Published online Aug 16, 2016. doi: 10.12998/wjcc.v4.i8.243
Peer-review started: February 14, 2016
First decision: March 30, 2016
Revised: April 24, 2016
Accepted: May 17, 2016
Article in press: May 27, 2016
Published online: August 16, 2016
Processing time: 181 Days and 23.2 Hours
Primary hepatic neuroendocrine tumors (PHNETs) are extremely rare and difficult to distinguish from other liver tumors, such as hepatocellular carcinoma (HCC) and cholangiocarcinoma, based on medical imaging findings. A 70-year-old man was referred for evaluation of liver mass incidentally discovered on abdominal computed tomography. The characteristic finding from dynamic liver magnetic resonance imaging led to a diagnosis of HCC. The patient underwent right hepatectomy. Histopathological and immunohistochemical examination revealed grade 2 neuroendocrine tumor. The postoperative 24-h urinary excretion of 5-hydroxy-indolacetic acid was within the normal range. Further imaging investigations were performed. No other lesions were found making probable the diagnosis of PHNET. This case shows that the diagnosis of PHNET is a medical challenge, requiring differentiation of PHNETs other hepatic masses and exclusion of occult primary neuroendocrine tumors. The diagnosis of PHNET can be ascertained after long term follow-up to exclude another primary origin.
Core tip: Primary hepatic neuroendocrine tumors (PHNETs) are extremely rare and difficult to diagnose before preoperative biopsy or surgery. This case report shows that the diagnosis of PHNET is a medical challenge. Thus differentiation of PHNET from other hepatic mass and exclusion of occult primary neuroendocrine tumors are necessary. The diagnosis of PHNET can be ascertained after long term follow-up to exclude another primary origin.
- Citation: Song JE, Kim BS, Lee CH. Primary hepatic neuroendocrine tumor: A case report and literature review. World J Clin Cases 2016; 4(8): 243-247
- URL: https://www.wjgnet.com/2307-8960/full/v4/i8/243.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v4.i8.243
Neuroendocrine tumors (NETs) are a rare type of tumor, originating in the cells of the neuroendocrine system. NETs arise preferentially in the bronchopulmonary tree (30%) or gastrointestinal tract (50%) and usually metastasis to the liver[1]. However, primary hepatic neuroendocrine tumor (PHNET), first described by Edmondson in 1958[2], is extremely rare, with only 94 cases described in the literature up to 2009[3]. This scarcity of cases makes it difficult for clinicians to diagnose PHNET accurately before biopsy or surgical resection of the tumor[4]. Therefore, PHNET is difficult to differentiate preoperatively from other solid mass, especially hepatocellular carcinoma (HCC). Herein, we report a rare case of PHNET suspected to be HCC before operation.
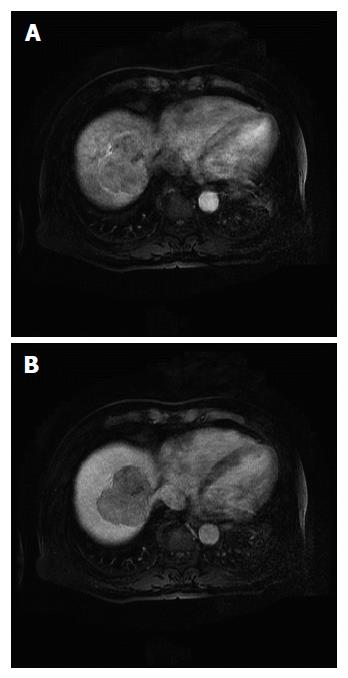
A 70-year-old man complained of left flank pain and gross hematuria. Abdominal computed tomography (CT) revealed a stone in the left uretero-pelvic junction and a 6.8-cm lobulated solid mass in the liver dome. He was referred for evaluation of the incidentally discovered liver mass. He did not have any other symptoms such as jaundice, vomiting, flushing, or diarrhea before admission. His medical history was not significant, except for type 2 diabetes. Physical examination and all biochemical laboratory results were within the normal limits, including tests for liver function and tumor markers (alpha-fetoprotein and carcinoembryonic antigen). No serologic evidence of hepatitis B or C virus infection was found. Endocrine studies were not performed. Liver magnetic resonance imaging (MRI) revealed a lobulated mass involving segments 7 and 8, with mild hypervascularity on arterial phase images and washout on delayed images (Figure 1). Based on the imaging findings, the presumed diagnosis was HCC.
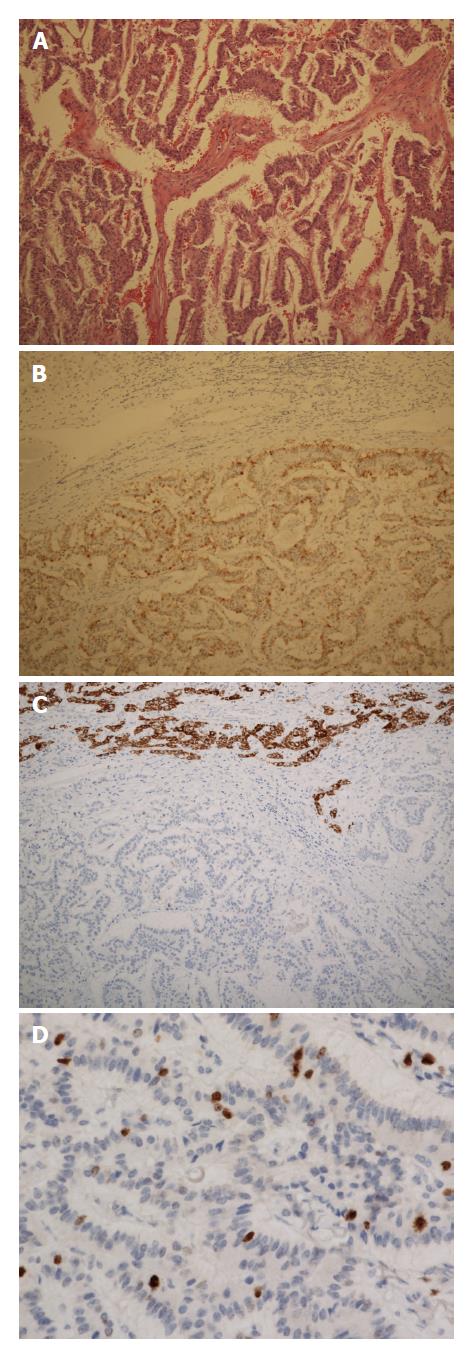
He underwent right hepatectomy and had an uneventful postoperative course. The resected specimen revealed a solid tumor measuring 8.3 cm × 6.5 cm, outlining a heavy cell proliferation with trabecular, glandular, and solid growth patterns. Immunohistochemical staining revealed that tumor cells were diffusely positive for synaptophysin, chromogranin A and CD56, with a Ki67 index of 10%, indicating nuclear reactivity. However, the staining was negative for Heppar-1 and alpha-fetoprotein (Figure 2). Given these findings, a grade 2 neuroendocrine tumor was made. The postoperative 24-h urinary excretion of 5-hydroxy-indole acetic acid (5HIAA) was within the normal range.
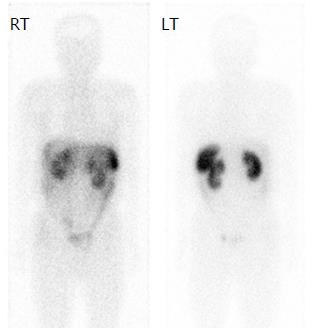
The tumor was considered as a metastatic NET, so further evaluation was undertaken to search for the primary tumor. Chest CT, and upper and lower gastrointestinal endoscopies were performed, and the results were negative for any tumor. The patient underwent an indium-111-Diethylenetriaminepentaacetic acid (DTPA)-octreotide scan, which revealed no lesions positive for somatostatin receptor (Figure 3). The final diagnosis was PHNET based on the pathological and imaging results. At 2-year follow-up the patients shows no signs of liver recurrence or appearance of another primary neuroendocrine tumor.
NETs derive from neuro-ectodermal cells that are dispersed throughout the body. The incidence rate of NET is 6.25 cases per 100000 individuals per year in the United States[5]. Between 54% and 90% of all NET cases arise from the gastrointestinal tract and a primary hepatic location is extremely rare (0.3% of all NET cases)[6].
The grading system used in the 2010 World Health Organization (WHO) classification of gastroenteropancreatic NET (GEP-NET) takes into account the number of mitoses per 10 high power microscopic fields or the percentage of neoplastic cells immunolabeled for the proliferation marker Ki67. These measures indicate the rate of proliferation and correlate with prognosis. GEP-NETs are classified into three types, namely well-differentiated tumors of low-grade malignancy with an indolent development and a good prognosis (grade 1), well-differentiated tumors of intermediate-grade neoplasms (grade 2), and poorly differentiated or high-grade neoplasms that have a poor prognosis (grade 3)[7]. So far, no classification system has been established for PHNETs. However, the categorization of PHNETs according to the 2010 WHO classification of GEP-NETs is useful for the assessment of the prognosis and malignant potential of the tumors[8]. In the present case, the patient was diagnosed as having a grade 2 NET according to the 2010 WHO GEP-NET classification.
PHNETs have a clinical presentation that distinguishes them from other NETs. PHNETs typically grow slowly and become clinically obvious only at an advanced stage[9]. In most cases, they are incidentally discovered, because they most often appear as an endocrinologically silent hepatic mass. Only 6.8% of patients with PHNETs present with the classic carcinoid syndrome, such as skin flushing, diarrhea, and abdominal pain[10]. The presentation of PHNETs can be contrasted with that of hepatic metastases from extrahepatic NETs, which are more commonly related to the typical carcinoid syndrome. However, why PHNETs are frequently endocrinologically silent while their metastatic counterparts are not. PHNETs are more often discovered based on symptoms associated with mass effects on the liver and adjacent organs, such as abdominal distension, vague pain, jaundice, and palpable right upper quadrant mass. In a recently reported review of literature (124 PHNET cases), the mean age at diagnosis was 51.9 years, and no apparent sex predilection was reported (50.8% women and 49.2% men)[10]. However, in another literature, PHNET was slightly more frequent in women (58.5%)[3]. Most tumors were solitary (76.6%) but could be multicentric, with right lobar preference (46.8%)[3]. Our patient was a 70-year-old man who had nonspecific symptoms for the liver tumor, which was located in the right lobe of the liver and found incidentally.
The diagnosis of PHNET is a continuum starting from preoperative to post-surgical stage including long term follow-up to search for extra-hepatic primary[10]. In preoperative imaging study, PHNETs are frequently misdiagnosed as HCC or cholangiocarcinoma (CCC). The gross radiological features of PHNET can be highly varied, with some lesions appearing solid or cystic, as well as having diffuse or well-circumscribed margin[11]. PHNETs have a rich blood supply from the hepatic artery, which is reflected in the type of dynamic enhancement curves. Wang et al[12] reported that all lesions were remarkably enhanced in the arterial phase and that the reconstruction of the arterial phase confirmed a rich blood supply. HCC has the typical patterns of marked arterial enhancement and washout in the portal and delayed phases, which easily confounds the diagnosis of PHNET.
Several studies has reported that preoperative diagnosis for PHNETs with needle biopsy[13-16], but the diagnostic accuracy is not high enough. The low diagnostic accuracy is obvious in reported cases that PHNETs are misdiagnosed as HCC or CCC[15,17,18]. It is still unclear on the value of liver biopsy therefore post-operative histological and immunohistochemical evaluation serves as the essential method for the definite diagnosis[10].
Radiological findings are similar for both primary and metastatic NETs[19]. Moreover, the pathological features of PHNET are difficult to distinguish from those of hepatic metastases. Therefore, careful investigations are required to exclude the presence of extrahepatic NET. These include CT, MRI, somatostatin scintigraphy, positron-emitting tomography (PET), gastroscopy, colonoscopy, bronchoscopy and operative exploration. When the primary tumor is still considered to be hepatic NET even after thorough investigation, long-term reexamination with CT, MRI, octreotide scan and PET are useful to detect a small extrahepatic tumor that may have initially been missed[20]. Occasionally, a very close post surgical long-term follow-up is needed for definite diagnosis of PHNET[21].
Gross pathological findings of PHNETs are gray-yellow in color and well demarcated mass with multiple irregular hemorrhagic lesions or with cystic area[22,23], ranging in size from 3.2 to 18 cm (mean: 8.6 ± 5.7 cm)[15]. Routine pathological examination using the hematoxylin-eosin staining method shows insular, nested, trabecular or mixed pattern of cell growth, but it is not specific for NETs and only beneficial to tumor classification. Neuron-specific enolase, chromogranin A and synaptophysin are generally accepted as highly sensitive immunohistochemical markers for the diagnosis of NETs. The tumor in our case was immunoreactive for synaptophysin and chromogranin A.
No treatment guideline for PHNET has been recently established, but surgical resection (e.g., wedge resection or formal lobectomy) is the treatment of choice that can provide a complete cure[14,24]. PHNETs are associated with a resectability rate of 70% and a 5-year survival rate after hepatectomy of 78%[5]. Recent study shows that the extent of the disease and type of surgery does not affect the survival rate[23]. In patients with unresectable disease, various palliative options exist, such as systemic 5 fluorouracil[25], hepatic artery embolization[26], and octreotide therapy[27]. However, data on these are limited. Currently, liver transplantation has been suggested to be a treatment option in selected patients with multiple lesions or impaired liver function[28].
The present case shows that the diagnosis of PHNET is a medical challenge. PHNETs are rare and asymptomatic. They are quite difficult to distinguish from other liver tumors, such as HCC and cholangiocarcinoma, based on medical imaging findings. PHNET should be suspected in patients with no chronic liver disease, with normal serum alpha-fetoprotein levels, and with solitary hypervascular tumor in imaging studies. Differentiation of PHNET from other hepatic mass and exclusion of occult primary neuroendocrine tumors are necessary. The diagnosis of PHNET can be ascertained after long term follow-up to exclude another primary origin.
A 70-year-old man was referred for evaluation of liver mass incidentally discovered on abdominal computed tomography.
The characteristic finding from dynamic liver magnetic resonance imaging led to a diagnosis of hepatocellular carcinoma.
Cholangiocarcinoma.
All biochemical laboratory results were within the normal limits, including tests for liver function and tumor markers (alpha-fetoprotein and carcinoembryonic antigen).
Liver magnetic resonance imaging revealed a lobulated mass involving segments 7 and 8, with mild hypervascularity on arterial phase images and washout on delayed images.
Immunohistochemical staining revealed that tumor cells were diffusely positive for synaptophysin, chromogranin A and CD56, with a Ki67 index of 10%, indicating nuclear reactivity. Given these findings, a grade 2 neuroendocrine tumor was made.
He underwent right hepatectomy.
This is a well written case report of a rare pathology. It lacks long term follow-up in order to support the immunohistological diagnosis of primary hepatic neuroendocrine tumor.
Manuscript source: Unsolicited manuscript
Specialty type: Medicine
Country of origin: South Korea
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C, C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Alimehmeti R, Donadon M, Rocca A S- Editor: Qi Y L- Editor: A E- Editor: Zhang FF
| 1. | Fenoglio LM, Severini S, Ferrigno D, Gollè G, Serraino C, Bracco C, Castagna E, Brignone C, Pomero F, Migliore E. Primary hepatic carcinoid: a case report and literature review. World J Gastroenterol. 2009;15:2418-2422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 48] [Cited by in RCA: 40] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 2. | Edmondson HA. Tumor of the liver and intrahepatic bile duct. Atlas of tumor pathology. 1958;7:105-109. |
| 3. | Lin CW, Lai CH, Hsu CC, Hsu CT, Hsieh PM, Hung KC, Chen YS. Primary hepatic carcinoid tumor: a case report and review of the literature. Cases J. 2009;2:90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 68] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 4. | Sano K, Kosuge T, Yamamoto J, Shimada K, Takayama T, Yamasaki S, Makuuchi M. Primary hepatic carcinoid tumors confirmed with long-term follow-up after resection. Hepatogastroenterology. 1999;46:2547-2550. [PubMed] |
| 5. | Knox CD, Anderson CD, Lamps LW, Adkins RB, Pinson CW. Long-term survival after resection for primary hepatic carcinoid tumor. Ann Surg Oncol. 2003;10:1171-1175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 105] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 6. | Camargo ÉS, Viveiros Mde M, Corrêa Neto IJ, Robles L, Rezende MB. Primary hepatic carcinoid tumor: case report and literature review. Einstein (Sao Paulo). 2014;12:505-508. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 7. | Klimstra DS, Modlin IR, Coppola D, Lloyd RV, Suster S. The pathologic classification of neuroendocrine tumors: a review of nomenclature, grading, and staging systems. Pancreas. 2010;39:707-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 776] [Cited by in RCA: 752] [Article Influence: 50.1] [Reference Citation Analysis (2)] |
| 8. | Zhao J, Yang B, Xu C, Zhang WS, Ji Y, Chen LL, Tan YS, Zeng HY, Zhu XZ. [Study on clinicopathologic grading system and prognosis of primary hepatic neuroendocrine neoplasms]. Zhonghua Bing Li Xue Za Zhi. 2012;41:102-106. [PubMed] |
| 9. | Jia C, Zhang Y, Xu J, Sun K. Experience in primary hepatic neuroendocrine tumor. Turk J Gastroenterol. 2012;23:546-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 10. | Quartey B. Primary Hepatic Neuroendocrine Tumor: What Do We Know Now? World J Oncol. 2011;2:209-216. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 11. | Kellock T, Tuong B, Harris AC, Yoshida E. Diagnostic Imaging of Primary Hepatic Neuroendocrine Tumors: A Case and Discussion of the Literature. Case Rep Radiol. 2014;2014:156491. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 12. | Wang LX, Liu K, Lin GW, Jiang T. Primary hepatic neuroendocrine tumors: comparing CT and MRI features with pathology. Cancer Imaging. 2015;15:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 37] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 13. | Dala R, Shoosmith J, Lilenbaum R, Cabello-Inchausti B. Primary hepatic neuroendocrine carcinoma: an underdiagnosed entity. Ann Diagn Pathol. 2006;10:28-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Iwao M, Nakamuta M, Enjoji M, Kubo H, Fukutomi T, Tanabe Y, Nishi H, Taguchi KI, Kotoh K, Nawata H. Primary hepatic carcinoid tumor: case report and review of 53 cases. Med Sci Monit. 2001;7:746-750. [PubMed] |
| 15. | Hwang S, Lee YJ, Lee SG, Kim CW, Kim KH, Ahn CS, Moon KM, Ko KH, Kim KW, Choi NK. Surgical treatment of primary neuroendocrine tumors of the liver. J Gastrointest Surg. 2008;12:725-730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 16. | Skagias L, Vasou O, Ntinis A, Kondi-Pafiti A, Koureas A, Politi E. Primary hepatic neuroendocrine tumor with exophytic growth: report of a case with diagnosis by fine needle aspiration biopsy. Acta Cytol. 2010;54:202-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 17. | Iimuro Y, Deguchi Y, Ueda Y, Tanaka A, Iwasa Y, Ishihara M, Mizuta K, Yamamoto Y, Ikai I, Shimahara Y. Primary hepatic carcinoid tumor with metachronous lymph node metastasis after long-term follow up. J Gastroenterol Hepatol. 2002;17:1119-1124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 18. | Moriura S, Ikeda S, Hirai M, Naiki K, Fujioka T, Yokochi K, Gotou S. Hepatic gastrinoma. Cancer. 1993;72:1547-1550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 19. | Baek SH, Yoon JH, Kim KW. Primary hepatic neuroendocrine tumor: gadoxetic acid (Gd-EOB-DTPA)-enhanced magnetic resonance imaging. Acta Radiol Short Rep. 2013;2:2047981613482897. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 20. | Landen S, Elens M, Vrancken C, Nuytens F, Meert T, Delugeau V. Giant hepatic carcinoid: a rare tumor with a favorable prognosis. Case Rep Surg. 2014;2014:456509. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 21. | Donadon M, Torzilli G, Palmisano A, Del Fabbro D, Panizzo V, Maggioni M, Santambrogio R, Montorsi M. Liver resection for primary hepatic neuroendocrine tumours: report of three cases and review of the literature. Eur J Surg Oncol. 2006;32:325-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 22. | Pilichowska M, Kimura N, Ouchi A, Lin H, Mizuno Y, Nagura H. Primary hepatic carcinoid and neuroendocrine carcinoma: clinicopathological and immunohistochemical study of five cases. Pathol Int. 1999;49:318-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 74] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 23. | Shetty PK, Baliga SV, Balaiah K, Gnana PS. Primary hepatic neuroendocrine tumor: an unusual cystic presentation. Indian J Pathol Microbiol. 2010;53:760-762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 24. | Yalav O, Ülkü A, Akçam TA, Demiryürek H, Doran F. Primary hepatic neuroendocrine tumor: Five cases with different preoperative diagnoses. Turk J Gastroenterol. 2012;23:272-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 25. | Andreola S, Lombardi L, Audisio RA, Mazzaferro V, Koukouras D, Doci R, Gennari L, Makowka L, Starzl TE, van Thiel DH. A clinicopathologic study of primary hepatic carcinoid tumors. Cancer. 1990;65:1211-1218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 26. | Krishnamurthy SC, Dutta V, Pai SA, Kane SV, Jagannath P, Desouza LJ, Deshpande R, Desai PB. Primary carcinoid tumor of the liver: report of four resected cases including one with gastrin production. J Surg Oncol. 1996;62:218-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 27. | Wängberg B, Nilsson O, Johanson V, Kölby L, Forssell-Aronsson E, Andersson P, Fjälling M, Tisell L, Ahlman H. Somatostatin Receptors in the Diagnosis and Therapy of Neuroendocrine Tumor. Oncologist. 1997;2:50-58. [PubMed] |
| 28. | Fenwick SW, Wyatt JI, Toogood GJ, Lodge JP. Hepatic resection and transplantation for primary carcinoid tumors of the liver. Ann Surg. 2004;239:210-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 70] [Article Influence: 3.3] [Reference Citation Analysis (0)] |