Published online Jul 26, 2021. doi: 10.12998/wjcc.v9.i21.5963
Peer-review started: November 30, 2020
First decision: March 11, 2021
Revised: March 30, 2021
Accepted: May 15, 2021
Article in press: May 15, 2021
Published online: July 26, 2021
Processing time: 232 Days and 17.8 Hours
A critically ill coronavirus disease 2019 (COVID-19) patient complicated by acute respiratory distress syndrome is reported. The patient survived following treatment with awake veno-venous extracorporeal membrane oxygenation (ECMO).
A 53-year-old male patient attended our hospital following a cough for 11 d and fever for 9 d. According to his computed tomography (CT) scan and real-time reverse transcription–polymerase chain reaction assay of a throat swab, nucleic acid was positive, confirming that he had COVID-19. He was subsequently transferred to the intensive care unit due to respiratory failure. The patient received antiviral drugs, a small dose of glucocorticoid, and respiratory support, including mechanical ventilation, but the treatment effect was poor. On the 28th day after admission, veno-venous ECMO and prone position ventilation (PPV) were performed, combined with awake ECMO and other comprehensive rehabilitation measures. On the 17th day of ECMO, the patient started to improve and his chest CT and lung compliance improved. ECMO was discontinued after 27 days, and mechanical ventilation was also discontinued after 9 days. The patient was then transferred to the rehabilitation department.
COVID-19 can damage lung tissues and cause evident inflammatory exudation, thus affecting oxygenation function. Awake ECMO, PPV, and comprehensive rehabilitation are effective in patients with critical COVID-19 and respiratory failure.
Core Tip: Coronavirus disease 2019 (COVID-19) symptoms include fever, cough, and breathing difficulties. Critically ill patients are often complicated by acute respiratory distress syndrome. Awake extracorporeal membrane oxygenation, prone position ventilation, and comprehensive rehabilitation are effective in patients with critical COVID-19 and respiratory failure.
- Citation: Zhang JC, Li T. Awake extracorporeal membrane oxygenation support for a critically ill COVID-19 patient: A case report. World J Clin Cases 2021; 9(21): 5963-5971
- URL: https://www.wjgnet.com/2307-8960/full/v9/i21/5963.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i21.5963
Coronavirus disease 2019 (COVID-19) is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), a novel coronavirus. The first case was confirmed in China, but COVID-19 has now become a pandemic. SARS-CoV2 infection leads to excessive exudation in pulmonary capillaries, manifesting as interstitial edema, and ground-glass opacities can be observed on imaging[1]. COVID-19 has various clinical manifestations, including fever and respiratory tract syndromes. Some COVID-19 patients experience mild symptoms, and no obvious abnormities are detected on chest computed tomography (CT), while a few patients have rapid severe respiratory failure, with a high mortality rate[2]. In these latter patients, a ventilator or extracorporeal membrane oxygenation (ECMO) is needed to support breathing[3].
ECMO can provide gas exchange and support cardiac function in patients with acute respiratory distress syndrome (ARDS) or cardiac failure. Following the CESAR trial in 2009, veno-venous (V-V) ECMO was established and is widely used to treat patients suffering from severe ARDS[4]. However, COVID-19 requires a relatively longer course of treatment than other known forms of viral pneumonia. In addition, it can progress quickly, thus contributing to respiratory failure[5]. Refractory hypoxemia is the most common clinical manifestation in critically ill COVID-19 patients. Previous studies have proved that awake ECMO can reduce the mortality rate of severe ARDS patients[6]. According to some reports, the combination of ECMO and prone position ventilation (PPV) can effectively cure viral pneumonia. From studies on its complications, it was discovered that the combination of ECMO and PPV is safe[7,8].
The prevalence of ARDS in COVID-19 patients is associated with extremely high mortality, especially before the introduction of effective antiviral treatment[9]. We report a patient with ARDS and COVID-19 who survived following the application of ECMO.
A 53-year-old man with a cough for 11 d, excess sputum, and fever for 9 d was admitted to our hospital.
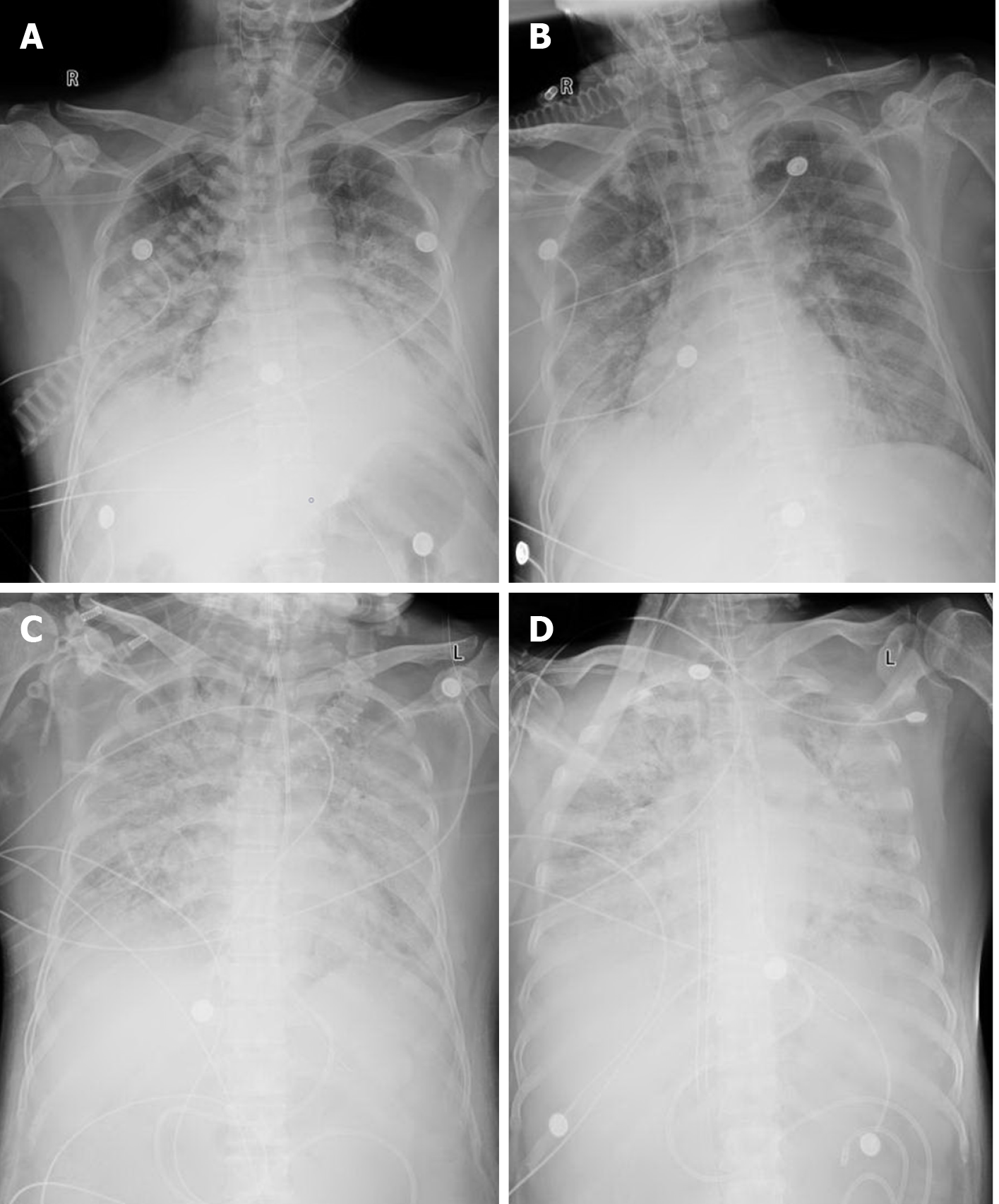
The patient attended our hospital with a respiratory tract infection. His chest X-ray indicated ground-glass opacities in the lower right lung (Figure 1A). SARS-CoV-2 infection was confirmed following real-time reverse transcription–polymerase chain reaction assay of a nasopharyngeal specimen. The patient was given high-flow nasal cannula oxygen therapy, arbidol, lopinavir + ritonavir, and methylprednisolone (40 mg/d for 8 d).
After 12 d of treatment, he developed respiratory failure, with partial pressure of oxygen/fraction of inspiration O2 (PaO2/FiO2) < 150 mmHg for 12 h and a respiratory rate > 30/min over 6 h, suggesting that the patient satisfied the criteria for mechanical ventilation and was admitted to the intensive care unit (ICU) for treatment.
He had a history of hypertension. The patient's respiratory function was normal without a history of smoking.
The patient did not have a history of drug allergies or genetic diseases.
On admission, symptoms included cough, expectoration of white sputum, and a body temperature of 38.7°C. Coarse breath sounds in both lungs with wet rales distributed at the base were identified on auscultation.
On hospital day 2, laboratory tests showed 185 mg/L of C-reactive protein, and the white blood cell count was 18.65 × 109/L. His renal and liver function tests were normal. Blood gas analysis after inserting the ventilator showed that the partial pressure of carbon dioxide and the partial pressure of oxygen were 47.00 mmHg and 59.00 mmHg, respectively.
A chest X-ray revealed increased density in the lower lung lobes, with the spread of plaques and a blurred border (Figure 1B). Over the next 96 h, lung protective strategies and fluid restriction were applied, but the patient’s condition did not improve, with poor lung compliance, hypoxemia, as well as dense opacifications due to diffuse lesions in the lungs (Figure 1C).
Critical COVID-19 requiring mechanical ventilation and ECMO, awake ECMO and rehabilitation treatment.
PPV was adopted, together with sedative and analgesic drugs, a muscle relaxant, lung recruitment and phlegm drainage, but no significant improvement was observed.
After 12 d of mechanical ventilation, a tracheotomy was performed, but testing showed that the patient was still positive for SARS-CoV-2. The patient’s PaO2/FiO2 dropped to 80 mmHg for 24 h, which met the standard for ECMO. The patient was given ECMO treatment on the 16th day of mechanical ventilation. In terms of the initiation parameter of ECMO, the mode was V-V, the intubation locations were the left femoral vein and right internal jugular vein, and the size of the tubes were 16F and 22F. A chest X-ray showed that the catheter was in a reasonable position and the pulmonary lesions were aggravated (Figure 1D).
During the running time, the activated partial thromboplastin time (APTT) was maintained at 40-60 (48.5 ± 5.3) s. The ECMO system was set at a blood flow of 3.5-4.0 L/min and a sweep flow rate of 4.0-7.0 L/min that were adjusted based on the PaCO2 obtained by blood gas analysis. The target coagulation profile was monitored by detecting the levels of APTT. During ECMO therapy, the target oxygenations were normal PaCO2 and PaO2.
With the support of ECMO, hemolysis occurred, and plasma-free hemoglobin was > 120 mg/dL. In addition, Enterococcus faecalis led to infectious shock and unstable blood volume, but there was no improvement after adequate fluid resuscitation. The ECMO catheter was replaced with right femoral vein and left internal jugular vein tubes 19F or 22F in size. The blood flow of ECMO subsequently improved, and hemolysis and infectious shock were controlled.
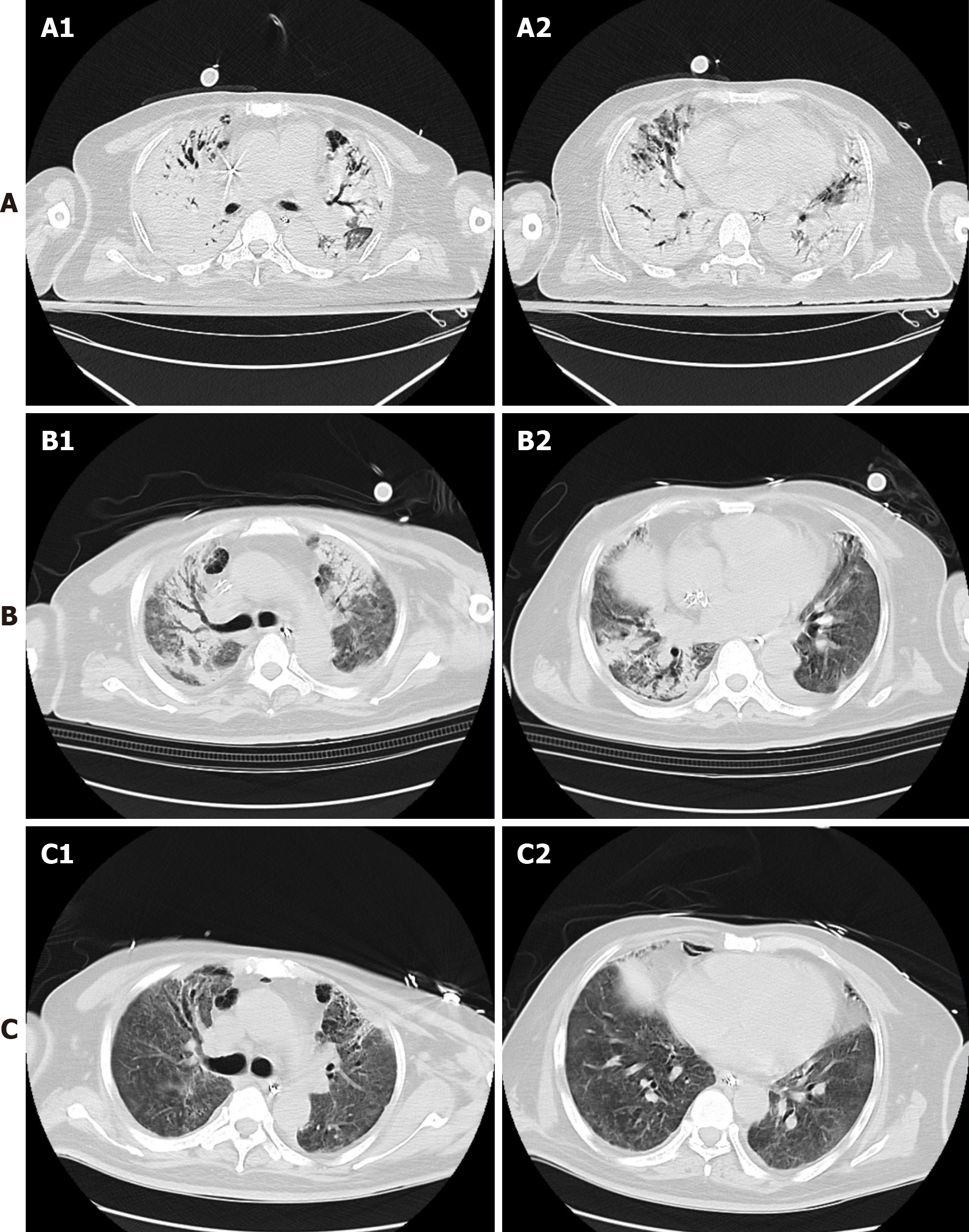
Re-examination of the CT scan illustrated lesions in both lungs and severe exudation (Figures 2A). In the early stage of ECMO, PPV was also carried out at the same time. The specific method of implementation was as follows: Four nurses helped to turn the patient, one nurse checked the ECMO regularly, and a respiratory physician was responsible for the management and protection of intubation. One doctor ensured the stability and flow of the ECMO tubes in case of pressure sores; therefore, materials were placed between the patient’s skin and the bed. To prevent pressure sores bandages were applied to fix the ECMO tube.
During ECMO (12 h/d), the ventilator mode setting was BIPAP, 26-30 cmH2O Peak inspiratory pressure, 6-8 cmH2O PEEP and 18-25 rpm RR. The parameters of PPV:PEEP were reduced to 2 cmH2O and 22-26 PIP, but other parameters were unchanged. In addition to the prone position, other care measures were also taken, including hand washing, closed endotracheal suction, and appropriate sedation to assist improvement (Figure 2B). On the 17th day after ECMO initiation, exhaled tidal volume increased to 6 mL/kg and chest radiography demonstrated that lung effusion had improved, after which the combination of awake ECMO and other comprehensive bed and floor rehabilitation exercises were adopted.
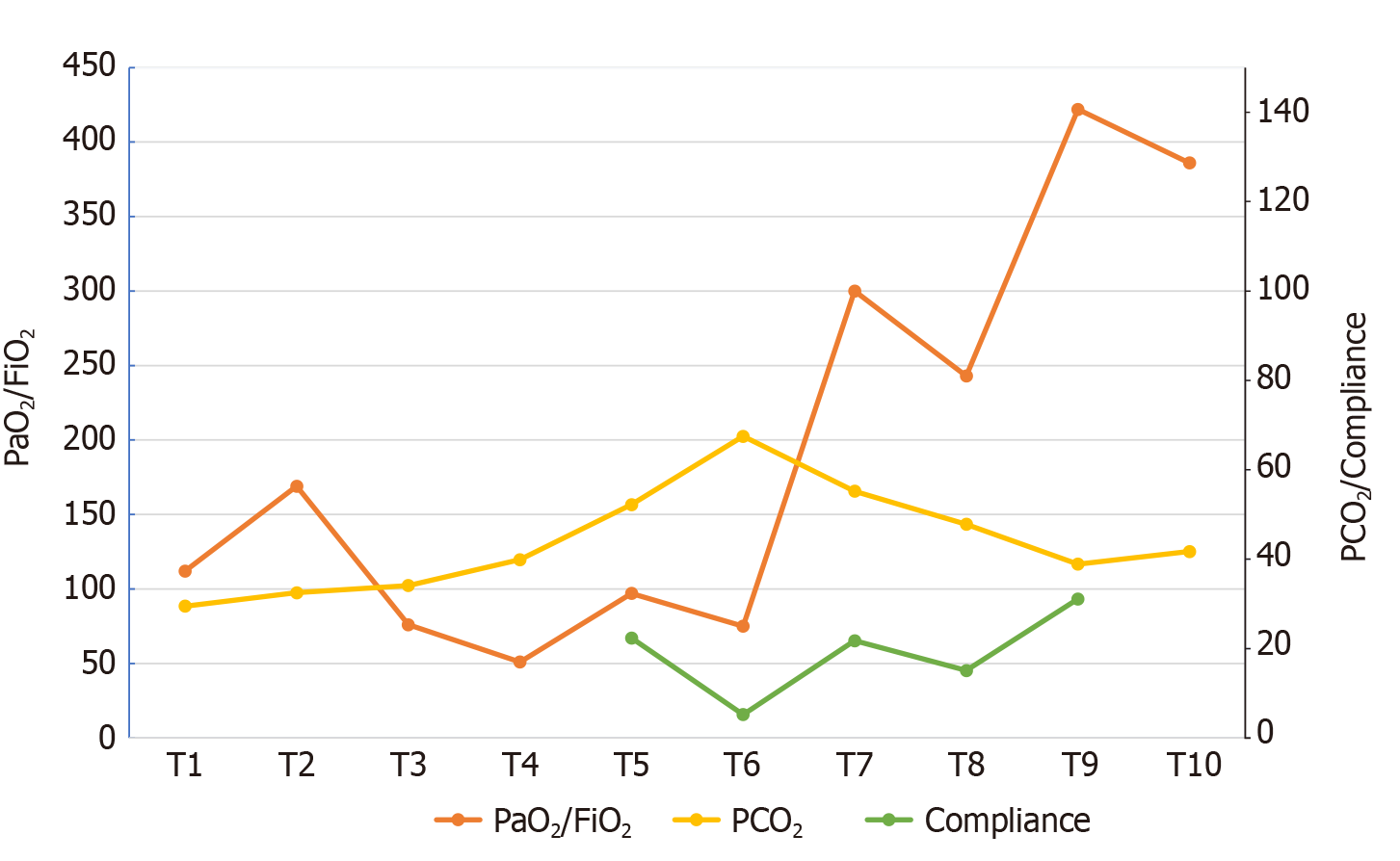
The patient performed rehabilitation therapy on the bed and floor. Bending arms and lowing legs were bed exercises. He also sat on the side of the bed for 30 min/day and performed passive functional leg exercises. Later, he also underwent training on his standing position. When the patient was transferred to the ICU, he received nutrition through a jejunal nutrient canal, aimed at 25-30 kcal/kg and the volume of food ingested was gradually increased. SARS-CoV-2 nucleic acid was negative 3 d after ECMO application. On the 27th day of ECMO support, chest CT showed improvement (Figure 2C), and the patient discontinued V-V ECMO. During the treatment period, the mechanical ventilator parameters and blood gas analysis were monitored (Figure 3). On the 9th day after ECMO removal, the patient was successfully closed casing. Following high flow nasal cannula oxygen therapy for 4 d, he was transferred to the rehabilitation department.
At present, initial reports from China and Italy suggest high mortality (2.16%) and stressed ICU capacity[10,11], with more than 10% mortality in some regions due to COVID-19. As a designated center, we admitted and treated a total of 104 patients, 78 of whom were seriously ill. Reports have indicated that COVID-19 patients who were hospitalized in the ICU rarely recovered. The patients supported with ECMO were reported to be less than one/week. Although this patient experienced long-term ECMO treatment, there are no reports on this treatment strategy.
SARS-CoV-2 belongs to the Coronaviridae family and betacoronavirus genus, and is the seventh coronavirus known to infect humans[12]. Coronaviruses are enveloped positive-sense, and single-stranded RNA viruses with mammalian and avian hosts. The SARS-CoV-2 virion is composed of a helical capsid formed by nucleocapsid (N) proteins bound to the RNA genome and an envelope made up of membrane (M) and envelope (E) proteins, coated with trimeric spike (S) proteins[13]. The S protein binds to the angiotensin-converting enzyme 2 receptor on the plasma membrane of type 2 pneumocytes and intestinal epithelial cells. After binding, the S protein is cleaved by a host membrane serine protease and TMPRSS2 to facilitate viral entry[14].
Multiple clinical trials are underway to define the potential roles of antiviral agents and specific immunomodulators[15]. Antiviral agents under investigation include inhibitors of endosome maturation (hydroxychloroquine), viral RNA-dependent RNA polymerase (remdesivir, favipiravir) and viral protein synthesis and maturation (lopinavir/ritonavir); immunomodulators under investigation include interferon-β and blockade of IL-6 receptor or IL-6 (tocilizumab, siltuximab, sarilumab)[16,17]. Passive immunization with convalescent plasma and active immunization strategies, involving live-attenuated virus, chimeric virus, subunits, nanoparticles, RNA, and DNA, are undergoing development and testing[18].
Our patient was an early case of COVID-19 transmission, with rapid progression and poor response to antiviral drugs after onset of the disease. Oxygenation becomes worse in patients after the use of non-invasive ventilation, which further confirmed that COVID-19 has a long disease course and oxygenation deteriorates rapidly, thus distinguishing it from traditional viral pneumonia[19]. Some COVID-19 patients may require the support of high-level pressure ventilation. However, due to high airway pressure, oxygen concentration, severe viral infection, along with protein exudation, mechanical ventilation can result in severe lung injury and decreased lung compliance[20].
The treatment for early critical patients is a conservative intubation strategy. However, when the disease progresses, with high systemic organ failure and a low cure rate, ECMO can be carried out to guarantee oxygenation. However, ECMO is an invasive procedure, with a high incidence rate, thereby requiring a well-equipped ECMO center and experienced and skilled medical personnel. In our center, 10 patients with severe COVID-19 received ECMO, and 7 of these patients were successfully treated. Our comprehensive treatment, with ECMO as a bridge, has resulted in significant improvements in patients suffering from COVID-19.
PPV can improve oxygenation in ARDS patients, especially when administered for a long period (17 h or 18 h)[21]. Two recent meta-analyses indicated that PPV can reduce the death rate of patients in the ICU[22,23]. Following ECMO treatment which improved oxygenation and stabilized circulation, our patient also received PPV. On CT, the distribution of lung lesions and atrophy of lung tissue were evident. Following PPV, the lung CT scan and clinical manifestations indicated that the treatment was effective.
Phlegm and alveolar lavage fluid were negative for SARS-CoV-2. On the 17th day, he accepted awake ECMO and his lungs markedly improved. During this process, an artificial airway and positive pressure ventilation cannot prevent ventilator-associated pneumonia and ventilator-induced lung injury[24]. In the early stage of his rehabilitation, sedative and analgesic drugs were reduced to maintain consciousness. Spontaneous ventilation can promote the even distribution of lung ventilation, and reduce the occurrence of ventilator-induced diaphragm dysfunction[25].
In addition to awake ECMO, the patient adopted various rehabilitation measures, including standing by the bed and riding a bike, after which his muscle power improved. Awake ECMO allowed the patient to change posture and he was encouraged to cough, which had apparent advantages in phlegm drainage[26]. The patient also required a high level of care and protection, which was difficult to implement during awake ECMO. It is difficult for elderly patients to perform rehabilitation exercise due to their poor basic ability; therefore, more health care staff and workers are needed, which is a massive challenge.
Multiple complications can develop during ECMO, and hemorrhage is the most common complication. In the present case, a conservative anticoagulation strategy was carried out, due to the long disease course, early hemorrhage in the digestive tract, and a high risk of bleeding. Some studies have demonstrated that in patients with severe COVID-19, as the ECMO time increases, the risk of bleeding rises[27,28]. An essential steps is to control bleeding during the ECMO process[29]. The APTT was set between 40 s and 60 s. In our experience, in addition to the combination of awake ECMO and active rehabilitation, a low level anticoagulation strategy is necessary. The patient should be closely monitored in case of hemorrhage complications which should be timely managed.
As a bridge to recovery, V-V ECMO was quickly introduced to support the patient, who was discharged on the 13th day fully recovered, and without adverse effects. Before he was discharged from our center, his lung function had improved and he did not receive oxygen therapy. Another 10 cases accepted combination treatment of ECMO and PPV, and 7 of these patients were able to breathe without ECMO. Therefore, this method deserves to be promoted.
Severe COVID-19 patients have a high rate of mortality, especially older patients. Conventional ventilator treatment is ineffective in critically ill patients. Although ECMO can lead to multiple complications, its clinical effects cannot be neglected. For some patients, if the effects of conventional ventilation are ineffective, ECMO treatment can be considered.
The authors would like to thank all participants in the study, the nurses and clinical staff who provided care for the patients. Finally, Zhang JC would particularly like to thank Zhao YW for invaluable support.
Manuscript source: Unsolicited manuscript
Specialty type: Critical care medicine
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Zelai NT S-Editor: Yan JP L-Editor: Webster JR P-Editor: Wang LYT
| 1. | Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P, Liu H, Zhu L, Tai Y, Bai C, Gao T, Song J, Xia P, Dong J, Zhao J, Wang FS. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420-422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5228] [Cited by in RCA: 5786] [Article Influence: 1157.2] [Reference Citation Analysis (2)] |
| 2. | Yao H, Lu X, Chen Q, Xu K, Chen Y, Cheng M, Chen K, Cheng L, Weng T, Shi D, Liu F, Wu Z, Xie M, Wu H, Jin C, Zheng M, Wu N, Jiang C, Li L. Patient-derived SARS-CoV-2 mutations impact viral replication dynamics and infectivity in vitro and with clinical implications in vivo. Cell Discov. 2020;6:76. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 3. | Yang X, Yu Y, Xu J, Shu H, Xia J, Liu H, Wu Y, Zhang L, Yu Z, Fang M, Yu T, Wang Y, Pan S, Zou X, Yuan S, Shang Y. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020;8:475-481. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6231] [Cited by in RCA: 6660] [Article Influence: 1332.0] [Reference Citation Analysis (0)] |
| 4. | Peek GJ, Mugford M, Tiruvoipati R, Wilson A, Allen E, Thalanany MM, Hibbert CL, Truesdale A, Clemens F, Cooper N, Firmin RK, Elbourne D; CESAR trial collaboration. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet. 2009;374:1351-1363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2740] [Cited by in RCA: 2332] [Article Influence: 145.8] [Reference Citation Analysis (0)] |
| 5. | Bhatraju PK, Ghassemieh BJ, Nichols M, Kim R, Jerome KR, Nalla AK, Greninger AL, Pipavath S, Wurfel MM, Evans L, Kritek PA, West TE, Luks A, Gerbino A, Dale CR, Goldman JD, O'Mahony S, Mikacenic C. Covid-19 in Critically Ill Patients in the Seattle Region - Case Series. N Engl J Med. 2020;382:2012-2022. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1819] [Cited by in RCA: 1862] [Article Influence: 372.4] [Reference Citation Analysis (0)] |
| 6. | Yeo HJ, Cho WH, Kim D. Awake extracorporeal membrane oxygenation in patients with severe postoperative acute respiratory distress syndrome. J Thorac Dis. 2016;8:37-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 7. | Schmidt M, Stewart C, Bailey M, Nieszkowska A, Kelly J, Murphy L, Pilcher D, Cooper DJ, Scheinkestel C, Pellegrino V, Forrest P, Combes A, Hodgson C. Mechanical ventilation management during extracorporeal membrane oxygenation for acute respiratory distress syndrome: a retrospective international multicenter study. Crit Care Med. 2015;43:654-664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 149] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 8. | Voelker MT, Jahn N, Bercker S, Becker-Rux D, Köppen S, Kaisers UX, Laudi S. [Prone positioning of patients during venovenous extracorporeal membrane oxygenation is safe and feasible]. Anaesthesist. 2016;65:250-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 9. | Zhang L, Li B, Jia P, Pu J, Bai B, Li Y, Zhu P, Li L, Zeng G, Zhao X, Dong S, Liu M, Zhang N. [An analysis of global research on SARS-CoV-2]. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2020;37:236-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 10. | Grasselli G, Pesenti A, Cecconi M. Critical Care Utilization for the COVID-19 Outbreak in Lombardy, Italy: Early Experience and Forecast During an Emergency Response. JAMA. 2020;323:1545-1546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1455] [Cited by in RCA: 1406] [Article Influence: 281.2] [Reference Citation Analysis (0)] |
| 11. | Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, Liu Y, Wei Y, Xia J, Yu T, Zhang X, Zhang L. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507-513. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14869] [Cited by in RCA: 12978] [Article Influence: 2595.6] [Reference Citation Analysis (1)] |
| 12. | Seyran M, Pizzol D, Adadi P, El-Aziz TMA, Hassan SS, Soares A, Kandimalla R, Lundstrom K, Tambuwala M, Aljabali AAA, Lal A, Azad GK, Choudhury PP, Uversky VN, Sherchan SP, Uhal BD, Rezaei N, Brufsky AM. Questions concerning the proximal origin of SARS-CoV-2. J Med Virol. 2021;93:1204-1206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 13. | Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, Li B, Huang CL, Chen HD, Chen J, Luo Y, Guo H, Jiang RD, Liu MQ, Chen Y, Shen XR, Wang X, Zheng XS, Zhao K, Chen QJ, Deng F, Liu LL, Yan B, Zhan FX, Wang YY, Xiao GF, Shi ZL. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270-273. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15248] [Cited by in RCA: 14131] [Article Influence: 2826.2] [Reference Citation Analysis (1)] |
| 14. | Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Müller MA, Drosten C, Pöhlmann S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020; 181: 271-280. e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11946] [Cited by in RCA: 14274] [Article Influence: 2854.8] [Reference Citation Analysis (0)] |
| 15. | Oberfeld B, Achanta A, Carpenter K, Chen P, Gilette NM, Langat P, Said JT, Schiff AE, Zhou AS, Barczak AK, Pillai S. SnapShot: COVID-19. Cell 2020; 181: 954-954. e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 94] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 16. | McCreary EK, Pogue JM. Coronavirus Disease 2019 Treatment: A Review of Early and Emerging Options. Open Forum Infect Dis. 2020;7:ofaa105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 203] [Cited by in RCA: 193] [Article Influence: 38.6] [Reference Citation Analysis (0)] |
| 17. | Khan Z, Ghafoor D, Khan A, Ualiyeva D, Khan SA, Bilal H, Khan B, Sajjad W. Diagnostic approaches and potential therapeutic options for coronavirus disease 2019. New Microbes New Infect. 2020;38:100770. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 18. | Zhao M, Zhang J, Li H, Luo Z, Ye J, Xu Y, Wang Z, Ye D, Liu J, Li D, Wang M, Wan J. Recent progress of antiviral therapy for coronavirus disease 2019. Eur J Pharmacol. 2021;890:173646. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 19. | Arentz M, Yim E, Klaff L, Lokhandwala S, Riedo FX, Chong M, Lee M. Characteristics and Outcomes of 21 Critically Ill Patients With COVID-19 in Washington State. JAMA. 2020;323:1612-1614. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1511] [Cited by in RCA: 1624] [Article Influence: 324.8] [Reference Citation Analysis (0)] |
| 20. | Borba MGS, Val FFA, Sampaio VS, Alexandre MAA, Melo GC, Brito M, Mourão MPG, Brito-Sousa JD, Baía-da-Silva D, Guerra MVF, Hajjar LA, Pinto RC, Balieiro AAS, Pacheco AGF, Santos JDO Jr, Naveca FG, Xavier MS, Siqueira AM, Schwarzbold A, Croda J, Nogueira ML, Romero GAS, Bassat Q, Fontes CJ, Albuquerque BC, Daniel-Ribeiro CT, Monteiro WM, Lacerda MVG; CloroCovid-19 Team. Effect of High vs Low Doses of Chloroquine Diphosphate as Adjunctive Therapy for Patients Hospitalized With Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection: A Randomized Clinical Trial. JAMA Netw Open. 2020;3:e208857. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 762] [Cited by in RCA: 688] [Article Influence: 137.6] [Reference Citation Analysis (0)] |
| 21. | Silversides JA, Ferguson ND. Clinical review: Acute respiratory distress syndrome - clinical ventilator management and adjunct therapy. Crit Care. 2013;17:225. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 22. | Sud S, Friedrich JO, Taccone P, Polli F, Adhikari NK, Latini R, Pesenti A, Guérin C, Mancebo J, Curley MA, Fernandez R, Chan MC, Beuret P, Voggenreiter G, Sud M, Tognoni G, Gattinoni L. Prone ventilation reduces mortality in patients with acute respiratory failure and severe hypoxemia: systematic review and meta-analysis. Intensive Care Med. 2010;36:585-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 458] [Cited by in RCA: 353] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 23. | Sud S, Sud M, Friedrich JO, Adhikari NK. Effect of mechanical ventilation in the prone position on clinical outcomes in patients with acute hypoxemic respiratory failure: a systematic review and meta-analysis. CMAJ. 2008;178:1153-1161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 103] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 24. | Nishimura M, Honda O, Tomiyama N, Johkoh T, Kagawa K, Nishida T. Body position does not influence the location of ventilator-induced lung injury. Intensive Care Med. 2000;26:1664-1669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 25. | Santana MC, Garcia CS, Xisto DG, Nagato LK, Lassance RM, Prota LF, Ornellas FM, Capelozzi VL, Morales MM, Zin WA, Pelosi P, Rocco PR. Prone position prevents regional alveolar hyperinflation and mechanical stress and strain in mild experimental acute lung injury. Respir Physiol Neurobiol. 2009;167:181-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 26. | Langer T, Santini A, Bottino N, Crotti S, Batchinsky AI, Pesenti A, Gattinoni L. "Awake" extracorporeal membrane oxygenation (ECMO): pathophysiology, technical considerations, and clinical pioneering. Crit Care. 2016;20:150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 145] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 27. | Ellouze O, Abbad X, Constandache T, Missaoui A, Berthoud V, Daily T, Aho S, Bouchot O, Bouhemad B, Guinot PG. Risk Factors of Bleeding in Patients Undergoing Venoarterial Extracorporeal Membrane Oxygenation. Ann Thorac Surg. 2021;111:623-628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 28. | Combes A, Hajage D, Capellier G, Demoule A, Lavoué S, Guervilly C, Da Silva D, Zafrani L, Tirot P, Veber B, Maury E, Levy B, Cohen Y, Richard C, Kalfon P, Bouadma L, Mehdaoui H, Beduneau G, Lebreton G, Brochard L, Ferguson ND, Fan E, Slutsky AS, Brodie D, Mercat A; EOLIA Trial Group; REVA, and ECMONet. Extracorporeal Membrane Oxygenation for Severe Acute Respiratory Distress Syndrome. N Engl J Med. 2018;378:1965-1975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1417] [Cited by in RCA: 1485] [Article Influence: 212.1] [Reference Citation Analysis (0)] |
| 29. | Murphy DA, Hockings LE, Andrews RK, Aubron C, Gardiner EE, Pellegrino VA, Davis AK. Extracorporeal membrane oxygenation-hemostatic complications. Transfus Med Rev. 2015;29:90-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 287] [Article Influence: 26.1] [Reference Citation Analysis (0)] |