Published online May 16, 2013. doi: 10.12998/wjcc.v1.i2.79
Revised: April 11, 2013
Accepted: April 17, 2013
Published online: May 16, 2013
Processing time: 119 Days and 3.9 Hours
In 2010 the World Health Organisation estimated the number of infections with Entamoeba histolytica at about 50 million cases including 100000 fatal courses. In most cases this infection is a subclinical event with few or none symptoms noticeable for the patient. Courses of this disease and incidence of this parasite in industrialised nations are not yet fully investigated. Our case reports about a 68-year-old male patient from Turkey who lives for more than 30 years in Germany and had not been abroad during the past 2 years. Resistant asymptomatic amoebic dormant bodies caused an emergency-laparoscopy and revealed the seldom complication of a colon perforation. In the age of globalisation all providers in the health care systems are urged to acquaint themselves also with non-typical syndromes for the countries they work in order to reduce preventable morbidity respectively mortality rates.
Core tip: This case shows how important it is that medicine providers expected rare diseases from other regions from the globalised world. The clinical signs of this patient have been wrongly interpreted. Itself the operation was not targeted. The histopathological examination of resected intestine had a surprising result, but not the source of the clinical signs. Only the use of the polymerase change reaction (PCR) identified the causal link between the clinical signs and the trigger. So the PCR should be the central feature in the diagnostic of unclear or undefined clinical signs.
- Citation: Redaèlli M, Mahmoohdzad J, Lang R, Schencking M. Globalised world, globalised diseases: A case report on an amoebiasis-associated colon perforation. World J Clin Cases 2013; 1(2): 79-81
- URL: https://www.wjgnet.com/2307-8960/full/v1/i2/79.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v1.i2.79
Infection with Entamoeba histolytica (E. histolytica) usually occurs by ingestion of contaminated water, unwashed fruits or vegetables. Faecal-oral transmission has also been described. An amoebiasis with bloody, mucousy stools with a frequency up to 40-50/d can result with intestinal pain, spasms and high fever[1].
As a resistant dormant body the amoebas can develop cysts and remain asymptomatic in the colon for years. For yet unknown reasons it can amount to a mutation of the amoebas’ DNA leading to a change of the enzymatic pattern[2,3]. These enzymes may allow penetration of the amoeba into the intestinal mucous membrane sometimes with fatal outcome. A possible intestinal finding is a perforation of the colon. Furthermore dissemination of amoebas to other organ-systems is possible due to the entrance of amoebas into the bloodstream. Most frequently liver and heart as well as the central nervous system and the urinary system are affected[4,5].
Perforation of the colon as a result of an amoebic colitis is a seldom complication but typically carries a high morbidity and mortality[6-8]. On the European continent the incidence of this illness is extremely rare, yet the increasing globalisation of trade and services as well as migration result also in a globalisation of infectious diseases and therefore an increasing number of amoebic colitis in Europe.
The worldwide relevance of infections with E. histolytica can be evaluated by the numbers estimated by the World Health Organisation of yearly 50 million with about 100000 fatal courses.
A 65-year-old Turkish man presented to the emergency department with spasmodic abdominal pain that had increased over several days and was therefore hospitalized. At the time of admission his body mass index was 33.6 (height: 185 cm, weight: 115 kg). He had been living in Germany for 30 years and had not travelled to any other country during the past 2 years. The patient did not suffer from diarrhoea or fever and had no pre-existing conditions. There were no visits to the general practitioner during the past 6 mo.
Physical examination revealed the patient’s general condition to be compromised with diffuse pain located throughout the abdomen and described as pressure. There were also peritoneal signs and muscular rigidity. The point of maximum tenderness was located in the right lower quadrant and midline. Peristalsis was reduced significantly; the rectal examination was without pathological findings.
The complete blood count was significant for leucocytosis (11000/L), the C-reactive protein was within the normal range with 0.7 mg/dL, and all other routine laboratory parameters were also without pathological findings.
Abdominal sonography revealed a narrow perihepatical margin of fluid. Abdominal X-rays in two planes did not show signs of free air or any typical findings of an ileus with the right colon stool-filled. Because of the clinical situation it was decided to utilize invasive diagnostics and invasive therapy if necessary.
An emergency-laparoscopy was carried out on the day of the admission because of suspected perforated appendicitis. The intraoperative findings included a long-segment, covered perforation of the coecum and the right hemicolon accompanied by a 2 quadrant-peritonitis. Because of the seriousness of the intraoperative findings a switch to an open laparotomy became necessary.
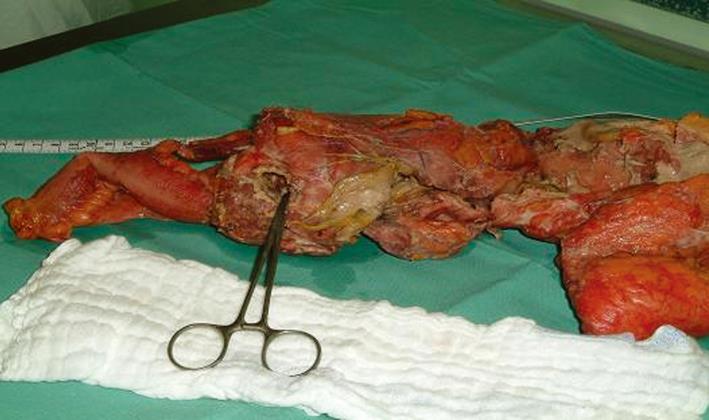
Following a median laparotomy the full extent of the patient’s findings became evident: a massive necrotising inflammation of the hemicolon including perforation, plus a “wooden phlegmon” of the mesocolon reaching up to the retroperitoneum. Because of suspected sarcoma of the mesocolon, a right hemicolectomy with a R0-resection was performed accomplished after extensive mobilisation of the right colon and the colon transversum (Figure 1). This procedure was followed by an ileotransversotomy, an end-to-end anastomosis with Vicryl-single button suture seromuscular, extra mucous and in a single row.
Initial histopathological examination of formalin fixed colon dissection samples showed the characteristics of a low malignant Non-Hodgkin-lymphoma. Following further pathological examination could ascertain that the perforation was caused by a severe amoebic colitis followed by a granulomatous inflammation of the mesocolon. A diagnosis of malignant lymphoma was ruled out.
The amoebic colitis was treated with metronidazole followed by paromomycin resulting in successful eradication of the amoebae. In addition methicillin resistant Staphylococcus aureus was cultured from intraoperative specimens. The patient was placed in contact isolation after the surgical intervention and provided with rehabilitation services. Repeated stool samples during the patient’s stay in hospital were all negative for amoebic infection.
During the postoperative course the patient developed an infection of the laparotomy wound accompanied by symptoms of an ileus and had to undergo another laparotomy on the 5th postoperative day. This surgery revealed an abdominal wall abscess without intra-abdominal findings. The subsequent postoperative course was uncomplicated. After 63 d of stay in hospital the patient could be discharged fit and healthy.
The mechanism of infection in this case is not apparent. With amoebic infections being very uncommon in Germany one can speculate that the patient’s occupation as a storekeeper for an airport company could have led to contact with contaminated materials or surfaces. In addition there is the possibility of this case representing a relapse. The patient could have been asymptomatically infected before his emigration to Germany or during visits to his home country (prevalence in Turkey: 15%)[9].
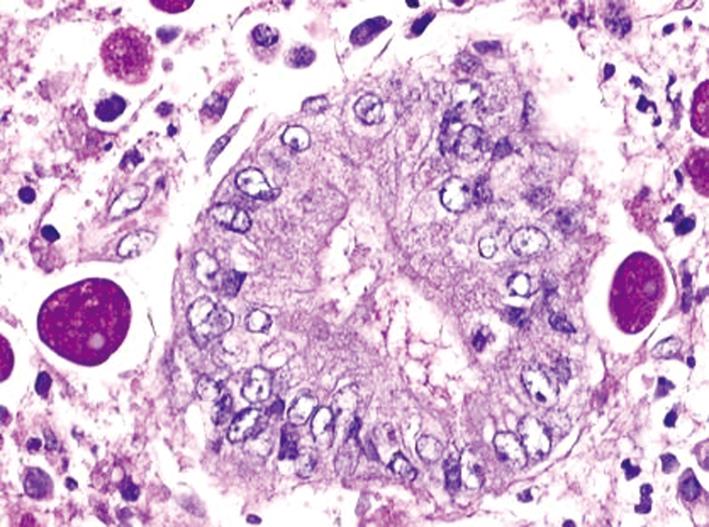
During laparotomy the intraoperative findings mimicked a sarcoma of the mesocolon. The diagnosis could not be assured until the final histopathological results were available: perforation because of a preexisting amoebic colitis. A granulomatous infection appearing as a “wood phlegmone” of the mesocolon has been described as a rare manifestation of infection with E. histolytica, which is the only pathogenic amoeba for humans. Figure 2 shows the intracellular phagocytosed erythrocytes, which are pathognomonic for this kind of amoeba.
Antibiotic therapy with metronidazole was recommended as first-line therapy in Germany at the time of hospital admission[10]. A Cochrane Review from 2009 suggests that tinidazole may be more effective[11]. If amoebas are found in the stool of asymptomatic individuals they are considered chronic carriers. Only patients, who are chronic carriers and near contact persons are considered notifiable cases in Germany.
Only a rare amount of case reports describing the E. histolytica induced symptoms of colitis or intestinal perforations due to wide necrosis of the intestine are existing in current scientific literature; in comparison to these cases our patient didn’t suffer of fever or diarrhoea and underwent a successful inpatient treatment[1,12].
An ulcerous colitis can originate from an amoebic infection. Therefore stool samples for this pathogen should always be sent for microscopy and, in addition, if available for molecular diagnostic tests such as testing by polymerase chain reaction. Diagnostic imaging can not provide a sufficient contribution to the causality amoebic infection and colitis.
In the age of globalisation all providers in the health care systems are urged to acquaint themselves also with non-typical syndromes for the countries they work in order to reduce preventable morbidity respectively mortality rates.
P- Reviewer Das UN S- Editor Wen LL L- Editor A E- Editor Zheng XM
| 1. | Yamada H, Matsuda K, Akahane T, Shimada R, Horiuchi A, Shibuya H, Aoyagi Y, Nakamura K, Hayama T, Iinuma H. A case of fulminant amebic colitis with multiple large intestinal perforations. Int Surg. 2010;95:356-359. [PubMed] |
| 2. | Lourenssen S, Houpt ER, Chadee K, Blennerhassett MG. Entamoeba histolytica infection and secreted proteins proteolytically damage enteric neurons. Infect Immun. 2010;78:5332-5340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 3. | Becker SM, Cho KN, Guo X, Fendig K, Oosman MN, Whitehead R, Cohn SM, Houpt ER. Epithelial cell apoptosis facilitates Entamoeba histolytica infection in the gut. Am J Pathol. 2010;176:1316-1322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 34] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 4. | Koo JS, Choi WS, Park DW. Fulminant amebic colitis mimicking pseudomembranous colitis. Gastrointest Endosc. 2010;71:400-401; discussion 401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 5. | Khan MA, Verma GR, Lokesh HM. Fulminant amebic colitis with lower GI bleed with liver abscess. Int J Colorectal Dis. 2010;25:535-537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 6. | Luvuno FM, Mtshali Z, Baker LW. Vascular occlusion in the pathogenesis of complicated amoebic colitis: evidence for an hypothesis. Br J Surg. 1985;72:123-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 7. | Ozdogan M, Baykal A, Aran O. Amebic perforation of the colon: rare and frequently fatal complication. World J Surg. 2004;28:926-929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 8. | Farhana F, Jamaiah I, Rohela M, Abdul-Aziz NM, Nissapatorn V. A ten year (1999-2008) retrospective study of amoebiasis in University Malaya Medical Centre (UMMC), Kuala Lumpur, Malaysia. Trop Biomed. 2009;26:262-266. [PubMed] |
| 9. | Malatyalı E, Ozçelik S, Celiksöz A. The investigation of Entamoeba histolytica prevalence in some villages of Sivas by ELISA method. Turkiye Parazitol Derg. 2011;35:6-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 10. | Burchard GD, Tannich E. Epidemiology, diagnosis and therapy of amoebiasis. Dtsch Arztebl. 2004;101:A 3036-A 3040 Available from: http://www.aerzteblatt.de/archiv/44171/Epidemiologie-Diagnostik-und-Therapie-der-Amoebiasis. |
| 11. | Gonzales ML, Dans LF, Martinez EG. Antiamoebic drugs for treating amoebic colitis. Cochrane Database Syst Rev. 2009;CD006085. [PubMed] |
| 12. | Ozer M, Ergul E, Donmez C, Sisman IC, Ulger BV, Kusdemir A. Amebic perforation of small bowel: an unexpected localization of a fatal complication. Bratisl Lek Listy. 2009;110:59-60. [PubMed] |