Published online Mar 6, 2021. doi: 10.12998/wjcc.v9.i7.1755
Peer-review started: November 30, 2020
First decision: December 13, 2020
Revised: December 18, 2020
Accepted: December 29, 2020
Article in press: December 29, 2020
Published online: March 6, 2021
Processing time: 91 Days and 1.1 Hours
Floating thrombus within the thoracic aorta is a rare entity but may cause systemic embolism. The pathogenesis of floating aortic thrombi is not yet fully understood. No definitive guidelines are available for the management of floating aortic thrombus.
We report a 48-year-old patient, without a history of trauma and infection, who presented with sudden severe back pain. A floating thrombus within the aortic arch was found by computed tomography angiography (CTA). No evidence of coagulopathies was found. However, with the assistance of a three dimensional-printed model, this floating thrombus was identified to be caused by occult aortic dissection (AD). Subsequently, an emergency thoracic endovascular repair was performed. The patient’s back pain was rapidly alleviated postoperatively. CTA at 1 year showed no filling defect in the stent-graft and aorta.
Occult AD is a potential factor causing floating aortic thrombi, endovascular stent-graft exclusion may be an optimal therapeutic choice with promising results. Moreover, the combination of CTA and three dimensional-printed models can contribute to the diagnosis and treatment of floating aortic thrombi due to occult AD.
Core Tip: Floating thrombus within the thoracic aorta is a rare but a life-threatening source of systemic emboli. The pathogenesis of floating aortic thrombi is not yet fully understood. Occult aortic dissection is a potential cause of floating aortic thrombus, which is most commonly detected at autopsy. Misdiagnosis may cause repeated recurrence of floating thrombus. We report a floating aortic thrombus caused by occult aortic dissection identified with the assistance of a three-dimensional printed model. Thoracic endovascular repair was the optimal treatment method in this patient, with promising results. Similar cases have not previously been reported.
- Citation: Wang TH, Zhao JC, Xiong F, Yang Y. Use of three dimensional-printing in the management of floating aortic thrombus due to occult aortic dissection: A case report. World J Clin Cases 2021; 9(7): 1755-1760
- URL: https://www.wjgnet.com/2307-8960/full/v9/i7/1755.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i7.1755
A floating thrombus within the thoracic aorta is rare and most commonly detected at autopsy[1]. With the frequent use of imaging over the past decades, floating thrombi are increasingly being identified as a cause of systemic embolism[2,3]. The pathogenesis of floating aortic thrombi is not yet fully understood. Hematological disorders have been associated with the formation of aortic thrombi, and diseases of the aorta itself (e.g., atherosclerosis, inflammatory vasculopathies, and dissection) have been suggested as the dominant causative factors[1,4,5]. Treatment options consist of anticoagulation, surgical thrombectomy, and endovascular treatment, but no definitive guidelines are available[6].
Some authors have reported that occult aortic dissection (AD) may lead to the formation of floating aortic thrombi, but diagnoses are usually delayed until they are confirmed by surgical thrombectomy[4,7]. Early identification of the etiology of floating thrombi can assist in treatment selection[1]. We report a rare case of a floating aortic thrombus due to occult AD, which was demonstrated by the combination of computed tomography angiography (CTA) and three-dimensional (3D) printing. The thrombus was successfully managed by endovascular treatment.
A 48-year-old male presented to our emergency department with sudden severe back pain without a history of trauma and infection.
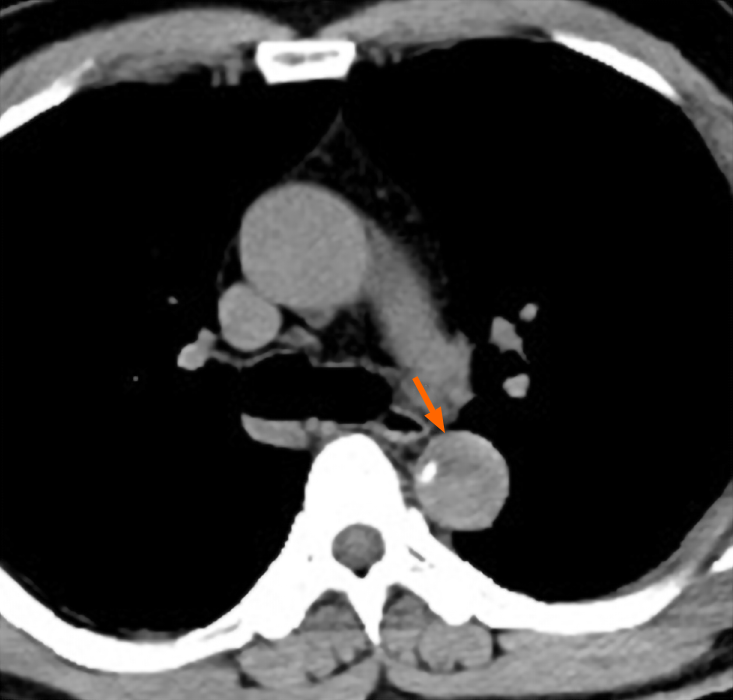
The patient, initially presenting with sudden severe back pain for about three day, was admitted to a local hospital. No abdominal pain, limb pain, syncope, or other neurologic changes occurred at this time. A chest computed tomography (CT) scan showed space occupying and calcification within the lumen of thoracic aorta (Figure 1). His back pain persisted after sedative and analgesic treatment. He was immediately transferred to our institution.
The patient was previously healthy, and had no history of trauma, infection, hypertension, or diabetes mellitus.
He denied a history of alcohol consumption and smoking.
Upon admission, his heart rate was 89 bpm, blood pressure was 123/70 mmHg, and respiratory rate was 21 breaths/min. Physical examination showed normal mental status, and no focal neurologic changes were noted. Abdominal tenderness and rebound tenderness were not detected. All peripheral pulses were present, and no carotid or subclavian bruit was observed.
The laboratory tests yielded a troponin T value of 5.7 ng/L, brain natriuretic peptide of 162 pg/mL, blood leukocyte count of 6.7 × 109/L, creatinine level of 78.2 µmol/L, alanine aminotransferase of 37.5 IU/L, and aspartate aminotransferase of 43.9 IU/L. Thrombin, prothrombin, and partial thromboplastin times, antithrombin III, protein C and S antigens, lupus anticoagulant, and anticardiolipin antibodies were examined and found to be within the normal limits. No evidence of coagulopathies was found. The electrocardiogram was unremarkable.
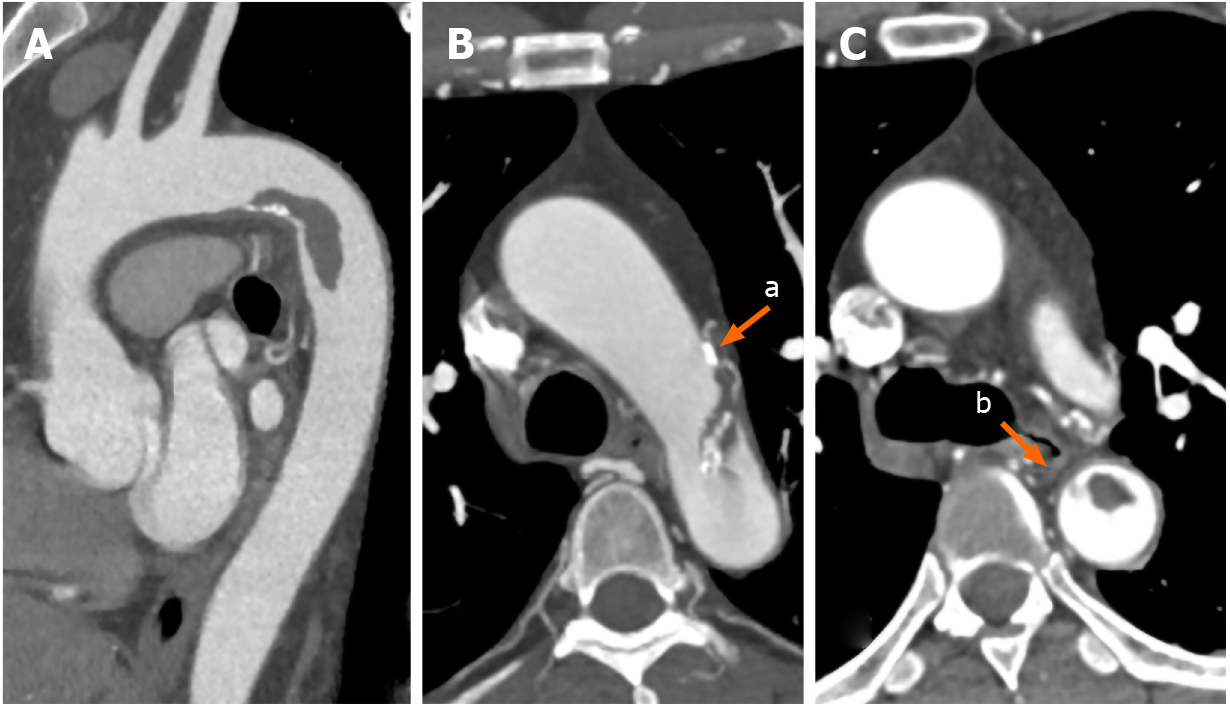
CTA revealed a hypermobile pedunculated thrombus attached to the lesser curve of the aortic arch and extending to the descending aorta (Figure 2A). Multiple calcified plaques were located at the aortic arch, and some calcified plaques were within the floating thrombus (Figure 2B). The floating thrombus was connected to the aortic wall by a pedicle (Figure 2C). The visceral, carotid and main peripheral arteries were patent.
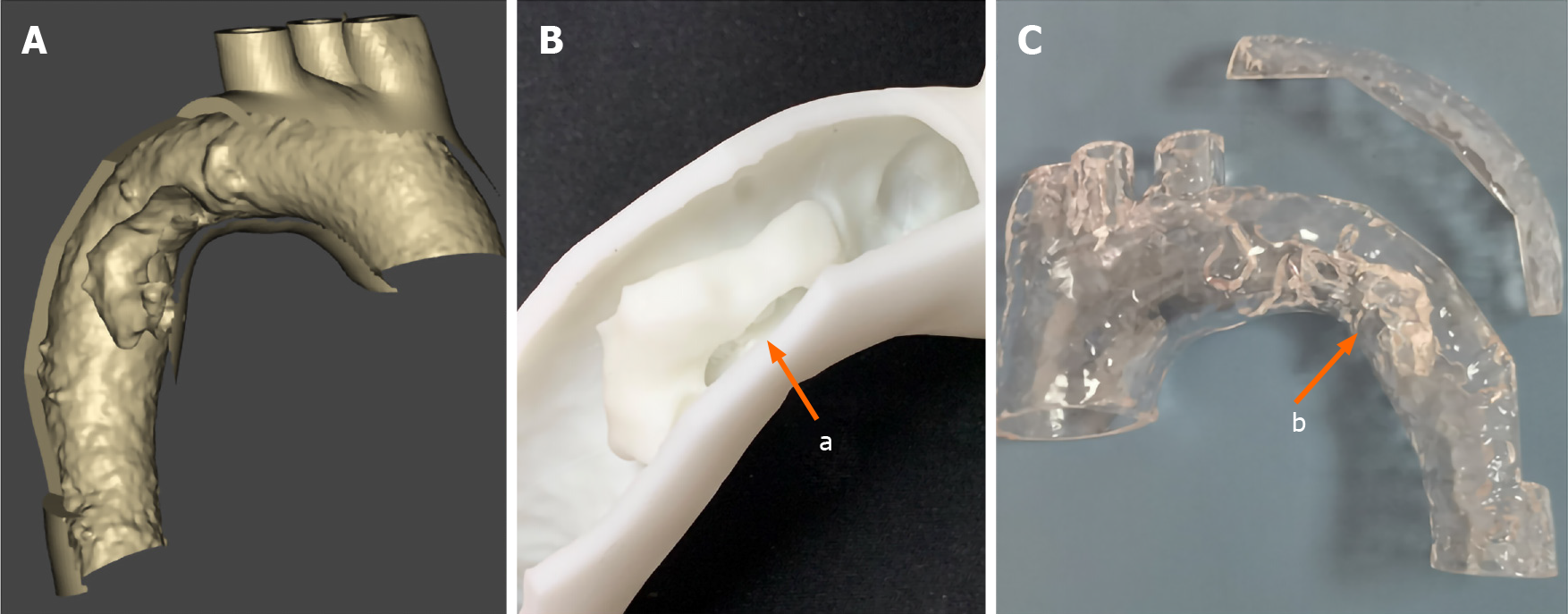
However, the etiology of this floating aortic thrombus remained unknown, and the CTA images were insufficiently clear as to make a definitive diagnosis. Subsequently, a 3D-printed model of the aorta and floating thrombus were made according to the imaging data. Using this model, a longitudinal thrombus in the lumen and an intimal defect in the adjacent aortic wall were found. Moreover, a longitudinal intimal flap connected the floating thrombus and adjacent abnormal aortic wall (Figure 3). Based on these findings, an occult AD resulting in a floating aortic thrombus was identified.
The final diagnosis in this patient was a floating aortic thrombosis resulting from occult AD, accompanied by slight aortic atherosclerosis.
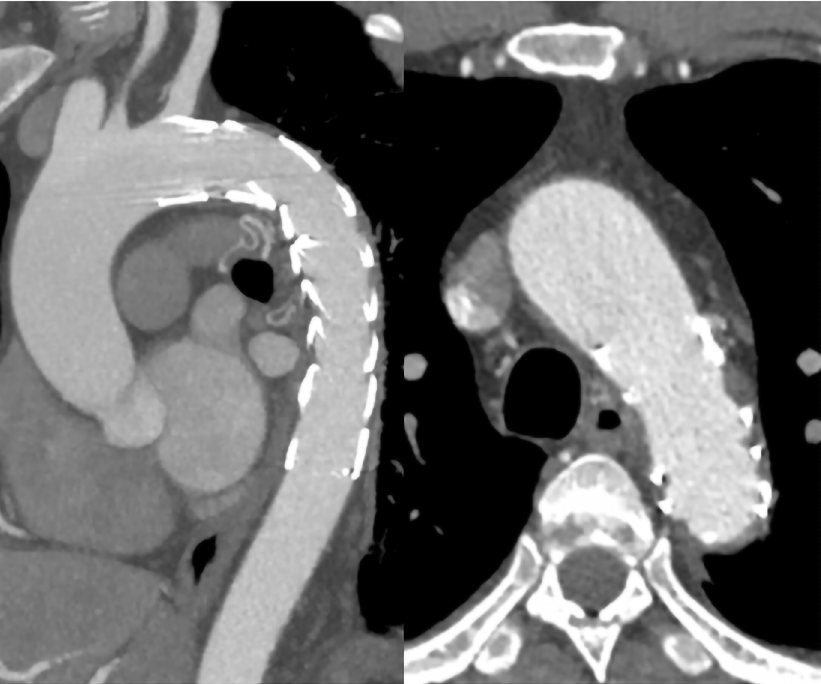
Systemic anticoagulation with heparin was performed upon verification of the floating aortic thrombosis. Due to the morphology of the thrombus and occult AD shown in the CTA and 3D-printed model, the decision for an emergency thoracic endovascular repair (TEVAR) was made. Right femoral accesses were obtained using a pre-close technique (ProGlide; Abbott Vascular, United States). With the assistance of the 3D-printed model, a suitable stent-graft (VAMF2626C150TE; Medtronic, United States) was selected and deployed from the distal part of the aortic arch to the descending aorta. The final angiogram revealed that the floating thrombus and dissection were completely covered.
The postoperative recovery of this patient was uneventful. No physical signs of systemic embolism were found, and his back pain was rapidly alleviated. He was discharged on postoperative day 3 with antiplatelet therapy. At 1 year of follow-up, CTA showed a well-formed stent graft, and no filling defect was found in the stent-graft and thoracic aorta (Figure 4).
Given the rarity of a floating aortic thrombus, few reports are available regarding its pathogenesis, diagnosis, and treatment[1]. The formation of a floating aortic thrombus, especially in the thoracic segment, creates a life-threating risk of stroke, as well as peripheral embolization[3]. Many risk factors such as atherosclerosis, trauma, coagulopathies, malignancy, and aortitis have been associated with floating aortic thrombi[1,2,8]. However, in patients with sudden severe back pain, the detection of a floating aortic thrombus should raise suspicion for occult AD[4]. Occult dissection is characterized by a stellate or linear intimal tear associated with exposure of the underlying aortic media but without the progression and separation of the medial layers, resulting in extensive undermining of the intimal layers. The inability to identify this occult AD may confer a dismal prognosis, such as progression of dissection, aortic rupture, or thrombosis[7,9]. In patients with intimal tears, the injured aortic wall or free intimal flap may induce thrombosis to form a floating thrombus[4].
Transesophageal echocardiography, CTA, and magnetic resonance angiography (MRA) are considered the optimal techniques to detect and characterize floating thrombi in thoracic aortic lesions[1]. Transesophageal echocardiography can be rapidly performed for hemodynamic unstable patients in emergency situations, but the portion of the aortic arch cannot be adequately visualized due to interference from the trachea. The process of MRA is time consuming and cannot be used in hemodynamic unstable patients. Moreover, severe artifacts of the aortic wall and thrombus are revealed by MRA. CTA is the most extensively used imaging technique for the assessment of AD and aortic mural thrombi[2,3]. However, the reconstruction of CTA imaging requires a computer screen for viewing, which is limited by an overall lack of realism[10]. Zurick et al[4] reported that CTA showed a floating aortic thrombus without aortic aneurysm or dissection, but the open repair and subsequent histopathology noted it was caused by occult AD.
In our case, the reconstruction images of CTA showed a floating thrombus originating from the aortic wall, and some calcified plaques within the thrombus (Figure 2A and B), indicating that this floating thrombus was derived from the mural thrombus of the aortic wall. However, sectional images showed a linear intimal flap connecting the aortic wall and thrombus along the long axis (Figure 2C). These different images enabled the distinctly different diagnosis in our patient. A 3D-printed model demonstrated a huge thrombus attached to the floating intimal flap and impaired aortic wall, which were caused by occult AD. Compared with computer-generated reconstruction, a 3D-printed model elucidates the complex vascular details and structural abnormalities. Individualized 3D-printed models can assist clinical diagnosis and selection of the optimal treatment strategy[11]. Therefore, a 3D-printed model is crucial for detecting the etiology of floating aortic thrombus, and it also plays an important role in the treatment of floating aortic thrombus caused by occult AD.
The best treatment for floating aortic thrombi is controversial[3]. Although thrombolysis has been proposed, a floating thrombus in the thoracic aorta carries a high risk of embolization. The choice of therapeutic method depends on the etiology of the floating aortic thrombus. Nonetheless, the etiology of floating aortic thrombi is relatively enigmatic and often deemed to be the result of hypercoagulation or abnormalities of the aortic wall[1]. The first-line therapy for an aortic mural thrombus is anticoagulation, and that for a hypermobile thrombus is surgical thrombectomy[12]. However, simplified thrombectomy cannot resolve the abnormalities of the aortic wall, and open surgical extirpation of the thoracic aorta is accompanied by extensive trauma. Given that the thrombus and aortic abnormalities can be completely covered with a stent graft, TEVAR provides a minimally invasive option to treat a floating aortic thrombus[4,5,13]. Our patient had a huge floating aortic thrombus caused by occult AD according to the 3D-printed model, so simplified surgical thrombus removal from the aorta may result in recurrent aortic thrombosis or progression of dissection. Recent studies have shown that endovascular treatment for a floating aortic thrombus has high technical success and no recurrences[1,3,6]. Moreover, TEVAR has been used as the standard treatment for Type-B AD, and a 3D-printed model can provide a simulation for the selection of a suitable stent-graft before the endovascular procedure. Therefore, stent grafting was the reasonable choice for this patient, and CTA at 1-year follow-up showed no filling defect in the aorta and stent-graft.
A floating aortic thrombus is an under-recognized but a life-threatening source of cerebral, visceral, and peripheral emboli. Occult AD is a potential cause of floating aortic thrombi, which can be verified by the combination of CTA and a 3D-printed model. Treatment should be performed using a patient-specific approach. According to the 3D-printed model, TEVAR was the optimal treatment method for this patient, with promising results.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, research and experimental
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Azzam AZ S-Editor: Zhang L L-Editor: Webster JR P-Editor: Ma YJ
| 1. | Weiss S, Bühlmann R, von Allmen RS, Makaloski V, Carrel TP, Schmidli J, Wyss TR. Management of floating thrombus in the aortic arch. J Thorac Cardiovasc Surg. 2016;152:810-817. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 42] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 2. | Klang E, Kerpel A, Soffer S, Zlotnik M, Shimon O, Rimon U, Konen E, Amitai MM. CT imaging features of symptomatic and asymptomatic floating aortic thrombus. Clin Radiol 2018; 73: 323.e9-323. e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 3. | Borghese O, Pisani A, Di Centa I. Symptomatic Aortic Mural Thrombus Treatment and Outcomes. Ann Vasc Surg. 2020;69:373-381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 4. | Zurick AO, Ramaiah C. Aortic Mural Thrombus in Association with Occult Aortic Dissection. CASE (Phila). 2017;1:62-64. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 5. | Baldini U, Chiaramonti F, Minzioni G, Galli M. A hanging mass in the ascending aorta diagnosed by transthoracic echocardiography in a totally asymptomatic subject. J Am Soc Echocardiogr 2009; 22: 1197.e5-1197. e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 6. | Yoon HJ, Jeong MH, Oho SK, Jeong IS, Ahn BH, Kim CS, Lee MG, Ko JS, Park KH, Sim DS, Yoon NS, Park HW, Hong YJ, Kim KH, Kim JH, Ahn Y, Cho JG, Park JC, Kang JC. Successful management of huge floating thrombus within aortic arch in a patient with old myocardial infarction. J Cardiol Cases. 2010;2:e1-e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 7. | Svensson LG, Labib SB, Eisenhauer AC, Butterly JR. Intimal tear without hematoma: an important variant of aortic dissection that can elude current imaging techniques. Circulation. 1999;99:1331-1336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 182] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 8. | Sosa C, Zuccarino F. Floating Aortic Thrombus in a Non-aneurysmal and Non-atherosclerotic Aorta. Eur J Vasc Endovasc Surg. 2018;55:491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 9. | Mussa FF, Horton JD, Moridzadeh R, Nicholson J, Trimarchi S, Eagle KA. Acute Aortic Dissection and Intramural Hematoma: A Systematic Review. JAMA. 2016;316:754-763. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 384] [Cited by in RCA: 334] [Article Influence: 37.1] [Reference Citation Analysis (0)] |
| 10. | Pergolini A, Zampi G, Tinti MD, Vallone A, Pino PG. Floating thrombus in the false lumen: an unusual finding in aortic dissection. Kardiol Pol. 2015;73:782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 11. | Ho D, Squelch A, Sun Z. Modelling of aortic aneurysm and aortic dissection through 3D printing. J Med Radiat Sci. 2017;64:10-17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 46] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 12. | Lopez S, Tarmiz A, Rousseau H, Fournial G. Floating aortic thrombus: aortic trauma treated by heparin and delayed covered stent. Ann Vasc Surg 2011; 25: 984.e1-984. e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 13. | Scott DJ, White JM, Arthurs ZM. Endovascular management of a mobile thoracic aortic thrombus following recurrent distal thromboembolism: a case report and literature review. Vasc Endovascular Surg. 2014;48:246-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |