Published online Aug 6, 2024. doi: 10.12998/wjcc.v12.i22.5229
Revised: May 23, 2024
Accepted: June 7, 2024
Published online: August 6, 2024
Processing time: 78 Days and 18.4 Hours
The gastrointestinal tract is a well-known extranodal site of lymphoma. B-cell lymph
A 45-year-old woman was admitted to our hospital for physical examination. Gastroenteroscopy revealed a visible pedunculated polyp in the transverse colon, for which endoscopic submucosal dissection (ESD) was performed. Pathology suggested highly active proliferation of T lymphocytes with atypical hyperplasia.
A middle-aged female patient was found to have colonic T-cell lymphoma by endoscopy. The lesion was successfully removed by ESD, and the surgical margin was negative. It is essential to raise awareness of colonic T-cell lymphoma and choose the appropriate treatment.
Core Tip: Primary colonic lymphoma is a very rare malignant tumor for which there is no standard treatment. We report a case of primary colonic T-cell lymphoma similar to a pedunculated polyp, and endoscopic submucosal dissection (ESD) was performed for diagnosis and treatment. ESD could be a feasible and economical choice for the treatment of colonic lymphomas. With the popularization of the concept of minimal invasiveness and the continuous development of minimally invasive technology, endoscopic surgery will have high application value for early gastrointestinal lesions.
- Citation: Sun YH, Lu SS, Fang Y, Xiong Z, Sun QY, Huang J. Rare primary colonic T cell lymphoma with curative resection by endoscopic submucosal dissection: A case report. World J Clin Cases 2024; 12(22): 5229-5235
- URL: https://www.wjgnet.com/2307-8960/full/v12/i22/5229.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v12.i22.5229
Approximately 40% of lymphomas involve the extranodal tract, the most common site of extranodal involvement being the gastrointestinal tract[1]. Primary colonic lymphoma is a very rare disease that can be histologically divided into Hodgkin's lymphoma and non-Hodgkin lymphoma. Among non-Hodgkin lymphomas, T-cell-lineage lymphomas account for only 13%-15%[2]. Of the distant organ metastases of colorectal malignancies, the liver and lungs are most commonly affected[3]. We report the case of a female patient with a primary colonic T-cell lymphoma-like pedunculated polyp who underwent endoscopic submucosal dissection (ESD). This may provide a new option for the treatment of colorectal T-cell lymphoma.
A 45-year-old woman was admitted to our hospital for a routine physical examination.
The patient had no obvious symptoms, such as abdominal pain, diarrhea, fever, or weight loss, and only came to the hospital for a routine physical examination.
The patient had undergone myomectomy 20 years ago and radical mastectomy for left breast cancer and cholecystectomy 10 years ago. She had a history of intestinal adhesions for 15 years. The patient recovered well after these surgeries, and there was no special history of other previous illnesses.
The patient denied any family history of malignant tumors.
On physical examination, the vital signs were as follows: Body temperature, 36.5 °C; blood pressure, 116/81 mmHg; heart rate, 81 bpm; and respiratory rate, 18 breaths/min. She had clear consciousness, no swelling of superficial lymph nodes, a normal-looking abdomen, visible old scars, and a flat and soft abdomen without tenderness.
No abnormalities were found on routine blood and urine analyses. Routine electrocardiogram revealed sinus rhythm, with an average of 79 bpm. Coagulation parameters were as follows: Activated partial thromboplastin time, 23.9 s; D-dimer, 5.73 mg/L; and Septin9 gene methylation negative. Serum levels of tumor markers were normal.
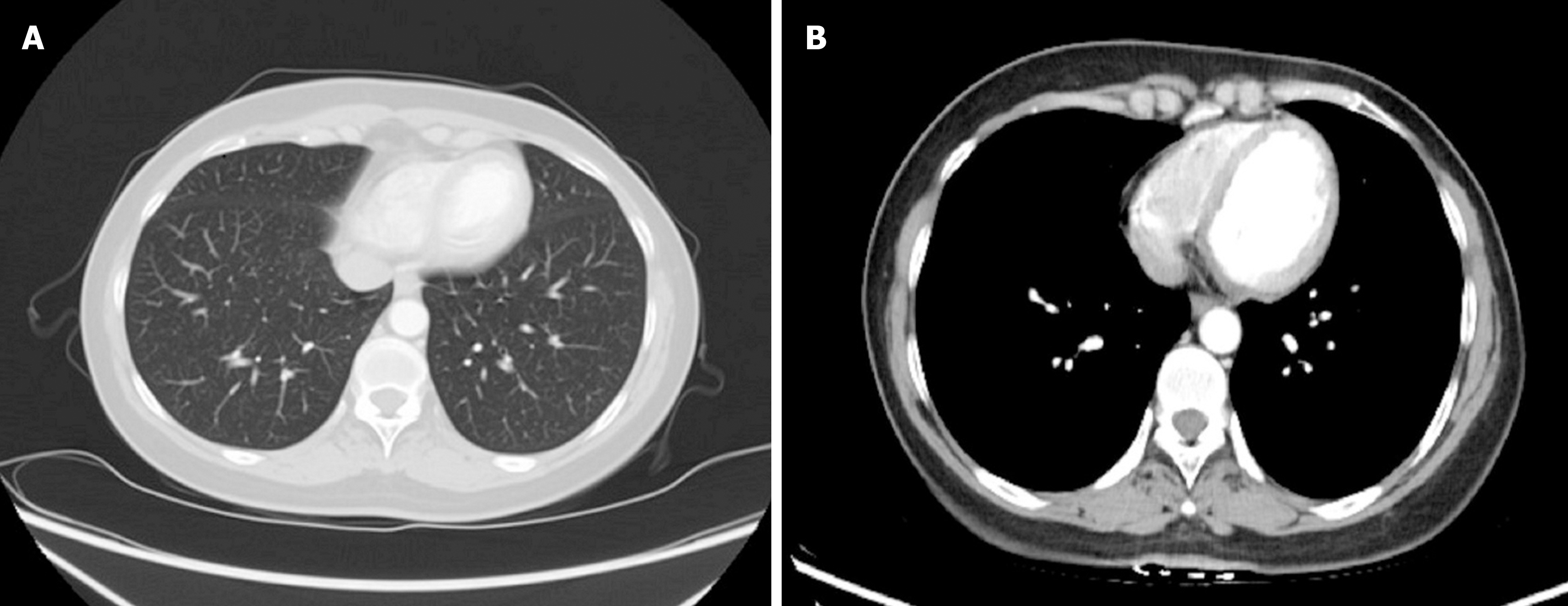
Chest-abdominal plain computed tomography (CT) showed small nodules in both lungs, without lung lymph node enlargement or visible masses or lymph nodes in the abdomen (Figure 1A and B). The patient later underwent bone marrow aspiration and positron emission tomography-CT (PET-CT), which did not reveal any significant abnormalities.
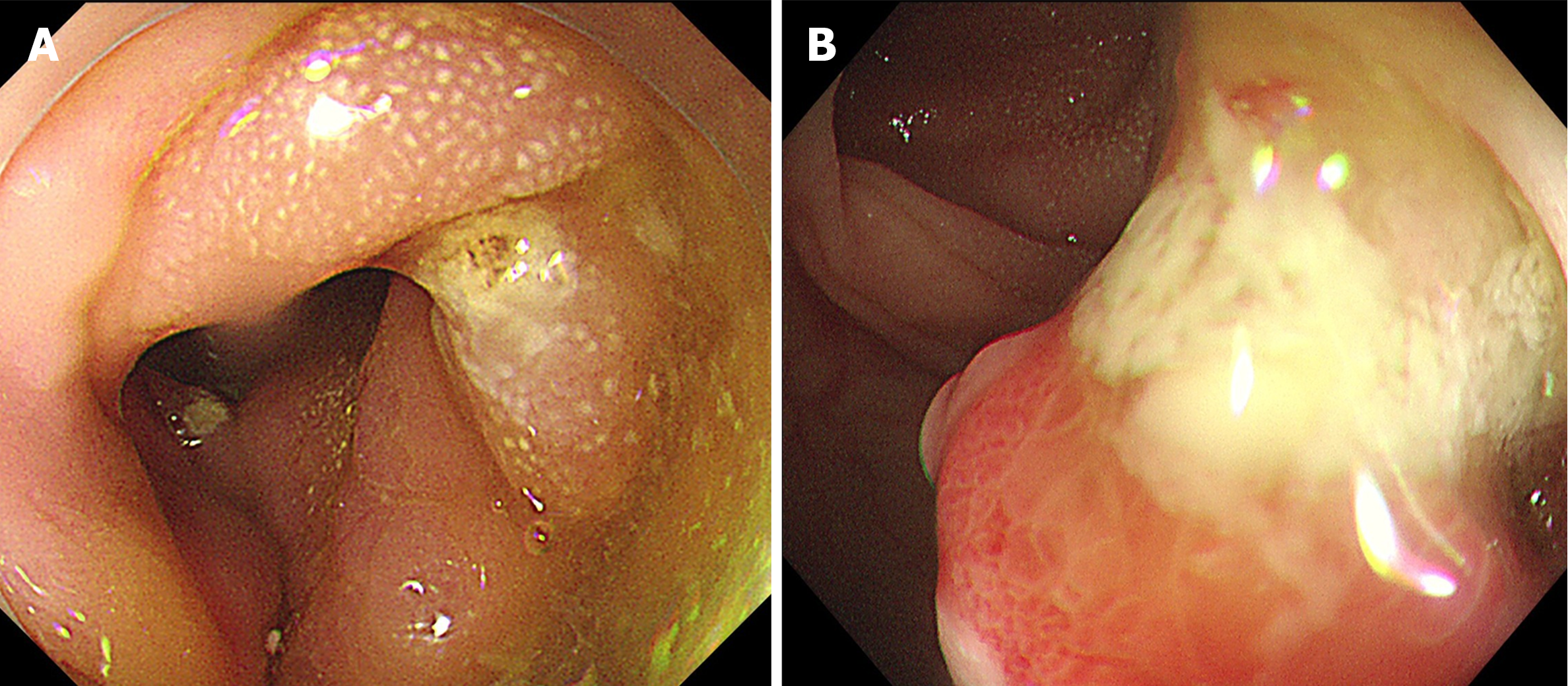
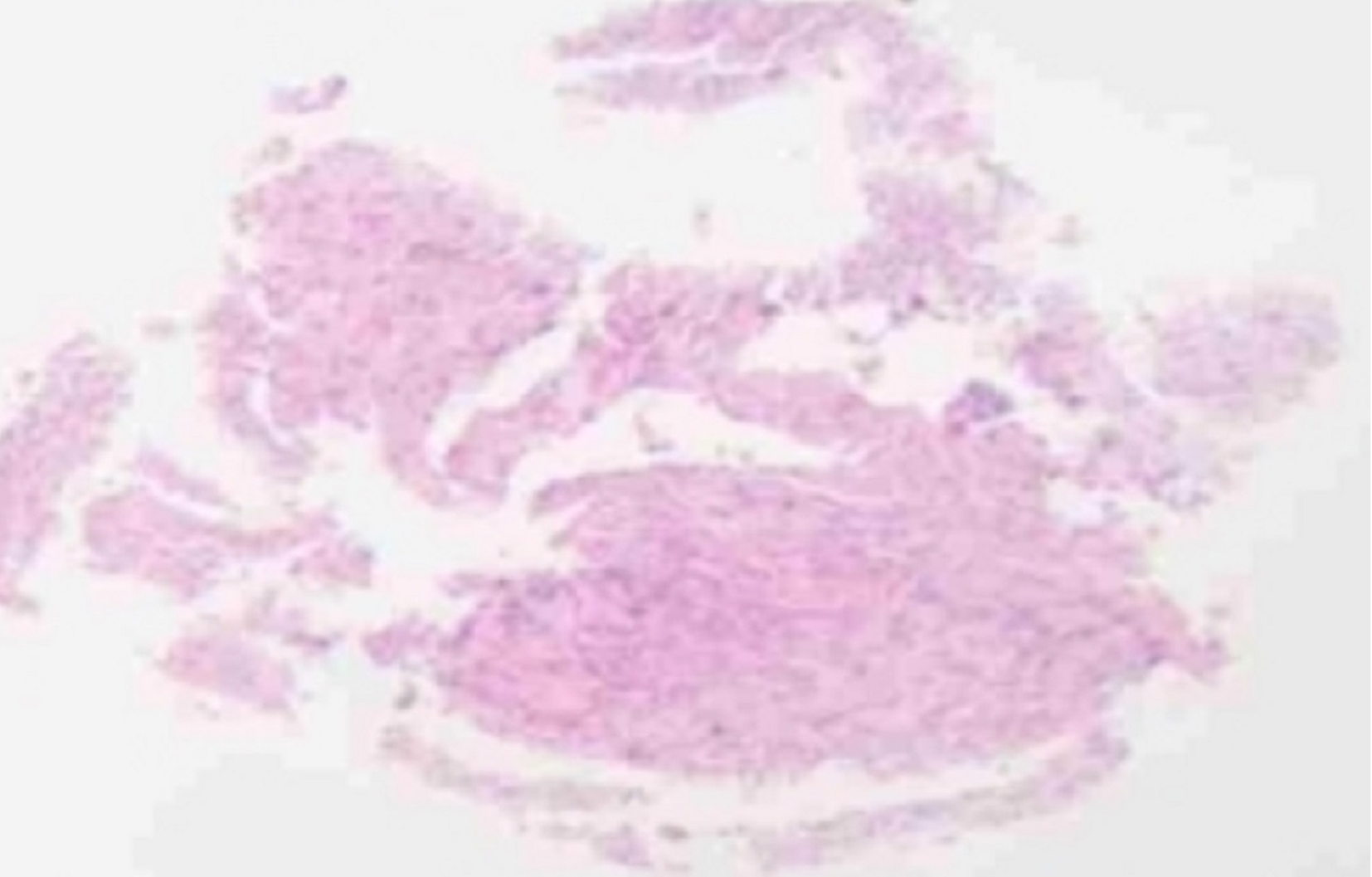
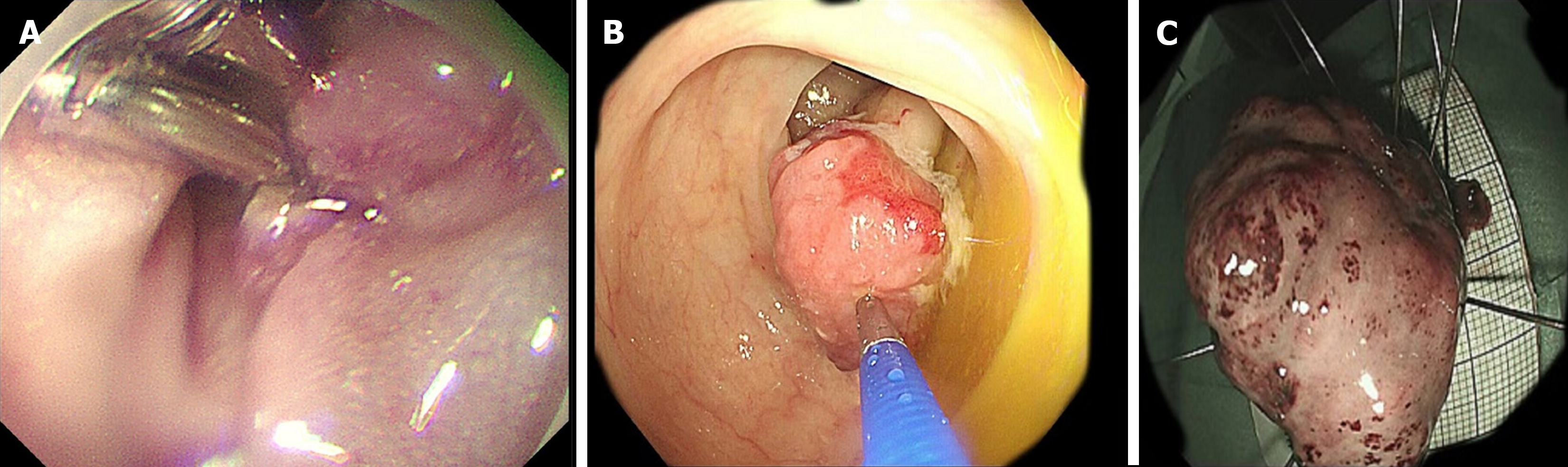
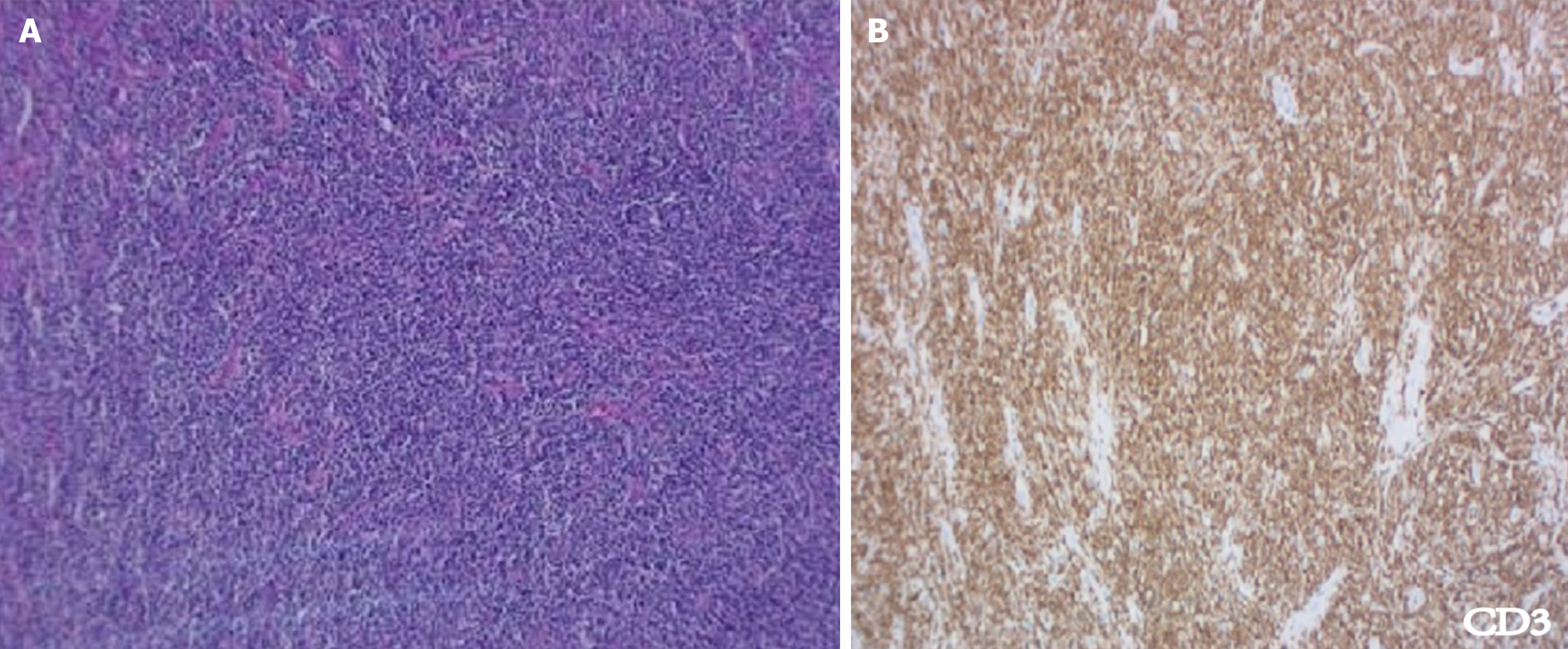
Gastroenteroscopy revealed a visible pedunculated polyp in the transverse colon, 1 cm in diameter and 5 cm in length, and local erosion on the surface (Figure 2A and B). The first impression was of a colon polyp with localized carcinoma. Histopathology of the colon mucosa showed a few heterogeneous nuclear cells in inflammatory granulation tissue, which were considered proliferating myofibroblasts and SMAs (+) based on immunohistochemistry (Figure 3A). We performed ESD for diagnosis and completely removed the lesion (Figure 4A-C). Histopathology revealed highly active proliferation of T-lymphocytes with atypical hyperplasia, infiltration into the mucosal layer (Figure 5A), and no involvement of the base cut margins. Immunohistochemistry detected CD20, CD3, CD5, CD2, CD7, CD4, CD8, and BCL-2 TIA (foci +); positivity for PD-1 and CXCL-13; and negativity for CKP, CD19, Pax-5, CD56, CD10, EBER, CD21, and CD23 (Figure 5B). After a multidisciplinary discussion between gastroenterology, pathology and hematology experts, the patient had no obvious clinical symptoms. Gastroenterology suggested the pedunculated polyp which was completely resected by ESD, and the histopathology suggested active T-cell proliferation, and immunohistochemistry suggested atypical proliferation of lymphocytes, which was suspected to be an inert intestinal T-cell lymphoma. TCR gene rearrangement showed a positive result in the clonality test for T-cell receptor genes, suggestive of clonal lymphocyte hyperplasia. PET-CT was further investigated to clarify the possible extent of the lesion, to further exclude the possibility of higher-grade T-cell lymphoma, and showed no abnormalities. Bone marrow aspiration and ultrasound of superficial lymph nodes in the hematology department showed no abnormalities.
The lesion was confirmed to be primary colonic T-cell lymphoma not otherwise specified by hematoxylin and eosin staining, immunohistochemical staining, and gene arrangement analysis.
The patient underwent ESD with complete debridement and no postoperative complications were observed.
ESD was performed for the lesion. Since T-cell lymphoma is a malignant tumor with a poorer prognosis than B-cell lymphoma and since our patient’s lesion was completely resected, a rational treatment and follow-up program was formulated after multidisciplinary discussion. Given the complete resection of the limited lesion and negative results of multiple investigations, the patient underwent repeat endoscopy and lymph node ultrasound at 6 and 9 months after ESD, which did not show any suspicious lesions. No additional treatment was considered for the time being. We advised the patient to undergo annual endoscopy as well as lymph node examinations annually. The patient has not experienced any tumor recurrence or metastasis.
Primary gastrointestinal T-cell lymphoma is a rare malignant tumor of the gastrointestinal tract. In terms of cellular origin, most lymphomas are B-cell lymphomas. T-cell lymphomas originating in the gastrointestinal tract are rare, especially if the primary lesion is located in the transverse colon[4]. The diagnostic difficulty and misdiagnosis rate of primary gastrointestinal lymphoma with atypical clinical symptoms are high, and it is usually confused with leather stomach or Crohn's disease. Some patients with gastrointestinal lymphoma are asymptomatic, like our patient, and others can have nonspecific symptoms, such as abdominal pain, bloating, weight loss, and changes in bowel habits[5].
Primary T-cell lymphomas encompass several entities, such as enteropathic T-cell lymphoma, monomorphic epitheliotropic intestinal T-cell lymphoma, and indolent T/natural killer cell lymphoproliferative disorders[3,4]. The diagnosis of colonic T-cell lymphoma was made by histopathology, immunohistochemistry, gene rearrangement analysis, and a negative history of intestinal disease. Based on the clinical characteristics of the patient as well as the pathology, we considered her lymphoma inert.
We report a case of primary gastrointestinal T-cell lymphoma originating from the transverse colon and a single lesion-like pedunculated polyp. Primary colonic T-cell lymphoma is a rare disease with diverse clinical and endoscopic presentations. The endoscopic findings are nonspecific, ranging from a single polypoid lesion to a subepithelial tumor type, an epithelial mass type, and an ileitis type[6]. In previous literature, lesions were macroscopically classified into the following types by colonoscopy: polypoid, ulcerative, multiple lymphomatous polyposis, diffuse, and mixed. Most macroscopic types are polypoid or ulcerative[7].
Currently, biopsy is the gold-standard diagnostic test and can be performed via endoscopic techniques. Histopathology is used to assess whether the lesion is localized to the mucosal level. To accurately identify and stage this diverse group of lymphomas, several procedures have been adopted, such as endoscopic biopsy, CT, magnetic resonance imaging, and PET-CT[8]. In this case, when we first performed gastroenteroscopy on the patient, we considered fibroblastic hyperplasia based on the colonoscopic multipoint biopsy.
ESD can aid in the diagnosis and complete debridement of the lesion for biopsy. Pathology revealed T-cell lymphoma located in the transverse colon. Compared with endoscopic biopsy, which has been used in previous studies, diagnostic ESD in this patient had the advantage of enabling diagnostic treatment and obtaining complete pathological specimens, which helped us confirm the depth of tumor infiltration and differentiation and evaluate patient prognosis[9].
The rarity of GI-TL and the lack of randomized controlled trials indicate that there is little consensus on the optimal therapy for the majority of GI-TLs. According to the literature, conventional chemotherapy remains the only option for treating pretreated T-cell lymphoma, and the R-CHOP chemotherapeutic protocol is the standard regimen for non-Hodgkin lymphoma[10]. Furthermore, chemotherapy after surgical resection may be an appropriate strategy for primary gastrointestinal natural killer/T-cell lymphoma[11]. Radiotherapy is usually a salvage therapy and is used less frequently[12]. For localized lesions, endoscopic resection could be prioritized to avoid unnecessary surgery or radiotherapy. When it is not possible to resect the lesion endoscopically, surgical resection may be considered as an alternative option.
There are few reports on the use of ESD for the treatment of colorectal T-cell lymphoma. In this case, we stripped the entire primary lesion intact by ESD, and the base and cutting edge were negative. We treated the lesion with standard care for early-stage cancers given that the lesion was confined to the transverse colon, with no metastasis or recurrence. Based on the discussion among the multidisciplinary medical team, no additional treatment was planned, and the patient was closely followed up. Endoscopy and lymph node ultrasound were performed at 6 and 9 mo after ESD, and no abnormalities were detected. We decided to perform annual endoscopic and lymph node ultrasound follow-up.
Based on the review of this case and the literature, when encountering primary gastrointestinal T-cell lymphoma in clinical practice, we should pay more attention to its clinical features, morphology, and immunohistochemistry. The etiology and pathogenesis of the disease are still unclear, and there is no consensus on its standard therapies. Treatment is chosen mainly based on its pathological characteristics, and appropriate treatment plans should be selected based on the condition of the lesion.
Previous research has indicated that surgical resection of localized colonic lymphoma provides better control of the conditions at the local level[13]. Local treatment is a reasonable approach. ESD seems to be a feasible and economical choice for the treatment of colonic lymphomas, on which little research has been done. However, the combination of ESD with another therapeutic regimen requires further clinical investigation[14]. Continuous monitoring is necessary, and chemotherapy and radiotherapy are recommended for patients who develop systemic conditions such as fever, gastrointestinal reactions, and worsening of their condition[15]. Due to the rarity of primary T-cell lymphomas, more evidence is needed to support ESD for the treatment of such tumors, and more data are urgently needed to provide better treatment strategies.
This patient presented with a primary colonic T lymphoma lesion similar to a pedunculated polyp, and ESD was performed for diagnosis and treatment. The patient will be followed up regularly. If recurrence or metastasis occurs, a combination of radiation and chemotherapy will be used.
We thank Dr. Zhou from the Department of Pathology for providing high-quality histopathological images.
| 1. | Nakamura S, Matsumoto T. Gastrointestinal lymphoma: recent advances in diagnosis and treatment. Digestion. 2013;87:182-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 66] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 2. | Nishimura MF, Nishimura Y, Nishikori A, Yoshino T, Sato Y. Primary Gastrointestinal T-Cell Lymphoma and Indolent Lymphoproliferative Disorders: Practical Diagnostic and Treatment Approaches. Cancers (Basel). 2021;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 3. | Bourmaf M, Motwani KK, McCool I, Singh Z, Kallen M, Koka M, Wong U. The Sigmoid Colon as an Unusual Primary Site of T Cell Lymphoma in a Patient Presenting With Brain Metastasis. ACG Case Rep J. 2023;10:e01172. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 4. | Okada M, Maeda K, Suzumiya J, Hagimoto T, Wakamatsu S, Ohshima K, Kanda M, Sonoda T, Sakamoto A, Tamura K. Primary colorectal T-cell lymphoma. J Gastroenterol. 2003;38:376-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 5. | Chen Y, Chen Y, Chen S, Wu L, Xu L, Lian G, Yang K, Li Y, Zeng L, Huang K. Primary Gastrointestinal Lymphoma: A Retrospective Multicenter Clinical Study of 415 Cases in Chinese Province of Guangdong and a Systematic Review Containing 5075 Chinese Patients. Medicine (Baltimore). 2015;94:e2119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 6. | Ishibashi H, Nimura S, Hirai F, Harada N, Iwasaki H, Kawauchi S, Oshiro Y, Matsuyama A, Nakamura S, Takamatsu Y, Yonemasu H, Shimokama T, Takeshita M. Endoscopic and clinicopathological characteristics of colorectal T/NK cell lymphoma. Diagn Pathol. 2020;15:128. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 7. | Yachida T, Matsuda T, Sakamoto T, Nakajima T, Kakugawa Y, Maeshima AM, Taniguchi H, Kushima R, Tobinai K, Kobara H, Masugata H, Masaki T, Saito Y. Endoscopic features of colorectal lymphoma according to histological type. JGH Open. 2022;6:257-262. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 8. | Tao Y, Nan Q, Lei Z, Miao YL, Niu JK. Rare primary rectal mucosa-associated lymphoid tissue lymphoma with curative resection by endoscopic submucosal dissection: A case report and review of literature. World J Clin Cases. 2022;10:7599-7608. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 1] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 9. | Park CH, Yang DH, Kim JW, Kim JH, Kim JH, Min YW, Lee SH, Bae JH, Chung H, Choi KD, Park JC, Lee H, Kwak MS, Kim B, Lee HJ, Lee HS, Choi M, Park DA, Lee JY, Byeon JS, Park CG, Cho JY, Lee ST, Chun HJ. Clinical Practice Guideline for Endoscopic Resection of Early Gastrointestinal Cancer. Clin Endosc. 2020;53:142-166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 108] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 10. | Inban P, Carredo CKC, Arslan F, Odoma VA, Okobia I, Adegbite A, Sharma S, Kc S, Zahdeh T, Rentiya ZS. Small Bowel Lymphoma Complicated With Ischemic Colitis: A Case Report. Cureus. 2023;15:e41792. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 11. | Hong H, Liang C, Huang H, Guo C, Tian Y, Liu T, Zhang M, Li X, Wang Z, Fang X, Lin J, Lin T. Surgical resection followed by chemotherapy may be an effective treatment strategy for primary gastrointestinal natural killer/T-cell lymphoma: a single center experience. Leuk Lymphoma. 2014;55:2649-2651. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 12. | Oka P, Sidhu R. Small bowel lymphoma: clinical update and challenges for the gastroenterologist. Curr Opin Gastroenterol. 2022;38:270-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 13. | Radman I, Kovacević-Metelko J, Aurer I, Nemet D, Zupancić-Salek S, Bogdanić V, Sertić D, Mrsić M, Pulanić R, Gasparović V, Labar B. Surgical resection in the treatment of primary gastrointestinal non-Hodgkin's lymphoma: retrospective study. Croat Med J. 2002;43:555-560. [PubMed] |
| 14. | Han J, Wang J, Xie HP. Gastric Mucosa-Associated Lymphoid Tissue Lymphomas Diagnosed by Jumbo Biopsy Using Endoscopic Submucosal Dissection: A Case Report. Front Med (Lausanne). 2021;8:668531. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 15. | Beaton C, Davies M, Beynon J. The management of primary small bowel and colon lymphoma--a review. Int J Colorectal Dis. 2012;27:555-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |