Published online Oct 26, 2021. doi: 10.12998/wjcc.v9.i30.9134
Peer-review started: March 22, 2021
First decision: July 5, 2021
Revised: July 13, 2021
Accepted: August 13, 2021
Article in press: August 13, 2021
Published online: October 26, 2021
Processing time: 213 Days and 2.5 Hours
Pancreatic cancer (PC) is a leading cause of cancer-related death, given its poor prognosis and the limited benefits of traditional therapies. As tumors become more genetically disorganized as they progress, genetic mutations might become new markers for us to predict their behavior. Nowadays, many inhibitors can selectively target gene products as a form of targeted therapy, with some showing promise as treatment for various types of cancer.
We describe a rare case of a PC patient with long-term survival of more than 8 yr. The patient was diagnosed with pancreatic ductal adenocarcinoma (PDAC) with BAP1 and PIK3CA gene mutations and Raf1 fusion and achieved partial response twice after treatment with apatinib in combination with chemotherapy.
BAP1, PIK3CA mutations, and Raf1 fusion are rare in PDAC. Patients with these three gene alterations of PDAC may achieve long-term survival with apatinib. Further research in other contexts is needed to determine whether apatinib has ideal efficacy for PC treatment.
Core Tip: We report a patient with pancreatic ductal adenocarcinoma (PDAC) possessing exceptionally rare RAF1, BAP1 and PIK3CA gene alterations who achieved partial response to apatinib combination therapy twice and experienced long-term survival. Until now, there have been no reports of a long-term PDAC patient with RAF1, BAP1 and PIK3CA aberrations who did not also have K-Ras, TP53, p16/ CDKN2A, or SMAD4 gene alterations. In such a rare case, we presume that PDAC with this special genetic alteration pattern might be converted to a kind of indolent cancer, which presents fewer symptoms and indicates a good prognosis for pancreatic cancer patients.
- Citation: Yang WW, Yang L, Lu HZ, Sun YK. Long-term survival of a patient with pancreatic cancer and lung metastasis: A case report and review of literature. World J Clin Cases 2021; 9(30): 9134-9143
- URL: https://www.wjgnet.com/2307-8960/full/v9/i30/9134.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i30.9134
Pancreatic cancer (PC) is the seventh most common cause of cancer -related mortality worldwide[1]. Its lethality results from its late presentation and poor prognosis remains a challenge for clinicians. PC is regarded as a silent disease because it is generally asymptomatic at early stages; thus, it is often undiagnosed until the tumor has already progressed to an advanced stage with a poor prognosis. As for laboratory tests, the level of serum carbohydrate antigen (CA)19-9 is a key predictor for pancreatic cancer, but its specificity is not high enough to establish a diagnosis. Because PC is rarely detected in its infancy, effective treatment options are limited. Only 15%-20% of patients diagnosed at an early stage are amenable to surgery, and the 5-year survival rate is only 20%[2,3]. It is known that PC is resistant to chemotherapy. Most patients who are diagnosed at advanced stages have a short median overall survival of 6 mo after optimum systemic therapy, with a 5-year survival rate of only 2%[4,5].
Cancer is defined as a disease caused by the accumulation of mutations. Different histological types of PC tend to express different genetic mutation patterns with different prognoses. Pancreatic ductal adenocarcinoma (PADC) is a major histological subtype of PC that accounts for 78.8% of PCs containing the four most common gene mutations, K-Ras, TP53, CDKN2A, and SMAD4[6]. With the emergence of next generation sequencing (NGS), these genetic alterations might become promising markers to predict tumor behavior, and might be used to diagnose and treat cancer. The treatment of lung cancer is a successful example. Various specific alterations in lung tumors, such as EGFR and ALK genetic mutations, can be targeted by specific agents[7]. Currently, several drugs as targeted therapy for PC treatment have been approved by the United States Food and Drug Administration, with some having achieved encouraging therapeutic efficacy. In this case report, we describe a PC patient with lung metastases who achieved an 8-year survival after the initial diagnosis and 5 years after the lung metastases emerged. The genetic alterations of BAP1, PIK3CA and Raf1 were detected in the tumor tissue, which is rare in PADC.
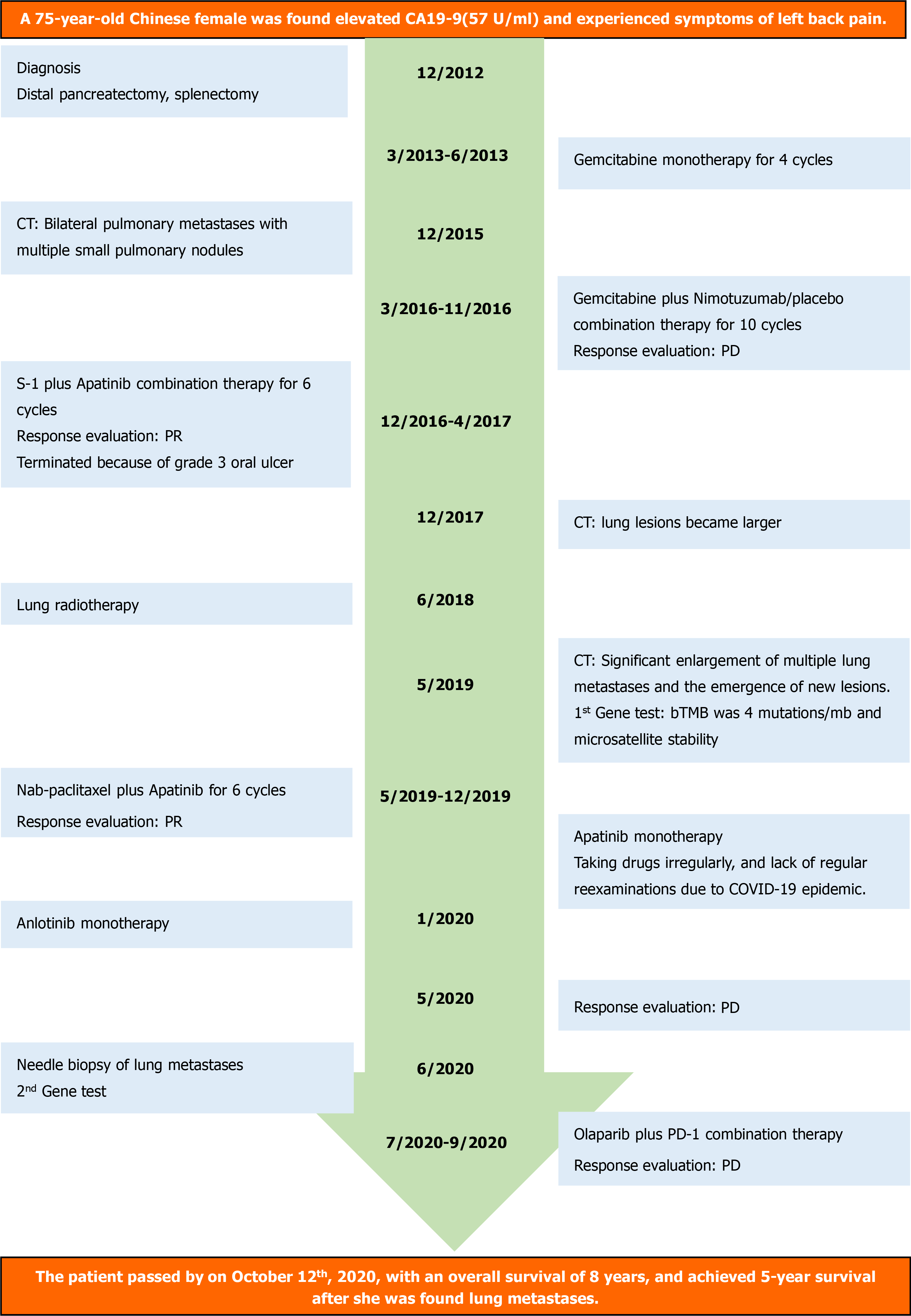
In April 2012, a 67-year-old Chinese woman was initially found to have an CA19-9 of 57 U/mL (normal range 0-35 U/mL) and experienced a symptom of left back pain.
The patient denied having left back pain, CA19-9 elevation or other clinical manifestations.
The patient had a clean medical history.
There was no family history of cancer.
No abnormality was found upon physical examination.
Blood analysis showed that CA19-9 was elevated to 57 U/mL (normal range 0-35 U/mL).
An initial imaging evaluation with a thoracic-abdominal computed tomography (CT) scan revealed a mass located in the pancreas.
In December 2012, she was diagnosed with PC at Peking Union Medical College Hospital. Distal pancreatectomy, splenectomy, and extended lymphadenectomy were performed, and the body and tail of the pancreas (9.5 cm × 3.5 cm × 1.5 cm) including a solid mass of about 2.0 cm × 1.1 cm × 2.5 cm was resected. Postoperative histopathological investigation found a moderately differentiated PADC, that was positive for alpha-1 antitrypsin, alpha 1-antichymotrypsin, CK7, AE1/AE3 by immunohistochemistry. There was invasion of peripancreatic adipose tissue, splenic parenchyma, and peripancreatic lymph and splenic hilar lymph nodes were positive. The surgical margins were negative. According to the Eighth edition of American Joint Committee on Cancer TNM staging system for PC, the tumor was classified as stage IIB (pT3N1M0).
After resection of lesions, four cycles of gemcitabine monotherapy (1300 mg on days 1, 8 and 15 of a 28 d cycle) was given from March to June 2013 at a local hospital, with regular reevaluation. Between June 2013 and February 2015, the patient did not undergo any treatment or examination. Multiple testing from February to December 2015 found persisting elevated levels of CA19-9. A thoracic-abdominal CT scan in December 2015showed bilateral pulmonary metastases with multiple small pulmonary nodules. The pulmonary metastasis was a clinical diagnosis but not approved by pathology, and the patient did not receive any form of therapy.
Genetic testing, completed as part of a clinical trial (CTR20131232) screening, found that she had a wild-type K-Ras gene. In the trial, she was administered gemcitabine (1300 mg on days 1, 8 and 15 of a 28 d cycle) plus nimotuzumab/placebo (400 mg on days 1, 8, 15 and 22 of a 28-d cycle), combination therapy for ten cycles from March to November 2016. Stable disease was achieved in the first eight cycles, but the lesions in her lung became larger after 10 cycles of treatment, which indicated progressive disease (PD).
From December 2016 to April 2017, she was treated with S-1 50 mg twice daily on days 1-7 of a 14 d cycle, plus apatinib 250 mg once daily of a 14 d cycle from the Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College and administered at the local hospital. However, in that period, she did not take drugs regularly because of an oral ulcer. The overall therapeutic effects were evaluated as stable disease after three cycles of treatment and partial response (PR) after 6 cycles of treatment. Unfortunately, the treatment was terminated because she developed a grade 3 oral ulcer (Common Terminology Criteria for Adverse Events v4.0).
Reevaluation at her local hospital in December 2017 found that the lung lesions were larger than before she had received any therapy. The lesions progressed slowly, and lung radiotherapy was performed in June 2018. Subsequent to treatment, she developed radiation pneumonitis, and was given symptomatic treatment with hormones and antibiotics. In May 2019, a restaging thoracic-abdominal CT scan revealed significant enlargement of multiple lung metastases and the emergence of new lesions, indicating that her disease had progressed. A peripheral blood sample was collected for genetic testing by NGS, which revealed that the blood tumor mutation burden included four mutations/mb and microsatellite stability. Both results signaled that the patient was not sensitive to immunotherapy.
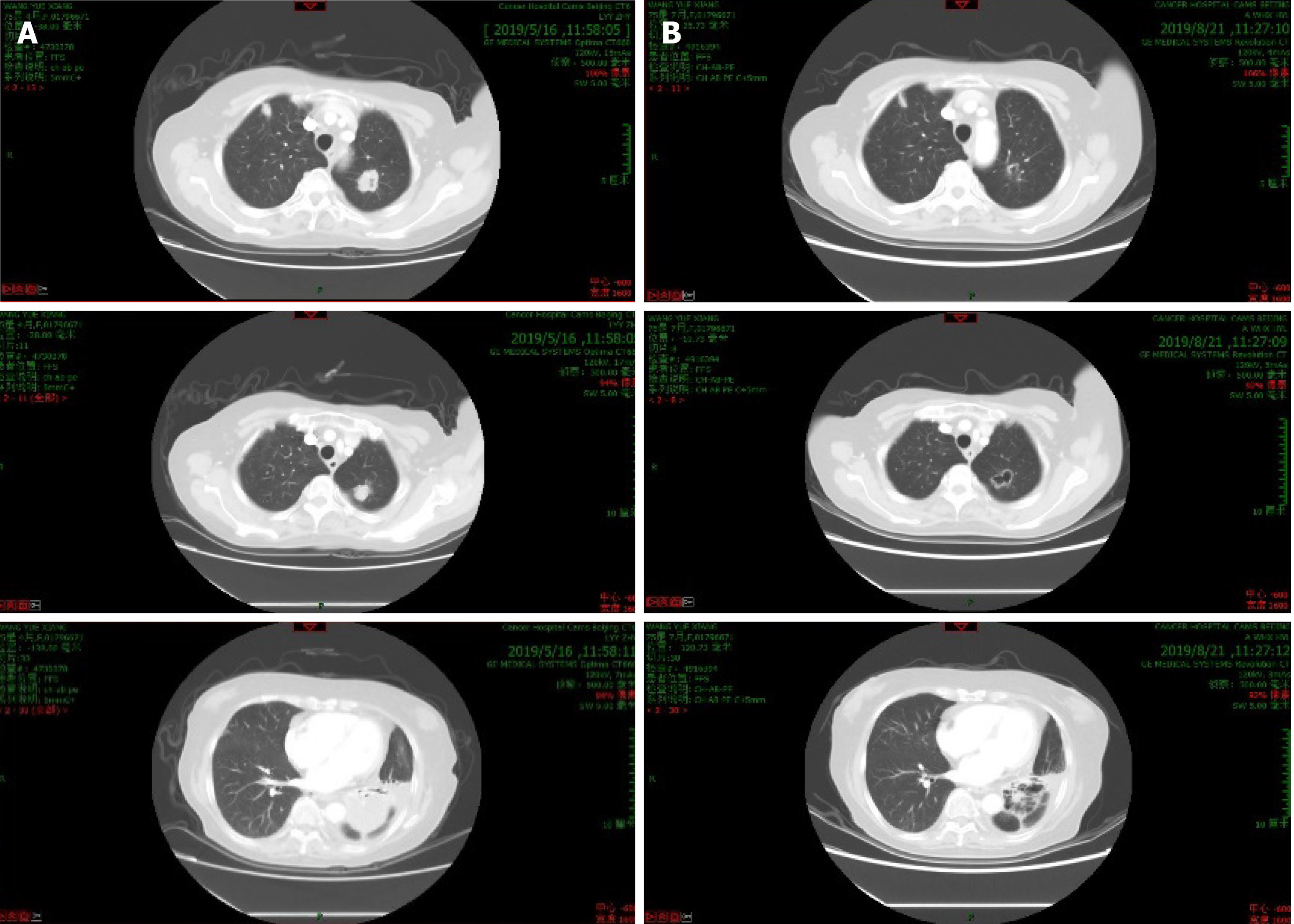
The patient was started on a nab-paclitaxel (180 mg on day 1 of a 14 d cycle) plus apatinib 250 mg once daily of a 14-d cycle that continued from May to December 2019. After six cycles of treatment, the lung lesions were classified as a PR (Figure 1). Subsequently, apatinib was administered as single agent maintenance therapy. However, this patient did not take drugs regularly, and she was unable to attend regular reexamination because of the emergence of the COVID-19 epidemic. At the beginning of 2020, she intermittently received oral anlotinib, also a multikinase inhibitor, (12 mg once daily on days 1-14 of a 21-d cycle).
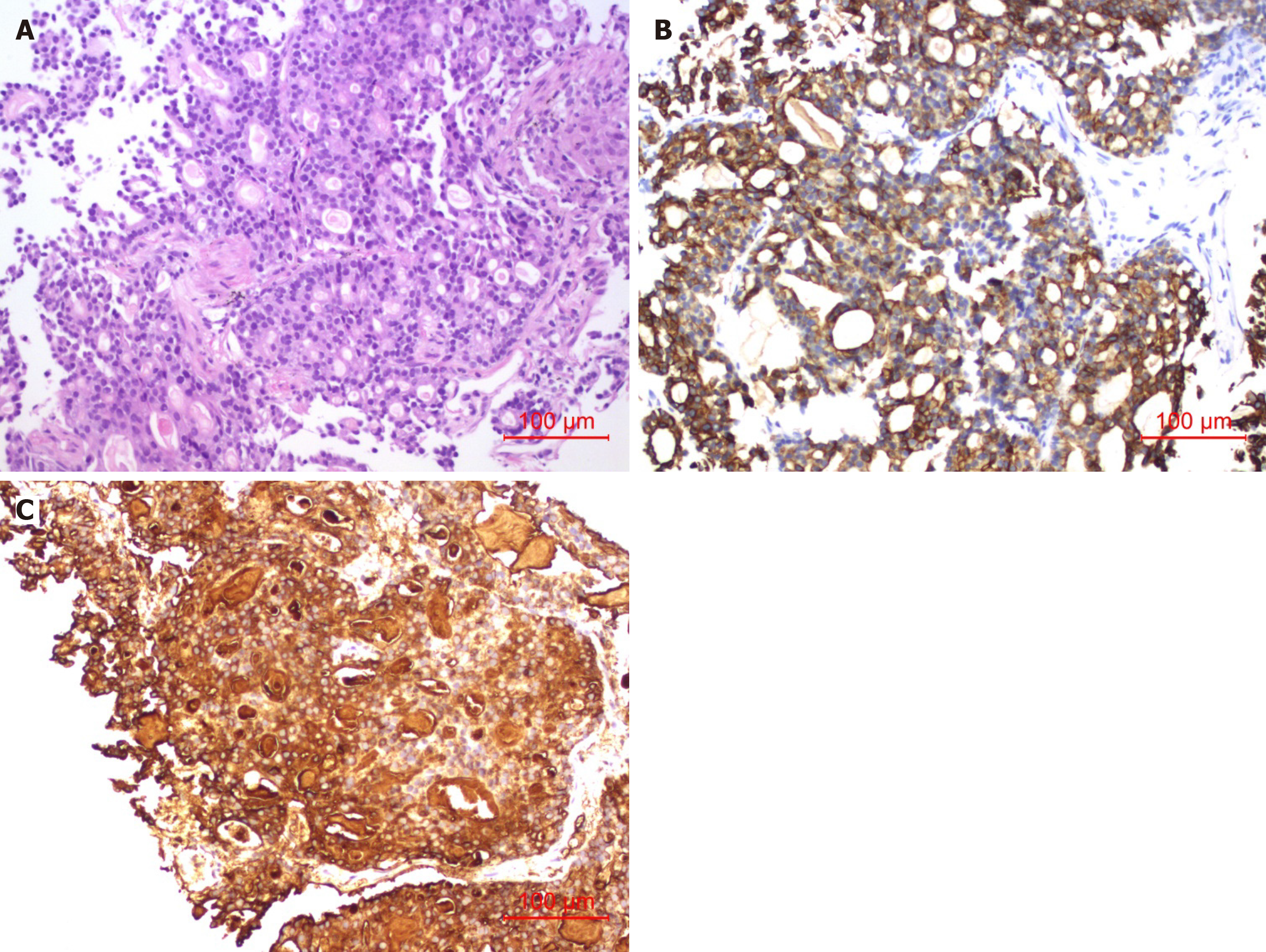
In May 2020, a restaging CT revealed progression of disease with multiple lung metastases that had increased in number and size. Needle biopsy, histopathological examination, and immunohistochemistry in June 2020 (Figure 2) revealed an adenocarcinoma that was positive for CA19-9, AE1/AE3, CK19 and Ki-67 (25%). Combined with her medical history, the examination confirmed that the lesions in her lung were the metastases of PC. The biopsy tissue was sent for genetic testing by NGS, which found that 40.68% of the lung biopsy tissue had PIK3CA genetic mutations and 62.07% mutations in the BAP1 gene and Raf1 gene fusion was detected in 49.82% of the biopsy tissue (Table 1). The patient then received olaparib plus PD-1 combination therapy from July to September 2020; and the last response evaluation was progressive disease.
| Genetic testing report | |||
| Tissue | Lung biopsy tissue | Date | June 2020 |
| Panel | 733 | Content of tumor tissue | 80% |
| Gene | Aberration | Mutation frequency/copy number | |
| PIK3CA | p.E545K Exon 10 | 40.68% | |
| RAF1 | AKAP9-RAF1 rearrangement | 49.82% | |
| BAP1 | p.T254Rfs*4 Exon9 | 62.07% | |
| TSC1 | Copy number decreasing | 1 | |
| PTCH1 | Copy number decreasing | 1 | |
| MLH1 | Copy number decreasing | 1 | |
| ALK | / | / | |
| BRAF | / | / | |
| BRCA1/2 | / | / | |
| PD-L1/2 | / | / | |
| EGFR | / | / | |
| HER2 | / | / | |
| FGFR2 | / | / | |
| KIT | / | / | |
| KRAS | / | / | |
| MET | / | / | |
| NRAS | / | / | |
| NTRK1/2/3 | / | / | |
| PDGFRA | / | / | |
| RET | / | / | |
| ROS1 | / | / | |
| POLD1 | / | / | |
| POLE | / | / | |
| TP53 | / | / | |
| RAD50 | / | / | |
| PBRM1 | / | / | |
| MDM2/4 | / | / | |
| DNMT3A | / | / | |
| JAK1/2 | / | / | |
| PTEN | / | / | |
| FGF3/4/19 | / | / | |
The patient passed away in October 2020, with an overall survival of 8 years and a 5-year survival after she was found to have lung metastases (Figure 3).
Even though much progress has been made in the management of other gastrointestinal tract cancers, the treatment of patients with PC has undergone little advancement in the last few years. In this report, we present a rare case of a 75-year-old woman diagnosed with PADC located in the body and tail of her pancreas. She underwent surgery 8 years ago and multiple lung metastases were detected 5 years ago. Three gene tests were conducted and indicated that she had wild-type K-Ras, BAP1, PIK3CA and Raf1 gene aberrations, all of which are rare in PADC. The patient was treated with surgery, chemotherapy, and targeted therapy, and lived for 8 years after the initial diagnosis, with a high quality of life. A recent meta-analysis found that the median overall survival of 819 patients with resectable PC was only 14.8 mo[8].
It is accepted that tumorigenesis is a process of accumulation of substantial genetic alterations; which could be regarded as predictors for prognoses of various types of cancer. Based on the three gene tests, we considered that the long-term survival of the patient was associated with the eccentric behavior of the genes. As mentioned above, the different histopathological types of PC tend to have different genetic alteration patterns. In PDAC, the most common gene mutations are K-Ras (over 90%), TP53, CDKN2A, SMAD4, but this patient did not have any of them. The downstream effectors of K-Ras signaling, which indicated by the genetic testing report in 2020, are shown in Table 1[9]. K-Ras is considered as a predictor of the prognosis of PC. In PDAC, patients with K-Ras mutations tend to have a poorer prognosis[10]. Mutant p53 and SMAD4 mutations are also associated with a poor outcome in PDAC patients[11,12]. Therefore, the long-term survival of this patient might be linked to the fact that none of these four widespread mutations (i.e. K-Ras, TP53, CDKN2A, SMAD4) were present. The gene aberrations present in the patient were PIK3CA E545K and BAP1 T254Rfs*4 mutation and AKAP9-Raf1 fusion, all of which are rare in PC. The PIK3CA gene encodes p110α protein, a subunit of phosphatidylinositol 3-kinase (PI3K), which activates the PI3K/Akt/mTOR pathway, with signaling transduction suppressing cell apoptosis and promoting cell proliferation and growth[13-16]. Mutations of PIK3CA may lead to the subsequent activation of PI3K, which then deregulates the signaling pathway and confers oncogenic potential to the cells[17]. Cancer Genome Atlas data indicates that patients with PIK3CA mutations tend to have a tendency for a decreased overall survival, the mutation only occurs in 0.8% of patients with PC, so few reports or studies of PIK3CA mutation-associated PC could be retrieved[18]. So far, PIK3CA mutations initiating tumorigenesis of PC have only been observed in mice, and the role it has in human PC oncogenesis needs to be explored[19]. The BAP1 gene encodes BRCA1-associated protein 1, which suppresses tumors by promoting the activity of the Hippo tumor suppressor pathway[20]. Therefore, BAP1-inactivating mutations contribute to tumorigenesis. In several types of cancer, such as clear-cell renal cell carcinoma, uveal melanoma, and colorectal cancer, the presence of mutations of the BAP1 gene is a herald of poor prognosis[21]. The frequency of BAP1 mutations occurring in PDAC is low, about 0.33%[22], and whether it contributes to the induction and progression of PC is still unclear. Raf1 rearrangement can consecutively activate the MAPK (i.e. Ras/Raf/MEK/Erk) signaling pathway), which leads to unlimited cell proliferation and suppressed apoptosis[23,24]. Thus the Raf1 fusion should contribute to oncogenesis and be associated with a poor prognosis. However, in PDAC, Raf1 fusion is rare, so we are unable to predict how the mutation might affect the prognosis of PC. Interestingly, Raf1 gene fusion is relatively common in pancreatic acinar cell carcinoma (PACC), with an incidence ranging from 14.3% to 18.5%, but rare in PDAC[25]. Unlike PDAC, PACC possesses less frequent K-Ras, TP53, p16/CDKN2A, and SMAD4, but the existence of BRCA1, BRCA2, B-Raf/Raf1, RB1, ATM, and GNAS gene mutations have been reported[26]. Moreover, patients with PACC have a better prognosis than patients with PDAC[27]. Our patient had Raf1 fusion without K-Ras, TP53, p16/CDKN2A, or SMAD4 mutation, which might indicate this PACC-like genetic alteration pattern is a herald of good prognosis. Furthermore, in the course of therapy, the patient twice achieved PR. The first PR was achieved after she took S-1 and apatinib and the second PR was after nab-paclitaxel plus apatinib. From that, we may infer that apatinib has an important role in advanced PC treatment.
In recent years, targeted therapy has become popular in the field of cancer treatment. Apatinib, also known as YN968D1, is a multiple kinase inhibitor that blocks vascular endothelial growth factor receptor-2 (VEGFR-2) to inhibit tumor angiogenesis[28]. It has been approved by the National Medical Products Administration of China for gastric cancer treatment and is currently under investigation for multiple indications, such as colon, breast, and liver cancer. PC is not an indication of apatinib, but tumor angiogenesis plays an important role in the oncogenesis of PC; thus, we might suppress the tumor growth by blocking the VEGFR-2 signaling pathway[29]. A preclinical study by He et al[30] found that apatinib promoted the apoptosis of CFPAC-1 and SW1990 PC cells and downregulated the expression of hypoxia-inducible factor-1α (HIF-1α) and vascular endothelial growth factor (VEGF). HIF-1α and VEGF both have roles in angiogenesis, so apatinib might suppress the growth of pancreatic tumors by decreasing their expression. A case report published in 2017 showed that a patient with pretreated metastatic PC had a significant response to apatinib, which reinforces our supposition. The patient was diagnosed with stage Ⅲ unresectable PC, with a 3.1 cm × 1.7 cm mass in the body of the pancreas. An endoscopic biopsy revealed a moderately differentiated adenocarcinoma[31].
We also considered whether the good response of our patient was associated with alterations of the RAF1, BAP1 and PIK3CA genes. Unfortunately, there is no evidence to verify that apatinib targets those genes or their related proteins, so the results are inconclusive. In clear-cell renal cell carcinoma, mutation of BAP1 is correlated with a decreased efficacy of anti-angiogenic therapy, thus warranting further research[32]. Until now, there have been no reports of long-term survival of a PDAC patient with RAF1, BAP1, and PIK3CA mutations who did not also have K-Ras, TP53, p16/CDKN2A, or SMAD4 gene alterations. In such rare cases, we presume that the rare genetic alteration pattern may convert PDAC to a kind of indolent cancer that presents with fewer symptoms and has a good prognosis. Whether this kind of gene map represents PC with a slow progression and good prognosis needs to be verified by further case studies and analysis. In the future, we are likely to find more gene maps that are indicative of good prognosis and ideal drug responses. The interpretation of this case has some limitations. For instance, we did not carry out genetic testing when we first found pulmonary nodules on CT, thus there should be a discussion of whether the lung metastases represented the primary PC that was found in 2012. Furthermore, this patient did not share her perspective on the treatments she received.
The patient had PADC that carried exceptionally rare RAF1, BAP1, and PIK3CA gene alterations, twice achieved partial responses to apatinib combination therapy, and experienced a long-term survival. We hold the opinion that various genetic mutations are associated with different tumor behaviors. With the emergence and development of NGS, we can to predict, diagnose, and treat cancers at early stages based on the genetic profiles. Even though many effects of genetic changes are still unclear, through the accumulation of experience and the promotion of research, more mysteries surrounding gene alterations will be revealed.
We reported a case of PDAC in a patient who achieved a long-term survival of 8 years after diagnosis. The patient possessed rare RAF1, BAP1 and PIK3CA gene alterations and twice achieved partial responses to apatinib combination therapy, which might indicate that PDAC with this rare genetic alteration pattern represents a kind of indolent cancer with a good prognosis. Further investigation is needed to confirm it.
Manuscript source: Unsolicited manuscript
Specialty type: Oncology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Casella C, Hamaya Y S-Editor: Ma YJ L-Editor: Filipodia P-Editor: Li JH
| 1. | Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53206] [Cited by in RCA: 55751] [Article Influence: 7964.4] [Reference Citation Analysis (132)] |
| 2. | Konstantinidis IT, Warshaw AL, Allen JN, Blaszkowsky LS, Castillo CF, Deshpande V, Hong TS, Kwak EL, Lauwers GY, Ryan DP, Wargo JA, Lillemoe KD, Ferrone CR. Pancreatic ductal adenocarcinoma: is there a survival difference for R1 resections vs locally advanced unresectable tumors? Ann Surg. 2013;257:731-736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 281] [Cited by in RCA: 307] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 3. | Kopper L, Zalatnai A, Tímár J. Genomics of pancreatic cancer: does it make any improvement in diagnosis, prognosis and therapy? Pathol Oncol Res. 2005;11:69-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 4. | Tabernero J, Chiorean EG, Infante JR, Hingorani SR, Ganju V, Weekes C, Scheithauer W, Ramanathan RK, Goldstein D, Penenberg DN, Romano A, Ferrara S, Von Hoff DD. Prognostic factors of survival in a randomized phase III trial (MPACT) of weekly nab-paclitaxel plus gemcitabine vs gemcitabine alone in patients with metastatic pancreatic cancer. Oncologist. 2015;20:143-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 114] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 5. | Chua YJ, Zalcberg JR. Pancreatic cancer--is the wall crumbling? Ann Oncol. 2008;19:1224-1230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 6. | Chen J, Baithun SI. Morphological study of 391 cases of exocrine pancreatic tumours with special reference to the classification of exocrine pancreatic carcinoma. J Pathol. 1985;146:17-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 108] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 7. | Morganti S, Tarantino P, Ferraro E, D'Amico P, Duso BA, Curigliano G. Next Generation Sequencing (NGS): A Revolutionary Technology in Pharmacogenomics and Personalized Medicine in Cancer. Adv Exp Med Biol. 2019;1168:9-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 132] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 8. | Versteijne E, Vogel JA, Besselink MG, Busch ORC, Wilmink JW, Daams JG, van Eijck CHJ, Groot Koerkamp B, Rasch CRN, van Tienhoven G; Dutch Pancreatic Cancer Group. Meta-analysis comparing upfront surgery with neoadjuvant treatment in patients with resectable or borderline resectable pancreatic cancer. Br J Surg. 2018;105:946-958. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 251] [Cited by in RCA: 386] [Article Influence: 55.1] [Reference Citation Analysis (1)] |
| 9. | Kanda M, Matthaei H, Wu J, Hong SM, Yu J, Borges M, Hruban RH, Maitra A, Kinzler K, Vogelstein B, Goggins M. Presence of somatic mutations in most early-stage pancreatic intraepithelial neoplasia. Gastroenterology. 2012;142:730-733.e9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 459] [Cited by in RCA: 545] [Article Influence: 41.9] [Reference Citation Analysis (0)] |
| 10. | Bournet B, Buscail C, Muscari F, Cordelier P, Buscail L. Targeting KRAS for diagnosis, prognosis, and treatment of pancreatic cancer: Hopes and realities. Eur J Cancer. 2016;54:75-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 134] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 11. | Xiang JF, Wang WQ, Liu L, Xu HX, Wu CT, Yang JX, Qi ZH, Wang YQ, Xu J, Liu C, Long J, Ni QX, Li M, Yu XJ. Mutant p53 determines pancreatic cancer poor prognosis to pancreatectomy through upregulation of cavin-1 in patients with preoperative serum CA19-9 ≥ 1,000 U/mL. Sci Rep. 2016;6:19222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 12. | Xu JZ, Wang WQ, Zhang WH, Xu HX, Gao HL, Zhang SR, Wu CT, Li S, Li H, Xu J, Yu XJ, Liu L. The Loss of SMAD4/DPC4 Expression Associated with a Strongly Activated Hedgehog Signaling Pathway Predicts Poor Prognosis in Resected Pancreatic Cancer. J Cancer. 2019;10:4123-4131. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 13. | Tuttle RL, Gill NS, Pugh W, Lee JP, Koeberlein B, Furth EE, Polonsky KS, Naji A, Birnbaum MJ. Regulation of pancreatic beta-cell growth and survival by the serine/threonine protein kinase Akt1/PKBalpha. Nat Med. 2001;7:1133-1137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 405] [Cited by in RCA: 415] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 14. | Arcaro A, Guerreiro AS. The phosphoinositide 3-kinase pathway in human cancer: genetic alterations and therapeutic implications. Curr Genomics. 2007;8:271-306. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 146] [Cited by in RCA: 198] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 15. | Manning BD, Cantley LC. AKT/PKB signaling: navigating downstream. Cell. 2007;129:1261-1274. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4880] [Cited by in RCA: 4814] [Article Influence: 267.4] [Reference Citation Analysis (0)] |
| 16. | Pacold ME, Suire S, Perisic O, Lara-Gonzalez S, Davis CT, Walker EH, Hawkins PT, Stephens L, Eccleston JF, Williams RL. Crystal structure and functional analysis of Ras binding to its effector phosphoinositide 3-kinase gamma. Cell. 2000;103:931-943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 468] [Cited by in RCA: 490] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 17. | Chalhoub N, Baker SJ. PTEN and the PI3-kinase pathway in cancer. Annu Rev Pathol. 2009;4:127-150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 996] [Cited by in RCA: 1140] [Article Influence: 71.3] [Reference Citation Analysis (0)] |
| 18. | Zhou L, Baba Y, Kitano Y, Miyake K, Zhang X, Yamamura K, Kosumi K, Kaida T, Arima K, Taki K, Higashi T, Imai K, Hashimoto D, Yamashita Y, Chikamoto A, Beppu T, Tan X, Baba H. KRAS, BRAF, and PIK3CA mutations, and patient prognosis in 126 pancreatic cancers: pyrosequencing technology and literature review. Med Oncol. 2016;33:32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 19. | Payne SN, Maher ME, Tran NH, Van De Hey DR, Foley TM, Yueh AE, Leystra AA, Pasch CA, Jeffrey JJ, Clipson L, Matkowskyj KA, Deming DA. PIK3CA mutations can initiate pancreatic tumorigenesis and are targetable with PI3K inhibitors. Oncogenesis. 2015;4:e169. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 58] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 20. | Brekken RA. Loss of BAP1 Leads to More YAPing in Pancreatic Cancer. Cancer Res. 2020;80:1624-1625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Luchini C, Nottegar A. The Roles of Chromatin Remodeling Genes in Pancreatic-Biliary Malignancies. Crit Rev Oncog. 2017;22:471-479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Tayao M, Andrici J, Farzin M, Clarkson A, Sioson L, Watson N, Chua TC, Sztynda T, Samra JS, Gill AJ. Loss of BAP1 Expression Is Very Rare in Pancreatic Ductal Adenocarcinoma. PLoS One. 2016;11:e0150338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 23. | Karnoub AE, Weinberg RA. Ras oncogenes: split personalities. Nat Rev Mol Cell Biol. 2008;9:517-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1183] [Cited by in RCA: 1147] [Article Influence: 67.5] [Reference Citation Analysis (0)] |
| 24. | Malumbres M, Barbacid M. RAS oncogenes: the first 30 years. Nat Rev Cancer. 2003;3:459-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1287] [Cited by in RCA: 1376] [Article Influence: 62.5] [Reference Citation Analysis (0)] |
| 25. | Prall OWJ, Nastevski V, Xu H, McEvoy CRE, Vissers JHA, Byrne DJ, Takano E, Yerneni S, Ellis S, Green T, Mitchell CA, Murray WK, Scott CL, Grimmond SM, Hofmann O, Papenfuss A, Kee D, Fellowes A, Brown IS, Miller G, Kumarasinghe MP, Perren A, Nahm CB, Mittal A, Samra J, Ahadi M, Fox SB, Chou A, Gill AJ. RAF1 rearrangements are common in pancreatic acinar cell carcinomas. Mod Pathol. 2020;33:1811-1821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 26. | Al-Hader A, Al-Rohil RN, Han H, Von Hoff D. Pancreatic acinar cell carcinoma: A review on molecular profiling of patient tumors. World J Gastroenterol. 2017;23:7945-7951. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 50] [Cited by in RCA: 56] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 27. | Schmidt CM, Matos JM, Bentrem DJ, Talamonti MS, Lillemoe KD, Bilimoria KY. Acinar cell carcinoma of the pancreas in the United States: prognostic factors and comparison to ductal adenocarcinoma. J Gastrointest Surg. 2008;12:2078-2086. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 132] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 28. | Tian S, Quan H, Xie C, Guo H, Lü F, Xu Y, Li J, Lou L. YN968D1 is a novel and selective inhibitor of vascular endothelial growth factor receptor-2 tyrosine kinase with potent activity in vitro and in vivo. Cancer Sci. 2011;102:1374-1380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 362] [Cited by in RCA: 427] [Article Influence: 30.5] [Reference Citation Analysis (0)] |
| 29. | Kuwahara K, Sasaki T, Kuwada Y, Murakami M, Yamasaki S, Chayama K. Expressions of angiogenic factors in pancreatic ductal carcinoma: a correlative study with clinicopathologic parameters and patient survival. Pancreas. 2003;26:344-349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 87] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 30. | He K, Wu L, Ding Q, Haider F, Yu H, Wang H, Xiang G. Apatinib Promotes Apoptosis of Pancreatic Cancer Cells through Downregulation of Hypoxia-Inducible Factor-1α and Increased Levels of Reactive Oxygen Species. Oxid Med Cell Longev. 2019;2019:5152072. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 31. | Li CM, Liu ZC, Bao YT, Sun XD, Wang LL. Extraordinary response of metastatic pancreatic cancer to apatinib after failed chemotherapy: A case report and literature review. World J Gastroenterol. 2017;23:7478-7488. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 15] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 32. | Hakimi AA, Voss MH, Kuo F, Sanchez A, Liu M, Nixon BG, Vuong L, Ostrovnaya I, Chen YB, Reuter V, Riaz N, Cheng Y, Patel P, Marker M, Reising A, Li MO, Chan TA, Motzer RJ. Transcriptomic Profiling of the Tumor Microenvironment Reveals Distinct Subgroups of Clear Cell Renal Cell Cancer: Data from a Randomized Phase III Trial. Cancer Discov. 2019;9:510-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 174] [Article Influence: 29.0] [Reference Citation Analysis (0)] |