Peer-review started: August 25, 2016
First decision: September 27, 2016
Revised: January 10, 2017
Accepted: February 8, 2017
Article in press: February 10, 2017
Published online: May 2, 2017
Processing time: 161 Days and 21.5 Hours
Scald injuries, which describe burns to living tissue from hot liquids, are a very common injury that occur across geographical, social, economic, and national boundaries. Despite their ubiquitous nature, a complete understanding of the conditions which are required to cause scald burns is not yet available. In addition, clear guidance to medical practitioners is available through various guidelines however in actual situations, the extent of the burn is not fully known and this lack of knowledge complicates care. Here, a comprehensive review is made of the available knowledge of temperatures and scald durations which lead to skin-burn injuries. The range of volumes and liquid temperatures are typical of those found in heated consumer beverages. This review can help medical practitioners design initial treatment protocols and can be used by manufacturers of hot-liquid products to avoid the most severe burns. Next, within the context of this ability to quantify burn depths, a review of current burn treatment guidelines is given. Included in this review is a visual recognition of the extent of burns into the dermal layer as well as decision guidelines for selection of patients which would benefit from referral to a dedicated burn center. It is hoped that by bringing together both the quantified burn-depth information and current treatment guidelines, this review can be used as a resource for persons in the medical, manufacturing, beverage service, and other industries to reduce the human impact of scald injuries.
Core tip: This paper presents a concise summary that relates hot-beverage spills to burn injury risk. Not only can this paper be used to predict the depth of burn injuries, but it can also show how service temperature and cooling time can be set to reduce the threat of injury. Results are presented in simple to use tables and graphs for ease to medical practitioners.
- Citation: Bourdon RT, Nelson-Cheeseman BB, Abraham JP. Review of the initial treatment and avoidance of scald injuries. World J Dermatol 2017; 6(2): 17-26
- URL: https://www.wjgnet.com/2218-6190/full/v6/i2/17.htm
- DOI: https://dx.doi.org/10.5314/wjd.v6.i2.17
Burn injuries are a common type of injury that can occur in many situations around the globe. Within the category of burn injuries, scald wounds caused by hot liquids are among the most common. Scald injuries can occur in kitchens, baths, industrial and manufacturing environments, restaurants, and other locations. The extent of scald injuries can vary from mild to death causing. Mild burns are almost always treated without medical attention, while extreme burn injuries often result in referral to dedicated burn centers. Because of the large variation in harm caused by burns, it is important to create environments which lessen burn injuries. It is also important for medical responders to quickly and correctly categorize a burn so that appropriate treatment is initiated.
Scald burns, which here refer to any thermal injury caused by heated liquid, can occur anywhere on exposed body; however, they are most common on the skin. Since the vast majority of scalds are skin burns, they will be the sole focus of this review.
The severity of a skin burn is quantified by two measurements. The first is the depth of the burn into the tissue. The second measure is an estimation of the size of the surface area involved. Both of these measures will now be discussed.
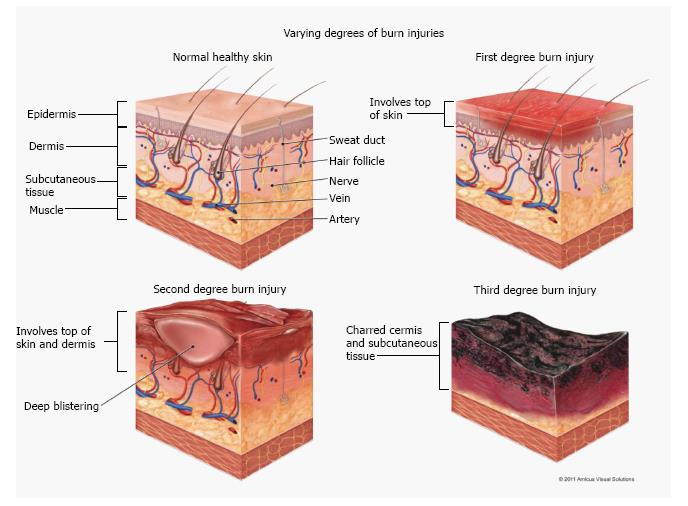
Both within the medical community as well as with the general public, the degree classification is most often used to describe burn depth. Within this classification, the description first-degree burn describes burns that kill tissue only within the outermost layer of skin (the epidermis). As a consequence, the burns generally heal quickly and without medical attention. The injuries may be painful and result in hyperemia and flaking of the necrosed epidermis following the incident.
Second-degree burns pass through the epidermal layer and into the dermis. They lead to thermal necrosis of tissue within that layer and consequently damage the skin structures which are housed there, such as hair follicles, sweat glands, capillaries, among others.
Third-degree burns pass through the dermis and enter the underlying hypodermis tissue and possibly into muscle. Since the burn completely destroys the dermis, the functions of this layer are also halted (such as blood flow and sensation). Consequently, the skin is ischemic. While it may appear hyperemic, when the skin is compressed, reperfusion of the tissue is very slow to occur if at all. The pain associated with third-degree burns is often less than that of second- or first-degree burns because the innervation to the tissue is compromised. For third-degree burns, slow healing occurs and medical attention such as surgical excision and grating is required. If large parts of the body are burned, shock, hypothermia, and infection can occur[1-3].
Within the academic and bioheat transfer community, different descriptions are used. In this community, superficial burns are the equivalent to burns of first degree. Superficial-partial-thickness burns are those that pass through the epidermis and into the external part of the dermal layer - generally not beyond the dermal midplane. Next, deep-partial-thickness burns pass through the dermal midplane and into the lower half of this layer. Finally, full-thickness burns are essentially the same as third-degree burns - they extend through the dermal layer and into underlying tissue. As with third-degree burns, they often require skin grafting[4-6]. The primary difference between the two modes of burn classification is that in the latter, a separate accounting is made for burns that either are confined to the outer half of the dermis and those that pass into the lower half of the dermal layer.
Regardless of the preferred nomenclature, it is a challenge to visually assess burn depth. Many times, the visual estimation is not accurate[5,7-10]. A summary image is provided in Figure 1 to bring together the various burn extents which have already been discussed in the accompanying text.
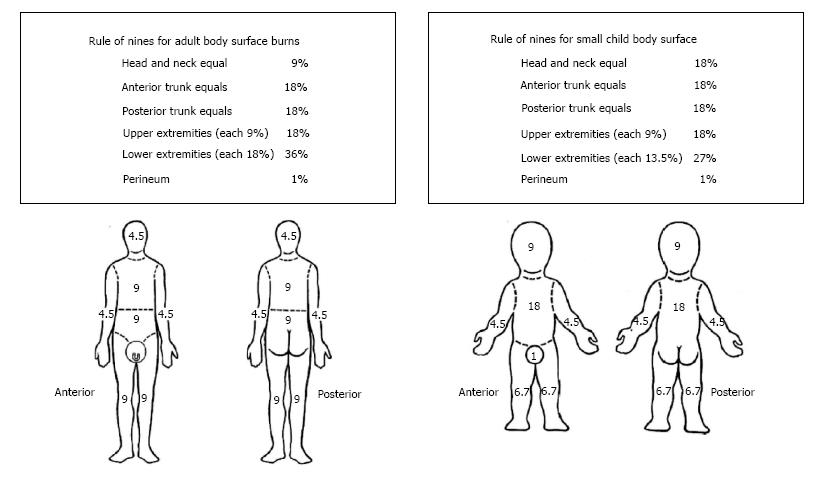
Total burn surface area (TBSA) is also an important measure of injury. TBSA is most commonly reported as a % of the body area that a burn covers. With this method, the body is subdivided into major regions (head, torso, legs, arms, etc.). Each region is allocated a numerical value which is a typical percentage of the region compared to the total body surface. An example of such a regional breakdown is provided in Figure 2. Since many of the body regions are multiples of 9%, this regional breakdown is often referred to as the Rule of Nines. The Rule of Nines is endorsed by both the American Burn Association and the European Practice Guidelines for Burn Care. While other burn area criteria have been studied, the widespread current usage of this metric motivated its inclusion here.
More information will be provided in a later section of this report related to decision making of medical professionals. First, however, quantitative information on the depth of the burn will be provided.
Burn depth studies have been performed since the late 1940s. Pioneering work[11-14] quantified the complex temperature-time relationship required for burns on porcine and human skin. It was discovered that the physiologic response to elevated temperatures was complex and that for small increases in exposure temperature, large changes in burn injury rates were observed. The outcome of this work was a quantification of the rate of cellular damage and the amount of viable cells still within the heated zone which is expressed mathematically as:
Damage index = Ω = ln [c (0)/c (t)] = ∫ Ae-(Ea/RT) dt (1)
Here, C represents the concentration of viable cells either initially or at some time t. A is the frequency factor (1/s), Ea is the activation energy (J/mol), R is the universal gas constant (8.3143 J/mol-K), T is the temperature in Kelvin, e is the constant 2.718, and t is the time (s)[15-31]. The fitted terms are obtained through experimental correlation, and they depend on the tissue type and body location. To illustrate the wide-ranging values of these parameters, Table 1 has been prepared.
| Ref. | A (1/s) | Ea (J/kmol) |
| [14,23] | 3.10e98 | 6.28e08 |
| [25] | 2.90e37 | 2.44e08 |
| [26] | 9.09e37 | 2.49e08 |
| [27] | 4.33e64, T < 50 | 24.19e8, T < 50 |
| 9.39e104, T > 50 | 6.70e8, T > 50 | |
| [13,24] | 3.10e98 | 6.28e08 |
| [28] | 2.19e124, T < 50 | 7.78e8, T < 50 |
| 1.82e51, T > 50 | 3.25e8, T > 50 | |
| [29] | 1.43e72 | 4.57e08 |
| [29] | 2.86e69 | 4.61e08 |
| [29] | 4.32e54, T < 50 | 4.16e8, T < 50 |
| 9.39e104, T < 60 | 6.65e8, T < 60 | |
| [30] | 3.1e98, T < 55 | 6.27e8, T < 55 |
| 5e45, T > 55 | 2.96e8, T > 55 | |
| [31] | 2.19e124, T < 50 | 7.82e8, T < 50 |
| 1.82e51, T > 50 | 3.27e8, T > 50 | |
| [27] | 4.33e64, T < 50 | 4.18e8, T < 50 |
| 9.39e104, T > 50 | 6.69e8, T > 50 |
The prediction of injury [through Equation (1)] is achieved by solving for the conduction of heat through perfused tissue using the Pennes model[32] which is expressed in Equation (2).
(ρc)t (Tt/t) = k (2Tt/x2) + Smet + (ρc)b ω(Tb - Tt) (2)
The calculation method outlined here has a long history of use and has been corroborated with experimental results. Results from those works have been adopted to predict skin burns using modern computational techniques[33-57]. An outcome of these efforts is the ability to predict the distribution of an injury parameter throughout the various layers of tissue that have been heated. Values of are taken to correspond to completely necrosed tissue as they generally represent tissue which has too few viable cells to regenerate.
In the present study, focus is given on a subclass of scald burns - those caused by hot beverages. While the spill scenario can vary dramatically (volume of liquid spilled, temperature of liquid, spill pattern, presence or absence of clothing, speed of spill, thickness of skin, ability of victim to remove heat and apply cooling duration of hot liquid cooling prior to spill, among others), it is possible to generalize the results in a way that can provide meaningful information to treating physicians. To enable the generalization, the following ranges of parameters were used in the experiments.
Volume of liquid spilled: 8-16oz (237-473 mL).
Temperature of liquid spilled: 158 ℉-203 ℉ (70 °C-95 °C).
Type of clothing: 1 layer of a cotton shirt.
Spills of cups that had caps as well as cups without caps.
Other parameters, such as the constitution of the cup, the presence or absence of an insulating sleeve, etc. had a very small impact on the results and so for simplicity are not included here[46].
The experiments, which were carried out involving spills on living human tissue and on a tissue surrogate are described in[46] and are not discussed in detail here. The experiments were carried out in typical ambient conditions of about 20 °C. The temperature during the cooling period for an 8 ounce (237 mL) beverages without a protective cover are shown below in Table 2. Corresponding information for covered beverages are provided in Table 3. These tables are followed immediately by information for larger volume beverages (16 ounces) both with and without protective caps (Tables 4 and 5).
| Cooling time (min) | Service temperature, °C (℉) | |||||
| 70 (158) | 75 (167) | 80 (176) | 85 (185) | 90 (194) | 95 (203) | |
| 0 | 70 | 75 | 80 | 85 | 90 | 95 |
| 5 | 58.1 | 61.9 | 61.9 | 69.5 | 73.4 | 77.2 |
| 10 | 50.4 | 53.4 | 53.4 | 59.5 | 62.5 | 65.5 |
| 15 | 45.1 | 47.6 | 47.6 | 52.6 | 55.2 | 57.7 |
| 20 | 42.4 | 44.6 | 44.6 | 49.1 | 51.4 | 53.6 |
| 25 | 42.2 | 44.4 | 44.4 | 48.9 | 51.1 | 53.3 |
| Cooling time (min) | Service temperature, °C (℉) | |||||
| 70 (158) | 75 (167) | 80 (176) | 85 (185) | 90 (194) | 95 (203) | |
| 0 | 70 | 75 | 80 | 85 | 90 | 95 |
| 5 | 62.5 | 66.7 | 66.7 | 75.2 | 79.5 | 83.7 |
| 10 | 56.9 | 60.6 | 60.6 | 67.9 | 71.6 | 75.3 |
| 15 | 52.5 | 55.8 | 55.8 | 62.3 | 65.6 | 68.8 |
| 20 | 49.5 | 52.5 | 52.5 | 58.4 | 61.3 | 64.3 |
| 25 | 47.7 | 50.5 | 50.5 | 56.1 | 58.8 | 61.6 |
| Cooling time (min) | Service temperature, °C (℉) | |||||
| 70 (158) | 75 (167) | 80 (176) | 85 (185) | 90 (194) | 95 (203) | |
| 0 | 70 | 75 | 80 | 85 | 90 | 95 |
| 5 | 61.5 | 65.7 | 69.8 | 74 | 78.1 | 82.3 |
| 10 | 55.9 | 59.5 | 63.1 | 66.7 | 70.3 | 73.9 |
| 15 | 51.9 | 55.1 | 58.3 | 61.5 | 64.7 | 67.9 |
| 20 | 49.6 | 52.5 | 55.5 | 58.5 | 61.4 | 64.4 |
| 25 | 48.9 | 51.8 | 54.7 | 57.5 | 60.4 | 63.3 |
| Cooling time (min) | Service temperature, °C (℉) | |||||
| 70 (158) | 75 (167) | 80 (176) | 85 (185) | 90 (194) | 95 (203) | |
| 0 | 70 | 75 | 80 | 85 | 90 | 95 |
| 5 | 65 | 69.5 | 74 | 78.5 | 83 | 87.5 |
| 10 | 61.5 | 65.7 | 69.8 | 74 | 78.1 | 82.3 |
| 15 | 58.8 | 62.7 | 66.6 | 70.5 | 74.4 | 78.3 |
| 20 | 57 | 60.7 | 64.4 | 68.1 | 71.8 | 75.5 |
| 25 | 56 | 59.6 | 63.2 | 66.8 | 70.4 | 74 |
The calculations were continued until the temperatures within the tissue reduced to levels which would no longer cause injury. Interested readers are invited to references[46] and which discuss both the cooling effect[56], the impact of rapid cooling after a burn, and the duration of time needed to bring tissue temperatures to safe levels.
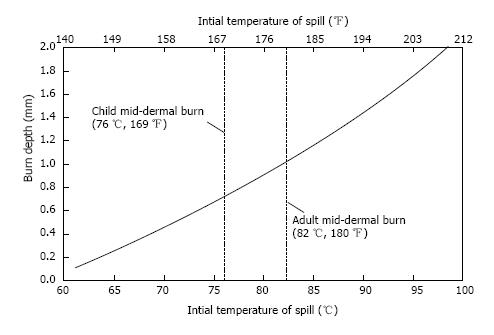
From the experiments and calculations completed[46], verified by a simplified model[56] and by the comparison between models and physical observations which were contained therein, it is possible to create a visual reference which allows the estimation of burn risk caused by hot-liquid spills. The reference is provided in Figure 3.
The information presented in Figure 3 and the preceding tables can be brought together in a simple to use manner as seen in Tables 6-9. These tables list the initial beverage temperature, the volume, and the cooling time which should occur to bring the beverage to a low enough temperature so that a threshold mid-dermal burn may not occur. Table 6 corresponds to small (237 mL) cups cooling with a protective cap; Table 7 is the counterpart for the no-cap situation. Tables 8 and 9 present information for 473 mL cups with and without a protective cap, respectively.
| Service temperature | Cooling time for adult mid-dermal burns (min) | Cooling time for children mid-dermal burns (min) | |
| (°C) | (℉) | ||
| 95 | 203 | 8 | 12 |
| 90 | 194 | 5 | 9 |
| 85 | 185 | 2 | 6 |
| 80 | 176 | Not applicable | 3 |
| Service temperature | Cooling time for adult mid-dermal burns (min) | Cooling time for children mid-dermal burns (min) | |
| (°C) | (℉) | ||
| 95 | 203 | 4 | 6 |
| 90 | 194 | 2 | 4 |
| 85 | 185 | 1 | 3 |
| 80 | 176 | Not applicable | 1 |
| Service temperature | Cooling time for adult mid-dermal burns (min) | Cooling time for children mid-dermal burns (min) | |
| (°C) | (℉) | ||
| 95 | 203 | 11 | 18 |
| 90 | 194 | 7 | 12 |
| 85 | 185 | 2 | 8 |
| 80 | 176 | Not applicable | 3 |
| Service temperature | Cooling time for adult mid-dermal burns (min) | Cooling time for children mid-dermal burns (min) | |
| (°C) | (℉) | ||
| 95 | 203 | 6 | 9 |
| 90 | 194 | 4 | 6 |
| 85 | 185 | 2 | 4 |
| 80 | 176 | Not applicable | 2 |
These data show that beverage service temperatures, which are often in excess of 180 ℉ are at levels which have the potential to cause serious physical harm. Furthermore, some beverage service temperatures are above the preferred temperature for the consumers[57-61]. It seems reasonable to promote the service of beverages at temperatures which are both preferred by consumers and safe so that serious mid-dermal burns are unlikely. The cooling results set forth here are corroborated by other mutually reinforcing studies[62-64].
It should also be noted that while the above tables correspond to typical adults and children (whose skin thickness is approximately 70% that of an adult), great care must be given to their use[55,65,66]. Also, as summarized in[55], skin thickness varies by body location[67-70]. For locations or persons whose skin is thinner than that used in the current study, lower temperatures or longer cooling durations are recommended. On the other hand, for persons whose skin is thicker than 2 mm, higher temperatures and/or shorter cooling durations can be used.
In addition to skin thickness, clothing type and the ability of someone to remove heat quickly and apply cool or cold temperatures should be a consideration. For persons whose mobility is limited, such as children or elderly, a spill may remain in contact with skin for a longer duration than for someone who quickly removes the source of heat (the spilled liquid and any saturated clothing). Consequently, for these mobility-challenged persons, lower temperatures and/or longer cooling durations are recommended.
With respect to clothing, it has two competing effects. First, clothing can insulate the skin against the hottest temperatures of the liquid. Second, the clothing can hold the hot liquid against the skin, extending the scald and delaying cooling. From a heat transfer perspective, a first responder should immediately remove the source of heat by removing saturated clothing if possible. In addition, the responder should apply cool/cold temperatures to quickly reduce the skin temperature. Room temperature liquids are often available and an excellent choice. It is recommended that if cool liquids such as room temperature water or other beverages are available nearby, they can be applied directly to the burn location and even through clothing. Burns occur very quickly and even a few second delay in the application of cooling can make an impact on burn depth.
Not only does cooling reduce temperatures and thereby reduce burn depth[55-56], but it also provides palliative relief[71-84]. Many of these studies have investigated the speed at which cooling should be applied to maximize the benefit. Others have considered the optimal temperatures for the cooling. While the consensus is that temperatures in the range of 10 °C-20 °C (50 ℉-70 ℉) are effective, in our opinion, speed is of critical importance. A delay of a second or two can make the difference between a superficial-partial-thickness burn and a deep-partial-thickness burn. In some instances, ice is used to cause cooling. Ice should be used with care, because very cold temperatures can cause vasoconstriction which inhibits blood flow to the injured region and slows healing. Extended application of cold temperatures can even cause cryological injuries[85-92].
Depth of the burn, TBSA involved, and the location of the burn all are necessary to develop a treatment plan. Burn depth occurs on a continuum and accurate assessment of burn depth is often difficult immediately after the injury[3,6-10,13-21]. Burn depth assessment helps predict which injuries will require excision and grafting as deep-partial-thickness burns and full-thickness burns often require this treatment.
First-degree (superficial) burns are red, have a dry surface, and typically are associated with discomfort. These burns do not blister. The affected skin will blanch with pressure and quickly reperfuse. A typical sunburn is an example of a first-degree burn.
Superficial second-degree (superficial-partial-thickness) burns are painful and form blisters. The surface under the blister is typically red, hypersensitive and moist. This surface will blanch with pressure.
Deep second-degree (deep-partial-thickness) burns blister and may be less painful than superficial partial thickness burns. The tissue underlying the blister will be moist and may appear mottled, red, or even white. Reperfusion after blanching is slow or absent. Sensation of the damaged tissue maybe diminished when tested with pin prick.
Third-degree (full-thickness) burns extend into subcutaneous fat or connective tissue. These injuries appear white or tan and are insensate. They do not blanch as there is no perfusion to this area of the burn due to damage of the capillaries.
Fourth-degree (full-thickness) burns extend into deep structures such as muscle or bone. These injuries are easily identified.
The size of the burn is estimated based on the percentage of total body surface area involved. Only partial-thickness burns and deeper are used when estimating TBSA burned. This estimation informs intravenous fluid resuscitation needs and is used to determine if a patient would benefit from a referral to a burn center. A common method for estimating TBSA burned is the Rule of Nines as described above. This method is endorsed by the American Burn Association, but several other methods exist. For smaller burns, the patient’s hand (palm and fingers) can be used as a reference as this surface area is approximately 1% TBSA.
The final aspect of the initial burn assessment involves noting the area of the body involved. Partial thickness and deep burns involving the face, hands, feet, perineum, genitals, and major joints often require consultation with a burn center.
Immediate removal of the burn source and cooling of the affected area is critical to prevent further tissue damage. Cooling is most easily accomplished by application of cool or even room temperature water. If immediate removal of any overlying clothing is possible this may be done before application of water. Otherwise, liquid can be poured directly on porous clothing to begin the cooling process. Cool water is a safe, effective, and soothing intervention. Therefore, continue with the application of cool water until the burning process is completely halted.
Serious burns require initial treatment at an emergency center. A complete review of the initial care of the burned patient is beyond the scope of this paper. The following text will therefore focus on how the burn assessment informs subsequent treatment. After any life threatening injuries and medical complications have been addressed, the burning process has been halted, and any necessary analgesics have been administered a burn assessment can be performed. The clinician will need to assess all burns noting location and depth. Next, TBSA burned can be estimated using only partial thickness burns and deeper for this calculation.
Fluid loss through damaged skin and loss into the interstitial space can result in hypovolemic shock and thus inadequate tissue perfusion. This can lead to not only viable skin becoming nonviable, but also to end organ dysfunction. Resuscitation with intravenous fluid is the mainstay for addressing and preventing burn shock. As noted previously, first degree burns are not included in the TBSA estimation for fluid resuscitation calculations[2,3]. Adult patients with less than 20% TBSA affected and pediatric patients will less than 10% TBSA affected can often be managed with oral hydration alone[3]. Larger burns require intravenous fluid resuscitation with crystalloids and ringer’s lactate solution is commonly used. Multiple formulas exist for estimating intravenous fluid needs in the first 24 to 48 h after a burn. The Parkland Formula is one of the most popular. This formula uses the variables TBSA burned and patient weight in kilograms. The Parkland formula estimates the milliliters of intravenous fluid to be given in the first 24 h after the burn occurs. Half of the volume is administered over the first 8 h and the second half of the volume is administered over the following 16 h.
The Parkland Formula: Milliliters of IV fluid = 4 x TBSA burned as percent x weight in kilograms (3)
The American Burn Association has published guidelines to help clinicians determine which patients would benefit from referral to a burn center[1].
Criteria recommended by the American Burn Association for burn center referrals are: (1) Partial thickness burns greater than 10% of body area; (2) burns of hands, feet, face, genitals, perineum, or major joints; (3) third degree burns; (4) electrical burns (including lightning); (5) chemical burns; (6) inhalation burns; (7) burns in patients with pre-existing conditions that could complicate management; (8) concomitant trauma which increases morbidity or mortality risk; (9) burns of minors treated in hospitals without qualified personnel or equipment; and (10) burn injury which requires social, emotional or rehabilitation intervention.
Here, a two-fold presentation of information is given. First, the relationship of liquid temperature to burn depth is showcased with an easy-to-use graph. Included in the results is information regarding how the cooling time between hot-beverage service and a spill incident will reduce burn depth. With this information, it is possible to predict, within a reasonable degree of certainty, the depth of a burn injury. In addition, required beverage cooling times to avoid mid-dermal burns are listed.
In addition, the information presented here can be used by the beverage service industry to make safer hot-liquid beverages. Often times, service temperatures are above those preferred by consumers and these elevated temperatures pose an unnecessary risk of injury.
While the focus in this study was on mid-dermal burns, it should be recognized that lesser burns can also be injurious. Also, readers should recognize that mid-dermal burns can be caused at lower temperatures on areas where the skin is thinner, when victim mobility is challenged, or when the situation makes difficult the removal of the source of heat and the application of cooling.
With respect to medical care, there are a number of care phases that occur. First care starts prior to arrival at a medical center and involves the removal of heat and application of cooling. Next, there is a stage of initial medical care which may include fluid resuscitation. For severe burn situations, referral to a burn center is made. Guidance for all these care phases is provided here with reference to published guidelines.
Manuscript source: Invited manuscript
Specialty type: Dermatology
Country of origin: United State
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Chen XL, Suliman MT S- Editor: Kong JX L- Editor: A E- Editor: Lu YJ
| 1. | Advanced Burn Life Support Course Provider Manual. USA: The American Burn Association, 2007. |
| 2. | Singer AJ, Taira BR, Lee CC. Rosen’s Emergency Medicine, 8th edition. Elsevier/Saunders: Thermal Burns 2014; Chapter 63. |
| 3. | Enoch S, Roshan A, Shah M. Emergency and early management of burns and scalds. BMJ. 2009;338:b1037. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 32] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 4. | Merz KM, Pfau M, Blumenstock G, Tenenhaus M, Schaller HE, Rennekampff HO. Cutaneous microcirculatory assessment of the burn wound is associated with depth of injury and predicts healing time. Burns. 2010;36:477-482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 35] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 5. | Johnson RM, Richard R. Partial-thickness burns: identification and management. Adv Skin Wound Care. 2003;16:178-187; quiz 188-189. [PubMed] |
| 6. | Monafo WW. Initial management of burns. N Engl J Med. 1996;335:1581-1586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 92] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 7. | Singer AJ, Berruti L, Thode HC, McClain SA. Standardized burn model using a multiparametric histologic analysis of burn depth. Acad Emerg Med. 2000;7:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 78] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 8. | Durrant CAT, Simpson AR, Williams G. Thermal injury the first 24 h. Current Anesthesia Crit Care. 2008;19:256-263. [RCA] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 9. | Heimbach DM, Afromowitz MA, Engrav LH, Marvin JA, Perry B. Burn depth estimation--man or machine. J Trauma. 1984;24:373-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 112] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 10. | Palla RL. A heat transfer analysis of scald injury. USA: National Technology Information Service, U.S. Department of Commerce 1981; . [DOI] [Full Text] |
| 11. | Henriques FC, Moritz AR. Studies of Thermal Injury: I. The Conduction of Heat to and through Skin and the Temperatures Attained Therein. A Theoretical and an Experimental Investigation. Am J Pathol. 1947;23:530-549. [PubMed] |
| 12. | Moritz AR, Henriques FC. Studies of Thermal Injury: II. The Relative Importance of Time and Surface Temperature in the Causation of Cutaneous Burns. Am J Pathol. 1947;23:695-720. [PubMed] |
| 13. | Moritz AR. Studies of Thermal Injury: III. The Pathology and Pathogenesis of Cutaneous Burns. An Experimental Study. Am J Pathol. 1947;23:915-941. [PubMed] |
| 14. | Henriques FC. Studies of thermal injury; the predictability and the significance of thermally induced rate processes leading to irreversible epidermal injury. Arch Pathol (Chic). 1947;43:489-502. [PubMed] |
| 15. | Pearce JA, Thomsen S. Kinetic models of laser-tissue fusion processes. Biomed Sci Instrum. 1993;29:355-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 16. | McMurray T, Pearce JA. Thermal damage quantification utilizing tissue birefringence color image analysis. Biomed Sci Instrum. 1993;29:235-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 17. | Sapareto SA, Dewey WC. Thermal dose determination in cancer therapy. Int J Radiat Oncol Biol Phys. 1984;10:787-800. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1719] [Cited by in RCA: 1530] [Article Influence: 37.3] [Reference Citation Analysis (0)] |
| 18. | Iwai M, Iwai Y, Suzumura S, Miyahara H, Imai S, Matsunaga T. Normal human salivary gland cells produce carcinoembryonic antigen-related antigen in collagen gels. In Vitro Cell Dev Biol. 1991;27A:759-762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 41] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 19. | Pearce JA. Comparative analysis of mathematical models of cell death and thermal damage processes. Int J Hyperthermia. 2013;29:262-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 107] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 20. | Lepock JR. Cellular effects of hyperthermia: relevance to the minimum dose for thermal damage. Int J Hyperthermia. 2003;19:252-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 251] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 21. | Arrhenius S. Uber die reaktionsgeschwindigkeit bei der inversion von rohrzucker durch sauren (on the reaction rate in the inversion of cane sugar by acids). Z Phys Chem. 1889;4:226-248. |
| 22. | Arrhenius S. Quantitative laws in biological chemistry. London: G. Bell & Sons 1915; 184. |
| 23. | Tropea BI, Lee RC. Thermal injury kinetics in electrical trauma. J Biomech Eng. 1992;114:241-250. [PubMed] [DOI] [Full Text] |
| 24. | Torvi DA, Dale JD. A finite element model of skin subjected to a flash fire. J Biomech Eng. 1994;116:250-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 84] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 25. | Taylor D. Physical mechanisms of cellular injury in electrical trauma. USA: PhD Thesis, MIT 1989; . |
| 26. | Moussa NA, McGrath JJ, Cravalho EG, Asimacopoulos PJ. Kinetics of Thermal Injury in Cells. J Biomech Eng. 1977;99:155-159. [RCA] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 27. | Takata A. Development of criterion for skin burns. Aerosp Med. 1974;45:634-637. |
| 28. | Weaver JA, Stoll AM. Mathematical model of skin exposed to thermal radiation. Aerosp Med. 1969;40:24-30. [PubMed] |
| 29. | Mehta AK, Wong F. Measurement of flammability and burn potential of fabrics. Technical Report, COM-73-10950, NTIS. 1973;. |
| 30. | Fugitt CE. A rate process of thermal injury. Armed Forces Special Weapons Project, AFSWP-606. 1955;. |
| 31. | Weaver JA, Stoll AM. Mathematical model of skin exposed to thermal radiation. Us Nav Air Develop Center Aviat Med Accel Lab Rep Mr: National Air Defense Command Memo Report 1967; 6708. |
| 32. | Pennes HH. Analysis of tissue and arterial blood temperatures in resting human forearm. J Applied Physiology. 1948;1:93-133. |
| 33. | Dai W, Yu H, Nassar R. A Fourth-Order compact finite-difference scheme for solving a 1-D Pennes’ bioheat transfer equation in a triple-layered skin structure. Num Heat Transfer B. 2004;46:447-461. [DOI] [Full Text] |
| 34. | Liu J, Zhu L, Xu LX. Studies on the three-dimensional temperature transients in the canine prostate during transurethral microwave thermal therapy. J Biomech Eng. 2000;122:372-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 14] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 35. | Diller KR. Modeling thermal skin burns on a personal computer. J Burn Care Rehabil. 1998;19:420-429. [PubMed] |
| 36. | Ng EY, Tan HM, Ooi EH. Boundary element method with bioheat equation for skin burn injury. Burns. 2009;35:987-997. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 24] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 37. | Diller KR, Hayes LJ, Blake GK. Analysis of alternate models for simulating thermal burns. J Burn Care Rehabil. 1991;12:177-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 21] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 38. | Ng EY, Chua LT. Prediction of skin burn injury. Part 1: Numerical modelling. Proc Inst Mech Eng H. 2002;216:157-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 25] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 39. | Ng EY, Chua LT. Prediction of skin burn injury. Part 2: Parametric and sensitivity analysis. Proc Inst Mech Eng H. 2002;216:171-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 37] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 40. | Ng EY, Chua LT. Comparison of one- and two-dimensional programmes for predicting the state of skin burns. Burns. 2002;28:27-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 94] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 41. | Abraham JP, Sparrow EM, Ramadhyani S. A mathematical model to predict tissue temperatures and necrosis during microwave thermal ablation of the prostate. New York: Numerical Heat Transfer 3: Numerical Implementation of Bioheat Models and Equations, Taylor and Francis 2009; . |
| 42. | Lovik RD, Abraham JP, Sparrow EM. Potential tissue damage from transcutaneous recharge of neuromodulation implants. Int J Heat Mass Trans. 2009;52:3518-3524. [DOI] [Full Text] |
| 43. | Smith DK, Lovik RD, Sparrow EM, Abraham JP. Human tissue temperatures achieved during recharging of new-generation neuromodulation devices. Int J Heat Mass Transfer. 2010;53:3292-3299. [DOI] [Full Text] |
| 44. | Abraham JP, Sparrow EM, Ramadhyani S. Numerical simulation of a bph thermal therapy - a case study involving TUMT. J Biomech Eng. 2007;129:548-557. [RCA] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 45. | Jiang SC, Ma N, Li HJ, Zhang XX. Effects of thermal properties and geometrical dimensions on skin burn injuries. Burns. 2002;28:713-717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 108] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 46. | Abraham JP, Nelson-Cheeseman BB, Sparrow E, Wentz JE, Gorman JM, Wolf SE. Comprehensive method to predict and quantify scald burns from beverage spills. Int J Hyperthermia. 2016;32:900-910. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 47. | Bourdon RT, Nelson-Cheeseman BB, Abraham JP. Prediction, identification, and initial treatment guidelines for scald injuries. Austin J Emerg Crit Care Med Special Issue on Burns. 2016;3:article no. 1043. |
| 48. | Diller KR, Hayes LJ. A finite element model of burn injury in blood-perfused skin. J Biomech Eng. 1983;105:300-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 72] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 49. | Viglianti BL, Dewhirst MW, Abraham JP, Gorman JM, Sparrow EM. Rationalization of thermal injury quantification methods: application to skin burns. Burns. 2014;40:896-902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 50. | Diller KR. Modeling of bioheat transfer processes at high and low temperatures. Advances in Heat Transfer. 1992;22:157-357. [RCA] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 64] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 51. | Vallez LJ, Plourde BD, Abraham JP. A new computational thermal model for the whole human body: applications to patient warming blankets. Num Heat Transfer A. 2016;69:227-241. [DOI] [Full Text] |
| 52. | Diller KR. Analysis of burns caused by long-term exposure to a heating pad. J Burn Care Rehabil. 1991;12:214-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 53. | Abraham JP, Hennessey MP, Minkowycz WJ. A simple algebraic model to predict burn depth and injury. Int Comm Heat Mass Transfer. 2011;38:1169-1171. [DOI] [Full Text] |
| 54. | Dai W, Wang H, Jordan PM, Mickens RE, Bejan A. A Mathematical Model for Skin Burn Injury Induced by Radiation Heating. Int J Heat Mass Trans. 2008;51; 5497-5510. [DOI] [Full Text] |
| 55. | Johnson NN, Abraham JP, Helgeson ZI, Minkowycz WJ, Sparrow EM. An archive of skin-layer thicknesses and properties and calculations of scald burns with comparisons to experimental observations. J Thermal Sci Engin Appl. 2011;3:011003. [DOI] [Full Text] |
| 56. | Abraham JP, Plourde B, Vallez L, Stark J, Diller KR. Estimating the time and temperature relationship for causation of deep-partial thickness skin burns. Burns. 2015;41:1741-1747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 33] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 57. | Brown F, Diller KR. Calculating the optimum temperature for serving hot beverages. Burns. 2008;34:648-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 48] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 58. | Pipatsattayanutong S, Lee HS, Lau S, O’Mahony M. Hedonic R-Index measurement of temperature preferences for drinking black coffee. J Sensor Studies. 2001;16:517-536. [RCA] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 59. | Borchgrevink CP, Susskind AM, Tarras JT. Consumer preferred hot beverage temperatures. Food Quality and Preference. 1999;10:117-121. [RCA] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 23] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 60. | Lee HS, O’Mahony M. At what temperature do consumers like to drink coffee?: Mixing methods. J Food Science. 2002;67:2774-2777. [RCA] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 46] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 61. | Jamnadas-Khoda B, See MS, Cubison CT, Dheansa BS. How would you like your tea, vicar? Burns. 2010;36:356-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 62. | Ramanathan C, Ekpenyong L, Stevenson JH. Scald burns in children caused by hot drinks--the importance of the type of cup. Burns. 1994;20:111-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 63. | Mercer NS. With or without? A cooling study. Burns Incl Therm Inj. 1988;14:397-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 64. | Warner RM, Wilson Y, Chester DL. Cooling properties of everyday liquids. Burns. 2012;38:1186-1191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 65. | Seidenari S, Giusti G, Bertoni L, Magnoni C, Pellacani G. Thickness and echogenicity of the skin in children as assessed by 20-MHz ultrasound. Dermatology. 2000;201:218-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 81] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 66. | Conti A, Schiavi ME, Seidenari S. Capacitance, transepidermal water loss and causal level of sebum in healthy subjects in relation to site, sex and age. Int J Cosmet Sci. 1995;17:77-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 44] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 67. | Laurent A, Mistretta F, Bottigioli D, Dahel K, Goujon C, Nicolas JF, Hennino A, Laurent PE. Echographic measurement of skin thickness in adults by high frequency ultrasound to assess the appropriate microneedle length for intradermal delivery of vaccines. Vaccine. 2007;25:6423-6430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 160] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 68. | Sandby-Møller J, Poulsen T, Wulf HC. Epidermal thickness at different body sites: relationship to age, gender, pigmentation, blood content, skin type and smoking habits. Acta Derm Venereol. 2003;83:410-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 434] [Cited by in RCA: 446] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 69. | Whitton JT, Everall JD. The thickness of the epidermis. Br J Dermatol. 1973;89:467-476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 150] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 70. | Southwood WF. The thickness of the skin. Plast Reconstr Surg (1946). 1955;15:423-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 109] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 71. | Saffle JR, Davis B, Williams P. Recent outcomes in the treatment of burn injury in the United States: a report from the American Burn Association Patient Registry. J Burn Care Rehabil. 1995;16:219-232; discussion 288-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 224] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 72. | Ahrenholz DH, Clayton MC, Solem LD. Burns and wound management. Otolaryngol Clin North Am. 1995;28:1039-1055. [PubMed] |
| 73. | Davies JW. Prompt cooling of burned areas: a review of benefits and the effector mechanisms. Burns Incl Therm Inj. 1982;9:1-6. [PubMed] |
| 74. | Wiedeman MP, Brigham MP. The effects of cooling on the microvasculature after thermal injury. Microvasc Res. 1971;3:154-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 75. | Yuan J, Wu C, Holland AJ, Harvey JG, Martin HC, La Hei ER, Arbuckle S, Godfrey TC. Assessment of cooling on an acute scald burn injury in a porcine model. J Burn Care Res. 2007;28:514-520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 48] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 76. | Rajan V, Bartlett N, Harvey JG, Martin HC, La Hei ER, Arbuckle S, Godfrey C, Holland AJ. Delayed cooling of an acute scald contact burn injury in a porcine model: is it worthwhile? J Burn Care Res. 2009;30:729-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 77. | Jandera V, Hudson DA, de Wet PM, Innes PM, Rode H. Cooling the burn wound: evaluation of different modalites. Burns. 2000;26:265-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 93] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 78. | Ross DC, Diller KR. Therapeutic effects of postburn cooling. J Biomech Eng. 1978;100:149-152. [RCA] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 79. | Baxter CR, Waeckerle JF. Emergency treatment of burn injury. Ann Emerg Med. 1988;17:1305-1315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 80. | Boykin JV, Eriksson E, Sholley MM, Pittman RN. Cold-water treatment of scald injury and inhibition of histamine-mediated burn edema. J Surg Res. 1981;31:111-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 30] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 81. | Nguyen NL, Gun RT, Sparnon AL, Ryan P. The importance of immediate cooling--a case series of childhood burns in Vietnam. Burns. 2002;28:173-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 78] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 82. | Jakobsson OP, Arturson G. The effect of prompt local cooling on oedema formation in scalded rat paws. Burns Incl Therm Inj. 1985;12:8-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 83. | Ernst E, Fialka V. Ice freezes pain? A review of the clinical effectiveness of analgesic cold therapy. J Pain Symptom Manage. 1994;9:56-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 138] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 84. | Werner MU, Lassen B, Pedersen JL, Kehlet H. Local cooling does not prevent hyperalgesia following burn injury in humans. Pain. 2002;98:297-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 85. | Shulman AG. Ice water as primary treatment of burns. Simple method of emergency treatment of burns to alleviate pain, reduce sequelae, and hasten healing. JAMA. 1960;173:1916-1919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 33] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 86. | Johnson NN, McCaffrey KL, Rose KM, Abraham JP. Cryosurgical treatments for terine fibroids. ASME. 2010;International Congress and Expo, Vancouver, CA, November 12-18, 2010. |
| 87. | Hoffmann NE, Bischof JC. Cryosurgery of normal and tumor tissue in the dorsal skin flap chamber: Part II--injury response. J Biomech Eng. 2001;123:310-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 41] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 88. | He X, Bischof JC. Quantification of temperature and injury response in thermal therapy and cryosurgery. Crit Rev Biomed Eng. 2003;31:355-422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 114] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 89. | Abraham JP, Plourde BD, Stark JR. Cryosurgical treatment of cancer: the importance of modeling. J Cancer Science Ther. 2014;26:124. |
| 90. | Hayes L, Diller KR, Chang HJ, Lee HS. Prediction of local cooling rates and cell survival during freezing of a cylindrical specimen. Cryobiology. 1988;25:67-872. [RCA] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 91. | Litvan GG. Mechanism of cryoinjury in biological systems. Cryobiology. 1972;9:182-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 0.6] [Reference Citation Analysis (0)] |