Published online Aug 26, 2020. doi: 10.12998/wjcc.v8.i16.3411
Peer-review started: May 1, 2020
First decision: June 18, 2020
Revised: July 3, 2020
Accepted: July 30, 2020
Article in press: July 30, 2020
Published online: August 26, 2020
Processing time: 115 Days and 13.4 Hours
Autoimmune pancreatitis has received considerable attention, especially due to the marked effect of corticosteroid therapy on its clinical course. Knowledge, especially regarding type 1 autoimmune pancreatitis, has significantly increased over the last decades, and despite significant differences in pathophysiology and outcomes, both type 1 and 2 autoimmune pancreatitis are still considered different types of the same disease. Some have proposed a different nomenclature reflecting these differences. Although the term steroid-responsive pancreatitides may be interpreted as synonymous to type 1 and 2 autoimmune pancreatitis, these are not the only pancreatic conditions that show a response to steroid therapy. Acute pancreatitis caused by vasculitis and connective tissue diseases and acute pancreatitis secondary to checkpoint inhibitors or programmed cell death receptor antibody-mediated blockage cancer therapy may also benefit from steroid treatment. This review presents current concepts on these disorders, aiming to increase awareness, analyze similarities and differences, and propose a new nomenclature that reflects their specific particularities, clustering them under the term “steroid-responsive pancreatitides”.
Core tip: This review presents the rationale on the current evidence on pancreatic disorders that respond to steroid therapy. It focuses not only on autoimmune pancreatitis 1 and 2 but also on less recognized but relevant conditions, such as acute pancreatitis related to immune checkpoint inhibitors and vasculitis and connective tissue diseases that often need and respond to corticosteroid therapy. The aims are to increase awareness, analyze clinical and pathophysiological similarities and differences, recognize research opportunities, and propose a new nomenclature that reflects their specific particularities, clustering them under the term steroid-responsive pancreatitides.
- Citation: Pelaez-Luna M, Soriano-Rios A, Lira-Treviño AC, Uscanga-Domínguez L. Steroid-responsive pancreatitides. World J Clin Cases 2020; 8(16): 3411-3430
- URL: https://www.wjgnet.com/2307-8960/full/v8/i16/3411.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v8.i16.3411
Thus far, no single medication has proven effective in treating, preventing or modifying the natural history and outcomes of acute and chronic pancreatitis. Current treatment considers a broad arsenal of therapeutic approaches, including supportive measurements, treating and avoiding exposure to predisposing factors and solving any presenting complications. Over the last decades, autoimmune pancreatitis (types 1 and 2) has received considerable attention, especially due to the dramatic effect of corticosteroid therapy on its clinical course.
Autoimmune pancreatitis (AIP) is not the only pancreatic condition with an indication and response to steroid therapy. Other less frequent and less noted pancreatopathies that benefit from steroid therapy are: (1) Acute pancreatitis caused by vasculitis and connective tissue diseases (VasC-AP), often associated with rheumatic diseases such as systemic lupus erythematous (SLE) or Wegener disease; and (2) Acute pancreatitis secondary to checkpoint inhibitors or programmed cell death receptor (PD-1) antibody-mediated blockage cancer therapy.
This review presents the rationale of current evidence on these pancreatic disorders in order to increase awareness, analyze clinical and pathophysiological similarities and differences, recognize research opportunities, and propose a new nomenclature that reflects their specific particularities, clustering them under the term “steroid-responsive pancreatitides” (Table 1).
| Acute pancreatitis | Chronic pancreatitis |
| Acute or subacute forms of IgG4 RD-AP | IgG4 related disease pancreatitis |
| Idiopathic duct centric pancreatitis | Idiopathic duct centric chronic pancreatitis |
| ICI-related pancreatitis | |
| VasC-related pancreatitis |
Autoimmune pancreatitis is considered a subtype of chronic pancreatitis (CP) and accounts for nearly 5% of all CP cases[1]. It usually presents as a focal mass resembling pancreatic cancer. The first description of AIP dates back to 1961 in France when Henri Sarles et al[2] described a nonalcoholic calcific CP case associated with hypergammaglobulinemia, but it was not until 1995, after Yoshida et al[3] described a case of CP presenting with hypergammaglobulinemia and serum autoantibodies that responded to steroid therapy, that the term AIP was coined. This was followed by several case series reports from Asia, Europe and North America[4-7].
Soon afterwards, differences in demographics, geography, pathology, and clinical presentation showed the existence of two distinct subtypes of AIP: Type 1 AIP (AIP-1) or lymphoplasmacytic sclerosing pancreatitis (LPSP) and type 2 AIP (AIP-2) or idiopathic duct centric chronic pancreatitis (IDCP)[8]. Since both disorders showed a dramatic response to steroids, AIP 1 and 2 were considered two different poles or presentations of AIP.
Later, marked differences in histology, demographics, clinical presentation and outcomes became evident, suggesting that each AIP subtype might be its own completely different and independent disease, sharing only the response to a specific treatment (e.g., steroids), a phenomenon that can be seen in other diseases (e.g., rheumatic disorders, oncology). This has led some authors to consider them as separate, even using a distinct nomenclature that takes into account their specific pathophysiology, histology and clinical picture: Autoimmune pancreatitis referring to AIP-1 and IDCP in reference to AIP-2[8].
This disorder has characteristic clinical, serum, and histological features and probably represents those early reports by Sarles et al[2] and Yoshida et al[3]. The presence of a dense lymphoplasmacytic infiltrate is responsible for its alternative name “LPSP”.
Currently, it is considered the most frequent manifestation of immunoglobulin G4-related disease (IgG4-RD), a systemic fibrotic condition that presents with fibrotic and tumor-like lesions in different organs and is associated with increased levels of IgG4 subclasses[9,10]. Approximately 41% of IgG4-RD cases present with pancreas involvement. Remarkably, in almost 50% of cases, the pancreas is the sole location for the manifestation of the disease; nearly 51% of IgG4-RD pancreatitis cases develop other organ involvement (OOI) during their course[8,11].
AIP or IgG4-RD pancreatitis is more common in Asia; it affects primarily men in the 7th decade with a male:female ratio of 3:1[7]. A set of Japanese surveys performed in different periods estimated an overall prevalence of 4.6 to 10.1 cases per 100000 inhabitants and an incidence of 1.4 to 3.1 new cases per 100000 inhabitants, suggesting that physician awareness and diagnostic tools have improved over time[12-14].
According to international surveys[10,15-17], it seems that IgG4-RD pancreatitis is more frequent in Asia than in Europe and North America, although no precise information about its global incidence and prevalence is provided.
Although several mechanisms and immunologic pathways have been explored, IgG4-RD pancreatitis pathogenesis remains unclear. The role of IgG4 subclasses has been under scrutiny ever since it was recognized that subjects with AIP (then known as sclerosing pancreatitis) present with increased serum levels[18].
The IgG family consists of four subclasses categorized according to the sequence of the heavy chain constant domains. IgG4 is present in lower amounts and has the weak capacity to bind to FcY receptors and C1q of complement, but through a Fab-arm exchange (the swap of a heavy chain and attached light chain), it has the ability to form “half antibodies”. These half antibodies are asymmetric and allow IgG4 to recognize two different antigens. This feature comes with the inability to crosslink antigens and form immune complexes, resulting in an overall anti-inflammatory function[19,20]. This anti-inflammatory nature is intriguing, considering the significant inflammatory infiltrate and fibrotic process observed in pancreas specimens from individuals affected by IgG4-RD pancreatitis.
The anti-inflammatory nature of IgG4, along with studies on T- and B cell-mediated acquired and innate immunity, raises the question of whether its increased serum levels and the presence of IgG4-positive plasma cells in involved organs are either a triggering factor or may represent an overexpressed anti-inflammatory phenotype.
T cells and B cells play a central role in IgG4-RD pancreatitis (AIP-1/LPSP) pathogenesis, where T helper 1 (Th1)/Th2 cells balance has an important role.
The observed inflammatory infiltrate in IgG4-RD-involved organs includes a significant number of regulatory T (Treg) cells characterized by an increase in Th2 cells and regulatory cytokines (interleukin 4 [IL-4], IL-5, IL-13, IL-10, and transforming growth factor beta [TGF-β). This particular cytokine profile combined with distinct Treg cell subpopulation counts (increased Treg CD4+CD25 and memory Treg cells) is responsible for a higher production of IgG4, suggesting that increased IgG4 levels are a consequence and not a disease driver[21-23].
It seems that IgG4 production is induced by increased peripheral Treg cells and elevated quantities of inducible costimulator (ICOS)-positive Treg cells via IL-10, while ICOS-negative Treg cells induce fibrosis via TGF-β. This suggests that IgG4 does not act as a triggering factor or as an anti-inflammatory factor[24].
Similar to T cells, an imbalance in different Breg cell subpopulations and plasmablasts seems to have a key role in IgG4-RD. This means that both, acquired and innate immunity play significant roles in the pathogenesis of the disease.
Pancreases from patients with AIP have positive Toll-like receptor (TLR) 2 and TLR4 basophil infiltrate[25-27]. Activation of these basophils may induce the presence of M2 macrophages that in turn could exert some influence on the previously described Th2 environment, affecting the production of IgG4 via TLR4. These observations support that the increase in IgG4 is a consequence and not a triggering factor.
Some reports indicate that neutrophil extracellular traps and peripheral eosinophils may also be involved in the development of AIP. However, it seems that the shift towards a Th2 response is responsible for such an increment in eosinophils and IgE levels. Interestingly, peripheral eosinophilia has been reported to occur in 12%-29% of IgG4-RD pancreatitis cases. Moderate to severe eosinophil infiltration has been observed in 67% of resected pancreas specimens with IgG4-RD pancreatitis. Although no correlation between peripheral and tissue eosinophils has been noted, some authors have proposed that elevated numbers of peripheral eosinophils or tissue infiltration can add diagnostic value to serum IgG4 levels[28-32].
Resembling other immune mediated disorders, the presence of different autoantibodies (e.g., lactoferrin and carbonic anhydrase II) and the expansion of oligoclonal plasmablasts of the IgG4 subtype advocate for an autoimmune mechanism.
However, the fact that IgG4-RD pancreatitis (AIP) predominantly affects elderly men, that the detected autoantibodies do not play a key role in the pathogenesis, that so far no IgG4 autoantibodies have been identified and that increased numbers of specific Treg populations have been observed (something not expected in autoimmune disorders where Treg numbers are expected to be low) are arguments against autoimmunity[26-28,33-35].
All the aforementioned immune profiles seem to occur in genetically susceptible individuals. Specific haplotypes or polymorphisms in the HLA (DRB1*0405-DQB1*0401) CTLA2 and FCRL3 genes have been described, but the triggering factor or factors remain elusive[36,37].
It has been suggested that Helicobacter pylori infection might be a risk or triggering factor. Some patients have antibodies against the plasminogen binding protein of the bacteria that could cross react with a highly expressed protein in acinar pancreatic cells. Other possible triggers are persistent exposure to external antigens, paraneoplastic reactions and allergies[38].
Although the majority of affected cases are nonatopic or the prevalence of atopy is no higher than that in the general population, some authors have suggested that IgG4-RD presents similar features seen in allergic reactions[39]. Even due to the presence of vasculitis-like lesions, IgG4-RD has been incorporated into the 2012 Revised Chapel Hill Consensus Conference as a cause of large vessel vasculitis[40].
These complex inflammatory, anti-inflammatory and immune response interactions suggest that IgG4-RD may be caused either by a B cell-mediated disease or a Th2/Treg-driven disorder or from a pro-or autoinflammatory and pro-fibrotic phenotype.
Uchida et al[41] and Kamisawa et al[42] have published a proposal of the pathophysiology of IgG4-RD pancreatitis (AIP). IgG4-RD pancreatitis can be considered a complex disease that most likely results from intricate immune and inflammatory interactions taking place simultaneously. What exactly triggers these interactions and whether they, along with their consequences are restricted to certain genetically predisposed individuals exposed to specific environmental factors has yet to be clarified.
IgG4 RD pancreatitis (AIP/LPSP) presents with characteristic histologic, imaging, clinical and serum features. Typically, patients seek medical attention due to the presence of a diffusely or focally enlarged pancreas resembling pancreatic cancer. Most of the time, this is an incidental finding or the result of the diagnostic work-up of jaundice, a sign present in up to 60% of cases. Other symptoms include abdominal (30%) or back pain (15%), acute pancreatitis (5%-10%), and weight loss (15%); some series have described peripancreatic vascular complications[10,13-15,43,44].
AIP (IgG4-RD pancreatitis) may have an acute presentation (e.g., obstructive jaundice and/or diffuse or focal pancreatic swelling) or late presentation (e.g., features of painless chronic pancreatitis). Those cases presenting with acute pancreatitis tend to be mild, with a low frequency of local or systemic complications[45].
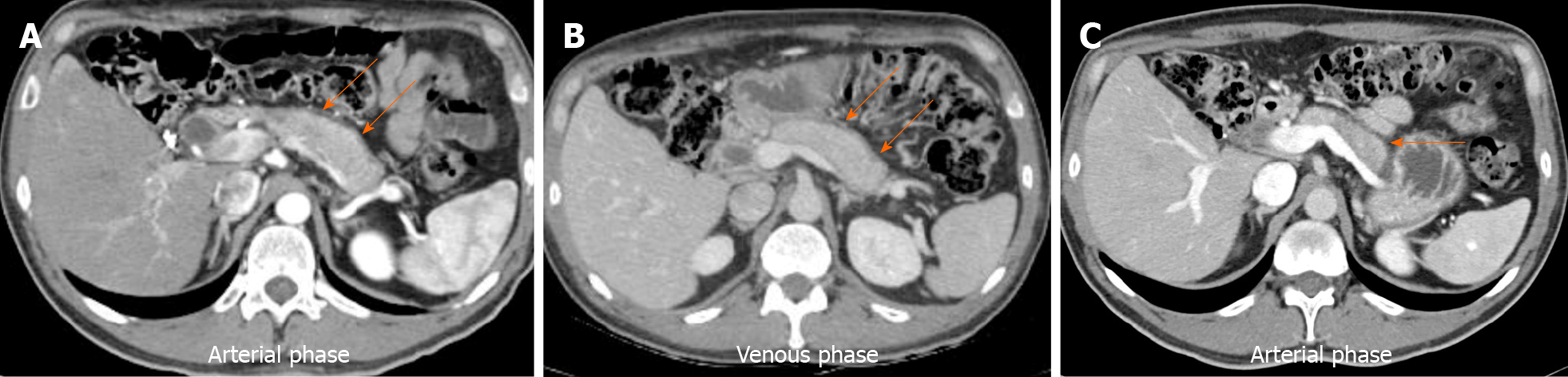
Since imaging abnormalities tend to be the first clinical manifestation, diagnosis can initially be suspected when certain specific or “typical” image characteristics such as the following are noted: Loss of lobularity and diffuse pancreatic enlargement defined as an increase in the size of the gland without density difference or a discrete mass greater than 2/3 of the entire pancreas (65%-70%), long main pancreatic duct (MPD) stenosis in the corresponding area without marked dilation of the upstream duct, poor contrast enhancement in the enlarged pancreas at the arterial phase that is usually recovered at the delayed phase on the dynamic CT (defined as delayed enhancement; different from pancreatic cancer in which the poor enhancement persists in the delayed phase), peripheral capsule-like rim (49%), and hyperintense bile duct wall (Figure 1).
If these “classic” characteristics are absent or are partially present (atypical presentation), then additional diagnostic clues must be found. Collateral evidence considers increased IgG4 serum levels (a diagnostic accuracy up to 95% is obtained with levels 2x above the upper limit of normal) as well as other organ involvement (OOI) (e.g., biliary strictures that are seen in up to 50% of cases, enlarged salivary glands, pulmonary nodules, retroperitoneal fibrosis, interstitial nephritis generally associated with a dense plasma cell IgG4+ infiltrate) that can appear before, synchronously or after the pancreas involvement[46-52].
We have reported that in 15 cases of histology-proven AIP (LPSP), the most common manifestation was weight loss (87%), followed by abdominal pain (60%) and bile duct stenosis (53%); contrary to the “typical” presentation, only 2 cases presented a 2 fold increase in serum IgG4. Overall, patients’ mean age of presentation was 47.5 years, with slight predominance (53%) of male gender[53].
Elevated serum IgG4 (> 135 mg/dL) have been reported in up to 70%-85% of cases. A cut-off point of > 2-fold elevation above the upper limit of normal in serum IgG4 has been recommended not only due to its increased diagnostic accuracy but also because less intense elevations of IgG4 may be seen in up to 10% of pancreatic cancer and other nonspecific inflammatory pancreato-biliary disorders[54-56].
Frequently, this additional information provides enough evidence to support the diagnosis of IgG4-RD pancreatitis, making histological examination (pancreas biopsy) seldom needed.
The recent IgG4-RD diagnostic consensus has suggested that 2 histopathological items that occur in up to 90% of AIP-1 and in only 5% of pancreatic cancer cases need to be present in tissue specimens to consider the diagnosis: > 40% of IgG positive plasma cells and > 10 IgG4 positive plasma cells per high powered field[57].
The ultimate diagnostic test once malignancy has been properly ruled out is assessment of the response to steroids[58,59].
Several diagnostic criteria have been published by different societies from Asia, North America and Europe. These criteria have been collected and graded in the International Consensus Diagnostic Criteria (ICDC)[60]. The ICDC considers typical and atypical presentations of each item (imaging, serum, clinical, histology, OOI). The probability of IgG4-RD pancreatitis (AIP) increases as the number of characteristics increases. The overall sensitivity, specificity and accuracy for all AIP cases are 89%-95%, 100% and 94%, respectively[61-63].
Thus far, endoscopic ultrasound-guided biopsy seems to be the method of choice for pancreatic tissue acquisition. Several reports on the advantages, limitations and utility of different techniques (e.g., fine-needle aspiration or fine-needle biopsy) have been published, concluding that the method and technique should be individualized and based on each endosonographer and center expertise[64-68].
In some cases, the IgG4+ to IgG+ plasma cell ratio in regular endoscopic biopsies obtained from the ampulla of Vater have helped to confirm the diagnosis of IgG4-RD pancreatitis and could be considered as a surrogate when endoscopic ultrasound guided biopsies or serum IgG4 are inconclusive or not available[69].
Frequently there is plenty of collateral evidence (serum, imaging, OOI) to reach a diagnosis, thus histology is usually not mandatory. It might be needed in the subset of cases deemed atypical/indeterminate AIP or not otherwise specified (AIP-NOS) in which clinical, serum, and imaging manifestations are inconclusive. Some of these cases may represent IDCP, but others may indeed be atypical cases of IgG4-RD pancreatitis (AIP)[70].
Steroids remain the cornerstone of AIP (IgG4-RD pancreatitis) treatment. Compared to patients who did not receive any therapy, corticosteroids achieve remission in up to 90%-99% of cases[71,72]. Unless contraindicated, steroids should be administered to all näive patients and to any patient attempting to induce remission. Other immunosuppressants, such as azathioprine and methotrexate, are ineffective in inducing remission and their use has been limited to maintenance therapy as steroid-sparing agents. Rituximab presents as an adequate option for inducing remission when steroids are contraindicated.
According to an international consensus[73], indications for treatment in symptomatic patients include pancreatic involvement, abdominal and back pain, and OOI such as obstructive jaundice due to bile duct stricture. Additionally, asymptomatic cases with persistent pancreatic masses on imaging and persistent liver function test abnormalities need to be treated.
In patients presenting with biliary stricture, if jaundice is mild and no signs of cholangitis are present, drainage might not be needed. If indicated, it presents an opportunity to acquire tissue to rule out malignancy in atypical cases.
Although 10%-20% of cases may improve spontaneously without any treatment, significant and irreversible complications such as pancreatic insufficiency and refractory biliary strictures, among others, have been documented in patients left untreated or when treatment was delayed.
Several steroid regimens have been used. The international consensus recommends an initial dose of prednisone of either 0.6-1.0 mg/kg/for a minimum of 20 mg/d. This should be administered for 2 to 4 wk and then tapered down at a rate of 5 mg per wk until stopped. The use of higher doses (30-40 mg/d) for 4 wk, which is the preferred approach of the authors and others, is associated with faster results. Regimens shorter than 4 wk and doses less than 20 mg/d or equivalents are not recommended.
Steroid response should be evaluated within 2 to 4 wk after its initiation, preferably with the same imaging technique used at diagnosis. Approximately 86%-100% of cases will show a decrease or disappearance of imaging findings (e.g., shrinkage of the enlarged pancreas, disappearance of hepatic or lung nodules, receding retroperitoneal fibrosis, etc). Such a response reconfirms the diagnosis and is an indication to complete treatment. Usually, 3 to 6 mo after steroid treatment, the thickness of the enlarged pancreas decreases by 60%-70%[74] (Figure 1).
Although the response to steroids is part of the diagnostic criteria, especially in atypical or IgG4-negative cases, it should never be considered before extensive work-up to rule out malignancy has been carried out (including endoscopic ultrasound guided fine needle aspiration biopsy). Additionally, when steroid response is not present, alternative diagnoses must be considered, and the patient will need to undergo diagnostic reassessment[74].
Relapse occurs in 24%-65% of AIP (IgG4-RD pancreatitis) cases[75]. This rate increases after discontinuation of steroids compared with that observed during tapering or maintenance therapy (67% vs 15% vs 18%, respectively).
Nationwide studies from Japan[72,76,77] favor maintenance therapy over the 12 wk regimen. These studies have shown a significantly lower relapse rate (26%) in patients on 5 mg/d of prednisone as maintenance therapy compared with the no maintenance therapy group (45% relapse). These numbers reached a plateau at 7 years and remained unchanged at the 10 years follow-up.
Despite the reported benefits, long-term steroid therapy-associated complications must be taken into account. Arguments against maintenance therapy include the lack of gain observed after 7 years of treatment and the onset of dose-and time-dependent steroid-related complications such as osteoporosis, myopathy and infections. Nearly 50% of patients receiving steroid maintenance therapy presented at least one steroid-related complication once a cumulative dose of 10000 mg was reached or exceeded or when therapy continued for > 5 year.
AIP (IgG4-RD pancreatitis) tends to affect elderly people; this group is more likely to present steroid-related complications than their younger counterparts. The international consensus and the authors recommendation is to individualize maintenance therapy, based on disease activity and the presence of high risk factors for relapse.
Serum IgG4, IgE, eosinophils and IL-2 soluble receptor have been reported as biomarkers of disease activity and, consequently, potential predictors of therapy response and relapse. A high risk of relapse has been associated with high pretreatment serum IgG4 (> × 4 upper limit of normal) persistent high serum IgG4 or minor decrease after steroid initiation, proximal biliary involvement and multiorgan involvement (> 2 organs)[78,79].
Considering the relevant side effects related to long-term steroid treatment, once remission has been achieved, individuals with a high risk of relapse or who have their 1st relapse episode should be placed on maintenance and relapse prevention therapy. The recommendation is to introduce steroid-sparing agents (e.g., azathioprine or 6 mercaptopurine). Doses and care are similar to those observed in other conditions in which they are prescribed (e.g., autoimmune hepatitis). Long-term use of thiopurines has been associated with a 5.3-fold risk of developing lymphoproliferative disorders (particularly in males over 65 years) among other neoplastic and hematologic complications.
In resistant cases of AIP (IgG4-RD pancreatitis) or in patients with contraindications or side effects related to the use of steroids and immune modulators, rituximab presents itself as an excellent alternative.
Rituximab is a chimeric monoclonal antibody that targets CD20 B lymphocytes. Different protocols of administration have been used with slight differences in primary outcomes. The rheumatoid arthritis protocol[80] administers two perfusions of 1000 mg of rituximab 15 d apart, and the Mayo Clinic protocol[81] uses 375 mg/m2 intravenous rituximab infusions weekly for 4 wk and then every 2 to 3 mo for 24 mo for an average of 10 perfusions. The reported efficacy rate favors the Mayo Clinic protocol (67% vs 83%, respectively), but the recurrence rate within 3 years after stopping rituximab is similar (40% vs 45%); although maintenance therapy with rituximab decreases recurrence, it is associated with an increased risk of infection. Follow up information beyond 3 year is not available.
Following the reports on LPSP (IgG4-RD pancreatitis) from Asia, reports from Europe and North America differed in their frequency of LPSP cases and described a particular non-IgG4-related pancreatitis variant. Due to similarities with AIP (IgG4-RD pancreatitis), especially its response to steroids, it was named type 2 autoimmune pancreatitis (AIP-2).
Earlier reports considered or found AIP-2 to be part of a group of masses forming chronic pancreatitides that resemble pancreatic cancer known as tumefactive chronic pancreatitis. A detailed description of these cases showed that AIP-2 has a unique clinical phenotype and histologic injury pattern that sets it apart from LPSP (IgG4-RD pancreatitis) and other forms of CP and from which it takes its current name “IDCP”[82].
Information is scarce, and the real incidence of IDCP might have been underestimated. According to a Japanese series[83], IDCP represents less than 5% of all cases of AIP of any subtype.
Data from Europe and North America[7,84-86] reported that IDCP accounts for 45% and 37% of all cases of AIP, respectively. At our institution, a tertiary referral center part of the National Mexican Institutes of Health System (unpublished data), the frequencies of LPSP and IDCP are almost equally distributed, with a slight difference favoring IgG4-related pancreatitis.
In a comparative study of resected pancreatic masses, those cases with IDCP presented similar demographics and clinical characteristics reported elsewhere. Compared to AIP (IgG4-RD pancreatitis) and pancreatic cancer, IDCP affects individuals a decade or more earlier (mean age of presentation ranges between the 4th and 5th decade), with no significant gender preference[87].
Although both disorders are responsive to steroids, the pathologic mechanisms responsible for IDCP are entirely different from those seen in IgG4-RD pancreatitis (AIP). Although little is known about the processes leading to IDCP, most of the pathogenic mechanisms can be inferred and understood from its histological characteristics. A IDCP pathognomonic histologic finding is a granulocytic epithelial lesion (GEL).
Microscopic examination of pancreas specimens shows that unlike AIP (IgG4-RD pancreatitis), IDCP presents deposition of C3c and IgG in the basement membrane of pancreatic ducts and acini, a patchy dense inflammatory infiltrate involving the lobules, a prominent neutrophil infiltrate involving the entire wall of the pancreatic ducts, all of which lead to ductal epithelial destruction and obstruction[29].
These findings suggest an immune complex-mediated response. The prominent neutrophil infiltrate appears to result from the chemotactic action of high concentrations of upregulated IL-8[88]. Interestingly, these changes in IL-8 expression and concentrations are possibly related to the overexpression of CD3/IL8 lymphocytes, a phenomenon that has also been observed in colonic biopsies of ulcerative colitis (UC). Altogether, these observations could explain GEL formation along with other histological abnormalities but also suggest a pathogenic link between IDCP and UC, which becomes relevant since it may support recent considerations of the role of UC as a diagnostic aid for IDCP[87].
In contrast to IgG4-RD pancreatitis (AIP), IDCP affects younger subjects and often presents with abdominal pain (60%-88%) and acute pancreatitis (40%-63%), and nearly one-third of these cases present with idiopathic recurrent episodes.
Jaundice (25%-33%) and either diffuse (28%; although a single series reported this could be as high as 78%) or focal (26%-30%) pancreatic enlargement are less frequent.
Similar to IgG4-RD pancreatitis (AIP), acute episodes of pancreatitis in IDCP tend to be mild (clinical and radiological). Disease activity-related changes (IgG4-RD pancreatitis or IDCP) in imaging studies can be differentiated from those related to acute pancreatitis. The presence of a capsule-like low-density rim, delayed enhancement and multifocal main pancreatic duct narrowing favors an immune etiology (IgG4 RD pancreatitis or IDCP), while peripancreatic fat infiltration and pancreatic fluid collections favor acute pancreatitis[45].
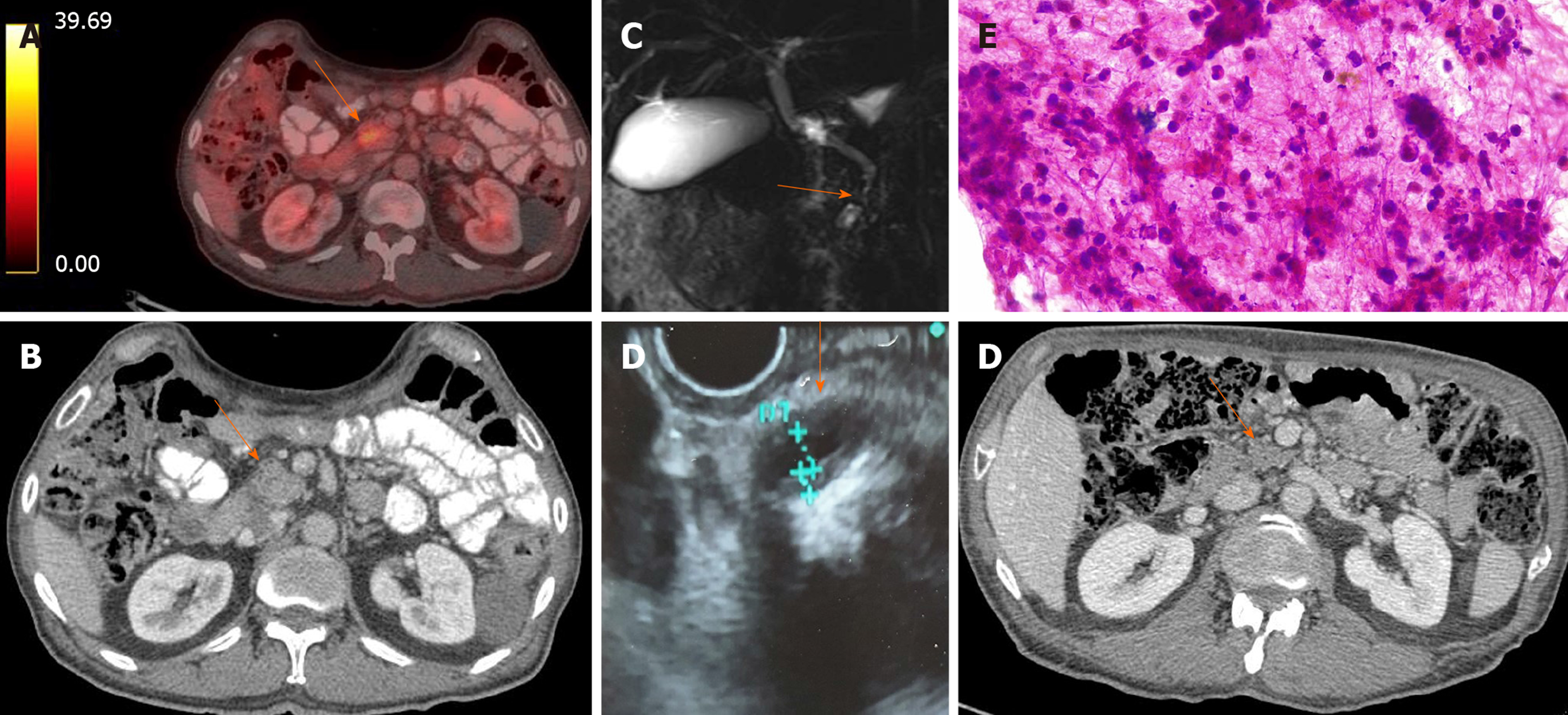
Other subtle but relevant image characteristics are MPD stenosis without upstream dilation and irregular and narrowed MPD with thickened walls observed during endoscopic ultrasound. A contrast enhanced CT scan may reveal hyperenhancement of the bile duct wall, hypoattenuation of the enlarged pancreas in the arterial phase and isoattenuation or hyperattenuation (compared to the spleen) in the portal venous phase, indicating delayed enhancement (some studies have objectively defined it as an increase of > 15 Hounsfield units from the arterial phase to the portal venous phase). IgG4 serum levels are usually within the normal range with, < 10% of cases presenting mildly elevated levels < 2 × upper limit of normal[45,89,90] (Figure 2).
Other organ involvement is not as common or varied as that in IgG4-RD pancreatitis (AIP). Inflammatory bowel disease (IBD) has been significantly associated with IDCP. UC is present in 33% to 44% of subjects, either before (47%), simultaneously (32%) or after (21%) the onset of IDCP, and most cases present pancolitis. Crohn’s disease is less frequent, but when present, intestinal involvement seems to be restricted to the colon[8,89,90].
The lack of a serum marker and OOI (except for those few cases presenting with IBD) pose a diagnostic challenge that may be overcome through pancreatic histology, which becomes almost mandatory in all cases.
Pancreatic biopsy provides valuable diagnostic information, but it might not be available everywhere, and even if it was, several technical caveats that are beyond the scope of this review surround and limit its systematic implementation.
Regardless of these dismal diagnostic pictures, it is possible to construct a clinical and imaging phenotype that suggests IDCP. In the clinical scenario of a pancreatic head mass, with no increased IgG4, no typical image characteristics and no OOI present, once a detailed diagnostic work-up has ruled out malignancy, the possibility of IDCP increases if the affected individual is younger than 50-years-old and presents with MPD stenosis lacking upstream dilation and history of acute pancreatitis or abdominal pain[86].
As mentioned previously, the presence of UC might increase diagnostic accuracy. Its presence in young patients presenting with presumed idiopathic recurrent acute pancreatitis suggests IDCP as the underlying cause of the pancreatic manifestations[88].
The ICDC are also used to diagnose IDCP. Compared to other criteria, the ICDC are highly sensitive (89%), specific (100%) and associated with adequate diagnostic accuracy (94%) for all autoimmune pancreatitides (IgG4-RD pancreatitis and IDCP) and for focal presentations (85%, 100% and 94%, respectively)[53].
Recent reports[88] suggest that UC should play a more important diagnostic role in IDCP. Thus far, a definite diagnosis of IDCP is made when GEL is demonstrated on pancreatic tissue. If GEL is not present, diagnosis can be considered based on the presence of level 2 histological findings, IBD and steroid responsiveness. IBD could increase diagnostic yield in those probable cases and no other specified cases (NOS).
AIP-NOS cases (those without solid criteria for IgG4-RD pancreatitis or IDCP) remain a challenge. Precisely in them, a thorough work-up focused on ruling out malignancy is imperative. If malignancy cannot be proven, collateral information (e.g., duct narrowing, age of presentation, history of abdominal pain or acute pancreatitis, presence of UC, among other) could suggest IDCP or IgG4-RD pancreatitis, and a steroid trial may be entertained. Whether AIP-NOS represents early or late stages of either IgG4-RD pancreatitis and IDCP or a different entity needs to be clarified[53,54].
Steroid regimens for IDCP are almost identical to IgG4-RD pancreatitis (AIP), but outcomes seem to be better, with fewer cases (< 10%) needing alternate and/or long-term maintenance steroid and nonsteroid therapies.
Recurrence rates in IDCP are low, ranging from 0% to 10%. A relapse rate as high as 25%, especially in those individuals with IDCP treated with steroids compared with those who underwent surgical resection. The reported cumulative relapse rate was 7.9% at 6 mo of follow-up, 10.6% at 1 year and 10.6% at 3 years. Acute pancreatitis was the most common relapse presentation.
Prompt diagnosis and treatment of both IDCP and IgG4-RD pancreatitis are encouraged since delay in initiating proper therapy results in exocrine pancreatic insufficiency in nearly half of the patients, and approximately 20% will develop diabetes mellitus[16,72,90,91].
Immune checkpoint inhibitors (ICIs) regulate T cell activation and proliferation and increase tumor CD8+ T cell activity, resulting in antitumor immunity. They include cytotoxic T lymphocyte antigen-4 (CTLA-4), PD-1 inhibitor and programmed death ligand-1 inhibitor, all of which have proven clinical efficiency in the treatment of multiple cancers, such as advanced melanoma and hepatocellular carcinoma[92,93] (Table 2).
| Immune checkpoint inhibitors | |
| Cytotoxic T lymphocyte antigen-4 | Ipilimumab |
| Programmed death-1 inhibitor | Nivolumab, pembrolizumab |
| Programmed death ligand-1 inhibitor | Atezolizumab, avelumab, durvalumab |
The consequent uncontrolled immune system activation mediated by the ICI can result in immune-related adverse events (iRAEs). These iRAEs commonly involve the gastrointestinal tract, lungs, liver, endocrine organs, and skin and represent an indication for discontinuing immunotherapy. The affected organ and iRAEs vary according to the type of ICI; colitis and hypophysitis are common with CTLA-4 blockade, while pneumonitis and thyroiditis are common with PD-1 blockade.
Prompt diagnosis and timely interventions in any iRAE, including corticosteroids and/or biologic therapy (e.g., mophetil mycophenolate or tacroliums), might prevent long-term toxicities and complications.
Due to the lack of information and novelty of this type of therapy, ICI-related pancreatic toxicity represents a diagnostic and therapeutic challenge. Pancreatic injury in this setting is usually asymptomatic; patients often present with normal imaging and increased lipase (frequently detected incidentally)[94].
The similarities and differences between “typical acute pancreatitis” and ICI-related acute pancreatitis (ICI-AP) remain to be clarified. According to Common Terminology Criteria for Adverse Events v4.03 from April 2011 through April 2018, any adverse effects are graded according to severity[95] (Table 3).
| Modified adverse events according to common terminology criteria for adverse events | ||
| Grade 1 | Mild | Asymptomatic or mild symptoms; clinical or diagnostic observations only |
| Grade 2 | Moderate | Enzyme elevation or radiologic findings only; minimal, local or noninvasive intervention indicated |
| Grade 3 | Severe | Lipase elevation > 2 times normal (60 U/L), severe pain, vomiting, needing medical intervention; hospitalization or prolongation of hospitalization indicated |
| Grade 4 | Life-threatening consequences | Urgent intervention indicated |
| Grade 5 | Death | Death related to adverse events |
Depending on serum and imaging findings, pancreatitis related to iRAEs can be considered grade 2 when the patient shows enzyme elevation or radiologic abnormalities and grade 3 when lipase elevation is > 2 times above the upper limit of normal, accompanied by severe abdominal pain, vomiting and the need for medical intervention (such as analgesic and nutritional support).
According to a recent systematic review and meta-analysis[96] that included trials with PD-1 inhibitors, CTLA-4 inhibitors and programmed death ligand-1 inhibitors in melanoma and nonmelanoma cancers, the incidence of asymptomatic lipase elevation after the use of ICIs was 2.7% (211/7702) and 1.9% for grade 2 pancreatitis.
Data from recent reports suggest that the type of ICI (e.g., CTL-4 inhibitors and PD-1 inhibitors) is associated with different clinical courses and outcomes. In solid tumors, the combination of CTLA-4 agents and other checkpoint inhibitors is associated with an increased incidence of any grade of lipase elevation and AP compared to monotherapy with any of these drugs[96]. Such an additive effect on the incidence of lipase elevation (14.29%) and grade 2 pancreatitis (10.60%) was noted when combining PD-1 and CTLA-4 inhibitors compared to the single use of either one. No reported deaths were attributed to immune-related pancreatitis. The incidence of any lipase elevation was significantly higher with CTLA-4 inhibitors than with PD-1 inhibitors (4.17% vs 1.26%). Patients treated with CTLA-4 inhibitors had an increased incidence of ICI-AP compared to patients who received PD-1 inhibitors (3.98%; 95%CI: 2.92-5.05 vs 0.94%; 95%CI: 0.48-1.40; P < 0.05).
In Su et al[97] meta-analysis CTLA 4 inhibitor marginally increased the risk for any lipase elevation (RR: 1.05; 95%CI: 1.01-2.24) but not the risk of acute pancreatitis compared to control groups. Melanoma patients were more prone to develop ICI-AP than nonmelanoma patients. Diabetes mellitus was documented in 14 cases (< 1%), but it was not associated with either ICI administration and/or pancreatitis. The incidence of lipase elevations and pancreatitis was significantly higher in the ICI group than in those receiving standard chemotherapy.
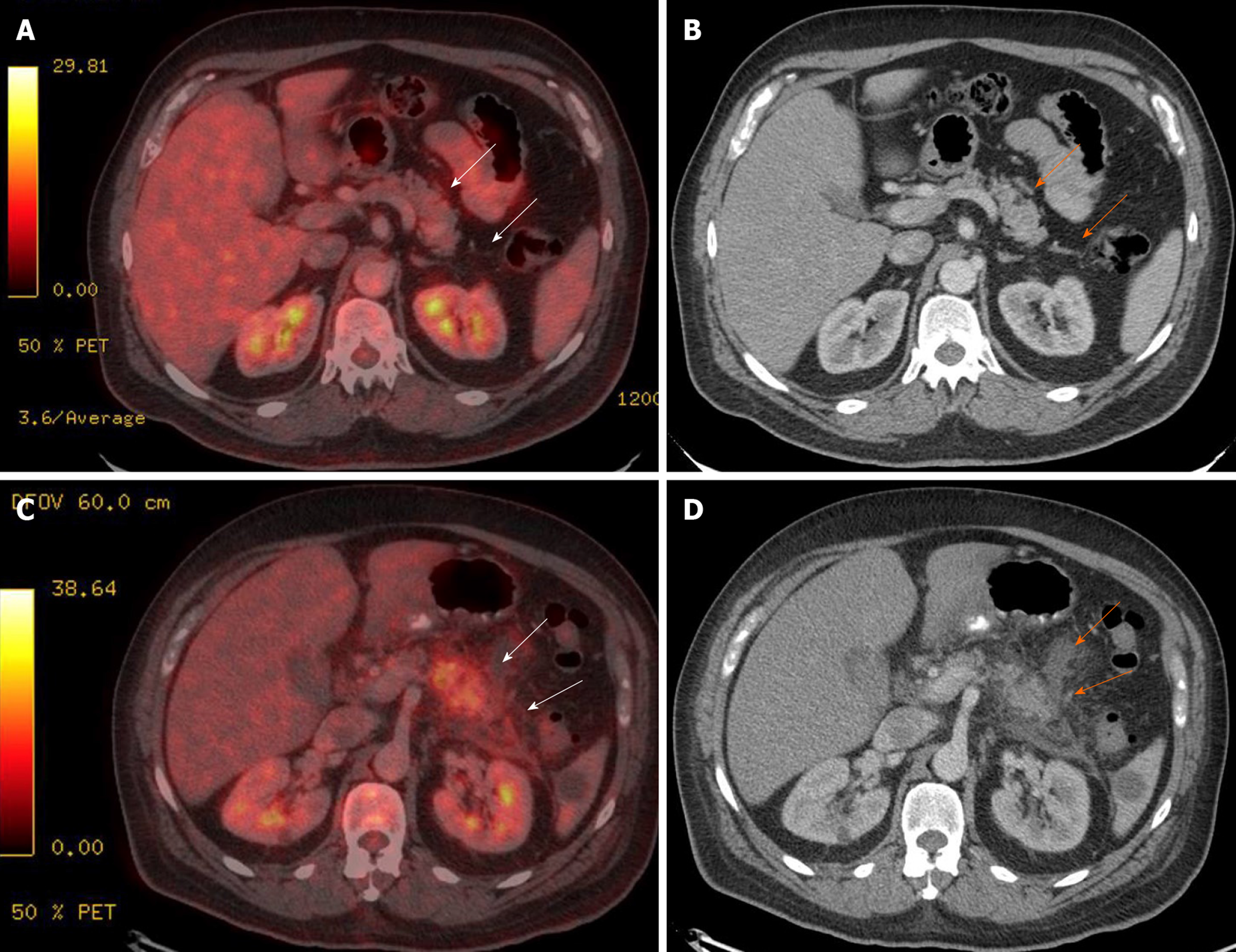
Symptoms of AP are present in up to 39% of cases, including epigastric pain (39%), nausea and vomiting (28%), fever (9%), and diarrhea (20%). Approximately 61% of cases present asymptomatic mild hyperlipasemia, but higher levels are seen in symptomatic pancreatitis. Additionally, CT findings suggestive of AP are more frequent in symptomatic than in asymptomatic patients (25% vs 6%, respectively) and include segmental parenchyma hypoenhancement, peripancreatic fat stranding, and pancreatic enlargement with heterogeneous enhancement. Short-and long-term outcomes and complications, such as pseudocysts (4%), chronic pancreatitis (4%) and diabetes mellitus (7%), are infrequent since most ICI–AP cases follow a mild course[98] (Figure 3).
The clinical significance of asymptomatic and isolated increases in pancreatic enzymes is unknown[99]. Pancreatic injury must be suspected in any patient complaining of new onset abdominal pain while on or after receiving ICI treatment.
The clinician should keep in mind that elevations of pancreatic enzyme serum levels are frequent and seldom fulfill the diagnostic criteria for AP. Only 10%-15% of cases will reach a grade 3 or higher pancreatic injury.
Lipase and amylase should be measured in the presence of any gastrointestinal symptom. Should any of them be elevated, abdominal imaging must be entertained.
ICI-AP management is similar to that suggested in different AP clinical guidelines; however, considering the effects of ICI on the immune system and observations in animal models suggesting that lesions in ICI-related pancreatitis resemble the injury and pathological mechanisms seen in AIP compared to those observed in usual AP etiologies, steroids are a plausible therapeutic alternative. Steroids in ICI-AP have demonstrated to be efficacious in limiting the intensity of the inflammatory process and subsequent complications[100].
As with any other ICI-related iRAEs, pancreatic injury treatment depends on the severity of the observed toxicity.
In grade 2 pancreatic injury, checkpoint inhibitors should be discontinued, and usual AP supportive and therapeutic measures should be started. If symptoms do not improve within 1 wk, steroids could be initiated (e.g., prednisone 0.5 mg/kg/d), and checkpoint inhibitors can be resumed once symptoms improve or severity is reduced to grade 1 or less.
For iRAEs grade 3 or higher ICI therapy should be discontinued permanently, and high doses of steroids (e.g., prednisone 1-2 mg/kg/d or equivalent) should be administered. These can be gradually tapered down over a month or more once symptoms disappear or reach grade 1 or less. As long as there are no other signs or symptoms of pancreatic inflammation, corticosteroid treatment is not indicated in patients with modest asymptomatic elevations in pancreatic enzymes[100,101].
No consensus on the type and dose of corticosteroid is available. Most authors recommend starting with high doses such as 1-2 mg/kg/d of methylprednisolone in single or divided doses followed by oral prednisone (40-60 mg/kg/d) with subsequent daily taper once the patient has improved. Response to corticosteroid therapy should be assessed in the next 48 to 72 h before considering moving to immunosuppressant. Some authors either increase the corticosteroid dose or switch to other immunosuppressive drugs including anti-TNF monoclonal antibodies[94,100,101].
Available information indicates that most ICI-AP cases follow a benign course, with low or no association with mortality; however, it has been reported that even several months after normalization of pancreatic serum enzymes and clinical remission, secondary pancreatic insufficiency, both exocrine and endocrine, may occur. Thus, patients will still benefit from prompt diagnosis and treatment since it would potentially reduce long-term complications.
No ICI-related toxicity predictive biomarker is available yet. Serum, immunologic and genetic candidates (e.g., IL-17, immunologically relevant gene expression, peripheral eosinophils) are being studied as a means to assess the risk of iRAEs[102,103]. Monitoring serum pancreatic enzymes in asymptomatic patients is not recommended unless pancreatitis is suspected clinically. As the use of these drugs increases, the iRAE frequency will rise. Clinical studies are needed to improve our understanding of ICI-related adverse effects.
Primary and secondary vasculitis can affect the gastrointestinal system. The most common primary disorders include: Anti-neutrophilic cytoplasmic antibodies, poliarteritis nodosa, Behcet’ SLE and rheumatoid arthritis, which are systemic diseases that can cause secondary vasculitis involving abdominal organs.
Patients with vasculitis affecting the gastrointestinal tract may present with fever, abdominal pain, nausea, vomiting, diarrhea, gastrointestinal bleeding and acute pancreatitis. Systemic vasculitis treatment is based on immunosuppressive therapy that has proven to increase survival and decrease related morbidities[104]. Among all gastrointestinal manifestations of vasculitis, AP is rare[105]. Available information is scant, with most of it coming from case reports and small series, with an estimated incidence < 5%[106-108].
Pancreatitis in these disorders may be due to disease activity causing direct vascular damage (vasculitis and connective tissue diseases related pancreatitis-VasC-AP) or indirectly secondary to pharmacological toxicity (drug-induced AP).
After exploring data from 895 patients attending SLE and from all AP cases admitted to our institution between January 1992 and July 2001, we identified 49 episodes of AP in 35 SLE patients (94% female). The estimated prevalence of AP in SLE patients was 3.5%, which is higher than that in the general population[109].
AP occurred in 14 patients within 2 years of the first clinical manifestation attributable to SLE and in 21 during the first 2 years of diagnosis (26 had a single episode of AP and 9 had recurrent attacks). Higher SLE activity was reported in those idiopathic episodes. Most of the episodes were severe (7 patients died during their first AP episode) and 40% of all cases occurred early in the course of SLE.
No clear causal relationship of drug (e.g., corticosteroids and azathioprine)-induced AP was established. Corticosteroids were administered at the onset of AP in 32 episodes. Overall, idiopathic AP was more frequent in SLE cases (46%) than in non-SLE AP controls (17%).
Similar observations were reported in 198 cases of pancreatitis in SLE[110]. A total of 173 patients (87.3%) were diagnosed with AP during the course of the autoimmune disease, but AP presented at the onset of SLE as one of the initial manifestations in 12.6% of cases. Symptoms included abdominal pain (98.9%), nausea or vomiting (24.1%) and fever (7.1%).
At the onset of AP, all patients showed evidence of active SLE. In the 25 cases that presented AP as the initial manifestation of SLE, 76% had hematological SLE activity (e.g., anemia, leukopenia and/or thrombocytopenia), 66% had malar rash, 33% had articular involvement, 28% had serositis and nearly 10% had neurological involvement.
SLE-related AP might present as subclinical pancreatitis (an increase in pancreatic enzyme levels without symptoms) or fulminant (with fatal outcome) or chronic disease, and the therapy of choice is high doses of corticosteroids.
SLE and vasculitis and connective tissue diseases patients with AP should undergo standardized AP management (e.g., analgesia, hydration, looking for common etiologies, prognostic evaluation, etc.) and simultaneously determine the presence and activity intensity of the underlying rheumatic disorder. Individual functional status and the damage that may have accrued as a consequence of the underlying rheumatic disease or its treatment must be assessed.
At presentation of AP, every vasculitis and connective tissue disorder of a patient requires a clinical evaluation looking for evidence of active vasculitis or damage that may be attributable to previous episodes of vasculitis. This must include a full assessment of the peripheral vasculature, complete ENT and fundoscopy exam, examination of the skin and nail beds, urine dipstick, and cardiorespiratory, abdominal, and neurological exam. Blood work should include liver function, acute phase reactants and estimation of renal function and hematology parameters[104,105]. The Birmingham Vasculitis Activity Score, which considers physician assessment to attribute clinical features to the presence of vasculitis may be used[111].
Once no other etiology (including drugs) is evident, SLE or other vasculitis and connective tissue disorder activity could account for the episode of AP, and treatment should include corticosteroids and/or other immune-suppressants. The participation of a rheumatology team is advisable even if vasculitis or a connective tissue disorder is not the cause. Both the causal factor of AP and the rheumatic disorder should be treated accordingly.
These observations suggest that patients suffering from SLE or any other vasculitis or connective tissue disorder are at an increased risk of idiopathic AP. These cases could be related to the underlying rheumatic disorder activity, acting as a cofactor causing either direct vascular damage or predisposing the pancreas to trigger an abnormal inflammatory response, which, according to prior observations, tends to be more severe and fatal. In cases of VasC-AP, steroid therapy has a key role; however, information pertaining to efficacy and outcomes is limited and needs to be further explored.
When comparing demographics, pathophysiology, clinical picture and outcomes of IDCP (AIP-2) and AIP-1 (IgG4-RD pancreatitis), both entities seem to only share the response to steroid therapy. This has led some authors to suggest that IDCP represents a totally different disorder and that the term AIP should be reserved only for AIP-1/LPSP.
A useful and correct definition is the one that states the meaning of a term, describing the concept and delineating the inclusion/exclusion boundaries. Current concepts regarding steroid-responsive pancreatitides only account for AIP and IDCP considering them as part of the same disease, despite evident and numerous differences.
A nomenclature showcasing each disorder´s particular pathophysiology (such as the recent mechanistic definition of chronic pancreatitis does)[112] and that includes other so far overlooked disorders that benefit from the same therapy would be clinically and scientifically relevant and beneficial.
We consider that the terms IgG4-RD pancreatitis and IDCP (rather than AIP/LPSP and AIP-2) better represent and allude to the particular clinical and pathological pictures of each disorder with both being part of a cluster of steroid-responsive pancreatitides that should include ICI-AP and VasC-AP.
A better understanding of the pathological and clinical characteristics of these entities could help us redefine and reclassify them. Even though AIP types 1 and 2 share a therapeutic response, each of them has different pathophysiology, epidemiology and clinical outcomes that suggest that they are 2 different entities. ICI-AP and VasC-AP have been overlooked, but they are part of a peculiar group of pancreatic disorders that need a special diagnostic approach and that benefit from adding corticosteroids to regular AP treatment. Further awareness and research are needed to increase their recognition and offer adequate and prompt therapeutics.
Manuscript source: Invited manuscript
Corresponding Author's Membership in Professional Societies: Asociación Mexicana de Gastroenterología; American Gastroenterological Association; American Society for Gastrointestinal Endoscopy; American Pancreatic Association; and American College of Gastroenterology.
Specialty type: Medicine, research and experimental
Country/Territory of origin: Mexico
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Anyfantakis D, Fraticelli P S-Editor: Zhang L L-Editor: Filipodia P-Editor: Liu JH
| 1. | Kim KP, Kim MH, Song MH, Lee SS, Seo DW, Lee SK. Autoimmune chronic pancreatitis. Am J Gastroenterol. 2004;99:1605-1616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 189] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 2. | SARLES H, SARLES JC, MURATORE R, GUIEN C. Chronic inflammatory sclerosis of the pancreas--an autonomous pancreatic disease? Am J Dig Dis. 1961;6:688-698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 536] [Cited by in RCA: 496] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 3. | Yoshida K, Toki F, Takeuchi T, Watanabe S, Shiratori K, Hayashi N. Chronic pancreatitis caused by an autoimmune abnormality. Proposal of the concept of autoimmune pancreatitis. Dig Dis Sci. 1995;40:1561-1568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1044] [Cited by in RCA: 925] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 4. | Uchida K, Okazaki K, Konishi Y, Ohana M, Takakuwa H, Hajiro K, Chiba T. Clinical analysis of autoimmune-related pancreatitis. Am J Gastroenterol. 2000;95:2788-2794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 138] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 5. | Klöppel G, Lüttges J, Löhr M, Zamboni G, Longnecker D. Autoimmune pancreatitis: pathological, clinical, and immunological features. Pancreas. 2003;27:14-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 154] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 6. | Hardacre JM, Iacobuzio-Donahue CA, Sohn TA, Abraham SC, Yeo CJ, Lillemoe KD, Choti MA, Campbell KA, Schulick RD, Hruban RH, Cameron JL, Leach SD. Results of pancreaticoduodenectomy for lymphoplasmacytic sclerosing pancreatitis. Ann Surg. 2003;237:853-8; discussion 858-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 129] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 7. | Notohara K, Burgart LJ, Yadav D, Chari S, Smyrk TC. Idiopathic chronic pancreatitis with periductal lymphoplasmacytic infiltration: clinicopathologic features of 35 cases. Am J Surg Pathol. 2003;27:1119-1127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 450] [Cited by in RCA: 382] [Article Influence: 17.4] [Reference Citation Analysis (1)] |
| 8. | Kamisawa T, Chari ST, Giday SA, Kim MH, Chung JB, Lee KT, Werner J, Bergmann F, Lerch MM, Mayerle J, Pickartz T, Lohr M, Schneider A, Frulloni L, Webster GJ, Reddy DN, Liao WC, Wang HP, Okazaki K, Shimosegawa T, Kloeppel G, Go VL. Clinical profile of autoimmune pancreatitis and its histological subtypes: an international multicenter survey. Pancreas. 2011;40:809-814. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 183] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 9. | Umehara H, Okazaki K, Masaki Y, Kawano M, Yamamoto M, Saeki T, Matsui S, Sumida T, Mimori T, Tanaka Y, Tsubota K, Yoshino T, Kawa S, Suzuki R, Takegami T, Tomosugi N, Kurose N, Ishigaki Y, Azumi A, Kojima M, Nakamura S, Inoue D; Research Program for Intractable Disease by Ministry of Health, Labor and Welfare (MHLW) Japan G4 team. A novel clinical entity, IgG4-related disease (IgG4RD): general concept and details. Mod Rheumatol. 2012;22:1-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 291] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 10. | Stone JH, Khosroshahi A, Deshpande V, Chan JK, Heathcote JG, Aalberse R, Azumi A, Bloch DB, Brugge WR, Carruthers MN, Cheuk W, Cornell L, Castillo CF, Ferry JA, Forcione D, Klöppel G, Hamilos DL, Kamisawa T, Kasashima S, Kawa S, Kawano M, Masaki Y, Notohara K, Okazaki K, Ryu JK, Saeki T, Sahani D, Sato Y, Smyrk T, Stone JR, Takahira M, Umehara H, Webster G, Yamamoto M, Yi E, Yoshino T, Zamboni G, Zen Y, Chari S. Recommendations for the nomenclature of IgG4-related disease and its individual organ system manifestations. Arthritis Rheum. 2012;64:3061-3067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 496] [Cited by in RCA: 488] [Article Influence: 40.7] [Reference Citation Analysis (0)] |
| 11. | Brito-Zerón P, Ramos-Casals M, Bosch X, Stone JH. The clinical spectrum of IgG4-related disease. Autoimmun Rev. 2014;13:1203-1210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 205] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 12. | Nishimori I, Tamakoshi A, Otsuki M; Research Committee on Intractable Diseases of the Pancreas, Ministry of Health, Labour, and Welfare of Japan. Prevalence of autoimmune pancreatitis in Japan from a nationwide survey in 2002. J Gastroenterol. 2007;42 Suppl 18:6-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 127] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 13. | Kanno A, Masamune A, Okazaki K, Kamisawa T, Kawa S, Nishimori I, Tsuji I, Shimosegawa T; Research Committee of Intractable Diseases of the Pancreas. Nationwide epidemiological survey of autoimmune pancreatitis in Japan in 2011. Pancreas. 2015;44:535-539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 109] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 14. | Masamune A, Kikuta K, Hamada S, Tsuji I, Takeyama Y, Shimosegawa T, Okazaki K; Collaborators. Nationwide epidemiological survey of autoimmune pancreatitis in Japan in 2016. J Gastroenterol. 2020;55:462-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 109] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 15. | Chari ST, Kloeppel G, Zhang L, Notohara K, Lerch MM, Shimosegawa T; Autoimmune Pancreatitis International Cooperative Study Group (APICS). Histopathologic and clinical subtypes of autoimmune pancreatitis: the Honolulu consensus document. Pancreas. 2010;39:549-554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 179] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 16. | Kamisawa T, Kim MH, Liao WC, Liu Q, Balakrishnan V, Okazaki K, Shimosegawa T, Chung JB, Lee KT, Wang HP, Lee TC, Choudhuri G. Clinical characteristics of 327 Asian patients with autoimmune pancreatitis based on Asian diagnostic criteria. Pancreas. 2011;40:200-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 69] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 17. | Hart PA, Kamisawa T, Brugge WR, Chung JB, Culver EL, Czakó L, Frulloni L, Go VL, Gress TM, Kim MH, Kawa S, Lee KT, Lerch MM, Liao WC, Löhr M, Okazaki K, Ryu JK, Schleinitz N, Shimizu K, Shimosegawa T, Soetikno R, Webster G, Yadav D, Zen Y, Chari ST. Long-term outcomes of autoimmune pancreatitis: a multicentre, international analysis. Gut. 2013;62:1771-1776. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 461] [Cited by in RCA: 374] [Article Influence: 31.2] [Reference Citation Analysis (0)] |
| 18. | Hamano H, Kawa S, Horiuchi A, Unno H, Furuya N, Akamatsu T, Fukushima M, Nikaido T, Nakayama K, Usuda N, Kiyosawa K. High serum IgG4 concentrations in patients with sclerosing pancreatitis. N Engl J Med. 2001;344:732-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2026] [Cited by in RCA: 1878] [Article Influence: 78.3] [Reference Citation Analysis (0)] |
| 19. | van der Neut Kolfschoten M, Schuurman J, Losen M, Bleeker WK, Martínez-Martínez P, Vermeulen E, den Bleker TH, Wiegman L, Vink T, Aarden LA, De Baets MH, van de Winkel JG, Aalberse RC, Parren PW. Anti-inflammatory activity of human IgG4 antibodies by dynamic Fab arm exchange. Science. 2007;317:1554-1557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 706] [Cited by in RCA: 714] [Article Influence: 39.7] [Reference Citation Analysis (0)] |
| 20. | Canfield SM, Morrison SL. The binding affinity of human IgG for its high affinity Fc receptor is determined by multiple amino acids in the CH2 domain and is modulated by the hinge region. J Exp Med. 1991;173:1483-1491. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 207] [Cited by in RCA: 212] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 21. | Miyoshi H, Uchida K, Taniguchi T, Yazumi S, Matsushita M, Takaoka M, Okazaki K. Circulating naïve and CD4+CD25high regulatory T cells in patients with autoimmune pancreatitis. Pancreas. 2008;36:133-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 147] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 22. | Zen Y, Fujii T, Harada K, Kawano M, Yamada K, Takahira M, Nakanuma Y. Th2 and regulatory immune reactions are increased in immunoglobin G4-related sclerosing pancreatitis and cholangitis. Hepatology. 2007;45:1538-1546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 526] [Cited by in RCA: 481] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 23. | Akitake R, Watanabe T, Zaima C, Uza N, Ida H, Tada S, Nishida N, Chiba T. Possible involvement of T helper type 2 responses to Toll-like receptor ligands in IgG4-related sclerosing disease. Gut. 2010;59:542-545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 68] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 24. | Kusuda T, Uchida K, Miyoshi H, Koyabu M, Satoi S, Takaoka M, Shikata N, Uemura Y, Okazaki K. Involvement of inducible costimulator- and interleukin 10-positive regulatory T cells in the development of IgG4-related autoimmune pancreatitis. Pancreas. 2011;40:1120-1130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 52] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 25. | Mattoo H, Mahajan VS, Della-Torre E, Sekigami Y, Carruthers M, Wallace ZS, Deshpande V, Stone JH, Pillai S. De novo oligoclonal expansions of circulating plasmablasts in active and relapsing IgG4-related disease. J Allergy Clin Immunol. 2014;134:679-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 259] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 26. | Zen Y, Britton D, Mitra V, Pike I, Heaton N, Quaglia A. A global proteomic study identifies distinct pathological features of IgG4-related and primary sclerosing cholangitis. Histopathology. 2016;68:796-809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 27. | Wallace ZS, Mattoo H, Carruthers M, Mahajan VS, Della Torre E, Lee H, Kulikova M, Deshpande V, Pillai S, Stone JH. Plasmablasts as a biomarker for IgG4-related disease, independent of serum IgG4 concentrations. Ann Rheum Dis. 2015;74:190-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 348] [Cited by in RCA: 319] [Article Influence: 31.9] [Reference Citation Analysis (0)] |
| 28. | Yanagawa M, Uchida K, Ando Y, Tomiyama T, Yamaguchi T, Ikeura T, Fukui T, Nishio A, Uemura Y, Miyara T, Okamoto H, Satoi S, Okazaki K. Correction to: Basophils activated via TLR signaling may contribute to pathophysiology of type 1 autoimmune pancreatitis. J Gastroenterol. 2018;53:582-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 29. | Mitsuyama T, Uchida K, Sumimoto K, Fukui Y, Ikeura T, Fukui T, Nishio A, Shikata N, Uemura Y, Satoi S, Mizuno N, Notohara K, Shimosegawa T, Zamboni G, Frulloni L, Okazaki K. Comparison of neutrophil infiltration between type 1 and type 2 autoimmune pancreatitis. Pancreatology. 2015;15:271-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 30. | Watanabe T, Yamashita K, Sakurai T, Kudo M, Shiokawa M, Uza N, Kodama Y, Uchida K, Okazaki K, Chiba T. Toll-like receptor activation in basophils contributes to the development of IgG4-related disease. J Gastroenterol. 2013;48:247-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 86] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 31. | Mohapatra S, Charilaou P, Sharma A, Singh DP, Sah RP, Murray D, Majumder S, Topazian MD, Chari ST. Significance of peripheral eosinophilia for diagnosis of IgG4-related disease in subjects with elevated serum IgG4 levels. Pancreatology. 2020;20:74-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 32. | Sah RP, Pannala R, Zhang L, Graham RP, Sugumar A, Chari ST. Eosinophilia and allergic disorders in autoimmune pancreatitis. Am J Gastroenterol. 2010;105:2485-2491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 41] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 33. | Löhr JM, Faissner R, Koczan D, Bewerunge P, Bassi C, Brors B, Eils R, Frulloni L, Funk A, Halangk W, Jesenofsky R, Kaderali L, Kleeff J, Krüger B, Lerch MM, Lösel R, Magnani M, Neumaier M, Nittka S, Sahin-Tóth M, Sänger J, Serafini S, Schnölzer M, Thierse HJ, Wandschneider S, Zamboni G, Klöppel G. Autoantibodies against the exocrine pancreas in autoimmune pancreatitis: gene and protein expression profiling and immunoassays identify pancreatic enzymes as a major target of the inflammatory process. Am J Gastroenterol. 2010;105:2060-2071. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 122] [Cited by in RCA: 106] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 34. | Frulloni L, Lunardi C, Simone R, Dolcino M, Scattolini C, Falconi M, Benini L, Vantini I, Corrocher R, Puccetti A. Identification of a novel antibody associated with autoimmune pancreatitis. N Engl J Med. 2009;361:2135-2142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 272] [Cited by in RCA: 248] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 35. | Okazaki K, Uchida K, Ohana M, Nakase H, Uose S, Inai M, Matsushima Y, Katamura K, Ohmori K, Chiba T. Autoimmune-related pancreatitis is associated with autoantibodies and a Th1/Th2-type cellular immune response. Gastroenterology. 2000;118:573-581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 410] [Cited by in RCA: 377] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 36. | Kawa S, Ota M, Yoshizawa K, Horiuchi A, Hamano H, Ochi Y, Nakayama K, Tokutake Y, Katsuyama Y, Saito S, Hasebe O, Kiyosawa K. HLA DRB10405-DQB10401 haplotype is associated with autoimmune pancreatitis in the Japanese population. Gastroenterology. 2002;122:1264-1269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 221] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 37. | Chang MC, Chang YT, Tien YW, Liang PC, Jan IS, Wei SC, Wong JM. T-cell regulatory gene CTLA-4 polymorphism/haplotype association with autoimmune pancreatitis. Clin Chem. 2007;53:1700-1705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 92] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 38. | Okazaki K, Uchida K. Current perspectives on autoimmune pancreatitis and IgG4-related disease. Proc Jpn Acad Ser B Phys Biol Sci. 2018;94:412-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 39. | Borges T, Silva S. IgG4-related disease: How to place it in the spectrum of immune-mediated and rheumatologic disorders? Mod Rheumatol. 2020;30:609-616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 40. | Jennette JC, Falk RJ, Bacon PA, Basu N, Cid MC, Ferrario F, Flores-Suarez LF, Gross WL, Guillevin L, Hagen EC, Hoffman GS, Jayne DR, Kallenberg CG, Lamprecht P, Langford CA, Luqmani RA, Mahr AD, Matteson EL, Merkel PA, Ozen S, Pusey CD, Rasmussen N, Rees AJ, Scott DG, Specks U, Stone JH, Takahashi K, Watts RA. 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 2013;65:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4416] [Cited by in RCA: 4256] [Article Influence: 354.7] [Reference Citation Analysis (0)] |
| 41. | Uchida K, Okazaki K. Clinical and pathophysiological aspects of type 1 autoimmune pancreatitis. J Gastroenterol. 2018;53:475-483. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 42. | Kamisawa T, Zen Y, Nakazawa T, Okazaki K. Advances in IgG4-related pancreatobiliary diseases. Lancet Gastroenterol Hepatol. 2018;3:575-585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 40] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 43. | Raina A, Yadav D, Krasinskas AM, McGrath KM, Khalid A, Sanders M, Whitcomb DC, Slivka A. Evaluation and management of autoimmune pancreatitis: experience at a large US center. Am J Gastroenterol. 2009;104:2295-2306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 181] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 44. | Ishikawa T, Itoh A, Kawashima H, Ohno E, Itoh Y, Nakamura Y, Hiramatsu T, Miyahara R, Ohmiya N, Haruta J, Goto H, Hirooka Y. Peripancreatic vascular involvements of autoimmune pancreatitis. J Gastroenterol Hepatol. 2012;27:1790-1795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 45. | Song TJ, Kim JH, Kim MH, Jang JW, Park DH, Lee SS, Seo DW, Lee SK, Yu E. Comparison of clinical findings between histologically confirmed type 1 and type 2 autoimmune pancreatitis. J Gastroenterol Hepatol. 2012;27:700-708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 46] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 46. | Frulloni L, Scattolini C, Falconi M, Zamboni G, Capelli P, Manfredi R, Graziani R, D'Onofrio M, Katsotourchi AM, Amodio A, Benini L, Vantini I. Autoimmune pancreatitis: differences between the focal and diffuse forms in 87 patients. Am J Gastroenterol. 2009;104:2288-2294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 154] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 47. | Negrelli R, Manfredi R, Pedrinolla B, Boninsegna E, Ventriglia A, Mehrabi S, Frulloni L, Pozzi Mucelli R. Pancreatic duct abnormalities in focal autoimmune pancreatitis: MR/MRCP imaging findings. Eur Radiol. 2015;25:359-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 51] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 48. | Zaheer A, Singh VK, Akshintala VS, Kawamoto S, Tsai SD, Gage KL, Fishman EK. Differentiating autoimmune pancreatitis from pancreatic adenocarcinoma using dual-phase computed tomography. J Comput Assist Tomogr. 2014;38:146-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 49. | Sahani DV, Kalva SP, Farrell J, Maher MM, Saini S, Mueller PR, Lauwers GY, Fernandez CD, Warshaw AL, Simeone JF. Autoimmune pancreatitis: imaging features. Radiology. 2004;233:345-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 312] [Cited by in RCA: 258] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 50. | Rasch S, Phillip V, Schmid RM, Algül H. Epidemiology, clinical presentation, diagnosis and treatment of autoimmune pancreatitis: A retrospective analysis of 53 patients. Pancreatology. 2016;16:73-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 51. | Takuma K, Kamisawa T, Anjiki H, Egawa N, Igarashi Y. Metachronous extrapancreatic lesions in autoimmune pancreatitis. Intern Med. 2010;49:529-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 52. | Kawa S, Okazaki K, Kamisawa T, Shimosegawa T, Tanaka M; Working members of Research Committee for Intractable Pancreatic Disease and Japan Pancreas Society. Japanese consensus guidelines for management of autoimmune pancreatitis: II. Extrapancreatic lesions, differential diagnosis. J Gastroenterol. 2010;45:355-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 47] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 53. | Bourlon MT, Bourlon C, Atisha-Fregoso Y, Chable-Montero F, Teliz MA, Angeles-Angeles A, Carrillo-Maravilla E, Llorente L, Uscanga LF. Clinical and Immunopathologic Profile of Mexican Patients with IgG4 Autoimmune Pancreatitis. ISRN Rheumatol. 2012;2012:164914. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 54. | Ghazale A, Chari ST, Smyrk TC, Levy MJ, Topazian MD, Takahashi N, Clain JE, Pearson RK, Pelaez-Luna M, Petersen BT, Vege SS, Farnell MB. Value of serum IgG4 in the diagnosis of autoimmune pancreatitis and in distinguishing it from pancreatic cancer. Am J Gastroenterol. 2007;102:1646-1653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 434] [Cited by in RCA: 362] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 55. | Raina A, Krasinskas AM, Greer JB, Lamb J, Fink E, Moser AJ, Zeh HJ, Slivka A, Whitcomb DC. Serum immunoglobulin G fraction 4 levels in pancreatic cancer: elevations not associated with autoimmune pancreatitis. Arch Pathol Lab Med. 2008;132:48-53. [PubMed] |
| 56. | Morselli-Labate AM, Pezzilli R. Usefulness of serum IgG4 in the diagnosis and follow up of autoimmune pancreatitis: A systematic literature review and meta-analysis. J Gastroenterol Hepatol. 2009;24:15-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 89] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 57. | Umehara H, Okazaki K, Masaki Y, Kawano M, Yamamoto M, Saeki T, Matsui S, Yoshino T, Nakamura S, Kawa S, Hamano H, Kamisawa T, Shimosegawa T, Shimatsu A, Nakamura S, Ito T, Notohara K, Sumida T, Tanaka Y, Mimori T, Chiba T, Mishima M, Hibi T, Tsubouchi H, Inui K, Ohara H. Comprehensive diagnostic criteria for IgG4-related disease (IgG4-RD), 2011. Mod Rheumatol. 2012;22:21-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 625] [Article Influence: 48.1] [Reference Citation Analysis (0)] |
| 58. | Chari ST, Takahashi N, Levy MJ, Smyrk TC, Clain JE, Pearson RK, Petersen BT, Topazian MA, Vege SS. A diagnostic strategy to distinguish autoimmune pancreatitis from pancreatic cancer. Clin Gastroenterol Hepatol. 2009;7:1097-1103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 224] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 59. | Kamisawa T, Imai M, Yui Chen P, Tu Y, Egawa N, Tsuruta K, Okamoto A, Suzuki M, Kamata N. Strategy for differentiating autoimmune pancreatitis from pancreatic cancer. Pancreas. 2008;37:e62-e67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 110] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 60. | Shimosegawa T, Chari ST, Frulloni L, Kamisawa T, Kawa S, Mino-Kenudson M, Kim MH, Klöppel G, Lerch MM, Löhr M, Notohara K, Okazaki K, Schneider A, Zhang L; International Association of Pancreatology. International consensus diagnostic criteria for autoimmune pancreatitis: guidelines of the International Association of Pancreatology. Pancreas. 2011;40:352-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1050] [Cited by in RCA: 1058] [Article Influence: 75.6] [Reference Citation Analysis (0)] |
| 61. | Naitoh I, Nakazawa T, Hayashi K, Miyabe K, Shimizu S, Kondo H, Yoshida M, Yamashita H, Umemura S, Hori Y, Ohara H, Joh T. Clinical evaluation of international consensus diagnostic criteria for type 1 autoimmune pancreatitis in comparison with Japanese diagnostic criteria 2011. Pancreas. 2013;42:1238-1244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 62. | Chang MC, Liang PC, Jan IS, Yang CY, Tien YW, Wei SC, Wong JM, Chang YT. Comparison and validation of International Consensus Diagnostic Criteria for diagnosis of autoimmune pancreatitis from pancreatic cancer in a Taiwanese cohort. BMJ Open. 2014;4:e005900. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 63. | Sugumar A, Levy MJ, Kamisawa T, Webster GJ, Kim MH, Enders F, Amin Z, Baron TH, Chapman MH, Church NI, Clain JE, Egawa N, Johnson GJ, Okazaki K, Pearson RK, Pereira SP, Petersen BT, Read S, Sah RP, Sandanayake NS, Takahashi N, Topazian MD, Uchida K, Vege SS, Chari ST. Endoscopic retrograde pancreatography criteria to diagnose autoimmune pancreatitis: an international multicentre study. Gut. 2011;60:666-670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 81] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 64. | Imai K, Matsubayashi H, Fukutomi A, Uesaka K, Sasaki K, Ono H. Endoscopic ultrasonography-guided fine needle aspiration biopsy using 22-gauge needle in diagnosis of autoimmune pancreatitis. Dig Liver Dis. 2011;43:869-874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 61] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 65. | Kanno A, Ishida K, Hamada S, Fujishima F, Unno J, Kume K, Kikuta K, Hirota M, Masamune A, Satoh K, Notohara K, Shimosegawa T. Diagnosis of autoimmune pancreatitis by EUS-FNA by using a 22-gauge needle based on the International Consensus Diagnostic Criteria. Gastrointest Endosc. 2012;76:594-602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 99] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 66. | Kerdsirichairat T, Saini SD, Chamberlain PR, Prabhu A. Autoimmune Pancreatitis Diagnosed with Core Biopsy Obtained from a Novel Fork-Tip EUS Needle. ACG Case Rep J. 2017;4:e7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 67. | Bang SJ, Kim MH, Kim DH, Lee TY, Kwon S, Oh HC, Kim JY, Hwang CY, Lee SS, Seo DW, Lee SK, Song DE, Jang SJ. Is pancreatic core biopsy sufficient to diagnose autoimmune chronic pancreatitis? Pancreas. 2008;36:84-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 60] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 68. | Hirano K, Fukushima N, Tada M, Isayama H, Mizuno S, Yamamoto K, Yashima Y, Yagioka H, Sasaki T, Kogure H, Nakai Y, Sasahira N, Tsujino T, Kawabe T, Fukayama M, Omata M. Diagnostic utility of biopsy specimens for autoimmune pancreatitis. J Gastroenterol. 2009;44:765-773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 69. | Sepehr A, Mino-Kenudson M, Ogawa F, Brugge WR, Deshpande V, Lauwers GY. IgG4+ to IgG+ plasma cells ratio of ampulla can help differentiate autoimmune pancreatitis from other "mass forming" pancreatic lesions. Am J Surg Pathol. 2008;32:1770-1779. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 33] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 70. | Ikeura T, Manfredi R, Zamboni G, Negrelli R, Capelli P, Amodio A, Caliò A, Colletta G, Gabbrielli A, Benini L, Okazaki K, Vantini I, Frulloni L. Application of international consensus diagnostic criteria to an Italian series of autoimmune pancreatitis. United European Gastroenterol J. 2013;1:276-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 71. | Kamisawa T, Shimosegawa T, Okazaki K, Nishino T, Watanabe H, Kanno A, Okumura F, Nishikawa T, Kobayashi K, Ichiya T, Takatori H, Yamakita K, Kubota K, Hamano H, Okamura K, Hirano K, Ito T, Ko SB, Omata M. Standard steroid treatment for autoimmune pancreatitis. Gut. 2009;58:1504-1507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 500] [Cited by in RCA: 485] [Article Influence: 30.3] [Reference Citation Analysis (0)] |
| 72. | Maire F, Le Baleur Y, Rebours V, Vullierme MP, Couvelard A, Voitot H, Sauvanet A, Hentic O, Lévy P, Ruszniewski P, Hammel P. Outcome of patients with type 1 or 2 autoimmune pancreatitis. Am J Gastroenterol. 2011;106:151-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 130] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 73. | Okazaki K, Chari ST, Frulloni L, Lerch MM, Kamisawa T, Kawa S, Kim MH, Lévy P, Masamune A, Webster G, Shimosegawa T. International consensus for the treatment of autoimmune pancreatitis. Pancreatology. 2017;17:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 161] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 74. | Gardner TB, Levy MJ, Takahashi N, Smyrk TC, Chari ST. Misdiagnosis of autoimmune pancreatitis: a caution to clinicians. Am J Gastroenterol. 2009;104:1620-1623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 60] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 75. | Kubota K, Kamisawa T, Okazaki K, Kawa S, Hirano K, Hirooka Y, Uchida K, Shiomi H, Ohara H, Shimizu K, Arakura N, Kanno A, Sakagami J, Itoi T, Ito T, Ueki T, Nishino T, Inui K, Mizuno N, Yoshida H, Sugiyama M, Iwasaki E, Irisawa A, Shimosegawa T, Takeyama Y, Chiba T. Low-dose maintenance steroid treatment could reduce the relapse rate in patients with type 1 autoimmune pancreatitis: a long-term Japanese multicenter analysis of 510 patients. J Gastroenterol. 2017;52:955-964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 72] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 76. | Wakabayashi T, Kawaura Y, Satomura Y, Watanabe H, Motoo Y, Sawabu N. Long-term prognosis of duct-narrowing chronic pancreatitis: strategy for steroid treatment. Pancreas. 2005;30:31-39. [PubMed] |
| 77. | Masamune A, Nishimori I, Kikuta K, Tsuji I, Mizuno N, Iiyama T, Kanno A, Tachibana Y, Ito T, Kamisawa T, Uchida K, Hamano H, Yasuda H, Sakagami J, Mitoro A, Taguchi M, Kihara Y, Sugimoto H, Hirooka Y, Yamamoto S, Inui K, Inatomi O, Andoh A, Nakahara K, Miyakawa H, Hamada S, Kawa S, Okazaki K, Shimosegawa T; Research Committee of Intractable Pancreas Diseases in Japan. Randomised controlled trial of long-term maintenance corticosteroid therapy in patients with autoimmune pancreatitis. Gut. 2017;66:487-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 141] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 78. | Matsubayashi H, Ishiwatari H, Imai K, Kishida Y, Ito S, Hotta K, Yabuuchi Y, Yoshida M, Kakushima N, Takizawa K, Kawata N, Ono H. Steroid Therapy and Steroid Response in Autoimmune Pancreatitis. Int J Mol Sci. 2019;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 79. | Lee HW, Moon SH, Kim MH, Cho DH, Jun JH, Nam K, Song TJ, Park DH, Lee SS, Seo DW, Lee SK. Relapse rate and predictors of relapse in a large single center cohort of type 1 autoimmune pancreatitis: long-term follow-up results after steroid therapy with short-duration maintenance treatment. J Gastroenterol. 2018;53:967-977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 59] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 80. | Soliman H, Vullierme MP, Maire F, Hentic O, Ruszniewski P, Lévy P, Rebours V. Risk factors and treatment of relapses in autoimmune pancreatitis: Rituximab is safe and effective. United European Gastroenterol J. 2019;7:1073-1083. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 81. | Hart PA, Topazian MD, Witzig TE, Clain JE, Gleeson FC, Klebig RR, Levy MJ, Pearson RK, Petersen BT, Smyrk TC, Sugumar A, Takahashi N, Vege SS, Chari ST. Treatment of relapsing autoimmune pancreatitis with immunomodulators and rituximab: the Mayo Clinic experience. Gut. 2013;62:1607-1615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 261] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 82. | Yadav D, Notahara K, Smyrk TC, Clain JE, Pearson RK, Farnell MB, Chari ST. Idiopathic tumefactive chronic pancreatitis: clinical profile, histology, and natural history after resection. Clin Gastroenterol Hepatol. 2003;1:129-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 97] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 83. | Notohara K, Nishimori I, Mizuno N, Okazaki K, Ito T, Kawa S, Egawa S, Kihara Y, Kanno A, Masamune A, Shimosegawa T. Clinicopathological Features of Type 2 Autoimmune Pancreatitis in Japan: Results of a Multicenter Survey. Pancreas. 2015;44:1072-1077. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 84. | Zamboni G, Lüttges J, Capelli P, Frulloni L, Cavallini G, Pederzoli P, Leins A, Longnecker D, Klöppel G. Histopathological features of diagnostic and clinical relevance in autoimmune pancreatitis: a study on 53 resection specimens and 9 biopsy specimens. Virchows Arch. 2004;445:552-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 503] [Cited by in RCA: 440] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 85. | Kamisawa T, Notohara K, Shimosegawa T. Two clinicopathologic subtypes of autoimmune pancreatitis: LPSP and IDCP. Gastroenterology. 2010;139:22-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 48] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 86. | Sugumar A, Klöppel G, Chari ST. Autoimmune pancreatitis: pathologic subtypes and their implications for its diagnosis. Am J Gastroenterol. 2009;104:2308-10; quiz 2311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 68] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 87. | Peláez-Luna M, Medina-Campos C, Uscanga-Domínuez L, Hernandez-Calleros J, Chan-Nuñez C, Negrete E, Angeles A. A Nondilated Main Pancreatic Duct Predicts Type 2 Autoimmune Pancreatitis: Comparative Study of Resected Pancreatic Head Masses. Digestion. 2020;101:137-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (2)] |
| 88. | Ku Y, Hong SM, Fujikura K, Kim SJ, Akita M, Abe-Suzuki S, Shiomi H, Masuda A, Itoh T, Azuma T, Kim MH, Zen Y. IL-8 Expression in Granulocytic Epithelial Lesions of Idiopathic Duct-centric Pancreatitis (Type 2 Autoimmune Pancreatitis). Am J Surg Pathol. 2017;41:1129-1138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 89. | Hart PA, Levy MJ, Smyrk TC, Takahashi N, Abu Dayyeh BK, Clain JE, Gleeson FC, Pearson RK, Petersen BT, Topazian MD, Vege SS, Zhang L, Chari ST. Clinical profiles and outcomes in idiopathic duct-centric chronic pancreatitis (type 2 autoimmune pancreatitis): the Mayo Clinic experience. Gut. 2016;65:1702-1709. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 55] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 90. | Vujasinovic M, Valente R, Maier P, von Beckerath V, Haas SL, Arnelo U, Del Chiaro M, Kartalis N, Pozzi-Mucelli RM, Fernandez-Moro C, Verbeke CS, Yu J, Ye W, Löhr JM. Diagnosis, treatment and long-term outcome of autoimmune pancreatitis in Sweden. Pancreatology. 2018;18:900-904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 44] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 91. | Kubota K, Kamisawa T, Hirano K, Hirooka Y, Uchida K, Ikeura T, Shiomi H, Ohara H, Shimizu K, Arakura N, Kanno A, Sakagami J, Itoi T, Ito T, Ueki T, Nishino T, Inui K, Mizuno N, Yoshida H, Sugiyama M, Iwasaki E, Irisawa A, Okazaki K, Kawa S, Shimosegawa T, Takeyama Y, Chiba T. Clinical course of type 1 autoimmune pancreatitis patients without steroid treatment: a Japanese multicenter study of 97 patients. J Hepatobiliary Pancreat Sci. 2018;25:223-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 92. | Reck M, Rodríguez-Abreu D, Robinson AG, Hui R, Csőszi T, Fülöp A, Gottfried M, Peled N, Tafreshi A, Cuffe S, O'Brien M, Rao S, Hotta K, Leiby MA, Lubiniecki GM, Shentu Y, Rangwala R, Brahmer JR; KEYNOTE-024 Investigators. Pembrolizumab versus Chemotherapy for PD-L1-Positive Non-Small-Cell Lung Cancer. N Engl J Med. 2016;375:1823-1833. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5948] [Cited by in RCA: 7525] [Article Influence: 836.1] [Reference Citation Analysis (0)] |
| 93. | Larkin J, Hodi FS, Wolchok JD. Combined Nivolumab and Ipilimumab or Monotherapy in Untreated Melanoma. N Engl J Med. 2015;373:1270-1271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 377] [Cited by in RCA: 569] [Article Influence: 56.9] [Reference Citation Analysis (0)] |
| 94. | Friedman CF, Clark V, Raikhel AV, Barz T, Shoushtari AN, Momtaz P, Callahan MK, Wolchok JD, Chapman PB, Hellmann MD, Postow MA. Thinking Critically About Classifying Adverse Events: Incidence of Pancreatitis in Patients Treated With Nivolumab + Ipilimumab. J Natl Cancer Inst. 2017;109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 53] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 95. | Cancer Therapy Evaluation Program. CTCAE: Common Terminology Criteria for Adverse Events. Available from: https://ctep.cancer.gov/protocolDevelopment/adverse_effects.htm. |
| 96. | George J, Bajaj D, Sankaramangalam K, Yoo JW, Joshi NS, Gettinger S, Price C, Farrell JJ. Incidence of pancreatitis with the use of immune checkpoint inhibitors (ICI) in advanced cancers: A systematic review and meta-analysis. Pancreatology. 2019;19:587-594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 79] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 97. | Su Q, Zhang XC, Zhang CG, Hou YL, Yao YX, Cao BW. Risk of Immune-Related Pancreatitis in Patients with Solid Tumors Treated with Immune Checkpoint Inhibitors: Systematic Assessment with Meta-Analysis. J Immunol Res. 2018;2018:1027323. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 40] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 98. | Abu-Sbeih H, Tang T, Lu Y, Thirumurthi S, Altan M, Jazaeri AA, Dadu R, Coronel E, Wang Y. Clinical characteristics and outcomes of immune checkpoint inhibitor-induced pancreatic injury. J Immunother Cancer. 2019;7:31. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 108] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 99. | Gullo L. Chronic nonpathological hyperamylasemia of pancreatic origin. Gastroenterology. 1996;110:1905-1908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 58] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 100. | Weber JS, Postow M, Lao CD, Schadendorf D. Management of Adverse Events Following Treatment With Anti-Programmed Death-1 Agents. Oncologist. 2016;21:1230-1240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 182] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 101. | Hsu C, Marshall JL, He AR. Workup and Management of Immune-Mediated Hepatobiliary Pancreatic Toxicities That Develop During Immune Checkpoint Inhibitor Treatment. Oncologist. 2020;25:105-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 102. | Callahan MK, Yang A, Tandon S, Xu Y, Subudhi SK, Roman RA, Heine AI, Pogoriler E, Kuk ED, Panageas K, Yuan JD, Allison JP, and Wolchok JD. Evaluation of serum IL-17 levels during ipilimumab therapy: Correlation with colitis. J Clin Oncol. 2011;29:2505-2505. [RCA] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 80] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 103. | Postow M, Wolchok J. Special consideration and toxicities associated with check point inhibitor immunotherapy. Up to date 2020. Available from: http://www.uptodate.com last accessed march 13th 2020. |
| 104. | McLaren JS, McRorie ER, Luqmani RA. Diagnosis and assessment of systemic vasculitis. Clin Exp Rheumatol. 2002;20:854-862. [PubMed] |
| 105. | Hatemi I, Hatemi G, Çelik AF. Systemic vasculitis and the gut. Curr Opin Rheumatol. 2017;29:33-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 106. | Pagnoux C, Mahr A, Cohen P, Guillevin L. Presentation and outcome of gastrointestinal involvement in systemic necrotizing vasculitides: analysis of 62 patients with polyarteritis nodosa, microscopic polyangiitis, Wegener granulomatosis, Churg-Strauss syndrome, or rheumatoid arthritis-associated vasculitis. Medicine (Baltimore). 2005;84:115-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 256] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 107. | Abu-Hilal M, Abu-Hilal M, McPhail MJ, Zeidan B, Bryant T, Bateman A, Johnson CD. Acute pancreatitis as the first presentation of Wegener's granulomatosis. JOP. 2008;9:300-304. [PubMed] |
| 108. | Matsubayashi H, Seki T, Niki S, Mizumura Y, Taguchi Y, Moriyasu F, Go K. Wegener's granulomatosis with onset of acute pancreatitis and rapid progress. A case report. Pancreatology. 2001;1:263-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 109. | Pascual-Ramos V, Duarte-Rojo A, Villa AR, Hernández-Cruz B, Alarcón-Segovia D, Alcocer-Varela J, Robles-Díaz G. Systemic lupus erythematosus as a cause and prognostic factor of acute pancreatitis. J Rheumatol. 2004;31:707-712. [PubMed] |
| 110. | Fasano S, Coscia MA, Valentini G. Subclinical Pancreatitis as Onset of Systemic Lupus Erythematosus: Case Report and Review of the Literature. J Clin Rheumatol. 2019;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 111. | Mukhtyar C, Lee R, Brown D, Carruthers D, Dasgupta B, Dubey S, Flossmann O, Hall C, Hollywood J, Jayne D, Jones R, Lanyon P, Muir A, Scott D, Young L, Luqmani RA. Modification and validation of the Birmingham Vasculitis Activity Score (version 3). Ann Rheum Dis. 2009;68:1827-1832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 856] [Cited by in RCA: 823] [Article Influence: 51.4] [Reference Citation Analysis (0)] |
| 112. | Whitcomb DC, Frulloni L, Garg P, Greer JB, Schneider A, Yadav D, Shimosegawa T. Chronic pancreatitis: An international draft consensus proposal for a new mechanistic definition. Pancreatology. 2016;16:218-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 359] [Cited by in RCA: 360] [Article Influence: 40.0] [Reference Citation Analysis (0)] |