Published online Nov 26, 2019. doi: 10.12998/wjcc.v7.i22.3821
Peer-review started: July 31, 2019
First decision: September 9, 2019
Revised: September 30, 2019
Accepted: October 5, 2019
Article in press: October 5, 2019
Published online: November 26, 2019
Processing time: 117 Days and 18 Hours
Concomitant allergic fungal rhinosinusitis (AFRS) and allergic bronchopulmonary aspergillosis (ABPA) are extremely rare, with no more than 20 cases reported in the English literature.
A 52-year-old female patient complained of right-sided nasal obstruction, rhinorrhea, sneezing, epistaxis, and hyposmia for a period of around 5 mo. Nasal examination detected paleness and edema of the nasal mucous membrane and a polyp in the right middle meatus. A computed tomography (CT) scan of the sinuses revealed a ground-glass opacity filling the right maxillary and ethmoid sinuses, along with bone absorption in the medial wall of the right maxillary sinus. Magnetic resonance images were obtained with T1-weighted, T2-weighted, and gadolinium-enhanced T1-weighted sequences. A well-defined mass, located in the right maxillary and ethmoid sinuses and displaying obvious hypointense features, was observed on both T1- and T2-weighted images, with peripheral enhancement on gadolinium-enhanced T1-weighted images. The patient also has a 20-year history of cough and dyspnea. Chest CT revealed columned and cystiform bronchiectasis in the bilateral bronchus, surrounded by a large number of spotted and funicular high-density lesions. The level of serum total IgE was > 5000 kU/L. Serum IgE levels related to house dust and aspergillus showed a positive result, with the values being 3.5 kU/L and 1.2 kU/L. We performed functional endoscopic sinus surgery under local anesthesia. After surgery, topical glucocorticoids and saline irrigation were applied in the nasal cavity until the present time. An oral glucocorticoid (methylprednisolone 16 mg/d) and antifungal agent (itraconazole 200 mg/d) were also used for a period of 4 wk. Montelukast was prescribed at 10 mg/d until the present time. An endoscopic examination showed that the patient was recovering well at 3 mo after surgery.
Since different specialists treat ABPA and AFRS, their coexistence may be overlooked. AFRS accompanied by ABPA requires surgical therapy combined with medical control to improve the symptoms.
Core tip: Concomitant allergic fungal rhinosinusitis (AFRS) and allergic bronchopulmonary aspergillosis (ABPA) are extremely rare. We describe the clinical presentation and treatment in one patient with AFRS accompanied by ABPA and review the English literature from 1970 to 2016. The most common pulmonary symptom was dyspnea. Twelve patients underwent surgery on the sinuses, all patients received oral glucocorticoids for at least 2 wk, and 11 patients received topical glucocorticoids. All patients achieved a good prognosis. Since different specialists treat ABPA and AFRS, their coexistence may be overlooked. AFRS accompanied by ABPA requires surgical therapy combined with medical control to improve the symptoms.
- Citation: Cheng KJ, Zhou ML, Liu YC, Zhou SH. Allergic fungal rhinosinusitis accompanied by allergic bronchopulmonary aspergillosis: A case report and literature review. World J Clin Cases 2019; 7(22): 3821-3831
- URL: https://www.wjgnet.com/2307-8960/full/v7/i22/3821.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v7.i22.3821
Allergic fungal rhinosinusitis (AFRS) is a particular type of chronic rhinosinusitis associated with allergic reactions to fungal antigens. This disease was first described in 1983[1]. AFRS affects around 1%–2% of the world’s population, with the incidence differing among regions[2,3]. Currently, patients are defined as having AFRS by meeting the following criteria: (1) Type I hypersensitivity; (2) Nasal polyposis; (3) Characteristic computed tomography (CT) findings; (4) Eosinophilic mucin without invasion; and (5) Positive fungal stain[4]. AFRS usually requires endoscopic surgery and long-term postoperative medical treatment. Immunotherapy is also suggested for AFRS[5].
Allergic bronchopulmonary aspergillosis (ABPA) is a type I hypersensitivity to the fungus Aspergillus, and presents as a necrotizing pneumonia[6]. This entity was first described by Hinson et al[7] in 1952. The pathological changes of ABPA are similar to those of AFRS. The major diagnostic criteria for ABPA are as follows: (1) Asthma; (2) Elevated total serum immunoglobulin E (IgE); (3) Elevated total serum immunoglobulin G (IgG); (4) Presence of transient pulmonary infiltrates; (5) Central/proximal bronchiectasis with normal tapering of the distal bronchi; and (6) Elevated serum IgE and IgG to A. fumigatus[8]. ABPA usually accompanies asthma and requires oral antifungal and glucocorticoid treatment[9]. AFRS has a close relationship with ABPA because of the location in the unified airway and the similarity in the pathogenesis. AFRS is an ENT equivalent of ABPA.
ABPA can accompany several types of rhinomycosis, including invasive sinus aspergillosis[10], aspergilloma[11], and AFRS[11]. However, concomitant AFRS and ABPA are extremely rare; this may be because the two different diseases might be overlooked by pulmonary physicians or otolaryngologists. Some articles mentioned the occurrence of concomitant AFRS and ABPA but provided no detailed information[9]. No more than 20 cases of AFRS accompanied by ABPA have been reported in the English literature. Whether AFRS causes ABPA, ABPA causes AFRS, or AFRS occurs concurrently with ABPA is still open to debate. The treatment options for this disease are controversial and its prognosis is uncertain. In this article, we describe a case of AFRS accompanied by ABPA and review the literature. The clinical manifestation, management, and prognosis of the disease are also discussed.
Right-sided nasal obstruction, rhinorrhea, sneezing, epistaxis, and hyposmia.
Five months.
The patient had a 20-year history of asthma with poor pulmonary function. She manifested obvious dyspnea and cough on exertion and the symptoms were not fully relieved after the application of asthma medication.
None.
Nasal examination detected paleness and edema of the nasal mucous membrane and a polyp in the right middle meatus.
A blood test indicated an elevation of eosinophil level by 5.2%. The level of serum total IgE was > 5000 kU/L. Serum IgE levels related to house dust and aspergillus showed a positive result, with the values being 3.5 kU/L and 1.2 kU/L, respectively.
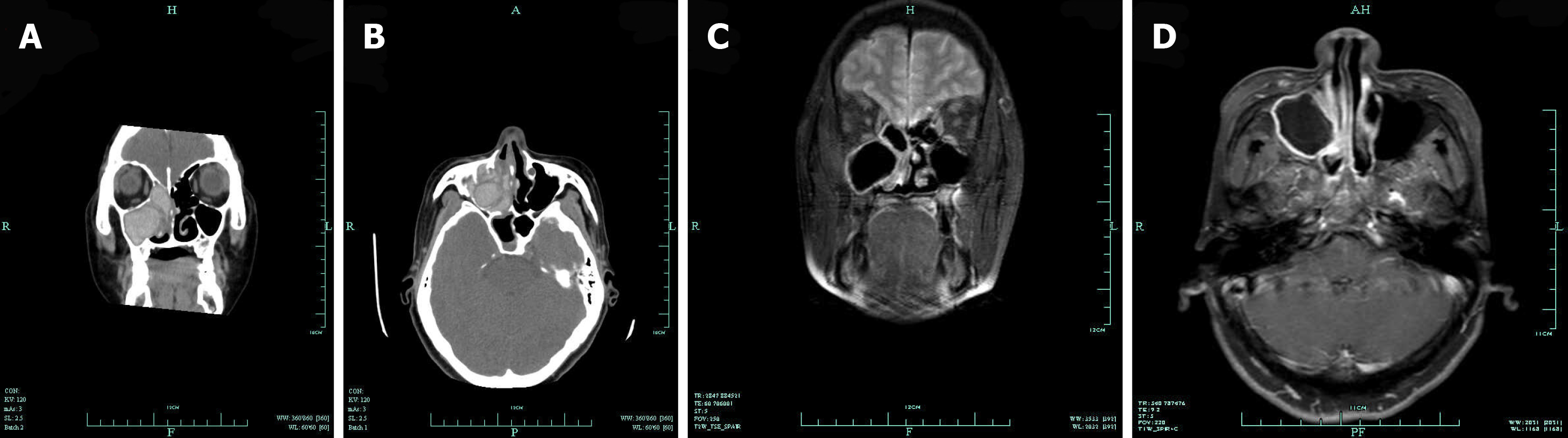
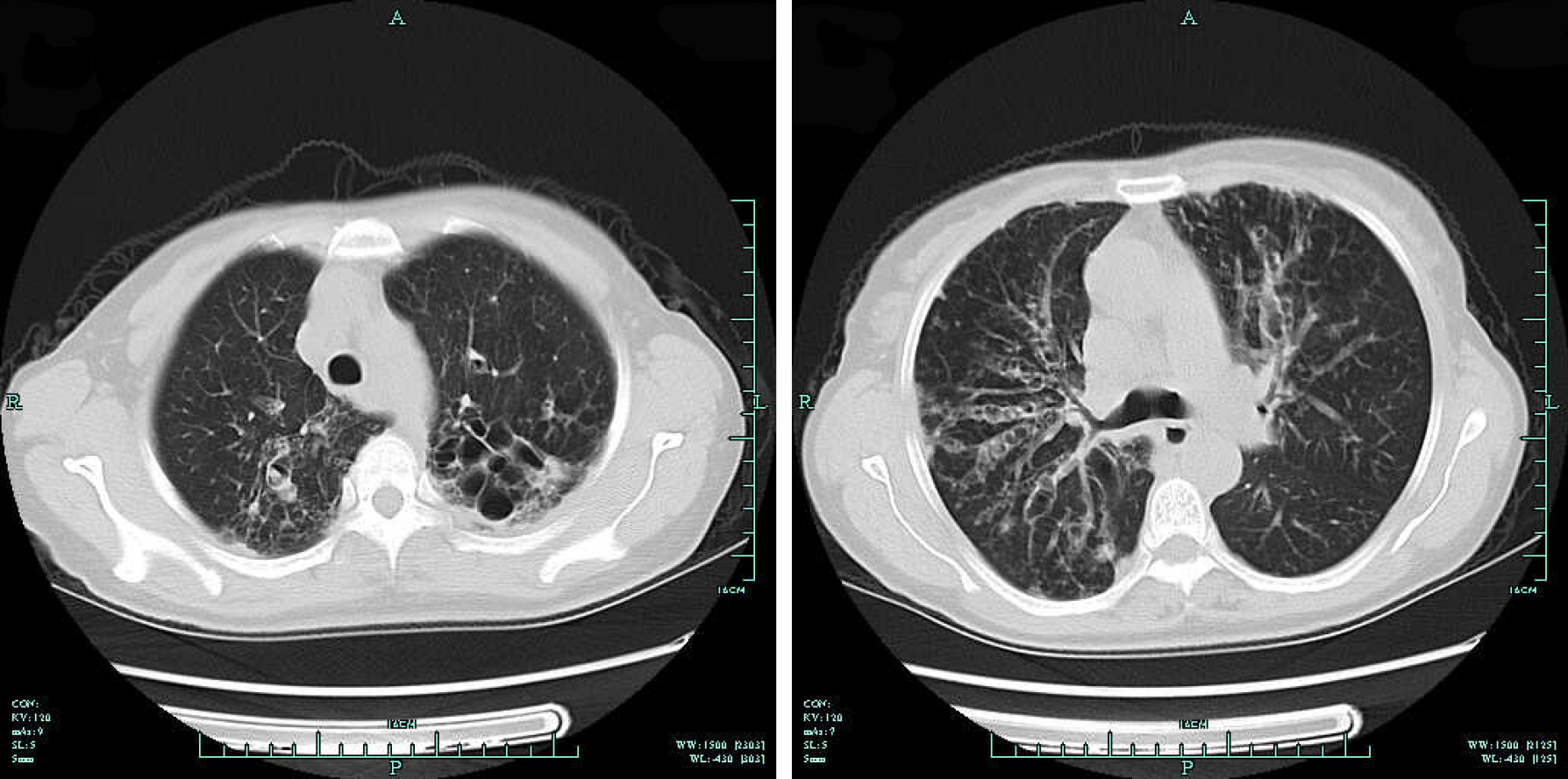
A CT scan of the sinuses revealed a ground-glass opacity filling the right maxillary and ethmoid sinuses, along with bone absorption in the medial wall of the right maxillary sinus (Figure 1A and B). The bones of the anterior skull base and lamina papyracea were unaffected. The other side of the sinuses also showed no involvement. Magnetic resonance images were obtained with T1-weighted, T2-weighted, and gadolinium-enhanced T1-weighted sequences. A well-defined mass, located in the right maxillary and ethmoid sinuses and displaying obvious hypointense features, was observed on both T1- and T2-weighted images, with peripheral enhancement on gadolinium-enhanced T1-weighted images (Figure 1C and D). A chest radiograph showed dilatation of the bilateral bronchus surrounded by a flocculent shadow and plaques. Chest CT revealed columned and cystiform bronchiectasis in the bilateral bronchus, surrounded by a large number of spotted and funicular high-density lesions (Figure 2A and B).
AFRS accompanied by ABPA.
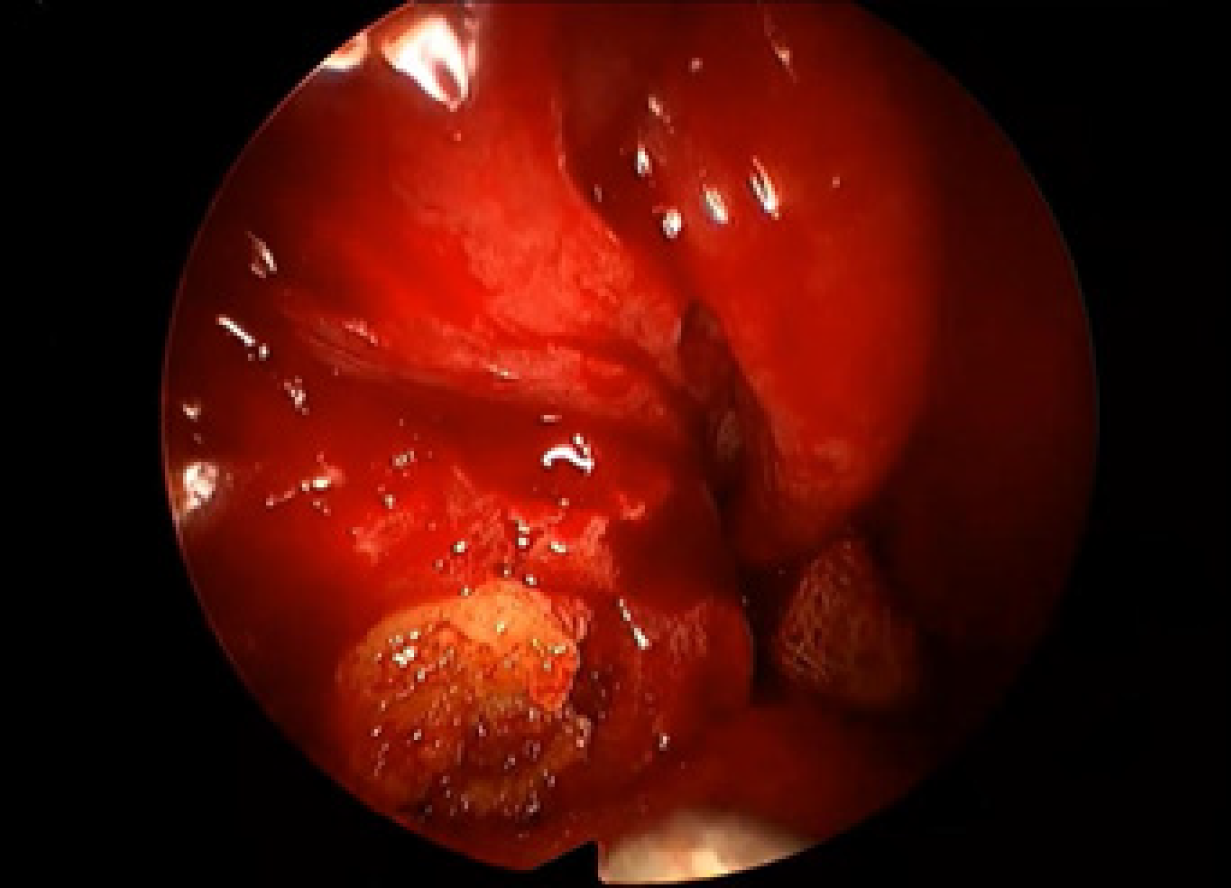
Because of the poor pulmonary function, the patient was unable to undergo surgery under general anesthesia. Therefore, we performed functional endoscopic sinus surgery under local anesthesia. We resected the polyp, opened the right maxillary and ethmoid sinus, and found a large amount of yellow, jam-like mucin in the right maxillary and ethmoid sinuses (Figure 3). We enlarged the ostium of the sinuses and cleared the mucin. The postoperative pathology found the aspergillus. The postoperative course was uneventful. After surgery, topical glucocorticoids and saline irrigation were applied in the nasal cavity until the present time. An oral glucocorticoid (methylprednisolone 16 mg/d) and antifungal agent (itraconazole 200 mg/d) were also used for a period of 4 wk. Montelukast was prescribed at 10 mg/d until the present time.
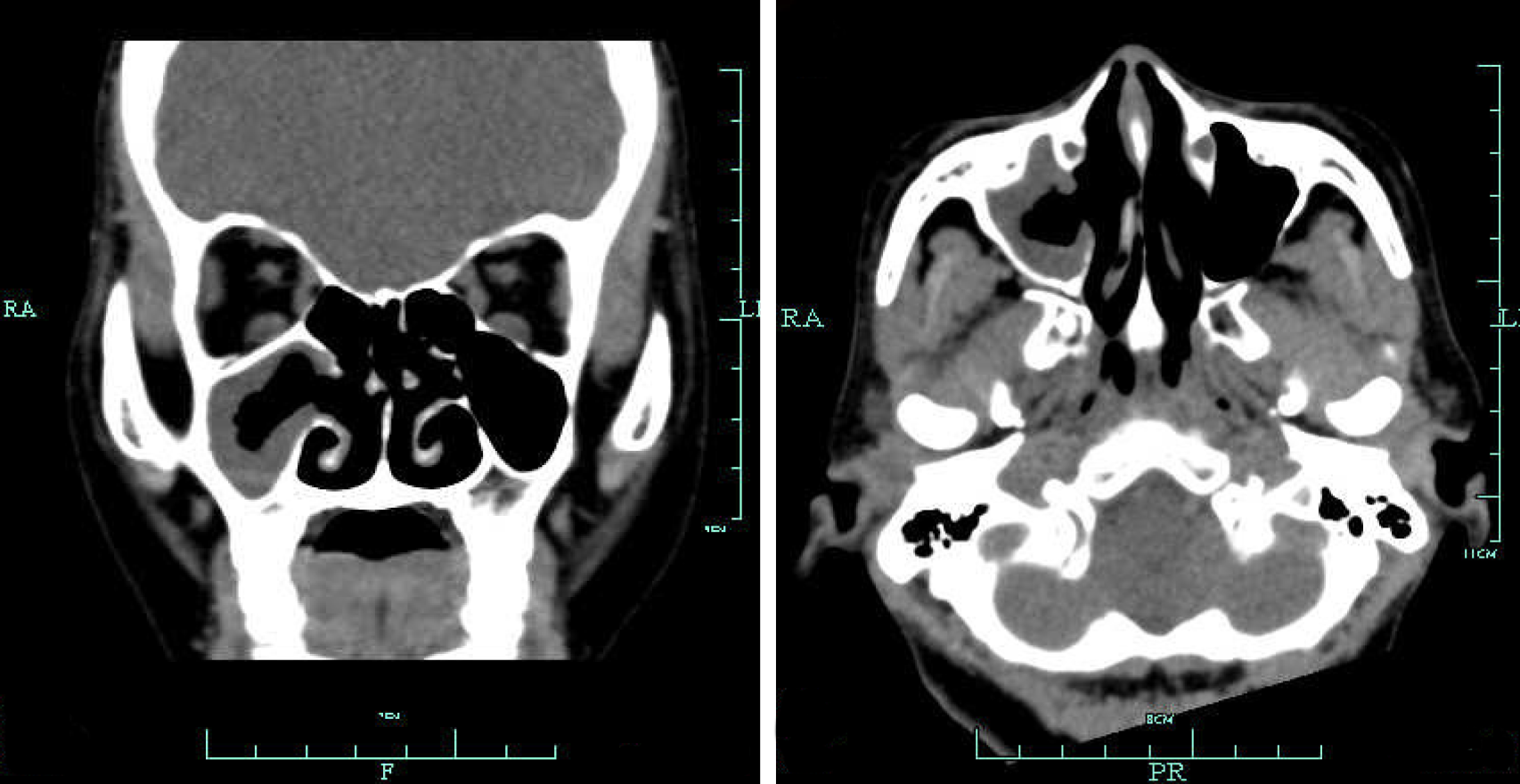
An endoscopic examination showed that the patient was recovering well at 3 mo after surgery. The ostium of the sinuses had opened well, and the mucin had disappeared. Sinus CT indicated a good recovery, with secretions in the right maxillary sinus after surgery (Figure 4). Moreover, after surgery the discomfort in the nasal cavity disappeared immediately and the patient felt that the dyspnea had also improved. A 25-month period of follow-up revealed no recurrence.
Fungi are almost ubiquitous and can be found in the nasal cavity and sinuses of healthy people. However, fungal infections of the airway are uncommon and may occur both in immunocompromised and immunocompetent individuals[12]. Fungal rhinosinusitis is the main type of fungal infection and inflammation of the upper airway, and can be divided into non-invasive and invasive forms[13]. The non-invasive types include localized fungal colonization, fungal ball, and AFRS[14]. AFRS is usually caused by Aspergillus spp., which is termed allergic aspergillus sinusitis. Fungal infections and inflammation of the lower airway are also mainly caused by Aspergillus spp., including ABPA, aspergilloma, chronic necrotizing pneumonia, and invasive pulmonary aspergillosis[15]. Other funguses can also lead to diseases such as ABPA, which is termed allergic bronchopulmonary mycosis (ABPM)[16]. ABPA belongs to ABPM. Recently, the morbidity of this allergic disease has increased perceptibly, in line with the increased prevalence of AFRS and ABPA. However, coexistence of AFRS and ABPA is still uncommon. Some authors have defined this type of disease as sinobronchial allergic mycosis[17]. We searched the PubMed database for reports of coexisting AFRS and ABPA for the period from 1970 to 2016 (keywords: allergic fungal rhinosinusitis, allergic bronchopulmonary aspergillosis; or allergic aspergillus sinusitis, allergic bronchopulmonary aspergillosis) and found 20 patients in 13 English articles (including the present case) that included clinical and imaging details (Tables 1 and 2)[18-28]. Until now, only one article recorded a case of coexisting AFRS and ABPM that did not originate with Aspergillus spp.[29]. Some articles mentioned the coexistence of AFRS and ABPA but did not include details (or the reports were written in other languages)[30-33].
| Ref. | Sex / Age | Region | Nasal symptoms/ duration | Pulmonary symptoms/ duration | Asthmas/ cystic fibrosis | Total IgE | Eosinophil count | Affected sinuses | Pulmonary imaging |
| Safirstein[20] | F/24 | United States | Rhinorrhea, blood-tinged casts | No obvious symptom | N/N | - | 12%, 1650/mm3 | - | Infiltration |
| Sher and Schwartz[19] | F/42 | United States | Symptoms of allergic rhinosinusitis /since childhood | Dyspnea, wheeze/2 yr | Y/N | 13084 ng/mL | 1355/mm3 | Left maxillary sinus | Bronchiectasis, interstitial scarring |
| Travis et al[26] | F/48 | United States | Nasal block | No obvious symptom | N/N | 794 IU/mL | 1350/mm3 | Bilateral frontal and ethmoid sinuses | Nodule |
| M/16 | United States | Nasal block/ 7 yr, right proptosis, and periorbital edema | Asthma-like symptoms/ 11 yr | Y/N | 785 IU/mL | Normal | Right maxillary sinus | Infiltration, bronchiolitis | |
| Bhagat et al[18] | F/38 | India | Rhinorrhea, nasal block, and discharge/ 5 yr | Pain, cough, wheezing, dyspnea/9 yr | Y/N | 280 IU/L | 12% | Bilateral maxillary sinuses | Pleural effusion, bronchiectasis, collapsed left lung |
| Shah et al[22] | M/55 | India | Rhinorrhea, nasal block, brownish plugs/12 yr | Dyspnea, wheeze, brownish plugs/2 yr | Y/N | - | 26% | Bilateral maxillary sinuses | Bronchiectasis, right lower zone opacity, transient infiltration |
| Shah et al[28] | M/24 | India | Rhinorrhea, sneeze, nasal block, postnasal discharge, plugs, headache/ 12 yr | Chest pain, dyspnea, wheeze, cough, plugs/6 yr | Y/N | 5600 IU/mL | 9% | Right maxillary sinus | Central bronchiectasis |
| M/45 | India | Rhinorrhea, sneeze, nasal block, postnasal discharge/ 1 yr | Cough, chest pain, dyspnea, haemoptysis/1 yr | Y/N | 680 IU/mL | 3% | Bilateral maxillary sinuses | Central bronchiectasis | |
| M/55 | India | Rhinorrhea, sneeze, nasal block, headache/ 12 yr | Chest pain, dyspnea, wheeze, cough, plugs/2 yr | Y/N | 540 IU/mL | 26% | Bilateral maxillary sinuses | Central bronchiectasis | |
| F/38 | India | Rhinorrhea, sneeze, nasal block, plugs/5 yr | Chest pain, dyspnea, wheeze, cough, plugs, haemoptysis/37 yr | Y/N | 280 IU/mL | 24% | Bilateral maxillary sinuses | Central bronchiectasis | |
| F/29 | India | Rhinorrhea, sneeze, nasal block, plugs, headache/ 5 yr | Dyspnea, wheeze, cough, plugs, haemoptysis/5 yr | Y/N | 590 IU/mL | 16% | Bilateral maxillary and frontal sinuses | Central bronchiectasis | |
| M/30 | India | Rhinorrhea, sneeze, nasal block, headache/ 1 yr | Chest pain, cough/1 yr | Y/N | 4560 IU/mL | 8% | Bilateral maxillary and frontal sinuses | Central bronchiectasis | |
| F/14 | India | Rhinorrhea, sneeze, nasal block/1.5 yr | Dyspnea, wheeze, cough, chest pain/1.5 yr | Y/N | 25216 IU/mL | 11% | Bilateral maxillary sinuses | Central bronchiectasis | |
| Shah et al[28] | M/26 | India | Rhinorrhea, nasal block, sneeze, postnasal discharge, brownish plugs/since childhood | Dyspnea, wheeze, cough, brownish plugs/since childhood | Y/N | 34132 IU/mL | 24%, | Left maxillary sinus | Bronchiectasis transient infiltration |
| Prasad et al[21] | M/52 | India | Rhinorrhea/ 25 yr | Hemoptysis/6 yr, | N/N | 5652 IU/mL | 12%, 1296/mm3 | Left maxillary, ethmoid sinuses | Bronchiectasis |
| Erwin and Fitzgerald[25] | M/57 | United States | Nasal block/since childhood | Tightness, wheeze, cough, brownish plugs/ since childhood | Y/N | 1987 IU/mL | 6.50% | Bilateral frontal and left sphenoid sinuses | Bronchiectasis with mucus plugging |
| Das et al[27] | M/9 | India | Brownish nasal discharge/ 6 yr | Chest pain, dyspnea, brownish sputum/5 yr | Y/N | 4838.5 IU/mL | 8% | Frontal and maxillary sinuses | - |
| Ghosh et al[24] | F/30 | India | Rhinorrhea, sneeze/6 yr | Dry cough/ 5 mo | N/N | 2500 ng/mL | 36% | Right maxillary sinus | Bilateral central bronchiectasis with mucus plugging |
| Upadhyay et al[23] | F/35 | India | Rhinorrhea, pain, tenderness/ 8 yr | Dyspnea, cough/8 yr | Y/N | 1000 IU/mL | 16%, 2240/ mm3 | Bilateral maxillary sinuses | Bilateral central cystic bronchiectasis |
| Current case | F/52 | China | Rhinorrhea, sneeze, nasal block, epistaxis, hyposmia/ 5 mo | Dyspnea, cough/20 yr | Y/N | > 5000 U/mL | 5.20% | Right maxillary and ethmoid sinuses | Bronchiectasis with mucus plugging |
| Ref. | Clinical stage of ABPA | Treatment | Outcome |
| Bhagat et al[18] | Acute | Oral glucocorticoid/2 wk | Improvement/1 mo |
| Sher and Schwartz[19] | Acute | Surgery for sinuses, oral glucocorticoid | Improvement |
| Safirstein[20] | Acute | Surgery for sinuses, oral glucocorticoid/6 mo | Improvement, recurrence after stopping oral glucocorticoid |
| Prasad et al[21] | Exacerbation | Surgery for sinuses, oral and topical glucocorticoid, montelukast sodium | Resolution |
| Shah et al[18] | Exacerbation | Oral glucocorticoid/2 wk | Resolution/1 mo |
| Shah and Panjabi[11] | Exacerbation | Oral glucocorticoid/tapered off | Resolution/3.5 yr |
| Upadhyay et al[23] | Exacerbation | Oral glucocorticoid, oral itraconazole/6 wk, then tapered off, omalizumab/6 wk | Improvement |
| Ghosh et al[24] | Remission | Oral glucocorticoid, oral itraconazole | Resolution |
| Erwin and Fitzgerald[25] | Corticosteroid dependent asthma | Surgery for sinuses, oral glucocorticoid/tapered off, topical glucocorticoid, oral itraconazole/2 mo, oral voriconazole/2 mo | Improvement/2 yr |
| Travis et al[26] | Remission | Surgery for sinuses, oral ketoconazole, surgery for lung | Improvement |
| Remission | Surgery for sinuses, lung biopsy | Improvement | |
| Das et al[27] | Exacerbation | Oral itraconazol/4 mo, oral glucocorticoid/tapered off 8 mo | Improvement |
| Shah et al[28] | - | Polypectomy, septoplasty, oral glucocorticoid/ tapered off 2 yr, topical glucocorticoid | Improvement |
| - | Oral glucocorticoid/tapered off 6 mo, topical glucocorticoid | Improvement | |
| - | Septoplasty, oral glucocorticoid/irregular, topical glucocorticoid | Improvement | |
| - | Polypectomy, oral glucocorticoid/tapered off 6 mo, topical glucocorticoid | Improvement | |
| Polypectomy, oral glucocorticoid/1-3 yr, topical glucocorticoid | Improvement | ||
| - | Polypectomy, oral glucocorticoid/1-3 yr, topical glucocorticoid | Improvement | |
| - | Oral glucocorticoid/1-3 yr, topical glucocorticoid | Improvement | |
| Current case | Exacerbation | Surgery for sinuses, oral glucocorticoid, oral itraconazol/4 wk, montelukast, topical glucocorticoid | AFRS/resolution, ABPA/ improvement |
In these studies, fungal disease usually had a regional susceptibility. Ferguson et al[34] found an increased prevalence of AFRS in the southeastern United States, where mold counts are notably high. Other studies suggested that AFRS was associated with lower income, rural counties, poor housing quality, and reduced access to healthcare[35,36]. In this review, 14 patients lived in India, 5 in the United States, and 1 in China. We inferred that the high prevalence of coexisting AFRS and ABPA might be due to the high temperature and humid climate in India. In this review, 10 female and 10 male patients had coexisting AFRS and ABPM, with no gender difference. The patients ranged in age from 9 to 57 years, with a mean age of 36.1 years, implying that this disease is very rare in older people.
The most common nasal symptom in our review was rhinorrhea, followed by nasal block, sneezing, and postnasal discharge; thus there was no difference in symptoms compared to the more common disease of chronic rhinosinusitis. The most common pulmonary symptom was dyspnea, followed by wheeze, cough, and chest pain. The duration of nasal symptoms ranged from 5 mo to 57 years, as did the duration of pulmonary symptoms. In this review, seven patients initially presented with nasal symptoms, four first showed pulmonary manifestations, seven showed concurrent nasal and pulmonary symptoms, and the symptoms were unknown in two cases. The upper airway is closely related to the lower airway. Based on the concept of “one airway one disease” in fungal allergy, one fungal antigen may therefore cause both AFRS and ABPA[37]. AFRS may lead to ABPA, and vice versa. Because of the lack of a convenient system for identifying funguses, diagnosis of coexisting AFRS and ABPA is usually difficult. Since different specialists treat ABPA and AFRS, their coexistence may be overlooked[38]. AFRS is a relatively common disease and with increasing focus on multi-disciplinary care, there should be more simultaneous cases. Moreover, the use of oral glucocorticoids and/or antifungal agents in either disorder may mask the manifestation of the other disorder[38]. We suggest that patients with AFRS should undergo pulmonary CT if they exhibit pulmonary symptoms. Conversely, patients with ABPA should undergo sinus CT if they show nasal symptoms. If this disease is recognized and treated appropriately, the inflammation process can be inhibited and irreversible lung destruction can be avoided.
ABPA usually accompanies asthma and cystic fibrosis (CF). Although rare, ABPA without asthma or CF can still occur[39]. In our review, 16 (80%) patients had asthma, but none had CF. This result implies that the coexistence of AFRS and ABPA might have nothing to do with CF. The CT findings of AFRS often demonstrate unilateral involvement of the sinuses[40], with the ethmoid sinus being the most commonly involved sinus[41]. In this review, the disease affected the bilateral sinuses in 11 patients. The most commonly affected sinus was the maxillary sinus, followed by the ethmoid, frontal, and sphenoid sinuses. The main manifestation of sinus CT was opacification, with two cases of destruction of bone.
All of the patients in this review demonstrated elevated total IgE and eosinophil counts in peripheral blood tests, which agrees with previous research[42,43]. The main manifestation of pulmonary CT was bronchiectasis, in agreement with another article[44]. According to conventional staging, APBA can be divided into five stages: Acute, remission, exacerbation, corticosteroid-dependent asthma, and fibrotic lung disease[45]. In this review, six patients belonged to the exacerbation stage, three to the acute stage, three to the remission stage, and one to the corticosteroid-dependent asthma stage; the stage was unknown in seven cases.
AFRS usually requires surgical therapy combined with medical control[40]. Surgery can remove the inciting fungal allergic mucin and enlarge the ostium of the involved sinuses. Medical therapy comprises corticosteroids, immunotherapy, and antifungal medications. However, there is limited evidence to support the use of topical or oral antifungal agents in patients with AFRS[46]. Oral corticosteroids and antifungal medications are the main treatments for ABPA[47]. In this review, 12 patients underwent surgery on the sinuses, all patients received oral glucocorticoids for at least 2 wk, and 11 patients received topical glucocorticoids. Seven patients received oral antifungal medications, and no patient was prescribed topical antifungal agents. All patients achieved a good prognosis, with 19 with an improved or resolved status and one case of recurrence after oral glucocorticoids were stopped.
In our opinion, surgery should be performed for sinusitis in patients with coexisting AFRS and ABPA to clear the fungal allergens and reduce the stimulation of allergic inflammation. Oral glucocorticoid and antifungal medications should be applied for a short time and then tapered off, due to the obvious side effects of these medicines. Topical glucocorticoids for the nasal cavity should be applied for longer periods. Until now, there has been no evidence of use of topical antifungal medications and immunotherapy to treat this disease.
Based on the concept of “one airway one disease” in fungal allergy, one fungal antigen may therefore cause both AFRS and ABPA. Since different specialists treat ABPA and AFRS, their coexistence may be overlooked. The patients with AFRS should undergo pulmonary CT if they exhibit pulmonary symptoms. Conversely, patients with ABPA should undergo sinus CT if they show nasal symptoms. AFRS accompanied by ABPA usually requires surgical therapy combined with medical control to improve the symptoms.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, research and experimental
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: El-Shazly A S-Editor: Wang J L-Editor: Wang TQ E-Editor: Wu YXJ
| 1. | Katzenstein AL, Sale SR, Greenberger PA. Allergic Aspergillus sinusitis: a newly recognized form of sinusitis. J Allergy Clin Immunol. 1983;72:89-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 371] [Cited by in RCA: 328] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 2. | Allphin AL, Strauss M, Abdul-Karim FW. Allergic fungal sinusitis: problems in diagnosis and treatment. Laryngoscope. 1991;101:815-820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 95] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 3. | Collins MM, Nair SB, Wormald PJ. Prevalence of noninvasive fungal sinusitis in South Australia. Am J Rhinol. 2003;17:127-132. [PubMed] |
| 4. | Bent JP, Kuhn FA. Diagnosis of allergic fungal sinusitis. Otolaryngol Head Neck Surg. 1994;580-588. |
| 5. | Doellman MS, Dion GR, Weitzel EK, Reyes EG. Immunotherapy in allergic fungal sinusitis: The controversy continues. A recent review of literature. Allergy Rhinol (Providence). 2013;4:e32-e35. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 6. | Shah A, Panjabi C. Allergic bronchopulmonary aspergillosis: a review of a disease with a worldwide distribution. J Asthma. 2002;39:273-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 30] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 7. | Hinson KF, Moon AJ, Plummer NS. Broncho-pulmonary aspergillosis; a review and a report of eight new cases. Thorax. 1952;7:317-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 519] [Cited by in RCA: 495] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 8. | Rosenberg M, Patterson R, Mintzer R, Cooper BJ, Roberts M, Harris KE. Clinical and immunologic criteria for the diagnosis of allergic bronchopulmonary aspergillosis. Ann Intern Med. 1977;86:405-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 483] [Cited by in RCA: 468] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 9. | Shah A. Allergic bronchopulmonary aspergillosis: an Indian perspective. Curr Opin Pulm Med. 2007;13:72-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 10. | Mohindra S, Mehta R, Bal A. ABPA Concomitantly Occurring with Invasive Sinus Aspergillosis: A Short Report on Two Patients. Indian J Otolaryngol Head Neck Surg. 2014;66:329-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 11. | Shah A, Panjabi C. Contemporaneous occurrence of allergic bronchopulmonary aspergillosis, allergic Aspergillus sinusitis, and aspergilloma. Ann Allergy Asthma Immunol. 2006;96:874-878. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 12. | Kini JR, Shetty SS, Kini H. Spectrum of paranasal sinus mycoses in coastal India. Ear Nose Throat J. 2012;91:E15-E18. [PubMed] |
| 13. | Callejas CA, Douglas RG. Fungal rhinosinusitis: what every allergist should know. Clin Exp Allergy. 2013;43:835-849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 14. | Ragab A, Samaka RM, Salem M. Impact of fungal load on diagnosis and outcome of allergic fungal rhinosinusitis. Eur Arch Otorhinolaryngol. 2014;271:93-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 15. | Thompson GR, Patterson TF. Pulmonary aspergillosis. Semin Respir Crit Care Med. 2008;29:103-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 16. | Fukutomi Y, Tanimoto H, Yasueda H, Taniguchi M. Serological diagnosis of allergic bronchopulmonary mycosis: Progress and challenges. Allergol Int. 2016;65:30-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 37] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 17. | Venarske DL, deShazo RD. Sinobronchial allergic mycosis: the SAM syndrome. Chest. 2002;121:1670-1676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 54] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 18. | Bhagat R, Shah A, Jaggi OP, Khan ZU. Concomitant allergic bronchopulmonary aspergillosis and allergic Aspergillus sinusitis with an operated aspergilloma. J Allergy Clin Immunol. 1993;91:1094-1096. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 19. | Sher TH, Schwartz HJ. Allergic Aspergillus sinusitis with concurrent allergic bronchopulmonary Aspergillus: report of a case. J Allergy Clin Immunol. 1988;81:844-846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 44] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 20. | Safirstein BH. Allergic bronchopulmonary aspergillosis with obstruction of the upper respiratory tract. Chest. 1976;70:788-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 100] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 21. | Prasad R, Garg R, Sanjay, Dua R. Concomitant allergic bronchopulmonary aspergillosis and allergic fungal sinusitis. Indian J Med Sci. 2007;61:532-534. [PubMed] |
| 22. | Shah A, Bhagat R, Panchal N, Jaggi OP, Khan ZU. Allergic bronchopulmonary aspergillosis with middle lobe syndrome and allergic Aspergillus sinusitis. Eur Respir J. 1993;6:917-918. [PubMed] |
| 23. | Upadhyay R, Kant S, Prakash V, Saheer S. Sinobronchial allergic aspergillosis with allergic bronchopulmonary aspergillosis: a less common co-existence. BMJ Case Rep. 2014;2014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 24. | Ghosh G, Sharma B, Chauhan A, Chawla MP. Coexistence of allergic bronchopulmonary aspergillosis and allergic aspergillus sinusitis in a patient without clinical asthma. BMJ Case Rep. 2013;2013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 25. | Erwin GE, Fitzgerald JE. Case report: allergic bronchopulmonary aspergillosis and allergic fungal sinusitis successfully treated with voriconazole. J Asthma. 2007;44:891-895. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 29] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 26. | Travis WD, Kwon-Chung KJ, Kleiner DE, Geber A, Lawson W, Pass HI, Henderson D. Unusual aspects of allergic bronchopulmonary fungal disease: report of two cases due to Curvularia organisms associated with allergic fungal sinusitis. Hum Pathol. 1991;22:1240-1248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 57] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 27. | Das P, Arya R, Shrivastava SK. Allergic bronchopulmonary aspergillosis with Aspergillus sinusitis-'9' year old boy. Lung India. 2008;25:158-159. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 28. | Shah A, Panchal N, Agarwal AK. Concomitant allergic bronchopulmonary aspergillosis and allergic Aspergillus sinusitis: a review of an uncommon association*. Clin Exp Allergy. 2001;31:1896-1905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 58] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 29. | Ogawa H, Fujimura M, Takeuchi Y, Makimura K. A case of sinobronchial allergic mycosis; possibility of basidiomycetous fungi as a causative antigen. Intern Med. 2011;50:59-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 30. | Schwartz HJ. Allergic fungal sinusitis: experience in an ambulatory allergy practice. Ann Allergy Asthma Immunol. 1996;77:500-502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 31. | Braun JJ, Pauli G, de Blay F. [Allergic fungal sinusitis and allergic broncho-pulmonary aspergillosis: a fortuitous association or a nosological rhino-bronchial entity?]. Rev Mal Respir. 2005;22:405-411. [PubMed] |
| 32. | Schubert MS, Goetz DW. Evaluation and treatment of allergic fungal sinusitis. I. Demographics and diagnosis. J Allergy Clin Immunol. 1998;102:387-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 122] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 33. | Braun JJ, Pauli G, Schultz P, Gentine A, Ebbo D, de Blay F. Allergic fungal sinusitis associated with allergic bronchopulmonary aspergillosis: an uncommon sinobronchial allergic mycosis. Am J Rhinol. 2007;21:412-416. [PubMed] |
| 34. | Ferguson BJ, Barnes L, Bernstein JM, Brown D, Clark CE, Cook PR, DeWitt WS, Graham SM, Gordon B, Javer AR, Krouse JH, Kuhn FA, Levine HL, Manning SC, Marple BF, Morgan AH, Osguthorpe JD, Skedros D, Rains BM, Ramadan HH, Terrell JE, Yonkers AJ. Geographic variation in allergic fungal rhinosinusitis. Otolaryngol Clin North Am. 2000;33:441-449. [PubMed] |
| 35. | Miller JD, Deal AM, McKinney KA, McClurg SW, Rodriguez KD, Thorp BD, Senior BA, Zanation AM, Ebert CS. Markers of disease severity and socioeconomic factors in allergic fungal rhinosinusitis. Int Forum Allergy Rhinol. 2014;4:272-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 36. | Lu-Myers Y, Deal AM, Miller JD, Thorp BD, Sreenath SB, McClurg SM, Senior BA, Zanation AM, Ebert CS. Comparison of Socioeconomic and Demographic Factors in Patients with Chronic Rhinosinusitis and Allergic Fungal Rhinosinusitis. Otolaryngol Head Neck Surg. 2015;153:137-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 38] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 37. | Ogawa H, Fujimura M, Ohkura N, Makimura K. A proposal of guidance for identification of Schizophyllum commune-associated sinobronchial allergic mycosis. Allergol Int. 2014;63:287-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 38. | Agarwal R, Bansal S, Chakrabarti A. Are allergic fungal rhinosinusitis and allergic bronchopulmonary aspergillosis lifelong conditions? Med Mycol. 2017;55:87-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 39. | Kim Y, Lee HY, Gu KM, Lee JY, Yoon SW, Park TY, Choi JC, Kim JY, Park IW, Shin JW, Choi BW, Jung JW. Delayed diagnosis of allergic bronchopulmonary aspergillosis due to absence of asthmatic symptoms. Asia Pac Allergy. 2016;6:187-191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 40. | Marple BF. Allergic fungal rhinosinusitis: current theories and management strategies. Laryngoscope. 2001;111:1006-1019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 154] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 41. | Glass D, Amedee RG. Allergic fungal rhinosinusitis: a review. Ochsner J. 2011;11:271-275. [PubMed] |
| 42. | Singh B, Denning DW. Allergic bronchopulmonary mycosis due to Alternaria: Case report and review. Med Mycol Case Rep. 2012;1:20-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 43. | Chowdhary A, Agarwal K, Kathuria S, Gaur SN, Randhawa HS, Meis JF. Allergic bronchopulmonary mycosis due to fungi other than Aspergillus: a global overview. Crit Rev Microbiol. 2014;40:30-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 146] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 44. | Knutsen AP. Allergic bronchopulmonary aspergillosis. Clin Exp Allergy. 2015;45:298-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 45. | Patterson R, Greenberger PA, Radin RC, Roberts M. Allergic bronchopulmonary aspergillosis: staging as an aid to management. Ann Intern Med. 1982;96:286-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 171] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 46. | Mistry SG, Kumar BN. The value of antifungal therapy in allergic fungal rhinosinusitis. Rhinology. 2014;52:9-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 47. | Elphick HE, Southern KW. Antifungal therapies for allergic bronchopulmonary aspergillosis in people with cystic fibrosis. Cochrane Database Syst Rev. 2016;11:CD002204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 14] [Article Influence: 1.6] [Reference Citation Analysis (0)] |