Published online May 26, 2024. doi: 10.12998/wjcc.v12.i15.2649
Revised: March 15, 2024
Accepted: April 9, 2024
Published online: May 26, 2024
Processing time: 87 Days and 18.6 Hours
Multiple primary carcinomas (MPCs) are defined as two or more independent primary cancers that occur simultaneously or sequentially in the same individual. Synchronous MPCs are rarer than solitary cancers or metachronous MPCs. Accurate diagnoses of synchronous MPCs and the choice of treatment are critical for successful outcomes in these cases.
A 64-year-old patient presented with dysphagia, without obvious cause. A diagnosis of synchronous esophageal squamous cell carcinoma and colon adenocarcinoma with liver metastasis was confirmed based on examination and laboratory results. After multi-disciplinary consultations, combination chemo
Successful cetuximab chemotherapy treatment provides a reference for the non-operative and homogeneous treatment of different pathological types of synchronous MCPs.
Core Tip: We report a case of a 64-year-old patient diagnosed with synchronous esophageal squamous cell carcinoma and colon adenocarcinoma with liver metastasis. After multi-disciplinary consultations, oxaliplatin + capecitabine chemotherapy and esophageal cancer radiotherapy were initiated. Based on genetic testing, treatment was switched to a regimen of leucovorin + fluorouracil + oxaliplatin and cetuximab for 8 cycles. Subsequently, capecitabine and bevacizumab were administered, and the tumor was stable at the most recent follow-up. This case demonstrates successful treatment of dual primary digestive tract tumors of differing pathological types using the same regimen, which supports non-surgical, simultaneous treatment for synchronous multiple primary cancers.
- Citation: Luo XX, Du YX, Zhang QQ, Zhang L, Zeng SY, Yu ZH, Shen P, Feng ZQ. Cetuximab combined with chemotherapy for simultaneous esophageal squamous cell carcinoma and colon adenocarcinoma: A case report. World J Clin Cases 2024; 12(15): 2649-2654
- URL: https://www.wjgnet.com/2307-8960/full/v12/i15/2649.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v12.i15.2649
Multiple primary carcinomas (MPCs), also known as multiple carcinomas, refer to two or more independent primary carcinomas occurring simultaneously or sequentially in the same individual. According to the time interval between occurrences, MPCs can be divided into metachronous MPCs or synchronous MPCs. The overall reported frequency of MPCs varies between 2.4%-17%[1]. The occurrence of MPCs may be related to factors such as the patient’s physical condition, lifestyle, environment, genetics, and treatment methods.
MPCs of the gastrointestinal tract have been treated by simultaneous surgical procedures[2]; however, few cases have been reported on the treatment of MPCs of different pathological types with non-operative regimens. Herein, we report a case of synchronous esophageal squamous cell carcinoma and colon adenocarcinoma with liver metastasis, successfully treated with a non-surgical regimen. This case may be used for guidance to improve the diagnosis and treatment of this disease.
A 64-year-old patient was admitted to the local gastroenterology department with a complaint of dysphagia, including difficulties with both eating and drinking.
In July 2022, the patient experienced worsening dysphagia, including difficulty swallowing food, and intermittent abdominal discomfort. These symptoms prompted the patient to seek medical attention.
In May 2022, the patient experienced dysphagia and mild difficulty defecating for approximately 1 month; he had no cough, intermittent abdominal discomfort, or diarrhea. These symptoms were found to have no obvious cause. The patient was not concerned and did not seek medical help.
The patient had no relevant family history.
On physical examination, no obvious abdominal mass or superficial lymph node swelling was detectable.
Prior to treatment, laboratory examinations revealed the following levels in peripheral blood samples: carcinoembryonic antigen, 5.17 ng/mL; carbohydrate antigen 125, 5.9 U/mL; carbohydrate antigen 19-9, 19.45 IU/mL; white blood cells, 4.8 × 109/L; neutrophils, 3.3 × 109/L; red blood cells, 4.07 × 1012/L; hemoglobin, 131 g/L. Routine urine, urine sediment, and routine fecal examinations, as well as occult blood testing, blood biochemistry, immune indicators, and infection indices showed no significant abnormalities.
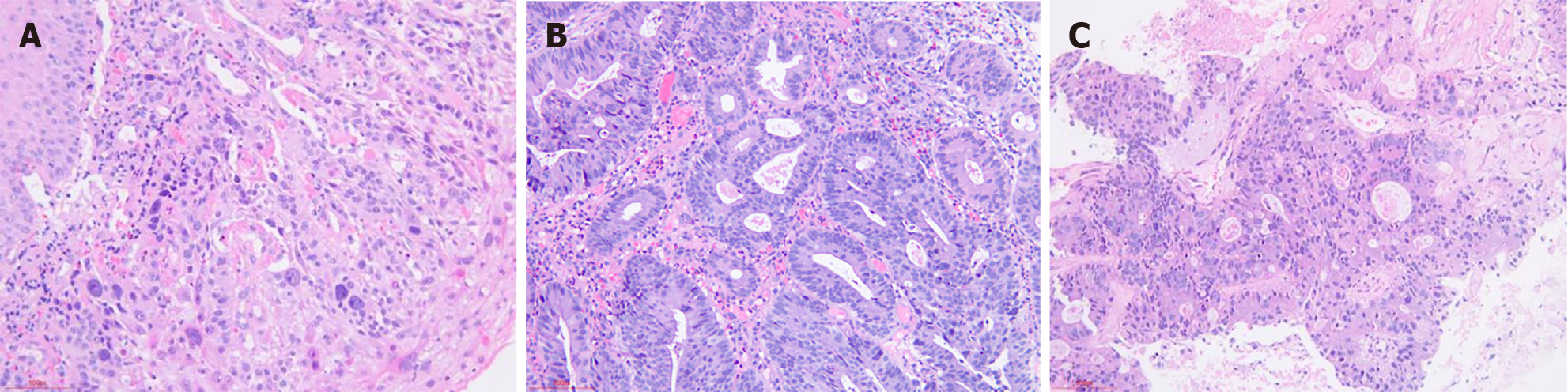
Gastroscopy and colonoscopy examinations were performed. Gastroscopic pathology (Figure 1A) revealed chronic moderate superficial gastritis (active), Helicobacter pylori (+), and squamous cell carcinoma (esophageal mucosa). Colonoscopic pathology (Figure 1B) revealed adenocarcinoma in the sigmoid colon, 60 cm from the anus. Immunohistochemistry was performed on the colon samples, with the following results: succinate dehydrogenase complex iron sulfur subunit B (+), S-100 (-), smooth muscle actin (+), desmin (+), CD34 (+), CD117 (-), Dog-1 (-), P53 (wildtype), Ki-67 (+, 2%), and submucosal spindle cell tumor formation. These findings were consistent with leiomyoma. Liver biopsy (Figure 1C) revealed adenocarcinoma, and this was considered to be metastatic intestinal adenocarcinoma.
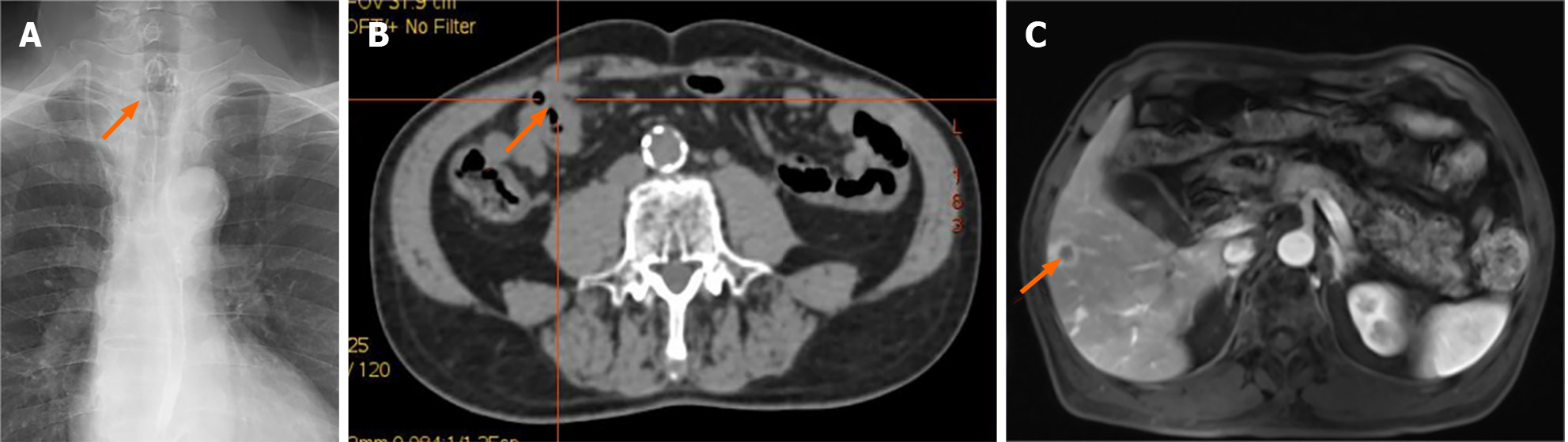
Esophageal meglumine diatrizoate angiography showed that the esophageal wall was locally thickened and stiffened in the anterior thoracic segment (Figure 2A). Positron emission tomography detected neoplasia in the anterior thoracic sigmoid colon 20 cm from the anus, occupying half of the intestinal cavity (Figure 2B). Multiple liver metastases were observed via magnetic resonance imaging (Figure 2C).
The patient was ultimately diagnosed with esophageal squamous cell carcinoma, colon adenocarcinoma with liver metastasis, and rectal leiomyoma.
After multidisciplinary consultations, on July 23, 2022, the patient was started on a 3-wk chemotherapy cycle with 212 mg oxaliplatin on day 1 and 1.5 g capecitabine twice daily on days 1-14. While waiting for the genetic testing results from the intestinal biopsy, radiotherapy was administered for esophageal carcinoma on July 29, 2022.
Reference genetic testing indicated that the patient was wildtype for Kirsten ras oncogene homolog (KRAS), neuroblastoma ras oncogene homolog (NRAS), and v-Raf murine sarcoma viral oncogene homolog B (BRAF), with fibroblast growth factor receptor 1 gene amplification and microsatellite stability (MSS). Owing to the MSS, immunotherapy was not considered. However, based on these results, treatment was switched to leucovorin + fluorouracil + oxaliplatin (mFOLFOX) and cetuximab for 8 cycles, from August 12, 2022, to December 17, 2022. The patient developed hand-foot syndrome (grade III), bone marrow depression (grade I), and skin rash (grade I) during chemotherapy.
After 9 cycles of chemotherapy, the patient was administered a temporary anti-infective treatment for coronavirus 2019 pneumonia. For convenience during this time and considering the curative effect as well as patient preference, the chemotherapy regimen was suspended. Thereafter, the patient was switched to a capecitabine oral cycle; capecitabine + bevacizumab continues to be administered at this time.
In December 2023, chest computed tomography showed slight thickening of the esophageal wall in the thoracic segment, which was improved compared to the anterior segment. At the same follow-up, colonoscopy revealed that the sigmoid colon lesion was 20 cm from the anus and that the neoplasia occupied a quarter of the intestinal cavity, demonstrating a reduction in size from pre-treatment imaging. Liver magnetic resonance imaging revealed complete clinical response of the liver metastases. Overall outcome assessment indicated a partial response (PR). As of January 2024, the patient’s condition remained stable.
Currently, the diagnosis of MPCs is based on the revised criteria of Warren and Gates[3]: (1) Malignancy must be confirmed pathologically for each tumor; (2) Each tumor must have its own unique pathological morphology, exist independently, and have no relationship to other tumors; (3) The tumors must occur in different sites or organs; and (4) Each tumor must be excluded as a metastatic focus from the other tumors. The interval between the occurrence of MPCs defines them as either synchronous (occurring within 6 months of each other) or metachronous (occurring more than 6 months apart).
The treatment of MPCs is similar to that of single primary carcinomas and significantly different from that of metastatic and recurrent carcinomas. In addition, the differences in pathological types and synchronicity influence the choice of treatment; therefore, accurate and timely diagnosis is crucial. Differential diagnosis of MPCs relies heavily on histopathology screening. Our patient presented with squamous cell carcinoma of the esophagus and sigmoid colon adenocarcinoma with metastatic adenocarcinoma of the liver. The pathological types of the two tumors were significantly different; moreover, the different primary sites and the occurrence of metastasis made it challenging to choose a treatment plan for simultaneous treatment. Only a few cases of concurrent malignant tumors of different pathological types have been successfully treated.
Colon cancer with liver metastasis can be treated with mFOLFOX and cetuximab based on the pathological type and results of genetic testing. Guidelines also suggest that mFOLFOX can be used to treat esophageal cancer, with successful cases reported in the literature. Therefore, we treated this case with an mFOLFOX and cetuximab regimen. Reference genetic testing indicated the patient was KRAS/NRAS/BRAF wild-type. From August 12, 2022, to December 17, 2022, eight cycles of mFOLFOX + cetuximab were administered, with PR achieved; after eight cycles, PR was re-assessed. At follow-up in January 2024, the patient’s condition remained stable.
MPCs of the first and second primary lesions are common in the digestive, respiratory, and urinary systems[4]. The digestive system is the most common, accounting for 48.7% of first and 44.0% of second primary lesions. Furthermore, the three most common sites for first and second primary lesions were found to be in the digestive system: the colorectum, stomach, and esophagus. Notably, studies[5-7] have suggested that patients with synchronous MPCs have a worse prognosis and lower overall survival than patients with metachronous MPCs. Therefore, to improve their prognosis, patients with MPCs should undergo an active treatment program promptly after diagnosis.
The primary treatments for MPCs are chemotherapy, radiotherapy, and surgery; however, a unified standard treatment strategy has not yet been established, and related research is insufficient. In the diagnosis and treatment of MPC, factors such as clinical stage and pathological type should be considered, and radical treatment should be adopted to the fullest extent, especially for localized tumors[8]. When a tumor cannot be resected, radiotherapy, chemotherapy, targeted therapy, and immunotherapy should be considered. Radiotherapy and chemotherapy, which have comprehensive anti-tumor activity, have the ability to concurrently control primary and metastatic lesions. Patients with locally advanced esophageal squamous cell carcinoma have been shown to be sensitive to chemoradiotherapy, with a high complete tumor regression rate and local tumor control[9].
The choice of drug therapy was based on the 2022 edition of the National Comprehensive Cancer Network (NCCN) guidelines[10], which recommends FOLFOX, CAPEOX, FOLFIRI or mFOLFIRINOX (leucovorin + fluorouracil + oxaliplatin, capecitabine + oxaliplatin, leucovorin + fluorouracil + irinotecan, and leucovorin + fluorouracil + irinotecan + oxaliplatin, respectively) as first-line treatment for metastatic colon cancer. If the patient is KRAS/NRAS/BRAF wildtype, they can be treated with chemotherapy + cetuximab. If mutations are found in the KRAS/NRAS genes, chemotherapy + bevacizumab can be administered. The first choice for patients with mismatch repair deficient tumors or high microsatellite instability is pembrolizumab.
The 2022 NCCN guidelines[10] recommend first-line treatment for locally advanced esophageal cancer, which is often selected because oxaliplatin is less toxic than cisplatin. Fluorouracil is also a commonly used drug for this cancer type. Chemotherapy + trastuzumab is recommended if the tumor is positive for human epidermal growth factor receptor 2 (HER2) overexpression and chemotherapy + immunotherapy if HER2 overexpression is negative.
For patients who have achieved PR after a certain number of cycles of chemotherapy, the existing maintenance treatment modes primarily include one of the following, based on the cumulative toxicity of the drugs: partial drug maintenance and dressing change maintenance, as in the original plan; chemotherapy termination; or targeted treatment.
The optimal radiotherapy dose for esophageal carcinoma remains controversial. In China, the conventional radiation dose for esophageal squamous cell carcinoma is 60 Gy, and its curative effect and long-term survival rate are consistent with relevant international studies[11]. The NCCN guidelines[10] consider 50.4 Gy as a safe and reliable dose for concurrent chemoradiotherapy. Recent studies[12] have demonstrated that an increased dose of radiotherapy combined with chemotherapy can improve the survival of patients with esophageal cancer. Therefore, 60 Gy was selected for treatment in this case.
In summary, imaging, endoscopic, and pathological examinations should be combined for the diagnosis of MPCs to differentiate primary malignant tumors from metastatic ones as accurately as possible. For multi-focal tumors, determining whether they are metastases is necessary. For the treatment of MPCs, the pathological characteristics of each tumor and the genetic status should be considered when choosing the individualized treatment scheme with the highest benefit for the patient. Surgery is the standard treatment for an early-stage tumor, with supplemental chemotherapy and biological therapy. The survival of patients with concurrent late metastasis of MPCs can be significantly prolonged by targeted chemotherapy or immunotherapy. This report describes the successful treatment of a case of simultaneous esophageal squamous cell carcinoma and colon adenocarcinoma with liver metastases. The characteristics and treatment plan of this case can be analyzed and used as a reference for the clinical treatment of similar MPCs.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Medicine, research and experimental
Country/Territory of origin: China
Peer-review report’s classification
Scientific Quality: Grade C, Grade D
Novelty: Grade C, Grade C
Creativity or Innovation: Grade C, Grade C
Scientific Significance: Grade C, Grade C
P-Reviewer: Adam CA, Romania S-Editor: Zheng XM L-Editor: A P-Editor: Yu HG
| 1. | Copur MS, Manapuram S. Multiple Primary Tumors Over a Lifetime. Oncology (Williston Park). 2019;33:629384. [PubMed] |
| 2. | Marano L. Dual primary gastric and colorectal cancer: A complex challenge in surgical oncology. World J Gastrointest Oncol. 2023;15:2049-2052. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 2] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 3. | Warren S, Gates O. Multiple primary malignant tumors: A survey of the literature and statistical study. Am J Cancer. 1932;16:1358-1414. |
| 4. | Cai YL. The clinical characteristics and prognosis of 234 multiple primary cancer patients. Ph.D. Dissertation, College of Medicine, Zhejiang University. 2018. Available from: https://kns.cnki.net/kcms2/article/abstract?v=Dl4MPbsAiWcWfY1btBpXwon0GWs-VxBEj_37ZWm1q_t3uH8sc0Wf3CRY5P8ml1XcPyCEDW2us9-bcEm85ynmrOOMQ8D7wZNipvgiEuAToM0SxI3-eDAiVH1yGarm0vZiTaoUWQn7XLQ=&uniplatform=NZKPT&flag=copy. |
| 5. | Natsugoe S, Matsumoto M, Okumura H, Ishigami S, Uenosono Y, Owaki T, Takao S, Aikou T. Multiple primary carcinomas with esophageal squamous cell cancer: clinicopathologic outcome. World J Surg. 2005;29:46-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 45] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 6. | Baba Y, Yoshida N, Kinoshita K, Iwatsuki M, Yamashita YI, Chikamoto A, Watanabe M, Baba H. Clinical and Prognostic Features of Patients With Esophageal Cancer and Multiple Primary Cancers: A Retrospective Single-institution Study. Ann Surg. 2018;267:478-483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 84] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 7. | Su XY, Yan Y, Jiang Q, Xia JH. [Overall survival analysis of double primary cancer patients with esophageal cancer as the first primary cancer]. Zhonghua Xiaohuabing Yu Yingxiang Zazhi. 2021;11:121-125. |
| 8. | Vogt A, Schmid S, Heinimann K, Frick H, Herrmann C, Cerny T, Omlin A. Multiple primary tumours: challenges and approaches, a review. ESMO Open. 2017;2:e000172. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 183] [Cited by in RCA: 369] [Article Influence: 46.1] [Reference Citation Analysis (1)] |
| 9. | Kitagawa Y, Uno T, Oyama T, Kato K, Kato H, Kawakubo H, Kawamura O, Kusano M, Kuwano H, Takeuchi H, Toh Y, Doki Y, Naomoto Y, Nemoto K, Booka E, Matsubara H, Miyazaki T, Muto M, Yanagisawa A, Yoshida M. Esophageal cancer practice guidelines 2017 edited by the Japan Esophageal Society: part 1. Esophagus. 2019;16:1-24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 377] [Cited by in RCA: 390] [Article Influence: 65.0] [Reference Citation Analysis (0)] |
| 10. | National Comprehensive Cancer Network. National Comprehensive Cancer Network guidelines: Colon cancer. Version 1. 2022. |
| 11. | Cheng G, Jiang XD, Zhang HP, Yang P, Liu H, Gu KS. [Clinical observation of 54 cases of non-surgical esophageal cancer treated with capecitabine rhythm chemotherapy combined with concurrent dose modulated radiation therapy]. Shandong Yiyao. 2017;57:87-89. |
| 12. | Cheng XY, Wu H, Zhang RR, Sun XM, Yan ZM, Liu X, Chen YS. [Prognostic analysis of 307 cases of stage II-III esophageal cancer treated with concurrent radiotherapy and chemotherapy]. Zhonghua Fangshe Zhongliuxue Zazhi. 2018;27:140-144. |