Published online Jun 6, 2023. doi: 10.12998/wjcc.v11.i16.3822
Peer-review started: November 26, 2022
First decision: February 7, 2023
Revised: February 24, 2023
Accepted: April 27, 2023
Article in press: April 27, 2023
Published online: June 6, 2023
Processing time: 187 Days and 23.5 Hours
We report a case of infective endocarditis (IE) in a patient with congenital heart valve lesions accompanied by IE, which was diagnosed based on blood culture analysis that revealed the presence of a gram-negative bacterium, Streptococcus gordonii.
The patient had a history of precordial valve disease diagnosed by cardiac ultrasound, as well as a 4-mo history of fever. He was subjected to comprehensive anti-infection and anti-heart failure treatment in the internal medicine department. Further examination revealed sudden dislodgement from and perforation through the aortic valve by the superfluous organisms, as well as occurrence of bacterial emboli dislodgement, which caused bacteremia and infectious shock. He recovered and was discharged from the hospital after surgical and postoperative anti-infection treatments.
We review the treatment process and highlight inspirations and reflections from this case; suggest possible future changes in treatment modalities.
Core Tip: Infective endocarditis is caused by pathogenic bacteria that infect the heart via the bloodstream. It is most common in heart valves but also in atrioventricular walls, tendons and other structures. Congenital heart disease is common in heart structures near the abnormal intracardiac shunt low-pressure cavities, and serious cases can be accompanied by abscesses, fistulas and other cardiac complications. The fallen flap or leaflet can cause embolism of various organs throughout the body, has a high mortality rate, and there are no specific clinical manifestations. It is very easy to misdiagnose, and missed diagnoses are common. The search for sensitive diagnostic methods and timely treatment are particularly important.
- Citation: Qu YF, Yang J, Wang JY, Wei B, Ye XH, Li YX, Han SL. Valve repair after infective endocarditis secondary to perforation caused by Streptococcus gordonii: A case report. World J Clin Cases 2023; 11(16): 3822-3829
- URL: https://www.wjgnet.com/2307-8960/full/v11/i16/3822.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v11.i16.3822
Infective endocarditis (IE) is a life-threatening cardiovascular disease with an annual incidence of 1.5/100000-15/100000 and a mortality rate of 20%-25%[1]. The disease is caused by a pathogenic microorganism that directly infects heart valves, the endocardium of the ventricular wall or the endocardium of the adjacent aorta via the bloodstream, often leading to superfluous organisms. Subacute IE is characterized by an insidious and slow onset, with a long course that usually lasts between 6 wk and 3 mo[2]. These patients mostly experience cardiac problems. Due to the insidious site of infection, notably, IE patients have diverse and nonspecific clinical manifestations, which subsequently lead to missed diagnoses and misdiagnoses that result in serious adverse consequences that threaten quality of life[3]. Therefore, early diagnosis coupled with timely treatment is crucial for management of the disease. In the present study, we report on a case of IE caused by Streptococcus gordonii, with fever as the main manifestation, at our hospital.
A 61-year-old man presented at our hospital with intermittent fever and malaise for 4 mo.
Four months before the visit, he had developed a fever (up to 40 C), mostly in the afternoon and at night, accompanied by chills and malaise. Although his body temperature normalized after 3 d of anti-infection treatment via self-treatment, the fever reappeared after stopping the treatment. The disease progressively worsened, and his blood pressure decreased and was accompanied by an increased heart rate. The patient was diagnosed with infectious shock and was subsequently transferred to our hospital for further treatment.
The patient was previously in good health.
He was previously healthy and had no family history of disease.
Physical examination revealed slightly pale skin and mucous membranes, with no enlargement of the jugular vein, but there was enlargement of the cardiac border to the left. There was also evidence of a grade III-IV diastolic murmur in the aortic valve area and peripheral vascular signs (+).
Laboratory test results revealed normal leukocytes and neutrophils, the patient had C-reactive protein and calcitonin levels of 53.1 mg/L and 2.66 ng/mL, respectively, as well as liver and renal function tests that were within normal limits. The Tuberculin purified protein derivative test results were negative. Paired blood cultures (asymmetric limbs, e.g., left upper limb and right lower limb) on 3 consecutive days showed gram-positive cocci. Macrogen next generation sequencing technology showed Streptococcus gordonii.
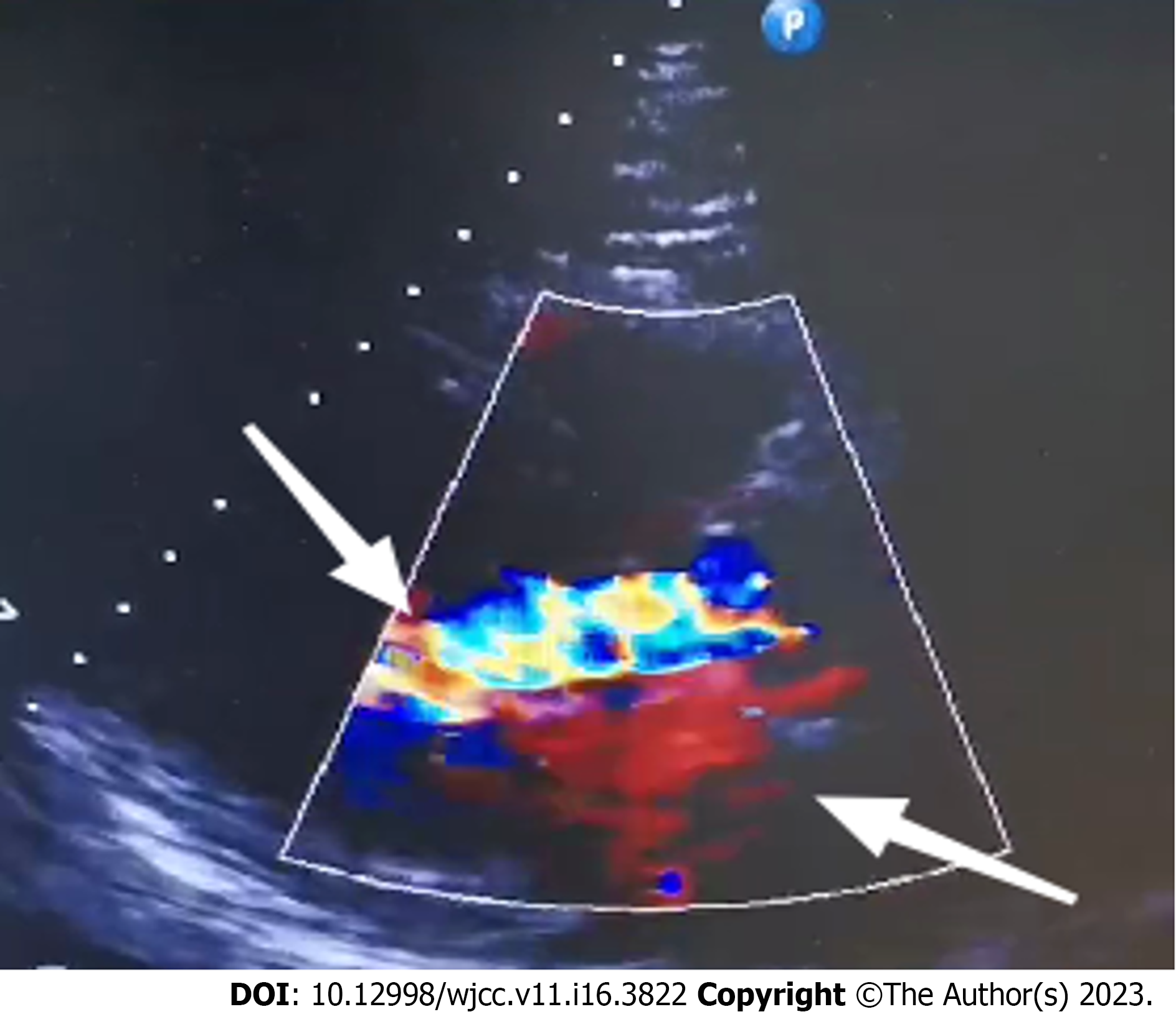
Cardiac ultrasonography showed an ejection fraction of 52% and aortic valve leaflet thickening and echogenic enhancement, a moderate regurgitant signal under the aortic valve in diastole, a small amount of regurgitant signal on the left atrial side of the mitral valve in systole, a small amount of regurgitant signal on the right atrial side of the tricuspid valve in systole, an ascending aortic internal diameter of 46 mm, an aortic valve Vmax of 2.2 m/s, an enlarged left atrium, aortic valve calcification with moderate regurgitation, and mild regurgitation of the second and tricuspid valves (Figure 1). The cardiac magnetic resonance imaging results were consistent with dilated cardiomyopathy changes, left heart insufficiency, a significantly reduced systolic function, aortic valve thickening and stenosis, incomplete closure of the mitral valve and tricuspid valve, a small amount of pericardial effusion, pericardial inflammatory changes, and a small amount of bilateral pleural effusion.
Flow cytometry showed an increase in the proportion of granulocytes and nucleated red cells and a mild decrease in the proportion of lymphocytes. The phenotype was not abnormal. Abnormal immunoglobulins and identification of multiple myeloma: Immunofixation electrophoresis (IFE) results showed that there was no “M protein” detected within the serum polyclonal immunoglobulins; there were no kap and lam light chain "M protein" detected in the urine. Serum protein and IFE revealed no significant "M protein", a reduction in albumin levels, and elevated levels of α1 and γ globulins. The serum immunoglobulin (Ig)G, IgA, IgM and kap, as well as lam light chain levels, were normal, but the IgE levels were high. We also found a moderate increase in the level of urine kap light chain and 24-h urine protein levels. Protein electrophoresis revealed no specific band in the urine. No light chain type "M protein" was detected in the urine via IFE, This result excluded abnormal immunoglobulinemia and multiple myeloma (Table 1).
| Name | Value | Rang | Unit |
| IgG | 1500 | 751-1560 | mg/dL |
| IgE | 126 | 0-100 | IU/mL |
| Urine kaP | 2.1 | < 1.9 | mg/dL |
| Stray lam | 38.2 | 8.3-27.0 | mg/L |
| Lamlight chain | 704 | 313-723 | mg/dL |
| IgA | 243 | 82-453 | mg/dL |
| Urine lam | < 0.5 | < 5.0 | mg/dL |
| FLC k/L | 0.56 | 0.13-1.56 | mg |
| IgM | 57.5 | 46-304 | mg/dL |
| 24-h urine protein | 1900 | 1000-2000 | mg |
| Stray kap | 21.4 | 6.7-22.4 | mg/L |
| Kaplight chain | 1310 | 629-1350 | mg/dL |
Infective endocarditis.
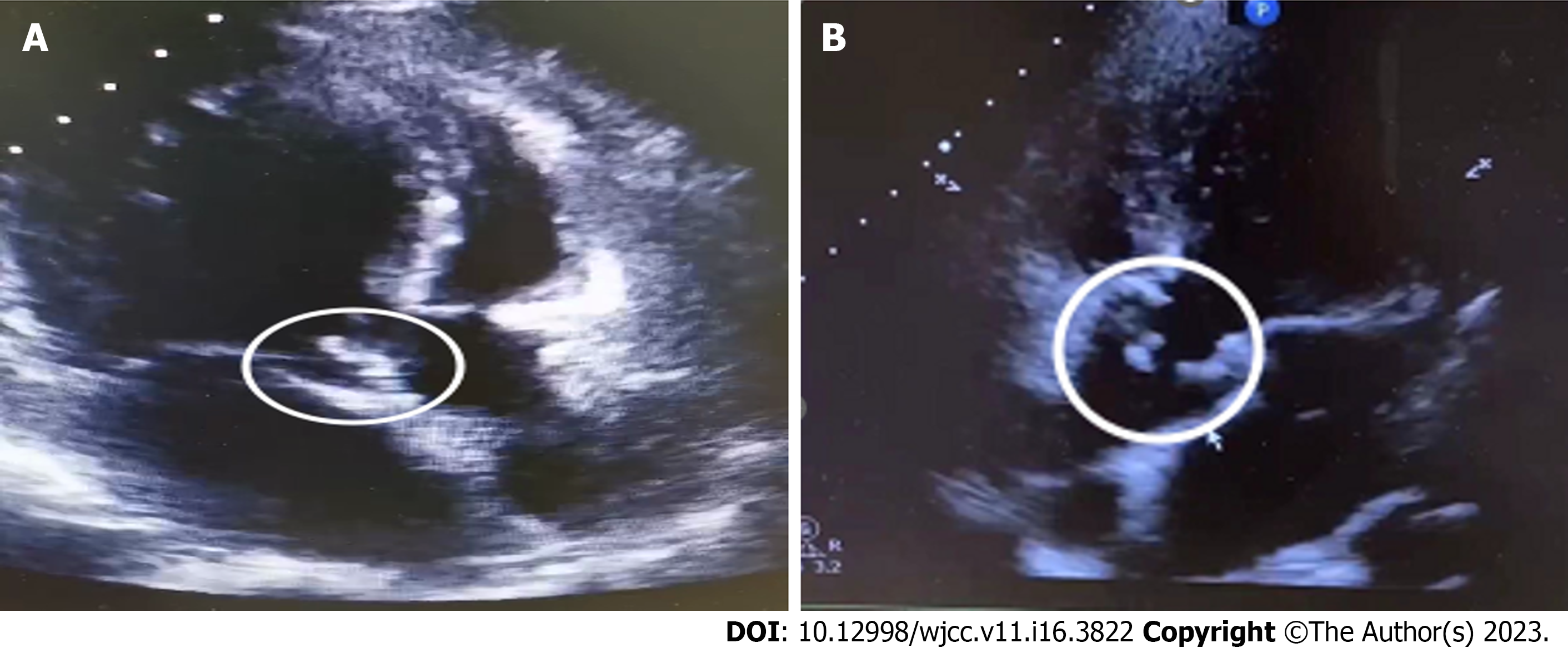
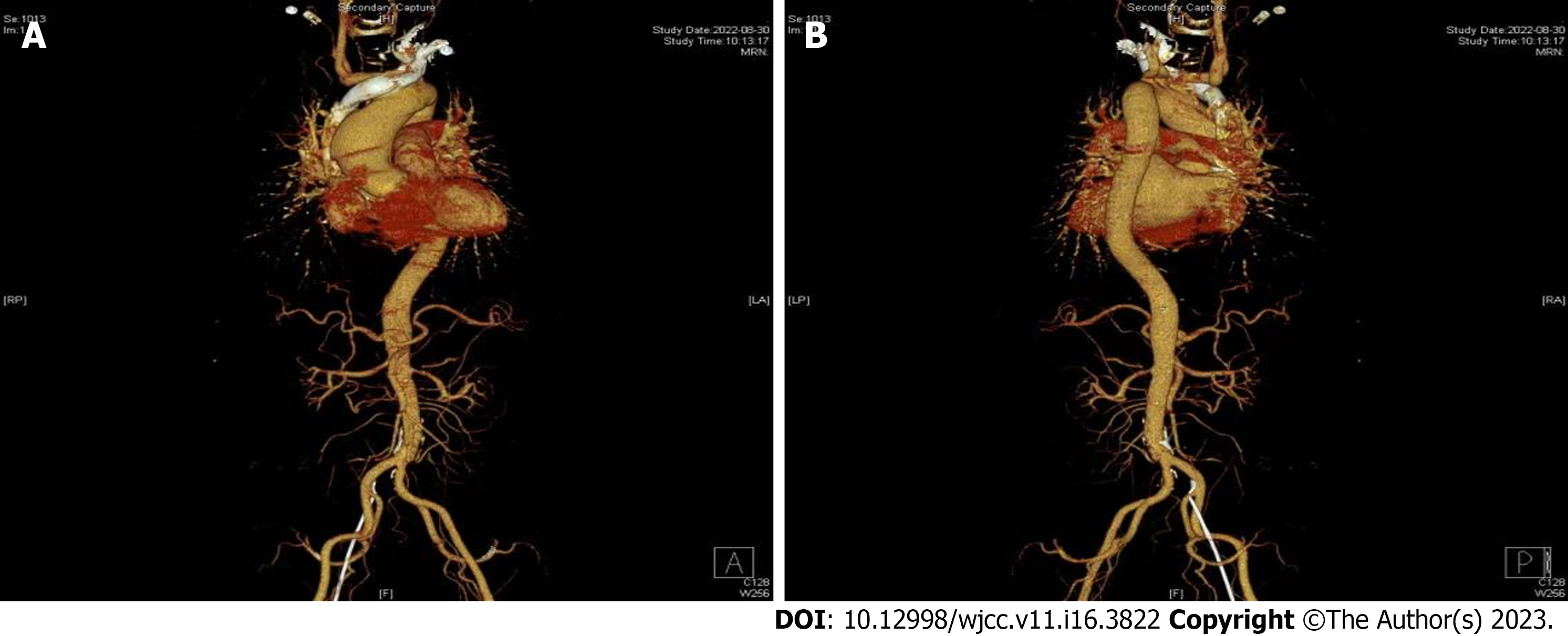
The patient's prolonged unexplained fever, coupled with blood culture and echocardiographic results, made us strongly suspect a subacute infection. The patient’s fever subsided after 9 d of continuous treatment, and his infection index was under control. Consequently, aortic valve replacement was recommended following anti-infection treatment. The patient then developed a preoperative sudden onset of chest and abdominal pain with profuse sweating and an emergency transesophageal ultrasonography was performed. The results are presented in Figure 2. In summary, we found evidence of aortic valve redundancy with perforation, while the emergency CTA results revealed moderate stenosis of the lumen at the beginning of the abdominal trunk. The possibility of bacterial esophageal involvement and visible penetrating ulcers was considered (Figure 3). Consequently, the surgical approach was changed from simple aortic valve replacement to aortic valve replacement surgery and aortic valvuloplasty. The intraoperative examination findings were as follows: Moderate levels of yellowish pericardial fluid were present, there was marked widening of the ascending aorta, marked malformation of the aortic bicuspid valve, leaflet destruction with perforation and massive regurgitation, complete leaflet removal, and compression of the artificial vessel to wrap the ascending aorta, and placement of aortic valve bioprosthesis was performed. The operation was successfully completed. Postoperative pathological analysis revealed fibrous connective tissue hyperplasia with focal glassy and mucinous degeneration, localized fibrin deposition, and evidence of calcified foci in the valve tissue.
The patient received anti-infection treatment for 1 mo after surgery. Reexamination revealed that his liver, kidney and cardiac functions had been restored back to normal.
In 2016, the American Association for Thoracic Surgery (AATS) published an expert consensus on the surgical management of IE, the most recent guiding document for the management of this condition. The AATS expert consensus indicates that the current DUKE criteria, which are based on the diagnostic criteria proposed in 1994 and have subsequently been modified several times, are the most practical for the diagnosis of IE[4]. The most common clinical IE manifestation is fever, with an insidious onset in subacute cases, and it can be associated with relatively mild toxic symptoms that may range from weeks to months. Although IE is a common cause of unexplained fever, only 18.6%-20.3% of patients present with typical manifestations described in the DUKE criteria, such as skin petechiae, emboli, splenomegaly, and Osler nodules. Approximately 30% of IE patients do not exhibit an obvious heart murmur during the early stages of disease development or during the course of treatment. The doctors who treated the patient in the present study before he came to our hospital thought that the heart murmur could be explained by mitral regurgitation and did not consider the possibility of IE. Notably, they were satisfied with improvement of his clinical symptoms and did not follow up on the disease change, a phenomenon that resulted in a delay of treatment of the disease.
Bacteremia, caused by pathogenic microorganisms in the blood, is the basis for the development of IE. Blood culture tests have diagnostic value and can help guide the selection of anti-infective drugs. The main pathogens and major routes of infection in IE have changed in recent years. Notably, virulent and drug-resistant staphylococci are the most common organisms and are more likely to occur in elderly patients, as well as in those with nosocomial chronic disease and intracardiac implants. In the present study, the blood culture results suggested that this patient had a Streptococcus gordonii infection. This pathogen belongs to the Gram-stain-positive retarded streptococcal group and is a partly anaerobic α-haemolytic streptococcus (VGS). Recent studies have shown that community-acquired autologous valvular endocarditis caused by VGS is also common in IE patients, particularly in developing countries[5], and many of them are caused by Streptococcus gordonii. Generally, Streptococcus gordonii colonizes the oral cavity and upper respiratory tract and is mainly involved in the composition of dental plaque; it is a conditional pathogen that is usually not pathogenic. In recent years, many cases of Streptococcus gordonii infections have been reported in China and abroad, and these cases include cases of sepsis, IE, splenic abscess and septic arthritis. These infections have subsequently been associated with poor oral hygiene, loose teeth, dental surgery and other invasive procedures. The patient in the present study exhibited upper respiratory tract infection symptoms, such as a sore and dry throat, 1 wk before the onset of fever. However, he did not pay attention to these symptoms and experienced overexertion during the same period[6]. It is worth noting that the patient’s medical history is imperative to obtain an adequate disease diagnosis.
Echocardiography is a key procedure for confirming an IE diagnosis[7] because it can not only detect superfluous organisms at an early stage but also accurately measure their sizes, thereby facilitating assessment of the disease severity, can be used to assess cardiac function, and can detect cardiac complications, among other factors[8]. However, echocardiography is influenced by subjective factors. In the present case, the initial echocardiography failed to detect superfluous organisms in this patient’s heart valve and relaxed our vigilance. We attributed this to the fact that the location and size of the flap were not easily detectable by the ultrasound beam and because of the physician’s inexperience. Transesophageal echocardiography can detect redundant organisms with a diameter of 1-1.5 mm and directly reveal the relationship between the redundant organisms and the valve from the posterior left atrium, achieving a positive detection rate of up to 90%. However, echocardiography is not an ideal detection tool for patients with severe valvular lesions, bulging valves < 2 mm, prolapsed valves, or valves without bulging valves. Based on this, a negative result does not completely exclude IE.
Successful IE treatment depends on the efficient removal of pathogenic microorganisms and surgery has shown promise in successfully eradicating these pathogens in patients[9]. Previous studies have shown that to reduce morbidity and mortality, 20%-50% of patients require cardiac surgery to remove infected tissue and restore valve function[10-12]. In fact, timely surgery is the only hope for saving a patient's life in a case of severe IE. Internal medicine and surgery have their own advantages and shortcomings during IE treatment. Although internal medicine-based treatments can clear most of the microorganisms, it was not efficacious with the patient in this case. The use of anti-infection treatment not only caused an impairment of his cardiac function but also resulted in hemodynamic abnormalities due to the destruction of the intracardiac structures and the emergence of dislodged bacterial emboli. Therefore, internal medicine treatment alone cannot prevent the occurrence of superfluous events, necessitating the application of surgical treatment that corrects congenital valve malformation and restores the heart valve. The patient in the current study was subjected to surgical treatment, which corrected his congenital valve malformation, restored his heart valve function, and improved his heart failure. The patient recovered. Choosing the right time for surgery is the key to effective surgical treatment. It is generally believed that patients with heart failure or hemodynamic disorders, persistent infections, uncontrolled intracardiac local infections, refractory microbial infections, persistent floppy biology > 10 mm with embolism or valve dysfunction should undergo surgery as soon as possible[13]. The echocardiography results of the patient in the present study revealed a floppy biology measuring 1.2 cm × 1.2 cm, with indications for surgery. There are also physicians who believe that surgical intervention is not appropriate in the acute phase due to the possibility of an increased occurrence of complications and high surgical risks and believe in conservative therapy. Notably, controversy still surrounds the timing and indications for surgery, necessitating further research explorations with large randomized controlled clinical trials. Although the application of cardiac surgery for the treatment of active IE is only a century old, recent advances in surgical techniques and perioperative management have made it the cornerstone of IE treatment. Moreover, there is no uniform standard for the indication, timing, and risk assessment of surgery in active IE due to the complexity and variable nature of the disease[14]. Therefore, clinical decision-making in complex cases must be fully individualized, taking into account the patient's demographic characteristics, comorbidities, disease severity, and stage, to determine whether and when to perform surgical intervention and to prepare for foreseeable risks after surgery. There is a need to consult hospitals with multidisciplinary teams specializing in IE in cases of complex IE. Given the large number of IE patients in China, there is a need for concerted efforts across clinical research to standardize IE treatment strategies by focusing on unresolved international issues. Since the population and pathogenic spectrum of IE patients in China are different from those in Europe and the United States, there is a need to develop active IE surgery strategies for the Chinese population through research on treatment and prospects.
IE is a very serious infectious disease which has an increasing incidence every year. The current treatment modalities are: (1) Antibiotic treatment; and (2) Surgical treatment. Antibiotic therapy is administered intravenously for a long period of time, usually 4-6 wk, depending on whether the pathogenic organism is cleared and whether the infection is in the primary or prosthetic valve. Currently, a randomized controlled trial in Denmark investigated the need for a full course of intravenous antibiotic therapy during the treatment of IE[15]. That study randomized patients with IE infected with Staphylococcus, Streptococcus, or Escherichia coli (infection controlled) to two groups, one group received continuing intravenous antibiotic therapy and the other group received oral antibiotic therapy. The results showed that the oral antibiotic treatment group did not have a higher incidence of death, embolism, bacteremia, and unplanned cardiac surgery than the full IV group, and the results were statistically significant. This finding may lead to future changes in the IE treatment guidelines, where the vast majority of patients with largely controlled infections can be transitioned to outpatient oral drug therapy due to the option of combination therapy, which means fewer hospital days and therefore potentially fewer hospital-acquired complications, and this may reduce financial stress for patients. In the next 5-10 years, more randomized clinical trials are planned to be completed to validate the feasibility of combination therapy[16]
IE is characterized by an insidious onset, a difficult diagnosis, an aggressive condition and susceptibility to floppy dislodgement events. Therefore, patients with recurrent fever of unknown origin need to be monitored closely.
There is a need for clinicians to raise awareness of IE, especially by performing comprehensive physical examinations and analysis. Clinicians should highly consider IE in patients with unexplained fever, especially those with underlying cardiac disease conditions.
Patients, who are suspected of having IE but who have negative results on echocardiogram should be retested or ultrasounded by a senior ultrasonographer. In cases where transthoracic echocardiogram suggests indirect hints, such as valve prolapse, incomplete closure, or septic lesions, transesophageal echocardiogram should be performed promptly to improve the detection rate.
The disease course in patients with active IE is often complex and variable. Although international guidelines have somewhat standardized the indications and timing of surgery in active IE patients, there is still a need for concerted efforts from an IE-specific multidisciplinary team during decision-making for specific patients. In fact, the discussion on the indication and timing of surgery is a game of risk vs expected benefit. Therefore, assessment of the risk of surgery in active IE patients is imperative for effective management of the disease.
I am grateful to Dr. Jun Yang and Dr. Jun-Yu Wang for inspiring me about the treatment of infective endocarditis and establishing a sound examination idea. I am grateful to Bing Wei, Si-Lu Han, Yi-Xuan Li, and Xing-Hua Ye for organizing the case contents and providing helpful examination results.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Medicine, research and experimental
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Gravina AG, Italy; Karcioglu Ö, Turkey S-Editor: Cai YX L-Editor: Filipodia P-Editor: Cai YX
| 1. | Werdan K, Dietz S, Löffler B, Niemann S, Bushnaq H, Silber RE, Peters G, Müller-Werdan U. Mechanisms of infective endocarditis: pathogen-host interaction and risk states. Nat Rev Cardiol. 2014;11:35-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 159] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 2. | Li JS, Sexton DJ, Mick N, Nettles R, Fowler VG Jr, Ryan T, Bashore T, Corey GR. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis. 2000;30:633-638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2699] [Cited by in RCA: 2827] [Article Influence: 113.1] [Reference Citation Analysis (0)] |
| 3. | Cahill TJ, Baddour LM, Habib G, Hoen B, Salaun E, Pettersson GB, Schäfers HJ, Prendergast BD. Challenges in Infective Endocarditis. J Am Coll Cardiol. 2017;69:325-344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 286] [Cited by in RCA: 428] [Article Influence: 53.5] [Reference Citation Analysis (0)] |
| 4. | AATS Surgical Treatment of Infective Endocarditis Consensus Guidelines Writing Committee Chairs; Pettersson GB, Coselli JS; Writing Committee, Pettersson GB, Coselli JS, Hussain ST, Griffin B, Blackstone EH, Gordon SM, LeMaire SA, Woc-Colburn LE. 2016 The American Association for Thoracic Surgery (AATS) consensus guidelines: Surgical treatment of infective endocarditis: Executive summary. J Thorac Cardiovasc Surg. 2017;153:1241-1258.e29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 283] [Article Influence: 35.4] [Reference Citation Analysis (0)] |
| 5. | Drost TF, Rosemurgy AS, Proctor D, Kearney RE. Outcome of treatment of combined orthopedic and arterial trauma to the lower extremity. J Trauma. 1989;29:1331-1334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 39] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 6. | Sevilla T, López J, Gómez I, Vilacosta I, Sarriá C, García-Granja PE, Olmos C, Di Stefano S, Maroto L, San Román JA. Evolution of Prognosis in Left-Sided Infective Endocarditis: A Propensity Score Analysis of 2 Decades. J Am Coll Cardiol. 2017;69:111-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 7. | Yong MS, Saxena P, Killu AM, Coffey S, Burkhart HM, Wan SH, Malouf JF. The preoperative evaluation of infective endocarditis via 3-dimensional transesophageal echocardiography. Tex Heart Inst J. 2015;42:372-376. [PubMed] |
| 8. | Xu H, Cai S, Dai H. Characteristics of Infective Endocarditis in a Tertiary Hospital in East China. PLoS One. 2016;11:e0166764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 9. | Dickerman SA, Abrutyn E, Barsic B, Bouza E, Cecchi E, Moreno A, Doco-Lecompte T, Eisen DP, Fortes CQ, Fowler VG Jr, Lerakis S, Miro JM, Pappas P, Peterson GE, Rubinstein E, Sexton DJ, Suter F, Tornos P, Verhagen DW, Cabell CH; ICE Investigators. The relationship between the initiation of antimicrobial therapy and the incidence of stroke in infective endocarditis: an analysis from the ICE Prospective Cohort Study (ICE-PCS). Am Heart J. 2007;154:1086-1094. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 237] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 10. | Rutherford SJ, Glenny AM, Roberts G, Hooper L, Worthington HV. Antibiotic prophylaxis for preventing bacterial endocarditis following dental procedures. Cochrane Database Syst Rev. 2022;5:CD003813. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 11. | Bouza E, Muñoz P, Burillo A. Gram-negative endocarditis: disease presentation, diagnosis and treatment. Curr Opin Infect Dis. 2021;34:672-680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 12. | Vieira ML, Grinberg M, Pomerantzeff PM, Andrade JL, Mansur AJ. Repeated echocardiographic examinations of patients with suspected infective endocarditis. Heart. 2004;90:1020-1024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 86] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 13. | Yepez C, Ríos J. Pulmonary valve reconstruction using Ozaki's technique for infective endocarditis. Eur J Cardiothorac Surg. 2021;59:917-919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 14. | Cecchi E, Ciccone G, Chirillo F, Imazio M, Cecconi M, Del Ponte S, Moreo A, Faggiano P, Cialfi A, Squeri A, Enia F, Forno D, De Rosa FG, Rinaldi M, Castiglione A. Mortality and timing of surgery in the left-sided infective endocarditis: an Italian multicentre study. Interact Cardiovasc Thorac Surg. 2018;26:602-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 15. | Iversen K, Ihlemann N, Gill SU, Madsen T, Elming H, Jensen KT, Bruun NE, Høfsten DE, Fursted K, Christensen JJ, Schultz M, Klein CF, Fosbøll EL, Rosenvinge F, Schønheyder HC, Køber L, Torp-Pedersen C, Helweg-Larsen J, Tønder N, Moser C, Bundgaard H. Partial Oral vs Intravenous Antibiotic Treatment of Endocarditis. N Engl J Med. 2019;380:415-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 361] [Cited by in RCA: 518] [Article Influence: 86.3] [Reference Citation Analysis (0)] |
| 16. | Bundgaard H, Ihlemann N, Gill SU, Bruun NE, Elming H, Madsen T, Jensen KT, Fursted K, Christensen JJ, Schultz M, Østergaard L, Rosenvinge F, Schønheyder HC, Helweg-Larsen J, Fosbøll EL, Køber L, Torp-Pedersen C, Tønder N, Moser C, Iversen K. Long-Term Outcomes of Partial Oral Treatment of Endocarditis. N Engl J Med. 2019;380:1373-1374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 51] [Article Influence: 8.5] [Reference Citation Analysis (0)] |