Published online Feb 26, 2021. doi: 10.12998/wjcc.v9.i6.1424
Peer-review started: October 13, 2020
First decision: November 24, 2020
Revised: December 8, 2020
Accepted: December 23, 2020
Article in press: December 23, 2020
Published online: February 26, 2021
Processing time: 115 Days and 20.3 Hours
Leiomyoma of the uterus is relatively common, but uterine leiomyoma of the greater omentum is rare.
Here, we report the case of a 22-year-old woman who presented with a 3 mo history of progressive abdominal distension and a hypervascular abdominopelvic mass. Due to a high serum concentration of CA125, the preoperative diagnosis was unclear. During surgery, 5 L of ascites was removed. An 18.8 cm solid mass, which was pedunculated from the uterine fundus and exhibited complex adhesion to the greater omentum, was removed. The CA125 level was reduced postoperatively, and a pathologic study confirmed that the mass was a leiomyoma that originated in the uterus.
Uterine leiomyoma can share vessels with the greater omentum. This case highlights the difficulty of diagnosing pseudo-Meigs syndrome and the importance of imaging and laboratory examinations.
Core Tip: We report a case of uterine leiomyoma of the greater omentum with a high serum concentration of CA125 and pseudo-Meigs syndrome. It is rarely seen in this condition. Upon entering the abdomen, up to 5000 mL of yellow ascites was removed by suction. A large solid cystic neoplasm that measured almost 20 cm was found and diagnosed as leiomyoma pathologically later.
- Citation: Wang YW, Fan Q, Qian ZX, Wang JJ, Li YH, Wang YD. Abdominopelvic leiomyoma with large ascites: A case report and review of the literature. World J Clin Cases 2021; 9(6): 1424-1432
- URL: https://www.wjgnet.com/2307-8960/full/v9/i6/1424.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i6.1424
Uterine leiomyomas are common benign smooth muscle tumors that affect up to 80% of women[1,2]. Common uterine leiomyomas principally result from the estrogen- and progesterone-dependent transformation of uterine smooth muscle[3]. Affected women may complain of bleeding, pain, pressure, or infertility, but few eventually develop uterine sarcoma[4,5].
Meigs syndrome is a clinical triad whose pathology is limited to ovarian fibroma, fibrothecoma, Brenner tumor, and granulosa cell tumor[6,7] and is a syndrome characterized by ascites and hydrothorax. Pseudo-Meigs syndrome is defined as a disorder similar to Meigs syndrome, in which neoplasms of different histological types, including uterine leiomyomas, are present[8-10]. Uterine leiomyomas that present as pseudo-Meigs syndrome have already been described in the literature[11-14]. However, few cases of pseudo-Meigs syndrome in which a leiomyoma is implanted on the omentum have been reported[15]. In this study, we report a massive abdominopelvic leiomyoma with large ascites in a 22-year-old woman.
A 22-year-old woman presented to the outpatient unit with abdominal pain.
The patient was not married and had never been pregnant. She reported no recent trauma, fall, or sexual intercourse. She reported a normal menstrual cycle with a menstrual period 18 d prior to her visit. The patient also reported a 3 mo history of increased abdominal circumference, which she considered a “pot belly”.
A review of systems was notable for findings of abdominal pain and a lack of appetite, but the woman denied clinical indications of constipation, diarrhea, vaginal bleeding, nausea, emesis, or urinary urgency or frequency.
The abdomen was gravid, soft, and firm. A rectovaginal examination revealed a movable mass at the front of the uterus.
The patient’s laboratory test results are shown in Table 1. Unexpectedly, the CA125 level was elevated to 569.5 U/mL.
| Variable | Reference range, IPMCH1 | 1 d after admission | 1 d after operation | 1 d before discharge |
| Hematocrit (%) | 33.5-45 | 38.8 | 25.8↓ | 25.7↓ |
| Hemoglobin (g/L) | 113-151 | 133 | 85↓ | 85↓ |
| White cell count (10^9/L) | 3.69-9.16 | 6.5 | 15.0↑ | 6.9 |
| Platelet count (10^9/L) | 101-320 | 337↑ | 188 | 500↑ |
| Sodium (mmoL/L) | 135-145 | 138 | 135 | 140 |
| Potassium (mmoL/L) | 3.5-5.5 | 4.37 | 4.15 | 4.75 |
| Chloride (mmoL/L) | 95-110 | 96 | 103 | 99 |
| Creatinine (μmoL/L) | 45-84 | 48 | 46 | 41 |
| Albumin (g/L) | 40-55 | 47.5 | 23.3↓ | 45.5 |
| Alanine aminotransferase (U/L) | 0-42 | 25 | 10 | 8 |
| Aspartate transaminase (U/L) | 0-42 | 32 | 17 | 17 |
| Prothrombin time (sec) | 11-14.5 | 13.0 | 14.7↑ | 14.1 |
| International normalized ratio | 0.95-1.125 | 1.00 | 1.17 | 1.11 |
| Activated partial thromboplastin time (sec) | 28-45 | 39.5 | 46.6↑ | 51.5↑ |
| D-dimer (mg/L) | 0-1.0 | 3.19↑ | 5.81↑ | 3.02↑ |
| CEA (μg/L) | < 5.2 | 0.8 | 0.8 | 0.8 |
| CA125 (U/mL) | < 35 | 569.5↑ | 192.3↑ | 162↑ |
| CA199 (U/mL) | < 35 | 13.7 | 8.7 | 10.2 |
| AFP (μg/L) | < 7.0 | 2.0 | 1.5 | 1.6 |
| BMI | 19-25 | 19.5 | 15.8 | 16.2 |
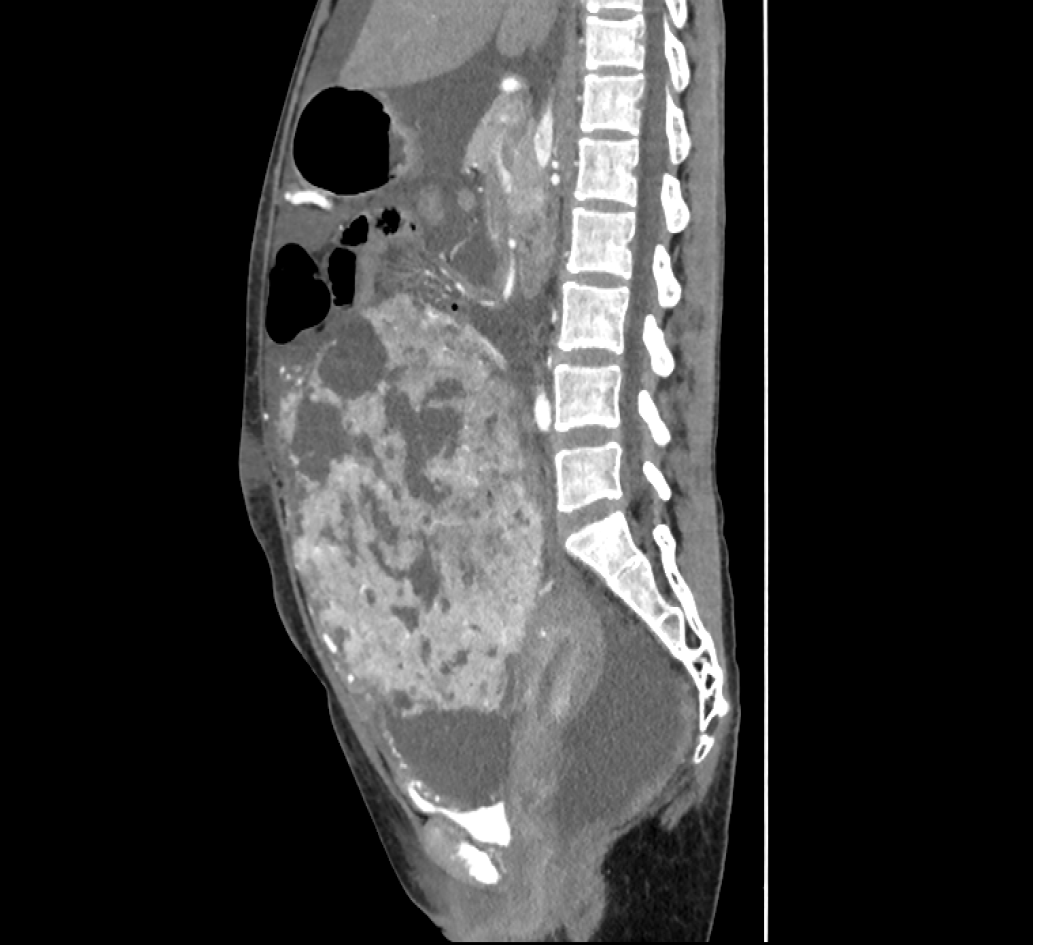
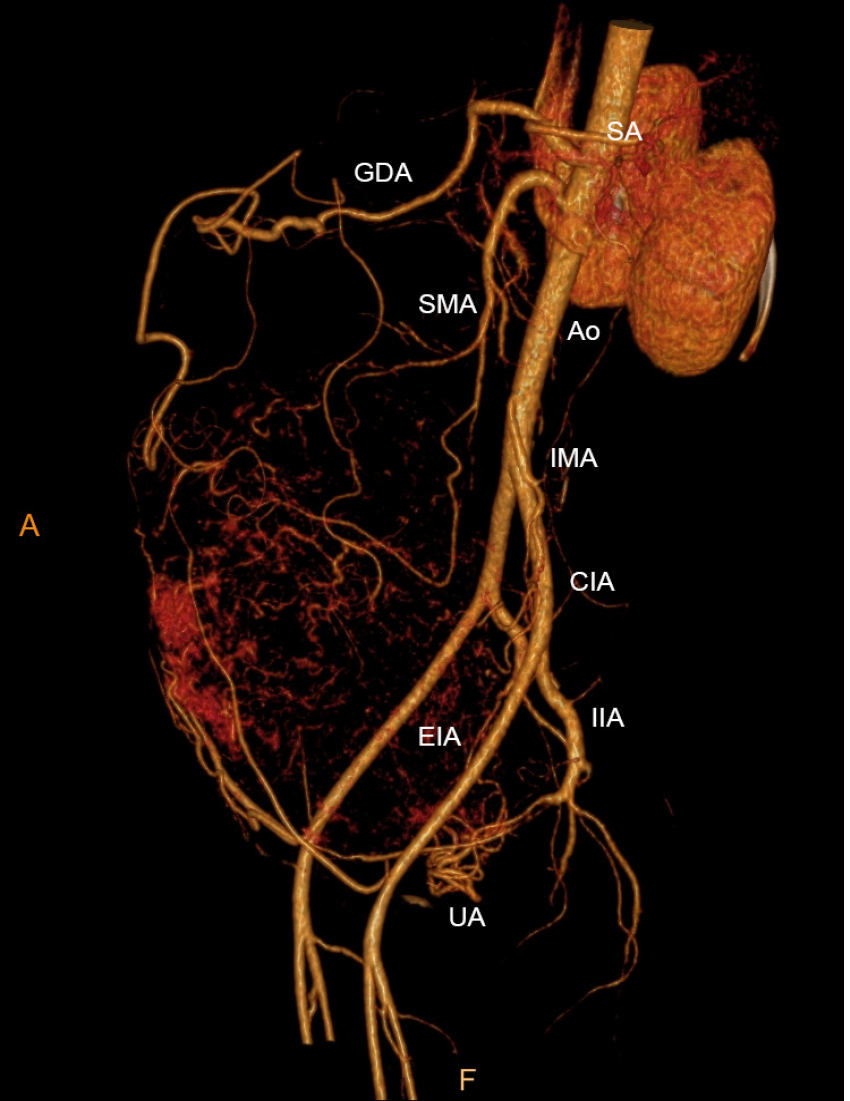
Computed tomography (CT) of the pelvis was performed after administration of an intravenous contrast agent (Figure 1), and magnetic resonance imaging (MRI) (data not shown) revealed a large mass with a maximum cross-section of 18.8 cm × 13.5 cm. Massive abdominal edema was also recorded. No evidence of swollen lymph nodes was found. Computed tomography angiography (CTA) revealed an abundant blood supply to the mass, which was provided by the uterine artery, superior mesenteric artery, and predominantly, by the gastroduodenal artery (Figure 2).
The mass was determined to be a uterine-derived leiomyoma that had implanted on the omentum majus.
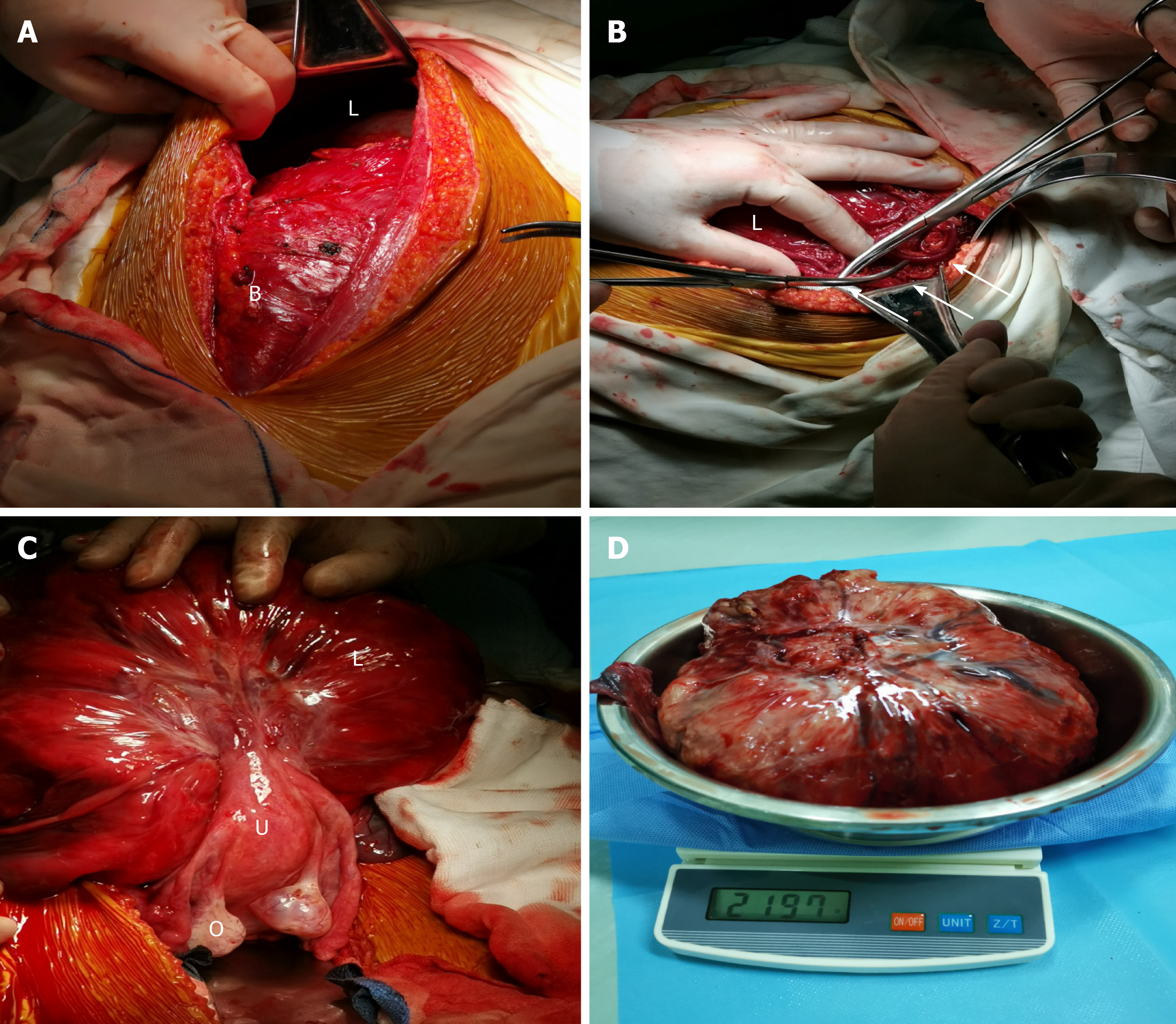
The patient underwent abdominal aortic balloon catheter placement in case of hemorrhage and later underwent exploratory laparotomy under combined spinal–epidural anesthesia. Upon entering the abdomen, up to 5000 mL of yellow ascites was removed by suction (Figure 3A). Ascitic fluid cytology revealed aggregated cells with inconspicuous atypia. A large solid cystic neoplasm that measured almost 20 cm was also found (Figure 3B). In addition, careful inspection after ligation of the omental vessels showed that the omental fat had vanished and that vessels from the greater omentum, which were shared with the mass, were engorged. Adhesions between the mass and the ileocecum, appendix, and descending colon were observed. The mass was found to be attached to the uterine fundus by a 3-cm-wide pedicle (Figure 3C). Resection of the mass and appendix was then performed (Figure 3D), and during surgery, the total blood loss was 1200 mL.
The patient was transferred to recovery in a stable condition, and on postoperative day 8, she was discharged from the hospital.
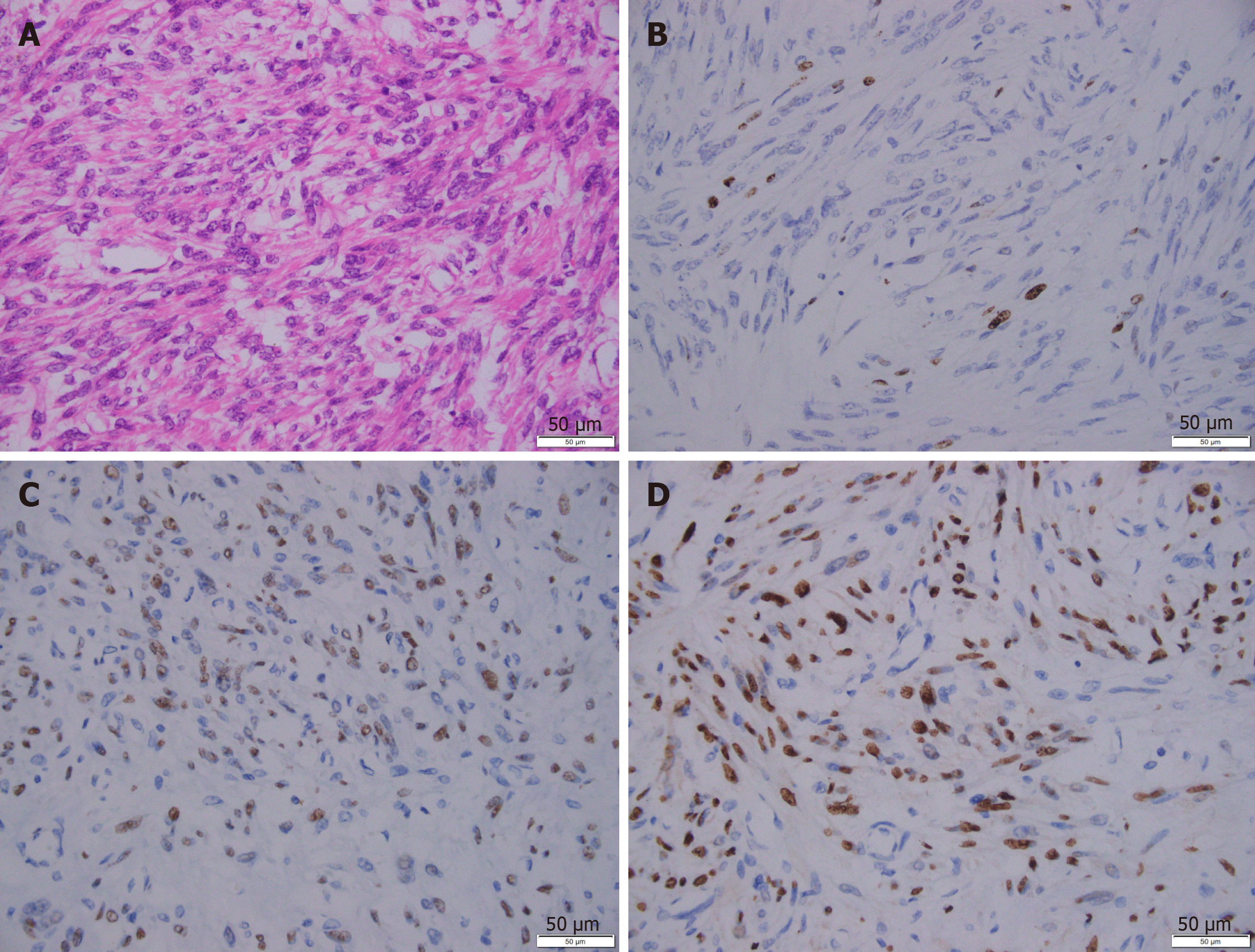
A pathologic study of the specimen confirmed a diagnosis of leiomyoma. Hydropic degeneration, hyaline degeneration, and cystic degeneration were recorded. An immunohistochemical analysis showed that the leiomyoma cells were positive for progesterone receptor, estrogen receptor, h-caldesmon, and smooth muscle actin (Figure 4). The tissue was negative for CD10, CD117, HMB45, Dog-1, and Ki67. The mass was determined to be a uterine-derived leiomyoma that had implanted on the omentum majus.
The unique features of the presented case can be summarized as follows: A massive leiomyoma with the “digested” greater omentum and an elevated CA125 level accompanied by pseudo-Meigs syndrome. Uterine leiomyoma is the most common benign gynecological neoplasm[16]. However, elevated CA125 levels, a massive leiomyoma, and large ascites, which are collectively known as pseudo-Meigs syndrome, are rarely seen in this condition[11-14,17-20]. In addition, no agreement on the pathogenesis of this rare condition has been established.
In this case, the increased abdominal circumference was considered to have resulted from pleural effusion and ascites. The pathogenesis of ascites in pseudo-Meigs syndrome is still not well defined[21]. According to several hypotheses, the release of inflammatory cytokines and growth factors may cause vascular permeability[22-24]. An imbalance among arterial flow, venous drainage, and direct pressure of the mass itself on lymphatic vessels may have caused the accumulation of nearly 5 L of ascites fluid in this case[25].
Removal of the large leiomyoma could resolve the ascites, but it is difficult to perform a complete excision when a leiomyoma is broadly adhered to adjacent structures. Full preparation, including detailed information about the size of the mass and its properties, could help in the design of a better surgical strategy[26]. Specifically, various imaging techniques, including CT and MRI, were used in this case, and both modalities indicated an 18-cm mass with abdominal edema.
CTA confirmed an abundant blood supply to the mass. During the surgery, distended blood vessels from the omentum (top), uterus (bottom), and colon (lateral) were observed and carefully ligated. Notably, the uterovesical peritoneal reflection was lifted, which was evidently due to the massive leiomyoma. Thus, preparations should have been made to reduce the risk of injury and to prevent uncontrolled bleeding or incomplete resection. Therefore, we performed abdominal aortic balloon catheter placement before surgery[27].
The elevated CA125 level is another complicating factor in this case. It is well known that as a high molecular weight glycoprotein, CA125 is expressed in a substantial proportion of ovarian cancers[28]. It has been reported that the serum CA125 levels range from 20 to over 1000 IU/mL in patients with leiomyomas[29-31]. Due to the significant overlap between malignancies and myomas, CA125 failed to provide a potential benefit for differential diagnosis[32]. The destruction of the peritoneum and severe adhesions of the omentum could explain why CA125 was overexpressed before surgery and reduced after surgery[33,34].
A preoperative diagnosis of a pelvic mass with pseudo-Meigs syndrome remains very challenging, especially in cases such as the one presented in this study. In these cases, an accurate diagnosis may only be made after surgery. For oncological safety, patients with myomas may undergo hysterectomy or ovariectomy, which impairs reproductive function. On the contrary, an overly optimistic presumption of a benign leiomyoma may lead to the intra-abdominal spread of an unexpected sarcoma, which could result in poor survival. Therefore, it is always important to consider a broad differential diagnosis and employ the necessary scrutiny using imaging techniques. Uterine sarcomas are rare gynecological neoplasms, and some researchers believe that sarcomas originate from benign leiomyomas because they contain the same epithelial progenitor cells[35]. Currently, no diagnostic technique is available to accurately distinguish this neoplasm. A meta-analysis revealed that the expression of CA125, lactate dehydrogenase (LDH), and GDF-15 proteins is altered in sarcomas and that their serum levels are statistically higher[35]. In our case, the CA125 level was elevated. However, we interpreted this finding and believed that the higher level was due to the implantation of the leiomyoma on the omentum rather than as a cancer-related signal. Deficiency of LDH and GDF-15 levels was not assessed in our case. In addition, MRI may help in the preoperative distinction of sarcomas from benign leiomyomas[36]. In our case, MRI revealed no swollen lymph nodes. However, the abundant blood supply to the mass observed on CTA imaging still roused our suspicion of a malignancy. Adenomyosis is another differential diagnosis of leiomyoma. A recent study has shown that MRI and elevated CA125 levels are fairly accurate for the diagnosis of adenomyosis, with a high sensitivity and specificity[29]. A mean serum CA125 level of 19 U/mL has been established as the cutoff value. We speculated that this cutoff did not apply to our case because of the size of the mass and the tight adhesion. Preoperative MRI revealed a suspected abdominopelvic mass with large ascites, which did not support the diagnosis of adenomyosis.
In conclusion, we have presented an unusual case of pseudo-Meigs syndrome with a uterine leiomyoma and an elevated CA125 level. CTA was used for the detection and ligation of the parasitic blood supply (in this case, mainly the blood vessels of the greater omentum). Surgeons should be aware of the malignant potential of this tumor and plan for the optimal surgical procedure, which would lead to a more appropriate treatment strategy.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, research and experimental
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Vij M S-Editor: Fan JR L-Editor: Wang TQ P-Editor: Li JH
| 1. | Marsh EE, Ekpo GE, Cardozo ER, Brocks M, Dune T, Cohen LS. Racial differences in fibroid prevalence and ultrasound findings in asymptomatic young women (18-30 years old): a pilot study. Fertil Steril. 2013;99:1951-1957. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 111] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 2. | Babacan A, Kizilaslan C, Gun I, Muhcu M, Mungen E, Atay V. CA 125 and other tumor markers in uterine leiomyomas and their association with lesion characteristics. Int J Clin Exp Med. 2014;7:1078-1083. [PubMed] |
| 3. | Commandeur AE, Styer AK, Teixeira JM. Epidemiological and genetic clues for molecular mechanisms involved in uterine leiomyoma development and growth. Hum Reprod Update. 2015;21:593-615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 129] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 4. | Quade BJ, Wang TY, Sornberger K, Dal Cin P, Mutter GL, Morton CC. Molecular pathogenesis of uterine smooth muscle tumors from transcriptional profiling. Genes Chromosomes Cancer. 2004;40:97-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 108] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 5. | Skubitz KM, Skubitz AP. Differential gene expression in leiomyosarcoma. Cancer. 2003;98:1029-1038. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 86] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 6. | Kawakubo N, Okido M, Tanaka R, Mitsugi K, Fukuhara M, Aishima S, Kato M, Ichimiya H. Pseudo-Meigs' syndrome associated with breast cancer metastasis to both ovaries: Report of a case. Surg Today. 2010;40:1148-1151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 7. | Meigs JV, Armstrong SH, Hamilton HH. A further contribution to the syndrome of fi broma of the ovary with fluid in the abdomen and chest: Meigs' syndrome. Am J Obstet Gynecol. 1943;46:19-37. |
| 8. | Schmitt R, Weichert W, Schneider W, Luft FC, Kettritz R. Pseudo-pseudo Meigs' syndrome. Lancet. 2005;366:1672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 9. | MEIGS JV. Pelvic tumors other than fibromas of the ovary with ascites and hydrothorax. Obstet Gynecol. 1954;3:471-486. [PubMed] |
| 10. | Kyo K, Maema A, Shirakawa M, Nakamura T, Koda K, Yokoyama H. Pseudo-Meigs' syndrome secondary to metachronous ovarian metastases from transverse colon cancer. World J Gastroenterol. 2016;22:4604-4609. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 7] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 11. | Tsao KC, Hong JH, Wu TL, Chang PY, Sun CF, Wu JT. Elevation of CA 19-9 and chromogranin A, in addition to CA 125, are detectable in benign tumors in leiomyomas and endometriosis. J Clin Lab Anal. 2007;21:193-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 12. | Yaguchi A, Ban K, Koshida Y, Fujikami Y, Ogura E, Terada A, Akagi K, Matsumoto H, Tobiume T, Okagaki A, Tatsumi K. Pseudo-Meigs Syndrome Caused by a Giant Uterine Leiomyoma with Cystic Degeneration: A Case Report. J Nippon Med Sch. 2020;87:80-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 13. | Dong R, Jin C, Zhang Q, Yang X, Kong B. Cellular leiomyoma with necrosis and mucinous degeneration presenting as pseudo-Meigs' syndrome with elevated CA125. Oncol Rep. 2015;33:3033-3037. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 14. | Buckshee K, Dhond AJ, Mittal S, Bose S. Pseudo-Meigs' syndrome secondary to broad ligament leiomyoma: a case report. Asia Oceania J Obstet Gynaecol. 1990;16:201-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 15. | Oguma T, Yamasaki N, Nakanishi K, Kinoshita D, Mitsuhashi T, Nakagawa S. Pseudo-Meigs' syndrome associated with hydropic degenerating uterine leiomyoma: a case report. J Obstet Gynaecol Res. 2014;40:1137-1140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 16. | Styer AK, Rueda BR. The Epidemiology and Genetics of Uterine Leiomyoma. Best Pract Res Clin Obstet Gynaecol. 2016;34:3-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 59] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 17. | Kebapci M, Aslan O, Kaya T, Yalcin OT, Ozalp S. Pedunculated uterine leiomyoma associated with pseudo-Meigs' syndrome and elevated CA-125 level: CT features. Eur Radiol. 2002;12 Suppl 3:S127-S129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 18. | Weinrach DM, Wang KL, Keh P, Sambasiva Rao M. Pathologic quiz case: a 40-year-old woman with a large pelvic mass, ascites, massive right hydrothorax, and elevated CA 125. Uterine symplastic leiomyoma associated with pseudo-Meigs syndrome and elevated CA 125. Arch Pathol Lab Med. 2004;128:933-934. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 19. | Ricci G, Inglese S, Candiotto A, Maso G, Piccoli M, Alberico S, Guaschino S. Ascites in puerperium: a rare case of atypical pseudo-Meigs' syndrome complicating the puerperium. Arch Gynecol Obstet. 2009;280:1033-1037. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 20. | Kurai M, Shiozawa T, Noguchi H, Konishi I. Leiomyoma of the ovary presenting with Meigs' syndrome. J Obstet Gynaecol Res. 2005;31:257-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 21. | Yip HK, Huang LW, Lin YH, Hwang JL. Massive ascites caused by a large pedunculated subserosal uterine leiomyoma that has feeding arteries from peripheral tissues and exhibits elevated CA125: a case report of atypical Pseudo-Meigs' syndrome. J Obstet Gynaecol. 2014;34:107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 22. | Tajima Y, Kameyama H, Yamada S, Yagi R, Nakano M, Nagahashi M, Shimada Y, Sakata J, Kobayashi T, Umezu H, Wakai T. Long-term survival in pseudo-Meigs' syndrome caused by ovarian metastases from colon cancer. World J Surg Oncol. 2016;14:286. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 23. | Yin H, Li XH, Xu HM, Lu YP. Pseudo-Meigs' syndrome secondary to bilateral ovarian endometrioid carcinomas. Int J Gynaecol Obstet. 1999;66:293-295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 24. | Ishiko O, Yoshida H, Sumi T, Hirai K, Ogita S. Vascular endothelial growth factor levels in pleural and peritoneal fluid in Meigs' syndrome. Eur J Obstet Gynecol Reprod Biol. 2001;98:129-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 27] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 25. | Migishima F, Jobo T, Hata H, Sato R, Ikeda Y, Arai M, Kuramoto H. Uterine leiomyoma causing massive ascites and left pleural effusion with elevated CA 125: a case report. J Obstet Gynaecol Res. 2000;26:283-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 26. | Roques F, Sanchez B, Bucher B, Larivière J. Role of pre-operative assessment in the surgical management of leiomyoma extended to the right heart chambers: a compendium of information from isolated reports. Eur J Cardiothorac Surg. 2001;19:522-524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 27. | Kingdom JC, Hobson SR, Murji A, Allen L, Windrim RC, Lockhart E, Collins SL, Soleymani Majd H, Alazzam M, Naaisa F, Shamshirsaz AA, Belfort MA, Fox KA. Minimizing surgical blood loss at cesarean hysterectomy for placenta previa with evidence of placenta increta or placenta percreta: the state of play in 2020. Am J Obstet Gynecol. 2020;223:322-329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 87] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 28. | He RH, Yao WM, Wu LY, Mao YY. Highly elevated serum CA-125 levels in patients with non-malignant gynecological diseases. Arch Gynecol Obstet. 2011;283 Suppl 1:107-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 29. | Kil K, Chung JE, Pak HJ, Jeung IC, Kim JH, Jo HH, Kim MR. Usefulness of CA125 in the differential diagnosis of uterine adenomyosis and myoma. Eur J Obstet Gynecol Reprod Biol. 2015;185:131-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 46] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 30. | Çınar M, Aksoy RT, Güzel Aİ, Tokmak A, Yenicesu O, Sarıkaya E, Evliyaoğlu Ö. The association between clinical parameters and uterine fibroid size in patients who underwent abdominal myomectomy. J Exp Ther Oncol. 2016;11:195-198. [PubMed] |
| 31. | Bast RC Jr, Badgwell D, Lu Z, Marquez R, Rosen D, Liu J, Baggerly KA, Atkinson EN, Skates S, Zhang Z, Lokshin A, Menon U, Jacobs I, Lu K. New tumor markers: CA125 and beyond. Int J Gynecol Cancer. 2005;15 Suppl 3:274-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 283] [Cited by in RCA: 315] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 32. | Yilmaz N, Sahin I, Kilic S, Ozgu E, Gungor T, Bilge U. Assessment of the predictivity of preoperative serum CA 125 in the differential diagnosis of uterine leiomyoma and uterine sarcoma in the Turkish female population. Eur J Gynaecol Oncol. 2009;30:412-414. [PubMed] |
| 33. | Tolman CJ, Vaid T, Schreuder HW. Extremely elevated CA-125 in benign ovarian disease due to stretch of the peritoneum. BMJ Case Rep. 2012;2012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 34. | Bottoni P, Scatena R. The Role of CA 125 as Tumor Marker: Biochemical and Clinical Aspects. Adv Exp Med Biol. 2015;867:229-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 121] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 35. | Zhang P, Zhang C, Hao J, Sung CJ, Quddus MR, Steinhoff MM, Lawrence WD. Use of X-chromosome inactivation pattern to determine the clonal origins of uterine leiomyoma and leiomyosarcoma. Hum Pathol. 2006;37:1350-1356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 44] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 36. | Kido A, Togashi K, Koyama T, Yamaoka T, Fujiwara T, Fujii S. Diffusely enlarged uterus: evaluation with MR imaging. Radiographics. 2003;23:1423-1439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 80] [Article Influence: 3.8] [Reference Citation Analysis (0)] |