Published online Sep 26, 2022. doi: 10.12998/wjcc.v10.i27.9556
Peer-review started: March 22, 2022
First decision: April 13, 2022
Revised: April 25, 2022
Accepted: August 24, 2022
Article in press: August 24, 2022
Published online: September 26, 2022
Processing time: 177 Days and 19.2 Hours
Coronavirus disease 2019 (COVID-19) is known to present with respiratory symptoms, which can lead to severe pneumonia and respiratory failure. However, it can have multisystem complications such as cardiovascular manifestations. The cardiovascular manifestations reported comprise myocarditis, cardiogenic shock, arrhythmias, pulmonary embolism, deep vein embolism, acute heart failure, and myocardial infarction. There is also an indirect impact of the pandemic on the management of cardiovascular care that has been shown clearly in multiple publications. In this review, we summarize the deadly relation of COVID-19 with cardiovascular events and the wider impact on several cardiovascular care areas by the pandemic situation
Core Tip: The pre-existing cardiovascular disease is an important risk factor for a severe clinical course of coronavirus disease 2019 (COVID-19) and is associated with adverse outcomes. Furthermore, COVID-19 may exacerbate underlying heart disease and is frequently aggravated by cardiovascular complications, such as thromboembolic events and myocardial injury between others. COVID-19 also has been associated with a direct damage of the cardiovascular system. In this review, we will focus on the direct and indirect impact of the pandemic in relation with cardiovascular diseases to show that cardiovascular disease and COVID-19 really were a deadly combination.
- Citation: Vidal-Perez R, Brandão M, Pazdernik M, Kresoja KP, Carpenito M, Maeda S, Casado-Arroyo R, Muscoli S, Pöss J, Fontes-Carvalho R, Vazquez-Rodriguez JM. Cardiovascular disease and COVID-19, a deadly combination: A review about direct and indirect impact of a pandemic. World J Clin Cases 2022; 10(27): 9556-9572
- URL: https://www.wjgnet.com/2307-8960/full/v10/i27/9556.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v10.i27.9556
The complete impact of the pandemic has been much greater than what is indicated by reported deaths due to coronavirus disease 2019 (COVID-19) alone. Indirect effects of the pandemic have been present on cardiovascular disease management and could justify partially the excess of death related with COVID-19[1].
The prior presence of cardiovascular disease is an important risk factor for a severe clinical course of COVID-19 and is associated with unfavorable outcomes[2,3]. Furthermore, COVID-19 may aggravate underlying heart disease and is frequently worsened by cardiovascular complications, such as thromboembolic events, malignant arrhythmia, and myocardial injury[4]. COVID-19 also has been associated with a direct damage of the cardiovascular system[5].
In this review, we will focus on the direct and indirect impact of the pandemic in relation with cardiovascular diseases to show that cardiovascular disease and COVID-19 really were a deadly combination.
Arterial and venous thromboembolic events are frequently observed in COVID-19 patients and contribute to increased morbidity and mortality[4,6].
The incidence rates of venous thrombotic events (VTE) reach more than 30% in cohorts of critically ill patients despite pharmacological prophylaxis[7]. As evidence of the activated coagulation system, D-dimer plasma levels are elevated in a relevant proportion of COVID-19 patients, associated with an adverse outcome[8]. Numerous randomized controlled trials have evaluated the role of therapeutic doses of heparin in reducing VTE events or mortality in patients hospitalized due to COVID-19. In the intensive care unit (ICU) scenario, these studies showed that heparin at therapeutic doses did not reduce mortality but may be associated with a higher risk of bleeding events; consequently, this approach is not recommended. According to the current guidelines, hospitalized COVID-19 patients should receive at a minimum routine thromboprophylaxis. Therapeutic-dose heparin should be used for hospitalized COVID-19 patients who have a D-dimer level above the upper limit of normal, require low-flow oxygen, and have no increased bleeding risk. In the patients receiving ICU level of care, prophylactic-dose heparin is recommended[9].
COVID-19 is an independent risk factor for the occurrence of ischemic stroke, with a higher risk in patients with a severe clinical course[10]. The reported stroke rate of COVID-19 patients is approximately 1%. Notably, patients with COVID-19 seem to have an increased risk for cryptogenic and large vessel stroke[4].
Furthermore, COVID-19 has been descried as a relevant risk factor for the development of acute myocardial infarction (AMI)[11]. A large fraction of patients presenting with AMI and accompanying severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection has type II MI caused by the primary infection inducing respiratory and/or hemodynamic derangement. It remains controversial whether SARS-CoV-2 can trigger type I MI; the potential underlying mechanisms might be cytokine-related plaque instability, immune-thrombosis, and endothelitis[4,12]. Patients presenting with SARS-CoV-2 infection and concomitant ST elevation myocardial infarction (STEMI) have a high thrombus burden, a high risk for stent thrombosis, and an increased risk for a poor outcome[13]. Importantly, the COVID-19 pandemic should not compromise timely reperfusion in STEMI, and management of patients with non-ST-segment elevation acute coronary syndromes (ACS) should be guided by clinical risk stratification[14].
Through the COVID-19 pandemic, a substantial increase in the incidence rate of Takotsubo syndrome was observed[15]. Notably, in one study, the majority of patients presenting with Takotsubo syndrome tested negative for SARS-CoV-2, potentially implying an increased level of stress in the general population due to the public health measure to reduce transmission rates (social distancing rules, self-isolation, and quarantine), economic stress, and fear of infection[4,15].
Progressive dyspnoea is both the hallmark symptom of acute heart failure (AHF) as well as severe COVID-19 and distinguishing these entities is challenging, as up to 12% of hospitalised COVID-19 patients might have an established diagnosis of chronic heart failure[16]. Natriuretic peptides might also be elevated in COVID-19 patients even in the absence of left ventricular systolic impairment, proving another challenge in differentiating both entities[14,17]. In line with this, COVID-19 might both trigger AHF in patients with a known history of heart failure as well as lead to a first episode of hospitalization in patients with occult heart failure[18]. Whether these factors are causative or just coincident is currently within the scope of scientific research. However, several factors induced by COVID-19 might be contributing to mechanisms of AHF such as acute myocardial injury defined as increase of circulating troponin levels (observed in 8% to 15% of COVID-19 patients), which has been described to be associated with disease severity[19]. Besides unspecific myocardial injury, the myocardium might also be deteriorated due to COVID-19 associated myocarditis, whereas both direct cardiomyocyte infection and autoimmune myocarditis have been described[20]. Acute respiratory distress syndrome (ARDS), hypoxemia, renal failure, volume expansion, increased sympathetic drive, fever, and systemic inflammatory response syndrome, factors that are commonly present in severe respiratory disease, might further induce or aggravate heart failure[21].
While AHF rates have consistently been reported to decline worldwide, there was an increase in symptom burden as well as a higher in-hospital mortality rate as compared to historical data observed during the COVID-19 pandemic[14,19,22,23]. Among patients with known heart failure, one third of patients hospitalised with COVID-19 and up to 50% of those that developed AHF died within the in-hospital stay[8]. Patients with heart failure and COVID-19 that had to be admitted to an ICU had an even higher mortality rate of up to 75%[16].
Besides left sided heart failure, acute right heart failure, which might occur secondary to acute pulmonary hypertension or ARDS, has been described in patients with COVID-19[24]. Even in the absence of manifestations of right heart failure, right ventricular dysfunction appears in nearly 15% of patients with COVID-19 and might contribute to impaired outcomes, irrespective of heart failure state[25]. Moreover, there has been recent data that patients following SARS-CoV-2 infection are at increased risk of developing cardiovascular disease even after the acute phase of infection often referred to as “long COVID”[26].
In addition to the increased risk of an adverse outcome of SARS-CoV-2 infection and to the cardiovascular effects of COVID-19, patients with cardiovascular disease suffer from indirect consequences of the pandemic. Profound adaptations of health care systems were necessary to cope with the high number of severely ill patients with SARS-CoV-2 infection. This included a deferral of a substantial number of elective procedures and affected the acute care of patients with cardiovascular diseases[27]. The New York Times asked in April 2020: “Where have all the heart attacks gone?” Multiple reports from different European countries, the United States, and China show a marked reduction in hospital admissions due to ACS[28-32]. A meta-analysis of 27 international studies corroborated these results, showing a 40%-50% reduction of hospital admissions due to ACS[33]. The interval between symptom onset and admission to the hospital was increased most likely because patients waited longer until they called the emergency services. An observed increase of the door-to-device-times[34,35] might be partly attributable to the recommendations regarding protective measures for the staff as well as adapted reperfusion strategies including fibrinolysis in STEMI and conservative strategies in non-STEMI[36,37]. Hospital admissions due to other acute cardiovascular conditions were also reduced during the pandemic[35,38]. Reports on the effects of the pandemic on in-hospital mortality of AMI are heterogeneous[27]. Some studies describe increased rates of mortality; others show no difference[29, 34,39,40]. The above-mentioned meta-analysis showed an increased AMI mortality from March to May 2020[33]. The observed increase in prehospital deaths and out-of-hospital cardiac arrests implies a negative impact of the pandemic on total mortality rates of AMI[41,42]. Table 1 gives an overview on potential causes for chances in acute cardiac care during the COVID-19 pandemic.
| Potential causes for changes in acute cardiac care during the coronavirus disease 2019 pandemic |
| Patients were afraid of infection with severe acute respiratory syndrome coronavirus-2 during hospitalization |
| Misinterpretation of thoracic complaints and/or dyspnea as non-cardiac by patients and doctors |
| Changed approach of AMI care with longer door-to-device times and adaption of reperfusion strategies |
| Planned reduction of elective procedures in order to keep resources for care of COVID-19 patients |
COVID-19-associated myocardial injury, defined as serum troponin level above the 99th percentile of the upper reference limit, was reported from the early days of the pandemic[43]. In several studies, myocardial injury was common among COVID-19 patients, with a very wide prevalence ranging from 8%-62% according to the study and the definition used[43,44]. Nevertheless, almost all studies have shown that these patients had a worse prognosis[44].
Interestingly, even after 2 years of the onset of the COVID-19 epidemic, the pathogenesis of myo
However, the ability of SARS-CoV-2 to directly cause cardiomyocyte infection and damage remains controversial. According to the current definition of myocarditis, proposed by the European Society of Cardiology (ESC) Working Group on Myocardial and Pericardial Diseases, a definitive diagnosis can only be made when a viral genome is proven in endomyocardial specimens along with the histological findings of active myocarditis[46]. In April 2020, a group from Padua University reported the first case of biopsy-proven viral myocardial involvement in a COVID-19 patient presenting with cardiogenic shock[47]. However, to this date, there have been limited reports with pathological evidence of COVID-19 direct myocardial invasion. The potential for long-term evolution into forms of inflammatory cardiomyopathy remains also unclear.
More recently, with the large-scale use of several COVID-19 vaccines, the attention shifted to COVID-19 vaccination-related myocarditis. Despite not being reported as an adverse event in the first vaccine clinical trials, several cases were observed soon after vaccination campaigns began, particularly with mRNA technology vaccines. In May 2021, the Centers for Disease Control and Prevention released a report stating a possible association between COVID-19 vaccination and myocarditis, regarding the BNT162b2 (Pfizer-BioNTech) and the mRNA-1273 (Moderna) vaccines[48]. Later, an analysis of 2.5 million vaccinated people from Israel expected the incidence of post-vaccination myocarditis to be 2.13 cases per 100000 vaccinated persons[49]. In this cohort of patients who received the BNT162b2 mRNA vaccine (Pfizer-BioNTech), the highest myocarditis incidence was among young male patients, and after the second vaccine dose[49]. Another large English study showed similar findings, namely, an increased risk of myocarditis in patients who received the 2nd dose, and in those aged below 40[50]. Across published reports, vaccine-associated myocarditis was mostly a self-limiting disease of mild to moderate severity[50,51]. It has been hypothesized that post-vaccination myocarditis, similarly to COVID-19 myocarditis itself, can result from immune-mediated, virus-independent immunopathologic mechanisms[50,52].
Although this topic gathered intense attention from the social media, it is known that SARS-CoV-2 infection in non-vaccinated people carries a much greater risk of hospitalization and death than the vaccine associated risks[51]. Vaccines have proved to be highly effective at preventing symptomatic and severe disease, and remain, to this moment, the most powerful instrument to halt the effect of this dramatic pandemic on public health and social and economic domains.
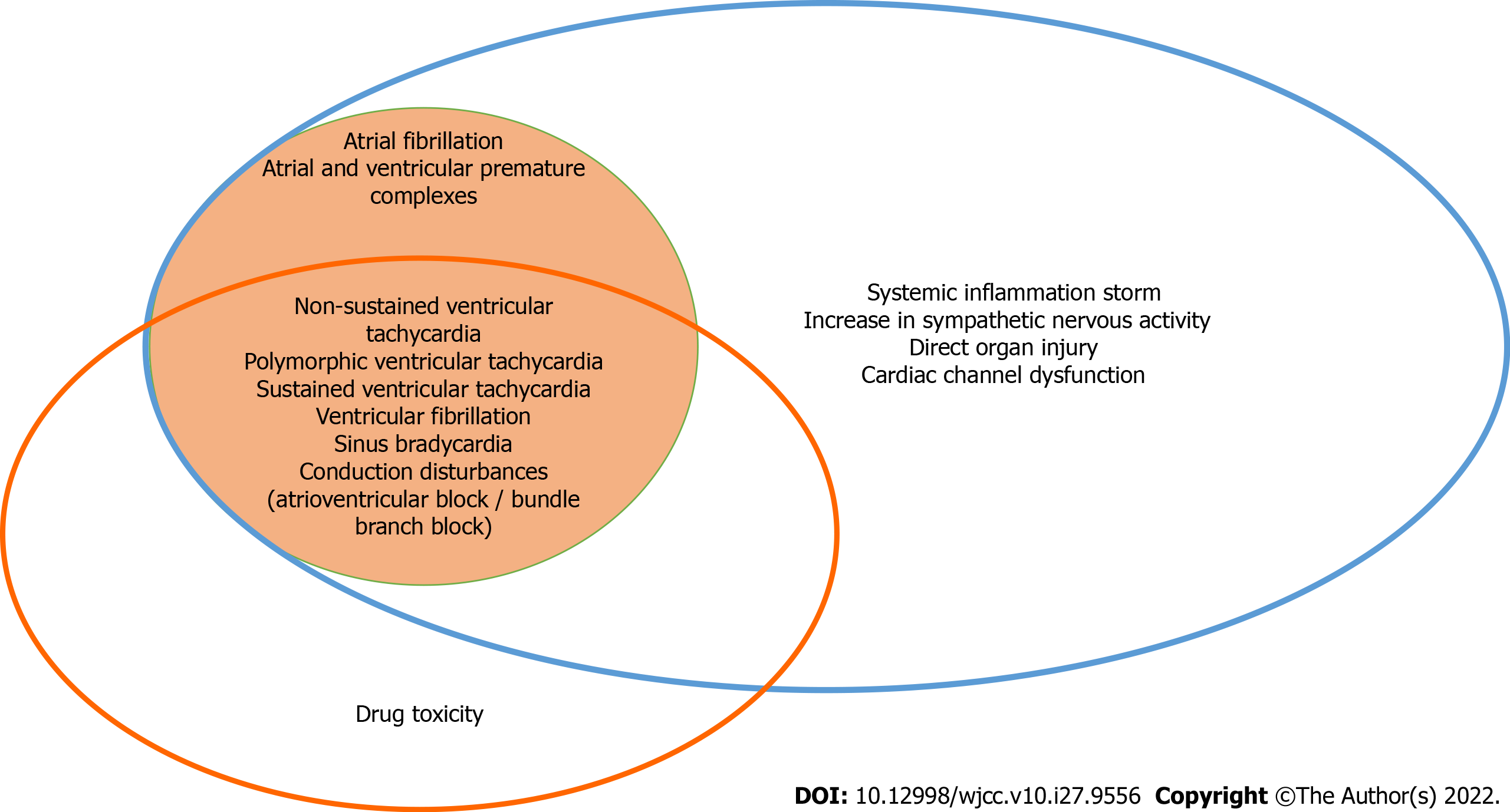
Cardiac arrhythmias may be not only the consequence of direct effects of SARS-CoV-2 infection but also the effects of the adverse reactions to medications used for the treatment of the infection and systemic illness (Figure 1). A metanalysis of 17 retrospective cohort studies of almost 6000 patients showed that the incidence for cardiac arrhythmias (Table 2) was 9.3%[53,54]. Table 3 describes the main mechanism of arrhythmogenicity in this setting[55].
| Supraventricular arrhythmias | Ventricular arrhythmias | Bradycardias |
| +Atrial fibrillation | +Ventricular premature complexes | +Sinus bradycardia |
| +Sinus tachycardia | +Non-sustained ventricular tachycardia | +Conduction disturbances (atrioventricular block / bundle branch block) |
| +Supraventricular tachycardia | +Polymorphic ventricular tachycardia (Torsade des pointes) | |
| +Atrial premature complexes | +Sustained ventricular tachycardia |
| Mechanisms of arrhythmogenicity |
| QT prolonging drugs (anti-coronavirus disease 2019 pharmacotherapies/antibiotic-associated diarrheas/other agents) |
| Drug-drug interactions |
| Previous heart rhythm conditions (long QT and Brugada syndrome) |
| Acute myocardial injury/myocarditis |
| Hypoxia |
| Systemic inflammation |
| Autonomic dysfunction (sympathetic/parasympathetic) |
| Electrolyte abnormalities |
| Cardiovascular comorbidities (hypertension, coronary artery disease, and cardiomyopathy) |
Atrial fibrillation was the most frequent cardiac arrhythmia detected in patients with SARS-CoV-2 infection. Virus-induced cardiac injury in the context of myocarditis, hypoxemia, systemic inflammatory response, and autonomic dysfunction are some of the mechanism implicated in the pathogenesis of ventricular fibrillation (VF) in this setting.
Ventricular arrhythmias (ventricular premature complexes, non-sustained and sustained ventricular tachycardia, polymorphic ventricular tachycardia, and ventricular fibrillation) may occur in the same context of the previous described AF, but also due to the proarrhythmic effects of COVID therapies. Ventricular arrhythmogenicity can be enhanced by acidosis, electrolyte disturbances, fever in Brugada patients, and pre-existing or acquired QT prolongation and induced by drugs.
In a retrospective cohort study of more than 1000 patients having a cTn level measured on admission, arrhythmias developed in 44 of the 170 (25.9%) that showed a high cTn level on admission, including six patients with ventricular tachycardia (VT) or VF. The mortality was 100% in this population[56].
Another report compared all the consecutive out-of-hospital cardiac arrests in 2 mo after the first documented case of COVID-19 in a region of Italy with those which occurred in the same time frame in 2019. The cumulative incidence of out-of-hospital cardiac arrest was 21 cases/100000 inhabitants, with a 52% rise as compared with the previous year (P < 0.001)[57]. Table 4 describes the main measures to prevent ventricular arrhythmias[2].
| Measures to prevent ventricular arrhythmias |
| Stop QT prolonging drugs in patients with baseline QTc > 500 ms or with known LQTS |
| Stop QT prolonging drugs when QTc increases to > 500 ms or if QTc is prolonged by > 60 ms compared to baseline measurement |
| Control effectively fever in Brugada patients |
| Avoid the use of chloroquine/hydroxychloroquine, macrolides, fluoroquinolones, and protease inhibitors in patients with known risk factors such as prolonged QTc and electrolyte abnormalities (hypokalemia and hypomagnesemia) |
| Avoid concomitant use of QT prolonging antiarrhythmic drugs, including class IA and class III agents |
| Avoid hypokalaemia and hypomagnesemia |
| Monitor QT via ECG or kardia mobile application |
Several agents used for potential prophylaxis and for treating SARS-CoV-2 infection prolong the QT interval and lead to polymorphic VT in the form of torsades de pointes (TDP) (Table 5)[2]. Some simple measures have been proposed to avoid TDP, like avoiding QT prolonging drugs in patients with baseline QTc > 500 ms or with known long QT syndrome, when QTc increases to > 500 ms or if QTc is prolonged by > 60 ms compared to baseline measurement, avoiding drugs of uncertain clinical effect against COVID-19 in patients with known risk factors such as prolonged QTc, and monitoring and avoiding hypomagnesemia, hypokalaemia, concomitant use of certain QT prolonging antiarrhythmic drugs, including class IA (procainamide and quinidine) and class III (sotalol and amiodarone), or bradycardia.
| Antiarrhythmics | Antibiotics | Antiviral agents | Antiemetics | Antipsychotics |
| +Class IA: Quinidine; Procainamide; +Class III: Amiodarone, Sotalol | +Chloroquine/Hydroxychloroquine; +Macrolides (Azithromycin); +Quinolones | +Lopinavir/Ritonavir; +Favipiravir; +Tocilizumab | +Domperidone | +Haloperidol |
A meta-analysis analyzing more than 4600 patients showed that patients with newly occurring arrhythmias and cardiac injury were at higher risk of requiring ICU admission or developing severe disease (relative risk [RR]: approximately 13, P < 0.001)[58].
Sinus tachycardia is the most frequent heart rhythm disturbance during the acute infection, due to fever, respiratory insufficiency, hemodynamic compromise, anxiety, and pain, among others. In the chronic context, the origin of sinus tachycardia is mainly due to physical deconditioning and autonomic dysfunction[59].
A study showed that the presence of atrial premature contractions, right bundle branch block, or intraventricular block increased the odds of death[60].
A German study showed that the pandemic was associated with an overall decline of device implantation rates of -2.6%, with a peak of almost -23%[61]. This situation was limited in time. The COVID-19 pandemic has led to a significant increase in the use of remote monitoring of cardiac implantable electronic devices[62].
Other pre-existing inflammatory conditions such as chronic coronary syndrome (CCS) may be associated with worse clinical outcomes in the context of COVID-19. In CCS, the cytokine storm triggered by SARS-CoV-2 infection may favour the rupture of a silent atheromatous plaque, leading to acute coronary syndrome and a sudden worsening of the patient's clinical condition[63].
CCS patients are generally at low risk of acute cardiovascular events, so diagnostic and/or interventional procedures can be deferred in most cases. In these patients, medical therapy should be optimized and/or intensified with the help of telemedicine. Remote clinical monitoring should be ensured to reassure patients and to detect possible changes in clinical status that may require hospitalization in selected patients with a high risk profile[14].
Therefore, during the first phase of the pandemic, it was necessary to continue follow-up in these patients, but with some restrictions.
Non-steroidal anti-inflammatory drugs may worsen the course of community-acquired pneumonia; on the contrary, their negative involvement in exacerbation of SARS-CoV-2 infections is not yet known. The possible effects of chronic aspirin therapy are not clearly understood. However, aspirin has only a very limited anti-inflammatory effect at the low dose administered in CCS. Therefore, CCS patients should not discontinue aspirin for secondary prevention[64].
In patients admitted with influenza or pneumonia, statin therapy has been variably associated with favorable outcomes. Alternatively, patients with COVID-19 have been reported to develop elevated liver enzymes or severe rhabdomyolysis and it may be advisable to temporarily suspend statin therapy[65,66].
During the initial phase of the COVID-19 pandemic, asymptomatic visits of patients with suspected CCS were often deferred as financial resources were allocated to the pandemic. In symptomatic patients with suspected coronary artery disease and a pre-test probability of 5%-15%, functional imaging examinations to detect myocardial ischaemia such as coronary computed tomography angiography were preferred to other imaging techniques such as stress echocardiography to avoid close contact between patients and medical staff[67].
After the initial phase, some changes were established in some local protocols, allowing cardiac testing of patients wearing facial masks, and this approach showed to be feasible, reaching the same levels of effort as in the prepandemic period for treadmill tests[68].
Association between SARS-CoV-2 infection and chronic heart failure (HF) may manifest as follows: (1) Patients with HF are at increased risk for severe and complicated course of COVID-19; (2) SARS-CoV-2 infection can exacerbate chronic HF; and (3) The COVID-19 pandemic is linked with dramatic changes in the delivery of outpatient care of HF patients
Patients with SARS-CoV-2 infection and pre-existing HF are more likely to be critically ill, with increased rates of ICU admission, renal replacement therapy, and mechanical ventilation[69]. Pre-existing HF represented an independent risk factor for mortality during COVID-19 hospitalization, with an adjusted odds ratio of 1.88 (95% confidence interval: 1.27-2.78)[70]. A large retrospective cohort study in United States veterans in the ambulatory setting testing positive for SARS-CoV-2 described that patients with COVID-19 and previously diagnosed HF had a greater risk of 30-d mortality and hospital admissions[71]. Interestingly, most of the clinical presentations of COVID-19 on top of advanced HF were dyspnoea and worsening of haemodynamic status instead of fever and other signs and symptoms of infection[72].
COVID-19 may cause or worsen HF through multiple mechanisms including myocardial ischemia or infarction, activation of the sympathetic nervous system, neurohormonal activation precipitating volume retention, elevations in pulmonary pressures, myocarditis, pulmonary embolism, stress cardiomyopathy, and inflammation leading to myocardial depression. The above mentioned mecha
Furthermore, it is not known whether the clinical course of COVID-19 differs depending on the left ventricular ejection fraction or background medication[73]. SARS-CoV-2 uses the angiotensin-converting enzyme (ACE)-2 receptors for cell entry and early data suggested that ACE inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) may upregulate ACE2, hypothetically increasing susceptibility to infection[74]. However, there is no clinical evidence linking ACEI/ARB treatment and susceptibility to infection or clinical course. Moreover, the available data do not support the discontinuation of ACEI/ARB in HF patients with COVID-19, which could increase the risk of death[75]. Consequently, it could be recommended that HF patients continue to take all prescribed guideline-appropriate medications (including ACEI, ARB, or sacubitril/valsartan) regardless of COVID-19[76]. Pneumococcal and influenza vaccination, as well as COVID-19 vaccination, when available, should be considered in patients with HF[77].
During COVID-19 pandemics, healthcare institutions have been forced to reconfigure the day-to-day routine ambulatory care. Adoption of restraint measures as an indirect impact of COVID-19 pandemics resulted in decreased hospital admissions for AHF and reduced number of self-referred AHF patients[78,79]. Altered medical care delivery was also confirmed in the multicentre, multinational PCHF-COVICAV registry, which demonstrated that COVID-19 impacted referral and hospitalizations of patients with acute HF and that HF was linked with a high mortality[80].
With an aim to diminish COVID-19 transmission during unnecessary hospitalizations of HF patients, and to maintain a healthy hospital workforce, medical facilities have broadly transitioned to noncontact care delivery methods for out-patient clinical care[81]. Several studies confirmed that this approach was able to keep a low proportion of admissions due to HF decompensation, without an increase in mortality. Results of these studies supported the implantation of telehealth outpatient visits in patients with HF and their safe incorporation into clinical practice[82].
Over the past decade, structural cardiac intervention has been increased worldwide, particularly transcatheter aortic valve implantation (TAVI), transcatheter mitral edge-to-edge repair (TEER), left atrial appendage closure (LAAC), atrial septal defect (ASD) closure, and patent foramen ovale (PFO) closure. This increase in structural cardiology is due to progressive improvements in technology, scientific support from several randomized controlled trials, and life expectancy.
The global COVID-19 pandemic profoundly impacted the treatment of patients eligible for coronary and structural cardiology interventions, as many hospitals had to adjust their internal organization and reallocate economic resources[83].
Structural interventions are usually elective and rarely performed in urgent cases as they require careful clinical and imaging examination such as CT and transthoracic (TT) or transoesophageal (TE) echocardiography. Hospitals changed their management, especially for all procedures requiring ventilators or intensive care. This forced restructuring led to significant constraints, delays, and, in some cases, deletion of procedures, especially in the first phase of the pandemic. TAVI, TEER, and LAAC procedures were reserved for urgent or highly symptomatic patients, while PFO closure, a procedure performed in the elective regime, was almost entirely suspended[84].
In addition, valvular heart disease (VHD) can exacerbate the course of COVID-19 and complicate treatment. An excess of mortality has been reported, particularly in patients with VHD infected with SARS-CoV-2; of 136 elderly patients with severe VHD (54% with aortic stenosis), 84.6% were treated conservatively, and the mortality rate was 41.8% after 30 d[85].
The first peak of the pandemic in England led to a considerable decrease in surgical valve interventions, 73%-76% for aortic valve replacement and 84%-85% for mitral valve replacement, while TAVI was less affected, with 35% and 18% decreases in April and May 2020, respectively[86].
The priority of valve interventions should therefore weigh the need for treatment, the immediate and short-term prognosis, available resources, and the risk to patients and healthcare professionals (HCPs) of hospital-acquired infections.
The ESC, the Society of Cardiovascular Angiography EuroIntervention and Interventions, and the Canadian Association of Interventional Cardiology have published their position statements on the management of structural heart surgery during the pandemic COVID-19[14,87,88].
As the pandemic continues, many centers have adopted a “minimalist” approach to TAVI, which is next day discharge (NDD) following transfemoral TAVI. NDD is a safe strategy for both balloon-expandable and self-expanding implants in selected cases[89].
It allows rapid discharge and avoids the risk of COVID-19 transfer to the patient in the hospital while ensuring patient treatment and medical care at a time of limited resources. NDD is unfortunately not performed in many centers, and for structural procedures, there is likely to be a long waiting list and a high burden for patients with symptomatic aortic stenosis when the pandemic recedes. Long waiting times can have a significant social and clinical impact, even in patients initially considered being at low risk of cardiovascular events. A possible solution should be to avoid standard referral to the ICU in centers with a high volume of procedures and expertise of surgeons using NDD, reserving it only for necessary cases, with all post-procedural care provided in a cardiovascular ward[90].
Treatment of mitral regurgitation (MR) differs according to aetiology and presentation. Chronic primary MR is usually well tolerated. In contrast, secondary MR is more variable and can lead to unstable HF syndromes that do not respond to medical treatment, especially in acute infections. Therefore, in the context of the pandemic, priority should be given to the treatment of acute primary MR complications (AMI or IE) and those with severe primary or secondary MR, which are symptomatic despite optimal medical therapy (OMT) and require hospitalization[91].
TEER requires general anesthesia (unlike transfemoral TAVI) and a longer TE echocardiograpy, exposing healthcare workers to the risk of COVID-19 transmission.
In contrast to patients affected by severe and symptomatic aortic stenosis, the majority of patients with severe MR can be managed by OMT, and indeed TEER has been deferred or reserved only for special cases during the pandemic. A web-based survey sent to EAPCI members from April 1 to 15, 2020 showed that TEER was discontinued in 73% of cases. A web-based survey sent to EAPCI members from April 1 to 15, 2020 showed that TEER was discontinued in 73% of cases[92].
Some changes in the classical organization of procedural protocols have been proposed to manage TEER better and avoid long waiting lists, especially in the postoperative phase. Same-day discharge (SDD) is increasingly practiced in larger centers, as shown by an observational study in which 89 patients who had an uncomplicated MitraClip inserted under moderate conscious sedation were discharged the same day without significant complications[93].
Chowdhury et al[94] developed a SDD protocol for patients treated during the pandemic by TEER. SDD reduced length of stay, resource utilization, and nosocomial SARS-CoV-2 infection risk. Patients were admitted 1 day before TEER, extubated in the cath lab, transferred to the recovery ward, and treated as outpatients within 2 h; then a TT echocardiogram was performed to assess outcomes and rule out pericardial effusion. If there was no immediate postoperative complication, they were discharged 3-4 h after the procedure. The protocol also included a telephone follow-up the next day and a follow-up at 2 wk and 30 d[94].
A new option for severe mitral regurgitation is transcatheter mitral valve replacement. This is a new and promising technique. Initial case series suggest that it is feasible and can lead to improvement in symptoms[95]. Numerous devices have been suggested in recent years, but unfortunately the use of this treatment has declined dramatically during the pandemic.
Other structural heart procedures, such as closure of PFO and ASD and LAAC, were unaffected in 9%, while complete cessation of activities was reported in 79%[92].
Two years after the pandemic, we are returning to normality, mainly due to the spread of the vaccine. Nevertheless, we have learned lessons from the first months of the pandemic that we are currently applying, such as avoiding extended hospital stays and promoting early discharge, avoiding intensive care unless absolutely necessary, and focusing on frequent follow-up.
Cardiovascular imaging (CVI) plays a pivotal role in the diagnostic pathway of both acute and chronic cardiovascular disease. The devastating impact of the COVID-19 pandemic on the treatment of patients with cardiovascular disease extended also to all cardiac imaging modalities, and likely contributed to delayed diagnosis of cardiovascular (CV) disease. Given its key role as a bedside test, and the “close contact” with the patient, echocardiography was the most affected cardiac imaging modality at the beginning[96]. Given the uncertainties of the disease since the beginning of the pandemic, the European Association of Cardiovascular Imaging issued specific recommendations for the use of cardiac imaging in this setting. Recommendations for the use of CVI were limited only to situations where it was likely to substantially change patient management or be lifesaving[96]. At that time, it was proposed that routine follow-up examinations and elective non-urgent procedures should be postponed or even cancelled and that more focused point-of-care examinations should be performed to minimize exposure time[96,97].
The INCAPS COVID survey was designed to assess the impact of COVID-19 on the use of cardiac imaging during the first lockdown. This survey reported a large reduction (45%-69%) in the total number of procedures in March and April 2020 in European countries[98]. Although we know that these numbers steadily recovered from Spring 2020, these results raised concern that the underutilization of CVI testing may have disrupted the implementation of primary and secondary strategies for CV disease prevention[99]. Patients deprived from prescribed CVI examinations could later present with more severe forms of disease, since they were probably not provided with appropriate care that would improve their long-term prognosis.
Furthermore, the COVID-19 pandemic also had a major impact on the well-being of healthcare professionals. A recently reported survey showed that CVI specialists experienced very high levels of stress, anxiety, and burnout during the COVID-19 pandemic, which highlights the psychological burden that these healthcare professionals have confronted and the importance to address proactively this problem[100].
Nevertheless, the pandemic also had some positive impact in cardiovascular imaging. We observed a broadened and more widespread use of pocket echocardiography and especially an increased awareness for the importance of lung ultrasound (LUS). Due to its high sensitivity, bedside availability, and steep learning curve, LUS already had previously well-established value as an important diagnostic and prognostic tool in heart failure and other acute cardiac care scenarios[101]. However, its imple
In summary, given the wide impact of the COVID-19 on cardiovascular imaging techniques, medium- and long-term consequences may be expected for some patients due to delayed diagnosis and treatment.
Today's technology, especially telemedicine, allows following patients with chronic cardiovascular diseases such as CCS and CHF. Some scientific societies focused on this approach due to the COVID-19 pandemic to suggest a better and wider use of telemedicine[103,104].
For known CCS patients, clinical follow-up should be conducted mainly via telemedicine. This would allow physicians to address most of the patient's concerns related to continuation or change of medical therapy. Possible occurrence/recurrence of unstable symptoms should be assessed as part of the patient's clinical history to weigh the need for hospitalization and diagnostic tests[14].
Several reports suggest a decrease in hospitalization rates for chronic HF in people without SARS-CoV-2 infection during the peak of the pandemic COVID-19 compared to 2019[22].
For the duration of the COVID-19 outbreak, patients with chronic HF should be advised to closely follow protective measures to prevent disease transmission. Outpatients with HF should avoid routine, non-urgent hospital visits, which has led to an increase in telemedicine and remote monitoring work. The increased use of telemedicine has been promoted to minimize infection risk and ensure continuity of care and timely optimization of medical treatment. Several papers have reported the effective use of this technology in medical consultation, treatment adjustment, and follow-up of outpatients HF during the COVID-19 outbreak[105]. Telemedicine has become an important tool for delivering of HF care to ensure continuity of care for the chronically ill while maintaining the safety of patients and HCPs[106].
The changes in hospitals during the pandemic have helped reduce barriers to telemedicine and facilitate its widespread adoption. The chronic heart failure-CePPORT (Canadian e-Platform to Promote Behavioral Self-Management in Chronic Heart Failure Trial) trial highlight an approach to supporting patients with chronic HF[107].
What this change represents for the future of HF management and the provision of HF services in outpatient scenarios worldwide remains warmly debated[108].
Nowadays, far-reaching projects have been proposed for the future management of HF to improve access to care by overcoming transport barriers, the excessive cost of clinical appointments, patient education, and remote home monitoring in more patients tailored ways[18].
The prior presence of cardiovascular disease is an important risk factor for a severe clinical course of COVID-19 and is associated with adverse outcomes. COVID-19 also has been associated with a direct damage of the cardiovascular system.
Although the pandemic seems to be near to its end, an effort must be made to enable the diagnosis of non-COVID-19 conditions that were overlooked during this period as these non-COVID-19 conditions untreated could explain the excess of death during this dramatic period. This should be a priority for policymakers while planning the recovery from these hazardous times. The lessons learnt during this period should serve as preparation for future challenges or impending pandemics that could be again a deadly combination as the COVID-19 was with the cardiovascular diseases.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Medicine, research and experimental
Country/Territory of origin: Spain
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: El Sayed S, Egypt; Sun XD, China S-Editor: Wang LL L-Editor: Wang TQ P-Editor: Wang LL
| 1. | COVID-19 Excess Mortality Collaborators. Estimating excess mortality due to the COVID-19 pandemic: a systematic analysis of COVID-19-related mortality, 2020-21. Lancet. 2022;399:1513-1536. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 430] [Cited by in RCA: 964] [Article Influence: 321.3] [Reference Citation Analysis (2)] |
| 2. | Driggin E, Madhavan MV, Bikdeli B, Chuich T, Laracy J, Biondi-Zoccai G, Brown TS, Der Nigoghossian C, Zidar DA, Haythe J, Brodie D, Beckman JA, Kirtane AJ, Stone GW, Krumholz HM, Parikh SA. Cardiovascular Considerations for Patients, Health Care Workers, and Health Systems During the COVID-19 Pandemic. J Am Coll Cardiol. 2020;75:2352-2371. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1381] [Cited by in RCA: 1366] [Article Influence: 273.2] [Reference Citation Analysis (0)] |
| 3. | Li B, Yang J, Zhao F, Zhi L, Wang X, Liu L, Bi Z, Zhao Y. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clin Res Cardiol. 2020;109:531-538. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1374] [Cited by in RCA: 1237] [Article Influence: 247.4] [Reference Citation Analysis (0)] |
| 4. | Burger AL, Kaufmann CC, Jäger B, Pogran E, Ahmed A, Wojta J, Farhan S, Huber K. Direct cardiovascular complications and indirect collateral damage during the COVID-19 pandemic : A review. Wien Klin Wochenschr. 2021;133:1289-1297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 5. | Rusu I, Turlacu M, Micheu MM. Acute myocardial injury in patients with COVID-19: Possible mechanisms and clinical implications. World J Clin Cases. 2022;10:762-776. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 15] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (2)] |
| 6. | Lodigiani C, Iapichino G, Carenzo L, Cecconi M, Ferrazzi P, Sebastian T, Kucher N, Studt JD, Sacco C, Bertuzzi A, Sandri MT, Barco S; Humanitas COVID-19 Task Force. Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thromb Res. 2020;191:9-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1298] [Cited by in RCA: 1535] [Article Influence: 307.0] [Reference Citation Analysis (0)] |
| 7. | Piazza G, Campia U, Hurwitz S, Snyder JE, Rizzo SM, Pfeferman MB, Morrison RB, Leiva O, Fanikos J, Nauffal V, Almarzooq Z, Goldhaber SZ. Registry of Arterial and Venous Thromboembolic Complications in Patients With COVID-19. J Am Coll Cardiol. 2020;76:2060-2072. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 152] [Cited by in RCA: 211] [Article Influence: 42.2] [Reference Citation Analysis (0)] |
| 8. | Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054-1062. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17476] [Cited by in RCA: 18199] [Article Influence: 3639.8] [Reference Citation Analysis (0)] |
| 9. | Coronavirus Disease 2019 (COVID-19) Treatment Guidelines Bethesda (MD), 2021. [cited 20 April 2022]. Available from: https://www.covid19treatmentguidelines.nih.gov/. |
| 10. | Belani P, Schefflein J, Kihira S, Rigney B, Delman BN, Mahmoudi K, Mocco J, Majidi S, Yeckley J, Aggarwal A, Lefton D, Doshi AH. COVID-19 Is an Independent Risk Factor for Acute Ischemic Stroke. AJNR Am J Neuroradiol. 2020;41:1361-1364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 137] [Article Influence: 27.4] [Reference Citation Analysis (0)] |
| 11. | Katsoularis I, Fonseca-Rodríguez O, Farrington P, Lindmark K, Fors Connolly AM. Risk of acute myocardial infarction and ischaemic stroke following COVID-19 in Sweden: a self-controlled case series and matched cohort study. Lancet. 2021;398:599-607. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 159] [Cited by in RCA: 279] [Article Influence: 69.8] [Reference Citation Analysis (0)] |
| 12. | Varga Z, Flammer AJ, Steiger P, Haberecker M, Andermatt R, Zinkernagel AS, Mehra MR, Schuepbach RA, Ruschitzka F, Moch H. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395:1417-1418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4227] [Cited by in RCA: 4582] [Article Influence: 916.4] [Reference Citation Analysis (0)] |
| 13. | Hamadeh A, Aldujeli A, Briedis K, Tecson KM, Sanz-Sánchez J, Al Dujeili M, Al-Obeidi A, Diez JL, Žaliūnas R, Stoler RC, McCullough PA. Characteristics and Outcomes in Patients Presenting With COVID-19 and ST-Segment Elevation Myocardial Infarction. Am J Cardiol. 2020;131:1-6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 76] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 14. | Task Force for the management of COVID-19 of the European Society of Cardiology. ESC guidance for the diagnosis and management of cardiovascular disease during the COVID-19 pandemic: part 2-care pathways, treatment, and follow-up. Eur Heart J. 2022;43:1059-1103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 95] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 15. | Jabri A, Kalra A, Kumar A, Alameh A, Adroja S, Bashir H, Nowacki AS, Shah R, Khubber S, Kanaa'N A, Hedrick DP, Sleik KM, Mehta N, Chung MK, Khot UN, Kapadia SR, Puri R, Reed GW. Incidence of Stress Cardiomyopathy During the Coronavirus Disease 2019 Pandemic. JAMA Netw Open. 2020;3:e2014780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 165] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 16. | Sokolski M, Reszka K, Suchocki T, Adamik B, Doroszko A, Drobnik J, Gorka-Dynysiewicz J, Jedrzejczyk M, Kaliszewski K, Kilis-Pstrusinska K, Konopska B, Kopec A, Larysz A, Lis W, Matera-Witkiewicz A, Pawlik-Sobecka L, Rosiek-Biegus M, Sokolska JM, Sokolowski J, Zapolska-Tomasiewicz A, Protasiewicz M, Madziarska K, Jankowska EA. History of Heart Failure in Patients Hospitalized Due to COVID-19: Relevant Factor of In-Hospital Complications and All-Cause Mortality up to Six Months. J Clin Med. 2022;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 17. | Zhang Y, Coats AJS, Zheng Z, Adamo M, Ambrosio G, Anker SD, Butler J, Xu D, Mao J, Khan MS, Bai L, Mebazaa A, Ponikowski P, Tang Q, Ruschitzka F, Seferovic P, Tschöpe C, Zhang S, Gao C, Zhou S, Senni M, Zhang J, Metra M. Management of heart failure patients with COVID-19: a joint position paper of the Chinese Heart Failure Association & National Heart Failure Committee and the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2020;22:941-956. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 95] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 18. | DeFilippis EM, Reza N, Donald E, Givertz MM, Lindenfeld J, Jessup M. Considerations for Heart Failure Care During the COVID-19 Pandemic. JACC Heart Fail. 2020;8:681-691. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 86] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 19. | Lippi G, Lavie CJ, Sanchis-Gomar F. Cardiac troponin I in patients with coronavirus disease 2019 (COVID-19): Evidence from a meta-analysis. Prog Cardiovasc Dis. 2020;63:390-391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 406] [Cited by in RCA: 477] [Article Influence: 95.4] [Reference Citation Analysis (0)] |
| 20. | Basso C, Leone O, Rizzo S, De Gaspari M, van der Wal AC, Aubry MC, Bois MC, Lin PT, Maleszewski JJ, Stone JR. Pathological features of COVID-19-associated myocardial injury: a multicentre cardiovascular pathology study. Eur Heart J. 2020;41:3827-3835. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 221] [Cited by in RCA: 353] [Article Influence: 88.3] [Reference Citation Analysis (0)] |
| 21. | Tomasoni D, Italia L, Adamo M, Inciardi RM, Lombardi CM, Solomon SD, Metra M. COVID-19 and heart failure: from infection to inflammation and angiotensin II stimulation. Searching for evidence from a new disease. Eur J Heart Fail. 2020;22:957-966. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 140] [Cited by in RCA: 192] [Article Influence: 38.4] [Reference Citation Analysis (0)] |
| 22. | Andersson C, Gerds T, Fosbøl E, Phelps M, Andersen J, Lamberts M, Holt A, Butt JH, Madelaire C, Gislason G, Torp-Pedersen C, Køber L, Schou M. Incidence of New-Onset and Worsening Heart Failure Before and After the COVID-19 Epidemic Lockdown in Denmark: A Nationwide Cohort Study. Circ Heart Fail. 2020;13:e007274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 85] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 23. | Bromage DI, Cannatà A, Rind IA, Gregorio C, Piper S, Shah AM, McDonagh TA. The impact of COVID-19 on heart failure hospitalization and management: report from a Heart Failure Unit in London during the peak of the pandemic. Eur J Heart Fail. 2020;22:978-984. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 157] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 24. | Creel-Bulos C, Hockstein M, Amin N, Melhem S, Truong A, Sharifpour M. Acute Cor Pulmonale in Critically Ill Patients with Covid-19. N Engl J Med. 2020;382:e70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 158] [Article Influence: 31.6] [Reference Citation Analysis (0)] |
| 25. | Kim J, Volodarskiy A, Sultana R, Pollie MP, Yum B, Nambiar L, Tafreshi R, Mitlak HW, RoyChoudhury A, Horn EM, Hriljac I, Narula N, Kim S, Ndhlovu L, Goyal P, Safford MM, Shaw L, Devereux RB, Weinsaft JW. Prognostic Utility of Right Ventricular Remodeling Over Conventional Risk Stratification in Patients With COVID-19. J Am Coll Cardiol. 2020;76:1965-1977. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 84] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 26. | Xie Y, Xu E, Bowe B, Al-Aly Z. Long-term cardiovascular outcomes of COVID-19. Nat Med. 2022;28:583-590. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1452] [Cited by in RCA: 1194] [Article Influence: 398.0] [Reference Citation Analysis (0)] |
| 27. | Zeymer U, Gitt A, Thiele H. [COVID-19 pandemic : Effects on clinical care of cardiovascular patients in spring 2020]. Herz. 2021;46:115-119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 28. | Garcia S, Albaghdadi MS, Meraj PM, Schmidt C, Garberich R, Jaffer FA, Dixon S, Rade JJ, Tannenbaum M, Chambers J, Huang PP, Henry TD. Reduction in ST-Segment Elevation Cardiac Catheterization Laboratory Activations in the United States During COVID-19 Pandemic. J Am Coll Cardiol. 2020;75:2871-2872. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 754] [Cited by in RCA: 908] [Article Influence: 181.6] [Reference Citation Analysis (0)] |
| 29. | Mafham MM, Spata E, Goldacre R, Gair D, Curnow P, Bray M, Hollings S, Roebuck C, Gale CP, Mamas MA, Deanfield JE, de Belder MA, Luescher TF, Denwood T, Landray MJ, Emberson JR, Collins R, Morris EJA, Casadei B, Baigent C. COVID-19 pandemic and admission rates for and management of acute coronary syndromes in England. Lancet. 2020;396:381-389. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 436] [Cited by in RCA: 492] [Article Influence: 98.4] [Reference Citation Analysis (0)] |
| 30. | Mohammad MA, Koul S, Olivecrona GK, Gӧtberg M, Tydén P, Rydberg E, Scherstén F, Alfredsson J, Vasko P, Omerovic E, Angerås O, Fröbert O, Calais F, Völz S, Ulvenstam A, Venetsanos D, Yndigegn T, Oldgren J, Sarno G, Grimfjärd P, Persson J, Witt N, Ostenfeld E, Lindahl B, James SK, Erlinge D. Incidence and outcome of myocardial infarction treated with percutaneous coronary intervention during COVID-19 pandemic. Heart. 2020;106:1812-1818. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 31. | Solomon MD, McNulty EJ, Rana JS, Leong TK, Lee C, Sung SH, Ambrosy AP, Sidney S, Go AS. The Covid-19 Pandemic and the Incidence of Acute Myocardial Infarction. N Engl J Med. 2020;383:691-693. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 465] [Cited by in RCA: 502] [Article Influence: 100.4] [Reference Citation Analysis (0)] |
| 32. | Xiang D, Xiang X, Zhang W, Yi S, Zhang J, Gu X, Xu Y, Huang K, Su X, Yu B, Wang Y, Fang W, Huo Y, Ge J. Management and Outcomes of Patients With STEMI During the COVID-19 Pandemic in China. J Am Coll Cardiol. 2020;76:1318-1324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 161] [Cited by in RCA: 169] [Article Influence: 33.8] [Reference Citation Analysis (0)] |
| 33. | Kiss P, Carcel C, Hockham C, Peters SAE. The impact of the COVID-19 pandemic on the care and management of patients with acute cardiovascular disease: a systematic review. Eur Heart J Qual Care Clin Outcomes. 2021;7:18-27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 116] [Article Influence: 29.0] [Reference Citation Analysis (0)] |
| 34. | De Luca G, Algowhary M, Uguz B, Oliveira DC, Ganyukov V, Zimbakov Z, Cercek M, Jensen LO, Loh PH, Calmac L, Roura-Ferrer G, Quadros A, Milewski M, Scotto di Uccio F, von Birgelen C, Versaci F, Ten Berg J, Casella G, Wong ASL, Kala P, Diez Gil JL, Carrillo X, Dirksen MT, Becerra-Muñoz VM, Kang-Yin Lee M, Juzar DA, de Moura Joaquim R, Paladino R, Milicic D, Davlouros P, Bakraceski N, Zilio F, Donazzan L, Kraaijeveld AO, Galasso G, Lux A, Marinucci L, Guiducci V, Menichelli M, Scoccia A, Yamac A, Ugur Mert K, Flores Rios X, Kovarnik T, Kidawa M, Moreu J, Flavien V, Fabris E, Lozano Martìnez-Luengas I, Boccalatte M, Bosa Ojeda F, Arellano-Serrano C, Caiazzo G, Cirrincione G, Kao HL, Sanchis Fores J, Vignali L, Pereira H, Manzo-Silberman S, Ordonez S, Özkan AA, Scheller B, Lehtola H, Teles R, Mantis C, Ylitalo A, Brum Silveira JA, Zoni R, Bessonov I, Savonitto S, Kochiadakis G, Alexopoulos D, Uribe C, Kanakakis J, Faurie B, Gabrielli G, Gutiérrez A, Bachini JP, Rocha A, Tam FC, Rodriguez A, Lukito A, Saint-Joy V, Pessah G, Tuccillo B, Cortese G, Parodi G, Bouraghda MA, Kedhi E, Lamelas P, Suryapranata H, Nardin M, Verdoia M; ISACS-STEMI COVID-19; Collaborators. COVID-19 pandemic, mechanical reperfusion and 30-day mortality in ST elevation myocardial infarction. Heart. 2022;108:458-466. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 32] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 35. | Kwok CS, Gale CP, Kinnaird T, Curzen N, Ludman P, Kontopantelis E, Wu J, Denwood T, Fazal N, Deanfield J, de Belder MA, Mamas M. Impact of COVID-19 on percutaneous coronary intervention for ST-elevation myocardial infarction. Heart. 2020;106:1805-1811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 76] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 36. | Chieffo A, Stefanini GG, Price S, Barbato E, Tarantini G, Karam N, Moreno R, Buchanan GL, Gilard M, Halvorsen S, Huber K, James S, Neumann FJ, Möllmann H, Roffi M, Tavazzi G, Ferré JM, Windecker S, Dudek D, Baumbach A. EAPCI Position Statement on Invasive Management of Acute Coronary Syndromes during the COVID-19 pandemic. EuroIntervention. 2020;16:233-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 37. | Welt FGP, Shah PB, Aronow HD, Bortnick AE, Henry TD, Sherwood MW, Young MN, Davidson LJ, Kadavath S, Mahmud E, Kirtane AJ; American College of Cardiology’s Interventional Council and the Society for Cardiovascular Angiography and Interventions. Catheterization Laboratory Considerations During the Coronavirus (COVID-19) Pandemic: From the ACC's Interventional Council and SCAI. J Am Coll Cardiol. 2020;75:2372-2375. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 257] [Cited by in RCA: 369] [Article Influence: 73.8] [Reference Citation Analysis (0)] |
| 38. | Bollmann A, Hohenstein S, Meier-Hellmann A, Kuhlen R, Hindricks G. Emergency hospital admissions and interventional treatments for heart failure and cardiac arrhythmias in Germany during the Covid-19 outbreak: insights from the German-wide Helios hospital network. Eur Heart J Qual Care Clin Outcomes. 2020;6:221-222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 39. | Woolf SH, Chapman DA, Sabo RT, Weinberger DM, Hill L, Taylor DDH. Excess Deaths From COVID-19 and Other Causes, March-July 2020. JAMA. 2020;324:1562-1564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 221] [Article Influence: 44.2] [Reference Citation Analysis (0)] |
| 40. | Scholz KH, Lengenfelder B, Thilo C, Jeron A, Stefanow S, Janssens U, Bauersachs J, Schulze PC, Winter KD, Schröder J, Vom Dahl J, von Beckerath N, Seidl K, Friede T, Meyer T. Impact of COVID-19 outbreak on regional STEMI care in Germany. Clin Res Cardiol. 2020;109:1511-1521. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 41. | Rashid Hons M, Gale Hons CP, Curzen Hons N, Ludman Hons P, De Belder Hons M, Timmis Hons A, Mohamed Hons MO, Lüscher Hons TF, Hains Hons J, Wu J, Shoaib A, Kontopantelis E, Roebuck C, Denwood T, Deanfield J, Mamas MA. Impact of Coronavirus Disease 2019 Pandemic on the Incidence and Management of Out-of-Hospital Cardiac Arrest in Patients Presenting With Acute Myocardial Infarction in England. J Am Heart Assoc. 2020;9:e018379. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 42. | Wu J, Mamas MA, Mohamed MO, Kwok CS, Roebuck C, Humberstone B, Denwood T, Luescher T, de Belder MA, Deanfield JE, Gale CP. Place and causes of acute cardiovascular mortality during the COVID-19 pandemic. Heart. 2021;107:113-119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 132] [Cited by in RCA: 129] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 43. | Nishiga M, Wang DW, Han Y, Lewis DB, Wu JC. COVID-19 and cardiovascular disease: from basic mechanisms to clinical perspectives. Nat Rev Cardiol. 2020;17:543-558. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 653] [Cited by in RCA: 886] [Article Influence: 177.2] [Reference Citation Analysis (0)] |
| 44. | Cenko E, Badimon L, Bugiardini R, Claeys MJ, De Luca G, de Wit C, Derumeaux G, Dorobantu M, Duncker DJ, Eringa EC, Gorog DA, Hassager C, Heinzel FR, Huber K, Manfrini O, Milicic D, Oikonomou E, Padro T, Trifunovic-Zamaklar D, Vasiljevic-Pokrajcic Z, Vavlukis M, Vilahur G, Tousoulis D. Cardiovascular disease and COVID-19: a consensus paper from the ESC Working Group on Coronary Pathophysiology & Microcirculation, ESC Working Group on Thrombosis and the Association for Acute CardioVascular Care (ACVC), in collaboration with the European Heart Rhythm Association (EHRA). Cardiovasc Res. 2021;117:2705-2729. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 97] [Article Influence: 24.3] [Reference Citation Analysis (0)] |
| 45. | Pirzada A, Mokhtar AT, Moeller AD. COVID-19 and Myocarditis: What Do We Know So Far? CJC Open. 2020;2:278-285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 79] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 46. | Caforio AL, Pankuweit S, Arbustini E, Basso C, Gimeno-Blanes J, Felix SB, Fu M, Heliö T, Heymans S, Jahns R, Klingel K, Linhart A, Maisch B, McKenna W, Mogensen J, Pinto YM, Ristic A, Schultheiss HP, Seggewiss H, Tavazzi L, Thiene G, Yilmaz A, Charron P, Elliott PM; European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2013;34:2636-2648. |
| 47. | Tavazzi G, Pellegrini C, Maurelli M, Belliato M, Sciutti F, Bottazzi A, Sepe PA, Resasco T, Camporotondo R, Bruno R, Baldanti F, Paolucci S, Pelenghi S, Iotti GA, Mojoli F, Arbustini E. Myocardial localization of coronavirus in COVID-19 cardiogenic shock. Eur J Heart Fail. 2020;22:911-915. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 608] [Cited by in RCA: 745] [Article Influence: 149.0] [Reference Citation Analysis (0)] |
| 48. | Boehmer TK, Kompaniyets L, Lavery AM, Hsu J, Ko JY, Yusuf H, Romano SD, Gundlapalli AV, Oster ME, Harris AM. Association Between COVID-19 and Myocarditis Using Hospital-Based Administrative Data - United States, March 2020-January 2021. MMWR Morb Mortal Wkly Rep. 2021;70:1228-1232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 205] [Article Influence: 51.3] [Reference Citation Analysis (0)] |
| 49. | Witberg G, Barda N, Hoss S, Richter I, Wiessman M, Aviv Y, Grinberg T, Auster O, Dagan N, Balicer RD, Kornowski R. Myocarditis after Covid-19 Vaccination in a Large Health Care Organization. N Engl J Med. 2021;385:2132-2139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 352] [Cited by in RCA: 444] [Article Influence: 111.0] [Reference Citation Analysis (0)] |
| 50. | Patone M, Mei XW, Handunnetthi L, Dixon S, Zaccardi F, Shankar-Hari M, Watkinson P, Khunti K, Harnden A, Coupland CAC, Channon KM, Mills NL, Sheikh A, Hippisley-Cox J. Risks of myocarditis, pericarditis, and cardiac arrhythmias associated with COVID-19 vaccination or SARS-CoV-2 infection. Nat Med. 2022;28:410-422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 409] [Cited by in RCA: 404] [Article Influence: 134.7] [Reference Citation Analysis (0)] |
| 51. | Heymans S, Cooper LT. Myocarditis after COVID-19 mRNA vaccination: clinical observations and potential mechanisms. Nat Rev Cardiol. 2022;19:75-77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 180] [Article Influence: 60.0] [Reference Citation Analysis (0)] |
| 52. | Dorward DA, Russell CD, Um IH, Elshani M, Armstrong SD, Penrice-Randal R, Millar T, Lerpiniere CEB, Tagliavini G, Hartley CS, Randle NP, Gachanja NN, Potey PMD, Dong X, Anderson AM, Campbell VL, Duguid AJ, Al Qsous W, BouHaidar R, Baillie JK, Dhaliwal K, Wallace WA, Bellamy COC, Prost S, Smith C, Hiscox JA, Harrison DJ, Lucas CD. Tissue-Specific Immunopathology in Fatal COVID-19. Am J Respir Crit Care Med. 2021;203:192-201. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 240] [Cited by in RCA: 224] [Article Influence: 56.0] [Reference Citation Analysis (0)] |
| 53. | Lavelle MP, Desai AD, Wan EY. Arrhythmias in the COVID-19 patient. Heart Rhythm O2. 2022;3:8-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 54. | Kunutsor SK, Laukkanen JA. Cardiovascular complications in COVID-19: A systematic review and meta-analysis. J Infect. 2020;81:e139-e141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 55. | Lazzerini PE, Boutjdir M, Capecchi PL. COVID-19, Arrhythmic Risk, and Inflammation: Mind the Gap! Circulation. 2020;142:7-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 187] [Article Influence: 37.4] [Reference Citation Analysis (0)] |
| 56. | Si D, Du B, Ni L, Yang B, Sun H, Jiang N, Liu G, Massé S, Jin L, Nanthakumar J, Bhaskaran A, Yang P, Nanthakumar K. Death, discharge and arrhythmias among patients with COVID-19 and cardiac injury. CMAJ. 2020;192:E791-E798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 57. | Baldi E, Sechi GM, Mare C, Canevari F, Brancaglione A, Primi R, Klersy C, Palo A, Contri E, Ronchi V, Beretta G, Reali F, Parogni P, Facchin F, Rizzi U, Bussi D, Ruggeri S, Oltrona Visconti L, Savastano S; Lombardia CARe researchers. COVID-19 kills at home: the close relationship between the epidemic and the increase of out-of-hospital cardiac arrests. Eur Heart J. 2020;41:3045-3054. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 185] [Cited by in RCA: 177] [Article Influence: 35.4] [Reference Citation Analysis (0)] |
| 58. | Li X, Pan X, Li Y, An N, Xing Y, Yang F, Tian L, Sun J, Gao Y, Shang H. Cardiac injury associated with severe disease or ICU admission and death in hospitalized patients with COVID-19: a meta-analysis and systematic review. Crit Care. 2020;24:468. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 52] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 59. | Chen Q, Xu L, Dai Y, Ling Y, Mao J, Qian J, Zhu W, Di W, Ge J. Cardiovascular manifestations in severe and critical patients with COVID-19. Clin Cardiol. 2020;43:796-802. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 63] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 60. | McCullough SA, Goyal P, Krishnan U, Choi JJ, Safford MM, Okin PM. Electrocardiographic Findings in Coronavirus Disease-19: Insights on Mortality and Underlying Myocardial Processes. J Card Fail. 2020;26:626-632. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 76] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 61. | Schwab JO, Wiese J, Hauser T. The influence of the 2020 COVID-19 pandemic on the implantation rates of cardiac implantable electronic devices in Germany: changes between 2020 Q1-Q3 and 2019 Q1-Q3. Eur Heart J Qual Care Clin Outcomes. 2022;8:104-112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 62. | Simovic S, Providencia R, Barra S, Kircanski B, Guerra JM, Conte G, Duncker D, Marijon E, Anic A, Boveda S. The use of remote monitoring of cardiac implantable devices during the COVID-19 pandemic: an EHRA physician survey. Europace. 2022;24:473-480. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 36] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 63. | Buicu AL, Cernea S, Benedek I, Buicu CF, Benedek T. Systemic Inflammation and COVID-19 Mortality in Patients with Major Noncommunicable Diseases: Chronic Coronary Syndromes, Diabetes and Obesity. J Clin Med. 2021;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 41] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 64. | Basille D, Plouvier N, Trouve C, Duhaut P, Andrejak C, Jounieaux V. Non-steroidal Anti-inflammatory Drugs may Worsen the Course of Community-Acquired Pneumonia: A Cohort Study. Lung. 2017;195:201-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 65. | Douglas I, Evans S, Smeeth L. Effect of statin treatment on short term mortality after pneumonia episode: cohort study. BMJ. 2011;342:d1642. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 62] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 66. | Xu L, Liu J, Lu M, Yang D, Zheng X. Liver injury during highly pathogenic human coronavirus infections. Liver Int. 2020;40:998-1004. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 622] [Cited by in RCA: 575] [Article Influence: 115.0] [Reference Citation Analysis (0)] |
| 67. | Task Force for the management of COVID-19 of the European Society of Cardiology. European Society of Cardiology guidance for the diagnosis and management of cardiovascular disease during the COVID-19 pandemic: part 1-epidemiology, pathophysiology, and diagnosis. Cardiovasc Res. 2021;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 32] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 68. | Barbeito-Caamaño C, Bouzas-Mosquera A, Peteiro J, López-Vázquez D, Quintas-Guzmán M, Varela-Cancelo A, Martínez-Ruiz D, Yañez-Wonenburger JC, Piñeiro-Portela M, Vázquez-Rodríguez JM. Exercise testing in COVID-19 era: Clinical profile, results and feasibility wearing a facemask. Eur J Clin Invest. 2021;51:e13509. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 69. | Pillai A, Lawson B. Coronavirus disease 2019 and cardiovascular diseases: collateral damage? Curr Opin Anaesthesiol. 2022;35:5-11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 70. | Bhatt AS, Jering KS, Vaduganathan M, Claggett BL, Cunningham JW, Rosenthal N, Signorovitch J, Thune JJ, Vardeny O, Solomon SD. Clinical Outcomes in Patients With Heart Failure Hospitalized With COVID-19. JACC Heart Fail. 2021;9:65-73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 100] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 71. | Rumery K, Seo A, Jiang L, Choudhary G, Shah NR, Rudolph JL, Wu WC, Erqou S. Outcomes of coronavirus disease-2019 among veterans with pre-existing diagnosis of heart failure. ESC Heart Fail. 2021;8:2338-2344. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 72. | Bocchi EA, Lima IGCV, Biselli B, Salemi VMC, Ferreira SMA, Chizzola PR, Munhoz RT, Pessoa RS, Cardoso FAM, Bello MVO, Hajjar LA, Gomes BR. Worsening of heart failure by coronavirus disease 2019 is associated with high mortality. ESC Heart Fail. 2021;8:943-952. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 73. | Vaduganathan M, Vardeny O, Michel T, McMurray JJV, Pfeffer MA, Solomon SD. Renin-Angiotensin-Aldosterone System Inhibitors in Patients with Covid-19. N Engl J Med. 2020;382:1653-1659. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1484] [Cited by in RCA: 1565] [Article Influence: 313.0] [Reference Citation Analysis (0)] |
| 74. | Furuhashi M, Moniwa N, Mita T, Fuseya T, Ishimura S, Ohno K, Shibata S, Tanaka M, Watanabe Y, Akasaka H, Ohnishi H, Yoshida H, Takizawa H, Saitoh S, Ura N, Shimamoto K, Miura T. Urinary angiotensin-converting enzyme 2 in hypertensive patients may be increased by olmesartan, an angiotensin II receptor blocker. Am J Hypertens. 2015;28:15-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 198] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 75. | Lafaurie M, Martin-Blondel G, Delobel P, Charpentier S, Sommet A, Moulis G. Outcome of patients hospitalized for COVID-19 and exposure to angiotensin-converting enzyme inhibitors and angiotensin-receptor blockers in France: results of the ACE-CoV study. Fundam Clin Pharmacol. 2021;35:194-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 76. | Seferovic PM, Ponikowski P, Anker SD, Bauersachs J, Chioncel O, Cleland JGF, de Boer RA, Drexel H, Ben Gal T, Hill L, Jaarsma T, Jankowska EA, Anker MS, Lainscak M, Lewis BS, McDonagh T, Metra M, Milicic D, Mullens W, Piepoli MF, Rosano G, Ruschitzka F, Volterrani M, Voors AA, Filippatos G, Coats AJS. Clinical practice update on heart failure 2019: pharmacotherapy, procedures, devices and patient management. An expert consensus meeting report of the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2019;21:1169-1186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 403] [Cited by in RCA: 438] [Article Influence: 73.0] [Reference Citation Analysis (0)] |
| 77. | Indolfi C, Barillà F, Basso C, Ciccone MM, Curcio A, Mancone M, Mercuro G, Muscoli S, Nodari S, Pedrinelli R, Romeo F, Sinagra G, Filardi PP. [Position paper of the Italian Society of Cardiology (SIC) on COVID-19 vaccine priority in patients with cardiovascular diseases]. G Ital Cardiol (Rome). 2021;22:363-375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 78. | Kubica J, Ostrowska M, Stolarek W, Kasprzak M, Grzelakowska K, Kryś J, Kubica A, Adamski P, Podhajski P, Navarese EP, Anielska-Michalak E, Brycht O, Curzytek A, Dudek A, Gromadziński L, Grzelakowski P, Kamiński L, Kleinrok A, Kostkiewicz M, Koziński M, Król P, Kulawik T, Minczew G, Mindykowski M, Pawlak A, Prokopczuk J, Skonieczny G, Sobkowicz B, Sowiński S, Stankala S, Szymański P, Wester A, Wilczewski P, Bartuś S, Budaj A, Gąsior M, Gruchała M, Drożdż J, Jaguszewski M, Jankowski P, Legutko J, Lesiak M, Leszek P, Mitkowski P, Nessler J, Tomaszuk-Kazberuk A, Tycińska A, Zdrojewski T, Kaźmierczak J. Impact of COVID-19 pandemic on acute heart failure admissions and mortality: a multicentre study (COV-HF-SIRIO 6 study). ESC Heart Fail. 2022;9:721-728. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 79. | Severino P, D'Amato A, Saglietto A, D'Ascenzo F, Marini C, Schiavone M, Ghionzoli N, Pirrotta F, Troiano F, Cannillo M, Mennuni M, Rognoni A, Rametta F, Galluzzo A, Agnes G, Infusino F, Pucci M, Lavalle C, Cacciotti L, Mather PJ, Grosso Marra W, Ugo F, Forleo G, Viecca M, Morici N, Patti G, De Ferrari GM, Palazzuoli A, Mancone M, Fedele F. Reduction in heart failure hospitalization rate during coronavirus disease 19 pandemic outbreak. ESC Heart Fail. 2020;7:4182-8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 80. | Sokolski M, Trenson S, Sokolska JM, D'Amario D, Meyer P, Poku NK, Biering-Sørensen T, Højbjerg Lassen MC, Skaarup KG, Barge-Caballero E, Pouleur AC, Stolfo D, Sinagra G, Ablasser K, Muster V, Rainer PP, Wallner M, Chiodini A, Heiniger PS, Mikulicic F, Schwaiger J, Winnik S, Cakmak HA, Gaudenzi M, Mapelli M, Mattavelli I, Paul M, Cabac-Pogorevici I, Bouleti C, Lilliu M, Minoia C, Dauw J, Costa J, Celik A, Mewton N, Montenegro CEL, Matsue Y, Loncar G, Marchel M, Bechlioulis A, Michalis L, Dörr M, Prihadi E, Schoenrath F, Messroghli DR, Mullens W, Lund LH, Rosano GMC, Ponikowski P, Ruschitzka F, Flammer AJ. Heart failure in COVID-19: the multicentre, multinational PCHF-COVICAV registry. ESC Heart Fail. 2021;8:4955-4967. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 81. | Oseran AS, Afari ME, Barrett CD, Lewis GD, Thomas SS. Beyond the stethoscope: managing ambulatory heart failure during the COVID-19 pandemic. ESC Heart Fail. 2021;8:999-1006. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 82. | Afonso Nogueira M, Ferreira F, Raposo AF, Mónica L, Simões Dias S, Vasconcellos R, Proença G. Impact of telemedicine on the management of heart failure patients during coronavirus disease 2019 pandemic. ESC Heart Fail. 2021;8:1150-1155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 83. | Cammalleri V, Muscoli S, Benedetto D, Stifano G, Macrini M, Di Landro A, Di Luozzo M, Marchei M, Mariano EG, Cota L, Sergi D, Bezzeccheri A, Bonanni M, Baluci M, De Vico P, Romeo F. Who Has Seen Patients With ST-Segment-Elevation Myocardial Infarction? J Am Heart Assoc. 2020;9:e017126. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 84. | Wood DA, Sathananthan J. "Minimalist" transcatheter aortic valve implantation during the COVID-19 pandemic: previously optional but now a necessity. EuroIntervention. 2020;16:e451-e452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 85. | Dvir D. Severe valvular heart disease and COVID-19: results from the multicenter international valve disease registry. [cited 20 April 2022]. Available from: https://www.tctmd.com/news/valve-disease-plus-covid-19-often-lethal-combination-registry-shows. |
| 86. | Mohamed MO, Banerjee A, Clarke S, de Belder M, Patwala A, Goodwin AT, Kwok CS, Rashid M, Gale CP, Curzen N, Mamas MA. Impact of COVID-19 on cardiac procedure activity in England and associated 30-day mortality. Eur Heart J Qual Care Clin Outcomes. 2021;7:247-256. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 51] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 87. | Shah PB, Welt FGP, Mahmud E, Phillips A, Kleiman NS, Young MN, Sherwood M, Batchelor W, Wang DD, Davidson L, Wyman J, Kadavath S, Szerlip M, Hermiller J, Fullerton D, Anwaruddin S. Triage considerations for patients referred for structural heart disease intervention during the COVID-19 pandemic: An ACC/SCAI position statement. Catheter Cardiovasc Interv. 2020;96:659-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 88. | Wood DA, Sathananthan J, Gin K, Mansour S, Ly HQ, Quraishi AU, Lavoie A, Lutchmedial S, Nosair M, Bagai A, Bainey KR, Boone RH, Liu S, Krahn A, Virani S, Mehta SR, Natarajan MK, Velianou JL, Dehghani P, Wijeysundera HC, Asgar AW, Virani A, Welsh RC, Webb JG, Cohen EA. Precautions and Procedures for Coronary and Structural Cardiac Interventions During the COVID-19 Pandemic: Guidance from Canadian Association of Interventional Cardiology. Can J Cardiol. 2020;36:780-783. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 89. | Costa G, Barbanti M, Picci A, Todaro D, La Spina K, Di Simone E, D'Arrigo P, Criscione E, Valvo R, Reddavid C, Deste W, Sgroi C, Tamburino C, Giuffrida A, Garretto V, Privitera G, Cannizzaro MT, Inserra C, Veroux P. Predictors and safety of next-day discharge in patients undergoing transfemoral transcatheter aortic valve implantation. EuroIntervention. 2020;16:e494-e501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 90. | Sathananthan J, Webb JG, Polderman J, Achtem L, Hensey M, Murdoch D, Moss R, Shook A, Bruce S, Blanke P, Bancroft C, Andrews H, Leipsic J, Wood D, Lauck S. Safety of Accelerated Recovery on a Cardiology Ward and Early Discharge Following Minimalist TAVR in the Catheterization Laboratory: The Vancouver Accelerated Recovery Clinical Pathway. Structural Heart. 2019;3:229-235. [RCA] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 91. | Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS, Falk V, González-Juanatey JR, Harjola VP, Jankowska EA, Jessup M, Linde C, Nihoyannopoulos P, Parissis JT, Pieske B, Riley JP, Rosano GMC, Ruilope LM, Ruschitzka F, Rutten FH, van der Meer P; ESC Scientific Document Group. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC)Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2016;37:2129-2200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10348] [Cited by in RCA: 9358] [Article Influence: 1039.8] [Reference Citation Analysis (3)] |
| 92. | Roffi M, Capodanno D, Windecker S, Baumbach A, Dudek D. Impact of the COVID-19 pandemic on interventional cardiology practice: results of the EAPCI survey. EuroIntervention. 2020;16:247-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 93. | Marmagkiolis K, Kilic ID, Ates I, Kose G, Iliescu C, Cilingiroglu M. Feasibility of Same-Day Discharge Approach After Transcatheter Mitral Valve Repair Procedures. J Invasive Cardiol. 2021;33:E123-E126. [PubMed] |
| 94. | Chowdhury M, Buttar R, Rai D, Tahir MW, Tan BE, Thakkar S, Ali H, Patel HP, Bhatt DL, Depta JP. Same-day discharge after transcatheter mitral valve repair using MitraClip in a tertiary community hospital: a case series. Eur Heart J Case Rep. 2021;5:ytab397. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 95. | Romeo F, Cammalleri V, Ruvolo G, Quadri A, De Vico P, Muscoli S, Marchei M, Meloni S, Conti F, Ussia GP. Trans-catheter mitral valve implantation for mitral regurgitation: clinical case description and literature review. J Cardiovasc Med (Hagerstown). 2016;17:85-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 96. | Skulstad H, Cosyns B, Popescu BA, Galderisi M, Salvo GD, Donal E, Petersen S, Gimelli A, Haugaa KH, Muraru D, Almeida AG, Schulz-Menger J, Dweck MR, Pontone G, Sade LE, Gerber B, Maurovich-Horvat P, Bharucha T, Cameli M, Magne J, Westwood M, Maurer G, Edvardsen T. COVID-19 pandemic and cardiac imaging: EACVI recommendations on precautions, indications, prioritization, and protection for patients and healthcare personnel. Eur Heart J Cardiovasc Imaging. 2020;21:592-598. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 192] [Cited by in RCA: 209] [Article Influence: 41.8] [Reference Citation Analysis (0)] |
| 97. | Fazzari F, Mantovani R, Donghi V, Curzi M, Bragato RM. From EACVI recommendations to the real-world experience: safety of performing echocardiography in the pandemic era. Eur Heart J Cardiovasc Imaging. 2021;22:e82-e83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 98. | Williams MC, Shaw L, Hirschfeld CB, Maurovich-Horvat P, Nørgaard BL, Pontone G, Jimenez-Heffernan A, Sinitsyn V, Sergienko V, Ansheles A, Bax JJ, Buechel R, Milan E, Slart RHJA, Nicol E, Bucciarelli-Ducci C, Pynda Y, Better N, Cerci R, Dorbala S, Raggi P, Villines TC, Vitola J, Malkovskiy E, Goebel B, Cohen Y, Randazzo M, Pascual TNB, Dondi M, Paez D, Einstein AJ; INCAPS COVID Investigators Group. Impact of COVID-19 on the imaging diagnosis of cardiac disease in Europe. Open Heart. 2021;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 99. | Dondi M, Milan E, Pontone G, Hirschfeld CB, Williams M, Shaw LJ, Pynda Y, Raggi P, Cerci R, Vitola J, Better N, Villines TC, Dorbala S, Pascual TNB, Giubbini R, Einstein AJ, Paez D; INCAPS COVID Investigators Group. Reduction of cardiac imaging tests during the COVID-19 pandemic: The case of Italy. Findings from the IAEA Non-invasive Cardiology Protocol Survey on COVID-19 (INCAPS COVID). Int J Cardiol. 2021;341:100-106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 100. | Joshi SS, Stankovic I, Demirkiran A, Haugaa K, Maurovich-Horvat P, Popescu BA, Cosyns B, Edvardsen T, Petersen SE, Carvalho RF, Cameli M, Dweck MR. EACVI survey on burnout amongst cardiac imaging specialists during the 2019 coronavirus disease pandemic. Eur Heart J Cardiovasc Imaging. 2022;23:441-446. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 101. | Picano E, Scali MC, Ciampi Q, Lichtenstein D. Lung Ultrasound for the Cardiologist. JACC Cardiovasc Imaging. 2018;11:1692-1705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 134] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 102. | Gargani L, Soliman-Aboumarie H, Volpicelli G, Corradi F, Pastore MC, Cameli M. Why, when, and how to use lung ultrasound during the COVID-19 pandemic: enthusiasm and caution. Eur Heart J Cardiovasc Imaging. 2020;21:941-948. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 94] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 103. | Barrios V, Cosín-Sales J, Bravo M, Escobar C, Gámez JM, Huelmos A, Ortiz Cortés C, Egocheaga I, García-Pinilla JM, Jiménez-Candil J, López-de-Sá E, Torres Llergo J, Obaya JC, Pallares-Carratalá V, Sanmartín M, Vidal-Pérez R, Cequier Á. Telemedicine consultation for the clinical cardiologists in the era of COVID-19: present and future. Consensus document of the Spanish Society of Cardiology. Rev Esp Cardiol (Engl Ed). 2020;73:910-918. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 104. | Gorodeski EZ, Goyal P, Cox ZL, Thibodeau JT, Reay RE, Rasmusson K, Rogers JG, Starling RC. Virtual Visits for Care of Patients with Heart Failure in the Era of COVID-19: A Statement from the Heart Failure Society of America. J Card Fail. 2020;26:448-456. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 139] [Article Influence: 27.8] [Reference Citation Analysis (0)] |
| 105. | Kerr B, Pharithi RB, Barrett M, Halley C, Gallagher J, Ledwidge M, McDonald K. Changing to remote management of a community heart failure population during COVID-19 - Clinician and patient perspectives'. Int J Cardiol Heart Vasc. 2020;31:100665. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 106. | Sayer G, Horn EM, Farr MA, Axsom K, Kleet A, Gjerde C, Latif F, Sobol I, Kelley N, Lancet E, Halik C, Takeda K, Naka Y, Yuzefpolskaya M, Kumaraiah D, Colombo PC, Maurer MS, Uriel N. Transition of a Large Tertiary Heart Failure Program in Response to the COVID-19 Pandemic: Changes That Will Endure. Circ Heart Fail. 2020;13:e007516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 107. | Nolan RP, Ross HJ, Farkouh ME, Huszti E, Chan S, Toma M, D'Antono B, White M, Thomas S, Barr SI, Perreault S, McDonald M, Zieroth S, Isaac D, Wielgosz A, Mielniczuk LM. Automated E-Counseling for Chronic Heart Failure: CHF-CePPORT Trial. Circ Heart Fail. 2021;14:e007073. [PubMed] |
| 108. | Giacalone A, Marin L, Febbi M, Franchi T, Tovani-Palone MR. eHealth, telehealth, and telemedicine in the management of the COVID-19 pandemic and beyond: Lessons learned and future perspectives. World J Clin Cases. 2022;10:2363-2368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 13] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (2)] |