Published online Jun 26, 2019. doi: 10.12998/wjcc.v7.i12.1475
Peer-review started: November 21, 2018
First decision: March 10, 2019
Revised: May 1, 2019
Accepted: May 11, 2019
Article in press: May 11, 2019
Published online: June 26, 2019
Processing time: 217 Days and 17.9 Hours
The most common lysosomal storage disorder is Gaucher disease (GD). It is a deficiency of lysosomal glucocerebrosidase (GBA) due to biallelic mutations in the GBA gene, characterized by the deposition of glucocerebroside in macrophage-monocyte system cells. The report targets clinical phenotypes of GD in order to correlate them with GBA gene mutations, as well as to identify GBA gene mutation in patients in Montenegro that are diagnosed with GD.
Five patients (4 male, 1 female) of type 1 GD (GD1) are reported. The age at diagnosis ranged from 7 to 40. Patients experienced delays of 1-12 years in diagnosis after the original onset of symptoms. The most common mode of presentation was a variable degree of splenomegaly and thrombocytopenia, while other symptoms included bone pain, hepatomegaly, abdominal pain and fatigue. Osteopenia was present in a majority of the patients: 4/5. All patients were found to have an asymptomatic Erlenmeyer flask deformity of the distal femur. On enzyme replacement therapy (ERT), the hematological and visceral parameters showed significant improvement, but no significant progression in bone mineral density was noticed. GBA gene sequencing revealed homozygosity for the N370S mutation in one patient. The genotypes of the other patients were N370S/55bp deletion, N370S/D409H (2 patients), and H255Q/N370S (1 patient).
The phenotypes of the GD1 encountered in Montenegro were severe but all responded well to ERT.
Core tip: This is the first report on Gaucher disease (GD) originating from Montenegro that presents clinical phenotypes of GD and glucocerebrosidase (GBA) gene mutations in patients in Montenegro that are diagnosed with GD and genotype/phenotype correlations. While GBA gene sequencing revealed a homozygosity for the N370S mutation in 1 patient, the genotypes of the other patients were N370S/55bp deletion, N370S/D409H (in 2 patients), and H255Q/N370S (1 patient). The phenotypes of the GD type 1 encountered were severe but all responded well to enzyme replacement therapy. Genetic testing for their progeny was also planned.
- Citation: Vujosevic S, Medenica S, Vujicic V, Dapcevic M, Bakic N, Yang R, Liu J, Mistry PK. Gaucher disease in Montenegro - genotype/phenotype correlations: Five cases report. World J Clin Cases 2019; 7(12): 1475-1482
- URL: https://www.wjgnet.com/2307-8960/full/v7/i12/1475.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v7.i12.1475
The most common lysosomal storage is Gaucher disease (GD), occurring in 1 in every 850 live births for Ashkenazi Jews[1] and approximately from 1 in every 57000 to 1 in 75000 worldwide[2]. The most prominent characteristic of the disease is the accumula-tion of glucosphingolipids glucosylceramide and glucosylsphingosine within the lysosomes of mononuclear phagocytes[3]. The defect itself is a deficiency of glucocere-brosidase (GBA) due to biallelic mutations in the GBA gene[4], where significant clinical heterogeneity differentiates into three clinical subtypes that give rise to the appearance of the characteristic Gaucher cells[5]: Type 1 (GD1)-nonneuronopathic GD, type 2-acute neuronopathic GD, and type 3-chronic neuronopathic GD. Type 1 disease is commonly presented in patients by splenomegaly, anemia, and thrombocytopenia accompanied by potential subsequent bleeding; in addition, patients may also present hepatomegaly, bone pain or pathologic fractures[6]. Type 2 GD is rare, but may be present at birth or during infancy. It is characterized by a rapid neurodegenerative course with extensive visceral involvement, resulting in death within the first 2 years of life[7,8]. Type 3 GD is characterized by the slower progression of neurologic symp-toms when compared to the acute type 2 GD, presenting with splenomegaly and/or hepatomegaly, seizures, skeletal malformations, as well as eye movement disorders, anemia, and respiratory problems. Patients suffering from type 3 GD often live into their early teens and adulthood[9]. Diagnosis can be confirmed by measuring GBA activity in peripheral blood leukocytes, whereby less than 15% of mean normal activity tests positive. Confirming the GBA gene mutation is also another means of diagnosis.
The aim of this study is to report clinical phenotypes of GD to correlate them with GBA gene mutations as well as to identify the GBA gene mutation in 5 patients (4 males and 1 female) in Montenegro that were diagnosed with GD. While an additional female patient was also diagnosed in Montenegro, the patient was lost to further follow-ups and is therefore not presented here.
Patient 1: Fatigue, left hip joint pain, poor quality of life.
Patient 2: Abdominal and lumbosacral pain, as well as loss of appetite.
Patient 3: Nasal bleeding, pain under the right costal arch.
Patient 4: Abdominal pain.
Patient 5: Abdominal pain, massive genital bleeding.
Patient 1: Patients symptoms were recognized 6 mo before admission with gradual worsening.
Patient 2: Patients symptoms were recognized 2 mo before admission with gradual worsening.
Patient 3: Patients symptoms were recognized 4 d before admission with gradual worsening.
Patient 4: Patients symptoms were recognized in early childhood; therapy was administrated at the age of 17.
Patient 5: Patients symptoms were recognized 3 mo before admission with gradual worsening.
Patient 1: Splenectomy was done at the age of 17 due to hypersplenism, previous chronic gastritis, gastroesophageal reflux disease, chronic hepatitis B.
Patient 2: Hypertension, pneumothorax in two occasions 1996 and 2003.
Patient 3: The patient experienced a spontaneous hemothorax on his right side at the age of 41 and 48. His pulmonary diffusion capacity for carbon monoxide was also low. Echocardiography showed a dilated left chamber. The patient was diagnosed with Parkinson’s disease at the age of 55.
Patient 4: No past illness of disease.
Patient 5: Hepatosplenomegaly was detected in childhood at the age of 11, further diagnostics were not carried out in the interim.
Patient 1: No family history of disease.
Patient 2: No family history of disease.
Patient 3: His brother had been previously diagnosed with GD.
Patient 4: No family history of disease.
Patient 5: Suture of a perineal rupture had been done following excessive bleeding after delivery. Two months subsequent to this incident, while undergoing a recons-tructive operation of the cloaca, massive bleeding also occurred. No family history of disease.
Patient 1: Normal vital signs, abdomen slightly soar to touch, hepatomegaly.
Patient 2: By physical examination a systolic murmur at apex was found, abdominal pain, hepatosplenomegaly.
Patient 3: Abdominal pain, hepatosplenomegaly.
Patient 4: Under routine examination, abdominal pain and splenomegaly.
Patient 5: Normal vital sings, abdominal pain on examination.
Patient 1: Thrombocytopenia (84 × 109/L), Gaucher cells findings in the sternal bone marrow aspirate. Low beta-glucosidase activity (0.74 U/mL) in the leucocytes, accompanied by markedly elevated plasma-chitotriosidase activity (8685 nmol/h/mL) and a confirmation of the mutation N370S/55bp deletion.
Patient 2: Elevated serum transaminases (AST 135, ALT 154, GGT 261) and thrombocytopenia were noted (96 × 109/L). A cholecystectomy was conducted, normalizing the transaminase level, but the thrombocytopenia remained. Finding of Gaucher cells from a bone marrow biopsy. Further low beta- glucosidase activity (0.58 U/mL) in the leucocytes and markedly elevated plasma-chitotriosidase activity (7752 nmol/h/mL). GBA gene sequencing revealed the genotype N370S/D409H.
Patient 3: Thrombocytopenia (79 × 109/L), Gaucher cells were found in bone marrow and liver biopsy. Low leucocyte-beta glucosidase activity (1.43 nmol/mg prot/h) and markedly elevated plasma-chitotriosidase activity (5397.5 nmol/h/mL). Subsequent GBA gene sequencing revealed the genotype N370S/D409H.
Patient 4: Slightly reduced platelet count (136 × 109/L), Gaucher cell in bone marrow biopsy, low leucocyte-beta glucosidase activity (0.32 U/mL) and markedly elevated plasma-chitotriosidase activity (28657 nmol/h/mL). The diagnosis was further confirmed by GBA gene sequencing for the genotype H255Q/N370S.
Patient 5: The laboratory analysis indicated a low platelet count (102 × 109/L), low leucocyte (3.1 × 109/L) beta-glucosidase (0.63 U/mL) activity and markedly elevated plasma-chitotriosidase activity (25578 nmol/h/mL), HBs, HCV, HIV negative. Gaucher cells in bone marrow biopsy. The GBA gene sequencing established homozy-gosity for the N370S mutation.
Patient 1: Abdominal ultrasound liver (15 cm × 13 cm × 19 cm), magnetic resonance imaging (MRI) of the femurs and lumbosacral spine visualized an Erlenmeye flask deformity of both the distal femurs, bone marrow infiltration of both the femurs and diffuse bone marrow infiltration of the lumbal spine.
Patient 2: Abdominal-multislice computed tomography (MSCT) showed an enlarged liver (18 cm × 16 cm × 21cm) and spleen (23.5 cm × 8.5 cm × 8cm). Nuclear magnetic resonance (NMR) of LS spine and pelvis showed no pathological finding. An endocranial MR showed no pathological finding.
Patient 3: Hepatosplenomegaly (spleen 167 mm), MRI of the lumbosacral spine showed a hypodense zone as a sign of infiltration in the trochanter region on both sides. NMR of head normal finding.
Patient 4: Splenomegaly (ultrasound 19 cm), an MRI of the patient’s bones indicated osteolytic leasions of both the femurs and the tibiae. No reduction in bone mineral density has since been found present by osteodensitometry.
Patient 5: Abdominal ultrasound liver 20 cm, spleen > 22 cm, pelvic CT scan no abnormalities.
GD 1.
Enzyme replacement therapy (ERT) by imiglucerase initiating when the patient was 19 years old (30 IU/kg once, bi-weekly).
ERT by imiglucerase was recommended, but was only initiated after three years due to financial difficulties.
Eliglustat treatment was started at 55 years of age. However, owing to financial difficulties, was discontinued. At the age of 57, ERT by imiglucerase was initiated.
ERT with imiglucerase was started at the age of 17. The current dose is 40 IU/kg (once, bi-weekly).
ERT with imiglucerase was initiated four months after the diagnosis. The current dose is 40 IU/kg (once, bi-weekly).
Treatment was noncompliant. Owing to financial difficulties, ERT was discontinued at the age of 38.
The current dose administered is 40 IU/kg (once, bi-weekly).
The current dose is 40 IU/kg (once, bi-weekly).
His spleen diameter and hepatogram have since normalized in the treatment.
Spleen and liver diameters as well as her hepatogram have since normalized.
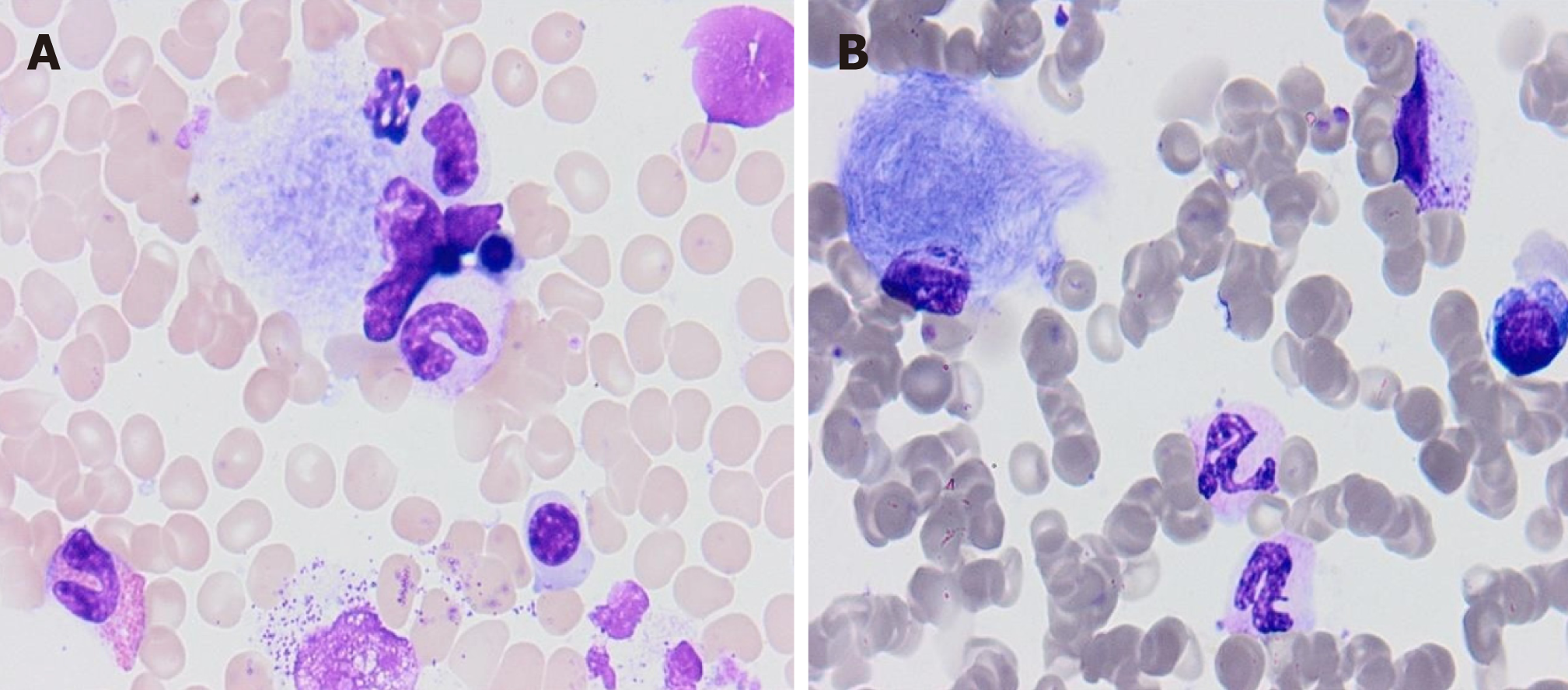
Concerning the cases covered in this report, whereas type 2 and 3 affect only 5% of patients, GD1 is the most common GD type[10]. It has been estimated that 66% of GD1 patients are diagnosed before the age of 20[11], but the age at diagnosis for these Montenegrin patients ranges from 7 to 40. Diagnosis thereof has been confirmed based on low leucocyte-acid beta-glucosidase activity and GBA gene mutation. The GBA gene is located on chromosome 1q21. Containing 11 exons and 10 introns and covering 7.6 kilobases of sequence, a highly homologous pseudogene is located 16 kb downstream where nearly 300 mutations and polymorphisms in GBA have been identified[12]. Mutations in saposin C, the β-GBA activator gene, have been reported in cases of GD[13] (Table 1.) The most distinct hallmark is the presence of Gaucher cells (Figure 1) in the macrophage monocyte system[14], in bone marrow or in liver biopsy samples. Patients generally experience delays of one to twelve years in diagnosis after the first onset of symptoms. The most common mode of presentation here is the variable degree of splenomegaly and thrombocytopenia, thought her symptoms include bone pain, hepatomegaly, abdominal pain and fatigue. Hematologic manife-stations of GD include anemia, thrombocytopenia and less frequent leucopenia[15]. One patient suffered from profuse bleeding due to thrombocytopenia. Hypersplenism, accompanied by an increased risk of infection, rupture and infracts are some of the possible splenomegaly complications[7]. Osteopenia is present in a majority of the patients under review here: 4/5. All patients show an asymptomatic Erlenmeyer-flask deformity of the distal femur. Unusual manifestations in GD1 noted are malignancy, Parkinsonism and pulmonary hypertension[7,16]. Parkinsonism in GD1 is believed to arise from synuclein aggregation within dopaminergic neurons that are induced either by the gain of function mutations in GBA1 that lead to protein misfolding (N370S is such a mutation) or the accumulation of lipids[16]. Recent publications have established a link between GD and impaired host- defense against microbial infections, up-regulation of T-helper (Th)1 and Th2 cytokines, the dysfunction of monocytes, as well as an increased risk for lymphoid malignancies (most strikingly, for multiplemyeloma)[17].
| Patient | Age at diagnosis | Symptoms and signs | Lab findings | Organomegaly; Bone disorder | Genetics | Children |
| Patient 1 Gender: M | 8 | Stunted growth; distended stomch; bone pain | Thrombocytopenia; Gaucher cells in BM; plasma: Chitotriosidase ↑; β- glucosidase ↓ | Splenomegaly; Erlenmeyer flask deformity of both femurs | N370S/55bp | No |
| Patient 2 Gender: M | 40 | Loss of appetite; Abdominal and lumbosacral pain | Thrombocytopenia; Gaucher cells in BM; plasma: Chitotriosidase ↑; β- glucosidase ↓ | Splenomegaly, hepatomegaly; No bone disorder | N370S/D409H | 2 |
| Patient 3 Gender: M | 36 | Nasal bleeding; pain under right costal arch | Thrombocytopenia; Gaucher cells in BM; plasma: Chitotriosidase ↑; β- glucosidase ↓ | Splenomegaly, hepatomegaly; Infiltration in the trochanter region on both sides and LS spine | N370S/D409H | 2 |
| Patient 4 Gender: M | 7 | None | Thrombocytopenia; Gaucher cells in BM; plasma: Chitotriosidase ↑; β- glucosidase ↓ | Splenomegaly; Both femurs and tibias were affected | H255Q/N370S | No |
| Patient 5 Gender: F | 23 | Massive bleeding after childbirth | Thrombocytopenia; Gaucher cells in BM; plasma: Chitotriosidase ↑; β- glucosidase ↓ | Splenomegaly, hepatomegaly; No bone disorder | Homozygosity N370S | 1 |
In addition to the reduced acid beta-glucosidase activity and genotyping at the GBA gene locus that may yield additional prognostic information, elevated plasma-chitotriosidase activity can be found in GD[18]. Several markers are used in therapeutic monitoring: chitotriosidase, ferritin, ACE and acid phosphatase, but no prognostic marker can predict long-term complications of GD. All these markers also increase with disease progression as well as decrease under ERT[19]. The prognosis for type 1 or type3 GD patients receiving ERT is good, where in normal life expectancy is common. One study has estimated life expectancy at birth type 1 GD individuals to be 68 years, compared to 77 years in the general population[20]. Type-2 GD patients usually die within the first years of life. Patients who have a splenectomy are at a higher mortality risk[21], which may worsen skeletal and lung manifestations[20]. In a clinical sense, GBA1 mutations might not prove to be a reliable prognostic indicator in Parkinson’s disease[22]. ERT is indicated for type 1 GD patients who also have anemia, thrombocy-topenia, skeletal disease or visceromegaly[12,23]. An alternative oral approach is substrate synthesis inhibition therapy, based on inhibiting glucosylceramide synthesis[12]. When undergoing ERT, the hematological and visceral parameters indicate marked improvement; no significant progression in bone mineral density was found in these cases. The delay in initiating treatment for these cases under review stemmed from financial reasons not in the treatment itself.
In patients who are Ashkenazi Jews, the mutations N370S, 84GG, L444P and IVS2+1G account for over 90% of disease alleles[24]. The two mutations of N370S and L444P are common in Jewish and non-Jewish patients alike, but the latter exhibits a much wider range of genotype, in which homozygosis for L444P results in neurono-pathic disease. The presence of a single mutant N370S allele, however, usually prevents neurological involvement[12]. The most prevalent disease genotype worldwide across many ethnicities is L444P. In those of European descent, it is the N370S/L444P mutation[16], which is often characterized by mild cytopenia and splenomegaly. Non-Jewish GD individuals are mostly compound heterozygotes. Patients who are homozygous for the N370S variant suffer from a milder disease than those who are compound heterozygous. GBA gene sequencing revealed homozygosity for the N370S mutation in 1 patient, while the genotypes of other patients were N370S/55bp deletion, N370S/D409H (in 2 patients) and H255Q/N370S (1 patient). Genetic testing for their progeny has also been planned.
This is the first report of GD from Montenegro. N370S was the most common mutation, occurring in all five patients. One patient was found to be homozygous while others were heterozygous. The phenotypes of GD type 1 encountered in Montenegro were severe; notwithstanding, they all responded well to ERT.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine, Research and Experimental
Country of origin: Montenegro
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): D, D
Grade E (Poor): 0
P-Reviewer: Ma K, Piccaluga PP, Ali I, Gheita TA, Tanabe S S-Editor: Dou Y L-Editor: A E-Editor: Wang J
| 1. | Mistry PK, Liu J, Sun L, Chuang WL, Yuen T, Yang R, Lu P, Zhang K, Li J, Keutzer J, Stachnik A, Mennone A, Boyer JL, Jain D, Brady RO, New MI, Zaidi M. Glucocerebrosidase 2 gene deletion rescues type 1 Gaucher disease. Proc Natl Acad Sci U S A. 2014;111:4934-4939. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 88] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 2. | Meikle PJ, Hopwood JJ, Clague AE, Carey WF. Prevalence of lysosomal storage disorders. JAMA. 1999;281:249-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1518] [Cited by in RCA: 1504] [Article Influence: 57.8] [Reference Citation Analysis (0)] |
| 3. | Mistry PK, Liu J, Yang M, Nottoli T, McGrath J, Jain D, Zhang K, Keutzer J, Chuang WL, Mehal WZ, Zhao H, Lin A, Mane S, Liu X, Peng YZ, Li JH, Agrawal M, Zhu LL, Blair HC, Robinson LJ, Iqbal J, Sun L, Zaidi M. Glucocerebrosidase gene-deficient mouse recapitulates Gaucher disease displaying cellular and molecular dysregulation beyond the macrophage. Proc Natl Acad Sci U S A. 2010;107:19473-19478. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 190] [Cited by in RCA: 187] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 4. | Grabowski GA. Gaucher disease and other storage disorders. Hematology Am Soc Hematol Educ Program. 2012;2012:13-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 40] [Reference Citation Analysis (0)] |
| 5. | Sidransky E. Gaucher disease: insights from a rare Mendelian disorder. Discov Med. 2012;14:273-281. [PubMed] |
| 6. | Andersson HC, Charrow J, Kaplan P, Mistry P, Pastores GM, Prakash-Cheng A, Rosenbloom BE, Scott CR, Wappner RS, Weinreb NJ, International Collaborative Gaucher Group U. S. Regional Coordinators. Individualization of long-term enzyme replacement therapy for Gaucher disease. Genet Med. 2005;7:105-110. [PubMed] [DOI] [Full Text] |
| 7. | Essabar L, Meskini T, Lamalmi N, Ettair S, Erreimi N, Mouane N. Gaucher's disease: report of 11 cases with review of literature. Pan Afr Med J. 2015;20:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 8. | Laks Y, Passwell J. The varied clinical and laboratory manifestations of type II Gaucher's disease. Acta Paediatr Scand. 1987;76:378-380. [PubMed] |
| 9. | Dreborg S, Erikson A, Hagberg B. Gaucher disease--Norrbottnian type. I. General clinical description. Eur J Pediatr. 1980;133:107-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 69] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 10. | Grabowski GA. Gaucher disease: lessons from a decade of therapy. J Pediatr. 2004;144:S15-S19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 11. | Grabowski GA, Andria G, Baldellou A, Campbell PE, Charrow J, Cohen IJ, Harris CM, Kaplan P, Mengel E, Pocovi M, Vellodi A. Pediatric non-neuronopathic Gaucher disease: presentation, diagnosis and assessment. Consensus statements. Eur J Pediatr. 2004;163:58-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 78] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 12. | Linari S, Castaman G. Clinical manifestations and management of Gaucher disease. Clin Cases Miner Bone Metab. 2015;12:157-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 13. | Tamargo RJ, Velayati A, Goldin E, Sidransky E. The role of saposin C in Gaucher disease. Mol Genet Metab. 2012;106:257-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 104] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 14. | Sgambato JA, Park TS, Miller D, Panicker LM, Sidransky E, Lun Y, Awad O, Bentzen SM, Zambidis ET, Feldman RA. Gaucher Disease-Induced Pluripotent Stem Cells Display Decreased Erythroid Potential and Aberrant Myelopoiesis. Stem Cells Transl Med. 2015;4:878-886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 15. | Chaabouni M, Aoulou H, Tebib N, Hachicha M, Ben Becher S, Monastiri K, Yacoub M, Sfar T, Elloumi M, Chakroun N, Miled M, Ben Dridi MF. [Gaucher's disease in Tunisia (multicenter study)]. Rev Med Interne. 2004;25:104-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 16. | Mistry PK, Cappellini MD, Lukina E, Ozsan H, Mach Pascual S, Rosenbaum H, Helena Solano M, Spigelman Z, Villarrubia J, Watman NP, Massenkeil G. A reappraisal of Gaucher disease-diagnosis and disease management algorithms. Am J Hematol. 2011;86:110-115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 136] [Cited by in RCA: 116] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 17. | Liu J, Halene S, Yang M, Iqbal J, Yang R, Mehal WZ, Chuang WL, Jain D, Yuen T, Sun L, Zaidi M, Mistry PK. Gaucher disease gene GBA functions in immune regulation. Proc Natl Acad Sci U S A. 2012;109:10018-10023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 69] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 18. | Mistry PK, Abrahamov A. A practical approach to diagnosis and management of Gaucher's disease. Baillieres Clin Haematol. 1997;10:817-838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 19. | Stirnemann J, Boutten A, Vincent C, Mekinian A, Heraoui D, Fantin B, Fain O, Mentré F, Belmatoug N. Impact of imiglucerase on the serum glycosylated-ferritin level in Gaucher disease. Blood Cells Mol Dis. 2011;46:34-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 20. | Nagral A. Gaucher disease. J Clin Exp Hepatol. 2014;4:37-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 67] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 21. | Weinreb NJ, Deegan P, Kacena KA, Mistry P, Pastores GM, Velentgas P, vom Dahl S. Life expectancy in Gaucher disease type 1. Am J Hematol. 2008;83:896-900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 62] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 22. | Lopez G, Kim J, Wiggs E, Cintron D, Groden C, Tayebi N, Mistry PK, Pastores GM, Zimran A, Goker-Alpan O, Sidransky E. Clinical course and prognosis in patients with Gaucher disease and parkinsonism. Neurol Genet. 2016;2:e57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 23. | Elstein D, Zimran A. Review of the safety and efficacy of imiglucerase treatment of Gaucher disease. Biologics. 2009;3:407-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 24. | Koprivica V, Stone DL, Park JK, Callahan M, Frisch A, Cohen IJ, Tayebi N, Sidransky E. Analysis and classification of 304 mutant alleles in patients with type 1 and type 3 Gaucher disease. Am J Hum Genet. 2000;66:1777-1786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 221] [Cited by in RCA: 224] [Article Influence: 9.0] [Reference Citation Analysis (0)] |