Published online Mar 26, 2023. doi: 10.12998/wjcc.v11.i9.2051
Peer-review started: November 27, 2022
First decision: January 12, 2023
Revised: January 15, 2023
Accepted: March 1, 2023
Article in press: March 1, 2023
Published online: March 26, 2023
Processing time: 109 Days and 22.9 Hours
Bacille Calmette-Guérin (BCG) instillation is recommended in patients with non-muscle-invasive bladder cancer who have intermediate-risk and high-risk tumors. However, granulomatous prostatitis is a rare complication induced by BCG instillation, which can easily be misdiagnosed as prostate cancer. Here, we report a case of granulomatous prostatitis that resembled prostate cancer.
A 64-year-old Chinese man with bladder cancer received BCG instillation. Three days later, he stopped BCG instillation and received anti-infective therapy due to the urinary tract infection. Three months after BCG restart, he had rising total prostate-specific antigen (PSA) (9.14 ng/mL) and decreasing free PSA/total PSA (0.09). T2-weighted images of magnetic resonance imaging (MRI) showed a 28 mm × 20 mm diffuse low signal abnormality in the right peripheral zone, which was markedly hyperintense on high b-value diffusion-weighted MRI and hypointense on apparent diffusion coefficient map images. Considering Prostate Imaging Reporting and Data System score of 5 and possibility of prostate cancer, a prostate biopsy was conducted. Histopathology showed typical features of granulomatous prostatitis. The nucleic acid test for tuberculosis was positive. He was finally diagnosed with BCG-induced granulomatous prostatitis. Thereafter, he stopped BCG instillation and received anti-tuberculosis treatment. During 10 mo follow-up, he had no evidence of tumor recurrence or symptoms of tuberculosis.
Temporarily elevated PSA and high followed by low signal abnormality on diffusion-weighted MRI are important indicators of BCG-induced granulomatous prostatitis.
Core Tip: Granulomatous prostatitis is a rare complication of BCG instillation, which can easily be misdiagnosed as prostate cancer. Here, we report a 64-year-old Chinese man with BCG-induced granulomatous prostatitis that resembles prostate cancer. Although histopathology remains the gold standard to accurately differentiate between the two diagnoses, some clues such as temporarily elevated PSA levels and a high signal followed by a low-signal abnormality on high b-value diffusion-weighted MRI are important indicators of BCG-induced granulomatous prostatitis.
- Citation: Yao Y, Ji JJ, Wang HY, Sun LJ, Zhang GM. Granulomatous prostatitis after bacille Calmette-Guérin instillation resembles prostate carcinoma: A case report and review of the literature. World J Clin Cases 2023; 11(9): 2051-2059
- URL: https://www.wjgnet.com/2307-8960/full/v11/i9/2051.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v11.i9.2051
Bladder cancer is the tenth most common malignancy worldwide and the sixth most common malignancy in men[1]. Approximately 75% of bladder cancer patients present with a disease confined to the mucosa or submucosa, which is defined as non-muscleinvasive bladder cancer (NMIBC)[2]. Compared with muscleinvasive bladder cancer, NMIBC is associated with better long-term survival and a lower risk of cancerspecific mortality[3,4]. Although transurethral resection of a bladder tumor (TURBT) can completely eradicate NMIBC, the high variability in the 3-month recurrence rate indicates that TURBT alone is inadequate[5]. Therefore, TURBT followed by intravesical chemotherapy or intravesical immunotherapy with bacille Calmette-Guérin (BCG) is recommended as a standard treatment for NMIBC[6].
Mycobacterium BCG, the sole tuberculosis vaccine that was first administered to a newborn baby in 1921, has saved tens of millions of lives over the past century[7]. BCG generates immunity and, therefore, has antitumor activity against tumors such as NMIBC[8]. BCG after TURBT was confirmed to be superior to TURBT plus chemotherapy or TURBT alone for preventing a recurrence of NMIBC in several recent meta-analyses[9-12]. A Cochrane systematic review also showed that mitomycin intravesical chemotherapy was less effective than BCG instillation in reducing the recurrence rate of NMIBC[13]. Therefore, intravesical BCG after TURBT is recommended in NMIBC patients with intermediate-risk and high-risk tumors.
However, BCG instillation is associated with more local and systemic side effects than intravesical chemotherapy, which may influence the treatment period[13,14]. Most patients only show mild local complications such as cystitis, urination frequency, and macroscopic hematuria[14]. The incidence of serious side effects is below 5% and nearly all cases can be treated effectively[15]. Only 1% of patients had a BCG infection in a registry-based cohort analysis[16]. One study that included BCG infection cases indicated that the top four sites of infection were the lungs, vascular tissue, the liver, and osteoarticular tissue[17]. Several cases of BCG infection after BCG instillation have been reported recently[18-21].
Compared with sites of infection such as the lungs and liver, genitourinary BCG infection is not common. Granulomatous prostatitis is a rare complication induced by BCG instillation and the exact incidence is unknown. Besides specific granulomatous prostatitis, it may be secondary to infections, surgery, associated to malacoplakia or to systemic granulomatous diseases such as sarcoidosis and Wegener’s granulomatosis[22,23]. Several cases have reported that granulomatous prostatitis presents as increased serum levels of prostate-specific antigen (PSA) and a node with an abnormal signal detected by multiparametric prostate magnetic resonance imaging (MP-MRI). Furthermore, previous cases involved nodular or diffusely firm enlargement on digital rectal examination, which was suggestive of prostate cancer and only resulted in a diagnosis of granulomatous prostatitis after prostate biopsy[24-27]. Here, we present a case of granulomatous prostatitis induced by BCG instillation and describe how to differentiate between granulomatous prostatitis induced by BCG instillation and prostate cancer.
A 64-year-old Chinese man with weekly BCG instillation visited our center in January 2022 for routinely examination of bladder cancer complaining of rising total PSA level (9.14 ng/mL) and decreasing free PSA/total PSA (0.09) with no symptom.
The patient was diagnosed with high-grade NMIBC in March 2020 and scheduled for gemcitabine intravesical chemotherapy. Owing to a pathologically T1G3 bladder carcinoma on histopathology, TURBT was repeated 3 mo later. Cystoscopy showed necrosis attached to the surface of the scar, and pathology suggested inflammatory necrosis with no tumor. Two months after the second TURBT, weekly BCG instillations were recommended to replace the gemcitabine intravesical chemotherapy. Three days after the first BCG instillation, the patient experienced fever up to 39 °C, gross hematuria, lower abdomen pain, and perineal pain. The physical examination and scrotal B-ultrasound indicated left hydrocele testis and epididymitis. Routine laboratory blood results indicated that total leukocyte counts were elevated to 18.25 × 109/L with increasing neutrophil granulocyte count (10.62 × 109/L) and decreasing hemoglobin levels (114 g/L). In addition, urinary testing revealed pyuria, hematuria, and bacteriuria. The patient was diagnosed with a urinary tract infection and received piperacillin and tazobactam as anti-infective therapy for 6 d until his temperature returned to normal. His urinary testing also became normal 1 wk later. He stopped BCG instillation and restarted gemcitabine intravesical chemotherapy. Following the second TURBT, cystoscopies and urinary B-ultrasounds were performed every 3 mo. The results showed no evidence of tumor recurrence. Sixteen months after the second TURBT, BCG intravesical immunotherapy was restarted.
The patient had no history of tuberculosis.
The patient and his family had no history of prostate cancer.
Digital rectal examination revealed a moderately enlarged, non-painful prostate gland with shallow central sulcus and normal density.
Laboratory tests revealed a rising serum total PSA level (9.14 ng/mL) and a decreasing serum ratio of free PSA/total PSA (0.09).
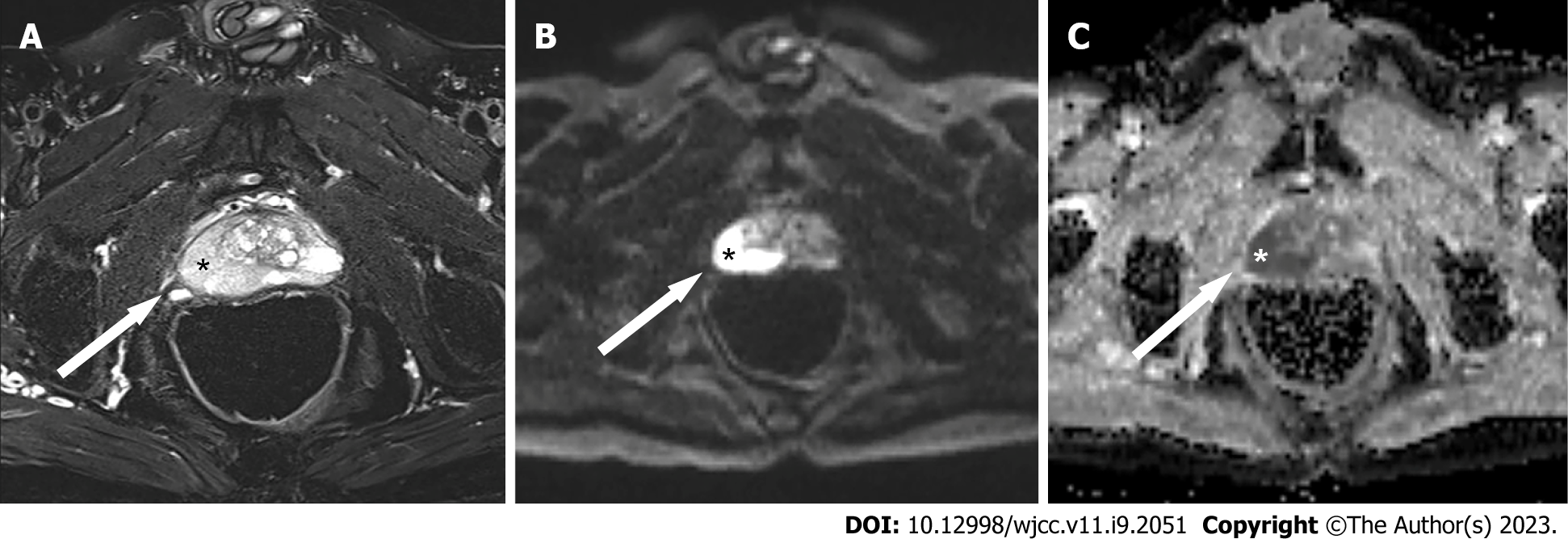
MP-MRI was performed. Axial T2-weighted images showed a 28 mm × 20 mm low-signal, diffuse abnormality in the right peripheral zone (Figure 1A). On diffusion-weighted MRI with a b-value at 1500, the lesion was seen as a markedly hyperintense focus (Figure 1B), which was consistent with a lesion with a Prostate Imaging Reporting and Data System (PI-RADS) score of 5, with hypointense signal on an apparent diffusion coefficient map image (Figure 1C).
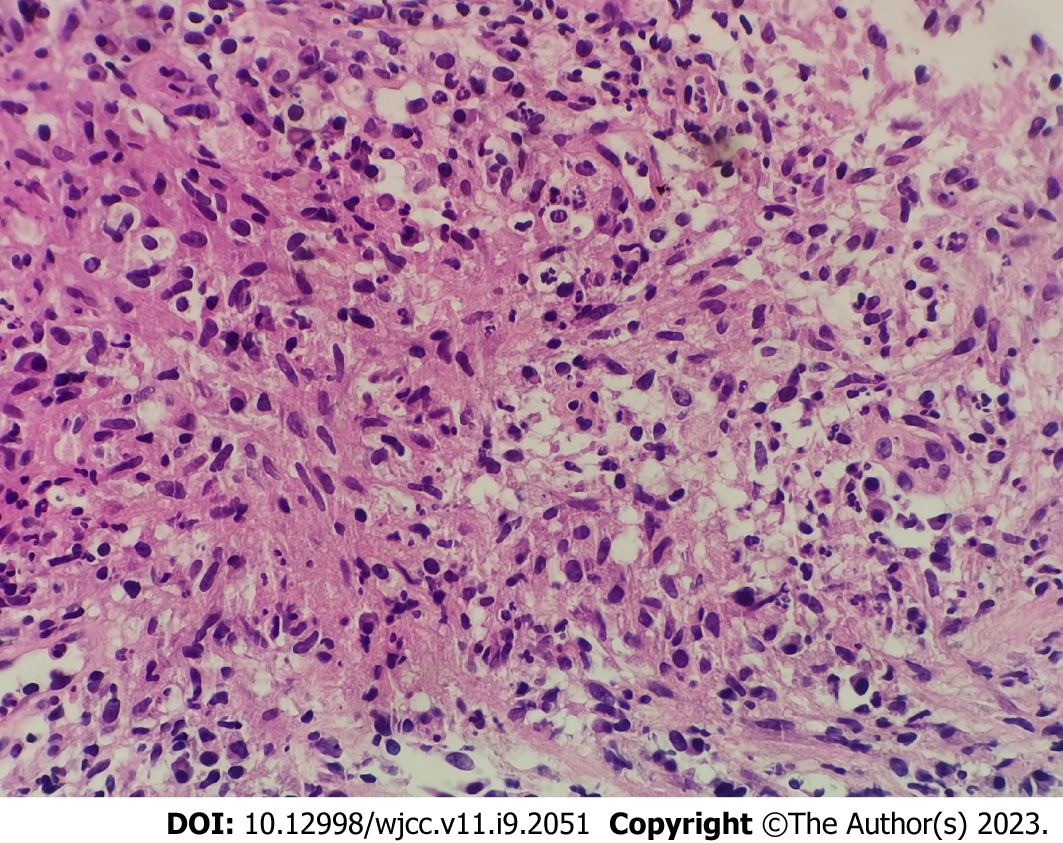
Considering the increasing PSA results and abnormal focus in MP-MRI, the patient was primarily diagnosed with prostate cancer. A transperineal prostate needle biopsy was conducted. Levofloxacin was used as antibiotic prophylaxis. However, the histopathological findings showed benign prostate tissue with typical features of granulomatous prostatitis with multinucleated giant cells, epithelioid cells, fibroblasts, and infiltration lymphocytes (Figure 2). A nucleic acid test of tuberculosis was subsequently performed and the result was positive (Ct = 37.18).
Based on the patient’s medical history, he was finally diagnosed with BCG-induced granulomatous prostatitis.
Thereafter, he received isoniazid, rifapentine, levofloxacin, and ethambutol as anti-tuberculosis treatment. The BCG instillation was stopped and replaced by epirubicin intravesical chemotherapy.
During a follow-up visit 10 mo after prostate biopsy, the patient had no evidence of tumor recurrence or symptoms of tuberculosis.
BCG instillation is used as standard immunotherapy in bladder cancer. As for complications, a tuberculosis infection in the urinary system is not common, as infection mostly occurs in the parenchymatous organs such as the testis, penis, kidneys, and prostate[17]. In recent articles, granulomatous prostatitis has been reported to account for 3.5% of BCG infections[28], and occurs in 1.06% of all bladder cancer patients who receive BCG instillation[29]. Most cases are asymptomatic and easily misdiagnosed as prostate cancer. Therefore, we present a case of granulomatous prostatitis induced by BCG instillations in order to investigate useful strategies for the differential diagnosis of the condition.
In two recent cases, elevated PSA levels ranging from 6.1 ng/mL to 8.4 ng/mL in patients with granulomatous prostatitis after BCG instillation[24,30]. In our patient, increasing total PSA levels and a decreasing ratio of free PSA/total PSA was found, which complicated the differentiation between prostate cancer and granulomatous prostatitis. Wang et al[31] discovered that half of all patients with BCG-induced granulomatous prostatitis had elevated PSA, while the ratio of free PSA/total PSA decreased to less than 0.16 in all patients. Although PSA has increased during immunotherapy for bladder cancer in some studies, the PSA elevation has been self-limited, and only PSA elevations for over 3 to 12 mo have led to recommendations for prostate biopsy[32,33]. Another study also reported that 40% of bladder cancer patients had elevated PSA after intravesical BCG therapy, but the PSA reverted to normal within 3 mo[34]. It seems that prostate biopsy should be delayed in these patients while PSA is monitored. However, two patients whose PSA level was elevated after BCG instillations were pathologically diagnosed with both granulomatous prostatitis and prostate cancer [26,27]. In an epidemiological study, concomitant prostate cancer was reported in 35.9% of patients with granulomatous prostatitis[17]. In another article, prostate cancer detection after intravesical BCG occurred frequently in patients with elevated PSA and a bladder tumor located far from the bladder neck[35]. These findings together suggest the need to develop patterns or curves of PSA levels after BCG instillation that could be used to distinguish between granulomatous prostatitis and prostate cancer. This strategy could reduce the frequency of unnecessary biopsies.
MP-MRI is widely used in the diagnosis of prostate cancer because it has good sensitivity for the localization and detection of over International Society of Urological Pathology grade 2 carcinomas[36-38]. With the inability to differentiate between granulomatous prostatitis and prostate cancer on transrectal ultrasound[25], specific characteristics of BCG-induced granulomatous prostatitis on MP-MRI have been proposed recently. In the case patient, an abnormal focus showed low signal in T2-weighted images and an apparent diffusion coefficient in the peripheral zone, which has been reported previously[24,26,27,31,39]. These characteristics could occur because the histology of granulomatous prostatitis includes stromal infiltration of chronic inflammatory cells and extracellular fluid surrounding the prostatic cells. However, both prostate cancer and granulomatous prostatitis present as destruction of the prostate gland with weakened water diffusion capability, and this is visualized as lower apparent diffusion coefficient values. The decreased signal intensity in T2-weighted images is also caused by the decreased water content present in both prostate cancer and granulomatous prostatitis[39]. Moreover, both BCG-related granulomatous prostatitis and prostate cancer mostly occurs in the peripheral zone of the prostate, which creates challenges in diagnosis[40,41].
On high b-value diffusion-weighted MRI, the lesion of the case patient had a markedly hyperintense focus with a PI-RADS version 2 score of 5. Several other studies support our finding of a PI-RADS version 2 score of 5[24,31,41]. However, Gottlieb et al[39] reported five patients with granulomatous prostatitis whose abnormalities on high b-value diffusion-weighted MRI were of low signal. Matsushima et al[42] described computed-tomography-detected asymptomatic abnormalities in the prostate after BCG therapy that naturally disappeared during the follow-up period. Similarly, one patient was reported to have temporarily elevated PSA after BCG instillations. One article demonstrated three different imaging patterns of BCG-related granulomatous prostatitis on MP-MRI, and all hyperintense focus on diffusion-weighted MRI reverted to hypointense during the follow-up period[40]. Therefore, we hypothesize that the signal on high b-value diffusion-weighted MRI is high in acute BCG-related granulomatous prostatitis and low in chronic BCG-related granulomatous prostatitis. Patients with a PI-RADS score of 3 or less on MRI should be recommended for a follow-up radiologic examination instead of undergoing prostate biopsy[39]. In one patient, an abnormality with increased fluorodeoxyglucose activity was detected by positron emission tomography computed tomography and pathologically diagnosed as BCG-related granulomatous prostatitis[43]. In granulomatous prostatitis, dynamic contrast-enhanced persistent time of prostate lesions on enhanced MRI was longer than that of prostate cancer[44]. These discoveries provide new clues for differential diagnosis.
Granulomas are clusters of macrophages surrounded by a mononuclear leukocytes and plasma cells. While the pathogenesis of BCG-related granulomatous prostatitis is uncertain, some evidence exists. BCG instillation induces a massive increase in lymphocytes, especially in the proportion of CD4+ Th1 cells[45]; this increase is an inflammatory reaction to BCG in the bladder. Miyashita et al[46] and Butel et al[47] noticed that BCG-related granulomatous prostatitis often radiated from close to the prostatic urethra toward the gland periphery in a wedge-shaped area related to one or more duct systems. A hypothesis was put forward that granulomatous prostatitis was caused by the intra-prostatic reflux of urine contaminated with BCG in the bladder[48]. Therefore, hypersensitivity reactions to BCG antigens from refluxed urine may cause infectious complications, including BCG-induced granulomatous prostatitis[49,50]. Anatomically, ducts in the peripheral zone enter the urethra at less obtuse angles than those from other zones and are likely to be more prone to refluxed urine and damage from potential BCG infections[47]. This could explain why most granulomatous prostatitis occurs in the peripheral zone. A multivariable regression analysis indicated that prostate volume and body mass index were significant risk factors for BCG-induced granulomatous prostatitis[51]. Despite the low incidence of this disease, the mechanisms and predictors of BCG-induced granulomatous prostatitis should be further explored. And histological evaluation remains the gold standard to differentiate granulomatous prostatitis from prostate cancer.
BCG-induced granulomatous prostatitis and prostatic abscesses tend to be intermediate complications that mostly occur 4 wk from the last instillation, whereas testicular and epididymal tuberculosis tend to be late complications (56 wk from the last instillation)[17]. However, our patient showed lower urinary tract symptoms, urinary hydrocele testis, and epididymitis only 3 d after the first instillation. Although he recovered after antibiotic treatment, we speculate that the BCG infection of the urogenital system occurred after the first instillation. BCG instillation was stopped, and the patient received anti-tuberculosis treatment after histopathological diagnosis with BCG-induced granulomatous prostatitis. Most BCG-induced granulomatous prostatitis is asymptomatic and does not require treatment[26,31]. The 4-quinolones and antituberculotics including isoniazid, rifapentine, pyrazinamide, and ethambutol are used in symptomatic BCG-induced granulomatous prostatitis and some BCG-induced prostatic abscesses[25,30,52-54]. If anti-tuberculosis treatment is ineffective, surgery such as transurethral resection of prostate is recommended[55]. The diagnosis and treatment of BCG-induced granulomatous prostatitis requires further study.
Granulomatous prostatitis is a rare complication of BCG instillation that can easily be misdiagnosed as prostate cancer. Although histopathology remains the gold standard to accurately differentiate between the two diagnoses, some clues such as temporarily elevated PSA levels and a high signal followed by a low-signal abnormality on high b-value diffusion-weighted MRI are important indicators of BCG-induced granulomatous prostatitis. High-quality studies should be designed to improve the diagnosis of BCG-induced granulomatous prostatitis.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Infectious diseases
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Crocetto F, Italy; Crocetto F, Italy; Koukoulaki M, Greece S-Editor: Chang KL L-Editor: A P-Editor: Chang KL
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75126] [Cited by in RCA: 64648] [Article Influence: 16162.0] [Reference Citation Analysis (176)] |
| 2. | Bladder cancer: diagnosis and management of bladder cancer: © NICE (2015) Bladder cancer: diagnosis and management of bladder cancer. BJU Int. 2017;120:755-765. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 117] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 3. | Burger M, Catto JW, Dalbagni G, Grossman HB, Herr H, Karakiewicz P, Kassouf W, Kiemeney LA, La Vecchia C, Shariat S, Lotan Y. Epidemiology and risk factors of urothelial bladder cancer. Eur Urol. 2013;63:234-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1158] [Cited by in RCA: 1459] [Article Influence: 112.2] [Reference Citation Analysis (0)] |
| 4. | Martini A, Fallara G, Pellegrino F, Cirulli GO, Larcher A, Necchi A, Montorsi F, Capitanio U. Neoadjuvant and adjuvant immunotherapy in renal cell carcinoma. World J Urol. 2021;39:1369-1376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 5. | Brausi M, Collette L, Kurth K, van der Meijden AP, Oosterlinck W, Witjes JA, Newling D, Bouffioux C, Sylvester RJ; EORTC Genito-Urinary Tract Cancer Collaborative Group. Variability in the recurrence rate at first follow-up cystoscopy after TUR in stage Ta T1 transitional cell carcinoma of the bladder: a combined analysis of seven EORTC studies. Eur Urol. 2002;41:523-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 465] [Cited by in RCA: 474] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 6. | Babjuk M, Burger M, Compérat EM, Gontero P, Mostafid AH, Palou J, van Rhijn BWG, Rouprêt M, Shariat SF, Sylvester R, Zigeuner R, Capoun O, Cohen D, Escrig JLD, Hernández V, Peyronnet B, Seisen T, Soukup V. European Association of Urology Guidelines on Non-muscle-invasive Bladder Cancer (TaT1 and Carcinoma In Situ) - 2019 Update. Eur Urol. 2019;76:639-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 651] [Cited by in RCA: 899] [Article Influence: 149.8] [Reference Citation Analysis (0)] |
| 7. | Lange C, Aaby P, Behr MA, Donald PR, Kaufmann SHE, Netea MG, Mandalakas AM. 100 years of Mycobacterium bovis bacille Calmette-Guérin. Lancet Infect Dis. 2022;22:e2-e12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 128] [Article Influence: 42.7] [Reference Citation Analysis (0)] |
| 8. | Mukherjee N, Julián E, Torrelles JB, Svatek RS. Effects of Mycobacterium bovis Calmette et Guérin (BCG) in oncotherapy: Bladder cancer and beyond. Vaccine. 2021;39:7332-7340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 9. | Malmström PU, Sylvester RJ, Crawford DE, Friedrich M, Krege S, Rintala E, Solsona E, Di Stasi SM, Witjes JA. An individual patient data meta-analysis of the long-term outcome of randomised studies comparing intravesical mitomycin C vs bacillus Calmette-Guérin for non-muscle-invasive bladder cancer. Eur Urol. 2009;56:247-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 416] [Cited by in RCA: 468] [Article Influence: 29.3] [Reference Citation Analysis (0)] |
| 10. | Shelley MD, Wilt TJ, Court J, Coles B, Kynaston H, Mason MD. Intravesical bacillus Calmette-Guérin is superior to mitomycin C in reducing tumour recurrence in high-risk superficial bladder cancer: a meta-analysis of randomized trials. BJU Int. 2004;93:485-490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 236] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 11. | Böhle A, Jocham D, Bock PR. Intravesical bacillus Calmette-Guerin vs mitomycin C for superficial bladder cancer: a formal meta-analysis of comparative studies on recurrence and toxicity. J Urol. 2003;169:90-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 403] [Cited by in RCA: 365] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 12. | Shelley MD, Kynaston H, Court J, Wilt TJ, Coles B, Burgon K, Mason MD. A systematic review of intravesical bacillus Calmette-Guérin plus transurethral resection vs transurethral resection alone in Ta and T1 bladder cancer. BJU Int. 2001;88:209-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 197] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 13. | Schmidt S, Kunath F, Coles B, Draeger DL, Krabbe LM, Dersch R, Kilian S, Jensen K, Dahm P, Meerpohl JJ. Intravesical Bacillus Calmette-Guérin vs mitomycin C for Ta and T1 bladder cancer. Cochrane Database Syst Rev. 2020;1:CD011935. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 14. | Brausi M, Oddens J, Sylvester R, Bono A, van de Beek C, van Andel G, Gontero P, Turkeri L, Marreaud S, Collette S, Oosterlinck W. Side effects of Bacillus Calmette-Guérin (BCG) in the treatment of intermediate- and high-risk Ta, T1 papillary carcinoma of the bladder: results of the EORTC genito-urinary cancers group randomised phase 3 study comparing one-third dose with full dose and 1 year with 3 years of maintenance BCG. Eur Urol. 2014;65:69-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 254] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 15. | van der Meijden AP, Sylvester RJ, Oosterlinck W, Hoeltl W, Bono AV; EORTC Genito-Urinary Tract Cancer Group. Maintenance Bacillus Calmette-Guerin for Ta T1 bladder tumors is not associated with increased toxicity: results from a European Organisation for Research and Treatment of Cancer Genito-Urinary Group Phase III Trial. Eur Urol. 2003;44:429-434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 190] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 16. | Larsen ES, Nordholm AC, Lillebaek T, Holden IK, Johansen IS. The epidemiology of bacille Calmette-Guérin infections after bladder instillation from 2002 through 2017: a nationwide retrospective cohort study. BJU Int. 2019;124:910-916. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 17. | Cabas P, Rizzo M, Giuffrè M, Antonello RM, Trombetta C, Luzzati R, Liguori G, Di Bella S. BCG infection (BCGitis) following intravesical instillation for bladder cancer and time interval between treatment and presentation: A systematic review. Urol Oncol. 2021;39:85-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 18. | Zaza SI, Ghasemzadeh A, Bennett KM. Mycobacterium Bovis Causing Mycotic Aneurysm Secondary to Intravesical Treatment with Bacillus Calmette-Guérin: A Case Report. Ann Vasc Surg. 2022;79:437.e1-437.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 19. | Notghi AAA, Hosseini F, Tsogas N. Severe diffuse proliferative bronchiolitis complicating culture-proven disseminated BCG infection after intravesical instillation for bladder cancer. BMJ Case Rep. 2022;15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 20. | Lupon E, Martin-Blondel G, Pollon T, Berthier C, Lellouch AG, Mansat P. BCGitis of the wrist after intravesical BCG therapy: A case report. Hand Surg Rehabil. 2020;39:585-587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 21. | Adami M, Marsteller I, Mazzucchelli L, Cerny A, Bernasconi E, Bertoli R. Granulomatous hepatitis after intravesical bacillus Calmette-Guérin treatment. Scand J Infect Dis. 2011;43:55-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 22. | De Luca L, Crocetto F, Barone B, Creta M, Pesce S, Aveta A, Campanino MR, Imbimbo C, Longo N. Granulomatous prostatitis mimicking prostate cancer in a patient with psoriatic arthritis: a case report. Future Sci OA. 2020;6:FSO591. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 23. | Crocetto F, Barone B, De Luca L, Creta M. Granulomatous prostatitis: a challenging differential diagnosis to take into consideration. Future Oncol. 2020;16:805-806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 24. | Garrido-Abad P, Rodríguez-Cabello MÁ, González-Gordaliza C, Vera-Berón R, Platas-Sancho A. BCG instillations can mimic prostate cancer on multiparametric MRI. Int Braz J Urol. 2018;44:835-837. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 25. | Białek W, Rudzki S, Iberszer P, Wronecki L. Granulomatous prostatitis after intravesical immunotherapy mimicking prostate cancer. J Ultrason. 2016;16:404-410. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 26. | Logan JK, Walton-Diaz A, Rais-Bahrami S, Merino MJ, Turkbey B, Choyke PL, Pinto PA. Changes observed in multiparametric prostate magnetic resonance imaging characteristics correlate with histopathological development of chronic granulomatous prostatitis after intravesical Bacillus Calmette-Guerin therapy. J Comput Assist Tomogr. 2014;38:274-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 27. | Dianat SS, Matoso A, Carter BH, Macura KJ. Multiparametric MRI findings of granulomatous prostatitis after intravesical bacillus Calmette-Guérin therapy in a patient undergoing active surveillance. Clin Genitourin Cancer. 2014;12:e215-e219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 28. | Pérez-Jacoiste Asín MA, Fernández-Ruiz M, López-Medrano F, Lumbreras C, Tejido Á, San Juan R, Arrebola-Pajares A, Lizasoain M, Prieto S, Aguado JM. Bacillus Calmette-Guérin (BCG) infection following intravesical BCG administration as adjunctive therapy for bladder cancer: incidence, risk factors, and outcome in a single-institution series and review of the literature. Medicine (Baltimore). 2014;93:236-254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 166] [Cited by in RCA: 204] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 29. | Coello Torà I, de la Cruz Ruiz M, Carrillo García P, Carmelo Pieras Ayala E. Epidemiological study of granulomatous prostatitis in a tertiary hospital: Associated risk factors and relationship with prostate cancer. Urologia. 2022;89:257-260. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 30. | Caulier P, Yombi JC, Dufaux M, Feyaerts A, Abi AA, Hainaut P. Prostate abscess following intravesical BCG therapy. Acta Clin Belg. 2009;64:436-437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 31. | Wang Z, Han C, Xu Y, Yu X, Kang W, Xiang Y, Yuan Y, Li L, Wang M. The role of prostate-specific antigen and multiparametric magnetic resonance imaging in the diagnosis of granulomatous prostatitis induced by intravesical Bacillus Calmette-Guérin vaccine therapy in patients with nonmuscle invasive bladder cancer. J Cancer Res Ther. 2021;17:625-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 32. | Serretta V, Gesolfo CS, Di Maida F, Caruana G, Rocchini L, Moschini M, Colombo R, Simonato A. The clinical value of PSA increase during intravesical adjuvant therapy for nonmuscle-invasive bladder cancer. Urologia. 2016;83:145-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 33. | Beltrami P, Ruggera L, Cazzoletti L, Schiavone D, Zattoni F. Are prostate biopsies mandatory in patients with prostate-specific antigen increase during intravesical immuno- or chemotherapy for superficial bladder cancer? Prostate. 2008;68:1241-1247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 34. | Leibovici D, Zisman A, Chen-Levyi Z, Cypele H, Siegel YI, Faitelovich S, Lindner A. Elevated prostate specific antigen serum levels after intravesical instillation of bacillus Calmette-Guerin. J Urol. 2000;164:1546-1549. [PubMed] |
| 35. | Hong S, Kim SC, Kwon T, Jeong IG, Kim CS, Ahn H, Hong JH. Is bladder tumor location associated with prostate cancer detection after intravesical bacillus Calmette-Guérin instillation? PLoS One. 2014;9:e103791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 36. | Borofsky S, George AK, Gaur S, Bernardo M, Greer MD, Mertan FV, Taffel M, Moreno V, Merino MJ, Wood BJ, Pinto PA, Choyke PL, Turkbey B. What Are We Missing? Radiology. 2018;286:186-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 182] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 37. | Le JD, Tan N, Shkolyar E, Lu DY, Kwan L, Marks LS, Huang J, Margolis DJ, Raman SS, Reiter RE. Multifocality and prostate cancer detection by multiparametric magnetic resonance imaging: correlation with whole-mount histopathology. Eur Urol. 2015;67:569-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 297] [Cited by in RCA: 352] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 38. | Bratan F, Niaf E, Melodelima C, Chesnais AL, Souchon R, Mège-Lechevallier F, Colombel M, Rouvière O. Influence of imaging and histological factors on prostate cancer detection and localisation on multiparametric MRI: a prospective study. Eur Radiol. 2013;23:2019-2029. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 227] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 39. | Gottlieb J, Princenthal R, Cohen MI. Multi-parametric MRI findings of granulomatous prostatitis developing after intravesical bacillus calmette-guérin therapy. Abdom Radiol (NY). 2017;42:1963-1967. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 40. | Lee S, Oh YT, Kim HM, Jung DC, Hong H. Imaging Patterns of Bacillus Calmette-Guérin-Related Granulomatous Prostatitis Based on Multiparametric MRI. Korean J Radiol. 2022;23:60-67. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 41. | Suzuki T, Takeuchi M, Naiki T, Kawai N, Kohri K, Hara M, Shibamoto Y. MRI findings of granulomatous prostatitis developing after intravesical Bacillus Calmette-Guérin therapy. Clin Radiol. 2013;68:595-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 42. | Matsushima M, Kikuchi E, Akita H, Miyajima A, Oya M, Jinzaki M. Natural course of asymptomatic abnormal prostate findings incidentally detected by CT after intravesical BCG therapy. Int J Clin Oncol. 2017;22:554-562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 43. | Wilkinson C, Chowdhury F, Scarsbrook A, Smith J. BCG-induced granulomatous prostatitis--an incidental finding on FDG PET-CT. Clin Imaging. 2012;36:413-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 44. | Quon JS, Moosavi B, Khanna M, Flood TA, Lim CS, Schieda N. False positive and false negative diagnoses of prostate cancer at multi-parametric prostate MRI in active surveillance. Insights Imaging. 2015;6:449-463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 45. | Ponticiello A, Perna F, Maione S, Stradolini M, Testa G, Terrazzano G, Ruggiero G, Malerba M, Sanduzzi A. Analysis of local T lymphocyte subsets upon stimulation with intravesical BCG: a model to study tuberculosis immunity. Respir Med. 2004;98:509-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 46. | Miyashita H, Troncoso P, Babaian RJ. BCG-induced granulomatous prostatitis: a comparative ultrasound and pathologic study. Urology. 1992;39:364-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 47. | Butel R, Ball R. The distribution of BCG prostatitis: A clue for pathogenetic processes? Prostate. 2018;78:1134-1139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 48. | Lamm DL, van der Meijden PM, Morales A, Brosman SA, Catalona WJ, Herr HW, Soloway MS, Steg A, Debruyne FM. Incidence and treatment of complications of bacillus Calmette-Guerin intravesical therapy in superficial bladder cancer. J Urol. 1992;147:596-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 507] [Cited by in RCA: 542] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 49. | Balasar M, Doğan M, Kandemir A, Taskapu HH, Cicekci F, Toy H, Gurbuz R. Investigation of granulomatous prostatitis incidence following intravesical BCG therapy. Int J Clin Exp Med. 2014;7:1554-1557. [PubMed] |
| 50. | Bevers RF, Kurth KH, Schamhart DH. Role of urothelial cells in BCG immunotherapy for superficial bladder cancer. Br J Cancer. 2004;91:607-612. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 105] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 51. | Kim TJ, Yu YD, Hwang SI, Lee HJ, Hong SK, Lee SE, Oh JJ. Analysis of risk factors for post-bacillus Calmette-Guerin-induced prostatitis in patients with non-muscle invasive bladder cancer. Sci Rep. 2020;10:9763. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Reference Citation Analysis (0)] |
| 52. | Ye HY, Xu QQ, Huang XB, Ma K, Wang XF. [Tuberculous prostatic abscess following intravesical bacillus Calmette-Guerin immunotherapy: a case report]. Beijing Da Xue Xue Bao Yi Xue Ban. 2015;47:1039-1041. [PubMed] |
| 53. | Doo SW, Kim JH, Yang WJ, Kim SI, Lee DW, Hong SS, Song YS. A case of tuberculous prostatitis with abscess. World J Mens Health. 2012;30:138-140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 54. | Aust TR, Massey JA. Tubercular prostatic abscess as a complication of intravesical bacillus Calmette-Guérin immunotherapy. Int J Urol. 2005;12:920-921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 55. | Okusa H, Irie A, Chin I, Baba S, Mitomi H, Shitara T. [Cases of bacillus Calmette-Guerin-induced granulomatous prostatitis and prostatic stromal invasion of the bladder carcinoma in situ, showing similar clinical findings]. Hinyokika Kiyo. 2003;49:555-557. [PubMed] |