is almost always the result of malabsorption[49,57]. The few exceptions are in strict vegetarians and hereditary transcobalamin II deficiency (a very infrequent disease). Among malabsorption causes of B12 vitamin deficiency, the most frequent is pernicious anemia. Therefore, the first step is to establish whether the serum levels of gastrin are highly elevated, because they are > 1000 pg/mL only in atrophic gastritis type A (pernicious anemia) and Zollinger-Ellison syndrome. In pernicious anemia, pepsinogen I levels are low, gastric pH is high and gastric acid secretion does not respond to stimulation with histamine or pentagastrin. Serum antibodies against intrinsic factor and parietal cells are not sensitive or specific enough to confirm the diagnosis. The exploration that leads directly to diagnosis is gastroscopy, with mucosal biopsy and study of antiparietal cell antibodies. Gastric biopsy has the added advantage of giving information about possible metaplasia or precancerous lesions[58]. In the absence of gastric disease, the most likely cause of malabsorption of vitamin B12 is Crohn’s disease, with involvement of the terminal ileum[59]. Hereditary causes of malabsorption (Immerslund syndrome) or transport (transcobalamin II deficiency) are extremely infrequent.
The list of causes of anemia is long and cumbersome, but most can fit into a very small number of diseases on which we must concentrate as a first diagnostic option (Table 6)[60-66].
IDA is the most prevalent form of anemia worldwide, especially in women and children. Thirty-percent of the world’s population, some 1300 million people, suffer from anemia. However, the prevalence of anemia worldwide is unequal (36% in underdeveloped and 8% in developed countries) About half of the patients with anemia have IDA, which is most prevalent in the general population and in outpatients. The most likely cause of IDA is malnutrition in children, bleeding in adult males (especially gastrointestinal), menstruation or lactation in fertile women, and bleeding in the elderly. The distribution of nutrient-deficiency anemia in the elderly is: 48% iron alone, 19% folate alone, 17% vitamin B12 alone, and the rest have combined deficiencies. Therefore, in young male adults and in both sexes older than 65 years, the most likely cause of IDA is chronic bleeding, especially from gastrointestinal lesions[67].
The hemogram, the concentration of iron, ferritin and transferrin in blood and investigation of fecal occult blood are sufficient to obtain a diagnosis in most cases.
ACD is the most prevalent form of anemia after IDA in the general population[40,68], but is even more common in hospitalized patients, regardless of sex and age[42,67,69,70].
The underlying causes of ACD vary greatly: acute and chronic infections, 18%-95%; cancer, 30%-77%; autoimmune diseases (including chronic inflammatory bowel disease), 8%-71%; rejection of solid organ transplantation, 8%-70%; and chronic renal disease, 23%-50%. ACD is found in 20% of elderly people with anemia (30% excluding nutritional deficiency), and a further 4.3% suffer from ACD and kidney failure. Therefore, we should keep in mind ACD as the most likely diagnosis in elderly and inpatients, and as the second most likely diagnosis in outpatients under 65 years of age[68]. The laboratory tests needed to confirm the diagnosis include serum iron, transferrin, transferrin saturation, ferritin, urea and creatinine.
The gastroenterologist must rule out chronic inflammatory bowel disease, tumors of the digestive tract and liver disease. In some patients, the cause of anemia is complex (chronic disease and chronic hemorrhage). In these cases, it is difficult to confirm the diagnosis and additional testing is required: soluble transferrin receptor and microscopic examination of bone marrow with special stains for iron.
The most common type of anemia after IDA and ACD is megaloblastic anemia (vitamin B12 deficiency, folic acid or both). It is important in gastroenterology because intestinal malabsorption and pernicious anemia are common causes of this form of anemia. The diagnosis is suspected when MCV is elevated, and is confirmed by measuring serum vitamin B12 and serum and erythrocyte folate.
Hemolytic anemia follows next in order of frequency. Acute hemolysis presents a distinctive clinical picture, but chronic hemolysis may go unnoticed. If hemolytic anemia is suspected, the appropriate laboratory tests are antiglobulin test (direct and indirect), LDH, haptoglobin, total and conjugated bilirubin in serum, and salts and bile pigments in urine. These tests can give us the generic diagnosis of hemolytic anemia, immune or not immune, but the etiological diagnosis should be resolved in collaboration with the hematologist.
Anemia is common in hematological malignancies, but with limited gastrointestinal symptoms, except for gastric or intestinal bleeding caused by thrombocytopenia or lymphoma. It is rare, but it should be noted that gastric or intestinal lymphoma sometimes presents as mild anemia with nonspecific gastrointestinal symptoms. Endoscopy usually provides the diagnosis.
Hereditary anemia is extremely rare, except for thalassemia and sickle cell disease in certain geographical areas. Sickle cell disease is restricted to blacks. When one suspects hereditary anemia, the cooperation of a hematologist should be sought, because the diagnosis is difficult and the laboratory tests should be carefully selected.
More than 100 diseases may cause anemia, but 90% belong to three groups: nutritional deficiencies (iron, vitamin B12 and folic acid), ACD (chronic inflammation, tumors), and bleeding (excluding chronic bleeding, which produces iron deficiency). Hemolytic anemia, although less frequent than the other, is the last option before considering the diagnosis of rare diseases.
A few laboratory tests, such as blood count, ESR, serum ferritin, and serum iron and transferrin, are sufficient to focus the diagnosis. According to initial results, additional laboratory tests should be ordered: serum vitamin B12 and serum and erythrocyte folate (in case of macrocytosis), tumor markers and acute phase reactants (if ACD is suspected), LDH, haptoglobin and antiglobulin test (if hemolytic anemia is suspected).
With these key data, the doctor can put in place more specific tests: imaging, endoscopy and additional laboratory tests. The next step should be to correct the cause of anemia when possible or correct anemia itself when the cause cannot be treated. The advantages are clear if the cause is a cancer that can be diagnosed at an early stage. On the other hand, we can improve the quality of life of patients by correcting the anemia with medical treatment or regular transfusions.
Peer reviewer: Giulio Marchesini, Professor, Department of Internal Medicine and Gastroenterology, “Alma Mater Studiorum” University of Bologna, Policlinico S. Orsola, Via Massarenti
S- Editor Tian L L- Editor Kerr C E- Editor Zheng XM
| 1. | Nutritional anaemias. Report of a WHO scientific group. World Health Organ Tech Rep Ser. 1968;405:5-37. |
| 2. | Beutler E, Waalen J. The definition of anemia: what is the lower limit of normal of the blood hemoglobin concentration? Blood. 2006;107:1747-1750. |
| 3. | Patel KV, Harris TB, Faulhaber M, Angleman SB, Connelly S, Bauer DC, Kuller LH, Newman AB, Guralnik JM. Racial variation in the relationship of anemia with mortality and mobility disability among older adults. Blood. 2007;109:4663-4670. |
| 4. | Irwin JJ, Kirchner JT. Anemia in children. Am Fam Physician. 2001;64:1379-1386. |
| 5. | Hercberg S, Galan P. Nutritional anaemias. Baillieres Clin Haematol. 1992;5:143-168. |
| 6. | Dallman PR, Yip R, Johnson C. Prevalence and causes of anemia in the United States, 1976 to 1980. Am J Clin Nutr. 1984;39:437-445. |
| 7. | Beghé C, Wilson A, Ershler WB. Prevalence and outcomes of anemia in geriatrics: a systematic review of the literature. Am J Med. 2004;116 Suppl 7A:3S-10S. |
| 8. | Bermejo F, García S. Anemia crónica de origen digestivo. Tratamiento de las enfermedades gastroenterológicas. 2nd ed. Madrid: Asociación Española de Gastroenterología 2006; 465-475. |
| 9. | Looker AC, Dallman PR, Carroll MD, Gunter EW, Johnson CL. Prevalence of iron deficiency in the United States. JAMA. 1997;277:973-976. |
| 10. | Andrews NC. Disorders of iron metabolism. N Engl J Med. 1999;341:1986-1995. |
| 11. | Rockey DC. Occult gastrointestinal bleeding. N Engl J Med. 1999;341:38-46. |
| 12. | Vives Corrons JL. La anemia, aspectos generales del diagnóstico. Hematología clínica. 5th ed. Madrid: Elsevier España 2006; 107-126. |
| 13. | Hillman RS. Characteristics of marrow production and reticulocyte maturation in normal man in response to anemia. J Clin Invest. 1969;48:443-453. |
| 14. | Hillman RS, Finch CA. Erythropoiesis: normal and abnormal. Semin Hematol. 1967;4:327-336. |
| 15. | Esrlev AJ. Clinical manifestations and classification of erytrocyte disorders. Williams hematology. 6th ed. New York: McGraw-Hill 2001; 369-374. |
| 16. | Rozman C, Feliu E, Grañena A, Monserrat E, Vives Corrons JL. Hematologia. Atlas practico para el medico general. Barcelona: Salvat 1981; 25-53. |
| 17. | Zucker S, Friedman S, Lysik RM. Bone marrow erythropoiesis in the anemia of infection, inflammation, and malignancy. J Clin Invest. 1974;53:1132-1138. |
| 18. | Iolascon A, Perrota S. Anemias diseritropoyéticas congénitas. Hematología. Madrid: Aran Ediciones 2003; . |
| 19. | Jandl JH. Blood: Textbook of hematology. 2nd ed. Boston: Little Brown and Co 1996; 251-288. |
| 20. | De Cruchy GC. Clinical haematology in medical practice. Oxford: Backwell Scientific Publication 1978; . |
| 21. | Tefferi A. Anemia in adults: a contemporary approach to diagnosis. Mayo Clin Proc. 2003;78:1274-1280. |
| 22. | Tefferi A, Hanson CA, Inwards DJ. How to interpret and pursue an abnormal complete blood cell count in adults. Mayo Clin Proc. 2005;80:923-936. |
| 23. | Guyatt GH, Oxman AD, Ali M, Willan A, McIlroy W, Patterson C. Laboratory diagnosis of iron-deficiency anemia: an overview. J Gen Intern Med. 1992;7:145-153. |
| 24. | Calvey HD, Castleden CM. Gastrointestinal investigations for anaemia in the elderly: a prospective study. Age Ageing. 1987;16:399-404. |
| 26. | McIntyre AS, Long RG. Prospective survey of investigations in outpatients referred with iron deficiency anaemia. Gut. 1993;34:1102-1107. |
| 27. | Bessman JD. Microcytic polycythemia. Frequency of nonthalassemic causes. JAMA. 1977;238:2391-2392. |
| 28. | Punnonen K, Irjala K, Rajamäki A. Serum transferrin receptor and its ratio to serum ferritin in the diagnosis of iron deficiency. Blood. 1997;89:1052-1057. |
| 29. | Weiss G. Iron and immunity: a double-edged sword. Eur J Clin Invest. 2002;32 Suppl 1:70-78. |
| 30. | Iron and immunity: a double-edged sword. Serum ferritin: a technical monograph. La Jolla, CA: National Health Laboratories 1989; . |
| 31. | Labbe RF, Rettmer RL. Zinc protoporphyrin: a product of iron-deficient erythropoiesis. Semin Hematol. 1989;26:40-46. |
| 32. | Barron BA, Hoyer JD, Tefferi A. A bone marrow report of absent stainable iron is not diagnostic of iron deficiency. Ann Hematol. 2001;80:166-169. |
| 33. | Raje D, Mukhtar H, Oshowo A, Ingham Clark C. What proportion of patients referred to secondary care with iron deficiency anemia have colon cancer? Dis Colon Rectum. 2007;50:1211-1214. |
| 34. | Ioannou GN, Rockey DC, Bryson CL, Weiss NS. Iron deficiency and gastrointestinal malignancy: a population-based cohort study. Am J Med. 2002;113:276-280. |
| 35. | Rockey DC. Occult gastrointestinal bleeding. Gastroenterol Clin North Am. 2005;34:699-718. |
| 36. | Rockey DC. Gastrointestinal evaluation for premenopausal women with iron deficiency anemia: what is appropriate? Am J Med. 1998;105:356-357. |
| 37. | Green BT, Rockey DC. Gastrointestinal endoscopic evaluation of premenopausal women with iron deficiency anemia. J Clin Gastroenterol. 2004;38:104-109. |
| 38. | Farrús Palou M, Pérez Ocaña A, Mayer Pujadas MA, Piquer Gibert M, Mundet Tudurí X, Iglesias Rodal M. [Anemia in primary care: etiology and morphological characteristics]. Aten Primaria. 2000;25:230-235. |
| 39. | Hirono A, Forman L, Beutler E. Enzymatic diagnosis in non-spherocytic hemolytic anemia. Medicine (Baltimore). 1988;67:110-117. |
| 40. | Cartwright GE. The anemia of chronic disorders. Semin Hematol. 1966;3:351-375. |
| 42. | Matzner Y, Levy S, Grossowicz N, Izak G, Hershko C. Prevalence and causes of anemia in elderly hospitalized patients. Gerontology. 1979;25:113-119. |
| 43. | Means RT Jr. Recent developments in the anemia of chronic disease. Curr Hematol Rep. 2003;2:116-121. |
| 44. | Mulherin D, Skelly M, Saunders A, McCarthy D, O'Donoghue D, Fitzgerald O, Bresnihan B, Mulcahy H. The diagnosis of iron deficiency in patients with rheumatoid arthritis and anemia: an algorithm using simple laboratory measures. J Rheumatol. 1996;23:237-240. |
| 45. | Sullivan PS, Hanson DL, Chu SY, Jones JL, Ward JW. Epidemiology of anemia in human immunodeficiency virus (HIV)-infected persons: results from the multistate adult and adolescent spectrum of HIV disease surveillance project. Blood. 1998;91:301-308. |
| 46. | Nissenson AR, Goodnough LT, Dubois RW. Anemia: not just an innocent bystander? Arch Intern Med. 2003;163:1400-1404. |
| 47. | van Iperen CE, van de Wiel A, Marx JJ. Acute event-related anaemia. Br J Haematol. 2001;115:739-743. |
| 48. | Davenport J. Macrocytic anemia. Am Fam Physician. 1996;53:155-162. |
| 49. | Colon-Otero G, Menke D, Hook CC. A practical approach to the differential diagnosis and evaluation of the adult patient with macrocytic anemia. Med Clin North Am. 1992;76:581-597. |
| 50. | Beck WS. Diagnosis of megaloblastic anemia. Annu Rev Med. 1991;42:311-322. |
| 51. | Bates CJ, Schneede J, Mishra G, Prentice A, Mansoor MA. Relationship between methylmalonic acid, homocysteine, vitamin B12 intake and status and socio-economic indices, in a subset of participants in the British National Diet and Nutrition Survey of people aged 65 y and over. Eur J Clin Nutr. 2003;57:349-357. |
| 52. | Pruthi RK, Tefferi A. Pernicious anemia revisited. Mayo Clin Proc. 1994;69:144-150. |
| 53. | Abramson SD, Abramson N. 'Common' uncommon anemias. Am Fam Physician. 1999;59:851-858. |
| 54. | Goddard AF, James MW, McIntyre AS, Scott BB. Guidelines for the management of iron deficiency anaemia. Vol. 2007. London: British Society of Gastroenterology 2005; 1-6. Available from: URL: http://www.bsg.org.uk/pdf_word_docs/iron_def.pdf. |
| 55. | Gonzalez-Hermoso F, Perez-Palma J, Marchena-Gomez J, Lorenzo-Rocha N, Medina-Arana V. Can early diagnosis of symptomatic colorectal cancer improve the prognosis? World J Surg. 2004;28:716-720. |
| 56. | Caunedo A, Barroso N, Herrerías H. Laboratorio en gastroenterología. Madrid: Sociedad Española de Patología Digestiva 2003; 267-289. |
| 57. | Hernández Nieto L, Hernández García MT, Pintado Cros T, Juncá Piera J, Vives Corrons JL, Martín Vega C. Medicina Interna. C Rozman (Dir). 15th ed. Madrid: Elsevier 2004; 1644-1669. |
| 58. | Orlando LA, Lenard L, Orlando RC. Chronic hypergastrinemia: causes and consequences. Dig Dis Sci. 2007;52:2482-2489. |
| 59. | Gasche C, Berstad A, Befrits R, Beglinger C, Dignass A, Erichsen K, Gomollon F, Hjortswang H, Koutroubakis I, Kulnigg S. Guidelines on the diagnosis and management of iron deficiency and anemia in inflammatory bowel diseases. Inflamm Bowel Dis. 2007;13:1545-1553. |
| 60. | Kepczyk T, Kadakia SC. Prospective evaluation of gastrointestinal tract in patients with iron-deficiency anemia. Dig Dis Sci. 1995;40:1283-1289. |
| 61. | Rockey DC, Cello JP. Evaluation of the gastrointestinal tract in patients with iron-deficiency anemia. N Engl J Med. 1993;329:1691-1695. |
| 62. | Cook IJ, Pavli P, Riley JW, Goulston KJ, Dent OF. Gastrointestinal investigation of iron deficiency anaemia. Br Med J (Clin Res Ed). 1986;292:1380-1382. |
| 63. | Zuckerman G, Benitez J. A prospective study of bidirectional endoscopy (colonoscopy and upper endoscopy) in the evaluation of patients with occult gastrointestinal bleeding. Am J Gastroenterol. 1992;87:62-66. |
| 64. | Hardwick RH, Armstrong CP. Synchronous upper and lower gastrointestinal endoscopy is an effective method of investigating iron-deficiency anaemia. Br J Surg. 1997;84:1725-1728. |
| 65. | Gisbert JP, Gomollón F. Common misconceptions in the diagnosis and management of anemia in inflammatory bowel disease. Am J Gastroenterol. 2008;103:1299-1307. |
| 66. | de la Morena F, Gisbert JP. [Anemia and inflammatory bowel disease]. Rev Esp Enferm Dig. 2008;100:285-293. |
| 67. | Guralnik JM, Eisenstaedt RS, Ferrucci L, Klein HG, Woodman RC. Prevalence of anemia in persons 65 years and older in the United States: evidence for a high rate of unexplained anemia. Blood. 2004;104:2263-2268. |
| 68. | Weiss G, Goodnough LT. Anemia of chronic disease. N Engl J Med. 2005;352:1011-1023. |
| 69. | Maury CP, Liljeström M, Laiho K, Tiitinen S, Kaarela K, Hurme M. Tumor necrosis factor alpha, its soluble receptor I, and -308 gene promoter polymorphism in patients with rheumatoid arthritis with or without amyloidosis: implications for the pathogenesis of nephropathy and anemia of chronic disease in reactive amyloidosis. Arthritis Rheum. 2003;48:3068-3076. |
| 70. | Denz H, Huber P, Landmann R, Orth B, Wachter H, Fuchs D. Association between the activation of macrophages, changes of iron metabolism and the degree of anaemia in patients with malignant disorders. Eur J Haematol. 1992;48:244-248. |