Published online Mar 26, 2020. doi: 10.12998/wjcc.v8.i6.1180
Peer-review started: December 17, 2019
First decision: January 13, 2020
Revised: February 10, 2020
Accepted: March 5, 2020
Article in press: March 5, 2020
Published online: March 26, 2020
Processing time: 99 Days and 11.5 Hours
Giant paraesophageal hiatal hernias (HH) are very infrequent, and their spectrum of clinical manifestations is large. Giant HH mainly occurs in elderly patients, and its relationship with anemia has been reported. For the surgical treatment of large HH, Nissen fundoplication is the most common antireflux procedure, and the reinforcement of HH repair with a patch (either synthetic or biologic) is still debatable.
We report on a case of giant paraesophageal HH in a middle-aged male patient with reflux symptoms and severe anemia. After performing a series of tests and diagnostic approaches, results showed a complete intrathoracic stomach associated with severe iron deficiency anemia. The patient underwent successful laparoscopic hernia repair with mesh reinforcement and Nissen fundoplication. Postoperatively, reflux symptoms were markedly relieved, and the imaging study showed complete reduction of the hernia sac. More importantly, anemia was resolved, and hemoglobin, serum iron and ferritin level were returned to the normal range. The patient kept regular follow-up appointments and remained in a satisfactory condition.
This case report highlights the relationship between large HH and iron deficiency anemia. For the surgical treatment of large HH, laparoscopic repair of large HH combined with antireflux procedure and mesh reinforcement is recommended.
Core tip: Giant paraesophageal hiatal hernia (HH) usually occurs in elderly patients and is relatively rare. We present a case of complete intrathoracic stomach in a 46-year-old male with reflux symptoms and severe iron deficiency anemia. The patient underwent successful laparoscopic hernia repair with mesh reinforcement and Nissen fundoplication. Postoperatively, reflux symptoms and iron deficiency anemia were resolved. The patient remained in a satisfactory condition. This case highlights that large HH is a potential cause of iron deficiency anemia. For the surgical treatment of large HH, laparoscopic repair of large HH combined with antireflux procedure and mesh reinforcement is recommended.
- Citation: Yasheng D, Wulamu W, Li YL, Tuhongjiang A, Abudureyimu K. Laparoscopic repair of complete intrathoracic stomach with iron deficiency anemia: A case report. World J Clin Cases 2020; 8(6): 1180-1187
- URL: https://www.wjgnet.com/2307-8960/full/v8/i6/1180.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v8.i6.1180
Classically, hiatal hernias (HH) are divided into four types according to the anatomic position of the gastric cardia. Among all types of HH, type I or sliding HH, is the most common with a prevalence of 95%, while the combination of types II, III and IV, or paraesophageal HH, account for around 5% of all HH[1]. Therefore, paraesophageal HH are relatively rare and usually occur in elderly patients. A giant HH is defined as a hernia that consists of > 30% of the stomach herniating through the diaphragmatic hiatus into the thorax[2], which makes it more uncommon among paraesophageal HH. Clinical manifestations of giant HH are unspecific, making their clinical diagnosis somewhat difficult. However, the relationship between large HH and anemia has previously been reported. Likewise, the association of gastroesophageal reflux disease and HH has long been established. Yet some reported that the patients with HH may have esophagitis or Barrett’s esophagus[3,4]. Hence, we report a case of a middle-aged patient with complete intrathoracic stomach, or a giant paraesophageal HH, who presented with reflux symptoms and anemia. Written consent was obtained from the patient, and the study was approved by the Ethics Committee of People's Hospital of Xinjiang Uygur Autonomous Region (Protocol number: KY2018122001).
A 46-year-old male was admitted to our hospital with chief complaints of heartburn, regurgitation and belching for the last 5 years, and symptoms could be worsened after having a meal. The main symptoms were as follows: Dizziness, hypodynamia and occasionally with nausea and vomiting as well as chest tightness.
Approximately 4 mo earlier, the patient noted that the symptoms worsened even with the medicines and was referred to our hospital.
A diagnosis of HH and iron deficiency anemia (IDA) was made by another hospital, and the patient received omeprazole (40 mg bid) and domperidone (10 mg tid) per day. In addition, the patient received several blood transfusions with the total volume of 1200 mL (the lowest hemoglobin level was 55 g/L). After discharge, the patient took the medicines for a long period of time. His conditions improved only while consistently taking the medicine.
The patient was 170 cm in height and 84 kg in weight with body mass index of 29.07 kg/m2. On examination, after admission to our hospital, the temperature was 36.7 °C, the pulse was 96 beats per minute, the blood pressure was 135/83 mmHg, and the respiratory rate was 18 breaths per minute. Bowel sounds were present. The remainder of the examination was normal. He did not have carotid bruits or jugular venous distention, nor did he have cardiac or pulmonary murmur or rub on auscultation.
Laboratory test results were significant for hemoglobin (105 g/L, normal 130-175 g/L), serum iron (6.06 μmol/L, normal range 11-30 μmol/L), serum ferritin (9.1 μg/L, normal range 15-200 μg/L), oxygen partial pressure (61 mmHg, normal range 80-105 mmHg) and oxygen saturation (91%, normal range 95%-98%).
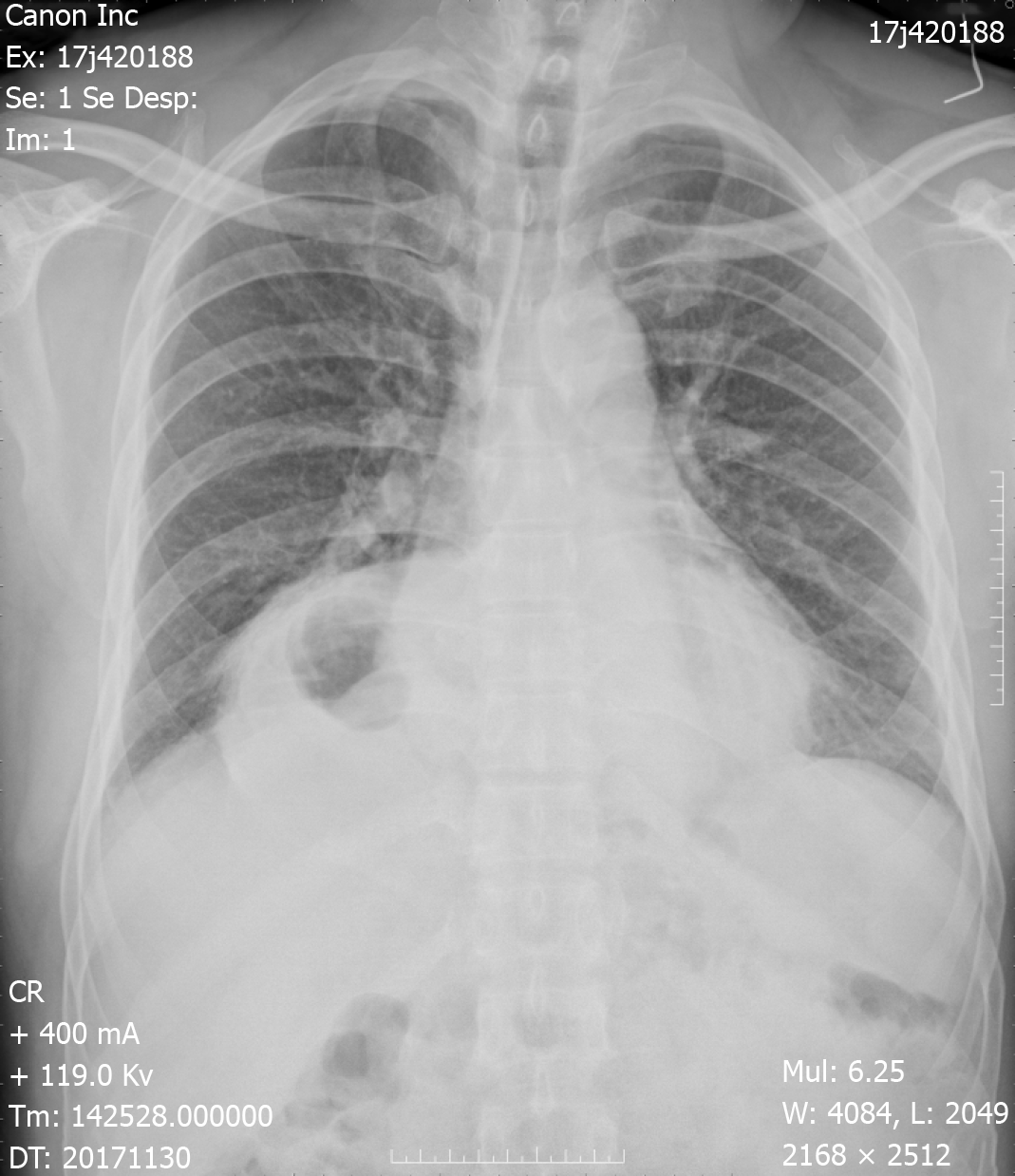
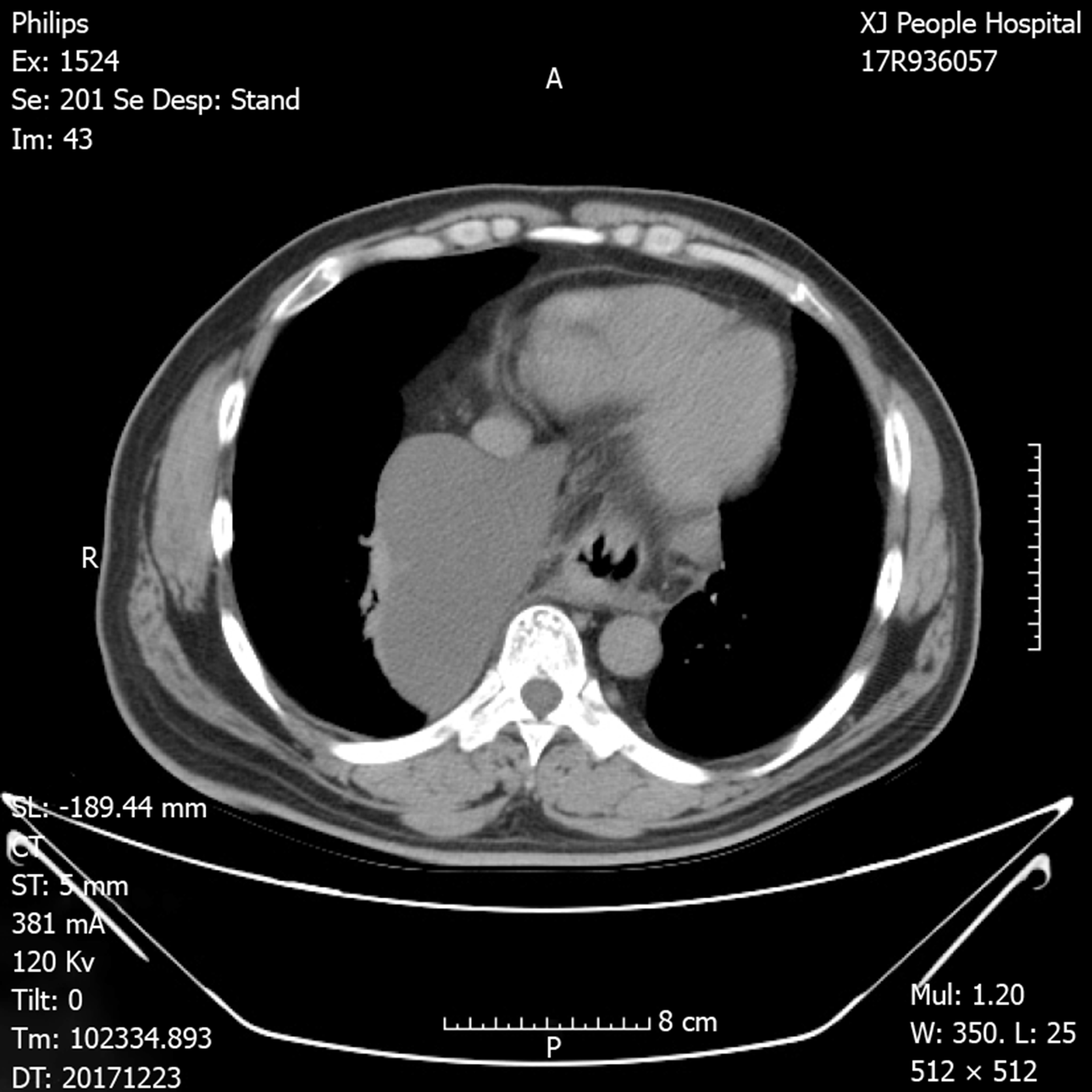
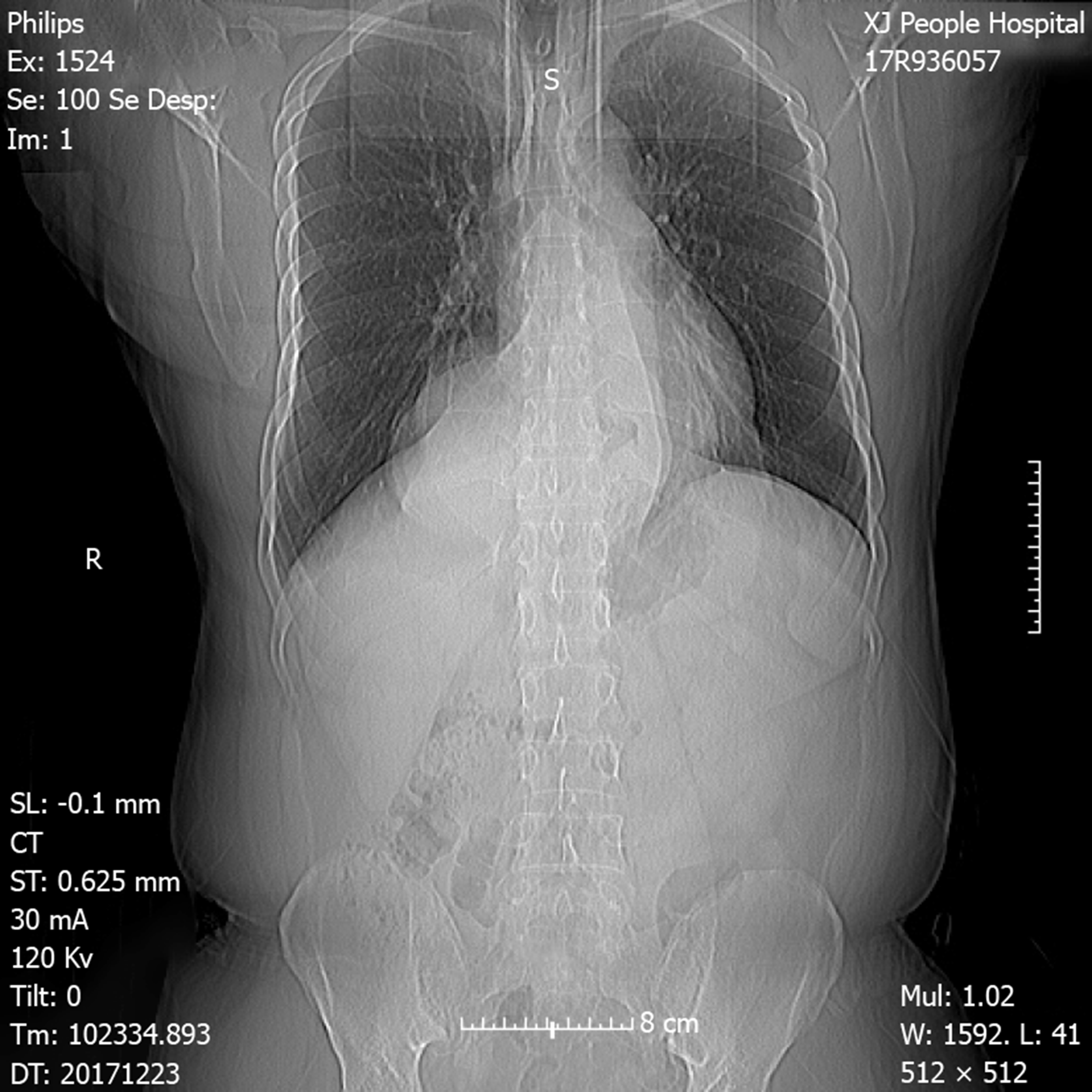
Chest X-ray demonstrated an intrathoracic gastric bubble with increased bilateral lung marking (Figure 1). Chest computed tomography with volumetric analysis demonstrated post-mediastinal location of the whole stomach along with peritoneal fat compressing both bilateral lung and heart (Figure 2). Furthermore, quantitative measurements for the size of the hernia sac, diameter of hernia port, volume of hernia sac and thoracic cavity as well as a ratio of volume of hernia sac to intrathoracic cavity were 13.4 cm × 18.6 cm, 6 cm, 1476.4 cm3, 4025 cm3 and 36.7%, respectively. Barium contrast radiography confirmed large HH and the configuration of the stomach within the hernia suggested an organoaxial volvulus (Figure 3).
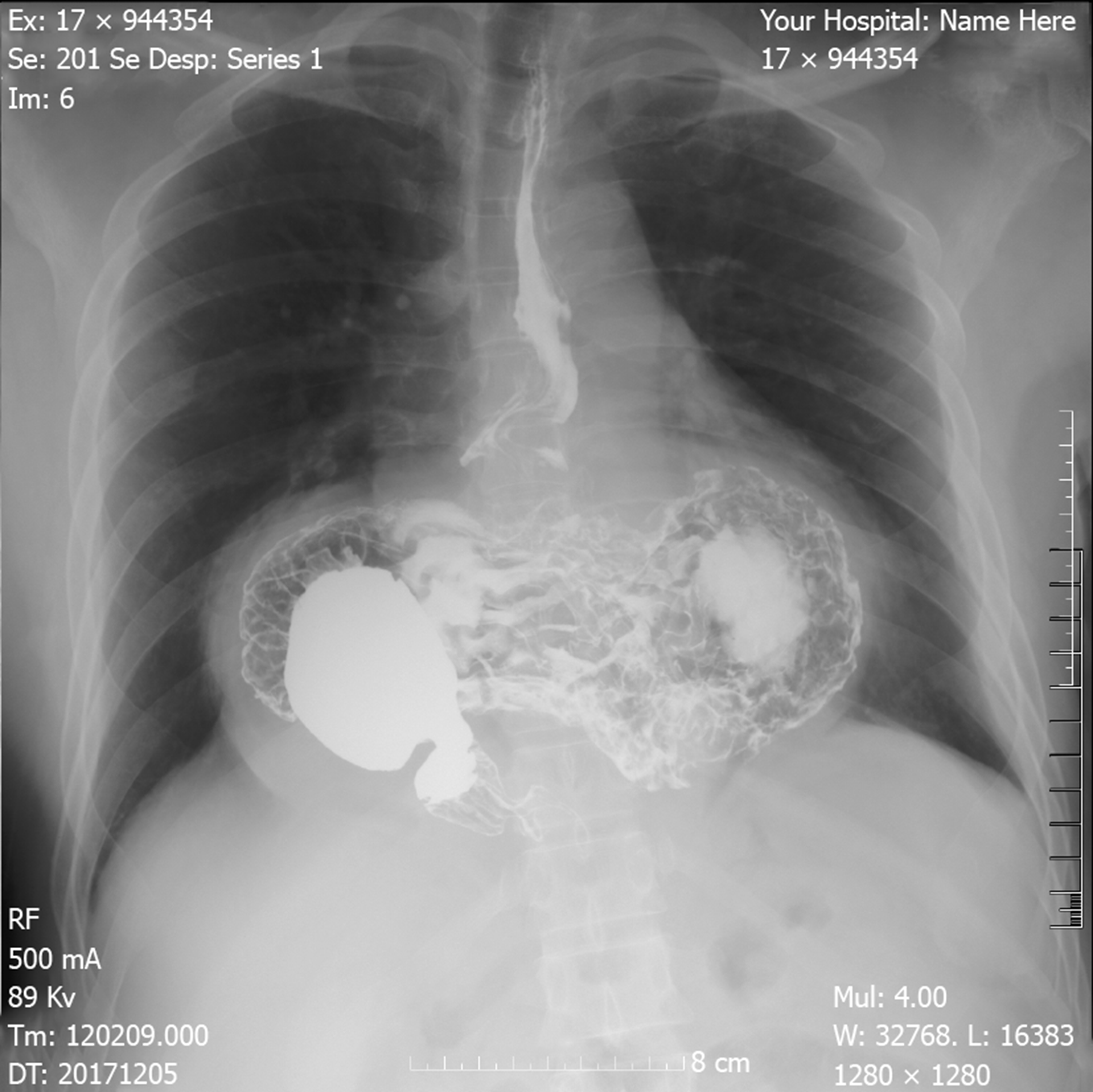
Electrocardiogram and cardiac ultrasonography did not show any abnormalities. To clarify the current and other related possible diagnosis, a series of studies were performed. However, esophageal high-resolution manometry and 24-hr multichannel intraluminal impedance-pH monitoring showed the presence of HH with an elevated level of lower esophageal sphincter of 9.6 cm high, pathological acid reflux and DeMesster score of 64.6 (Figure 4).
The final diagnosis of the presented case is giant paraesophageal HH and IDA.
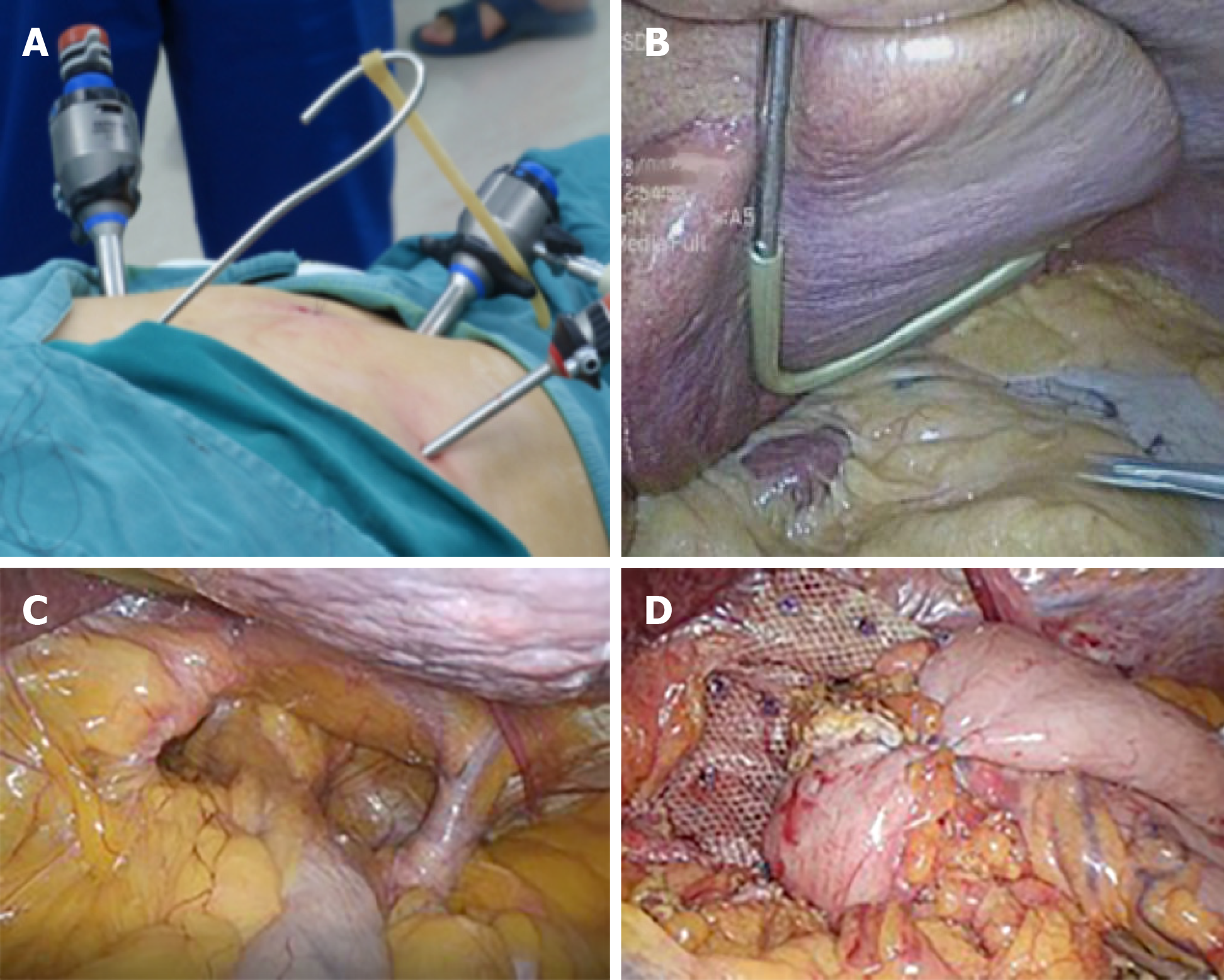
After careful preoperative evaluation for surgical repair, a successful laparoscopic hernia repair with mesh reinforcement and Nissen fundoplication was carried out in accordance with the guidelines recommended by the Society of American Gastrointestinal and Endoscopic Surgeons[5]. The patient was positioned in supine, split-leg position, and the chief surgeon stood between the patient’s legs, while the assistant surgeon stood on the patient’s left. Four ports and a homemade liver retractor were used for surgical access. The initial port of 12 mm was placed supra-umbilically for the laparoscope. After entry, the abdomen was explored looking for iatrogenic injury and the presence of intra-abdominal adhesions that would hinder subsequent port placement. A 12 mm port was then placed just below the left costal margin in the mid-clavicular line as the main working port. The other two 5 mm ports were also placed, one just below the right costal margin in the mid-clavicular line, and the other in the left flank. A separate 3 mm subxiphoid incision was made for the reverse “7” shaped, homemade liver retractor as shown in the pictures (Figure 5A and 5B).
Firstly, an atraumatic grasper was used to grasp the anterior epigastric fat pad. Then the stomach was retracted downward and toward the left lower quadrant to reposition. Subsequently, dissection was preformed until diaphragmatic crura were well displayed, along with the preservation of hepatic branch of the anterior vagus nerve. Then, 3 cm of tension-free esophagus was repositioned intra-abdominally (Figure 5C). The hiatus was then repaired posteriorly with interrupted nonabsorbable sutures. As the patient’s hiatal defect reached 8 cm, we preformed mesh reinforcement using Parietex™ Composite hiatal mesh provided by Medtronic (Minneapolis, MN, United States). Finally, Nissen fundoplication was completed. The 360° wrap was created by grasping the right and left portion of the mobile funds and pulling them behind the esophagus and sutured together in front of the anterior part of the abdominal portion of the esophagus. The length of the wrap was 2 cm with three sutures. At the end, a gastropexy was performed by suturing the posterior fundus to the inferior crus with three interrupted permanent sutures (Figure 5D). Estimated blood loss was 20 mL, and it took 80 min for the surgical procedure.
For the first day after surgery, abdominal examination revealed normal bowel sounds, then the gastric tube was removed. In the following days, the patient started a liquid diet. According to the patient’s statement, almost all of the preoperative discomforts were gradually resolved. After discharge from the hospital, the patient presented to our department at 1-mo post-surgery for follow-up. Neither distinctive abnormalities nor hernia recurrence were observed from the chest X-ray (Figure 6). The patient had an increase in postoperative hemoglobin, serum iron and serum ferritin level (135 g/L, 18.3 μmol/L and 92.4 μg/L, respectively).
Paraesophageal HH are defined as the condition in which gastroesophageal junction and components of the abdominal cavity, most commonly the stomach, herniated via esophageal hiatus into the mediastinum. However, most large paraoesophageal HH occur in elderly patients with the incidence rate of > 60% above the age of 70 years and is a relatively rare condition[6]. Interestingly, our case was a middle-aged male patient, and it indicates that the physicians should be aware of the presence of HH when making clinical diagnosis for the patient with atypical characteristics. Some studies have reported that the formation of HH is related to obesity[7]. Therefore, high body mass index can be one of the risk factors contributing to large HH in this patient, whose body mass index (29.07 kg/m2) is close to the category of obesity.
It is crucial to distinguish between symptomatic paraesophageal HH and asymptomatic or minimally symptomatic HH. Generally, symptomatic patients are recommended for surgical repair to prevent subsequent acute complications, such as strangulation, perforation and bleeding. In addition to HH repair, laparoscopic antireflux procedure is a well-established treatment for patients suffering from reflux disease associated with HH, especially for large paraoesophageal HH. Nissen fundoplication is the most commonly used procedure for the treatment of gastroesophageal reflux disease due to its good postoperative long-term reflux control results in approximately 90% of patients[8].
In view of these facts, the patient underwent successful HH repair reinforced by a “U” shaped mesh with Nissen fundoplication. Even though the reinforcement of the HH repair with a patch (either synthetic or biologic) is still debatable, a meta-analysis reported by Targarona et al[9] concluded that prosthetic reinforcement was beneficial with an acceptable rate of secondary complications. Another meta-analysis of three randomized studies also reported that prosthetic reinforcement had a four-fold decrease in 1-year risk of recurrence[10]. Therefore, according to related studies and our experience, HH repair with mesh reinforcement is recommended for symptomatic HH patients, especially for large paraoesophageal HH patients.
The relationship of IDA with HH has been studied, and it is reported that HH is a cause of gastrointestinal bleeding and increases the risk of subsequent IDA[11]. Gray et al[12] suggested that the prevalence of Cameron lesion, which is considered to be a source of gastrointestinal bleeding, is known to vary with HH size, with the highest prevalence occurring in large HH patients. They identified large HH as a major risk factor for IDA. In our case, the patient’s reflux-related symptoms completely resolved, and his hemoglobin level was returning to a normal range after the operation. Consequently, this case report complements the other studies and strengthens the evidence that large HH may be a cause of anemia.
Complete intrathoracic stomach, or giant HH, is very infrequent, and its spectrum of clinical manifestations is large. This report presented a case of giant HH in a middle-aged male patient with reflux symptoms and severe anemia. Although studies have reported that large HH mainly occurs in elderly patients, one possible factor contributing to the situation in this relatively young patient might be his high body mass index.
According to the literature mentioned above and our case report, there appears to be a relationship between large HH and IDA and indicates that large HH may be a potential cause of IDA. Surgical repair of large HH relieves IDA symptoms as reported by others[13-15] and as seen in our patient. More importantly, for the patients suffering from reflux-related symptoms with large HH, antireflux procedure and mesh reinforcement are recommended.
Manuscript source: Unsolicited manuscript
Corresponding Author's Membership in Professional Societies: Chinese College of Surgeons of GERD Committee (Chairman); Chinese Hernia College of Surgeons (Vice Chairman).
Specialty type: Medicine, research and experimental
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Bandyopadhyay SK, Sugimoto H S-Editor: Dou Y L-Editor: Filipodia E-Editor: Qi LL
| 1. | Abbara S, Kalan MM, Lewicki AM. Intrathoracic stomach revisited. AJR Am J Roentgenol. 2003;181:403-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 30] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 2. | Mitiek MO, Andrade RS. Giant hiatal hernia. Ann Thorac Surg. 2010;89:S2168-S2173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 96] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 3. | Jobe BA, Horvath KD, Swanstrom LL. Postoperative function following laparoscopic collis gastroplasty for shortened esophagus. Arch Surg. 1998;133:867-874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 91] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 4. | Lin E, Swafford V, Chadalavada R, Ramshaw BJ, Smith CD. Disparity between symptomatic and physiologic outcomes following esophageal lengthening procedures for antireflux surgery. J Gastrointest Surg. 2004;8:31-9; discussion 38-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 35] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 5. | Stefanidis D, Hope WW, Kohn GP, Reardon PR, Richardson WS, Fanelli RD; SAGES Guidelines Committee. Guidelines for surgical treatment of gastroesophageal reflux disease. Surg Endosc. 2010;24:2647-2669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 348] [Cited by in RCA: 252] [Article Influence: 16.8] [Reference Citation Analysis (1)] |
| 6. | Winans CS. Hiatus hernia. Its significance in the elderly patient. Geriatrics. 1972;27:69-78. [PubMed] |
| 7. | Valezi AC, Herbella FAM, Schlottmann F, Patti MG. Gastroesophageal Reflux Disease in Obese Patients. J Laparoendosc Adv Surg Tech A. 2018;28:949-952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 8. | Obeidat FW, Lang RA, Knauf A, Thomas MN, Hüttl TK, Zügel NP, Jauch KW, Hüttl TP. Laparoscopic anterior hemifundoplication and hiatoplasty for the treatment of upside-down stomach: mid- and long-term results after 40 patients. Surg Endosc. 2011;25:2230-2235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 9. | Targarona EM, Bendahan G, Balague C, Garriga J, Trias M. Mesh in the hiatus: a controversial issue. Arch Surg. 2004;139:1286-1296; discussion 1296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 80] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 10. | Antoniou SA, Antoniou GA, Koch OO, Pointner R, Granderath FA. Lower recurrence rates after mesh-reinforced versus simple hiatal hernia repair: a meta-analysis of randomized trials. Surg Laparosc Endosc Percutan Tech. 2012;22:498-502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 59] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 11. | Ruhl CE, Everhart JE. Relationship of iron-deficiency anemia with esophagitis and hiatal hernia: hospital findings from a prospective, population-based study. Am J Gastroenterol. 2001;96:322-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 12. | Gray DM, Kushnir V, Kalra G, Rosenstock A, Alsakka MA, Patel A, Sayuk G, Gyawali CP. Cameron lesions in patients with hiatal hernias: prevalence, presentation, and treatment outcome. Dis Esophagus. 2015;28:448-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 13. | Asti E, Bonavina L, Lombardi M, Bandera F, Secchi F, Guazzi M. Reversibility of cardiopulmonary impairment after laparoscopic repair of large hiatal hernia. Int J Surg Case Rep. 2015;14:33-35. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Naoum C, Puranik R, Falk GL, Yiannikas J, Kritharides L. Postprandial left atrial filling is impaired in patients with large hiatal hernia and improves following surgical repair. Int J Cardiol. 2015;182:291-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 15. | Bjelović M, Babic T, Gunjić D, Veselinović M, Spica B. Laparoscopic repair of hiatal hernias: experience after 200 consecutive cases. Srp Arh Celok Lek. 2014;142:424-430. [PubMed] |