Published online Jun 16, 2021. doi: 10.12998/wjcc.v9.i17.4388
Peer-review started: February 2, 2021
First decision: February 28, 2021
Revised: March 14, 2021
Accepted: April 8, 2021
Article in press: April 8, 2021
Published online: June 16, 2021
Processing time: 112 Days and 19.7 Hours
Primary intratracheal schwannoma is an extremely rare type of benign airway tumor, especially in adolescents. The presenting symptoms are typically prolonged cough and wheezing that can be misdiagnosed as asthma in adolescent patients.
A 16-year-old adolescent girl admitted to a local hospital with symptoms of an irritating cough and wheezing was diagnosed with bronchial asthma and treated with budesonide and formoterol. Over the next year, the patient's wheezing and coughing symptoms gradually worsened and the antiasthma treatment was ineffective. One week prior to this admission, the patient developed dyspnea after catching a cold and was transferred to our hospital with a diagnosis of severe asthma. However, chest computed tomography and bronchoscopy showed a mass in the trachea. Primary intratracheal schwannoma was diagnosed by biopsy. Her symptoms were relieved by endoscopic resection by electrosurgical snaring combined with argon plasma coagulation. No relapse occurred during an 18 mo follow-up.
Primary intratracheal schwannoma should be considered in the differential diagnosis in adolescents with recurrent asthma-like attacks.
Core Tip: Primary intratracheal schwannoma is a rare benign airway tumor in adolescents. The presenting symptoms are typically prolonged cough and wheezing, and antiasthma treatment was ineffective in this patient. Chest computed tomography and bronchoscopy should be performed as soon as possible to clarify the diagnosis. Here we report a 16-year-old female patient with a primary intratracheal schwannoma misdiagnosed as severe asthma. This case report is intended to increase awareness for it and help avoid future misdiagnoses by physicians.
- Citation: Huang HR, Li PQ, Wan YX. Primary intratracheal schwannoma misdiagnosed as severe asthma in an adolescent: A case report. World J Clin Cases 2021; 9(17): 4388-4394
- URL: https://www.wjgnet.com/2307-8960/full/v9/i17/4388.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i17.4388
Primary tracheal tumors can be either benign or malignant, but malignant tumors are more common. Benign tracheal tumors have a low incidence, and the common types include papillomas, hamartomas, and hemangiomas. Primary intratracheal schwannoma is an extremely rare benign airway tumor, with an incidence of about 0.2% and few published reports of cases in child or adolescents[1-3]. Primary intratracheal schwannoma has no characteristic clinical manifestations in the early stage, but when the tumor grows to a certain size, it can cause symptoms associated with blockage of the tracheal lumen that can be easily misdiagnosed. Here we report an adolescent patient with a primary intratracheal schwannoma misdiagnosed as severe asthma.
A 16-year-old adolescent girl had an intermittent cough with shortness of breath and wheezing for 1 year that had become worse in the previous week.
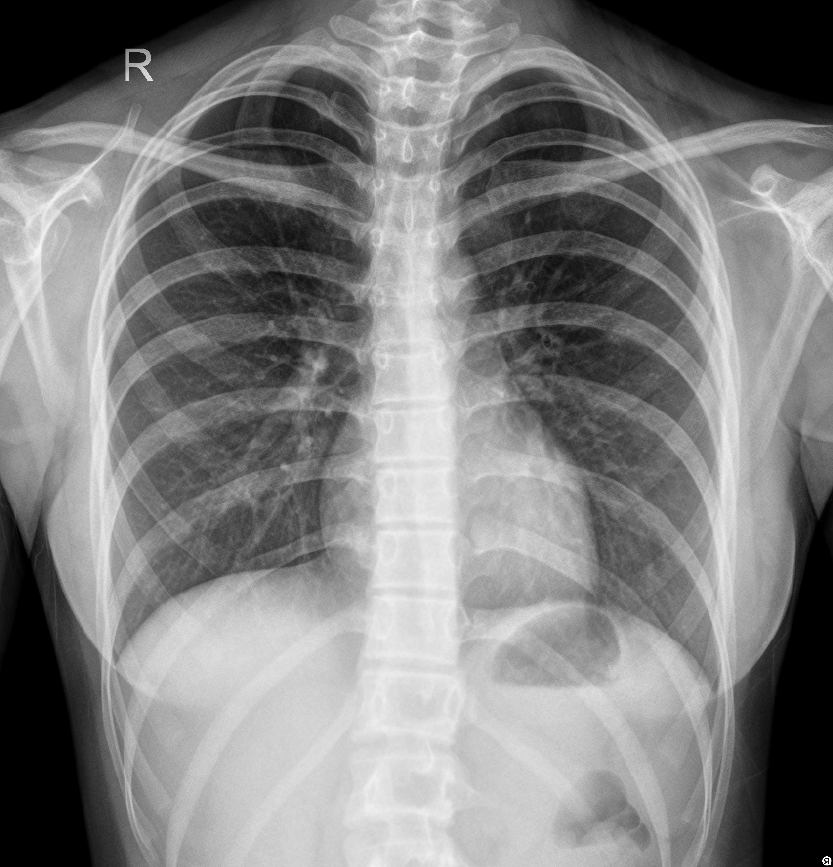
The patient visited a local hospital with symptoms of an irritating cough and wheezing. A chest X-ray showed no abnormalities. She was diagnosed with bronchial asthma and treated with budesonide and formoterol. Over the next year, the wheezing and cough symptoms gradually worsened, and the treatment was not effective. One week before admission, the patient's cough and wheezing symptoms worsened after catching a cold, and she experienced wheezing and a feeling of suffocation even when resting. A chest X-ray revealed blurred markings in both lungs (Figure 1). Allergen testing was not performed. She was diagnosed with acute onset of asthma and treated with Ventolin (salbutamol sulfate solution for inhalation) and intravenous methylprednisolone for 1 wk, but the symptoms were not relieved. She was then transferred to the respiratory department of our hospital.
The patient had no other previous medical history.
The patient had no relevant family medical history.
The patient had a body temperature of 36.7 ℃, blood pressure of 128/80 mmHg, heart rate of 103 beats/min, respiratory rate of 28 breaths/min, and an SPO2 of 98%. The patient had clear consciousness and was in a semi-supine position. The three concave sign was positive, her inspiratory phase was prolonged, and bidirectional wheezing could be heard.
Routine blood tests showed a white blood cell count of 12.0 × 109/L, hemoglobin of 122 g/L, and a neutrophil ratio of 85%. Arterial blood gas analysis showed a pH of 7.45, a PaO2 60 mmHg, and a PaCO2 of 33 mmHg. Liver and kidney function was normal and neuron-specific enolase was 16.5 ng/mL (normal range: 0-15.2 ng/mL). The patient did not undergo pulmonary function tests due because of her dyspnea.
Chest computed tomography (CT) at the T2/T3 vertebral level showed a nodular mass in the trachea that obstructed about 90% of the lumen without invading the tracheal wall and its surrounding tissues (Figure 2).
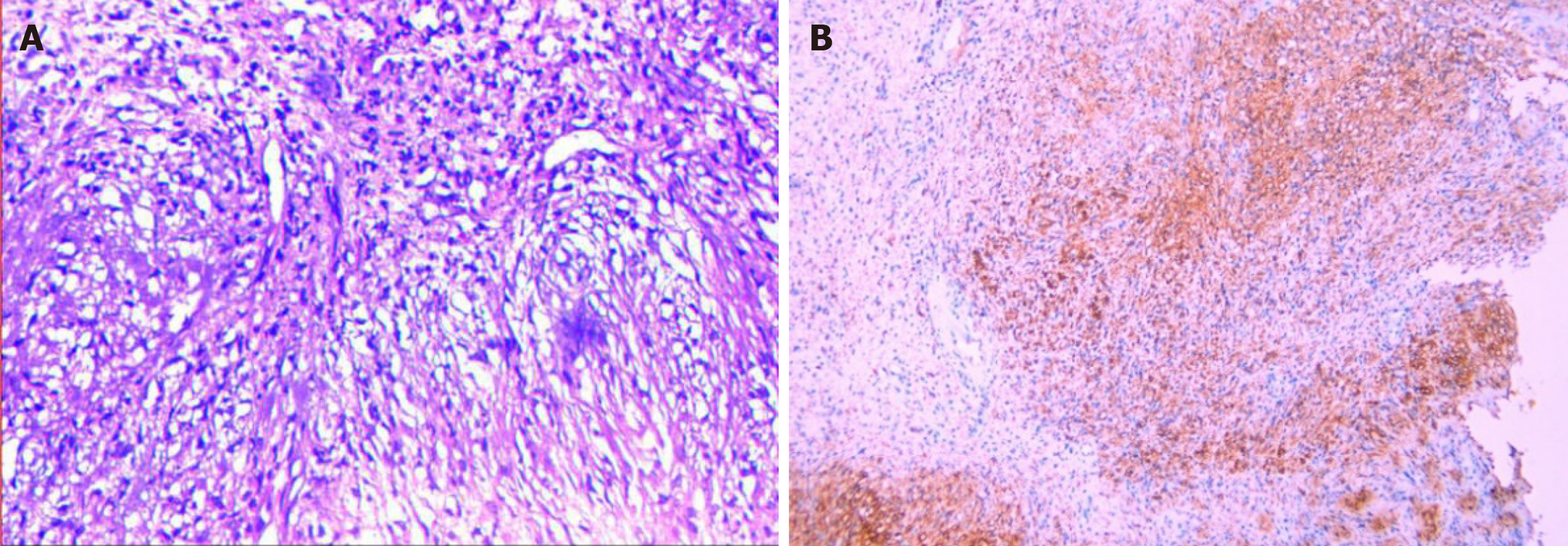
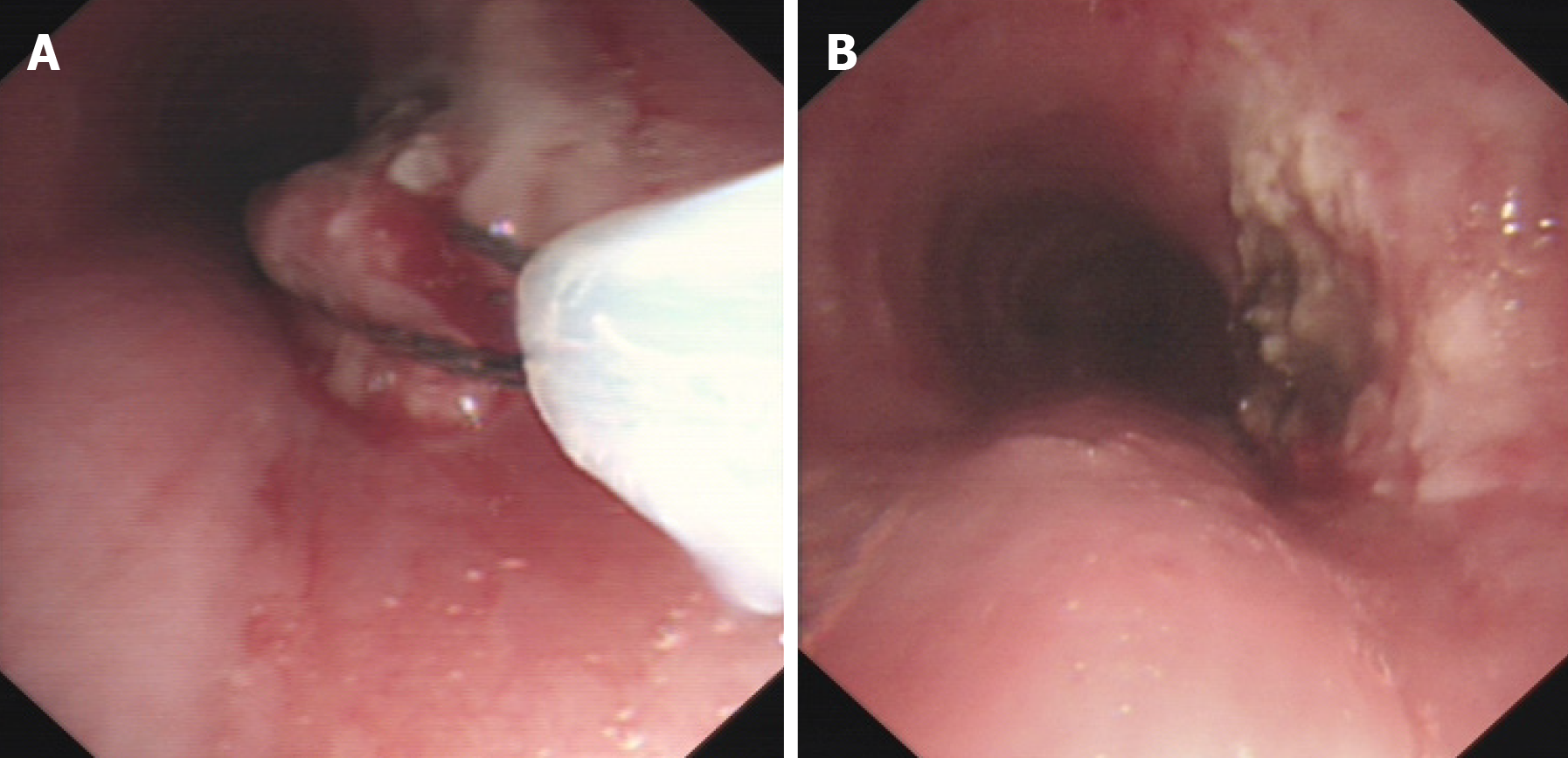
A bronchoscopy was done with the patient's consent to determine the nature of the intratracheal mass. The bronchoscopy showed a pedicled dumbbell-shaped reddish-brown mass with a smooth surface and small blood vessels in the middle part of the trachea, about 4 cm below the vocal cords (Figure 3). The mass obstructed about 90% of the airway. Pathological examination of the tracheal mass showed that the tumor cells were long and shuttle-shaped, with an uneven distribution and fenestrated arrangement in some areas (Figure 4A). The sample cells were incubated with mouse antihuman monoclonal antibodies against S100, desmin, CD117, and Ki-67 (MXB Biotechnologies, China). Immunohistochemistry (IHC) was performed with a VENTANA BenchMARK Ultra automated staining instrument following the manufacturer’s instructions. IHC revealed S-100 (++), desmin (-), CD117 (-), and a Ki-67 positivity of < 1%. The pathological diagnosis was tracheal schwannoma.
Primary intratracheal schwannoma.
Using an ERBR ICC-200 high-frequency electrosurgical unit, the tumor was removed by electronic snaring with a flexible bronchoscope, followed by further clearance of residual tissue at the base of the tumor by argon plasma coagulation. After the tumor was removed, its root was visible in the mid trachea at approximately a 5 o'clock position (Figure 5).
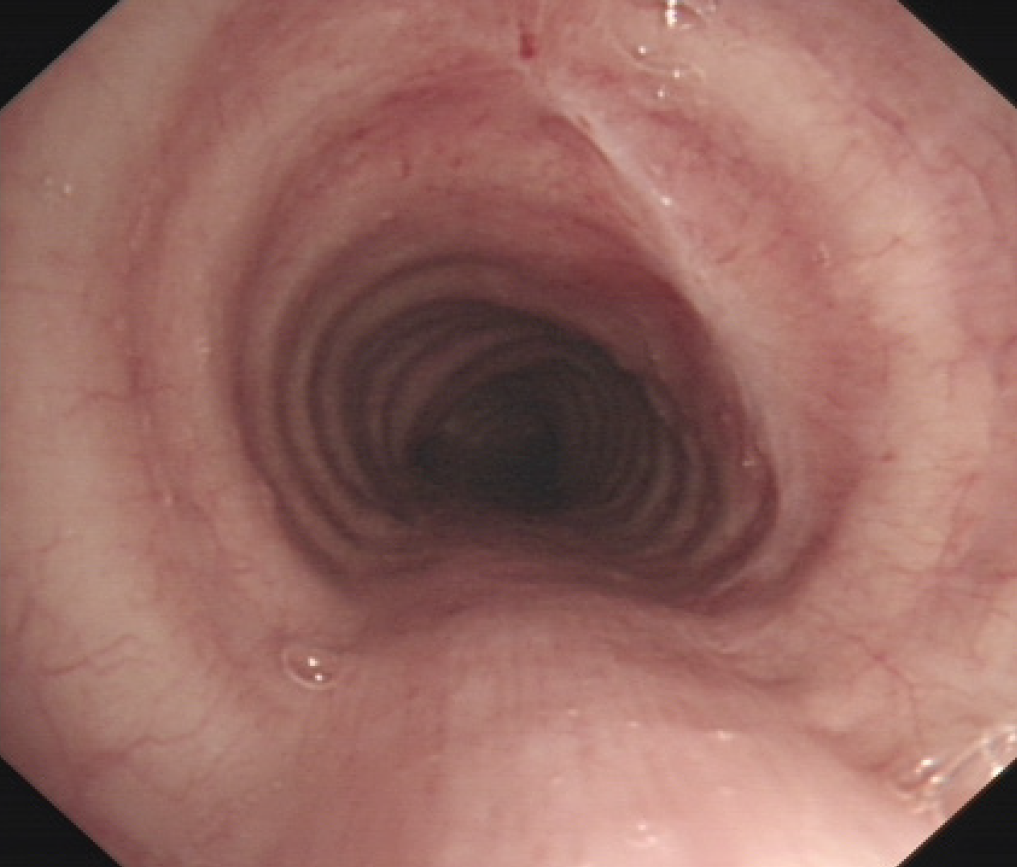
The patient's cough and dyspnea symptoms disappeared immediately following endoscopic resection of the tumor. During an 18-mo postoperative follow-up, the patient had no early or late complications or tumor recurrence. Bronchoscopic surveillance showed a minor scar on the trachea at the site of the original lesion (Figure 6). The patient is still being followed.
Schwannomas, also known as neurilemmomas, originate from the cell membranes of neurons derived from the embryonic neural crest. Schwannomas have a low incidence and can occur in all parts of the body, but are more common in the head/neck and the flexors of all four extremities[4]. Schwannomas occur rarely in the airway. Intratracheal schwannomas were first described by Straus and Guckien[5] in 1951. Intratracheal schwannomas are benign tumors, most often located near the ramus of the lower 1/3 of the trachea, followed by the upper 1/3 and then the middle 1/3 of the trachea. They have an intact envelope, grow slowly, and cause no symptoms in the early stage[6-10]. In the present case, the tumor was located in the middle portion of the trachea, a site that is rarely encountered in the clinical setting, and the diagnosis can be easily missed.
Intratracheal schwannomas are more common in young adults, especially in women. The common clinical manifestations of intratracheal schwannomas include cough, shortness of breath, dyspnea, and other symptoms of upper airway obstruction, which can be related to body position. Occasionally, patients also suffer from hemoptysis, hoarseness, and chest pain[11]. Due to the nonspecific nature of these symptoms, diagnosis of this rare condition is often delayed. The average time to diagnosis from the onset of initial symptoms was approximately 17 mo[7,12], and half the patients are reported to be misdiagnosed with asthma before the diagnosis is finally confirmed[13]. In the early stage, the tumor is small and grows slowly, often presenting as an irritating dry cough that can persist for months or even years. As the tumor grows and obstructs more than 1/2 of the tracheal lumen or when the resistance increases significantly, the patient gradually develops shortness of breath, dyspnea, and other symptoms. At that stage, chest X-ray films, especially anteroposterior films, show no obvious abnormalities in 30%-75% of the patients because the tumor is still covered by mediastinal soft tissues[8,14]. As early treatment with glucocorticoids and bronchodilator can temporarily relieve symptoms, intratracheal schwannomas are often misdiagnosed as asthma, and further diagnosis and treatment are often delayed. When the tracheal obstruction worsens and the above treatments are ineffective, the disease is easily misdiagnosed as refractory asthma or severe asthma. However, there are differences in symptoms and signs between those two conditions. Wheezing caused by schwannomas is mostly manifested as progressive aggravation, and both expiratory and inspiratory wheezing can be heard during auscultation. The sound is strong in the proximal part of the airway and gradually decreases in the distal part, showing inspiratory dyspnea. In contrast, asthma is reversible, episodic, and recurrent, causing expiratory dyspnea, and the sound is mostly diffuse and in the expiration phase. This patient was misdiagnosed with bronchial asthma at a local hospital for up to 1 year. The possibility of an intratracheal schwannoma should be considered in adolescent patients with a long-term irritating cough accompanied mostly by inspiratory dyspnea, especially in adolescents who have failed to respond to conventional treatments.
Thoracic CT and 3-dimensional reconstruction are valuable in identifying the mass, assessing the location and size of the tracheal tumor, the degree of airway obstruction, the involvement of the mediastinum, and in determining treatment options[7,10,15]. The CT presentation of schwannomas is diverse, especially multiple forms of heterogeneous enhancement that can be observed on contrast-enhanced CT. Because of the lack of specific symptoms, intratracheal schwannomas can be easily misdiagnosed as other benign tumors of the trachea[10].
Bronchoscopy is the most effective method to diagnose intratracheal schwannomas. The main bronchoscopic manifestations include: (1) A broad base, with the mass growing along the longitudinal axis of the tracheobronchus and a round or oval protrusion into the lumen; (2) A pedicled tumor with a polyp-like growth into the lumen; and (3) A dumbbell-shaped tumor growing into the lumen. The final confirmation of the diagnosis depends on histopathological evaluation. Biopsy sampling is often difficult because of the tough texture of the tumor. If the biopsy site is shallow, microscopy may only reveal chronic inflammation of the mucosa. Therefore, the biopsy site should be sufficiently deep. If necessary, resected tissues may also be sent for pathological examination. Pathologically, schwannomas often have an intact envelope, a well-arranged internal structure, cell-rich bundles, (an Antoni type-A area), and a loose mucus-like reticular zone (an Antoni type-B area. The tumor cells have the morphological characteristics of Schwann cells in terms of immunophenotype and ultrastructure. S-100(+) immunohistochemical staining also helps to confirm the diagnosis[7,16].
The current treatment options for intratracheal schwannomas include surgical resection plus airway reconstruction and interventional bronchoscopic resection[7,8]. The choice of treatment depends on tumor location, relationship of the mass with surrounding tissues, tracheal wall infiltration, risk of tracheal resection, and presence of extratracheal components[6,17]. With the rapid development of respiratory interventional techniques in recent years, bronchoscopic cutting with electrocautery snaring, argon plasma coagulation, cryotherapy, and laser resection have also become available[18-20]. High-frequency electrocoagulation ablation is a safe treatment for endotracheal tumors, with a low incidence of complications.
Intratracheal schwannoma is a relatively rare benign airway tumor. Its diagnosis is difficult because of the lack of specific clinical presentations and nonspecific chest X-ray findings. The incidence of intratracheal schwannoma is low in children and adolescents, and misdiagnosis may easily occur. If standard treatment is ineffective in patients with recurrent asthma-like attacks, especially children or adolescents, then chest CT and bronchoscopy should be performed as soon as possible to clarify the diagnosis.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, research and experimental
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Elpek GO, Li XM S-Editor: Gong ZM L-Editor: Filipodia P-Editor: Wang LL
| 1. | Yukawa T, Shimizu K, Hirami Y, Okita R, Saisho S, Maeda A, Yasuda K, Nakata M. A case report of intrapulmonary schwannoma. Gen Thorac Cardiovasc Surg. 2014;62:252-254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 2. | Ohtsuka T, Nomori H, Naruke T, Orikasa H, Yamazaki K, Suemasu K. Intrapulmonary schwannoma. Jpn J Thorac Cardiovasc Surg. 2005;53:154-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 3. | Zhang L, Tang W, Hong QS, Lv PF, Jiang KM, Du R. Case report: A tracheobronchial schwannoma in a child. Respir Med Case Rep. 2020;30:101047. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 4. | Grillo HC, Mathisen DJ. Primary tracheal tumors: treatment and results. Ann Thorac Surg. 1990;49:69-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 296] [Cited by in RCA: 268] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 5. | Straus GD, Guckien JL. Schwannoma of the tracheobronchial tree. A case report. Ann Otol Rhinol Laryngol. 1951;60:242-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 6. | Kang HJ, Shin SJ, Kang ES. Schwannomas of the upper extremity. J Hand Surg Br. 2000;25:604-607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 78] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 7. | Sharma PV, Jobanputra YB, Perdomo Miquel T, Schroeder JR, Wellikoff A. Primary intratracheal schwannoma resected during bronchoscopy using argon plasma coagulation. BMJ Case Rep. 2018;2018. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 8. | Ge X, Han F, Guan W, Sun J, Guo X. Optimal treatment for primary benign intratracheal schwannoma: A case report and review of the literature. Oncol Lett. 2015;10:2273-2276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 28] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 9. | Han DP, Xiang J, Ye ZQ, He DN, Fei XC, Wang CF, Che JM, Hang JB, Li HC. Primary tracheal schwannoma treated by surgical resection: a case report. J Thorac Dis. 2017;9:E249-E252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 10. | Miyake KK, Nakamoto Y, Kataoka TR, Ueshima C, Higashi T, Terashima T, Nakatani K, Saga T, Minami S, Togashi K. Clinical, Morphologic, and Pathologic Features Associated With Increased FDG Uptake in Schwannoma. AJR Am J Roentgenol. 2016;207:1288-1296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 38] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 11. | Righini CA, Lequeux T, Laverierre MH, Reyt E. Primary tracheal schwannoma: one case report and a literature review. Eur Arch Otorhinolaryngol. 2005;262:157-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 25] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 12. | Hamouri S, Novotny NM. Primary tracheal schwannoma a review of a rare entity: current understanding of management and followup. J Cardiothorac Surg. 2017;12:105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 19] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 13. | Horovitz AG, Khalil KG, Verani RR, Guthrie AM, Cowan DF. Primary intratracheal neurilemoma. J Thorac Cardiovasc Surg. 1983;85:313-317. [PubMed] |
| 14. | Sughrue ME, Yeung AH, Rutkowski MJ, Cheung SW, Parsa AT. Molecular biology of familial and sporadic vestibular schwannomas: implications for novel therapeutics. J Neurosurg. 2011;114:359-366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 34] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 15. | Meyers BF, Mathisen DJ. Management of Tracheal Neoplasms. Oncologist. 1997;2:245-253. [PubMed] |
| 16. | Tang LF, Chen ZM, Zou CC. Primary intratracheal neurilemmoma in children: case report and literature review. Pediatr Pulmonol. 2005;40:550-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 17. | Uhlmann EJ, Plotkin SR. Neurofibromatoses. Adv Exp Med Biol. 2012;724:266-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 18. | Shi XQ, Luo JF, Yang Y, Chen X, Qu WS, Wang YB, Cheng FB. [A case of schwannoma in cervical trachea]. Lin Chung Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2017;31:810-811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 19. | Rusch VW, Schmidt RA. Tracheal schwannoma: management by endoscopic laser resection. Thorax. 1994;49:85-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 20. | Jung YY, Hong ME, Han J, Kim TS, Kim J, Shim YM, Kim H. Bronchial schwannomas: clinicopathologic analysis of 7 cases. Korean J Pathol. 2013;47:326-331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 19] [Article Influence: 1.6] [Reference Citation Analysis (0)] |