Published online Nov 19, 2021. doi: 10.5498/wjp.v11.i11.954
Peer-review started: February 25, 2021
First decision: May 13, 2021
Revised: June 5, 2021
Accepted: September 19, 2021
Article in press: September 19, 2021
Published online: November 19, 2021
Processing time: 264 Days and 4.8 Hours
Insufficient sleep has been correlated to many physiological and psychoneurological disorders. Over the years, our understanding of the state of sleep has transcended from an inactive period of rest to a more active state involving important cellular and molecular processes. In addition, during sleep, electrophysiological changes also occur in pathways in specific regions of the mamm
Core Tip: Sleep hygiene is thought to be necessary for memory consolidation and learning while sleep disturbances can alter both synaptic plasticity and memory consolidation. Recent findings indicate that the expression of G-protein coupled receptors (GPCRs) like metabotropic glutamate receptors, gamma-amino butyric acid-B receptors and serotonin (5-hydroxytryptamine) receptors as well as synaptic plasticity are altered following a short episode of sleep deprivation. GPCRs are involved in a variety of central nervous system functions and are targets for therapeutic agents in psychiatry and other neurological disorders. Therefore, a deeper understanding of these receptors in association with the state of sleep and its related functions and disorders is needed.
- Citation: Parmar S, Tadavarty R, Sastry BR. G-protein coupled receptors and synaptic plasticity in sleep deprivation. World J Psychiatr 2021; 11(11): 954-980
- URL: https://www.wjgnet.com/2220-3206/full/v11/i11/954.htm
- DOI: https://dx.doi.org/10.5498/wjp.v11.i11.954
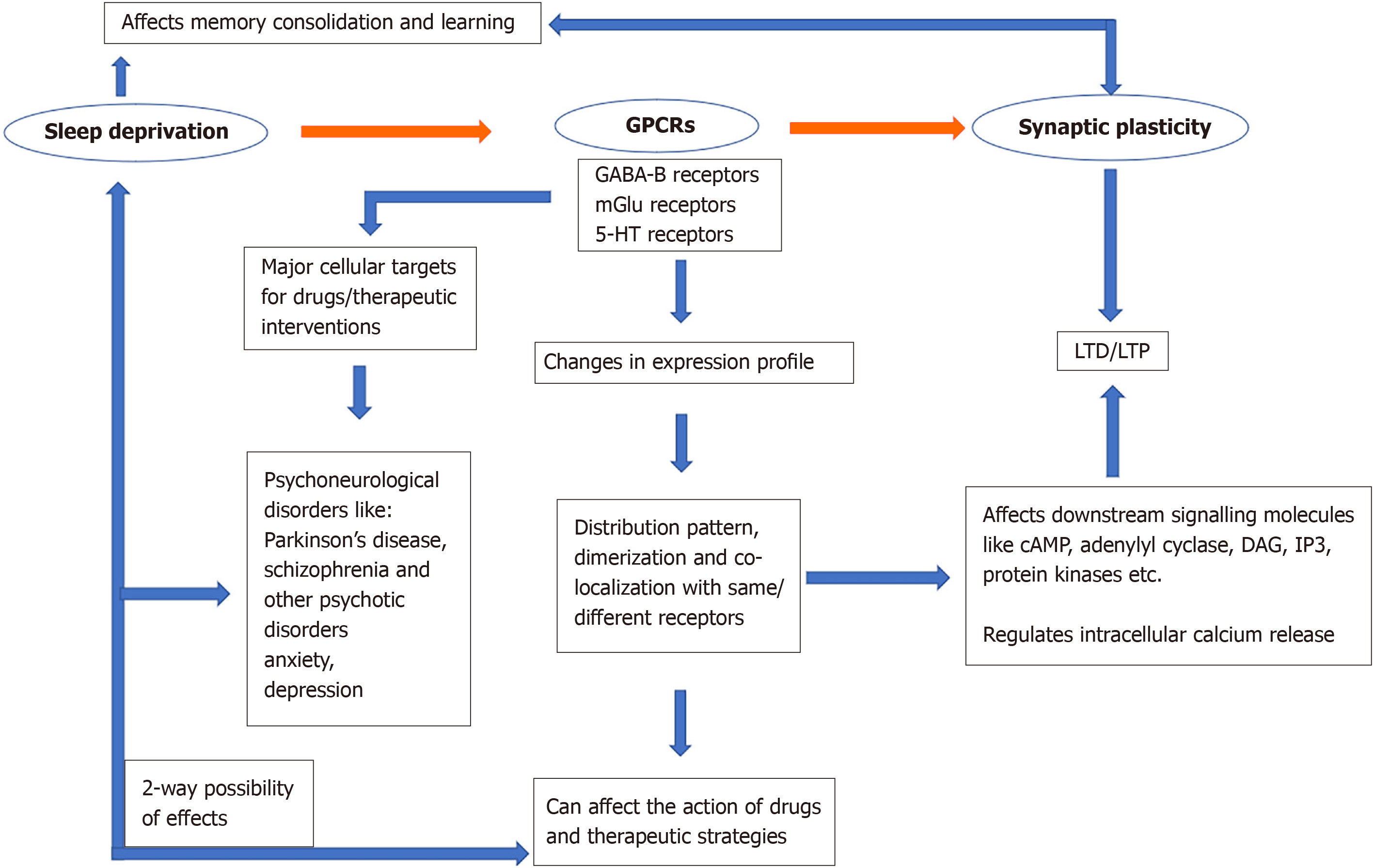
Sleep is one of the fundamental needs of most conscious living beings. Earlier sleep was more analogous to just the idea of resting and having our mind and body recharge for the activities in our consecutive state of wakefulness. We have moved far along from this initial notion as we now know that even in the state of rest, our body is constantly working right from the molecular to the more visible physiological and psychological levels. The implications associated with sleep deprivation manifest themselves both behaviourally as well as physiologically. In a way, both these implications are correlated considering external stimuli enables an animal to act in a certain way depending on what kind of signals the brain has processed. The processing of such signals is mediated by cellular components like genes and proteins. In different neurological disorders like depression, loss of memory, psychosis, hallucination, anxiety, etc., certain biomolecules are differentially affected. Synaptic plasticity which is thought to be a cellular correlate involved in memory consolidation and learning, is also affected following sleep deprivation.
In this review, therefore, we aim to understand and highlight the fundamental properties of sleep and sleep deprivation by keeping memory consolidation and synaptic plasticity as representative functions which get affected by the different states of sleep. In addition, any changes in G-protein coupled receptors (GPCRs) caused by sleep disturbance are emphasised.
Sleep is indispensable for most animals. Extensive studies in this area have now led us to believe that sleep is not a static phenomenon but it is heterogenous and dynamic even if its physical nature appears completely passive. There are interconnections between the thalamus, cortex and hippocampus regions of the brain and this interplay of networks is known to be operated by stage specific oscillations that take place while we are asleep[1,2]. Aserinsky was one of the first researchers whose studies overcame the concept of the cerebral cortex being dormant in sleep and gravitated the scientific community towards the reality of the brain being in an active state even while we are asleep[3]. There are stages or periods in sleep which transition alternatively from an active period, which is manifested by rapid eye movements (REM) to an inactive or quiescent period, also known as the slow-wave sleep (SWS)[3]. The oscillation patterns in the brain fluctuate during these two stages of sleep with high frequency and low amplitude waves being a characteristic of the REM/Late-night paradoxical sleep and the SWS portraying the contrary wave pattern[2,3]. Advancement in this area has revealed many more biological functions like respiration, thermoregulation, etc., along with the neurobiological processes of learning and memory formation in association with the alternating stages of REM sleep and SWS[4].
Disturbances in regular sleep cycles were assessed through the monitoring of EEG patterns. Waves of low amplitude and high frequency, associated with wakefulness, also occur during REM sleep. Deprivation of sleep is usually followed by a compe
The functional significance of sleep guides us towards the negative implications of sleep deprivation. A balanced and sufficient sleep cycle acts as one of the major factors that determine the quality of human life. There are a wide range of environmental, psychological and physiological factors that lead to sleep deprivation. Environmental changes have influenced many aspects of our day-to-day life and sleep disturbances can arise due to an increase in surrounding noise as well as fluctuations in light and temperature[12]. These factors add on to the list of causes that negatively burden the state of mind. Mental health also plays a crucial role in maintaining a regular sleep pattern since the prevalence of stress, anxiety, depression, etc., affects regular sleep cycle which can translate into insomniac conditions. Psychosis disorders like schizophrenia or neurodegenerative diseases like Alzheimer’s have often been associated with issues related to sleep deprivation like a reduced REM sleep cycle as well as a lowered sleep spindle activity[13,14]. Apart from mental disorders, pathophysiological illnesses (e.g., cancer, diabetes, respiratory disorders etc.) also often result in sleep disturbances due to manifestations like pain or difficulty in breathing. Interestingly, lack of sleep can also increase the prevalence of such physiological disorders since an important function of sleep is the regulation of the immune system[15]. Modern lifestyle changes like uneven working hours, over consumption of caffeine along with an increase in screen-time exposure have potentially interluded the quality and quantity of sleep. Homeostasis of the normal biological circadian rhythm is required for better cognition and task performance. For example, studies have implicated that night shift-workers, especially in chronic situations, experience varying levels of cognitive impairment and task performance[16,17]. Drug abuse and alcohol consumption which can lead to substance use disorder have also been recognized as a growing cause of sleep disruption. For instance, alcohol consumption is indicated to suppress REM sleep which in turn causes impairment in performing procedural tasks[18]. Conversely, lack of sleep affects the activity balance of important neurotransmitters like dopamine (DA) in the brain and this has been indicated to increase vulnerability to the use of drugs[19]. The above mentioned negative influences of sleep deprivation are just few of the many but it helps in bringing the importance of sleep deprivation into perspective.
Without sufficient amount of sleep, the brain cannot adequately perform the processes that take place during the state of sleep. Sleep deprivation affects the ability to concentrate, intake information and mediate that information through neuronal signalling, learn as well as process memories for consolidation. Furthermore, it is affected by the signalling and expression profiles of many biological molecules including the GPCRs[20,21]. These functions are related to the different stages of sleep and hence there are stage specific implications of sleep deprivation. For example, lack of REM sleep has been directed towards to an impairment in the development and expression of emotional and spatial memories[22,23]. Additionally, disruption of SWS is suggested to reduce attention span, affect motor activity and task performance[24]. Since learning and task performance are dependent on the memory processing of various responsible stimuli, the concept of memory consolidation has been highlighted to understand the significance of sleep and its associated cellular machinery.
Memory is the ability of living beings to retain information that they have acquired through their various day to day experiences and activities. The brain consolidates memories in different stages starting first from acquiring the memory through learning experiences, encoding and then consolidating those memories to be recalled or retrieved upon stimulus[25]. Interestingly, even the processed memories can become transient overtime and a reconsolidation of those memories is required for which sleep is essential[26]. While we sleep, the brain is actively carrying out its functions and ‘offline-reprocessing’ of memories appears to be one of them[3]. The presence of the rapid and spontaneous oscillations in the cortical networks of the brain during wake and sleep periods have been known to get triggered upon sensory stimuli[27,28] which can get incorporated in the brain in form of a memory. This gradual incorporation of acquired memories into the different regions of the brain is more crucial for long term memory than the short-term memory, which points towards a quicker mechanism of action[29,30]. REM sleep and SWS are generally associated with the consolidation of long-term implicit (non-declarative) and explicit (declarative) memories, respectively[31-33], both working in a double-step process. In support of the alternate nature of the REM sleep and the SWS, work done on the memory consolidation function of these stages has revealed that SWS also has its part to play in reactivation and redistribution of the spatio-temporal patterns of the neurons which are observed during the encoding stage of memory function when we are awake[31,34-36]. When there is distraction or disruption of the periods immediately after the learning or teaching stimulus, the formation of memory is interrupted and conse
The discussed oscillation and wave patterns during the state of sleep are at least partly a result of the electrical impulses that are transmitted between neurons via the synapses. This transmission modifies the synapses and hence synaptic plasticity is an important consideration while studying about the various functions of sleep including memory consolidation. To better understand this correlation, we will first discuss the regions and cells of the brain that are associated with sleep dependent functions and how these regions communicate via signal transmission.
As mentioned earlier, activities in the brain during the state of sleep are in conjunction with changes in the oscillation and wave-patterns in specific regions of the brain. These complex wave-sequences form a characteristic component of the neocortical and the thalamic system, which co-ordinate in order to exhibit the various functions of sleep, including memory consolidation. Although the mechanisms that are involved in the functions and regulation of memory consolidation are yet unclear, various cellular processes in brain regions have been studied as potential candidates. Amongst these, the hippocampus is one of the main centers for processing sleep dependent memories through neurons like granule cells and pyramidal cells[40]. The significance of the hippocampus and its sub-regions may be attributed to its function of independently giving the neuronal networks the ability to process multimodal information and building an integrated system that can represent the newly acquired information for its consequent storage in the higher centers of the brain in the form of 'memories'[41]. Inspired by this finding, a two-staged memory consolidation concept was put forward which essentially highlighted the events wherein there occurred a transmission of information between the hippocampus (short-term storage) and the neocortex (long-term storage) through the exploratory theta burst stimulations of the granule cells and sharp waves of the pyramidal cells[42]. While the hippocampus is concerned with high frequency oscillations and sharp wave ripples in the CA1 and CA3 regions, the neocortex shows slow oscillations and spindles during SWS[43]. During SWS, the signal from the hippocampus to the neocortex is transmitted through the CA1 neurons whereas during REM sleep the information enters the hippocampus from the neocortex through the CA3 neurons; thus, causing the CA1 neurons to hyperpolarize the CA3 neurons through the entorhinal cortex[43]. This rhythmic communication of oscillations gives rise to coupling as well as consistent spike timing relationship between the neocortex and the hippocampus which further strengthens the information processed for learning and memory consolidation[43-45].
As discussed above, there seems to be a pattern of relay of information that is associated with sleep dependent functions between the hippocampus and the neocortex. Since the relayed signals go through synapses, any plasticity at the junctions can significantly alter the above mentioned patterns. Therefore, in this review, a discussion on the phenomenon is made.
The active nature of brain during the state of sleep so far has been advocated by multiple studies on the specific regions of the brain which primarily show distinct wave patterns and relay information through signal transmission. Transcending from the Hebbian[46] theory, the strength of these signals has been associated with the continuous firing of action potentials from a pre-synaptic neuron to a post-synaptic neuron. It has appeared that a signal gets stronger when it is repeatedly transmitted from one synapse to another and this is where the phenomenon of synaptic plasticity comes into play. When there is continuous firing of signals between neurons, there are changes that take place at the synapses of these cells, which means that the cellular components at these junctions also undergo changes and consequently affect the strength of the signal being transmitted. This concept can be appreciated by looking at instances wherein it appears that by repeatedly performing a task, we get better and better at it. In a similar way, a repeated stimulus to the brain results in repeated signal transmission from one neuron to another and this can have the potential to strengthen that signal[46]. This takes place through synaptic plasticity which entails molecular as well as electrophysiological changes at the synapses. The electrophysiological changes can be characterized by long term depression (LTD) and long term potentiation (LTP), each relating to the persistent weakening and strengthening of the synapses, respectively. Initial work done in this area revealed that at the excitatory synapses, there occurred a use-dependent and long term strengthening of the synapse upon high frequency stimulation of the pre-synaptic fibres in the hippocampus, which was termed as LTP[47]. Analogous to the Hebbian[48] findings, experimental data at cellular level has indicated that synaptic plasticity in the form of LTP in the hippo
| Prominent brain regions | Receptors involved | Induction | Maintenance | Association with sleep | Functional significance | |
| LTP: Strengthening of synaptic transmission due to a transient high frequency stimulation of the synapses | Cerebellum, hippocampus, cerebral cortex | Ionotropic: NMDARs, AMPARs | Requires activation of both pre- and post-synaptic neurons at the same time for glutamate to fully activate the NMDA receptors [51]. | Secondary activation of receptors like AMPARs by glutamate needed for maintenance of LTP[51]. | Active nature of LTP likely associated with active state of sleep. | Enhances synaptic response as well as neuronal excitability. |
| Metabotropic: Group I mGluRs | Ionotropic induction requires activation of NMDARs by glutamate released from pre-synaptic neurons for higher | Requires activation of intermediate protein kinases like CaMKIV, protein kinase M-ζ, PKA etc. by high Ca2+ concentration to release membrane obscured AMPARs[56]. | More commonly linked with REM sleep associated cellular and molecular modulation of synaptic plasticity[57]. | Involved in memory consolidation and learning: Strengthened synapses promote long-term memory storage[58]; Promotes associative and spatial learning[59]; Involved in motor learning and task reperformance[60,61]. | ||
| Metabotropic induction works via an increase in intracellular Ca2+ release through mGluR activated phospholipase C and synthesis of secondary messengers IP3 and DAG[54,55]. | ||||||
| LTD: Weakening of the synaptic strength due to a relatively low frequency stimulation of the synapses | Cerebellum, hippocampus, cerebral cortex | Ionotropic: NMDARs | Activation of pre-synaptic neuron sufficient to trigger a moderate response of NMDA receptors and does not require both synapses to be activated at the same time[51]. | Deactivation of AMPARs involved in maintenance of LTD[131]. | Suppressive nature of LTD more likely to be associated with quiescent state of sleep. | Interferes with LTP thereby providing a counteractive balance to prevent hyperexcitability of neurons[52]. |
| Metabotropic: GPCRs like mGlu1Rs, mGlu5Rs, GABA-B | Ionotropic induction requires moderate activation of NMDA receptors by glutamate for Ca2+ influx in post-synaptic neuron which is lower than that for LTP induction[52]. | Requires activation of intermediate protein phosphatases like protein phosphatase 1, 2 and calcineurin by low Ca2+ concentration to inhibit the release of membrane obscured AMPARs[53]. | More commonly linked with SWS associated cellular and molecular modulation of synaptic plasticity[64]. | Involved in memory consolidation and learning: Weakened state of synapses implicated in forgetting old memories in order to make space for new ones [65]; Involved in novelty acquisition and spatial learning[66]. | ||
| Metabotropic induction through mGlu1Rs and mGlu5Rs causes endocytosis of the expressed AMPARs and/or a decrease in phospholipase C via reduced adenylyl cyclase activity[62]. | ||||||
| Enhancement of GABA-B mediated inhibitory effects may result in LTD[63]. |
The process of learning and memory consolidation that takes place while we are asleep can also be attributed to further molecular changes that can take place following the synaptic strengthening. A disruption of these due to sleep deprivation can, thus, have a negative consequence.
In spite of having a breakthrough discovery about LTP and LTD causing long-lasting modification in the synapses in the 1970s[47,67], the functional implications of LTD and LTP in learning and memory processing are still under a lot of conjecture. While the initial studies relied more on the active state of synapses through LTP as the focal model for learning, subsequent studies also implicated the role of LTD by highlighting the fact that a consequent increase and decrease in signal strength is what allows the phenomenon of synaptic plasticity to function without getting worn out[52,58,59,68]. For instance, Tsumoto[65]’s work highlights LTD in cerebral cortex being responsible in 'forgetting' certain memories in order to make place for the new ones. Looking back at Hebbian[46] and the follow-up findings, the role of synapses and the existence of synaptic plasticity as cellular correlates of learning and memory consolidation become apparent. As discussed in the previous sections, learning and memory consolidation are a result of collective cellular communications through the processing of the learning stimuli. The cortical-hippocampal dialogue is thought to drive these learning stimuli forward in the brain to encode, process and store the information acquired through those stimuli. Several lines of studies demonstrate the correlation of memory acquisition with the processing of spatial learning to LTP and LTD[65,66,69]. Furthermore, exploratory behaviours and learning are known to promote information acquisition[70] and experimental studies on rats that were exposed to hole board exploration resulted in events that reversed LTP upon exposure to a novel environment with inductive LTD mechanisms being activated. These rats showed habituation upon a second exposure to that environment indicating LTD’s possible role in remembering the previously acquired information of that environment[66]. On the contrary, work done on adult rats has also revealed the significance of LTD and LTP by demonstrating that the preservation of LTP is hampered upon an increase to LTD susceptibility and this also interrupts the CNS functions including learning and information retention[71,72]. Therefore, there are debatable influences of LTD on learning but its role in novelty acquisition and spatial learning[66,73] deserves further investigation.
What exactly is harnessing this LTP and LTD in these regions of the brain and how these activity dependent synaptic changes relate to memory consolidation and learning? In this review, an attempt is made to find some answers. An early hypothesis put forward by Lynch and Baudry[74] on the biochemistry of memory involved an examination of calcium proteinase-receptor interaction. They demonstrated that in the forebrain sub-synaptic membranes, an influx of Ca2+ causes a long lasting increase in the number of glutamate receptors through its activation of the enzyme calpain which is a proteinase. The activated form of this enzyme degrades the membrane-anchored cytoskeleton protein fodrin and as a result, exposes the obstructed glutamate receptors. The effects of this Ca2+ proteinase-receptor interaction could functionally modify neuronal circuits as well as showed similar biochemical effects that are seen post learning[74]. This and other related studies examining molecular mechanisms were, therefore, examined to find correlations between synaptic strength and memory consolidation.
Most work done on excitatory neurons like the hippocampal pyramidal cells reveal that tetanic stimulation of inputs can readily induce LTP that lasts for hours, days and can even be permanent, making it a good cellular correlate of memory and learning[75-77]. In vivo and in vitro induction of LTP and LTD through external theta burst stimulations in model organisms[47,49,78] has revealed the various cellular components that present themselves as functional entities in the process of memory formation and learning. It is already well established that receptors like metabotropic glutamate receptors (mGluR), ionotropic NMDA and AMPA receptors are mediators for LTP and LTD through the increase or decrease in the Ca2+ concentration, respectively[51,79,80]. Both ionotropic (e.g., NMDA, AMPA receptors) and metab
The active state of sleep has been associated with many functions, including synaptic plasticity (LTP and LTD), cognitive learning and memory processing[87]. In fact, it is also reasonable to suggest that learning paradigms that promote sleep and synaptic plasticity also enhances brain development[57,88].
The above notions have risen from earlier hypotheses that sleep affects the patterns of neuronal circuits in the brain[42,89]. REM sleep is associated with long-term memory consolidation and learning events promote synaptic plasticity. Moreover, REM is required for brain development and an increase in intensive learning activity significantly enhances the duration and number of REM sleep cycles and promotes synaptic plasticity[90,91]. Correlating sleep cycles and synaptic plasticity, LTP has been credited with many learning functions of the brain, especially in the hippocampus. It appears that sleep deprivation results in an inhibition or decrease in the level of LTP and an impairment in the associated learning[92,93]. This trend has also been observed in rats during sleep fragmentation where in there was a decrease in LTP through NMDA receptors in the CA1 neurons of the hippocampus[94]. Interestingly, the reduction in LTP is also associated with a downregulation of cortactin, which is a dendritic cytoskeleton protein, pointing towards the role of LTP in the structural modification of neurons and a disruption upon sleep deprivation[95]. While the impact of sleep deprivation on LTP seems clear, the fate of LTD has not been adequately identified. However, LTD is known to protect neurons by regulating neuronal hyperexcitability and preventing a saturation of network activity[59]. Additionally, LTD’s role in memory consolidation has been seen through events like novelty acquisition and spatial learning or even in making space for new memories through the concept of ‘forgetting’[65,66,73]. Since these aspects of memory processing are sleep dependent, it is logical to suggest that sleep deprivation can affect LTD. In agreement with this line of thinking, an increase in hippocampal LTD following sleep deprivation was observed in rats[96].
In conclusion, sleep seems to promote synaptic plasticity, especially LTP, and memory consolidation, while sleep disturbances appear to adversely affect both. LTD seems to be enhanced following sleep deprivation and, thus, may be associated with interference with memory consolidation.
After a brief introduction of a few cellular components involved in the functioning of synaptic plasticity in section II, some detailed events in relation to the state of sleep are described here. Going back to the fundamentals of electrophysiology, an electrical signal is transmitted when a neurotransmitter gets released from the pre-synaptic cell and activates its specific receptor on the post-synaptic cell. This further invites a series of downstream reactions that ultimately give rise to the required effect. Neurotransmitters and receptors are cellularly synthesized molecules and if the signal pattern changes during stages of sleep and wakefulness, changes in the expression of genes and proteins, that are precursors for these molecules, can also occur. In sleep and sleep deprivation, glutamate, acetylcholine, DA, serotonin, etc., are important transmitters to pay attention to. Acetylcholine, for instance, is known to be correlated with the amplitude of theta burst oscillations that take place in the hippocampus. Events that lower the levels of acetylcholine can reduce the amplitude of such oscillations[97]. Since sleep sensitive processes of learning, like memory consolidation demonstrate oscillatory changes, neurotransmitters like acetylcholine are significant to such brain functions. Additionally, imbalances in the levels of neurotransmitters like DA and serotonin are associated with many neurogenic issues like depression, anxiety and psychosis; all of which are symptoms often seen in patients that have been sleep deprived[98-100].
Focusing more on the receptors involved in mediating synaptic plasticity in excitatory glutamatergic pathways, the glutamate receptors present themselves as frontline players and amongst these, the ionotropic NMDA and AMPA receptors are known to depolarize/excite the post-synaptic neuron through an influx and efflux of Na+ and K+ ions, respectively[83]. The main inhibitory neurotransmitter of the CNS is GABA and it acts upon the two types of GABA receptors: Ionotropic GABA-A/GABA-C receptors and metabotropic GABA-B Rs. GABA’s contribution to modulating LTP and LTD in excitatory pathways, owing to the strategic location of its receptors to regulate input and output signals in the pyramidal neurons, is well documented[101-103]. LTP and LTD are carried out by the long term excitatory and inhibitory actions of these receptors including GPCRs, which will be looked into in the next section. Ca2+ has been a crucial factor and its increase has shown to depend on the activation of NMDA receptors by glutamate, Ca2+ release from intracellular stores and its influx through the voltage gated calcium channels[104]. The binding of the released calcium to calmodulin and then further to two other proteins: Ca2+/calmodulin dependent protein kinase type 2 (CAMK II) and calcineurin, results into LTP and LTD, respectively[104].
In continuation with the molecular changes associated with synaptic plasticity, protein synthesis and post-synaptic biochemical changes have been implicated in different phases of LTP[105]. For instance, brain derived neurotrophic factor, protein kinase Mζ, calcium/CAMK II, and activity-related cytoskeletal protein, have so far been indicated in the induction or maintenance of late phase LTP[106-109]. Whether these or other factors participate during sleep-induced consolidation of memory is unknown and needs future investigation. Another not so conventional molecule that is speculated to promote synaptic plasticity is nitric oxide (NO). What is intriguing about this molecule is that the enzyme which synthesizes it is activated by an increase in Ca2+ concentrations which again is dependent on receptors like NMDA which promotes synaptic plasticity[57,110]. More insight on such accessory molecules needs to be put forward through detailed experimental work and NO has so many other important physiological functions like decreasing vascular resistance and increasing cerebral blood flow and oxygenation rate[111] or playing a key role in mediation of an immune response against infectious diseases[112]. Thus, its definitive role in sleep deprivation through synaptic plasticity can be a major contribution to this field of research.
In summary, these are just few of many cellular components which have been discussed here to help understand and appreciate the intricate network of cellular molecules in driving synaptic plasticity which also play an important part in carrying out sleep associated functions. The functions carried out by these regulatory molecules have been known to get altered during the state of sleep and sleep deprivation. This means that the genes that transcribe the proteins for these receptor molecules also undergo changes. Differential gene expression has been observed during both the states of sleep and wakefulness. In fact, modulation in the gene expressions during these two states have shown that around 10% of the total cerebral cortex transcripts are differentially expressed[113]. For instance, the mRNAs for the receptors of the inhibitory neurotransmitter GABA, shows a higher expression during the state of sleep as compared to the state of wakefulness[113,114].
GPCRs by definition are receptors that mediate their effects after binding to G proteins which are heterotrimeric molecules made up of alpha, beta and gamma subunits that function by phosphorylating the nucleotide guanosine triphosphate to guanosine diphosphate. Structurally, GPCRs are transmembrane proteins with multiple transmembrane domains and the homology of these domains categorize them into 4 classes (A, B, C and F). About 90% of the non-sensory GPCRs are expressed in the brain and appear to play regulatory roles in various neurological processes. The expression profile of these receptors is extremely high in the hippocampus (around 300 GPCRs) and about 20 of these GPCRs display their potential role in synaptic plasticity[115]. The signalling pathways that are induced by the GPCRs regulate both pre and post-synaptic components which ultimately can affect synaptic plasticity as well as the release of pre-synaptic vesicular molecules[116]. They are also known to be involved in events of structural plasticity as well as cognitive development[117]. GPCRs can also alter NMDA receptors by a direct action at the CA1 synapses, thus indirectly affecting NMDA receptor-mediated synaptic plasticity[118], suggesting the possibility for receptor co-expression. Furthermore, GPCRs also seem to elicit their effects intracellularly while modulating synaptic plasticity[119]. The range of neuronal functions that are attributed to GPCRs needs a more coherent and concrete investigation to be well defined in relation to sleep deprivation, synaptic plasticity and memory consolidation. For the purposes of this review, two major types of GPCRs known as the mGluRs and GABA-B Rs are discussed in more detail. Other GPCRs, especially those that are important for drugs used in psychiatry, are also discussed.
The mGluRs belong to the class C of GPCRs. They are categorized into three groups based on their signal transduction pathways. Group I (mGlu1R and mGlu5R), Group II (mGluR2 and mGluR3) and Group III (mGluR4, mGluR6, mGluR7, mGluR8)[120]. The subdivisions are based on the difference in their physiological activity and structure. The group I receptors display a post-synaptic location and are known to act via the Gq protein through the activation of phospholipase C protein and the synthesis of the secondary messengers such as inositol-1,4,5-triphosphate (IP3) and diacylglycerol (DAG). The general pathway involving these secondary messengers results in the release of intracellular Ca2+ stores which is a prerequisite for the induction of synaptic plasticity[54,55]. These receptors are also involved in the modulation of neuronal excitability in the hippocampus pyramidal cells wherein they activate cationic conductance through the reduction in resting K+ current and inhibition of Ca2+ ion channels[121,122]. On the other hand, mGluRs were also seen to induce IP3-mediated release of Ca2+ from intracellular stores through stimulation of CA3 pyramidal neurons. Unlike the Group I mGluRs, Group II and III mediate their functions via the Gi/Go protein. They are negatively coupled to adenylyl cyclase and inhibit the production of cyclic adenosine monophosphate Cyclic adenosine monophosphate (cAMP)[123] which results into activation of K+ channels and inhibition of Ca2+ channels. These two groups have a very distinct pre-synaptic localization pattern with Group II being predominant in extra synaptic sites whereas Group III in the synaptic sites. Additionally, the Group III receptors also appeared to be segregated in correlation to their target post-synaptic neurons[124].
The metabotropic GABA-B Rs are widely known for their inhibitory action in the CNS and like mGluRs, also belong to the class C GPCRs. Structurally, these are obligate heterodimers made up of GABA-B1 and GABA-B2 receptor subunits which are genetically co-expressed and are homologous to the structure of mGluRs[125,126]. Gene expression studies for this receptor have shown that heterodimerization is essential for the GABA-B Rs to elicit their function since the individual expression of the GABA-B1 subunit in the target mammalian cells results in immature and low functionality receptors as compared to the heterodimer[127]. These receptors show a wide cellular and sub cellular localization pattern through their expression on pre-synaptic, post-synaptic as well as extra synaptic regions in the brain[128]. Based on work performed on rat hippocampus, the two subunits of GABA-B Rs: GABA-B1a/b and GABA-B2, appear to be more on the post-synaptic regions as compared to the pre-synaptic ones. Furthermore, they also displayed an abundant distribution of GABA-B1a/b on glutamatergic synapses on the spines and on dendritic shafts of pyramidal cells[128]. GABA-B2 on the other hand, has a dominant extra-synaptic localization. These localizations are indicative of their functions and considering the range of cellular and subcellular occurrences of these receptors, they show a promising involvement in a great number of neurogenic processes. For example, their presence at the glutamatergic terminals points towards their involvement in glutamatergic neurotransmission[128].
Functionally, GABA-B Rs induce their slow and long lasting action via the Gαi/o proteins through the inhibition of adenylate cyclase and like the mGluRs, they too affect the Ca2+ and K+ conductance[129]. GABA-B Rs are known to control the calcium dependent neurological processes through the inhibition of voltage sensitive Ca2+ channels[85,130]. On the pre-synaptic sites, GABA-B Rs appear to be negatively coupled to Ca2+ channels and block the Cav2.2 (N- type) and Cav2.1 (P and Q type) voltage gates channels via the action of G- proteins. They also have the ability to affect K+ channel directly without the involvement of a G protein. This controls the release of neurotransmitters from the pre-synaptic neurons and thus GABA-B Rs have the potential to regulate the onset of a stimulatory signal[85,130]. At the post-synaptic sites, these receptors mediate their effects via the G proteins through a slow hyperpolarization by inward rectification of K+ channels such as the GIRK channels as well as Ca2+ channels[131]. GABA-B Rs are, therefore, diverse both in their structural as well as functional mediation of inhibitory signals and show immense potential to regulate synaptic plasticity in association with sleep deprivation.
The serotonin, DA and epinephrine group of GPCRs are very important in psychiatry considering their ligands are responsible for a wide range of CNS functions like cognitive learning and sleep-wake behaviours and are implicated in many neurological disorders. Serotonin receptors, also known as the 5-hydroxytyrptamine (5-HTA) receptors are classified into 7 families (5-HT1 to 5-HT7), all of which are GPCRs except the 5-HT3 receptor[132]. These are further divided into their respective 14 sub-types [e.g., 5-hydroxytyrptamine subtype 1A (5-HT1A) to 5-HT1F] based on their function and location[132]. They exhibit their function mainly through the Gαi, Gαq/11, and Gαs proteins followed by modulation of Ca2+ concentrations via the regulation of adenylyl cyclase and cAMP or activation of phospholipase C producing IP3 and DAG[133]. In the CNS, 5-HT1 and 5-HT5 are associated with inhibitory signal transduction and the others are excitatory in nature; with pre- and post-synaptic localizations. For instance, the 5-HT1A receptors, which are widely implicated in depression, anxiety and learning, are found in both pre-synaptic (auto-receptors) and post-synaptic (heteroreceptors) locations[134]. Additionally, structural significance in this class of receptor through dimerization with each other and other GPCRs has also been indicated as a functional prerequisite for the cellular trafficking and functioning of 5-HT receptors[135].
DA receptors are GPCRs divided into 5 subtypes (D1-D5) and exhibit their action through the Gs/olf and Gi/o proteins by activating or inactivating adenylyl cyclase, respectively[136]. These receptors have a wide pre and post-synaptic distribution pattern in the CNS which contributes to the functional significance of DA[137]. For instance, the D1 and D2 receptors are abundantly localized in the striatum and substantia nigra region of the brain and are involved in the nigrostriatal pathway which is responsible for the control of bodily movements[138]. These are also majorly distributed in the ventral tegmental area (VTA) of the brain which is connected to the ventral striatum and this connection promotes reward-associated behaviour via the meso-limbic pathway[139]. Furthermore, along with a localization pattern observed in both pre- and post-synaptic regions, co-expression is also a general trend seen with DA receptors[140]. Lastly, the GPCRs that are the target for the catecholamine norepinephrine/noradrenaline (NA) are classified into alpha-1, alpha-2, beta-1, beta-2 and beta-3 receptors and they are localized on both pre- and post- synaptic sites. Alpha-1 follows the Gq protein mediated elevation of calcium levels by activation of phospholipase C producing IP3 and DAG and the other three subtypes work via the modulation of adenylyl cyclase pathway[141]. Interestingly, co-localization patterns have also been observed between DA and NA receptors in the rat prefrontal cortex[142]. Whether such co-localizations and/or dimerization have implications for pharmacological actions of therapeutic agents need further investigation.
As introduced above, GPCRs appear to be involved in processes that regulate the transduction of action potential especially in the cells of the hippocampus region of the brain. Both LTD and LTP have demonstrated a pattern of being modulated by such regulatory molecules. It is already well established that Type I mGluRs induce LTD through the synaptic endocytosis of AMPA receptors in the hippocampus[143]. Similar lines of studies have pointed towards mGluRs in the CA1 pyramidal cells being involved in the induction of LTD through post-synaptic elevation of Ca2+ levels[144]. Interestingly, studies on the cerebellar Purkinje fibres have also demonstrated that activation of GABA-B Rs enhances LTD through an association with mGluRs[63]. Even though the mechanism of action of these receptors may be related, many of their molecular machineries have their own distinctive features while promoting cellular LTD. For instance, in the Purkinje fibres, LTD mediated via GABA-B relied on the beta gamma subunits of the G-protein whereas mGluR mediated LTD depended on the alpha subunit[63]. So far, it appears that these GPCRs are associated with the induction of LTD and reviewing other related studies, we know that NMDARs are the main mediators of LTP. Contrary to these findings, recent experimental work has put forward a type of LTP that is induced in the absence of NMDARs and requires the activation of Type I mGluRs[145]. As for the role of GABA-B Rs in LTP, its auto receptors depress their own activity through negative feedback and promotes NMDAR mediated LTP, thus maintaining a balance between LTP and LTD[146]. Considering how the hippocampus and the cortex communicate and manifest their functions of learning and memory consolidation in the state of sleep through synaptic plasticity, it is reasonable to imply that these GPCRs modulating LTD and LTP are significant for this purpose. It also seems that co-expression of GPCRs and their heteromeric subunits is a repeated trend seen in studies that show the association of GPCRs with LTD[63,125,126]. The functionality of GPCR dimerization has also been put forward in earlier demonstrative studies which showed that the GABA-B1 subunit needs the GABA-B2 subunit to reach the cell surface and that GABA-B1 is responsible for agonist interaction whereas the B2 subunit works towards the G-protein activation[147,148]. Furthermore, gene knock-out or elimination studies for these subunits have also indicated the dependence on dimerization for the functioning of this receptor and that in some regions of the brain, GABA-B subunit gene elimination does not affect the functionality of the receptor which implies that it is replaceable by other G-protein activating molecules[149] and thus may have the potential to associate with other GPCRs to mediate their actions. Progressing from these initial findings, recent work done on rats that were sleep deprived using gentle prodding and tapping has demonstrated that induced LTD of population EPSP in the hippocampus requires activation of mGluRs and GABA-B Rs along with an increase in Ca2+ released from intracellular stores[150]. In sleep deprived conditions, western blot analysis and co-immunoprecipitation studies revealed that there were elevated expression levels of mGlu1αR and GABA-B1 receptor subunit as well as enhanced co-expression and heterodimerization between mGlu1αR and GABA-B R1 subunit and mGlu1αR and GABA-B R2 subunit[150].
As introduced earlier, GPCRs for serotonin are diverse both in their localization and functions. Their effects are mediated by calcium levels, which again is crucial for synaptic plasticity. Furthermore, 5-HT1A receptors are known to decrease NMDA receptor activity whereas 5-HT2A tends to increase it[151,152], thereby playing an opposing role in regulating NMDA mediated LTP. These are also co-expressed in hippocampal pyramidal cells and may direct studies towards their reciprocal action in regulating synaptic plasticity. Similar cross-talks have also been suggested between 5-HT and GABA-B receptors[153], which as already discussed, are involved in modu
DA and NA receptors demonstrate somewhat analogous modulations of synaptic plasticity. In rat prefrontal cortex, DA receptors appear to modulate both LTD and LTP through glutamatergic synapses[157]. In the hippocampus, opposing effect of D1/D5 and D2/D3/D4 receptors on NMDA receptor modulation, regulates LTD and LTP[158]. Furthermore, D1/D5 have also been implicated in novelty acquisition through hippocampal LTD and LTP[159]. The induction of synaptic plasticity via NA receptors is more prevalent through the beta group receptors and they do so by modulating both NMDA and AMPA receptors in the pyramidal cells and perirhinal cortex[158,160]. Consequently, the modulation of synaptic plasticity via DA and NA receptors can be extended towards sleep associated functions. Studies in rats have indicated that sleep deprivation leads to a differential expression of DA receptors[161] and chronic sleep restriction results in changes in the density of NA receptors[162]. Considering the range of these GPCRs and their functions, their cross-talk properties and effects during the state of sleep through synaptic plasticity require further detailed investigation.
Another intriguing and related aspect of GPCRs that signifies its structural implications in synaptic plasticity is the allosteric modulation of these receptors. Binding at sites other than the active site of the GPCR, it has been suggested that allosteric modulators can selectively stimulate the homomeric and heteromeric forms of the receptor. For instance, in the CNS, heterodimerization is seen between mGlu2R and mGlu4R with allosteric modulation being targeted to the homomeric forms relative to the heteromeric receptors[163,164]. Furthermore, LTP and LTD and spatial learning at the excitatory synapses in the hippocampus have appeared to be enhanced by the positive allosteric modulation of mGlu5Rs[165]. Considering even GABA-B receptors demonstrate their sleep dependent synaptic changes by forming such heteromeric complexes and associations with mGluRs[150], the importance of such structural modifications and associations needs more attention with respect to sleep dependent functions.
Sleep is important for normal physiological and neurological functions and sleep deprivation affects majority of these functions. The negative effects of sleep deprivation can be better managed when we are able to specifically target the cellular and molecular machinery that drives these functions and, in this review, GPCRs are examined as a pivotal molecular machinery that impacts sleep associated functions through synaptic plasticity. For example, memory consolidation and learning as a model involving hippocampal-neocortical dialogue was looked at. The dialogue, in turn brings forth distinct electrophysiological patterns that are active during the SWS and REM sleep stages. Activity mediated synaptic plasticity has been widely correlated to sleep associated functions of cognitive learning, memory processing and over all brain development. GPCRs like GABA-B Rs and mGluRs are types of receptors that have shown a potential to be involved in these processes through their actions on LTD and LTP. What is even more intriguing is the fluctuation in their distribution, heterodimerization and co-localization following sleep deprivation, suggesting that these receptors can exist in one condition during normal sleep and change with sleep deprivation. GPCRs are one of the major cellular targets for drug interaction and therefore changes in the receptor expression profile can affect drug action.
For instance, the antipsychotic drug clozapine is one of the common and effective drugs used in treating disorders like schizophrenia. The cellular targets for this drug are GPCRs like 5-HT2A and DA D2 receptors with a higher affinity for the former receptor than the latter. It acts as an antagonist of DA D2 receptors in the mesolimbic pathway and although initially classified as an antagonist, clozapine is also known to be an inverse agonist of the 5-HT2A receptor present in the prefrontal cortex of the brain[166-168]. Other antipsychotic drugs like olanzapine, risperidone, aripiprazole, etc., have also been indicated to elicit their effects through these receptors[166]. Interestingly, 5-HT2A and mGluR2 receptors are suggested to form heterodimers which raise the possibility of testing if clozapine and other such antipsychotic drugs modulate similar complex formation[156]. Moreover, drugs like clozapine are known to have sleep-inducing effects[169] and since disorders like schizophrenia and other psychiatric diseases are associated with sleep disturbances[170], potential interplay between the molecular events of sleep deprivation and actions of antipsychotic drugs needs to be investigated. Other examples of drugs that act on GPCRs include ropinirole for Parkinson’s disease which acts as an agonist on DA D2 receptors[171] and baclofen, a GABA-B receptor agonist suggested for treatment of depression and anxiety[172]. Whether their actions change with sleep deprivation and vice versa also needs to be tested. With regards to synaptic plasticity, antipsychotics and antidepressants are suggested to affect LTP upon both acute and chronic use and interestingly, these effects are different for each of these situations which may suggest differences in network behaviour with acute vs chronic exposure to the drugs (see Table 2[173-211]). Moreover, their effect on LTD has not been sufficiently explored and hence similar studies on the differential effects of chronic vs acute use of such drugs on network behaviour are required with regards to LTD. Table 2 provides a brief summary on antipsychotic, antidepressant and anxiolytic drugs with their mechanisms of action and association with synaptic plasticity, learning and sleep.
The relation between a drug’s mechanism of action and sleep can be directly observed in drugs that are used for treating sleeping disorders like insomnia. Characterized by a lack of both quality and quantity of sleep, insomniac conditions are treated with many drugs that target GPCRs. For example, melatonin is a hormone that has been known to play a key role in the sleep-wake cycle and promoting sleep. Agonist drugs like ramelteon act on the melatonin GPCRs type 1 and 2 (MT1 and MT2) which results in reduced sleep latency in chronic insomniac patients[212]. Intriguingly, the MTI and MT2 receptors have been known to form dimers among themselves as well as heterodimers with other GPCRs like the serotonin 5-HT2C receptor[213,214]. Thus, differences in ligand selectivity for these homomeric and heteromeric forms of the target receptors can have important consequences if the expression profile of such GPCRs change upon sleep deprivation. Other drug targets for insomnia include neuropeptides like orexins which promote wakefulness and are synthesized by neurons in the hypothalamus region of the brain. Drugs like suvorexant act as antagonists of the orexin GPCRs type 1 and 2 (OX1R and OX2R) thereby blocking their wakefulness promoting effects[215]. These GPCRs known to be associated with other GPCRs like the cannabinoid receptor type 1 (CB1) and GABA-B receptors. They form heterodimers with the CB1 receptors[216] which have been implicated in affecting memory formation and maintenance of mood. Moreover, orexin and GABA-B receptor activity are indicated to have a balancing interplay wherein inhibitory GABA-B modulates the wakefulness promoting properties in the orexin producing neurons[217]. If expression profiles of GABA-B receptors change with sleep deprivation, can there be consequences for actions of orexins? In summary, changes in receptor profiles associated with sleep deprivation can have consequences for drug action and need a thorough investigation to understand the mechanisms involved so that CNS disorders can be treated with more rationally based therapeutics.
Unlike receptor redistribution and co-localization, heterodimerization can have serious implications for drug-receptor interactions and consequently, its action. On the other hand, if co-localization of different receptors leads to common transduction pathways, it can significantly affect the actions of such drugs. These consequences can vary depending on whether pre- or post-synaptic receptors are changed with sleep deprivation. Since this process is dynamic and the receptors may be in different states, an understanding of the mechanisms involved in clearly needed. Moreover, since GPCRs among 5-HT, norepinephrine, DA, etc., may be affected, drug therapy involving antidepressants and antipsychotics should take the occurrence of receptor plasticity with sleep deprivation into account as drug-receptor interactions may be changed (Figure 1).
| Class of drugs | Drug examples | Target GPCRs | Mechanism of action | Therapeutic application | Possible association with synaptic plasticity and learning | Implications in sleep and associated functions | |
| Antipsychotics | 1st generation / typical antipsychotic drugs | Chlorpromazine | DA D1/D2/D3, 5-HT2A, Histamine H1, α1-Adrenergic, Muscarinic receptors. | 1 Acts as an antagonist by blocking post-synaptic DA D2 receptors. | 1 In the treatment of psychotic disorders like schizophrenia, bipolar disorder, acute mania, dementia, etc. | 1 Synaptic plasticity: Acute treatment with haloperidol can impair induction of LTP but not its maintenance in rabbit hippocampus. Chronic treatment with haloperidol suggested to enhance LTP in corticostriatal brain slices of rats[174,175]. | 1 Associated with sedation and often promote sleep inducing effects. |
| Haloperidol | 2 Can also block serotoninergic, histaminergic, cholinergic and noradrenergic receptors[173]. | 2 Reduce psychosis symptoms like hallucinations and delusion. | 3 Learning Functions: Haloperidol and Chlorpromazine can impair spatial learning in rats[176, 177]. | (1) Haloperidol, loxapine and mesoridazine increase REM sleep latency. Withdrawal of drug causes significant disruption of sleep with reduced REM and total sleep; | |||
| Loxapine | (2) No significant effect on SWS[178-180]; | ||||||
| Mesoridazine | (3) Chlorpromazine enhances SWS along with a dose dependent effect on REM sleep - lower doses increasing and higher doses reducing or having no effect on REM sleep[181]. | ||||||
| 2nd generation /atypical antipsychotic drugs | Clozapine | DA D2, 5-HT2A, 2C receptors | 1 Antagonistic effects by blocking 5-HT2A/2C receptors and agonistic effects by promoting 5-HT1A receptor mediated actions. | 1 In the treatment of psychotic disorders like schizophrenia, bipolar disorder, acute mania, dementia etc. | 1 Synaptic Plasticity: Without tetanization, acute clozapine administration induces LTP in rabbit hippocampus[184]. Chronic treatment with clozapine impairs LTP in rat prelimbic cortex[185]; Acute administration of risperidone impairs LTP in rabbit hippocampus. LTP induction post tetanization takes places in rat hippocampus upon chronic treatment with olanzapine[186,187]. | 1 Associated with an increase in the total amount of sleep but show an uneven trend in their stage specific effects. | |
| Risperidone | 2 Transiently block DA D2 receptors[182]. | 2 Reduce psychosis symptoms like hallucinations and delusion, disordered thinking, social withdrawal etc. | 2 Learning Functions: Clozapine and olanzapine impair memory retrieval in mice[188] while risperidone improves learning and memory processing in humans[189]. | (1) Clozapine increases REM sleep density and improves NREM sleep. However, its effect on SWS is inconsistent[180]; | |||
| Olanzapine | |||||||
| 3rd Generation antipsychotic drugs | Aripiprazole | DA D2 receptors | 1 Partial agonist of DA D2 and 5-HT1A receptors and an antagonist of 5-HT2A receptors. | (2) Olanzapine improves SWS as well as REM sleep latency. Risperidone increases SWS but reduces REM sleep[180,190,191]. | |||
| 2 DA D2 antagonist in the mesolimbic pathway[183]. | |||||||
| Antidepressants and anxiolytics | Fluoxetine | 5-HT-1A receptors | 1 Blocks the serotonin reuptake and increases serotonin action via 5-HT1A receptors. | In the treatment of depression, obsessive control disorders, eating disorders, substance use disorders etc. | 1 Synaptic Plasticity: (1) Acute treatment with fluoxetine before stressful stimuli indicated to suppress stress induced LTD in CA1 region of rat hippocampus[194]; | 1 Associated with uneven side effects on sleep like somnolence, sedation and insomnia. | |
| 2 Suggested to cause reversible internalization of 5-HT1A auto -receptors[192]. | (2) Chronic fluoxetine treatment enhances neurogenesis dependent LTP in mice hippocampal granule cells and also upregulates Brain-Derived Neurotrophic Factor-LTP associated genes. Acute treatment showed no such upregulation[195,196]; | (1) Fluoxetine reduces overall sleep continuity with a reduction in REM sleep but increases REM latency. It either increases or has no effect on SWS[197]; | |||||
| Mirtazapine | 5-HT1 and 2 receptors, α2-adrenergic receptors | 1 Blocks 5-HT2 receptors in order to increase 5-HT1 mediated transmission. | (3) Acute treatment of vortioxetine enhances LTP in the CA1 region of rat hippocampus by increasing pyramidal cell output[199];of memory processing[207,208]; | (2) Mirtazapine increases sleep continuity and SWS but does not affect REM sleep[197]; | |||
| 2 Antagonist of α2-adrenergic receptors to increase adrenergic neurotransmission[193]. | (4) Acute treatment of buspirone as an agonist of 5-HT1A results in the reversal of LTP in a time -dependent manner in rat hippocampal slices[202]; | (3) Vortioxetine causes a reduction in REM sleep but increases its latency. It’s effects on SWS are unclear[197]; | |||||
| Vortioxetine | 5-HT1A, 1B, 1D, 5-HT7 receptors. | Antagonist of 5-HT1D and H-HT7 receptors and a partial agonist of 5-HT1A and 1B receptors[198]. | In the treatment of Major Depressive Disorder. | (5) Acute treatment of baclofen induces GABA-B receptor mediated LTD enhancement of a glutamate-evoked current in the cerebellar cortical neurons. It also enhances mGluR1-coupled intracellular Ca2+ release[63]. | (4) Vilazodone causes sleep disturbances with a decrease in REM sleep and an increase in wakefulness and SWS[209]; | ||
| Vilazodone | 5-HT1A receptors | Blocks the serotonin reuptake inhibitor and also acts as a partial agonist of 5-HT1A receptor and stimulates it[200]. | 2 Learning Functions: (1) Fluoxetine reverses memory impairment in rats and enhances memory processing in mice[203,204]. Chronic use of mirtazapine and vortioxetine improve cognitive functions in humans[205,206,208,209]; | (5) Buspirone increases REM sleep density and latency[210]; | |||
| Buspirone | 5-HT1A, DA D2 auto receptors | Partial agonist of 5-HT1A receptor and antagonist of DA D2 auto receptors with low affinity[201]. | For treating of Generalized Anxiety Disorder and side effects of Parkinson’s and Alzheimer’s disease. | (2) Buspirone and baclofen are implicated in the impairment of memory processing[207,208]. | (6) Baclofen prolongs total sleep time with an increase in duration for REM sleep and SWS[211]. | ||
| Baclofen | GABA-B receptors | GABA-B agonist to increase inhibitory signals[172]. | Suggested use in treating depression and anxiety as well as a muscle relaxant. | ||||
The diverse nature of GPCRs, both in their structure and functions has made them front line players in understanding many cellular processes. With the advent of sleep being credited with a wide range of psychological and physiological functions, and with changes happening in GPCRs following sleep deprivation, it is important to understand how the receptors and the functions they regulate, like synaptic plasticity and memory consolidation, are affected. GPCRs are one of the largest groups in the mammalian CNS. In psychiatry, most currently used antidepressants and antip
Firstly, even though LTP and LTD have both been emphasized in manifesting the neurological changes during the state of sleep and wakefulness, more work is needed with regards to LTD. In an attempt to bridge that gap, a better understanding of the inhibitory GPCRs like GABA-B receptors is highly crucial. Additionally, GABA-B and mGluRs seem to have an associative mode of action and based on the studies that are reviewed so far; it appears that co-localization and heterodimerization of these two specific receptor types occurs with sleep deprivation[150]. Hence, the driving mole
Apart from memory consolidation and learning, GPCRs are also implicated in other psychoneurological occurrences. Around 60% of drugs target membrane proteins and 30% of them are GPCRs[219]. They are especially important as targets for antipsychotic and antidepressant drugs, considering some of their ligands like serotonin, DA and norepinephrine are associated with pathways that are important in psychiatry. Additionally, mGluRs and GABA-B Rs have an almost ubiquitous involvement in major cellular pathways which need to be better understood since they are affected in neurological disorders like Alzheimer’s disease, Parkinson’s disease, stress related disorders, etc.[220,221]. Changes in sleep patterns are often observed in many neurological disorders and considering these major GPCRs are altered during the state of sleep, an understanding of this correlation can affect the way we approach therapeutics in this area. For example, even though there was a temporary elevation observed in the expression of the serotonin 5-HT1A receptor upon sleep deprivation[155], it is universally correlated to disorders like depression, anxiety and psychosis and changes in its expression profile upon sleep deprivation does warrant for further studies on this GPCR and its other subtypes. Additionally, related GPCRs like the DA receptor D1 are also known to interact directly with LTP inducing NMDA receptors[118] which are important in many neurological disorders. Furthermore, since the D1 and D2 DA receptors are involved in the nigrostriatal and mesolimbic pathways, changes in their expression profiles affect movements and behavioural patterns that are also implicated in Parkinson’s disease and schizophrenia[222,223]. Therefore, DA GPCRs are highly potential drug targets and sleep associated changes in their expression profile needs more attention. Drugs used in psychiatry for such disorders, e.g., aripiprazole, are known to target 5-HTA and DA receptors[224] and if the expression profile of these receptors is changing upon sleep deprivation, could it also affect the action of such drugs? Disorders like schizophrenia and depression often encompass sleep deprivation and drugs used for these conditions which target such GPCRs, may get negatively affected if receptor plasticity (co-localization and/or heterodimerization with other receptor subunits, etc.) occurs with associated sleep disorders. For example, the antidepressant drug mirtazapine exhibits its action through GPCRs by blocking 5-HT2 and adrenergic α2 receptors, leading to an increase in the activation of 5-HT1A receptor mediated activity as well as NA release, respectively[193]. This drug is known to have sleep-promoting properties and has been associated with problems like day-time somnolence[225] potentially disrupting the normal sleep cycle. Interestingly, there are off-label therapies that employ antidepressants like mirtazapine in treating sleeping disorders due to its sedative effect[226]. Since antidepressants and antipsychotic drugs appear to directly or indirectly change activity at GPCRs, drug therapy for these disorders can affect sleep while sleep disturbances can also necessitate changes in that therapy. Therefore, sleep disturbance-induced plasticity and cross-talk between GPCRs can have consequences for drug therapy, the mechanisms for which need to be thoroughly examined. Lastly, allosteric modulation, which is another upcoming molecular interaction that is important for drug designing, can be applied to the homo and heterodimerization of GPCRs for various conditions. Hence, studies on structural, functional, receptor co-localization and remodelling, etc., will yield new insights into mechanisms involved and help improve therapeutics for a variety of CNS disorders, including in psychiatry.
Conclusion and future scope are included in the main text under heading "Intro
Provenance and peer review: Invited article; Externally peer reviewed
Corresponding Author's Membership in Professional Societies: Society for Neuroscience
Specialty type: Neurosciences
Country/Territory of origin: Canada
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Jarmasz JS S-Editor: Liu M L-Editor: A P-Editor: Yu HG
| 1. | Born J. Slow-wave sleep and the consolidation of long-term memory. World J Biol Psychiatry. 2010;11 Suppl 1:16-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 94] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 2. | Siegel JM. The REM sleep-memory consolidation hypothesis. Science. 2001;294:1058-1063. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 372] [Cited by in RCA: 315] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 3. | ASERINSKY E, KLEITMAN N. Regularly occurring periods of eye motility, and concomitant phenomena, during sleep. Science. 1953;118:273-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1552] [Cited by in RCA: 1142] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 4. | Siegel JM. Clues to the functions of mammalian sleep. Nature. 2005;437:1264-1271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 697] [Cited by in RCA: 586] [Article Influence: 29.3] [Reference Citation Analysis (0)] |
| 5. | Suchecki D, Tiba PA, Machado RB. REM Sleep Rebound as an Adaptive Response to Stressful Situations. Front Neurol. 2012;3:41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 74] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 6. | Borbély AA, Baumann F, Brandeis D, Strauch I, Lehmann D. Sleep deprivation: effect on sleep stages and EEG power density in man. Electroencephalogr Clin Neurophysiol. 1981;51:483-495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 720] [Cited by in RCA: 671] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 7. | Dijk DJ, Hayes B, Czeisler CA. Dynamics of electroencephalographic sleep spindles and slow wave activity in men: effect of sleep deprivation. Brain Res. 1993;626:190-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 223] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 8. | Finelli LA, Borbély AA, Achermann P. Functional topography of the human nonREM sleep electroencephalogram. Eur J Neurosci. 2001;13:2282-2290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 228] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 9. | Knoblauch V, Martens WL, Wirz-Justice A, Cajochen C. Human sleep spindle characteristics after sleep deprivation. Clin Neurophysiol. 2003;114:2258-2267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 61] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 10. | Borbély AA, Tobler I, Hanagasioglu M. Effect of sleep deprivation on sleep and EEG power spectra in the rat. Behav Brain Res. 1984;14:171-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 230] [Cited by in RCA: 237] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 11. | Vyazovskiy VV, Tobler I. Theta activity in the waking EEG is a marker of sleep propensity in the rat. Brain Res. 2005;1050:64-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 137] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 12. | Johnson DA, Billings ME, Hale L. Environmental Determinants of Insufficient Sleep and Sleep Disorders: Implications for Population Health. Curr Epidemiol Rep. 2018;5:61-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 164] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 13. | Extent and Health Consequences of Chronic Sleep Loss and Sleep Disorders. In: Sleep Disorders and Sleep Deprivation: An Unmet Public Health Problem. Colten HR, Altevogt BM, editor. Washington (DC): National Academies Press (US), 2006. |
| 14. | Winsky-Sommerer R, de Oliveira P, Loomis S, Wafford K, Dijk DJ, Gilmour G. Disturbances of sleep quality, timing and structure and their relationship with other neuropsychiatric symptoms in Alzheimer's disease and schizophrenia: Insights from studies in patient populations and animal models. Neurosci Biobehav Rev. 2019;97:112-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 15. | Besedovsky L, Lange T, Born J. Sleep and immune function. Pflugers Arch. 2012;463:121-137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 471] [Cited by in RCA: 597] [Article Influence: 42.6] [Reference Citation Analysis (0)] |
| 16. | Akerstedt T, Wright KP Jr. Sleep Loss and Fatigue in Shift Work and Shift Work Disorder. Sleep Med Clin. 2009;4:257-271. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 446] [Cited by in RCA: 361] [Article Influence: 22.6] [Reference Citation Analysis (0)] |
| 17. | Rouch I, Wild P, Ansiau D, Marquié JC. Shiftwork experience, age and cognitive performance. Ergonomics. 2005;48:1282-1293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 126] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 18. | Smith C, Smith D. Ingestion of ethanol just prior to sleep onset impairs memory for procedural but not declarative tasks. Sleep. 2003;26:185-191. [PubMed] |
| 19. | Roehrs TA, Roth T. Sleep Disturbance in Substance Use Disorders. Psychiatr Clin North Am. 2015;38:793-803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 65] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 20. | Ly S, Pack AI, Naidoo N. The neurobiological basis of sleep: Insights from Drosophila. Neurosci Biobehav Rev. 2018;87:67-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 21. | Wang L, Gu Y, Zhang J, Gong L. Effects of Sleep Deprivation (SD) on Rats via ERK1/2 Signaling Pathway. Med Sci Monit. 2019;25:2886-2895. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 22. | Peever J, Fuller PM. Neuroscience: A Distributed Neural Network Controls REM Sleep. Curr Biol. 2016;26:R34-R35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 36] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 23. | Rasch B, Born J. About sleep's role in memory. Physiol Rev. 2013;93:681-766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1723] [Cited by in RCA: 1725] [Article Influence: 143.8] [Reference Citation Analysis (0)] |
| 24. | Groeger JA, Stanley N, Deacon S, Dijk DJ. Dissociating effects of global SWS disruption and healthy aging on waking performance and daytime sleepiness. Sleep. 2014;37:1127-1142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 25. | Squire LR. Mechanisms of memory. Science. 1986;232:1612-1619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 790] [Cited by in RCA: 671] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 26. | Stickgold R, Walker MP. Memory consolidation and reconsolidation: what is the role of sleep? Trends Neurosci. 2005;28:408-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 225] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 27. | Amzica F, Neckelmann D, Steriade M. Instrumental conditioning of fast (20- to 50-Hz) oscillations in corticothalamic networks. Proc Natl Acad Sci U S A. 1997;94:1985-1989. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 25] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 28. | Steriade M. Grouping of brain rhythms in corticothalamic systems. Neuroscience. 2006;137:1087-1106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 812] [Cited by in RCA: 878] [Article Influence: 46.2] [Reference Citation Analysis (0)] |
| 29. | Muller GE, Pilzecker A. Experimental contributions to the theory of memory. Z Psychol Z Angew Psychol. 1900;1:1-300. |
| 30. | McGaugh JL. Memory--a century of consolidation. Science. 2000;287:248-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2830] [Cited by in RCA: 2733] [Article Influence: 109.3] [Reference Citation Analysis (0)] |
| 31. | Maquet P. The role of sleep in learning and memory. Science. 2001;294:1048-1052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 677] [Cited by in RCA: 623] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 32. | Plihal W, Born J. Effects of early and late nocturnal sleep on priming and spatial memory. Psychophysiology. 1999;36:571-582. [PubMed] |
| 33. | Smith C. Sleep states and memory processes in humans: procedural vs declarative memory systems. Sleep Med Rev. 2001;5:491-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 210] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 34. | Diekelmann S, Born J. The memory function of sleep. Nat Rev Neurosci. 2010;11:114-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2092] [Cited by in RCA: 2379] [Article Influence: 158.6] [Reference Citation Analysis (0)] |
| 35. | McClelland JL, McNaughton BL, O'Reilly RC. Why there are complementary learning systems in the hippocampus and neocortex: insights from the successes and failures of connectionist models of learning and memory. Psychol Rev. 1995;102:419-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2911] [Cited by in RCA: 2730] [Article Influence: 91.0] [Reference Citation Analysis (0)] |
| 36. | Walker MP. The role of slow wave sleep in memory processing. J Clin Sleep Med. 2009;5:S20-S26. [PubMed] |
| 37. | Stickgold R, James L, Hobson JA. Visual discrimination learning requires sleep after training. Nat Neurosci. 2000;3:1237-1238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 458] [Cited by in RCA: 426] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 38. | Walker MP, Brakefield T, Morgan A, Hobson JA, Stickgold R. Practice with sleep makes perfect: sleep-dependent motor skill learning. Neuron. 2002;35:205-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 879] [Cited by in RCA: 852] [Article Influence: 37.0] [Reference Citation Analysis (0)] |
| 39. | Walker MP, Brakefield T, Seidman J, Morgan A, Hobson JA, Stickgold R. Sleep and the time course of motor skill learning. Learn Mem. 2003;10:275-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 304] [Cited by in RCA: 290] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 40. | Buzsáki G. Hippocampal sharp waves: their origin and significance. Brain Res. 1986;398:242-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 582] [Cited by in RCA: 616] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 41. | Nakazawa K, Quirk MC, Chitwood RA, Watanabe M, Yeckel MF, Sun LD, Kato A, Carr CA, Johnston D, Wilson MA, Tonegawa S. Requirement for hippocampal CA3 NMDA receptors in associative memory recall. Science. 2002;297:211-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 804] [Cited by in RCA: 787] [Article Influence: 34.2] [Reference Citation Analysis (0)] |
| 42. | Buzsáki G. Two-stage model of memory trace formation: a role for "noisy" brain states. Neuroscience. 1989;31:551-570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1112] [Cited by in RCA: 1162] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 43. | Siapas AG, Wilson MA. Coordinated interactions between hippocampal ripples and cortical spindles during slow-wave sleep. Neuron. 1998;21:1123-1128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 654] [Cited by in RCA: 685] [Article Influence: 25.4] [Reference Citation Analysis (0)] |
| 44. | Sirota A, Csicsvari J, Buhl D, Buzsáki G. Communication between neocortex and hippocampus during sleep in rodents. Proc Natl Acad Sci U S A. 2003;100:2065-2069. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 657] [Cited by in RCA: 644] [Article Influence: 29.3] [Reference Citation Analysis (0)] |
| 45. | Wierzynski CM, Lubenov EV, Gu M, Siapas AG. State-dependent spike-timing relationships between hippocampal and prefrontal circuits during sleep. Neuron. 2009;61:587-596. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 255] [Cited by in RCA: 221] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 46. | Hebbian DO. The organization of behavior: A neuropsychological theory. New York: John Wiley and Sons, 1949. |
| 47. | Bliss TV, Lomo T. Long-lasting potentiation of synaptic transmission in the dentate area of the anaesthetized rabbit following stimulation of the perforant path. J Physiol. 1973;232:331-356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4427] [Cited by in RCA: 4276] [Article Influence: 82.2] [Reference Citation Analysis (0)] |
| 48. | Kelso SR, Ganong AH, Brown TH. Hebbian synapses in hippocampus. Proc Natl Acad Sci U S A. 1986;83:5326-5330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 307] [Cited by in RCA: 254] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 49. | Sastry BR, Goh JW, Auyeung A. Associative induction of posttetanic and long-term potentiation in CA1 neurons of rat hippocampus. Science. 1986;232:988-990. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 135] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 50. | Wigström H, Gustafsson B. Postsynaptic control of hippocampal long-term potentiation. J Physiol (Paris). 1986;81:228-236. [PubMed] |
| 51. | Lüscher C, Malenka RC. NMDA receptor-dependent long-term potentiation and long-term depression (LTP/LTD). Cold Spring Harb Perspect Biol. 2012;4:a005710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 535] [Cited by in RCA: 723] [Article Influence: 55.6] [Reference Citation Analysis (0)] |
| 52. | Bear MF, Malenka RC. Synaptic plasticity: LTP and LTD. Curr Opin Neurobiol. 1994;4:389-399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 922] [Cited by in RCA: 934] [Article Influence: 30.1] [Reference Citation Analysis (0)] |
| 53. | Lee HK, Barbarosie M, Kameyama K, Bear MF, Huganir RL. Regulation of distinct AMPA receptor phosphorylation sites during bidirectional synaptic plasticity. Nature. 2000;405:955-959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 781] [Cited by in RCA: 846] [Article Influence: 33.8] [Reference Citation Analysis (0)] |
| 54. | Fagni L, Chavis P, Ango F, Bockaert J. Complex interactions between mGluRs, intracellular Ca2+ stores and ion channels in neurons. Trends Neurosci. 2000;23:80-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 214] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 55. | Lujan R, Nusser Z, Roberts JD, Shigemoto R, Somogyi P. Perisynaptic location of metabotropic glutamate receptors mGluR1 and mGluR5 on dendrites and dendritic spines in the rat hippocampus. Eur J Neurosci. 1996;8:1488-1500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 610] [Cited by in RCA: 687] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 56. | Abraham WC, Williams JM. Properties and mechanisms of LTP maintenance. Neuroscientist. 2003;9:463-474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 118] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 57. | Benington JH, Frank MG. Cellular and molecular connections between sleep and synaptic plasticity. Prog Neurobiol. 2003;69:71-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 141] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 58. | Bliss TV, Collingridge GL. A synaptic model of memory: long-term potentiation in the hippocampus. Nature. 1993;361:31-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8184] [Cited by in RCA: 8455] [Article Influence: 264.2] [Reference Citation Analysis (0)] |
| 59. | Dayan P, Willshaw DJ. Optimising synaptic learning rules in linear associative memories. Biol Cybern. 1991;65:253-265. [PubMed] [DOI] [Full Text] |
| 60. | Iriki A, Pavlides C, Keller A, Asanuma H. Long-term potentiation in the motor cortex. Science. 1989;245:1385-1387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 209] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 61. | Whitlock JR, Heynen AJ, Shuler MG, Bear MF. Learning induces long-term potentiation in the hippocampus. Science. 2006;313:1093-1097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1269] [Cited by in RCA: 1370] [Article Influence: 72.1] [Reference Citation Analysis (0)] |
| 62. | Gladding CM, Fitzjohn SM, Molnár E. Metabotropic glutamate receptor-mediated long-term depression: molecular mechanisms. Pharmacol Rev. 2009;61:395-412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 174] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 63. | Kamikubo Y, Tabata T, Kakizawa S, Kawakami D, Watanabe M, Ogura A, Iino M, Kano M. Postsynaptic GABAB receptor signalling enhances LTD in mouse cerebellar Purkinje cells. J Physiol. 2007;585:549-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 45] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 64. | Tononi G, Cirelli C. Sleep and synaptic homeostasis: a hypothesis. Brain Res Bull. 2003;62:143-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 724] [Cited by in RCA: 753] [Article Influence: 35.9] [Reference Citation Analysis (0)] |
| 65. | Tsumoto T. Long-term depression in cerebral cortex: a possible substrate of "forgetting" that should not be forgotten. Neurosci Res. 1993;16:263-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 52] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 66. | Manahan-Vaughan D, Braunewell KH. Novelty acquisition is associated with induction of hippocampal long-term depression. Proc Natl Acad Sci U S A. 1999;96:8739-8744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 269] [Cited by in RCA: 294] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 67. | Stent GS. A physiological mechanism for Hebb's postulate of learning. Proc Natl Acad Sci U S A. 1973;70:997-1001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 423] [Cited by in RCA: 378] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 68. | Lynch GS, Dunwiddie T, Gribkoff V. Heterosynaptic depression: a postsynaptic correlate of long-term potentiation. Nature. 1977;266:737-739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 411] [Cited by in RCA: 393] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 69. | Barnes CA, McNaughton BL. An age comparison of the rates of acquisition and forgetting of spatial information in relation to long-term enhancement of hippocampal synapses. Behav Neurosci. 1985;99:1040-1048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 28] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 70. | O’Keefe J, Nadel L. The hippocampus as a cognitive map. Oxford: Oxford University Press, 1978. |
| 71. | Foster TC, Kumar A. Susceptibility to induction of long-term depression is associated with impaired memory in aged Fischer 344 rats. Neurobiol Learn Mem. 2007;87:522-535. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 85] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 72. | Norris CM, Korol DL, Foster TC. Increased susceptibility to induction of long-term depression and long-term potentiation reversal during aging. J Neurosci. 1996;16:5382-5392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 211] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 73. | McNaughton BL, Barnes CA, Rao G, Baldwin J, Rasmussen M. Long-term enhancement of hippocampal synaptic transmission and the acquisition of spatial information. J Neurosci. 1986;6:563-571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 222] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 74. | Lynch G, Baudry M. The biochemistry of memory: a new and specific hypothesis. Science. 1984;224:1057-1063. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 838] [Cited by in RCA: 773] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 75. | Abraham WC. How long will long-term potentiation last? Philos Trans R Soc Lond B Biol Sci. 2003;358:735-744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 175] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 76. | Larson J, Lynch G. Induction of synaptic potentiation in hippocampus by patterned stimulation involves two events. Science. 1986;232:985-988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 410] [Cited by in RCA: 410] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 77. | Staubli U, Lynch G. Stable hippocampal long-term potentiation elicited by 'theta' pattern stimulation. Brain Res. 1987;435:227-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 255] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 78. | Levy WB, Steward O. Synapses as associative memory elements in the hippocampal formation. Brain Res. 1979;175:233-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 403] [Cited by in RCA: 362] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 79. | Cull-Candy S, Kelly L, Farrant M. Regulation of Ca2+-permeable AMPA receptors: synaptic plasticity and beyond. Curr Opin Neurobiol. 2006;16:288-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 346] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 80. | Harney SC, Rowan M, Anwyl R. Long-term depression of NMDA receptor-mediated synaptic transmission is dependent on activation of metabotropic glutamate receptors and is altered to long-term potentiation by low intracellular calcium buffering. J Neurosci. 2006;26:1128-1132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 79] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 81. | Collingridge GL, Peineau S, Howland JG, Wang YT. Long-term depression in the CNS. Nat Rev Neurosci. 2010;11:459-473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 767] [Cited by in RCA: 705] [Article Influence: 47.0] [Reference Citation Analysis (0)] |
| 82. | Lau CG, Takeuchi K, Rodenas-Ruano A, Takayasu Y, Murphy J, Bennett MV, Zukin RS. Regulation of NMDA receptor Ca2+ signalling and synaptic plasticity. Biochem Soc Trans. 2009;37:1369-1374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 95] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 83. | Dingledine R, Borges K, Bowie D, Traynelis SF. The glutamate receptor ion channels. Pharmacol Rev. 1999;51:7-61. [PubMed] |
| 84. | Bortolotto ZA, Collingridge GL. Characterisation of LTP induced by the activation of glutamate metabotropic receptors in area CA1 of the hippocampus. Neuropharmacology. 1993;32:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 190] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 85. | Anwyl R. Modulation of vertebrate neuronal calcium channels by transmitters. Brain Res Brain Res Rev. 1991;16:265-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 128] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 86. | Kemp A, Manahan-Vaughan D. Hippocampal long-term depression: master or minion in declarative memory processes? Trends Neurosci. 2007;30:111-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 231] [Cited by in RCA: 259] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 87. | Bailey CH, Kandel ER. Structural changes accompanying memory storage. Annu Rev Physiol. 1993;55:397-426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 581] [Cited by in RCA: 595] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 88. | Smith C, Wong PT. Paradoxical sleep increases predict successful learning in a complex operant task. Behav Neurosci. 1991;105:282-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 45] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 89. | Buzsáki G. Memory consolidation during sleep: a neurophysiological perspective. J Sleep Res. 1998;7 Suppl 1:17-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 349] [Cited by in RCA: 350] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 90. | Marks GA, Shaffery JP, Oksenberg A, Speciale SG, Roffwarg HP. A functional role for REM sleep in brain maturation. Behav Brain Res. 1995;69:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 165] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 91. | Smith C, Lapp L. Increases in number of REMS and REM density in humans following an intensive learning period. Sleep. 1991;14:325-330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 98] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 92. | Campbell IG, Guinan MJ, Horowitz JM. Sleep deprivation impairs long-term potentiation in rat hippocampal slices. J Neurophysiol. 2002;88:1073-1076. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 130] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 93. | McDermott CM, LaHoste GJ, Chen C, Musto A, Bazan NG, Magee JC. Sleep deprivation causes behavioral, synaptic, and membrane excitability alterations in hippocampal neurons. J Neurosci. 2003;23:9687-9695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 289] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 94. | Tartar JL, Ward CP, McKenna JT, Thakkar M, Arrigoni E, McCarley RW, Brown RE, Strecker RE. Hippocampal synaptic plasticity and spatial learning are impaired in a rat model of sleep fragmentation. Eur J Neurosci. 2006;23:2739-2748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 157] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 95. | Davis CJ, Meighan PC, Taishi P, Krueger JM, Harding JW, Wright JW. REM sleep deprivation attenuates actin-binding protein cortactin: a link between sleep and hippocampal plasticity. Neurosci Lett. 2006;400:191-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 96. | Tadavarty R, Kaan TK, Sastry BR. Long-term depression of excitatory synaptic transmission in rat hippocampal CA1 neurons following sleep-deprivation. Exp Neurol. 2009;216:239-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 97. | Hasselmo ME. Neuromodulation: acetylcholine and memory consolidation. Trends Cogn Sci. 1999;3:351-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 527] [Cited by in RCA: 535] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 98. | Bosboom JL, Wolters ECh. Psychotic symptoms in Parkinson's disease: pathophysiology and management. Expert Opin Drug Saf. 2004;3:209-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 99. | Dale E, Pehrson AL, Jeyarajah T, Li Y, Leiser SC, Smagin G, Olsen CK, Sanchez C. Effects of serotonin in the hippocampus: how SSRIs and multimodal antidepressants might regulate pyramidal cell function. CNS Spectr. 2016;21:143-161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 106] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 100. | Geyer MA, Vollenweider FX. Serotonin research: contributions to understanding psychoses. Trends Pharmacol Sci. 2008;29:445-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 285] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 101. | Steele PM, Mauk MD. Inhibitory control of LTP and LTD: stability of synapse strength. J Neurophysiol. 1999;81:1559-1566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 95] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 102. | Stelzer A. GABAA receptors control the excitability of neuronal populations. Int Rev Neurobiol. 1992;33:195-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 103. | Wagner JJ, Alger BE. GABAergic and developmental influences on homosynaptic LTD and depotentiation in rat hippocampus. J Neurosci. 1995;15:1577-1586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 185] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 104. | Blackwell KT, Jedrzejewska-Szmek J. Molecular mechanisms underlying neuronal synaptic plasticity: systems biology meets computational neuroscience in the wilds of synaptic plasticity. Wiley Interdiscip Rev Syst Biol Med. 2013;5:717-731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 105. | Scharf MT, Woo NH, Lattal KM, Young JZ, Nguyen PV, Abel T. Protein synthesis is required for the enhancement of long-term potentiation and long-term memory by spaced training. J Neurophysiol. 2002;87:2770-2777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 149] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 106. | Guzowski JF, Lyford GL, Stevenson GD, Houston FP, McGaugh JL, Worley PF, Barnes CA. Inhibition of activity-dependent arc protein expression in the rat hippocampus impairs the maintenance of long-term potentiation and the consolidation of long-term memory. J Neurosci. 2000;20:3993-4001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 731] [Cited by in RCA: 734] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 107. | Lisman J, Yasuda R, Raghavachari S. Mechanisms of CaMKII action in long-term potentiation. Nat Rev Neurosci. 2012;13:169-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 687] [Cited by in RCA: 843] [Article Influence: 64.8] [Reference Citation Analysis (0)] |
| 108. | Mei F, Nagappan G, Ke Y, Sacktor TC, Lu B. BDNF facilitates L-LTP maintenance in the absence of protein synthesis through PKMζ. PLoS One. 2011;6:e21568. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 63] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 109. | Pastalkova E, Serrano P, Pinkhasova D, Wallace E, Fenton AA, Sacktor TC. Storage of spatial information by the maintenance mechanism of LTP. Science. 2006;313:1141-1144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 595] [Cited by in RCA: 623] [Article Influence: 32.8] [Reference Citation Analysis (0)] |
| 110. | Garthwaite J, Boulton CL. Nitric oxide signaling in the central nervous system. Annu Rev Physiol. 1995;57:683-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1244] [Cited by in RCA: 1241] [Article Influence: 41.4] [Reference Citation Analysis (0)] |
| 111. | Faraci FM, Brian JE Jr. Nitric oxide and the cerebral circulation. Stroke. 1994;25:692-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 384] [Cited by in RCA: 353] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 112. | Burgner D, Rockett K, Kwiatkowski D. Nitric oxide and infectious diseases. Arch Dis Child. 1999;81:185-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 58] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 113. | Cirelli C, Tononi G. The search for the molecular correlates of sleep and wakefulness. Sleep Med Rev. 2001;5:397-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 114. | Cirelli C, Gutierrez CM, Tononi G. Extensive and divergent effects of sleep and wakefulness on brain gene expression. Neuron. 2004;41:35-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 445] [Cited by in RCA: 455] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 115. | Regard JB, Sato IT, Coughlin SR. Anatomical profiling of G protein-coupled receptor expression. Cell. 2008;135:561-571. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 568] [Cited by in RCA: 534] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 116. | Gerber KJ, Squires KE, Hepler JR. Roles for Regulator of G Protein Signaling Proteins in Synaptic Signaling and Plasticity. Mol Pharmacol. 2016;89:273-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 91] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 117. | Leung CCY, Wong YH. Role of G Protein-Coupled Receptors in the Regulation of Structural Plasticity and Cognitive Function. Molecules. 2017;22:1239. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 35] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 118. | Lee FJ, Liu F. Direct interactions between NMDA and D1 receptors: a tale of tails. Biochem Soc Trans. 2004;32:1032-1036. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 119. | Jong YI, Harmon SK, O'Malley KL. Intracellular GPCRs Play Key Roles in Synaptic Plasticity. ACS Chem Neurosci. 2018;9:2162-2172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 120. | Schoepp DD, Jane DE, Monn JA. Pharmacological agents acting at subtypes of metabotropic glutamate receptors. Neuropharmacology. 1999;38:1431-1476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 821] [Cited by in RCA: 838] [Article Influence: 32.2] [Reference Citation Analysis (0)] |
| 121. | Guérineau NC, Gähwiler BH, Gerber U. Reduction of resting K+ current by metabotropic glutamate and muscarinic receptors in rat CA3 cells: mediation by G-proteins. J Physiol. 1994;474:27-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 119] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 122. | Swartz KJ, Bean BP. Inhibition of calcium channels in rat CA3 pyramidal neurons by a metabotropic glutamate receptor. J Neurosci. 1992;12:4358-4371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 121] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 123. | Schoepp DD, Conn PJ. Metabotropic glutamate receptors in brain function and pathology. Trends Pharmacol Sci. 1993;14:13-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 564] [Cited by in RCA: 566] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 124. | Shigemoto R, Kinoshita A, Wada E, Nomura S, Ohishi H, Takada M, Flor PJ, Neki A, Abe T, Nakanishi S, Mizuno N. Differential presynaptic localization of metabotropic glutamate receptor subtypes in the rat hippocampus. J Neurosci. 1997;17:7503-7522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 782] [Cited by in RCA: 809] [Article Influence: 28.9] [Reference Citation Analysis (0)] |
| 125. | Jones KA, Borowsky B, Tamm JA, Craig DA, Durkin MM, Dai M, Yao WJ, Johnson M, Gunwaldsen C, Huang LY, Tang C, Shen Q, Salon JA, Morse K, Laz T, Smith KE, Nagarathnam D, Noble SA, Branchek TA, Gerald C. GABA(B) receptors function as a heteromeric assembly of the subunits GABA(B)R1 and GABA(B)R2. Nature. 1998;396:674-679. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 817] [Cited by in RCA: 786] [Article Influence: 29.1] [Reference Citation Analysis (0)] |
| 126. | Kaupmann K, Malitschek B, Schuler V, Heid J, Froestl W, Beck P, Mosbacher J, Bischoff S, Kulik A, Shigemoto R, Karschin A, Bettler B. GABA(B)-receptor subtypes assemble into functional heteromeric complexes. Nature. 1998;396:683-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 905] [Cited by in RCA: 868] [Article Influence: 32.1] [Reference Citation Analysis (0)] |
| 127. | White JH, Wise A, Main MJ, Green A, Fraser NJ, Disney GH, Barnes AA, Emson P, Foord SM, Marshall FH. Heterodimerization is required for the formation of a functional GABA(B) receptor. Nature. 1998;396:679-682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 913] [Cited by in RCA: 863] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 128. | Kulik A, Vida I, Luján R, Haas CA, López-Bendito G, Shigemoto R, Frotscher M. Subcellular localization of metabotropic GABA(B) receptor subunits GABA(B1a/b) and GABA(B2) in the rat hippocampus. J Neurosci. 2003;23:11026-11035. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 163] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 129. | Knight AR, Bowery NG. The pharmacology of adenylyl cyclase modulation by GABAB receptors in rat brain slices. Neuropharmacology. 1996;35:703-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 61] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 130. | Mintz IM, Bean BP. GABAB receptor inhibition of P-type Ca2+ channels in central neurons. Neuron. 1993;10:889-898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 209] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 131. | Ladera C, del Carmen Godino M, José Cabañero M, Torres M, Watanabe M, Luján R, Sánchez-Prieto J. Pre-synaptic GABA receptors inhibit glutamate release through GIRK channels in rat cerebral cortex. J Neurochem. 2008;107:1506-1517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 65] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 132. | Hoyer D, Martin G. 5-HT receptor classification and nomenclature: towards a harmonization with the human genome. Neuropharmacology. 1997;36:419-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 298] [Cited by in RCA: 259] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 133. | McCorvy JD, Roth BL. Structure and function of serotonin G protein-coupled receptors. Pharmacol Ther. 2015;150:129-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 250] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 134. | Altieri SC, Garcia-Garcia AL, Leonardo ED, Andrews AM. Rethinking 5-HT1A receptors: emerging modes of inhibitory feedback of relevance to emotion-related behavior. ACS Chem Neurosci. 2013;4:72-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 79] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 135. | Herrick-Davis K. Functional significance of serotonin receptor dimerization. Exp Brain Res. 2013;230:375-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 44] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 136. | Pierce KL, Premont RT, Lefkowitz RJ. Seven-transmembrane receptors. Nat Rev Mol Cell Biol. 2002;3:639-650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2002] [Cited by in RCA: 2021] [Article Influence: 87.9] [Reference Citation Analysis (0)] |
| 137. | Levey AI, Hersch SM, Rye DB, Sunahara RK, Niznik HB, Kitt CA, Price DL, Maggio R, Brann MR, Ciliax BJ. Localization of D1 and D2 dopamine receptors in brain with subtype-specific antibodies. Proc Natl Acad Sci U S A. 1993;90:8861-8865. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 421] [Cited by in RCA: 470] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 138. | Hersch SM, Ciliax BJ, Gutekunst CA, Rees HD, Heilman CJ, Yung KK, Bolam JP, Ince E, Yi H, Levey AI. Electron microscopic analysis of D1 and D2 dopamine receptor proteins in the dorsal striatum and their synaptic relationships with motor corticostriatal afferents. J Neurosci. 1995;15:5222-5237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 344] [Cited by in RCA: 333] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 139. | Baik JH. Dopamine signaling in reward-related behaviors. Front Neural Circuits. 2013;7:152. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 235] [Cited by in RCA: 338] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 140. | Ridray S, Griffon N, Mignon V, Souil E, Carboni S, Diaz J, Schwartz JC, Sokoloff P. Coexpression of dopamine D1 and D3 receptors in islands of Calleja and shell of nucleus accumbens of the rat: opposite and synergistic functional interactions. Eur J Neurosci. 1998;10:1676-1686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 127] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 141. | Strosberg AD. Structure, function, and regulation of adrenergic receptors. Protein Sci. 1993;2:1198-1209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 165] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 142. | Mitrano DA, Pare JF, Smith Y, Weinshenker D. D1-dopamine and α1-adrenergic receptors co-localize in dendrites of the rat prefrontal cortex. Neuroscience. 2014;258:90-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 143. | Lüscher C, Huber KM. Group 1 mGluR-dependent synaptic long-term depression: mechanisms and implications for circuitry and disease. Neuron. 2010;65:445-459. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 505] [Cited by in RCA: 473] [Article Influence: 31.5] [Reference Citation Analysis (0)] |
| 144. | Bolshakov VY, Siegelbaum SA. Postsynaptic induction and presynaptic expression of hippocampal long-term depression. Science. 1994;264:1148-1152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 326] [Cited by in RCA: 386] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 145. | Wang H, Ardiles AO, Yang S, Tran T, Posada-Duque R, Valdivia G, Baek M, Chuang YA, Palacios AG, Gallagher M, Worley P, Kirkwood A. Metabotropic Glutamate Receptors Induce a Form of LTP Controlled by Translation and Arc Signaling in the Hippocampus. J Neurosci. 2016;36:1723-1729. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 56] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 146. | Davies CH, Starkey SJ, Pozza MF, Collingridge GL. GABA autoreceptors regulate the induction of LTP. Nature. 1991;349:609-611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 415] [Cited by in RCA: 442] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 147. | Couve A, Filippov AK, Connolly CN, Bettler B, Brown DA, Moss SJ. Intracellular retention of recombinant GABAB receptors. J Biol Chem. 1998;273:26361-26367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 155] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 148. | Galvez T, Duthey B, Kniazeff J, Blahos J, Rovelli G, Bettler B, Prézeau L, Pin JP. Allosteric interactions between GB1 and GB2 subunits are required for optimal GABA(B) receptor function. EMBO J. 2001;20:2152-2159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 297] [Cited by in RCA: 286] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 149. | Gassmann M, Shaban H, Vigot R, Sansig G, Haller C, Barbieri S, Humeau Y, Schuler V, Müller M, Kinzel B, Klebs K, Schmutz M, Froestl W, Heid J, Kelly PH, Gentry C, Jaton AL, Van der Putten H, Mombereau C, Lecourtier L, Mosbacher J, Cryan JF, Fritschy JM, Lüthi A, Kaupmann K, Bettler B. Redistribution of GABAB(1) protein and atypical GABAB responses in GABAB(2)-deficient mice. J Neurosci. 2004;24:6086-6097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 191] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 150. | Tadavarty R, Rajput PS, Wong JM, Kumar U, Sastry BR. Sleep-deprivation induces changes in GABA(B) and mGlu receptor expression and has consequences for synaptic long-term depression. PLoS One. 2011;6:e24933. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 38] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 151. | Yuen EY, Jiang Q, Chen P, Feng J, Yan Z. Activation of 5-HT2A/C receptors counteracts 5-HT1A regulation of n-methyl-D-aspartate receptor channels in pyramidal neurons of prefrontal cortex. J Biol Chem. 2008;283:17194-17204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 105] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 152. | Zhong P, Yuen EY, Yan Z. Modulation of neuronal excitability by serotonin-NMDA interactions in prefrontal cortex. Mol Cell Neurosci. 2008;38:290-299. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 57] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 153. | Pineda JC, Góngora-Alfaro JL. Cross-Modulation Between GABAB and 5-HT Receptors: A Link Between Anxiety and Depression? In: Monoaminergic Modulation of Cortical Excitability. Tseng KY, Atzori M, editor. Boston (MA): Springer US, 2007: 103–111. |
| 154. | Monti JM. Serotonin control of sleep-wake behavior. Sleep Med Rev. 2011;15:269-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 310] [Cited by in RCA: 386] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 155. | Azizi H, Hwang J, Suen V, Kang NZ, Somvanshi R, Tadavarty R, Kumar U, Sastry BR. Sleep deprivation induces changes in 5-HT actions and 5-HT1A receptor expression in the rat hippocampus. Neurosci Lett. 2017;655:151-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 156. | González-Maeso J, Ang RL, Yuen T, Chan P, Weisstaub NV, López-Giménez JF, Zhou M, Okawa Y, Callado LF, Milligan G, Gingrich JA, Filizola M, Meana JJ, Sealfon SC. Identification of a serotonin/glutamate receptor complex implicated in psychosis. Nature. 2008;452:93-97. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 677] [Cited by in RCA: 623] [Article Influence: 36.6] [Reference Citation Analysis (0)] |
| 157. | Otani S, Daniel H, Roisin MP, Crepel F. Dopaminergic modulation of long-term synaptic plasticity in rat prefrontal neurons. Cereb Cortex. 2003;13:1251-1256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 129] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 158. | Palacios-Filardo J, Mellor JR. Neuromodulation of hippocampal long-term synaptic plasticity. Curr Opin Neurobiol. 2019;54:37-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 111] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 159. | Lemon N, Manahan-Vaughan D. Dopamine D1/D5 receptors gate the acquisition of novel information through hippocampal long-term potentiation and long-term depression. J Neurosci. 2006;26:7723-7729. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 268] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 160. | Laing M, Bashir ZI. β-Adrenoceptors and synaptic plasticity in the perirhinal cortex. Neuroscience. 2014;273:163-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 161. | Lim MM, Xu J, Holtzman DM, Mach RH. Sleep deprivation differentially affects dopamine receptor subtypes in mouse striatum. Neuroreport. 2011;22:489-493. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 162. | Kim Y, Elmenhorst D, Weisshaupt A, Wedekind F, Kroll T, McCarley RW, Strecker RE, Bauer A. Chronic sleep restriction induces long-lasting changes in adenosine and noradrenaline receptor density in the rat brain. J Sleep Res. 2015;24:549-558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 163. | Kammermeier PJ. Functional and pharmacological characteristics of metabotropic glutamate receptors 2/4 heterodimers. Mol Pharmacol. 2012;82:438-447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 62] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 164. | Yin S, Noetzel MJ, Johnson KA, Zamorano R, Jalan-Sakrikar N, Gregory KJ, Conn PJ, Niswender CM. Selective actions of novel allosteric modulators reveal functional heteromers of metabotropic glutamate receptors in the CNS. J Neurosci. 2014;34:79-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 101] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 165. | Ayala JE, Chen Y, Banko JL, Sheffler DJ, Williams R, Telk AN, Watson NL, Xiang Z, Zhang Y, Jones PJ, Lindsley CW, Olive MF, Conn PJ. mGluR5 positive allosteric modulators facilitate both hippocampal LTP and LTD and enhance spatial learning. Neuropsychopharmacology. 2009;34:2057-2071. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 181] [Cited by in RCA: 181] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 166. | Mauri MC, Paletta S, Maffini M, Colasanti A, Dragogna F, Di Pace C, Altamura AC. Clinical pharmacology of atypical antipsychotics: an update. EXCLI J. 2014;13:1163-1191. [PubMed] |
| 167. | Meltzer HY. The mechanism of action of novel antipsychotic drugs. Schizophr Bull. 1991;17:263-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 292] [Cited by in RCA: 255] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 168. | Weiner DM, Burstein ES, Nash N, Croston GE, Currier EA, Vanover KE, Harvey SC, Donohue E, Hansen HC, Andersson CM, Spalding TA, Gibson DF, Krebs-Thomson K, Powell SB, Geyer MA, Hacksell U, Brann MR. 5-hydroxytryptamine2A receptor inverse agonists as antipsychotics. J Pharmacol Exp Ther. 2001;299:268-276. [PubMed] |
| 169. | Hinze-Selch D, Mullington J, Orth A, Lauer CJ, Pollmächer T. Effects of clozapine on sleep: a longitudinal study. Biol Psychiatry. 1997;42:260-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 55] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 170. | Monti JM, Monti D. Sleep in schizophrenia patients and the effects of antipsychotic drugs. Sleep Med Rev. 2004;8:133-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 139] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 171. | Eden RJ, Costall B, Domeney AM, Gerrard PA, Harvey CA, Kelly ME, Naylor RJ, Owen DA, Wright A. Preclinical pharmacology of ropinirole (SKandF 101468-A) a novel dopamine D2 agonist. Pharmacol Biochem Behav. 1991;38:147-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 98] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 172. | Frankowska M, Filip M, Przegaliński E. Effects of GABAB receptor ligands in animal tests of depression and anxiety. Pharmacol Rep. 2007;59:645-655. [PubMed] |
| 173. | Miller R. Mechanisms of action of antipsychotic drugs of different classes, refractoriness to therapeutic effects of classical neuroleptics, and individual variation in sensitivity to their actions: Part I. Curr Neuropharmacol. 2009;7:302-314. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 174. | Centonze D, Usiello A, Costa C, Picconi B, Erbs E, Bernardi G, Borrelli E, Calabresi P. Chronic haloperidol promotes corticostriatal long-term potentiation by targeting dopamine D2L receptors. J Neurosci. 2004;24:8214-8222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 79] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 175. | Jibiki I, Wakita S, Kubota T, Kurokawa K, Fukushima T, Yamaguchi N. Haloperidol-induced blockade of induction of long-term potentiation in perforant path-dentate gyrus pathway in chronically prepared rabbits. Pharmacol Biochem Behav. 1993;46:847-852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 176. | Terry AV Jr, Hill WD, Parikh V, Waller JL, Evans DR, Mahadik SP. Differential effects of haloperidol, risperidone, and clozapine exposure on cholinergic markers and spatial learning performance in rats. Neuropsychopharmacology. 2003;28:300-309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 67] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 177. | Terry AV Jr, Warner SE, Vandenhuerk L, Pillai A, Mahadik SP, Zhang G, Bartlett MG. Negative effects of chronic oral chlorpromazine and olanzapine treatment on the performance of tasks designed to assess spatial learning and working memory in rats. Neuroscience. 2008;156:1005-1016. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 178. | Adam K, Allen S, Carruthers-Jones I, Oswald I, Spence M. Mesoridazine and human sleep. Br J Clin Pharmacol. 1976;3:157-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 179. | Branchey MH, Brebbia DR, Cooper TB, Simpson GM. Effects of loxapine on the sleep of chronic schizophrenics. Psychopharmacology (Berl). 1979;62:201-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 180. | Cohrs S. Sleep disturbances in patients with schizophrenia : impact and effect of antipsychotics. CNS Drugs. 2008;22:939-962. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 295] [Cited by in RCA: 285] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 181. | Lester BK, Coulter JD, Cowden LC, Williams HL. Chlorpromazine and human sleep. Psychopharmacologia. 1971;20:280-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 182. | Meltzer HY, Massey BW. The role of serotonin receptors in the action of atypical antipsychotic drugs. Curr Opin Pharmacol. 2011;11:59-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 262] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 183. | de Bartolomeis A, Tomasetti C, Iasevoli F. Update on the Mechanism of Action of Aripiprazole: Translational Insights into Antipsychotic Strategies Beyond Dopamine Receptor Antagonism. CNS Drugs. 2015;29:773-799. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 126] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 184. | Kubota T, Jibiki I, Kishizawa S, Kurokawa K. Clozapine-induced potentiation of synaptic responses in the perforant path-dentate gyrus pathway in chronically prepared rabbits. Neurosci Lett. 1996;211:21-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 185. | Gemperle A, Olpe HR. Effects of subchronic clozapine treatment on long-term potentiation in rat prefrontal cortex. Eur Neuropsychopharmacol. 2004;14:340-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 186. | Kubota T, Jibiki I, Kurokawa S. Effects of risperidone, an atypical antipsychotic drug, on excitatory synaptic responses in the perforant path-dentate gyrus pathway in chronically prepared rabbits. Pharmacol Biochem Behav. 2001;70:237-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 187. | Shim SS, Hammonds MD, Tatsuoka C, Feng IJ. Effects of 4-weeks of treatment with lithium and olanzapine on long-term potentiation in hippocampal area CA1. Neurosci Lett. 2012;524:5-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 188. | Mutlu O, Ulak G, Celikyurt IK, Tanyeri P, Akar FY, Erden F. Effects of olanzapine and clozapine on memory acquisition, consolidation and retrieval in mice using the elevated plus maze test. Neurosci Lett. 2011;501:143-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 189. | Houthoofd SA, Morrens M, Sabbe BG. Cognitive and psychomotor effects of risperidone in schizophrenia and schizoaffective disorder. Clin Ther. 2008;30:1565-1589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 49] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 190. | Giménez S, Clos S, Romero S, Grasa E, Morte A, Barbanoj MJ. Effects of olanzapine, risperidone and haloperidol on sleep after a single oral morning dose in healthy volunteers. Psychopharmacology (Berl). 2007;190:507-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 60] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 191. | Sharpley AL, Vassallo CM, Cowen PJ. Olanzapine increases slow-wave sleep: evidence for blockade of central 5-HT(2C) receptors in vivo. Biol Psychiatry. 2000;47:468-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 82] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 192. | Descarries L, Riad M. Effects of the antidepressant fluoxetine on the subcellular localization of 5-HT1A receptors and SERT. Philos Trans R Soc Lond B Biol Sci. 2012;367:2416-2425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 69] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 193. | Davis R, Wilde MI. Mirtazapine : A Review of its Pharmacology and Therapeutic Potential in the Management of Major Depression. CNS Drugs. 1996;5:389-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 85] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 194. | Han H, Dai C, Dong Z. Single fluoxetine treatment before but not after stress prevents stress-induced hippocampal long-term depression and spatial memory retrieval impairment in rats. Sci Rep. 2015;5:12667. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 195. | Alme MN, Wibrand K, Dagestad G, Bramham CR. Chronic fluoxetine treatment induces brain region-specific upregulation of genes associated with BDNF-induced long-term potentiation. Neural Plast. 2007;2007:26496. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 76] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 196. | Wang JW, David DJ, Monckton JE, Battaglia F, Hen R. Chronic fluoxetine stimulates maturation and synaptic plasticity of adult-born hippocampal granule cells. J Neurosci. 2008;28:1374-1384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 379] [Cited by in RCA: 418] [Article Influence: 24.6] [Reference Citation Analysis (0)] |
| 197. | Wichniak A, Wierzbicka A, Walęcka M, Jernajczyk W. Effects of Antidepressants on Sleep. Curr Psychiatry Rep. 2017;19:63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 219] [Cited by in RCA: 290] [Article Influence: 36.3] [Reference Citation Analysis (0)] |
| 198. | Koesters M, Ostuzzi G, Guaiana G, Breilmann J, Barbui C. Vortioxetine for depression in adults. Cochrane Database Syst Rev. 2017;7:CD011520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 199. | Dale E, Zhang H, Leiser SC, Xiao Y, Lu D, Yang CR, Plath N, Sanchez C. Vortioxetine disinhibits pyramidal cell function and enhances synaptic plasticity in the rat hippocampus. J Psychopharmacol. 2014;28:891-902. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 75] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 200. | Cruz MP. Vilazodone HCl (Viibryd): A Serotonin Partial Agonist and Reuptake Inhibitor For the Treatment of Major Depressive Disorder. P T. 2012;37:28-31. [PubMed] |
| 201. | Loane C, Politis M. Buspirone: what is it all about? Brain Res. 2012;1461:111-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 145] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 202. | Huang CC, Liang YC, Hsu KS. A role for extracellular adenosine in time-dependent reversal of long-term potentiation by low-frequency stimulation at hippocampal CA1 synapses. J Neurosci. 1999;19:9728-9738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 96] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 203. | Flood JF, Cherkin A. Fluoxetine enhances memory processing in mice. Psychopharmacology (Berl). 1987;93:36-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 80] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 204. | Lyons L, ElBeltagy M, Umka J, Markwick R, Startin C, Bennett G, Wigmore P. Fluoxetine reverses the memory impairment and reduction in proliferation and survival of hippocampal cells caused by methotrexate chemotherapy. Psychopharmacology (Berl). 2011;215:105-115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 87] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 205. | Borkowska A, Drozdz W, Ziółkowska-Kochan M, Rybakowski J. Enhancing effect of mirtazapine on cognitive functions associated with prefrontal cortex in patients with recurrent depression. Neuropsychopharmacol Hung. 2007;9:131-136. [PubMed] |
| 206. | McIntyre RS, Harrison J, Loft H, Jacobson W, Olsen CK. The Effects of Vortioxetine on Cognitive Function in Patients with Major Depressive Disorder: A Meta-Analysis of Three Randomized Controlled Trials. Int J Neuropsychopharmacol. 2016;19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 140] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 207. | Stackman RW, Walsh TJ. Baclofen produces dose-related working memory impairments after intraseptal injection. Behav Neural Biol. 1994;61:181-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 50] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 208. | Leong KC, Goodman J, Packard MG. Buspirone blocks the enhancing effect of the anxiogenic drug RS 79948-197 on consolidation of habit memory. Behav Brain Res. 2012;234:299-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 209. | Murck H, Frieboes RM, Antonijevic IA, Steiger A. Distinct temporal pattern of the effects of the combined serotonin-reuptake inhibitor and 5-HT1A agonist EMD 68843 on the sleep EEG in healthy men. Psychopharmacology (Berl). 2001;155:187-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 32] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 210. | Ott GE, Rao U, Lin KM, Gertsik L, Poland RE. Effect of treatment with bupropion on EEG sleep: relationship to antidepressant response. Int J Neuropsychopharmacol. 2004;7:275-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 211. | Finnimore AJ, Roebuck M, Sajkov D, McEvoy RD. The effects of the GABA agonist, baclofen, on sleep and breathing. Eur Respir J. 1995;8:230-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 59] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 212. | Simpson D, Curran MP. Ramelteon: a review of its use in insomnia. Drugs. 2008;68:1901-1919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 44] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 213. | Ayoub MA, Levoye A, Delagrange P, Jockers R. Preferential formation of MT1/MT2 melatonin receptor heterodimers with distinct ligand interaction properties compared with MT2 homodimers. Mol Pharmacol. 2004;66:312-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 158] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 214. | Kamal M, Gbahou F, Guillaume JL, Daulat AM, Benleulmi-Chaachoua A, Luka M, Chen P, Kalbasi Anaraki D, Baroncini M, Mannoury la Cour C, Millan MJ, Prevot V, Delagrange P, Jockers R. Convergence of melatonin and serotonin (5-HT) signaling at MT2/5-HT2C receptor heteromers. J Biol Chem. 2015;290:11537-11546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 87] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 215. | Patel KV, Aspesi AV, Evoy KE. Suvorexant: a dual orexin receptor antagonist for the treatment of sleep onset and sleep maintenance insomnia. Ann Pharmacother. 2015;49:477-483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 45] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 216. | Ellis J, Pediani JD, Canals M, Milasta S, Milligan G. Orexin-1 receptor-cannabinoid CB1 receptor heterodimerization results in both ligand-dependent and -independent coordinated alterations of receptor localization and function. J Biol Chem. 2006;281:38812-38824. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 171] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 217. | Matsuki T, Nomiyama M, Takahira H, Hirashima N, Kunita S, Takahashi S, Yagami K, Kilduff TS, Bettler B, Yanagisawa M, Sakurai T. Selective loss of GABA(B) receptors in orexin-producing neurons results in disrupted sleep/wakefulness architecture. Proc Natl Acad Sci U S A. 2009;106:4459-4464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 99] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 218. | Gold SJ, Ni YG, Dohlman HG, Nestler EJ. Regulators of G-protein signaling (RGS) proteins: region-specific expression of nine subtypes in rat brain. J Neurosci. 1997;17:8024-8037. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 321] [Cited by in RCA: 334] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 219. | Overington JP, Al-Lazikani B, Hopkins AL. How many drug targets are there? Nat Rev Drug Discov. 2006;5:993-996. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2616] [Cited by in RCA: 2584] [Article Influence: 143.6] [Reference Citation Analysis (0)] |
| 220. | Crupi R, Impellizzeri D, Cuzzocrea S. Role of Metabotropic Glutamate Receptors in Neurological Disorders. Front Mol Neurosci. 2019;12:20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 166] [Article Influence: 27.7] [Reference Citation Analysis (0)] |
| 221. | Kumar K, Sharma S, Kumar P, Deshmukh R. Therapeutic potential of GABA(B) receptor ligands in drug addiction, anxiety, depression and other CNS disorders. Pharmacol Biochem Behav. 2013;110:174-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 82] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 222. | Hisahara S, Shimohama S. Dopamine receptors and Parkinson's disease. Int J Med Chem. 2011;2011:403039. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 56] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 223. | Ledonne A, Mercuri NB. Current Concepts on the Physiopathological Relevance of Dopaminergic Receptors. Front Cell Neurosci. 2017;11:27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 55] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 224. | Pae CU, Serretti A, Patkar AA, Masand PS. Aripiprazole in the treatment of depressive and anxiety disorders: a review of current evidence. CNS Drugs. 2008;22:367-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 55] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 225. | Fava M. Daytime sleepiness and insomnia as correlates of depression. J Clin Psychiatry. 2004;65 Suppl 16:27-32. [PubMed] |