Published online Sep 16, 2021. doi: 10.12998/wjcc.v9.i26.7893
Peer-review started: April 8, 2021
First decision: June 4, 2021
Revised: June 5, 2021
Accepted: July 22, 2021
Article in press: July 22, 2021
Published online: September 16, 2021
Processing time: 155 Days and 2.2 Hours
Hepatocellular carcinoma (HCC) with inferior vena cava and right atrium thrombus is rare, accounting for approximately 1.4%-4.9% of cases. These patients are rarely reported, but the condition is being increasingly discovered with advances in imaging techniques, and their prognosis is extremely pessimistic with no current effective treatment. This condition is further associated with unexpected sudden death by cardiac arrest and acute large area pulmonary embolism.
A 34-year-old man with advanced HCC with a hepatic vein thrombus extending into the right atrium had a long-term, disease-free survival following 5-mo sequential treatment combined with transcatheter arterial chemoembolization and curative liver resection. No severe adverse effects were encountered, such as massive hemorrhage or pulmonary embolism. The proper selection of operative method is an important factor.
HCC with a tumor thrombus extending into the right atrium has a significant impact on the survival of patients. Thrombectomy combined with adjuvant therapy may be beneficial for these patients.
Core Tip: Hepatocellular carcinoma with a tumor thrombus extending into the right atrium has a huge impact on the survival of the patients. It is further associated with unexpected sudden death by cardiac arrest and acute large area pulmonary embolism. Thrombectomy combined with adjuvant therapy may bring light for these patients.
- Citation: Liu J, Zhang RX, Dong B, Guo K, Gao ZM, Wang LM. Hepatocellular carcinoma with inferior vena cava and right atrium thrombus: A case report. World J Clin Cases 2021; 9(26): 7893-7900
- URL: https://www.wjgnet.com/2307-8960/full/v9/i26/7893.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i26.7893
Hepatocellular carcinoma (HCC) is a highly metastatic and malignant cancer. Vascular invasion into the main vessels occurs most frequently in the advanced stage, in approximately 44%-62.2% of cases[1]. Tumor thrombus in the inferior vena cava (IVC) and right atrium (RA) is rare, approximately 1.4%-4.9%[2-4], and is an important prognostic factor and predictor of HCC. Such patients often survive less than 3 mo if untreated[5-6]. A major vein tumor is always associated with intrahepatic metastases and satellite nodules, which increases the recurrence rate after curative resection. Patients are also occasionally complicated by cardiac arrest, heart failure, lung metastasis, pulmonary artery embolism, or secondary Budd-Chiari syndrome[7-9], which are associated with a risk of death at any time. Macrovascular invasion would probably be classified as the Barcelona Clinic Liver Cancer stage C; hence, tyrosine kinase inhibitors, such as sorafenib or lenvatinib, should be given as per standard therapy[10]. We treated him successfully with systemic therapy, followed by transcatheter arterial chemoembolization (TACE) and liver resection. We also collected pathology results after TACE, which is important for this type of downstage therapy. At the end of the paper, we discuss which therapies should be considered and which details should be mentioned and avoided when facing such cases.
A 34-year-old man complained of abdominal pain for 2 mo.
The patient complained of episodes of continuous distending pain in his right upper abdomen for 2 mo. He denied other symptoms, including fever, vomiting, diarrhea, hematochezia, and skin rash, as well as recent medication intake or contact history.
The patient had a 30-year history of hepatitis B virus infection.
The patient’s mother had a history of hepatitis B virus infection.
On examination, the patient showed facial acne and facial hair. He used to be a boxer and was strongly built with a body mass index of 31 kg/m². On deep palpation, he had increased mild tenderness of the right upper abdominal quadrant.
An admission test was significant for AFP: 231.73 ng/mL. The patient’s liver function tests were as follows: Child-Pugh A; ALT: 52.6 U/L; AST: 26.4 U/L; TB: 20.2 µmol/L; Alb: 46.35 g/L; PT: 12.9 s; and INR: 0.97.
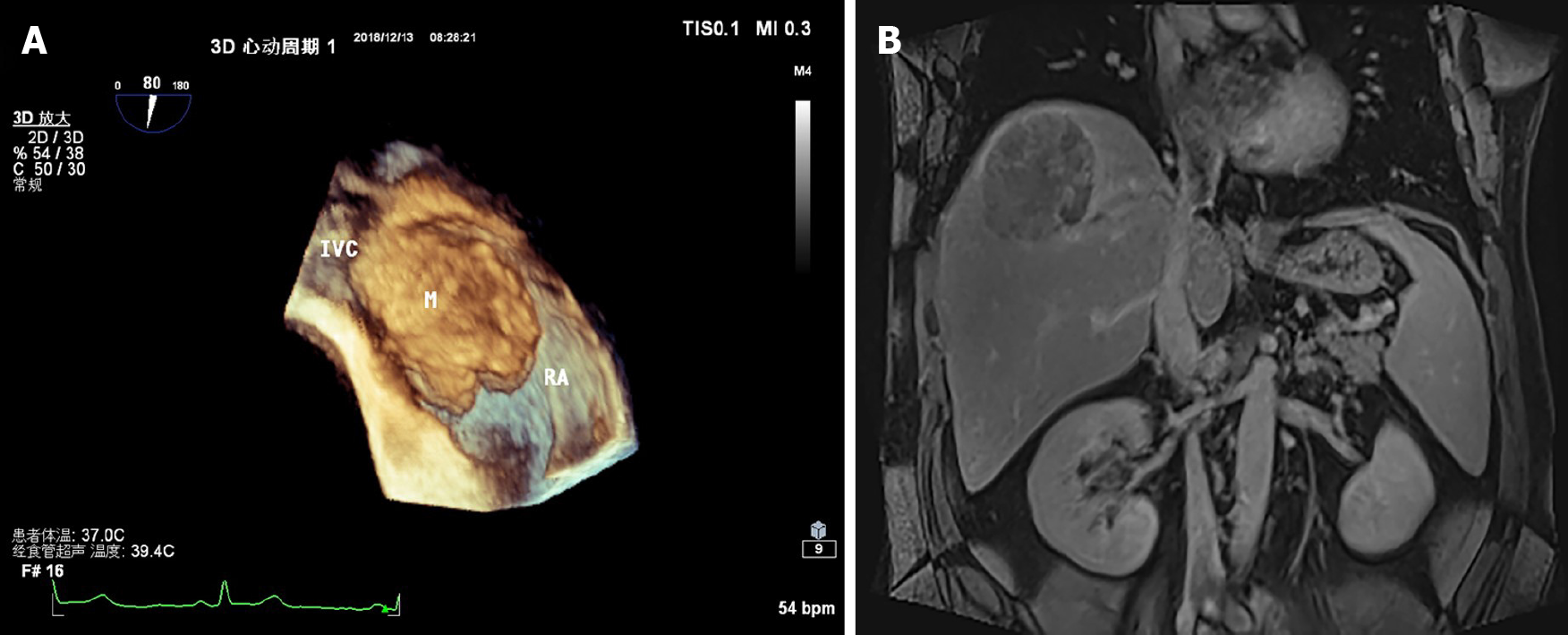
Ultrasound showed a medium echo of thrombus in the RA, with a range of approximately 56.0 mm × 42.0 mm, attached to the entrance of the IVC and RA (Figure 1A). Enhanced magnetic resonance imaging showed a mass, which was considered to be primary HCC with hemorrhage. Carcinoma thrombus grew from the hepatic segment to the IVC as well as the RA (Figure 1B).
The multidisciplinary team recommended sternotomy for resection of the carcinoma thrombus in consideration of the chances of unexpected sudden death by cardiac arrest and acute large area pulmonary embolism.
HCC with hemorrhage was ultimately diagnosed. Carcinoma thrombus grew from the hepatic segment to the IVC as well as the RA.
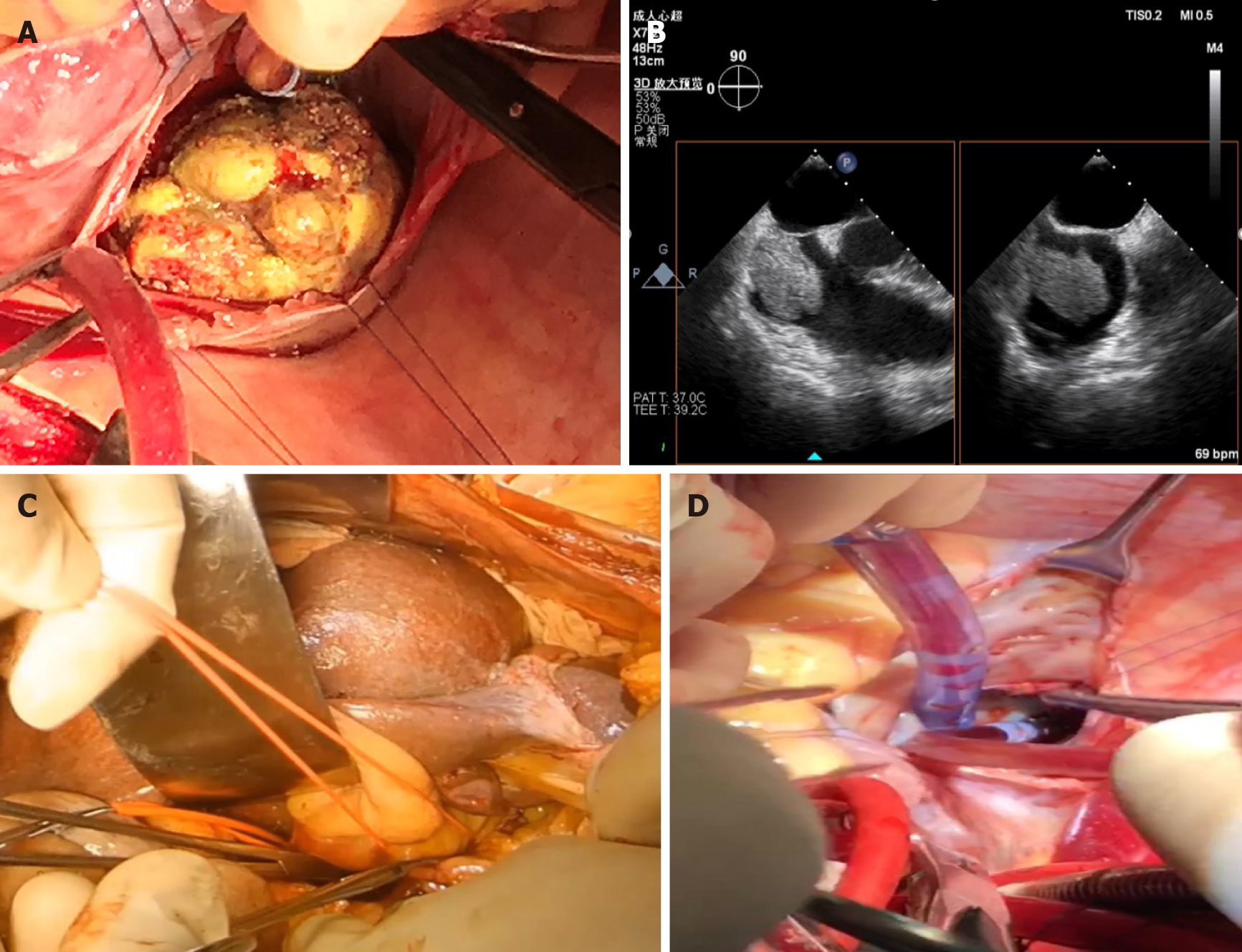
We first performed TACE twice to decrease his stage and reduce the recurrence rate. The patient underwent transcatheter arterial chemoembolization with 100 mg of cisplatin particles in 20 min via the right hepatic artery every month. The AFP decreased to within the normal range after the second session. Computed tomography showed that the main tumor and the tumor thrombus shrunk. After 2 mo, the patient received live resection and thrombectomy under cardiopulmonary bypass. The diameter of the tumor was approximately 10.5 cm. Conical secondary thrombus was found in the IVC, with a length of approximately 8 cm (Figure 2A). Ascending aorta, superior vena cava, and right femoral vein intubation bypass was carried out during surgery in collaboration with heart surgeons. To avoid an undesirable pulmonary tumor embolism, transesophageal echocardiography was monitored during the entire procedure (Figure 2B). After the supra-hepatic IVC was blocked, the RA was opened, and the tumor thrombus was completely removed with cardiac arrest (for further details regarding the surgical procedures, please refer to the Video). In addition, we reconstructed the defect of the cava. Intraoperative cardiac arrest lasted for 19 min. After releasing the ascending aorta in the pericardium, the heart started pumping again, and then cardiopulmonary bypass was performed. The cardiopulmonary bypass procedure lasted for 1 h and 6 min. Right hepatic lobectomy was performed with an ultrasonic dissector to decrease bleeding and occlude the hepatic portal vessels discontinuously with fingers.
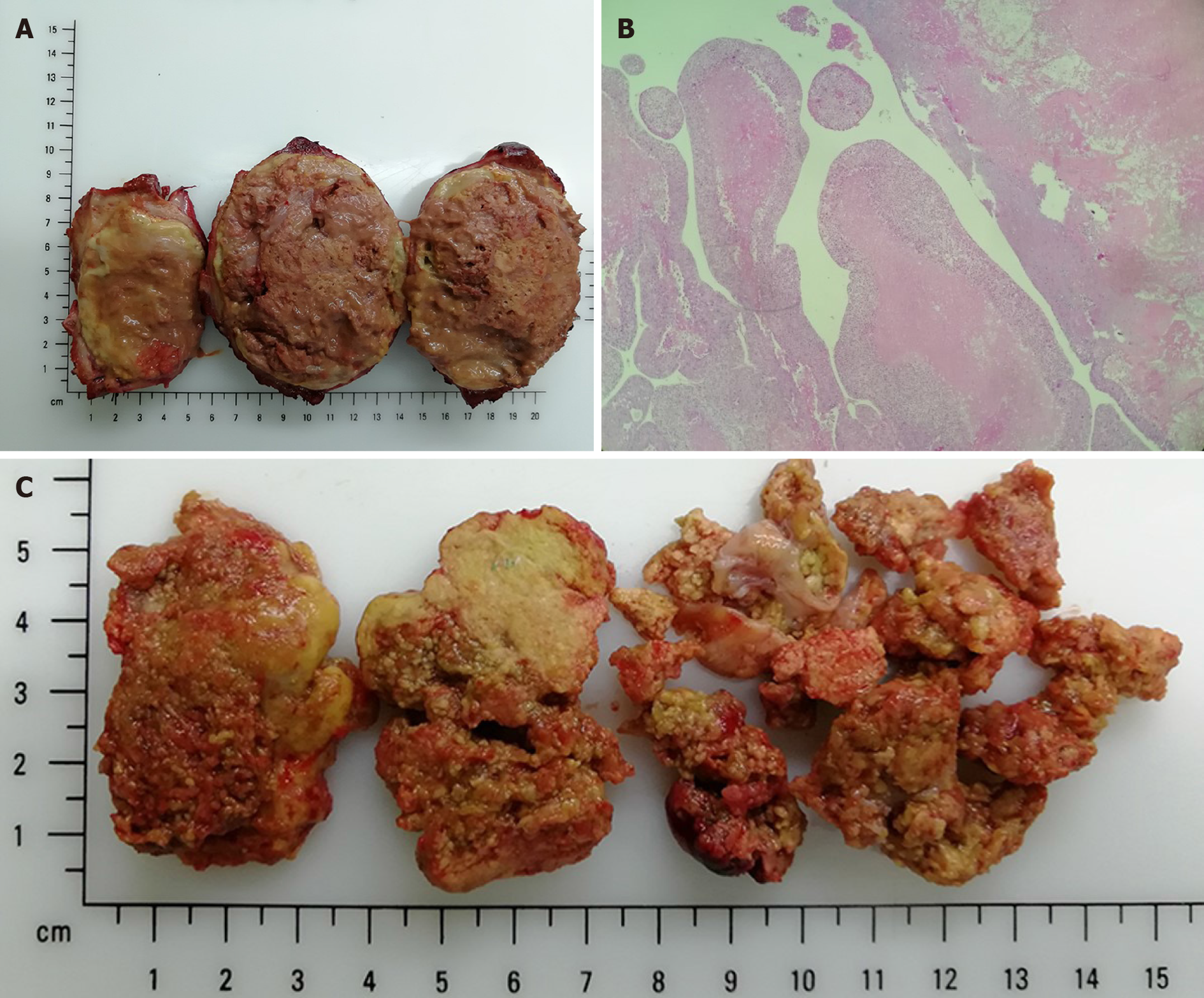
All IVC thrombi were removed under hepatic vascular exclusion (HVE) (Figure 2C and D). The total HVE was performed over 16 min. The operation time was 215 min. During the procedure, the blood loss was 3200 mL. We transferred 2400 mL of filtered autologous blood for transfusion, 1800 mL of suspended red cell fluid, and 1200 mL of plasma. The postoperative pathological findings were as follows: Moderately differentiated HCC, thick fibrous capsule with blue embolus particles visible in the vessels, and a necrotic area exceeding 95% (Figure 3A). Regarding the atrial thrombus, active cancer cells could be seen around the focal capsule, and necrosis was over 75% (Figure 3B and C).
The patient recovered uneventfully. After 1.5 mo of patient recovery, diluted tropi
HCC with a hepatic vein thrombus extending to the IVC and RA is rarely reported. The prognosis of patients presenting with IVC or RA thrombus is poor due to the chances of unexpected sudden death by cardiac arrest and acute large area pulmonary embolism. Patients are recommended to take sorafenib and lenvatinib, which are the only highly evidenced therapeutics recommended for advanced HCC treatment, although the median overall survival was less than 6.5 mo[3].
As cardiac arrest, heart failure, lung metastasis, and pulmonary artery embolism, which are associated with a risk of death at any time, were highly probable for this patient, it was extremely critical for him to choose a treatment option. We found that a series of cases could be referenced. Wang et al[4] reported that patients achieved a median survival duration of 19 mo through thrombectomy. Others applied TACE combined with or without radiotherapy (11.7 mo vs 4.7 mo) or no treatment (4.0 mo)[6,11]. The decision process was meticulous because of the high risk of the surgery. However, the patient and his parents were very positive regarding surgical outcomes. We collaborated with a multidisciplinary team to make a detailed plan.
One can observe the significance of surgery for these patients; however, there are no standardized opinions or procedures for these diseases, as not enough case reports have been published with detailed surgical procedures. We developed a personalized surgical plan for this patient focusing on some key points during surgery with a multidisciplinary team, including cardiothoracic surgeons, intervention surgeons, anesthetists, and oncologists.
Recently, surgeons have attempted thrombectomy without CPB. There have been five previous reports of cases where HCC with a thrombus extending into the IVC was successfully removed without CPB[7]. Some studies have aimed to distinguish this type, such as the classification of “vv” in Japan and “I/II/III” in China[12,13]. We recommend the Chinese standard based on its value for evaluating progression and the choice of therapy. We believe that the location and size of the main tumor and thrombus of this patient should be ranked to the intracardiac type (type III), in which the thrombus extends above the diaphragm and into the RA according to preoperative ultrasonography. Regarding type III, CPB is necessary to maintain a no-blood condition during an open-heart procedure. In addition, we need to pay attention to CPB complications (the internal environment is disordered, characterized by coagulation and circulatory, respiratory, and renal dysfunction) during the operation.
Concerning the risk of uncontrollable bleeding during lobectomy after heparinization in CPB, most surgeons choose to perform hepatectomy prior to RA thrombectomy. However, with the neutralization of heparin, the amount of hemorrhage during hepatectomy following thrombectomy is comparable to the general consensus or even less[14].
We opted to perform cava-atrial thrombectomy first for this patient to have more manipulated space and confidence to handle unexpected dropped embolus and heart arrest.
We even observed that inflow obstruction by the ball-valve effect may cause more uncontrollable bleeding when right hepatectomy is performed without first performing a thrombectomy[15]. In fact, one case of bleeding over 30 L was reported. Moreover, the ball-valve effect increased the incidence rates of sudden cardiac arrest[7]. In 2004, Sugimoto et al[13] first reported the performance of RA thrombectomy prior to hepatectomy to decrease death rates due to unexpected pulmonary embolism and cardiac arrest[14].
It is worth noting that oncological surgeons should weigh the pros and cons of liver surgery for patients with HCC, especially in thrombectomy prior to liver resection, with the need to cut the thrombus in the IVC going against the principle of “en bloc”. This procedure may increase the recurrence of lung metastasis, which occurs in Sugimoto’s and some other cases. However, the rate of lung metastasis did not decrease when managing hepatectomy prior to thrombectomy. According to reports, five of the 22 cases had lung metastasis after hepatectomy prior to thrombectomy[15]. Therefore, systemic sequential therapy, such as TACE, radiotherapy, and molecular therapy, is important for avoiding lung metastasis.
Some studies have reported procedures for thrombectomy that do not need cardiac arrest. Although there is no need to block the coronary blood circulation, which can minimize the risk of myocardial ischemia and shorten the time of blockage in liver blood flow to reduce reperfusion injury, the tumor thrombus may be disseminated with the heart beating during thrombectomy. We chose cardiac arrest during the operation for this patient, as he was a young man with good cardiac reserve function and a lower probability of spreading the embolus. To avoid dissemination of the tumor, some experts believe that placing a filter is useful for decreasing the incidence of metastasis before surgery. Considering the possible complications and prolonged waiting times with this patient, we did not place the filter before surgery.
HCC with a tumor thrombus extending into RA has a significant impact on the survival of patients. Studies focusing on this type of HCC are insufficient, and more studies are warranted to further elucidate this condition.
We appreciate the patient’s generous support for this report.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Karaca CA, Veerankuttyh FH S-Editor: Wu YXJ L-Editor: Wang TQ P-Editor: Liu JH
| 1. | Koo JE, Kim JH, Lim YS, Park SJ, Won HJ, Sung KB, Suh DJ. Combination of transarterial chemoembolization and three-dimensional conformal radiotherapy for hepatocellular carcinoma with inferior vena cava tumor thrombus. Int J Radiat Oncol Biol Phys. 2010;78:180-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 81] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 2. | Cuevas C, Raske M, Bush WH, Takayama T, Maki JH, Kolokythas O, Meshberg E. Imaging primary and secondary tumor thrombus of the inferior vena cava: multi-detector computed tomography and magnetic resonance imaging. Curr Probl Diagn Radiol. 2006;35:90-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 41] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 3. | Kokudo T, Hasegawa K, Matsuyama Y, Takayama T, Izumi N, Kadoya M, Kudo M, Kubo S, Sakamoto M, Nakashima O, Kumada T, Kokudo N; Liver Cancer Study Group of Japan. Liver resection for hepatocellular carcinoma associated with hepatic vein invasion: A Japanese nationwide survey. Hepatology. 2017;66:510-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 140] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 4. | Wang Y, Yuan L, Ge RL, Sun Y, Wei G. Survival benefit of surgical treatment for hepatocellular carcinoma with inferior vena cava/right atrium tumor thrombus: results of a retrospective cohort study. Ann Surg Oncol. 2013;20:914-922. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 117] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 5. | Llovet JM, Bustamante J, Castells A, Vilana R, Ayuso Mdel C, Sala M, Brú C, Rodés J, Bruix J. Natural history of untreated nonsurgical hepatocellular carcinoma: rationale for the design and evaluation of therapeutic trials. Hepatology. 1999;29:62-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 839] [Cited by in RCA: 907] [Article Influence: 34.9] [Reference Citation Analysis (1)] |
| 6. | Chang JY, Ka WS, Chao TY, Liu TW, Chuang TR, Chen LT. Hepatocellular carcinoma with intra-atrial tumor thrombi. A report of three cases responsive to thalidomide treatment and literature review. Oncology. 2004;67:320-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 55] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 7. | Tsang J, Chan A, Chok K, Tsang F, Lo CM. Combined cavo-atrial thrombectomy and hepatectomy in hepatocellular carcinoma. Hepatobiliary Pancreat Dis Int. 2017;16:329-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 8. | Papp E, Keszthelyi Z, Kalmar NK, Papp L, Weninger C, Tornoczky T, Kalman E, Toth K, Habon T. Pulmonary embolization as primary manifestation of hepatocellular carcinoma with intracardiac penetration: a case report. World J Gastroenterol. 2005;11:2357-2359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 22] [Cited by in RCA: 19] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 9. | Sung AD, Cheng S, Moslehi J, Scully EP, Prior JM, Loscalzo J. Hepatocellular carcinoma with intracavitary cardiac involvement: a case report and review of the literature. Am J Cardiol. 2008;102:643-645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 55] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 10. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol. 2018;69:182-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5593] [Cited by in RCA: 6064] [Article Influence: 866.3] [Reference Citation Analysis (3)] |
| 11. | Kyokane T, Iyomasa S, Sawasaki N, Tojima Y, Matsuda M. A Case of Hepatocellular Carcinoma with Intra-atrial Tumor Thrombus Presenting Pulmonary Thromboemboli. Jpn J Gastroenterol Surg. 2010;43:398-404. [RCA] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 12. | Li AJ, Zhou WP, Lin C, Lang XL, Wang ZG, Yang XY, Tang QH, Tao R, Wu MC. Surgical treatment of hepatocellular carcinoma with inferior vena cava tumor thrombus: a new classification for surgical guidance. Hepatobiliary Pancreat Dis Int. 2013;12:263-269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 34] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 13. | Sugimoto H, Inoue S, Mori T, HirotaM, Takeda S, Kaneko T, Nakao A. Tumor Thrombus Removal with Hepatic Resection Using Cardiopulmonary Bypass in a Patient with Hepatocellular Carcinoma Extending into Right Atrium. Jpn J Gastroenterol Surg. 2004;37. [RCA] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 14. | Ariizumi SI, Kikuchi C, Tokitou F, Yamashita S, Kotera Y, Omori A, Kato T, Nemoto S, Niinami H, Yamamoto M. Cavo-atrial thrombectomy prior to hepatectomy for hepatocellular carcinoma with tumor thrombus in the right atrium: a case report. Surg Case Rep. 2019;5:57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 15. | Wakayama K, Kamiyama T, Yokoo H, Kakisaka T, Kamachi H, Tsuruga Y, Nakanishi K, Shimamura T, Todo S, Taketomi A. Surgical management of hepatocellular carcinoma with tumor thrombi in the inferior vena cava or right atrium. World J Surg Oncol. 2013;11:259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 68] [Article Influence: 5.7] [Reference Citation Analysis (0)] |