Published online Sep 28, 2009. doi: 10.3748/wjg.15.4547
Revised: August 1, 2009
Accepted: August 8, 2009
Published online: September 28, 2009
AIM: To study the effects of lysophosphatidic acid (LPA) on proliferation, adhesion, migration, and apoptosis in the human colon cancer cell line, SW480, and its mechanisms of action.
METHODS: Methyl tetrazolium assay was used to assess cell proliferation. Flow cytometry was employed to detect cell apoptosis. Cell migration was measured by using a Boyden transwell migration chamber. Cell adhesion assay was performed in 96-well plates according to protocol.
RESULTS: LPA significantly stimulated SW480 cell proliferation in a dose-dependent and time-dependent manner compared with the control group (P < 0.05) while the mitogen-activated protein kinase (MAPK) inhibitor, PD98059, significantly blocked the LPA stimulation effect on proliferation. LPA also significantly stimulated adhesion and migration of SW480 cells in a dose-dependent manner (P < 0.05). Rho kinase inhibitor, Y-27632, significantly inhibited the up-regulatory effect of LPA on adhesion and migration (P < 0.05). LPA significantly protected cells from apoptosis induced by the chemotherapeutic drugs, cisplatin and 5-FU (P < 0.05), but the phosphoinositide 3-kinase (PI3K) inhibitor, LY294002, significantly blocked the protective effect of LPA on apoptosis.
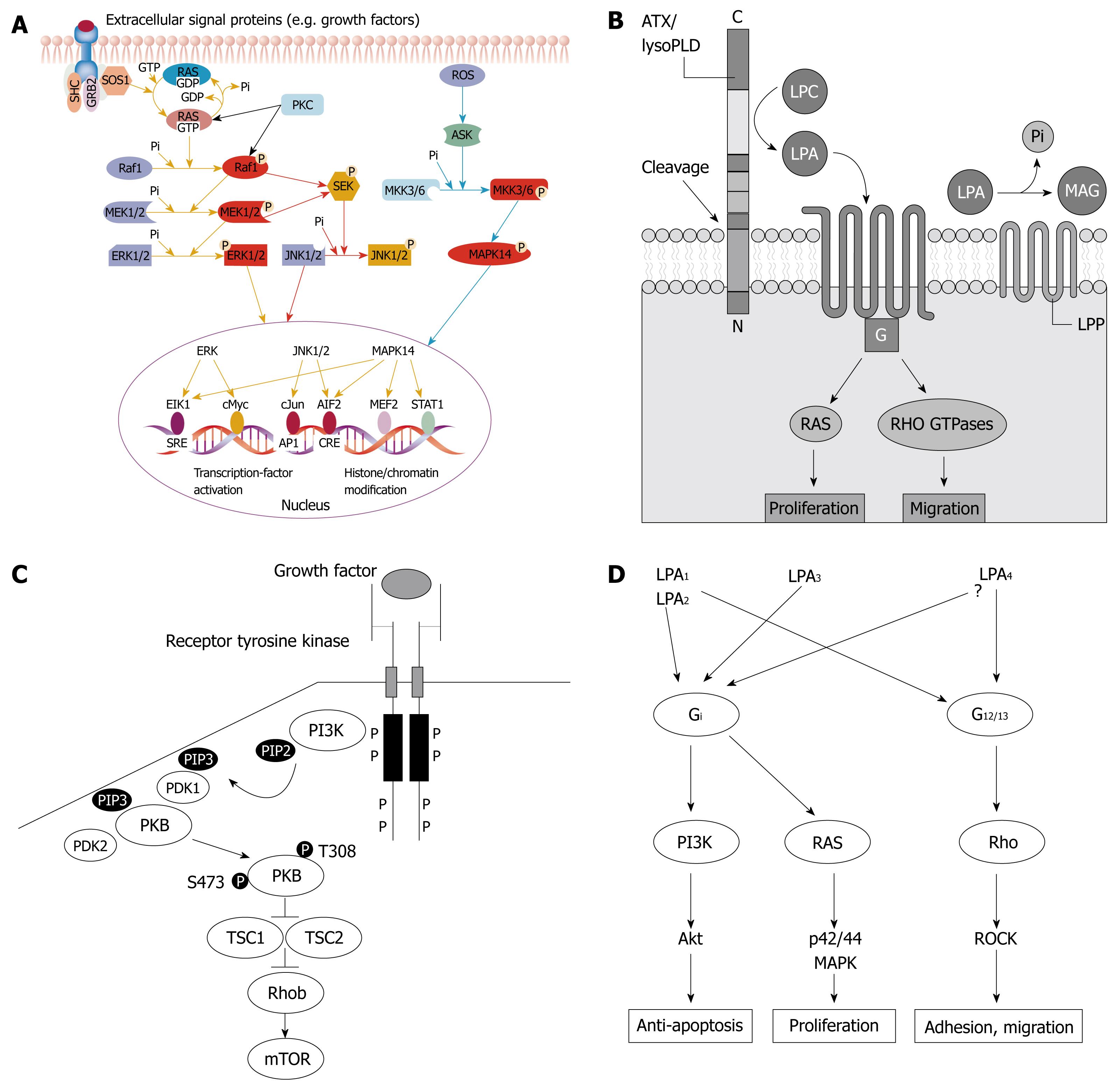
CONCLUSION: LPA stimulated proliferation, adhesion, migration of SW480 cells, and protected from apoptosis. The Ras/Raf-MAPK, G12/13-Rho-RhoA and PI3K-AKT/PKB signal pathways may be involved.
- Citation: Sun H, Ren J, Zhu Q, Kong FZ, Wu L, Pan BR. Effects of lysophosphatidic acid on human colon cancer cells and its mechanisms of action. World J Gastroenterol 2009; 15(36): 4547-4555
- URL: https://www.wjgnet.com/1007-9327/full/v15/i36/4547.htm
- DOI: https://dx.doi.org/10.3748/wjg.15.4547
Colorectal cancer (CRC) is a common form of cancer and a major cause of cancer death. The incidence of CRC has been rapidly increasing in recent years. Although the incidence of CRC was substantially lower in Asia than in the USA in the mid-twentieth century, the incidence in Japan and China has been rapidly increasing[1,2]. Thus, CRC is now a leading cancer killer worldwide.
Lysophosphatidic acid (LPA) was first found in the ascitic fluid from ovarian cancer patients. It is a bioactive glycerophospholipid generated and released by platelets, macrophages, epithelial cells, and some tumor cells. Studies have shown the presence of high levels of LPA in the ascitic fluid of patients with ovarian cancer[3] and LPA is known to be an “ovarian cancer activating factor”, which exerts growth factor-like effects through four specific G protein-coupled receptors (LPA1-4).
LPA is a potent mediator with a broad range of cellular responses, including regulation of cell proliferation, protection from apoptosis, modulation of chemotaxis and transcellular migration[4,5], which mediates survival of ovarian cancer cells, macrophages, fibroblasts, and neonatal cardiac myocytes. Some of these cellular responses indicate that LPA is a mediator of tumor progression.
In a recent study we found that plasma levels of several LPAs, including 18:1-LPA and 18:2-LPA, were significantly increased in CRC patients compared with controls[6]. This is the first report of high levels of LPA in plasma of CRC patients. It implies that LPA may play roles in CRC development. In order to clarify these roles of LPA in CRC development, the LPA effect on the CRC cell line, SW480, was studied in vitro.
LPA was found firstly to be increased in the body fluids of ovarian cancer patients, so the roles of LPA in ovarian cancer have been widely studied. A few preliminary studies of LPA in CRC have been reported, but not in the cell line SW480. Our previous study revealed a high expression of LPA receptors on SW480 cells, especially LPA receptor 2[7,8]. This study aimed to investigate the effect of LPA on proliferation, migration, adhesion, and apoptosis in the CRC cell line, SW480.
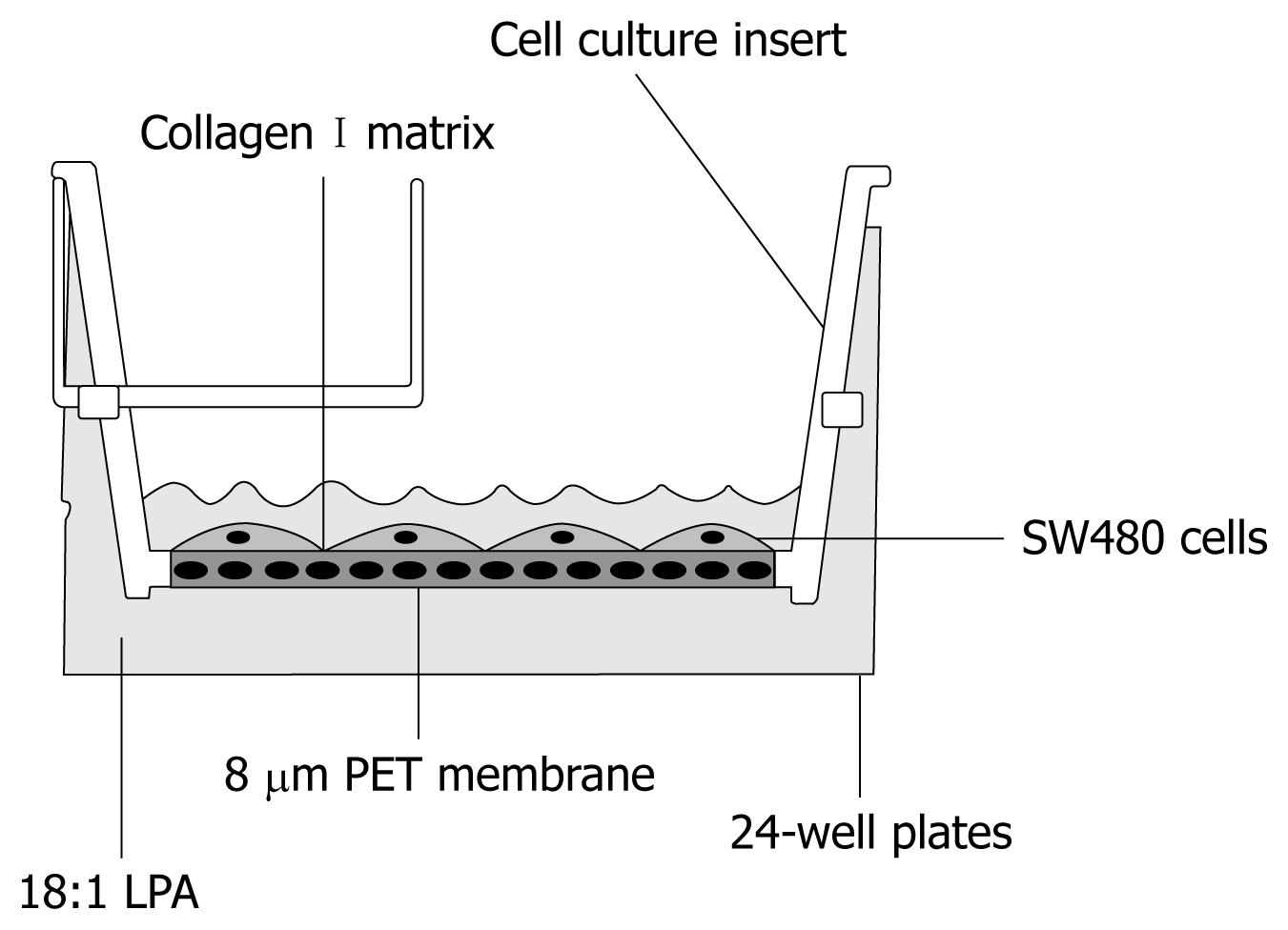
1-Oleoyl LPA (18:1 LPA) was purchased from Avanti Polar Lipids (Birmingham, Al, USA). Inhibitor of phosphoinositide 3-kinase (PI3K), LY290042, and inhibitor of mitogen-activated protein kinase (MAPK), PD98059, are from Cell Signaling (Beverly, MA, USA). Rho kinase inhibitor, Y-27632, was from Biomol (Beverly, MA, USA). Boyden transwell migration chambers and 24-well plates were from Corning Costar Corporation (Cambridge, MA).
SW480 cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 100 mL/L fetal bovine serum, streptomycin (100 mg/L), and penicillin (100 kU/L) at 37°C in 50 mL/L CO2 incubator. Cells were serum starved for 12 h before LPA treatment.
Methyl tetrazolium (MTT) colorimetry assay was employed to measure cell proliferation. SW480 cells (2 × 103/well) were seeded in 96-well plates. After cells were starved for 12 h, DMEM containing LPA supplemented with 1 g/L bovine serum albumin was put into the wells. After 24, 48, 72 and 96 h of culture, 20 µL of MTT solution (5 g/L) was added to each well. Four hours later, the medium was removed and 150 μL of dimethyl sulfoxide was added to each well. Absorbance value was measured at 490 nm on a Microplate Reader (EXL800). Each assay was performed in quintuplicate.
After treating with cisplatin or 5-FU, LPA and/or inhibitors, the cells were resuspended in binding buffer (10 mmol/L HEPES/NaOH, pH 7.4, 140 mmol/L NaCl, 2.5 mmol/L CaCl2). Then the cells were stained with 5 μL of annexin-FITC and 5 mg/L propidium iodide (PI), and then analyzed by flow cytometry (FACSCalibur cytometer, BD Biosciences), and CellQuest (BD Biosciences) was used to quantify the apoptotic cells. Experiments were performed in triplicate.
Flat-bottom 96-well plates were coated with 2 μg of collagen I (0.04 g/L) (Sigma Chemical Co.) in phosphate-buffered saline overnight at 4°C. Plates were blocked with 2 g/L BSA for 2 h at room temperature followed by washing three times with DMEM. SW480 cells (4 × 104/well) were added to each well. Four hours later, unbound cells were removed, washing twice with DMEM. Bound cells were fixed by methanol and stained with crystal blue. Stained cells were counted with a phase contrast microscope. Experiments were performed in triplicate.
Migration assays were performed in Costar transwell cell culture chamber inserts (coated with collagen I; Corning Costar Corporation, Cambridge, MA) with an 8 μm pore size as described as Figure 1. Briefly, SW480 cells (5 × 104 cells in 100 μL of starvation medium) were used for cell migration, which was conducted for 4 h at 37°C. Migrated cells were fixed, stained and counted in five randomly (× 200) selected fields with a phase contrast microscope, and the average numbers of cells per field were counted.
SW480 cells were starved in serum-free DMEM for 12 h and treated with LPA at different doses, and then reconstituted in DMEM containing 10 g/L BSA. All inhibitors including LY294002 (50 μmol/L), PD98059 (10 μmol/L), and Y-27632 (10 μmol/L) were applied to cells 30 min before the action of LPA.
Statistical significance was assessed by one-way ANOVA using SPSS software. Data are presented as the mean ± SE.
SW480 cells were starved in serum-free DMEM for 12 h and treated with LPA at different doses. After different time periods, MTT assay was performed to evaluate the activity of cell growth. It was found that LPA significantly stimulated the proliferation of SW480 cells in a dose- and time-dependent manner (P < 0.001, Figure 2A). LPA, especially when the concentration was ≥ 10 μmol/L, remarkably stimulated cell growth compared with the control group.
In order to investigate the signal pathways which mediated the stimulation effect of LPA on SW480 cells, inhibitors against key molecules of several signal transduction pathways were applied to the LPA-treated group. Three inhibitors were employed including PI3K inhibitor (LY290042), MAPK inhibitor (PD98059), and Rho kinase inhibitor (Y-27632). It was found that after applying the inhibitors, the stimulation effect of LPA on cell growth was significantly blocked by PD98059 and LY290042 (P < 0.001, Figure 2B); especially PD98059. This indicated that the Ras/Raf-MAPK signal pathway and the PI3K-AKT/PKB signal pathway may be involved in the LPA stimulation effect on proliferation of SW480 cells.
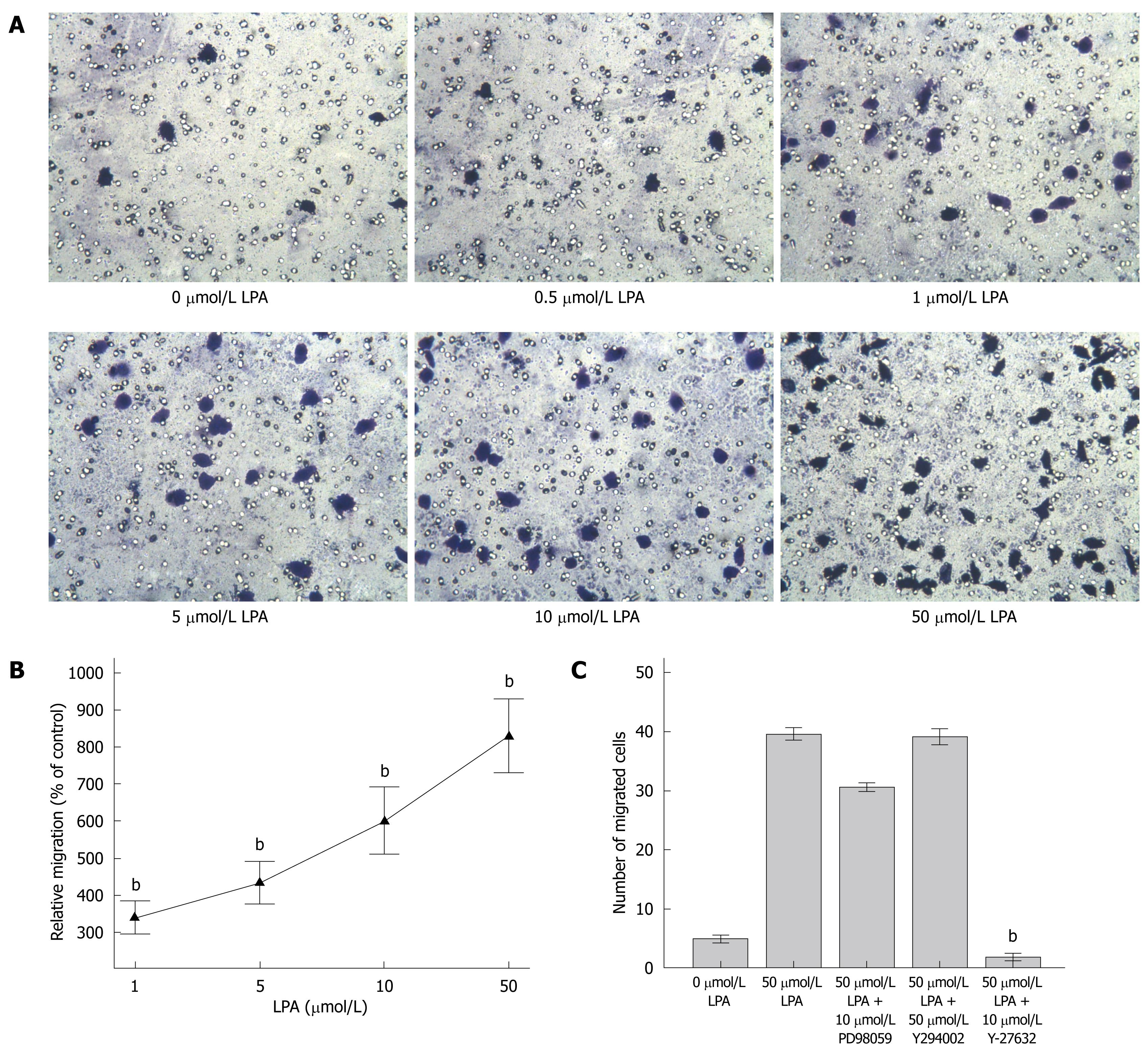
SW480 cells (1 × 105 cells in 100 μL of starvation medium) were seeded on the transwell inserts with an 8 μm pore size. Different doses of LPA in DMEM were added to the lower chamber of the transwell. Cells were then incubated at 37°C for 4 h. Cells migrated to the lower surface of inserts were fixed, stained, and quantified. It was found that LPA significantly enhanced SW480 cell migration toward the lower chamber of the transwell in a dose-dependent manner compared with the control (P < 0.001, Figure 3A and B). This indicates that LPA has a significant chemotactic effect on SW480 cells.
In order to investigate the signal pathways which mediated the chemotactic effect of LPA on SW480 cells, some inhibitors against key molecules of signal transduction pathways were employed. It was demonstrated that Rho kinase inhibitor (Y-27632 at 10 μmol/L) dramatically blocked the chemotactic effect of LPA on SW480 cells (P < 0.001, Figure 3C). This indicated that Rho kinase and G12/13-Rho-RhoA signal pathways may mediate the LPA effect on SW480 cell migration.
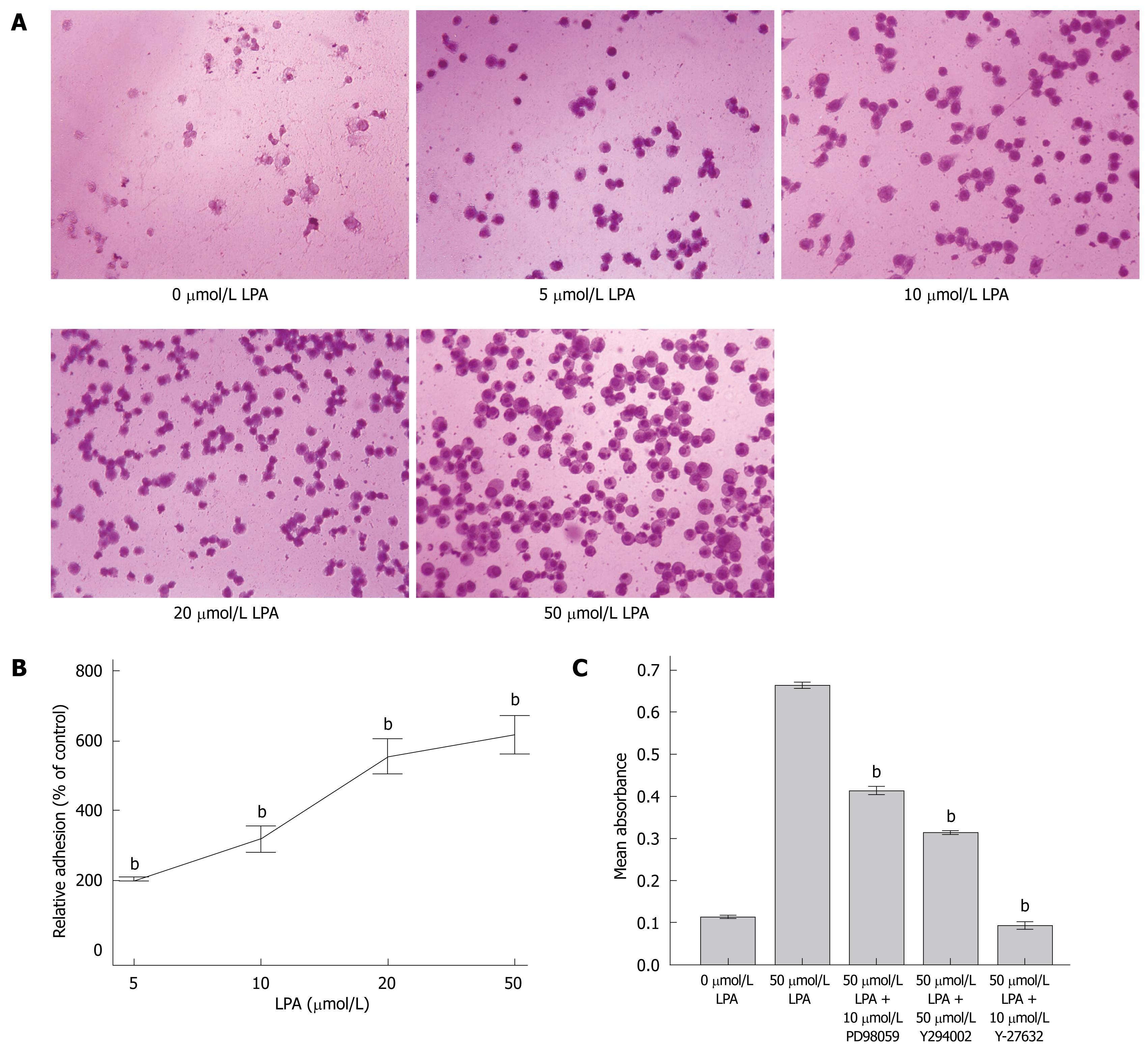
SW480 cells were seeded in 96-well plates. After the cells had undergone 12 h of starvation, LPA at different doses was added to the cells. SW480 cells were allowed to adhere to the plates for 4 h at 37°C in the incubator. Unbound cells were washed away twice. Adhered cells were fixed, stained, and quantified. Images of adhered cells under different doses of LPA were taken (Figure 4A). It was demonstrated that LPA significantly increased SW480 cell adhesion to extracellular matrix (ECM) in a dose-dependent manner compared with controls (P < 0.001, Figure 4B).
Some inhibitors were used to determine the mechanisms which mediated the LPA effect on adhesion. It was found that Rho kinase inhibitor (Y-27632) and LY294002 dramatically inhibited LPA upregulation of adhesion; especially Y-27632 (P < 0.001, Figure 4C and D). This indicated that the G12/13-Rho-RhoA signal pathway and the PI3K-AKT/PKB signal pathway may participate in the LPA effect on the adhesion of SW480 cells.
SW480 cells (1 × 105/well) seeded in 24-well culture plates were starved for 24 h and then treated with cisplatin (10 mg/L) or 5-FU (8 mg/L) for 24 h in the absence or presence of LPA (20 μmol/L). Inhibitors including LY294002 (50 μmol/L) and PD98059 (10 μmol/L) were added to the LPA-treated group.
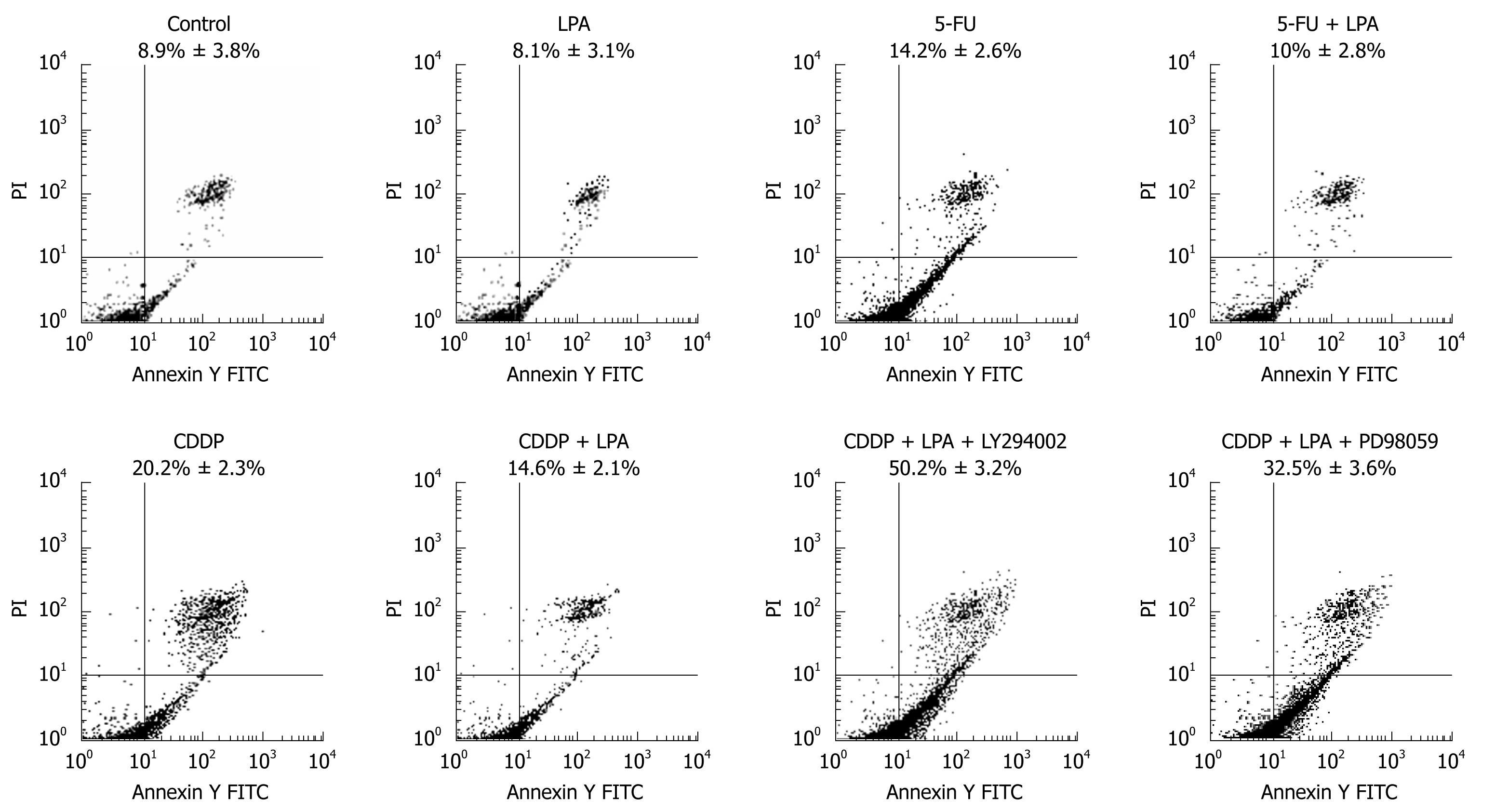
Apoptotic cells were detected by flow cytometry after Annexin V and PI staining. Apoptotic cells were defined as Annexin-positive, PI-negative (Figure 5). After cells were exposed to cisplatin and 5-FU, the apoptotic population was 20.2% ± 2.3% and 14.2% ± 2.6%, respectively. However, after the action of LPA, the apoptotic population dropped to 14.6% ± 2.1% in the cisplatin-treated group and 10% ± 2.8% in the 5-FU-treated group. LPA protected 27.7% of cells from cisplatin-induced apoptosis and protected 29.6% of cells from 5-FU-induced apoptosis. This suggests that LPA effectively protected SW480 cells from apoptotic death induced by the chemotherapeutic agents.
In order to elucidate the mechanisms of LPA protection from apoptosis, LY294002 and PD98059 were added to the LPA-treated group. Apoptotic population increased to 50.2% ± 3.2% and 32.5% ± 3.6% respectively after exposure to LY294002 (PI3K inhibitor) and PD98059 (MAPK inhibitor). This indicated that the PI3K, MAPK, PI3K-AKT/PKB signal pathways and the Ras/Raf-MAPK signal pathway may be involved in the LPA apoptotic-protection effect.
LPA, the simplest glycerophospholipid, was initially found in the ascites of ovarian cancer patients at significant levels (2-80 μmol/L), and plays an important role in the development of ovarian cancer. LPA exerts growth factor-like effects through four specific G protein-coupled receptors (LPA1-4). The effects include mitogenesis, secretion of proteolytic enzymes, and migration activity, which are accompanied by stress fiber formation and focal adhesion assembly in ovarian cancer cells.
We found recently that the level of LPA increases not only in the body fluid of ovarian cancer patients, but also in the plasma of patients with CRC. There are many studies focused on the LPA role in ovarian cancer cells. It has been found that LPA plays important roles in the progression of ovarian cancer and acts as an ovarian cancer promoter. Since LPA also increases in the plasma of CRC patients, what are the roles of LPA in CRC?
There are few studies regarding LPA effects on CRC. Furthermore, our previous study has shown that LPA receptors (LPA2, 4) are highly expressed in SW480 cells. In the present study, we preliminarily investigated the roles of LPA in the proliferation, migration, adhesion and apoptosis of SW480 cells and its mechanisms of action. We found that LPA significantly stimulated the proliferation of SW480 cells in a dose-dependent and time-dependent manner. This is consistent with the reports in ovarian cancer, in which LPA promotes growth of ovarian cancer similar to growth factor[9].
We found that the MEK1 inhibitor, PD98059, significantly inhibited the LPA effect on the proliferation of SW480 cells. MEK1, a MAPK, is a key molecule of the Ras/Raf1/MEK/ERK signal pathway[10]. We also found that the PI3K inhibitor, LY290042, partially inhibited the effect of LPA on the cell proliferation. This indicated that LPA stimulates the proliferation of SW480 cells through the Ras/Raf1/MEK/ERK pathway, and that the PI3K-AKT/PKB signal pathway may also be partially involved in the LPA effect on proliferation.
MAPK transfers major cell proliferation signals from the cell surface to the nucleus. There are three major subfamilies of MAPK, including the extracellular-signal-regulated kinase (Ras/Raf1/MEK/ERK or ERK MAPK), the c-Jun N-terminal or stress-activated protein kinase (JNK or SAPK), and MAPK14[11]. The Ras/Raf1/MEK/ERK pathway is one of the most important pathways for cell proliferation. Several lines of evidence suggest that, in CRC, the Ras/Raf1/MEK/ERK pathway, but not the JNK pathway or the p38 MAPK pathway, is the major regulator of cell proliferation. There is growing evidence that activation of the Ras/Raf1/MEK/ERK pathway is involved in the pathogenesis, progression, and oncogenic behavior of human CRC[12]. This supports our observations that LPA stimulated growth of CRC cell line SW480 through the Ras/Raf1/MEK/ERK pathway.
There are several studies reporting LPA’s effect on the proliferation of CRC cells. Zhang et al[13] has found that LPA facilitates proliferation of colon cancer cells via induction of Krüppel-like Factor 5 (KLF5). KLF5 is a transcriptional factor highly expressed in the crypt compartment of the intestinal epithelium. LPA stimulated the KLF5 expression in colon cancer cells, SW480 and HCT116. Moreover, LPA-mediated KLF5 induction was partially blocked by inhibition of MAPK kinase and protein kinase C (PKC). This also indirectly indicated that the MAPK signaling pathway is involved in the proliferation of SW480 cells.
Yang et al[14] reported that LPA-induced colon cancer cell proliferation requires the β-catenin signaling pathway. LPA activated the main signaling events in the β-catenin pathway, but inhibition of PKC blocked the effects, suggesting PKC involvement in LPA-induced activation of the β-catenin pathway. This also indirectly indicated that the PKC signaling pathway is involved in the proliferation of SW480 cells.
Balavenkatraman et al[15] reported that DEP-1 protein tyrosine phosphatase inhibits proliferation and migration of colon carcinoma cells and is upregulated by protective nutrients. Upregulation of DEP-1 expression, and in turn inhibition of cell growth and migration, may present a previously unrecognized mechanism of chemoprevention by nutrients. This result contradicts other reports.
In our study, it was found that LPA significantly enhanced the migration and adhesion of SW480 cells in a dose-dependent manner. The stimulation effect of LPA on cell adhesion, invasion and migration has been reported in other cancer types, including ovarian cancer[16-19], pancreatic cancer[18], and breast cancer[20,21]. Enhanced migration activity and increased adherence to ECM are two major factors which contribute to tumor metastasis. Cell-ECM adhesions can alter the cell’s capacity to attach and migrate through surrounding tissues. Changes of the expression and activities of the components of such adhesions could make an important contribution to preventing cancer invasion. Our study showed that LPA stimulated both migration and adhesion to ECM of colon cancer SW480 cells. This means that LPA significantly promotes the metastatic potential of SW480 cells. In our study, Rho kinase inhibitor, Y-27632, significantly inhibited cell migration and adhesion induced by LPA. LY294002 partially inhibited the LPA effect on adhesion. This indicated that the G12/13-Rho-GEFs-RhoA signal pathway may mediate the effect of LPA on both migration and adhesion, and that the PI3K-AKT/PKB pathway may partially mediate the LPA effect on adhesion.
Rho GTPase family proteins, including Rho, Rac1, and Cdc42, control a wide variety of cellular processes, such as cell adhesion, motility, proliferation, differentiation, and apoptosis[22]. One of the best effectors of Rho is Rho-associated kinase (ROCK). ROCK is a target effect molecule downstream of RhoA. Rho activates ROCK by phosphorylation of Ser854 and Thr697, and induces a series of actions downstream to stimulate adhesion and migration. Y-27632 is a novel and specific inhibitor of ROCK, which is cell permeable and inhibits ROCK-I and ROCK-II by competing with ATP.
It has been reported in other tumors that LPA drives the formation of focal adhesions, the tyrosine phosphorylation of focal adhesion proteins, and ROCK. Our results suggested that in colon cancer SW480 cells, the G12/13-Rho-GEFs-RhoA pathway may mediate migration and adhesion induced by LPA.
Several studies have also found that LPA promotes the metastasic potential of CRC.
Tatsuta et al[23] found that LPA significantly increased the peritoneal and pleural metastases of intestinal adenocarcinomas induced by azoxymethane through RhoA activation. This is consistent with our study, in which the Rho signaling pathway was found to be involved in the LPA effect on metastasis. Other mechanisms have also been found.
Shiada et al[24] found that cross-talk between LPA1 and epidermal growth factor receptors mediates up-regulation of sphingosine kinase 1 to promote gastric cancer cell motility and invasion. Down-regulation of SphK1 attenuated LPA-stimulated migration and invasion of MNK1 cells. Shiada et al[25] also reported that LPA acts as a potent stimulator of colon cancer progression, although the binding to LPA1 and LPA2 induced slightly different responses. Komuro et al[26] found that LPA1 expression was increased in the early stage of adenoma.
We also found that LPA significantly protected SW480 cells from apoptosis induced by chemotherapeutic drugs, while LY294002 and PD98059 effectively blocked the LPA effect on apoptosis, indicating that the PI3K-AKT/PKB and the Ras/Raf-MAPK signal pathways may mediate the LPA effect on apoptosis; especially the PI3K-AKT/PKB pathway. The apoptosis protection roles of LPA have been reported in ovarian cancer cells.
PI3K can be divided into three classes. Class I PI3K is the most studied class of PI3K, consisting of an 110 kDa catalytic subunit and a regulatory subunit of 85 kDa. The activity of PI3K protein family is associated with cytoskeletal organization, cell division, inhibition of apoptosis and glucose uptake. The phospholipid products of PI3K activate downstream targets, including PDK, Akt and PKC. LY294002 blocks PI3 kinase-dependent Akt phosphorylation and kinase activity. In SW480 cells we observed that the PI3K-AKT/PKB pathway may partially mediate the effect of LPA on proliferation.
In this study, it has been found that LPA significantly stimulated the proliferation, adhesion, and migration of human colon cells, SW480, and protected their apoptosis. The Ras/Raf-MAPK signal pathway may be involved in the LPA effect on proliferation. The G12/13-Rho-RhoA signal pathway may be associated with the LPA effect on adhesion and migration. The PI3K-AKT/PKB signal pathway may participate in the anti-apoptotic effect of LPA. This indicates that LPA probably acts as a promoter of the development of CRC. To decrease the LPA level in CRC patients and to block the LPA action (Figure 6) could be the aim of new strategies of treatment and prevention of CRC. The pathways involved in the LPA effects which we have discovered in this study could be new treatment targets of CRC.
Colorectal cancer (CRC) is a major cause of cancer death worldwide. The incidence has been rapidly increasing in recent years. Lysophosphatidic acid (LPA) was initially found in the ascites of ovarian cancer patients. Recently the authors group found that LPA levels increase not only in the body fluid of ovarian cancer patients, but also in the plasma of patients with CRC. There are many studies of LPA roles in ovarian cancer cells which have found that LPA stimulates the progression of ovarian cancer. Since LPA increases in the plasma of CRC patients as well, what are the roles of LPA in CRC? This study preliminarily investigated the roles of LPA in the proliferation, migration, adhesion and apoptosis of SW480 cells and its mechanisms of action.
LPA was firstly found in the ascitic fluid from ovarian cancer patients. It is a bioactive glycerophospholipid generated and released by platelets, macrophages, epithelial cells, and some tumor cells. Studies have shown the presence of high levels of LPA in the ascitic fluid of patients with ovarian cancer and LPA is known to be an “ovarian cancer activating factor”, which exerts growth factor-like effects through four specific G protein-coupled receptors (LPA1-4). LPA is a potent mediator with a broad range of cellular responses, including regulation of cell proliferation, protection from apoptosis, modulation of chemotaxis and transcellular migration, which also mediates survival of ovarian cancer cells, macrophages, fibroblasts, and neonatal cardiac myocytes. Some of these cellular responses indicate that LPA is a mediator of tumor progression.
Since LPA was found in the ascites of ovarian cancer patients, there have been many studies on LPA, but most of the studies have focused on ovarian cancer. There are some studies of LPA in colon cancer, but this present study has some different findings from other studies. Firstly, because the expression level of LPA receptor varies in different colon cancer cell lines, and different LPA receptors mediate different responses to LPA, so LPA effects on SW480 cells are different from other colon cancer cell lines. Secondly, some mechanisms found in this study are not completely the same as findings in other studies. They found some pathways which mediate the LPA effect on proliferation, migration, and adhesion which are different from other studies.
In order to completely block the growth, metastasis and progression of CRC, the mechanisms for its development need to be clarified. The provided some information about the LPA effects on colon cancer cells and some mechanisms of action. These results will help to design targeted strategies to block LPA’s stimulation effect on colon cancer.
LPA is associated with inflammation and has been thought to be one of the mediators of inflammation-induced promotion of cancer. LPA is one of the possible keys for inflammation-induced carcinogenesis in GI tract. Actually there are at least 511 papers on LPA and cell proliferation, among which there are nine papers on LPA and colon cancer. In this particular manuscript, the study was conducted carefully on many aspects including proliferation, apoptosis, cell adhesion, migration etc, of colon cancer cells. Title, abstract, methods and results were carefully written.
Peer reviewer: Shingo Tsuji, MD, PhD, AGAF, Professor, Department of Internal Medicine and Therapeutics, Osaka University Graduate School of Medicine (A8), 2-2 Yamadaoka, Suita, Osaka 565-0871, Japan
S- Editor Tian L L- Editor Logan S E- Editor Zheng XM
| 1. | Lu JB, Sun XB, Dai DX, Zhu SK, Chang QL, Liu SZ, Duan WJ. Epidemiology of gastroenterologic cancer in Henan Province, China. World J Gastroenterol. 2003;9:2400-2403. |
| 2. | Yiu HY, Whittemore AS, Shibata A. Increasing colorectal cancer incidence rates in Japan. Int J Cancer. 2004;109:777-781. |
| 3. | Erickson JR, Hasegawa Y, Fang X, Eder A, Mao M, Furui T, Aoki J, Morris A, Mills GB. Lysophosphatidic acid and ovarian cancer: a paradigm for tumorogenesis and patient management. Prostaglandins. 2001;64:63-81. |
| 4. | Ishii I, Fukushima N, Ye X, Chun J. Lysophospholipid receptors: signaling and biology. Annu Rev Biochem. 2004;73:321-354. |
| 5. | Panetti TS. Differential effects of sphingosine 1-phosphate and lysophosphatidic acid on endothelial cells. Biochim Biophys Acta. 2002;1582:190-196. |
| 6. | Zhao Z, Xiao Y, Elson P, Tan H, Plummer SJ, Berk M, Aung PP, Lavery IC, Achkar JP, Li L. Plasma lysophosphatidylcholine levels: potential biomarkers for colorectal cancer. J Clin Oncol. 2007;25:2696-2701. |
| 7. | Shida D, Watanabe T, Aoki J, Hama K, Kitayama J, Sonoda H, Kishi Y, Yamaguchi H, Sasaki S, Sako A. Aberrant expression of lysophosphatidic acid (LPA) receptors in human colorectal cancer. Lab Invest. 2004;84:1352-1362. |
| 8. | Kishi Y, Okudaira S, Tanaka M, Hama K, Shida D, Kitayama J, Yamori T, Aoki J, Fujimaki T, Arai H. Autotaxin is overexpressed in glioblastoma multiforme and contributes to cell motility of glioblastoma by converting lysophosphatidylcholine to lysophosphatidic acid. J Biol Chem. 2006;281:17492-17500. |
| 9. | Lee CW, Kim NH, Choi HK, Sun Y, Nam JS, Rhee HJ, Chun J, Huh SO. Lysophosphatidic acid-induced c-fos up-regulation involves cyclic AMP response element-binding protein activated by mitogen- and stress-activated protein kinase-1. J Cell Biochem. 2008;104:785-794. |
| 10. | Mukai M, Imamura F, Ayaki M, Shinkai K, Iwasaki T, Murakami-Murofushi K, Murofushi H, Kobayashi S, Yamamoto T, Nakamura H. Inhibition of tumor invasion and metastasis by a novel lysophosphatidic acid (cyclic LPA). Int J Cancer. 1999;81:918-922. |
| 11. | Hommes DW, Peppelenbosch MP, van Deventer SJ. Mitogen activated protein (MAP) kinase signal transduction pathways and novel anti-inflammatory targets. Gut. 2003;52:144-151. |
| 12. | Wang X, Wang Q, Hu W, Evers BM. Regulation of phorbol ester-mediated TRAF1 induction in human colon cancer cells through a PKC/RAF/ERK/NF-kappaB-dependent pathway. Oncogene. 2004;23:1885-1895. |
| 13. | Zhang H, Bialkowska A, Rusovici R, Chanchevalap S, Shim H, Katz JP, Yang VW, Yun CC. Lysophosphatidic acid facilitates proliferation of colon cancer cells via induction of Krüppel-like factor 5. J Biol Chem. 2007;282:15541-15549. |
| 14. | Yang M, Zhong WW, Srivastava N, Slavin A, Yang J, Hoey T, An S. G protein-coupled lysophosphatidic acid receptors stimulate proliferation of colon cancer cells through the {beta}-catenin pathway. Proc Natl Acad Sci USA. 2005;102:6027-6032. |
| 15. | Balavenkatraman KK, Jandt E, Friedrich K, Kautenburger T, Pool-Zobel BL, Ostman A, Böhmer FD. DEP-1 protein tyrosine phosphatase inhibits proliferation and migration of colon carcinoma cells and is upregulated by protective nutrients. Oncogene. 2006;25:6319-6324. |
| 16. | Hu YL, Albanese C, Pestell RG, Jaffe RB. Dual mechanisms for lysophosphatidic acid stimulation of human ovarian carcinoma cells. J Natl Cancer Inst. 2003;95:733-740. |
| 17. | Li H, Ye X, Mahanivong C, Bian D, Chun J, Huang S. Signaling mechanisms responsible for lysophosphatidic acid-induced urokinase plasminogen activator expression in ovarian cancer cells. J Biol Chem. 2005;280:10564-10571. |
| 18. | Fishman DA, Liu Y, Ellerbroek SM, Stack MS. Lysophosphatidic acid promotes matrix metalloproteinase (MMP) activation and MMP-dependent invasion in ovarian cancer cells. Cancer Res. 2001;61:3194-3199. |
| 19. | Symowicz J, Adley BP, Woo MM, Auersperg N, Hudson LG, Stack MS. Cyclooxygenase-2 functions as a downstream mediator of lysophosphatidic acid to promote aggressive behavior in ovarian carcinoma cells. Cancer Res. 2005;65:2234-2242. |
| 20. | Yamada T, Sato K, Komachi M, Malchinkhuu E, Tobo M, Kimura T, Kuwabara A, Yanagita Y, Ikeya T, Tanahashi Y. Lysophosphatidic acid (LPA) in malignant ascites stimulates motility of human pancreatic cancer cells through LPA1. J Biol Chem. 2004;279:6595-6605. |
| 21. | Boucharaba A, Serre CM, Guglielmi J, Bordet JC, Clézardin P, Peyruchaud O. The type 1 lysophosphatidic acid receptor is a target for therapy in bone metastases. Proc Natl Acad Sci USA. 2006;103:9643-9648. |
| 23. | Tatsuta M, Iishi H, Baba M, Uedo N, Ishihara R, Higashino K, Mukai M, Ishiguro S. Induction by lysophosphatidic acid of peritoneal and pleural metastases of intestinal cancers induced by azoxymethane in Wistar rats. Cancer Lett. 2005;219:137-145. |
| 24. | Shida D, Fang X, Kordula T, Takabe K, Lépine S, Alvarez SE, Milstien S, Spiegel S. Cross-talk between LPA1 and epidermal growth factor receptors mediates up-regulation of sphingosine kinase 1 to promote gastric cancer cell motility and invasion. Cancer Res. 2008;68:6569-6577. |
| 25. | Shida D, Kitayama J, Yamaguchi H, Okaji Y, Tsuno NH, Watanabe T, Takuwa Y, Nagawa H. Lysophosphatidic acid (LPA) enhances the metastatic potential of human colon carcinoma DLD1 cells through LPA1. Cancer Res. 2003;63:1706-1711. |
| 26. | Komuro Y, Watanabe T, Kitayama J, Yamaguchi H, Tsuno N, Nagawa H. The Immunohistochemical expression of endothelial cell differentiation gene-2 receptor in human colorectal adenomas. Hepatogastroenterology. 2003;50:1770-1773. |
| 27. | Fang JY, Richardson BC. The MAPK signalling pathways and colorectal cancer. Lancet Oncol. 2005;6:322-327. |