Published online Oct 16, 2025. doi: 10.12998/wjcc.v13.i29.108608
Revised: May 27, 2025
Accepted: July 29, 2025
Published online: October 16, 2025
Processing time: 132 Days and 10.4 Hours
Hepatic venous outflow obstruction (HVOO) is a rare but serious complication of liver transplantation, particularly in piggyback liver transplantation techniques where the inferior vena cava (IVC) is preserved.
A transplanted liver patient underwent retransplantation due to hepatic artery thrombosis and subsequently developed HVOO caused by graft compression of the IVC. A novel approach using a retrohepatic tissue expander effectively relieved the IVC compression, restored venous outflow, and stabilized hemodynamics. We discuss this case in the context of current treatment options and advances in HVOO management, from endovascular interventions such as balloon dilation and stenting to innovative surgical solutions such as graft repositioning and retrohepatic implants.
This case shows how important personalized treatments are for managing HVOO and how tissue expanders can be an adjustable and less invasive option.
Core Tip: Hepatic venous outflow obstruction (HVOO) is a serious complication of liver transplantation, particularly with the piggyback technique where the inferior vena cava (IVC) is preserved. A unique approach using a retrohepatically placed tissue expander may successfully relieve IVC compression caused by graft malposition and stabilize hemodynamics without needing major surgery. This minimally invasive method demonstrates adjustable and effective decompression of the hepatic veins, suggesting a new therapeutic option for complex vascular complications after liver transplant. Our report highlights the importance of proactive monitoring and individualized interventions in managing HVOO to improve graft function and patient outcomes.
- Citation: Petrovic I, Brekalo K, Romic I, Silovski H, Sesa V, Galunic Cicak R, Bulic K, Koltay OM, Strajher IM, Mrzljak A. Tissue expanders – an innovative approach in the management of hepatic venous outflow obstruction after liver transplantation: A case report. World J Clin Cases 2025; 13(29): 108608
- URL: https://www.wjgnet.com/2307-8960/full/v13/i29/108608.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v13.i29.108608
Liver transplantation (LT) has transformed from an experimental procedure into a life-saving intervention, providing hope to thousands of patients suffering from irreversible liver disease, which contributes to 4% of global mortality[1]. Vascular issues are the most dangerous complications after LT, often resulting in graft failure, which could lead to a fatal outcome. While there have been marked improvements in vascular techniques since the beginnings of LT, the incidence of vascular complications remains around 8% in deceased donor LT and around 10% in living donor LT[2]. Vascular complications are classified according to the location of the lesion, in the inflow vessels: Hepatic artery and/or portal vein, or outflow vessels – the hepatic veins and/or inferior vena cava (IVC)[3]. Hepatic venous outflow obstruction (HVOO) is a rare complication, with an incidence of 3%–4% when the IVC is preserved, as in piggyback LT (PBLT) technique[4]. Numerous factors can impact the development of HVOO: Surgical technique used, size and fit of the graft, or the recipient’s underlying vascular anatomy and physiology[5,6]. The incidence may be even higher (4%–9%) in the pediatric population due to smaller anastomosis diameter and a potential size mismatch between the donor and recipient vessels[7]. The clinical manifestations of HVOO include refractory ascites, lower extremity edema, hepatic congestion, and abnormal liver function tests. Doppler ultrasound is crucial for diagnosing HVOO and typically shows reduced hepatic venous flow with dampened phasicity[8]. The treatment of HVOO depends both on the time of presentation and the etiology of the venous outflow obstruction. Potential treatment methods include endovascular procedures such as balloon dilatation or venous stenting, surgical procedures such as transplantation or cavoplasty, or conservative measures with diuretics and blood pressure management. Here, we present a novel approach of HVOO management using a retrohepatically placed tissue expander and discuss it in the context of currently available treatment options.
A 58-year-old woman with alcoholic liver disease required frequent hospitalization due to decompensated liver cirrhosis with sequelae including ascites, portal encephalopathy, renal injury and esophageal varices.
She had been abstinent for > 1 year when she was evaluated and placed on the transplant list with a Model for End-Stage Liver Disease (MELD) score of 19.
No other illnesses were present.
She was diagnosed with cirrhosis for the last 2 years. No liver disorders were present in her family.
Physical examination demonstrated no specific findings. The patient was obese and had mild pain in the upper right abdomen on palpation.
The laboratory examinations on admission showed creatinine 2.1 mg/dL, bilirubin 1.1 mg/dL, international normalized ratio 1.6 and sodium 137 meQ/L, which resulted in MELD score 19. She had elevated alkaline phosphatase (160 U/L) and normal aspartate aminotransferase (20 U/L), alanine aminotransferase (29 U/L) and -glutamyl transpeptidase (30 U/L). Other findings were normal.
After 2 months on the list, LT was performed using a whole graft from a donor after brain death using the PBLT technique. Due to the origin of the donor right hepatic artery (RHA) from the superior mesenteric artery, the RHA was transected and an end-to-end anastomosis between the RHA and the gastroduodenal artery was performed during graft preparation on the back table. Vascular anastomoses included a wide end-to-side hepatocaval anastomosis, an end-to-end porto-portal anastomosis, and an end-to-end arterial anastomosis.
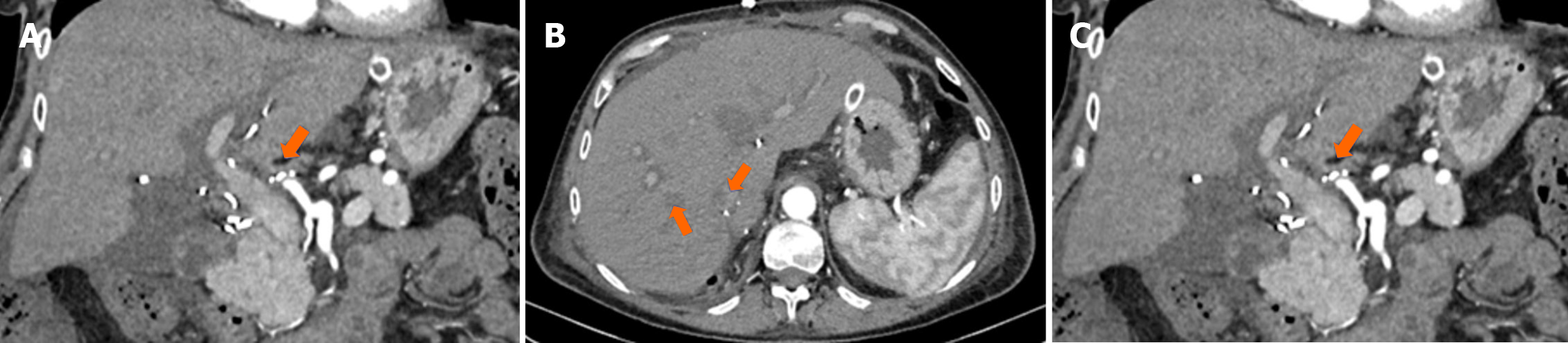
On postoperative day 7, elevated liver enzymes prompted computed tomography (CT) angiography which showed a lack of opacification in the hepatic arteries suggestive of hepatic artery thrombosis (HAT) at the anastomosis site and localized ischemic zones in liver segment VI (Figure 1A and 1B). The finding was discussed with the patient, as well as treatment options including retransplantation, but the patient insisted on being discharged. One month later, she returned complaining of severe pain. CT scans confirmed the presence of peripheral wedge infarcts in graft segments VI and VII (Figure 1C), which resulted in the patient being relisted due to HAT.
HVOO.
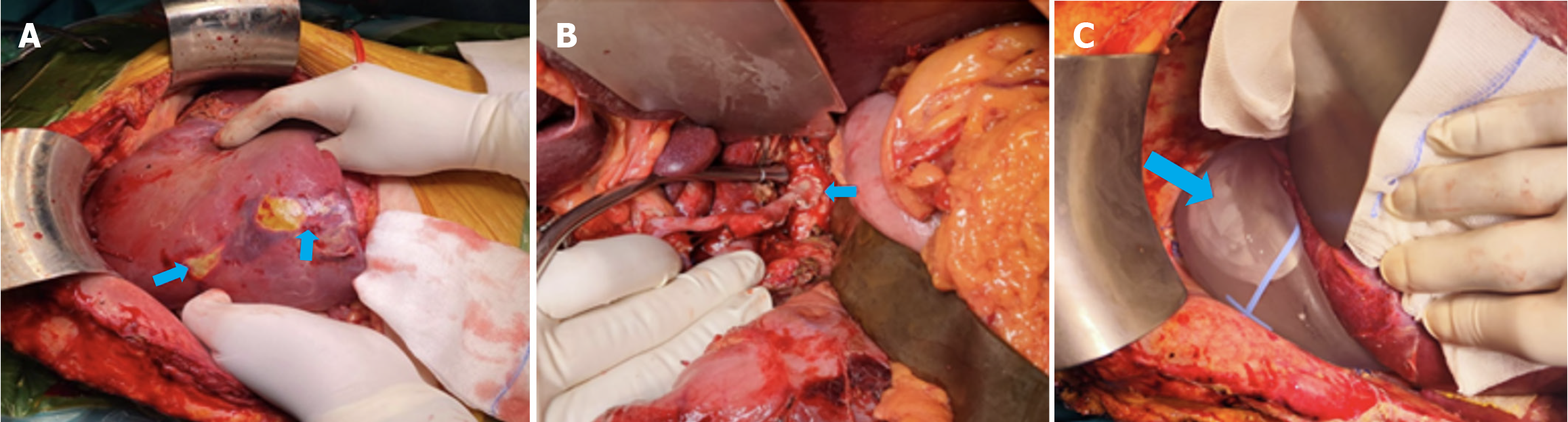
One week after the patient’s relisting, retransplantation was performed using a whole liver graft from a donor after brain death (measured liver diameter in the midclavicular line was 16 cm, Donor Risk Index was 1.35 and cold ischemic time 3.7 h). Intraoperatively, the previous graft had ischemic lesions in segments VI and VII (Figure 2A). Following adhesiolysis, liver mobilization and hepatectomy, the new liver graft was placed in the abdomen. A wide end-to-side hepatocaval anastomosis, and an end-to-end portoportal anastomosis were created. Due to thrombosis and poor quality of the recipient arterial wall at the site of the previous anastomosis, a direct end-to-end arterial anastomosis was abandoned and a direct end-to-side anastomosis of the donor aorta and celiac trunk as a patch on the recipient’s supraceliac aorta was performed (Figure 2B).
The procedure was complicated by a drop in blood pressure when placing the liver in its anatomical position in the right hemiabdomen. Upon intraoperative color Doppler ultrasound, IVC flow decreased, but improved after the liver was lifted off the IVC. A decision was made to place the liver more to the left by suturing the remaining teres hepatis ligament to the abdominal wall, which resulted in improvement of blood pressure.
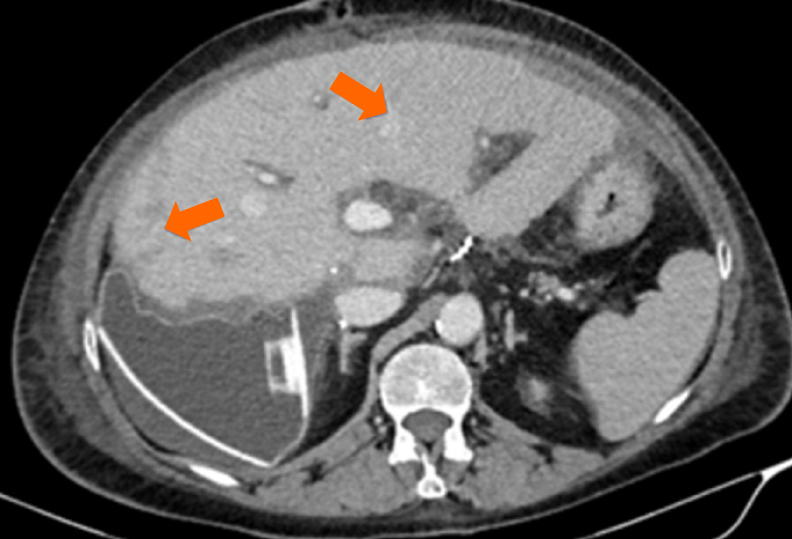
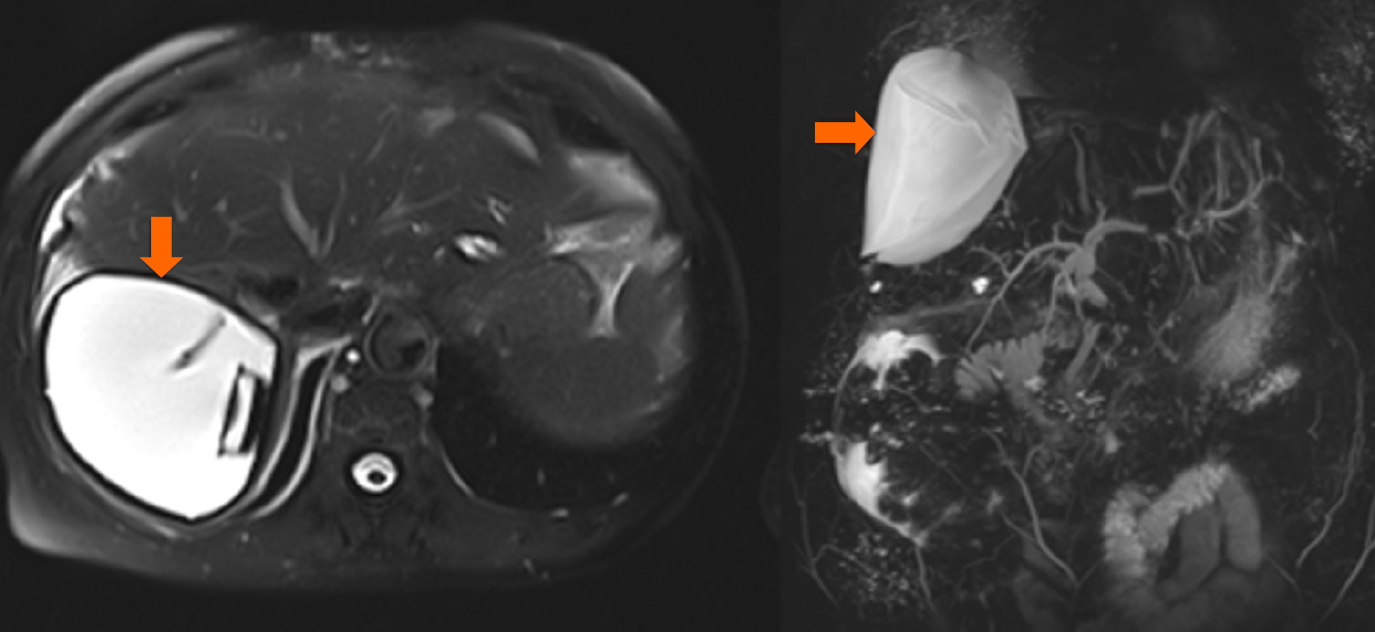
In the intensive care unit, the patient was persistently hypotensive with elevated lactate and transaminase levels despite substantial volume support and vasopressors, and Doppler ultrasound showed significant long-distance stenosis of the IVC caused by external compression of the donor liver. Immediately, the patient underwent relaparotomy, during which a laterolateral anastomosis of the donor and recipient IVC was performed. Additionally, a tissue expander was placed under the right hemiliver to lift it off the IVC (Figure 2C). CT examination performed 1 week after final surgery revealed narrow confluent ischemic areas in the VII segment, IVa segment and the caudate lobe of the liver graft (Figure 3). Three weeks later, the patient showed no clinical deterioration and was discharged from hospital. Follow-up CT performed 3 months later showed significant regression of the previously described ischemic areas. After 1 year of follow-up, the patient’s liver function was stable (alkaline phosphatase 160 U/L, aspartate aminotransferase 20 U/L, alanine aminotransferase 29 U/L and -glutamyl transpeptidase 30 U/L) with occasional discomfort in the upper right quadrant. Magnetic resonance imaging showed an optimal position of the tissue expander with patent vessels and no new ischemic lesions (Figure 4).
In the presented case, HVOO developed immediately after the second LT. Persistent postoperative hypotension and Doppler imaging demonstrated dampened flow consistent with venous compression by the graft, which were indicative of the diagnosis. Early HVOO (within 28 days post-LT) is often related to technical factors such as graft size[9], graft malposition, tight anastomosis, or kinking[10]. We found five single institution studies describing HVOO cases after LT (Table 1). Overall, these studies comprised 52 patients. Narrowing at the venous anastomosis was the predominant cause of HVOO and, in 10 cases, some type of ectopic material was used to manage HVOO. Pitchaimuthu et al[11] reported HVOO in patients who had undergone liver retransplantation, which might be explained by the technical aspects of the procedure, with possible kinking or twisting of the IVC anastomosis, performed usually on the IVC cuff remaining from the failed graft. However, this observation was based on small numbers since only two (3.3%) of 60 patients who underwent retransplantation developed HVOO. In all 27 patients with HVOO (10 with clinically significant HVOO), endovascular intervention was performed. The study shows that endovascular intervention is an effective and safe procedure providing symptom relief with long-lasting primary patency[11]. In our case, liver retransplantation (along with liver malposition due to weight and shape) was the probable cause of HVOO, in addition to altered anatomy of the right subphrenic region, inflammatory and fibrotic reactions, and repetitive manipulation of IVC, which contribute to IVC compression or blood flow disturbances at the anastomosis. Most authors highlight the importance of careful graft sizing and positioning during the transplant procedure to minimize the risk of outflow obstruction. In contrast, late-onset HVOO (beyond 28 days post-LT) is typically associated with neointimal hyperplasia or fibrosis at the anastomosis site[10]. It is important to keep in mind that syndromes like veno-occlusive disease, Budd–Chiari syndrome and congestive hepatopathy, can all manifest as HVOO. Veno-occlusive disease occurs at the level of sinusoids and terminal venules, Budd–Chiari syndrome occurs from the hepatic veins to the superior end of the IVC, and congestive hepatopathy occurs at heart level[12]. Since all these syndromes usually develop gradually, patients more commonly present with abdominal pain caused by hepatomegaly, jaundice and ascites, rather than the dramatic hypotension as seen in our case of extravascular compression caused by the large-for-size graft.
| Arudchelvam et al[8] | Sambommatsu et al[10] | Lim et al[9] | Khorsandi et al[4] | Wang et al[14] | ||
| Number of HVOO cases | 19 | 16 | 3 | 7 | 7 | |
| LT | 17 | 14 | 3 | 7 | 7 | |
| Retransplants | 2 | 2 | 0 | 0 | 0 | |
| Indication for LT | Acute liver failure | 5 | 2 | 0 | 0 | 0 |
| Hepatitis B or C | 3 | 2 | 0 | 2 | 2 | |
| HCC | 4 | 1 | 1 | 2 | 0 | |
| Alcoholic cirrhosis | 1 | 3 | 1 | 0 | 0 | |
| Other | 6 | 8 | 1 | 5 | 5 | |
| HVOO etiology | Narrowing at anastomosis | 17 | 14 | 0 | ||
| Thrombosis | 2 | 0 | 0 | |||
| Kinking | 0 | 2 | 3 | |||
| HVOO classification | Early onset | 11 | 2 | 3 | 3 | |
| Late onset | 8 | 14 | 0 | 4 | ||
| Outcome | Stenting | 18 | 16 | 1 | 6 | 7 |
| Required liver re-transplantation due to HVOO | 1 | 0 | 1 | 1 | 0 | |
| Type of intervention | ||||||
| Interventional radiology | Balloon angioplasty | 1 | 10 | |||
| Stenting | 17 | 5 | ||||
| Placement of ectopic material | Breast implant | 2 | ||||
| Surgical pads | 1 | |||||
| Tissue expander | 4 | |||||
| Foley catheter | 3 | |||||
| Cavoplasty | 4 | |||||
| Other surgical interventions | 1 | |||||
| Conservative management (diuretics) | 2 | |||||
| Liver re-transplantation | 1 | 1 | 1 | |||
Postoperative follow-up with Doppler ultrasound is essential to monitor for signs of HVOO and allows prompt intervention to prevent graft failure. However, the treatment of HVOO depends on the time of presentation as well as on the etiology. Balloon dilation and stenting are appropriate choices when the cause of the HVOO is kinking of the veins or anastomotic stenosis[8]. Primary stent placement has been suggested as the appropriate treatment for most cases of HVOO after LDLT[13]. However, outflow obstruction caused by large-for-size whole liver graft compression may not be as effective in these cases. Several innovative surgical techniques have been used to address this issue.
In work presented by Wang et al[14], the treatment of HVOO involved the intraoperative use of a tissue expander and/or Foley catheter to reposition the liver graft and relieve mechanical venous obstruction caused by malposition. Doppler ultrasonography identified inadequate hepatic and portal venous flow in seven patients, prompting interventions. Tissue expanders (filled with 300–770 mL saline) or Foley catheters (filled with 15–75 mL of saline) were placed in the right subphrenic space to mechanically adjust the graft position and improve venous drainage. Post-intervention Doppler imaging confirmed significant improvement in venous outflow. The prostheses were gradually deflated starting from postoperative day 6–27 and removed between day 19–56 (mean: 38 days). All complications (e.g., chylous ascites, bile leak and infections) were managed nonoperatively, and no recurrent outflow obstruction was observed after device removal. Patient and graft survival rates at 1 and 2 years were 100%, demonstrating that this approach was safe and effective for managing HVOO in LT recipients.
In the case series presented by Lim et al[9], HVOO during LT involving large-for-size grafts was treated intraoperatively through retrohepatic placement of supportive materials to relieve IVC compression and restore venous flow. Three patients experienced severe graft congestion upon reperfusion, attributed to mechanical compression by oversized grafts. In two cases, breast implants were placed in the right subphrenic space, effectively repositioning the liver to restore venous outflow. In one case, the implants were later removed without complication, while in the other, they remained in place due to their bio-inert nature. In the third case, four abdominal pads were similarly used, although the patient required retransplantation and died from multiorgan failure. This approach aligns with prior techniques utilizing inflatable or inert materials (e.g., Foley catheters, Blakemore balloons and surgical gloves), with breast implants offering a durable, infection-resistant option that can be left indefinitely if needed.
In the report by Nawabi et al[15], treatment of HVOO post-LT involved a combination of surgical anastomotic revision and placement of a breast implant tissue expander to mechanically stabilize the graft position. The patient, who had right diaphragmatic eventration due to phrenic nerve palsy, experienced HVOO caused by allograft rotation following PBLT. Upon reoperation, a side-to-side cavo-cavostomy was performed to revise the anastomosis. Due to persistent graft malposition from diaphragmatic eventration, an 850-cm3 breast implant tissue expander was placed posterior to the liver graft to prevent further rotation and maintain hepatic venous outflow with no recurrence of HVOO. The breast implant remained in place without complications, and the patient maintained excellent graft function for 10 years, demonstrating the utility of bio-inert implants for long-term management of graft positioning in HVOO.
Gastaca et al[16] demonstrated three cases of early post-transplant HVOO due to graft malposition or IVC anastomotic kinking that were successfully treated with surgical placement of a 300-cm3 silicone gel-filled breast implant in the right hepatic fossa. In all patients, initial management with angioplasty showed elastic (nonfibrotic) stenosis and was clinically ineffective, prompting surgical reintervention. The breast implant repositioned the graft, resolved massive ascites and renal impairment, and restored hepatic outflow confirmed by Doppler ultrasonography and venography. No prosthesis-related complications were observed over 30–58 months of follow-up.
In our patient a tissue expander filled up to 400 mL was placed in the retrohepatic position, which successfully relieved the compression on the IVC, restored the blood flow, and stabilized the patients hemodynamic. Vigilant monitoring demonstrated relief of the compression, and the patient remained normotensive with no signs of graft failure. This approach confirms the utility of tissue expanders as a minimally invasive option to manage graft-related HVOO. The main advantage of expander is its adjustability because it can be gradually filled to gently elevate the graft and decompress the hepatic veins, without the need for major revisional surgeries. In addition, expanders are available in different sizes and shapes, these are biologically inert and can be easily removed if necessary. As our literature review demonstrates, there were only four reported cases of tissue expander application in HVOO, so there is a lack of scientific data on this topic. Therefore, our case may contribute to better understanding of this specific management of post-transplant HVOO. Complications related to tissue expanders are predominantly studied in breast surgery, trauma and burns, and these include seroma, hematoma, migration or rupture[17,18]. The infection rate is 2.5%–5% and overall complication rate < 12%, and the majority of complications are minor. These data suggest that tissue expander may be effective for HVOO treatment after LT. To achieve optimal long-term outcomes, clinicians should be aware of the importance of proactive surveillance and multidisciplinary approach to managing this complex complication of LT.
This case underscores the complexities and challenges inherent in LT, particularly when dealing with vascular complications such as HAT and HVOO. HVOO is a complex and challenging complication of LT that requires prompt diagnosis and tailored treatment. While advances in surgical techniques and endovascular interventions have improved outcomes, management of HVOO remains a significant challenge. Further research is needed to refine our strategies for preventing and treating this serious complication, and to improve the long-term survival and quality of life for LT recipients. Potential specific areas of investigation may include prevention strategies (such as preoperative CT volumetry and three-dimensional graft reconstruction or optimal surgical techniques for anastomoses) and therapeutic strategies (interventional radiology, cavoplasty or pharmacological measures). Due to the rarity of this complication, every single case should be analyzed and published, since it will contribute to better understanding of the etiology and management of HVOO after LT.
| 1. | Devarbhavi H, Asrani SK, Arab JP, Nartey YA, Pose E, Kamath PS. Global burden of liver disease: 2023 update. J Hepatol. 2023;79:516-537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 849] [Reference Citation Analysis (4)] |
| 2. | Khalaf H. Vascular complications after deceased and living donor liver transplantation: a single-center experience. Transplant Proc. 2010;42:865-870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 86] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 3. | Pandhi MB, Lipnik AJ, Niemeyer MM. Endovascular Treatment of Hepatic Venous Outflow Obstruction after Liver Transplant. Dig Dis Interv. 2019;3:277-286. [DOI] [Full Text] |
| 4. | Khorsandi SE, Athale A, Vilca-Melendez H, Jassem W, Prachalias A, Srinivasan P, Rela M, Heaton N. Presentation, diagnosis, and management of early hepatic venous outflow complications in whole cadaveric liver transplant. Liver Transpl. 2015;21:914-921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 5. | Estrin JA, Belani KG, Ascher NL, Lura D, Payne W, Najarian JS. Hemodynamic changes on clamping and unclamping of major vessels during liver transplantation. Transplant Proc. 1989;21:3500-3505. [PubMed] |
| 6. | Tzakis A, Todo S, Starzl TE. Orthotopic liver transplantation with preservation of the inferior vena cava. Ann Surg. 1989;210:649-652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 459] [Cited by in RCA: 408] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 7. | Choi JW, Jae HJ, Kim HC, Yi NJ, Lee KW, Suh KS, Chung JW. Long-term outcome of endovascular intervention in hepatic venous outflow obstruction following pediatric liver transplantation. Liver Transpl. 2015;21:1219-1226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 8. | Arudchelvam J, Bartlett A, McCall J, Johnston P, Gane E, Munn S. Hepatic venous outflow obstruction in piggyback liver transplantation: single centre experience. ANZ J Surg. 2017;87:182-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 9. | Lim C, Osseis M, Tudisco A, Lahat E, Sotirov D, Salloum C, Azoulay D. Hepatic venous outflow obstruction after whole liver transplantation of large-for-size graft: versatile intra-operative management. Ann Hepatobiliary Pancreat Surg. 2018;22:321-325. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 10. | Sambommatsu Y, Hirukawa K, Shimata K, Honda M, Sakurai Y, Ishii M, Ibuki S, Isono K, Irie T, Kawabata S, Hirao H, Sugawara Y, Tamura Y, Ikeda O, Hirai T, Inomata Y, Morinaga J, Hibi T. Hepatic venous outflow obstruction after adult living donor liver transplantation. Liver Transpl. 2023;29:1292-1303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 11. | Pitchaimuthu M, Roll GR, Zia Z, Olliff S, Mehrzad H, Hodson J, Gunson BK, Perera MT, Isaac JR, Muiesan P, Mirza DF, Mergental H. Long-term follow-up after endovascular treatment of hepatic venous outflow obstruction following liver transplantation. Transpl Int. 2016;29:1106-1116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 12. | Bayraktar UD, Seren S, Bayraktar Y. Hepatic venous outflow obstruction: three similar syndromes. World J Gastroenterol. 2007;13:1912-1927. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 92] [Cited by in RCA: 87] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 13. | Ko GY, Sung KB, Yoon HK, Kim JH, Song HY, Seo TS, Lee SG. Endovascular treatment of hepatic venous outflow obstruction after living-donor liver transplantation. J Vasc Interv Radiol. 2002;13:591-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 85] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 14. | Wang CC, Concejero AM, Yong CC, Chen YS, Wang SH, Lin CC, Liu YW, Yang CH, Lin TS, Hung KC, Jawan B, Cheng YF, Ibrahim S, Chen CL. Improving hepatic and portal venous flows using tissue expander and Foley catheter in liver transplantation. Clin Transplant. 2006;20:81-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 15. | Nawabi A, Lambou KR, Meta H, Nawabi AZ, Kumer S. Utilization of Breast Implant Tissue Expander for Hepatic Venous Outflow Obstruction Post Liver Transplant: A Case Report. Am J Biomed Sci Res. 2024;22:802. |
| 16. | Gastaca M, Valdivieso A, Ruiz P, Gonzalez J, Ventoso A, de Urbina JO. Venous outflow obstruction after orthotopic liver transplantation: use of a breast implant to maintain graft position. Clin Transplant. 2011;25:E320-E326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 17. | Wang SL, Sze DY, Busque S, Razavi MK, Kee ST, Frisoli JK, Dake MD. Treatment of hepatic venous outflow obstruction after piggyback liver transplantation. Radiology. 2005;236:352-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 63] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 18. | Cunha MS, Nakamoto HA, Herson MR, Faes JC, Gemperli R, Ferreira MC. Tissue expander complications in plastic surgery: a 10-year experience. Rev Hosp Clin Fac Med Sao Paulo. 2002;57:93-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 70] [Article Influence: 3.0] [Reference Citation Analysis (0)] |