Published online Aug 24, 2017. doi: 10.5500/wjt.v7.i4.243
Peer-review started: September 17, 2016
First decision: October 21, 2016
Revised: May 29, 2017
Accepted: June 19, 2017
Article in press: June 20, 2017
Published online: August 24, 2017
Processing time: 340 Days and 10.8 Hours
The familiar amyloid polyneuropathy (FAP) is a rare autosomal-dominant systemic amyloidosis. Amyloid deposition occurs more frequently and extensively in the vitq. The increase in intraocular pressure (IOP) is a result of deposition of transthyretin (TTR) in trabecular meshwork. Rarely, the amyloid deposition in anterior segment can be more exuberant than in posterior segment. A 42 years old man, with FAP (Val30Met mutation), liver transplantation in 1997. He was asymptomatic, without any significant ocular abnormality until 2011. In 2011 he had an episode of pain in right eye (RE). Scalloped pupils, pupillary amyloid deposits and subtle vitreous opacities were detected. The IOP was 40 mmHg in RE and 28 mmHg in left eye (LE) with open angle. Optical coherence tomography detected a temporal superior retinal nerve fiber layer defect in LE and perimetry was normal. Topical timolol was initiated, and brimonidine was subsequently added to improve IOP control, which was achieved with topical medication until last evaluation. No progression occurred since 2011. Actually, with longer life expectancies, there is an increased risk of ocular involvement in FAP, even after liver transplantation. Although rare, a more exuberant amyloid deposition in anterior segment vs posterior segment can occur, and supports an important role of amyloid production in ciliary pigment epithelium in these patients. Medical control of IOP and a stable course are unusual in this secondary glaucoma. Ophthalmologists have an important task in the follow-up of patients and early diagnosis of risk factors for secondary glaucoma, such as scalloped pupils with amyloid deposits.
Core tip: Ocular manifestations of familial amyloidotic polyneuropathy (FAP) can appear after liver transplantation due to de novo ocular production of amyloid. Rarely, amyloid deposition in vitreous is relatively less exuberant than in anterior segment. Our case illustrates this asymmetry of amyloid deposition and emphasizes the association between scalloped pupils and glaucoma, a major ocular complication of FAP. Our case had a stable course, with excellent visual function and the intraocular pressure was controlled by medical therapy, which are unusual in this type of glaucoma. This case-report also highlights the importance of the long-term ophthalmological follow-up in FAP patients.
- Citation: Gama IF, Almeida LD. De novo intraocular amyloid deposition after hepatic transplantation in familial amyloidotic polyneuropathy. World J Transplant 2017; 7(4): 243-249
- URL: https://www.wjgnet.com/2220-3230/full/v7/i4/243.htm
- DOI: https://dx.doi.org/10.5500/wjt.v7.i4.243
Transthyretin (TTR)-related familial amyloid polyneuropathy (FAP) is a group of autosomal-dominant diseases of variable penetrance caused by the deposition of polymerized mutated TTR in the peripheral nerves, gastrointestinal tract, heart, ocular tissues, and other organs. These protein aggregates have affinity for Congo red stain and apple-green birefringence when viewed under polarized light[1-3]. They are caused by mutations of TTR gene (18q11.2-12.1)[2]. Peripheral neuropathy is progressive and frequently the first manifestation of the disease[1-3]. Type 1 FAP, the Portuguese type (FAP1) was described for the first time in 1952 by Corino de Andrade[4]. FAP1 is the most frequent type of FAP and caused by a mutational substitution of the valine for methionine in position 30 of TTR gene (Val30Met)[2].
There are many ophthalmological manifestations of FAP caused by deposition of amyloid in various intra-ocular tissues: Vitreous, iris, pupillary border, anterior capsule and trabecular meshwork. The pupillary margin may have a scalloped/indented configuration (scalloped pupils) and pupils may be slow or nonreactive to both light and near stimulation, caused by disturbance of autonomic innervation[1-3,5-8]. Fleck deposits resembling pseudoexfoliation (PEX) may be found on the anterior lens capsule and pupillary margin[1-3,7]. Pseudopodia lentis is a hallmark of vitreous amyloidosis, where multiple small dots or footplates are formed on the posterior lens surface[2]. Trabecular meshwork deposition of amyloid causes obstruction of aqueous humor outflow and subsequent elevation of intra-ocular pressure (IOP)[9]. Secondary glaucoma can develop rapidly with high IOP, which if left untreated it can lead to severe damage[3,9]. Other manifestations include dry eye by decreased tear production, conjunctival microaneurysms and reduced corneal sensitivity with subsequent neurotrophic corneal ulcers[2].
TTR is a normal constituent of blood plasma, acts as a thyroxine transport protein and is important in vitamin A transport[2,3]. TTR is synthesized mainly in liver (90%), but there is also intra-ocular production[1,3,6,7,10-12]. Retinal pigment epithelium (RPE) is a source of TTR synthesis in rat eyes[10]. Recently, it was demonstrated that TTR production also occurs in the ciliary pigment epithelium (CPE)[12].
Liver transplantation (LT) improved the quality and survival of FAP patients, but does not prevent ocular manifestations of FAP, because of persistent intra-ocular production of amyloidotic TTR (ATTR). A case of vitreous amyloidosis appearing 2 years after LT was described and mutant protein ATTR was detected in aqueous humor of a Japanese patient after LT[13,14].
Secondary glaucoma is a major complication of FAP, which can be the first ocular manifestation and cause irreversible visual loss. Thus, early diagnosis is fundamental to avoid rapid progression of glaucoma[9].
The authors want to emphasize the importance of the recognition of ophthalmological signs that are associated with increased risk of ocular hypertension and glaucoma in FAP1 patients after LT as well as to report an unusual asymmetric pattern of intraocular amyloid deposition, with a case report and bibliographic revision.
A 42-year-old man had a diagnosis of FAP since 1995, with a positive genetic test for ATTR Val30Met mutation, and was subjected to LT in 1997. The peripheral neuropathy improved after LT. His brother and mother had type 1 FAP. The patient did not have any other previous ophthalmological diagnosis besides myopia. No FAP-related ophthalmological abnormalities were detected on routine ophthalmology evaluations for 14 years after LT.
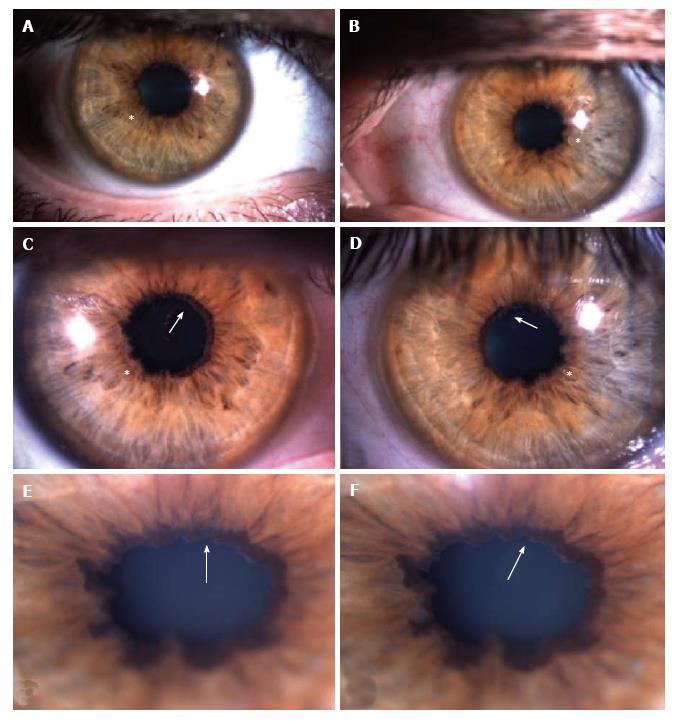
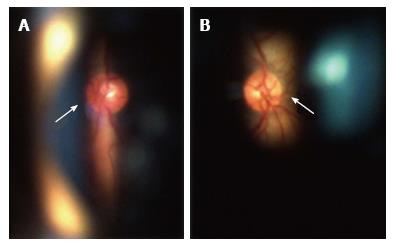
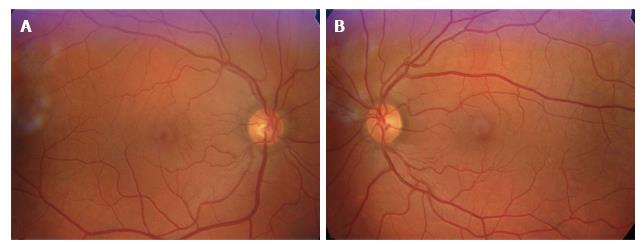
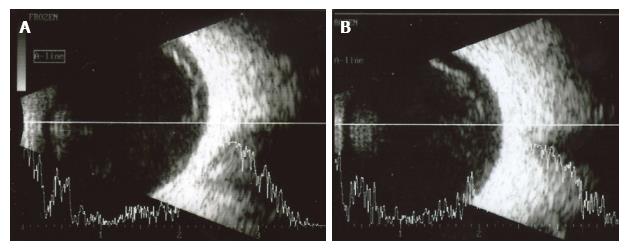
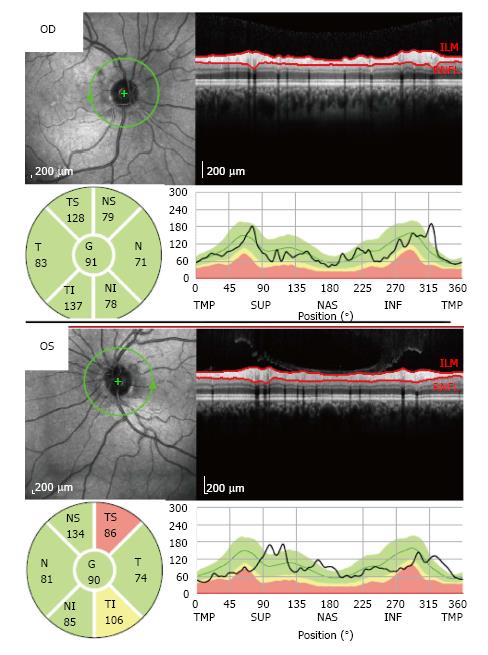
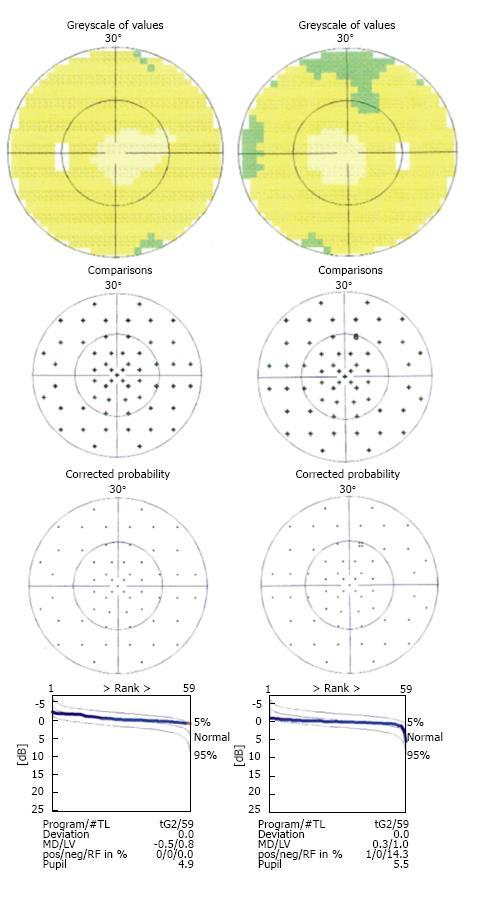
In November 2011, he had an episode of ocular pain in right eye (RE) and attended the emergency room. Best-corrected visual acuity was 20/20 in RE and left eye (LE). Pupils were isochoric with slow pupillary responses to light and near stimulation. Ocular movements were normal. Biomicroscopy showed bilateral whitish fleck flocculent deposits of amyloid in the pupillary borders, scalloped pupils and few deposits in anterior vitreous (Figure 1). The detection of abnormalities led to the measurement of intraocular pressure (IOP) by Goldmann applanation tonometry (GAT), being 40 mmHg in RE and 28 mmHg in LE. Gonioscopy showed open angles - Shaffer grade of 4. Fundoscopy and retinography showed few vitreous opacities and clearly visible normal posterior poles, with normal appearing optic discs (Figures 2 and 3). Central corneal thickness was 559 μm in RE and 550 μm in LE. Ophthalmic ultrasound (US) showed few vitreous opacities bilaterally (Figure 4). Optical coherence tomography (OCT) only showed a superior-temporal peripapillary retinal fiber layer retinal nerve fiber layer (RNFL) defect in LE (Figure 5). Automated perimetry was unremarkable in both eyes (Figure 6). Topical monotherapy with timolol 0.5% was initiated at that time, and the IOP lowered to 26 mmHg in RE and to 21 mmHg in LE. To optimize IOP control, brimonidine was associated with timolol further lowering the IOP to 14 mmHg in both eyes. The patient was followed up closely in the glaucoma clinic until present, with controlled IOP. Last CSP and OCT exams excluded glaucoma progression.
Although ATTR levels after LT decline to < 1% of pre-transplant levels, FAP patients are still at risk of ophthalmological complications of the disease because of continued intra-ocular production in RPE and CPE and continued amyloid deposition in various ocular tissues, such as vitreous, pupil, anterior lens capsule and trabecular meshwork[1,3,6,7,10,12-18].
LT improved survival and consequently there is increased risk of ocular complications of FAP in transplantation era, because ocular manifestations are dependent of the duration of systemic disease. Glaucoma is a major ocular complication of FAP and a major cause of visual loss in these patients[1,3,19,20].
The study of Kimura et al[1] reported glaucoma in 24% of all FAP patients and in 17% of patients with Val30Met mutation, but the prevalence of glaucoma differs in various studies, from 5.4% to 27%[1,20,21]. Glaucoma is secondary to amyloid deposition in trabecular meshwork and if not recognized or treated adequately can have a rapid progression and devastating visual consequences in these patients, who have already a great morbidity from the systemic disease[1,3,19,20]. The pathophysiology of glaucoma in FAP1 after LT is related to the deposition of amyloid fibrillar aggregates in intertrabecular spaces of corneoscleral and uveoscleral meshworks and degeneration of endothelium cells of trabecular meshwork[6]. The trabecular outflow resistance increases, which raises IOP. Perivascular deposition of amyloid in conjunctival and scleral tissues can increase episcleral pressure and, consequently, the outflow resistance, but this mechanism is mainly dependent on systemic production of ATTR, playing an important role only before LT[6].
Most of the ophthalmological studies of FAP are focused in vitreous opacities (VO) and there are only few studies about secondary glaucoma. In the study of Kimura et al[1], VO were found in 35% of patients and amyloid deposition in pupil and anterior lens capsule in 31% of patients. Scalloped pupils are caused by autonomic abnormalities, which are associated with a higher degree of amyloid deposition in anterior segment and can also predict glaucoma[3]. They occur in 8% of FAP patients and glaucoma in 20% of patients. Glaucoma was diagnosed in all cases (100%) with scalloped pupils and in 57% of cases with amyloid deposition in anterior segment (pupil and anterior lens capsule). Only 49% of cases with VO had glaucoma[1]. Vitreous opacities are a classic ocular manifestation of FAP, but accordingly to the studies of Kimura et al[1] and Sandgren et al[3], the association between VO and glaucoma is weaker than between glaucoma and pupillary abnormalities (scalloped pupils, ATTR deposition in pupillary margin)[1,3]. This finding is supported by our clinical case.
Sandgren et al[3,7] suggested that amyloid deposits in pupil and anterior lens capsule are more precocious than in the vitreous, which can explain the existence of rare cases, such as our case, which have much more ATTR deposition in anterior segment than in vitreous. These rare cases, such as our clinical case, corroborate the hypothesis raised by the study from Kawaji et al[12] postulating that the ATTR accumulated in anterior segment may have origin in CPE. This hypothesis can explain this asymmetry between ATTR deposition in anterior and posterior segments, as occurred our clinical case[12].
Amyloid is transported in the aqueous. Thus, pupillary amyloid deposits are an indirect sign of exuberant amyloid deposition in anterior segment, including the trabecular meshwork. This results in an increased resistance to aqueous humor outflow[19]. Kimura et al[1] have found that pupillary amyloid deposits have preceded the diagnosis of glaucoma by an average period of 2.55 ± 1.43 years (range 0.2-4.0 years). In the presented clinical case, the recognition of the pupillary abnormalities raised the clinical suspicion of glaucoma that was confirmed by appropriate investigation. Preperimetric glaucoma was confirmed by the finding of a localized defect of nerve fiber layer without perimetric functional repercussion.
Most cases of glaucoma secondary to FAP are usually refractory to medical treatment and have a fast progression and bad prognosis. This type of glaucoma usually requires surgical treatment[8]. Tube shunts, specially the Ahmed valve have been extensively used for surgical treatment of FAP1-related glaucoma in Portugal[8]. Recently minimal invasive options for glaucoma treatment are available for primary open-angle glaucoma and some types of secondary glaucoma, having the advantage of being less traumatic to the eye. However, prospective studies of efficacy in FAP-related glaucoma are lacking. Our clinical case had an unusual clinical course, with a good IOP control with medical treatment and stable visual fields and RNFL thicknesses.
Pars plana vitrectomy can be performed if vitreous opacities impair visual acuity, but this was not the case of our patient. Also, glaucoma can occur or be aggravated after pars plana vitrectomy in FAP patients, which is an important aspect to consider when managing ocular manifestations of FAP patients also affected by secondary glaucoma.
In an era that FAP patients have a greater life expectance with liver transplant, there is an increased probability of serious ocular disease caused by FAP, such as glaucoma that requires a regular ophthalmologic follow-up.
A 41-year-old man with type 1 familiar amyloid polyneuropathy (FAP) subjected to liver transplantation in 1997, presented with ocular pain.
Ophthalmological examination showed ocular hypertension, scalloped pupils associated to exuberant amyloid pupillary deposits, which contrasted with the mild vitreous opacities on ultrasound.
FAP-related secondary open-angle glaucoma, FAP-related secondary ocular hypertension, pseudoexfoliation glaucoma, ocular hypertension associated to pseudoexfoliation syndrome.
Ocular ultrasound showed mild vitreous opacities due to amyloid deposition. Retinography showed normal posterior poles. Optical coherence tomography only showed a peripapillary temporal-superior retinal nerve fiber layer defect in OS. Perimetry did not show significant visual field abnormalities.
Treatment with topical timolol and brimonidine achieved intraocular pressure (IOP) control. This treatment was continued, permitting disease stabilization with IOP control. This is a rare clinical course of this disease.
De novo intraocular amyloid synthesis and deposition occurs after liver transplantation, having the potential to cause serious ocular complications. Most reported cases of FAP-related secondary glaucoma with scalloped pupils have exuberant vitreous amyloid deposition. The asymmetry between exuberant amyloid deposition in anterior segment vs mild vitreous deposition that was reported in this clinical case is rare, and suggests a role of ciliary pigment epithelium in intraocular amyloid synthesis. This clinical case had a rare clinical course.
FAP-related glaucoma after liver transplantation is a secondary type of glaucoma, caused by an increase in trabecular outflow resistance associated to trabecular amyloid deposition, with amyloid fibrillar aggregates in intertrabecular spaces of corneoscleral and uveoscleral meshworks and degeneration of endothelium cells of trabecular meshwork.
Rarely, amyloid deposition in anterior segment can be much more exuberant than vitreous deposition. This asymmetry supports a significant role of the ciliary pigmented epithelium in the intraocular amyloid synthesis in these cases. Pupillary amyloid deposition and scalloped pupils have a stronger correlation to glaucoma than other ocular manifestations. Rarely, FAP-related glaucoma can be stable and well controlled by medical treatment alone.
This case is very rare and an interesting case.
Manuscript source: Invited manuscript
Specialty type: Transplantation
Country of origin: Portugal
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Hong YJ, Nowak MS, Salvadori M S- Editor: Ji FF L- Editor: A E- Editor: Lu YJ
| 1. | Kimura A, Ando E, Fukushima M, Koga T, Hirata A, Arimura K, Ando Y, Negi A, Tanihara H. Secondary glaucoma in patients with familial amyloidotic polyneuropathy. Arch Ophthalmol. 2003;121:351-356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 47] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 2. | Rutar TRM. Diseases of the Vitreous. Albert & Jakobiec’s Principles and Practice of Ophthalmology. Philadelphia: W. B. Saunders Company 2008; 2391-2398. |
| 3. | Sandgren O, Kjellgren D, Suhr OB. Ocular manifestations in liver transplant recipients with familial amyloid polyneuropathy. Acta Ophthalmol. 2008;86:520-524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 4. | Andrade C. A peculiar form of peripheral neuropathy; familiar atypical generalized amyloidosis with special involvement of the peripheral nerves. Brain. 1952;75:408-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 794] [Cited by in RCA: 731] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 5. | Lessell S, Wolf PA, Benson MD, Cohen AS. Scalloped pupils in familial amyloidosis. N Engl J Med. 1975;293:914-915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 6. | Rosa AM, Quadrado MJ, Ferrão J, Manifesta ções Oculares de Polineuropatia Amiloidótica Familiar Tipo I em Doentes Submetidos a Transplante Hepático. Oftalmol (Port Ophthalmol Soc Journal). 2009;33:177-183. |
| 7. | Sandgren O. Ocular amyloidosis, with special reference to the hereditary forms with vitreous involvement. Surv Ophthalmol. 1995;40:173-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 87] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 8. | Sampaio I, Queirós J, Borges P, Reimão P, Beirão M. MMAC. Glaucoma em Doentes Portugueses com Polineuropatia Amiloidótica Familiar. Oftalmol (Port Ophthalmol Soc Journal). 2011;35:311-318. |
| 9. | Doft BH, Machemer R, Skinner M, Buettner H, Clarkson J, Crock J, McLeod D, Michels R, Scott J, Wilson D. Pars plana vitrectomy for vitreous amyloidosis. Ophthalmology. 1987;94:607-611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 37] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 10. | Cavallaro T, Martone RL, Dwork AJ, Schon EA, Herbert J. The retinal pigment epithelium is the unique site of transthyretin synthesis in the rat eye. Invest Ophthalmol Vis Sci. 1990;31:497-501. [PubMed] |
| 11. | Futa R, Inada K, Nakashima H, Baba H, Kojima Y, Okamura R, Araki S. Familial amyloidotic polyneuropathy: ocular manifestations with clinicopathological observation. Jpn J Ophthalmol. 1984;28:289-298. [PubMed] |
| 12. | Kawaji T, Ando Y, Nakamura M, Yamamoto K, Ando E, Takano A, Inomata Y, Hirata A, Tanihara H. Transthyretin synthesis in rabbit ciliary pigment epithelium. Exp Eye Res. 2005;81:306-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 50] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 13. | Munar-Qués M, Salva-Ladaria L, Mulet-Perera P, Solé M, López-Andreu FR, Saraiva MJ. Vitreous amyloidosis after liver transplantation in patients with familial amyloid polyneuropathy: ocular synthesis of mutant transthyretin. Amyloid. 2000;7:266-269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 14. | Haraoka K, Ando Y, Ando E, Sun X, Nakamura M, Terazaki H, Misumi S, Tanoue Y, Tajiri T, Shoji S. Presence of variant transthyretin in aqueous humor of a patient with familial amyloidotic polyneuropathy after liver transplantation. Amyloid. 2002;9:247-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 15. | Ando E, Ando Y, Haraoka K. Ocular amyloid involvement after liver transplantation for polyneuropathy. Ann Intern Med. 2001;135:931-932. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 16. | Ando Y, Ando E, Tanaka Y, Yamashita T, Tashima K, Suga M, Uchino M, Negi A, Ando M. De novo amyloid synthesis in ocular tissue in familial amyloidotic polyneuropathy after liver transplantation. Transplantation. 1996;62:1037-1038. [PubMed] |
| 17. | Hara R, Kawaji T, Ando E, Ohya Y, Ando Y, Tanihara H. Impact of liver transplantation on transthyretin-related ocular amyloidosis in Japanese patients. Arch Ophthalmol. 2010;128:206-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 57] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 18. | Neto E, Ferreira A, Almeida L, Pinto F GM. Paramiloidose ocular após transplante hepático. Oftalmol (Port Ophthalmol Soc Journal). 2009;33:51-56. |
| 19. | Silva-Araújo AC, Tavares MA, Cotta JS, Castro-Correia JF. Aqueous outflow system in familial amyloidotic polyneuropathy, Portuguese type. Graefes Arch Clin Exp Ophthalmol. 1993;231:131-135. [PubMed] |
| 20. | Tsukahara S, Matsuo T. Secondary glaucoma accompanied with primary familial amyloidosis. Ophthalmologica. 1977;175:250-262. [PubMed] |
| 21. | Ando E, Ando Y, Okamura R, Uchino M, Ando M, Negi A. Ocular manifestations of familial amyloidotic polyneuropathy type I: long-term follow up. Br J Ophthalmol. 1997;81:295-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 101] [Article Influence: 3.6] [Reference Citation Analysis (0)] |