Published online Mar 6, 2019. doi: 10.12998/wjcc.v7.i5.628
Peer-review started: December 25, 2018
First decision: December 29, 2018
Revised: January 9, 2019
Accepted: January 26, 2019
Article in press: January 26, 2019
Published online: March 6, 2019
Processing time: 73 Days and 20 Hours
The association of two congenital coronary artery anomalies (CAAs) is extremely rare but represents one of the main cause of sudden cardiac death in young athletes. Although coronary angiography (CX-A) is still widely used in childhood, cardiac magnetic resonance (C-MRI) and cardiac computed tomography (C-CT) have recently taken on an increasing diagnostic role in early detection of CAAs and concomitant congenital cardiac malformations.
A healthy 10-year-old male patient was referred to the Radiological Department of our Institution due to no evidence of left coronary artery in echocardiographic examination. With C-MRI was detected marked myocardial trabeculation and was suspected anomalous origin and course of left circumflex (LCx) artery and of left anterior descending (LAD) artery. With third generation Dual Source C-CT 192x2-sections (SOMATOM Force, Siemens, Germany) was confirmed anomalous origin of LCx artery from right pulmonary artery associated with anomalous origin of LAD artery from right coronary artery with course in front of right ventricular outflow tract. The patient underwent surgical treatment with reimplantation of the anomalous LCX and LAD arteries into the wall of ascending aorta, with no postoperative complications. The patient remained asymptomatic and follow-up C-MRI scan four months after operation showed complete success of surgery treatment.
This case highlights the diagnostic potential of C-CT and C-MRI in evaluation of CAAs and of cardiac morphology and functionality, with very low radiation dose and without the risks related to invasive procedure.
Core tip: Congenital coronary artery anomalies (CAAs) represent one of the main causes of sudden cardiac death in young athletes. For this reason, early detection of CAAs is essential. We present an extremely rare case of association of two congenital coronary anomalies, characterized by anomalous origin of left circumflex artery from right pulmonary artery and anomalous origin of left anterior descending artery from right coronary artery with course in front of right ventricular outflow tract. This case highlights the diagnostic potential of cardiac computed tomography and cardiac magnetic resonance in evaluation of CAAs and of cardiac morphology and functionality, with very low radiation dose and without the risks related to invasive procedure.
- Citation: Schicchi N, Fogante M, Giuseppetti GM, Giovagnoni A. Diagnostic detection with cardiac tomography and resonance of extremely rare coronary anomaly: A case report and review of literature. World J Clin Cases 2019; 7(5): 628-635
- URL: https://www.wjgnet.com/2307-8960/full/v7/i5/628.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v7.i5.628
Congenital coronary artery anomalies (CAAs) are uncommon and frequently associated with other cardiac malformations. Extremely rare is the contemporary association of two CAAs[1,2]. With adequate coronary collateralisation, the clinical course may be silent but, sometimes, patients may had severe clinical outcomes, including sudden cardiac death (SDC), and need surgical treatment in infancy. For these reasons, early detection and exact delineation of coronary origin and course are essential[3,4]. Most of CAAs are diagnosed by invasive procedure, like coronary angiography (CX-A), or during surgical treatment.
Here we describe the first case of a pediatric male patient with origin of left circumflex (LCx) artery from right pulmonary artery (RPA) associated with origin of left anterior descending (LAD) artery from right coronary artery (RCA). These anomalies were suspected with cardiac magnetic resonance (C-MRI) and confirmed with cardiac computed tomography (C-CT) with very low radiation dose and without the risks related to invasive procedure, like CX-A.
A healthy 10-year-old male patient (weight 53.3 kg and height 158.4 cm) was referred to the Radiological Department of our Institution.
He had not familiarity and risk factors for cardio-vascular diseases, congenital heart disease, arrhythmias and SCD. Karate practitioner (3 times/wk) without problems. Asymptomatic for chest pain, dyspnea, palpitation and syncope episodes.
The patient had a free previous medical history. In particular, born at term (38 wk), childbirth without any complications and birth weight was 2.4 kg. Regular growth.
Physical examination revealed no sign of cardiopathy and without any
cardiac added noises at chest auscultation. The patient’s temperature was 36 °C, heart rate was 72 bpm, respiratory rate was 13 breaths per minute, blood pressure was 110/70 mmHg and oxygen saturation in room air was 98%.
Blood analysis revealed normal haematocrit and platelet count. The blood biochemistries, as well as urine analysis were normal. Creatinine value was 0.9 mg/dL and creatinine clearance was 106 mL/min (estimated with Cockcroft-Gault formula).
Electrocardiography showed sinus rhythm, correct atrial-ventricular conduction (QRS 0.09 ms) and non-specific alterations of ventricular recovery (QTc 0.39 ms). Echocardiographic findings included normal volumes of left ventricle and atrium. Normal function of atrioventricular, aortic e pulmonary valves. No septal defects. Normal diameter of ascending aorta. No pericardial effusion. RCA origin high and ectatic. Left coronary artery origin and course not detect. For these reasons, patient was undergoing to C-MRI.
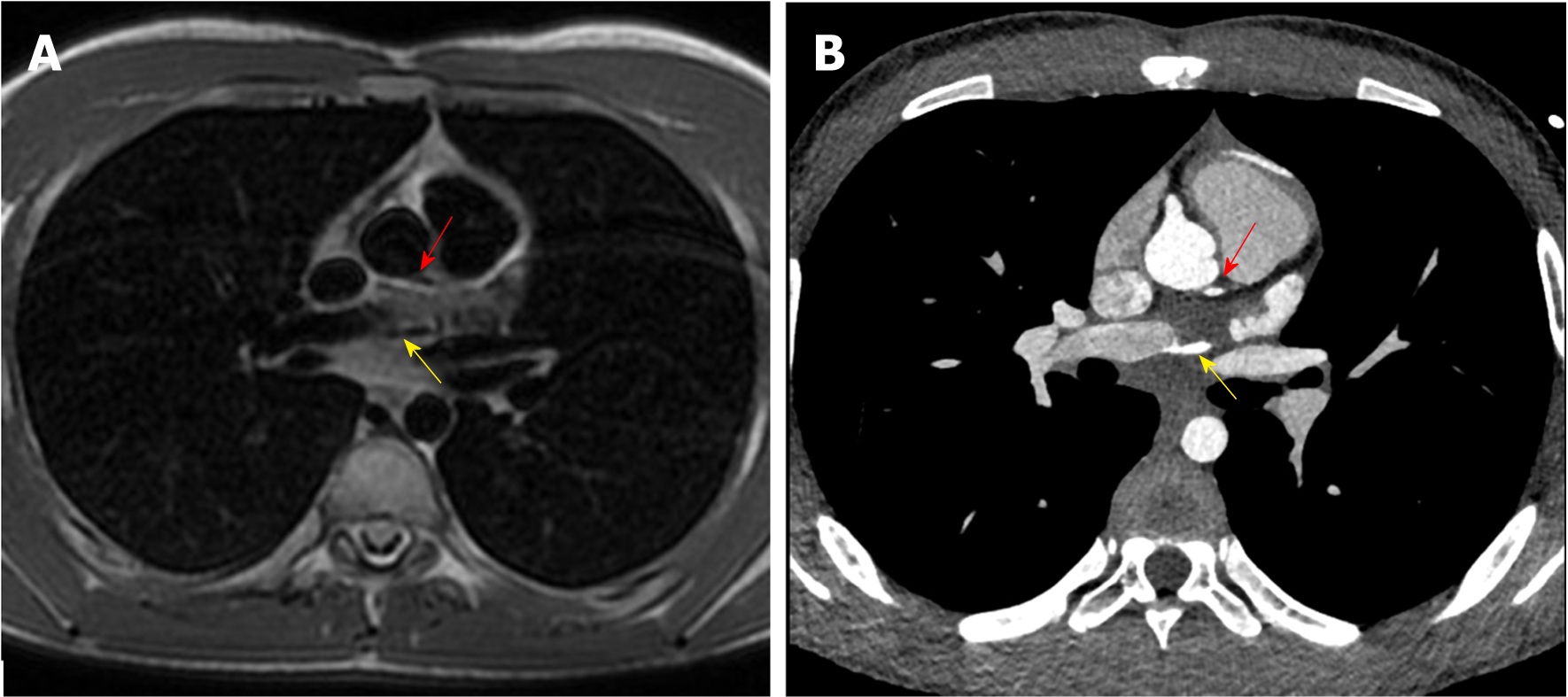
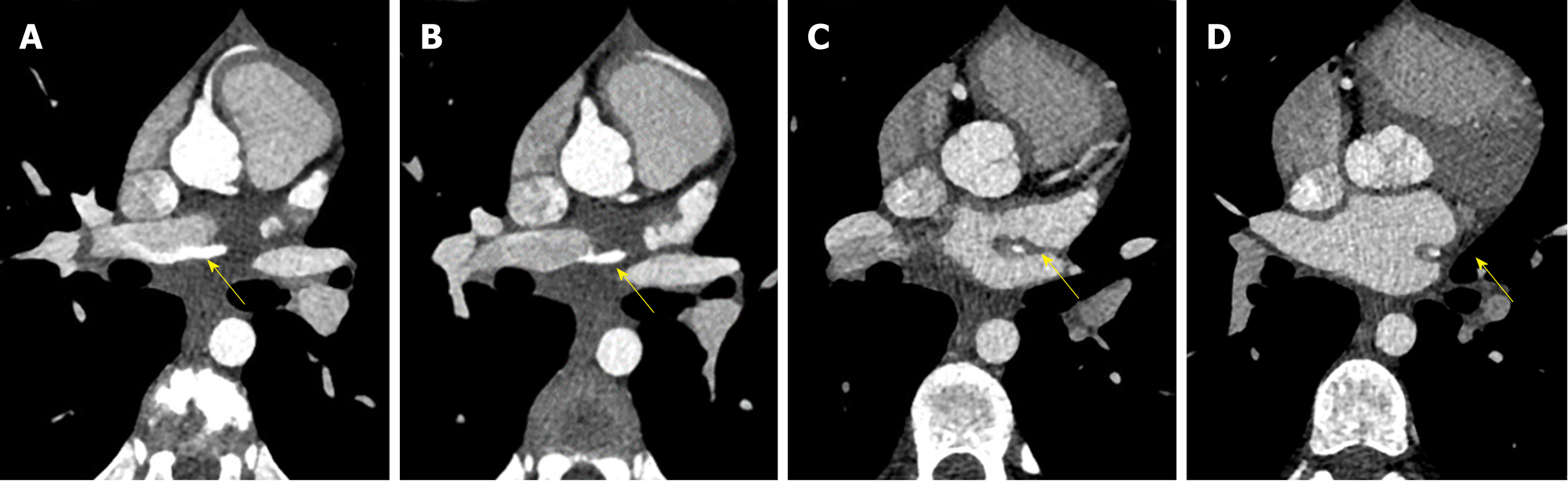
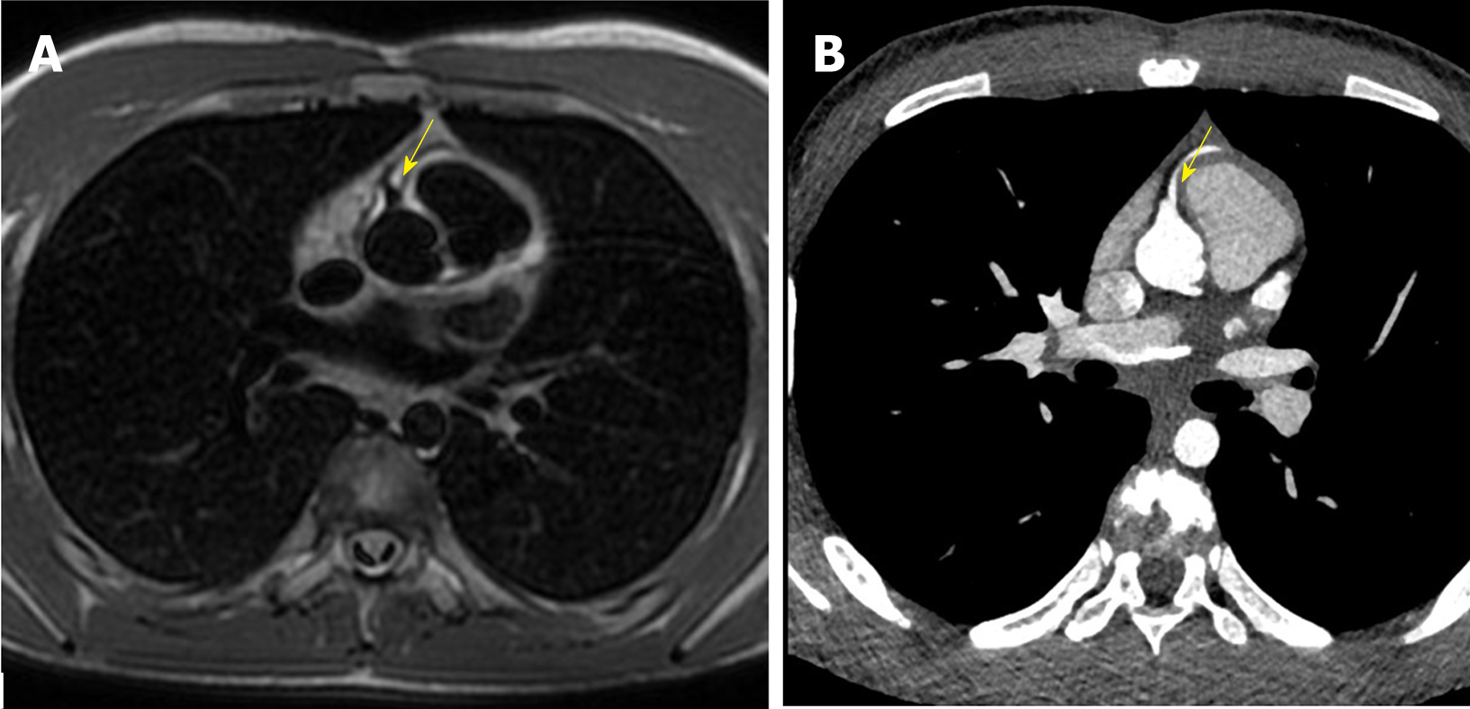
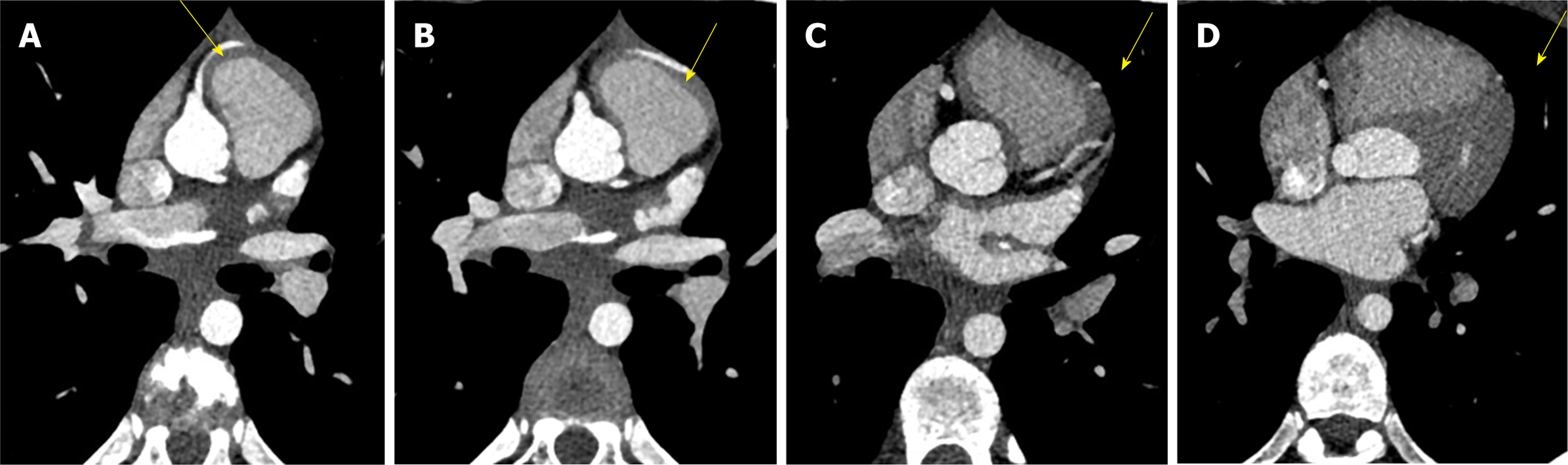
C-MRI (Achieva, Philips Medical Systems, The Netherlands) using a 1.5-T scanner was performed. Was used a 32-channel MR cardiac array coil with patient in prone position. C-MRI protocol consisted of balanced steady-state free precession (BSSFP) cine-imaging, T2-weighted and T1-weighted inversion recovery images, and sequences for late enhancement evaluation. Contrast agent dose was 0.2 mL/kg (gadobutrol, 1.0 mmol/mL) with flow injection rate of 2.0 mL/s. BSSFP sequences were acquired in the three cardiac axis, T2-weighted and T1-weighted in transversal cardiac axis, and sequences after contrast agent in short cardiac axis covering the entire left ventricle. The exam showed concordance in atrial-ventricular and ventricular-artery communication, regular return of superior vena cava and pulmonary veins. Left ventricle normal for volume, thickening of posterior wall with multiple mitral accessory ropes. Regular contractile functionality (ejection fraction of 71%). Accentuated endocardial trabeculation. Regular volume of left atrium. Regular right cardiac sections. Probable anomalies of origin and course of LCx and LAD arteries. For this reason, the patient was scanned with third generation Dual Source C-CT 192 × 2-sections (SOMATOM Force, Siemens, Germany). Patient remained in sinus rhythm before examination without receiving βblocker. Patient received a non-ionic low-osmolality contrast agent Visipaque 320 mgI/mL (iodixanol; GE Healthcare Life Sciences, Chalfont, United Kingdom), with a volume of 30 mL and flow injection rate of 5 mL/sec followed by saline injection at same volume and velocity. The scanning area began from the upper limit of the sternum to 1 cm below the diaphragm with superior to inferior direction. The parameters used for the exam were: 70 kV of tube voltage; automatic tube current modulation technique (CAREdose); rotation time, 0.28 sec; detector array, 192 × 0.6 mm; slice thickness, 0.75 mm and convolution kernel of B26. Was applied prospective ECG-triggered axial coronary protocol in a step-and-shoot scan mode and the exposure time was adjusted between 40%-70% of the cardiac cycle. After the examination, radiation CT dose index, dose length product and effective dose were recorded. Then the raw data was post-processed and was included the capture of the optimal images, multi-plane and maximum intensity projection reconstructions and volume rendering representations. C-CT confirmed anomalous origin of LCx artery from RPA associated with anomalous origin of LAD artery from RCA with proximal-medium course in front of RVOT and presences of coronary interconnections. Moreover, the examination showed the origin of a small diagonal branch from the left coronary sinus for the vascularization of the left ventricle wall (Figures 1-5).
The final diagnosis of the presented case is anomalous origin of LCx artery from RPA associated with anomalous origin of LAD artery from RCA with proximal-medium course in front of RVOT and presences of coronary interconnections.
The young patient underwent surgical treatment. The procedure began with standard median sternotomy, chest opening, thymus removal and pericardium opening. Then were cannulated the innominate artery and right atrium for cardiopulmonary bypass. After pulmonary arteries were clamped, the child was cooled down to 16 °C core temperature and circulatory arrest was commenced. Primarily, LCX was dissected from the RPA with a generous cuff, and was re-implanted into the wall of the aorta. Secondly, LAD was dissected from RCA and re-implated into the wall of aorta. Finally, after full rewarming, the operation was concluded and the sternum was closed.
The patient had an uneventful postoperative clinical course. At follow-up visit, two months after surgical treatment (one month after hospital discharge), the patient was asymptomatic. Follow-up C-MRI, four months after operation, showed complete success of surgery treatment.
Based on scientific works present in literature, the prevalence of CAAs in general population is around 1%; with a range from 0.3% to 5.6% in studies on patients undergoing CX-A, and in approximately 1% of routine autopsy. About 26% of CAAs are associated with other congenital cardiac malformations[5-7]. Separate origin of the LAD and LCX represents the commonest CAA with an incidence of 0.41%, followed by LCX arising from the RCA, with an incidence of 0.37%[8-10]. These anomalies, could be asymptomatic, but in some cases may lead to myocardial infarction, arrhythmia, ventricular aneurysm, mitral regurgitation, cardiomyopathy and SCD and they represent the second most common cause of SDC in young athletes. Therefore, the symptoms depend to the expansion of collateral vessels from coronary arteries. For this reason, patients need surgical treatment in early infancy[3,4]. Some potential pathogenetic mechanisms have been proposed to explain ischaemia and SCD, such as endothelial injury of the anomalous coronary artery with consequent sudden coronary spasm or modification of physiologic blood circulation. Another possible cause of ischemia is the acute angle of take-off of the anomalous vessel and the related slit-like orifice, which may become kinked and occluded during exercise[11-14]. Otherwise, it is interesting to note that in nearly 15% of patients with CAAs, myocardial ischemia can develop in the absence of atherosclerosis. For this reasons, early detection of CAAs and concomitant congenital cardiac malformations are crucial[15-17].
Although CX-A is still widely used in childhood, C-MRI and C-CT have recently taken on an increasing role in the diagnosis of CAAs. Indeed the association, of these two non- invasive examinations, allows the evaluation of cardiac morphology and functionality, with high spatial and temporal resolution, and permits to assess, exactly, coronary anatomy, without the risks related to invasive procedure and patient sedation and with a very low radiation dose[18-25].
Here we reported the case of a 10-year-old male patient with anomalous origin of LCx artery from RPA associated with anomalous origin and course of LAD artery from RCA in front of RVOT. This association was suspected with C-MRI and confirmed with C-CT without the necessity of CX-A, or other invasive procedures. To the best of our knowledge, the presented case is the first one in scientific literature that describes this anomaly association. Moreover, of 12 similar cases of CAAs reported in literature, only 2 were detected and confirmed with C-MRI or C-CT, the others were diagnosed with CX-A or during surgical treatment[26-37]. Our patient was a healthy child with no previous serious or recurrent or unusual cardiac symptoms. Electrocardiography examination was regular but with echocardiography, left coronary artery origin and course was not detect. For these reasons patient was undergoing to C-MRI. This exam provided informations on cardiac morphology and functionality and allowed to suspect anomalous origin and course of coronary arteries. Subsequently, third generation dual source C-CT electrocardiogram-gated, with high spatial resolution, guaranteed the detection of exact origin and course of coronary arteries, estimation of their diameter, and visualization of collateral vessels from coronary arteries. The radiation dose was very low, as summarized in Table 1, with effective dose of 3.87 mSv and without risks related to an invasive procedure and patient sedation. The patient had an uneventful postoperative clinical course and follow-up C-MRI showed complete success of surgery treatment.
| kV | mAs / ref. | CTDIvol (mGy) | DLP (mGycm) | |
| Scout_AP | 120 | 19 | 0.07 | 2.5 |
| Test Bolus | 70 | 70 | 5.62 | 5.6 |
| DS_CorAdSeq | 70 | 428/588 | 6.54 | 102.0 |
| Total | 110.0 |
Although CX-A is still widely used in childhood for the diagnosis of CAAs, C-MRI allows the evaluation of cardiac morphology and functionality and C-CT, with high spatial resolution, allows to evaluate, with high diagnostic accuracy, CAAs in children, without the risks related to invasive procedure and patient sedation and with a very low radiation dose.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, research and experimental
Country of origin: Italy
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Altarabsheh SE, Falconi M, Kharlamov AN, Najafi M S- Editor: Ji FF L- Editor: A E- Editor: Bian YN
| 1. | Angelini P. Coronary artery anomalies: an entity in search of an identity. Circulation. 2007;115:1296-1305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 466] [Cited by in RCA: 527] [Article Influence: 29.3] [Reference Citation Analysis (2)] |
| 2. | Goo HW. Coronary artery imaging in children. Korean J Radiol. 2015;16:239-250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 45] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 3. | Spicer DE, Henderson DJ, Chaudhry B, Mohun TJ, Anderson RH. The anatomy and development of normal and abnormal coronary arteries. Cardiol Young. 2015;25:1493-1503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 4. | Gavrielatos G, Letsas KP, Pappas LK, Antonellis I, Kardaras F. Anomalous origin of the entire coronary system with separate ostia within the right sinus of valsalva: a rare congenital anomaly and a review of the literature. Cardiology. 2007;107:209-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 5. | Young PM, Gerber TC, Williamson EE, Julsrud PR, Herfkens RJ. Cardiac imaging: Part 2, normal, variant, and anomalous configurations of the coronary vasculature. AJR Am J Roentgenol. 2011;197:816-826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 37] [Article Influence: 2.6] [Reference Citation Analysis (2)] |
| 6. | Angelini P. Coronary artery anomalies--current clinical issues: definitions, classification, incidence, clinical relevance, and treatment guidelines. Tex Heart Inst J. 2002;29:271-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 7. | Angelini P, Velasco JA, Flamm S. Coronary anomalies: incidence, pathophysiology, and clinical relevance. Circulation. 2002;105:2449-2454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 602] [Cited by in RCA: 579] [Article Influence: 25.2] [Reference Citation Analysis (1)] |
| 8. | Yamanaka O, Hobbs RE. Coronary artery anomalies in 126,595 patients undergoing coronary arteriography. Cathet Cardiovasc Diagn. 1990;21:28-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1278] [Cited by in RCA: 1361] [Article Influence: 38.9] [Reference Citation Analysis (1)] |
| 9. | Yurtdaş M, Gülen O. Anomalous origin of the right coronary artery from the left anterior descending artery: review of the literature. Cardiol J. 2012;19:122-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 10. | Alexander RW, Griffith GC. Anomalies of the coronary arteries and their clinical significance. Circulation. 1956;14:800-805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 257] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 11. | Frescura C, Basso C, Thiene G, Corrado D, Pennelli T, Angelini A, Daliento L. Anomalous origin of coronary arteries and risk of sudden death: a study based on an autopsy population of congenital heart disease. Hum Pathol. 1998;29:689-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 368] [Cited by in RCA: 351] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 12. | Camarda J, Berger S. Coronary artery abnormalities and sudden cardiac death. Pediatr Cardiol. 2012;33:434-438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 13. | Burke AP, Farb A, Virmani R, Goodin J, Smialek JE. Sports-related and non-sports-related sudden cardiac death in young adults. Am Heart J. 1991;121:568-575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 218] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 14. | Drory Y, Turetz Y, Hiss Y, Lev B, Fisman EZ, Pines A, Kramer MR. Sudden unexpected death in persons less than 40 years of age. Am J Cardiol. 1991;68:1388-1392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 305] [Cited by in RCA: 283] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 15. | Basso C, Maron BJ, Corrado D, Thiene G. Clinical profile of congenital coronary artery anomalies with origin from the wrong aortic sinus leading to sudden death in young competitive athletes. J Am Coll Cardiol. 2000;35:1493-1501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 791] [Cited by in RCA: 749] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 16. | Maron BJ, Thompson PD, Puffer JC, McGrew CA, Strong WB, Douglas PS, Clark LT, Mitten MJ, Crawford MD, Atkins DL, Driscoll DJ, Epstein AE. Cardiovascular preparticipation screening of competitive athletes: addendum: an addendum to a statement for health professionals from the Sudden Death Committee (Council on Clinical Cardiology) and the Congenital Cardiac Defects Committee (Council on Cardiovascular Disease in the Young), American Heart Association. Circulation. 1998;97:2294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 34] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 17. | Taylor AJ, Rogan KM, Virmani R. Sudden cardiac death associated with isolated congenital coronary artery anomalies. J Am Coll Cardiol. 1992;20:640-647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 556] [Cited by in RCA: 539] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 18. | Sun Z, Lin C, Davidson R, Dong C, Liao Y. Diagnostic value of 64-slice CT angiography in coronary artery disease: a systematic review. Eur J Radiol. 2008;67:78-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 102] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 19. | Abdulla J, Abildstrom SZ, Gotzsche O, Christensen E, Kober L, Torp-Pedersen C. 64-multislice detector computed tomography coronary angiography as potential alternative to conventional coronary angiography: a systematic review and meta-analysis. Eur Heart J. 2007;28:3042-3050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 243] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 20. | Mowatt G, Cook JA, Hillis GS, Walker S, Fraser C, Jia X, Waugh N. 64-Slice computed tomography angiography in the diagnosis and assessment of coronary artery disease: systematic review and meta-analysis. Heart. 2008;94:1386-1393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 387] [Cited by in RCA: 340] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 21. | Miller JM, Rochitte CE, Dewey M, Arbab-Zadeh A, Niinuma H, Gottlieb I, Paul N, Clouse ME, Shapiro EP, Hoe J, Lardo AC, Bush DE, de Roos A, Cox C, Brinker J, Lima JA. Diagnostic performance of coronary angiography by 64-row CT. N Engl J Med. 2008;359:2324-2336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1383] [Cited by in RCA: 1340] [Article Influence: 78.8] [Reference Citation Analysis (0)] |
| 22. | Min JK, Shaw LJ, Berman DS. The present state of coronary computed tomography angiography a process in evolution. J Am Coll Cardiol. 2010;55:957-965. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 136] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 23. | Bonello B, Kilner PJ. Review of the role of cardiovascular magnetic resonance in congenital heart disease, with a focus on right ventricle assessment. Arch Cardiovasc Dis. 2012;105:605-613. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 24. | Fratz S, Chung T, Greil GF, Samyn MM, Taylor AM, Valsangiacomo Buechel ER, Yoo SJ, Powell AJ. Guidelines and protocols for cardiovascular magnetic resonance in children and adults with congenital heart disease: SCMR expert consensus group on congenital heart disease. J Cardiovasc Magn Reson. 2013;15:51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 334] [Cited by in RCA: 346] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 25. | Kilner PJ. The role of cardiovascular magnetic resonance in adults with congenital heart disease. Prog Cardiovasc Dis. 2011;54:295-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 26. | Honey M, Lincoln JC, Osborne MP, de Bono DP. Coarctation of aorta with right aortic arch. Report of surgical correction in 2 cases: one with associated anomalous origin of left circumflex coronary artery from the right pulmonary artery. Br Heart J. 1975;37:937-945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 38] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 27. | Garcia CM, Chandler J, Russell R. Anomalous left circumflex coronary artery from the right pulmonary artery: first adult case report. Am Heart J. 1992;123:526-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 28. | Sarioglu T, Kinoglu B, Saltik L, Eroglu A. Anomalous origin of circumflex coronary artery from the right pulmonary artery associated with subaortic stenosis and coarctation of the aorta. Eur J Cardiothorac Surg. 1997;12:663-665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 29. | Korosoglou G, Ringwald G, Giannitsis E, Katus HA. Anomalous origin of the left circumflex coronary artery from the pulmonary artery. A very rare congenital anomaly in an adult patient diagnosed by cardiovascular magnetic resonance. J Cardiovasc Magn Reson. 2008;10:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 30. | Danov V, Kornovski V, Hazarbasanov D, Panayotov P. Anomalous origin of left circumflex coronary artery from the right pulmonary artery in adult. Thorac Cardiovasc Surg. 2009;57:114-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 31. | Jayagopal PB, Pai BV, Jayasree HN, Thomas A. Anomalous origin of left circumflex artery from pulmonary artery. Indian Heart J. 2014;66:214-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 32. | Jaworski R, Kansy A, Łaniewski-Wołłk P. Anomalous origin of circumflex coronary artery from right pulmonary artery in a hypoplastic left heart syndrome child. Kardiochir Torakochirurgia Pol. 2014;11:210-212. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 33. | Sekelyk R, Mykychak Y, Fedevych O, Yemets I. Anomalous origin of circumflex coronary artery from right pulmonary artery associated with coarctation of the aorta: a case report of surgical treatment. World J Pediatr Congenit Heart Surg. 2014;5:97-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 34. | Liu B, Fursevich D, O'Dell MC, Flores M, Feranec N. Anomalous Left Circumflex Coronary Artery Arising from the Right Pulmonary Artery: A Rare Cause of Aborted Sudden Cardiac Death. Cureus. 2016;8:e499. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 35. | Al-Muhaya MA, Syed A, Najjar AHA, Mofeed M, Al-Mutairi M. Anomalous origin of circumflex coronary artery from right pulmonary artery associated with atrial septal defect. J Saudi Heart Assoc. 2017;29:219-222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 36. | Daylan A, Ertugay S, Apaydın AZ, Oğuz E. Circumflex coronary artery originating from the right pulmonary artery in adult. Asian Cardiovasc Thorac Ann. 2017;25:528-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 37. | Bo I, Semple T, Cheasty E, Rubens MB, Ho SY, Rigby ML, Nicol ED. Left circumflex coronary artery from the pulmonary artery in scimitar syndrome. Pediatr Radiol. 2018;48:632-637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |