Published online Mar 18, 2017. doi: 10.5312/wjo.v8.i3.221
Peer-review started: September 1, 2016
First decision: September 29, 2016
Revised: December 2, 2016
Accepted: December 13, 2016
Article in press: December 14, 2016
Published online: March 18, 2017
Processing time: 200 Days and 4.6 Hours
Total loss of talus due to trauma or avascular necrosis, for example, still remains to be a major challenge in foot and ankle surgery with severely limited treatment options. Implantation of a custom made total talar prosthesis has shown promising results so far. Most important factors for long time success are degree of congruence of articular surfaces and ligamentous stability of the ankle. Therefore, our aim was to develop an optimized custom made prosthesis for total talus replacement providing a high level of primary stability. A custom made hemiprosthesis was developed using computed tomography and magnetic resonance imaging data of the affected and contralateral talus considering the principles and technology for the development of the S.T.A.R. prosthesis (Stryker). Additionally, four eyelets for fixation of artificial ligaments were added at the correspondent footprints of the most important ligaments. Two modifications can be provided according to the clinical requirements: A tri-articular hemiprosthesis or a bi-articular hemiprosthesis combined with the tibial component of the S.T.A.R. total ankle replacement system. A feasibility study was performed using a fresh frozen human cadaver. Maximum range of motion of the ankle was measured and ligamentous stability was evaluated by use of standard X-rays after application of varus, valgus or sagittal stress with 150 N. Correct implantation of the prosthesis was technically possible via an anterior approach to the ankle and using standard instruments. Malleolar osteotomies were not required. Maximum ankle dorsiflexion and plantarflexion were measured as 22-0-28 degrees. Maximum anterior displacement of the talus was 6 mm, maximum varus tilt 3 degrees and maximum valgus tilt 2 degrees. Application of an internally braced prosthesis for total talus replacement in humans is technically feasible and might be a reasonable procedure in carefully selected cases with no better alternatives left.
Core tip: Implantation of a total talar prosthesis has shown promising results so far. The aim was to develop an optimized prosthesis providing a high level of primary stability. A custom made hemiprosthesis was developed using computed tomography and magnetic resonance imaging data. Four eyelets for fixation of artificial ligaments were added at the footprints of important ligaments. Correct implantation of the prosthesis in a cadaver model was possible via an anterior approach. Maximum ankle dorsiflexion and plantarflexion were measured as 22-0-28 degrees. Maximum anterior displacement of the talus was 6 mm, maximum varus tilt 3 degrees and maximum valgus tilt 2 degrees.
- Citation: Regauer M, Lange M, Soldan K, Peyerl S, Baumbach S, Böcker W, Polzer H. Development of an internally braced prosthesis for total talus replacement. World J Orthop 2017; 8(3): 221-228
- URL: https://www.wjgnet.com/2218-5836/full/v8/i3/221.htm
- DOI: https://dx.doi.org/10.5312/wjo.v8.i3.221
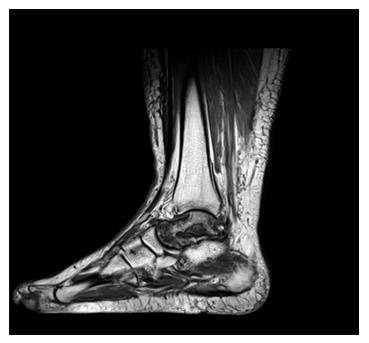
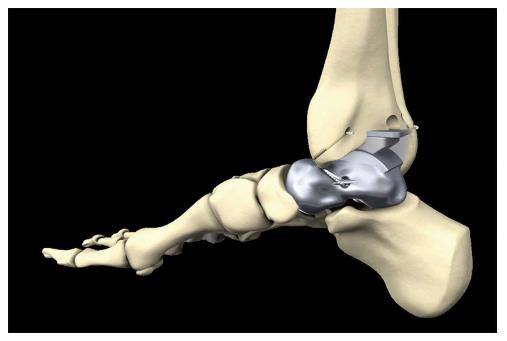
Total loss of talus due to trauma[1-5] or avascular necrosis[6-9], for example, still remains to be a major challenge in foot and ankle surgery with severely limited treatment options[2,4,5,8,9]. Furthermore, collapse of the talar body as a complication of total ankle arthroplasty[10], talectomy in infection and septic talus necrosis[11] or severe bone defects due to tumor resection[12] may result in the need for total talar replacement, especially in younger and active patients (Figure 1).
Because arthrodesis of the ankle or the complete rearfoot as well as tibiocalcaneal fusion after talectomy can produce severe disability of the ankle and the foot, different study groups have already developed a prosthesis to replace the talar body[13-16] or even the complete talus[10,17-20]. Lampert et al[12] and Ketz et al[21] combined a custom total talar prosthesis with the tibial component of a standard total ankle prosthesis, and Giannini et al[22] recently reported on a custom-made total talonavicular replacement in a professional rock-climber.
Implantation of a custom made talar body[13-16] or total talar prosthesis[10,17-20] in humans has already shown promising results so far. In 1997 Harnroongroj et al[16] was the first to report on a series of 16 patients treated by use of a quite primitive talar body prosthesis with has been implanted by a medial transmalleolar approach. Eight of nine patients who were evaluated 11 to 15 years postoperatively had a satisfactory result, which is quite comparable to the results reported after standard total ankle replacement at that period of time. The exceptional patient in this series had an unsatisfactory result because the prosthetic stem had sunk into the talar neck and needed revision surgery 13 years after the index operation[16]. Total talar replacement with a prosthesis was first performed in Japan in 1999. Several subsequent prosthetic design revisions have resulted in improved outcomes after prosthesis implantation[14]. Taniguchi et al[14] reported favorable results in eight of 14 patients after a mean follow-up period of 83 mo using a second-generation prosthesis which only partially replaced the talar body. As mentioned in their report, the third-generation prosthesis completely replacing the talus is currently recommended and has been associated with much better outcomes than the second-generation prosthesis[14]. Tsukamoto et al[10] first reported treatment of talar collapse after total ankle arthroplasty in a patient with rheumatoid arthritis by talar replacement with a third-generation prosthesis. However, this type of prosthesis still had a subtalar stem for fixation to the calcaneus by use of bone cement. Magnan et al[13] extended this procedure using a total talar prosthesis and combined it with the standard S.T.A.R. total ankle arthroplasty system (Waldemar Link, Hamburg, Germany) in a 45-year-old professional male skier and rock-climber. Stevens et al[17] even reported on a 14-year-old girl who underwent total talar replacement after an open talar dislocation. And again it was Harnroongroj to report on the largest series of 33 patients with by far the longest follow-up period of 10-36 years after implantation of a talar body prosthesis[15]. In this series published in 2014, 28 of the 33 prostheses were still in place at the time of final follow-up while five had failed prior to five years.
Advantages of total talar replacement include preservation of joint mobility, a relatively short period of restricted weight bearing, rapid pain relief and preservation of limb length[18]. A native talus is well-seated within the ankle, fitted firmly between the tibia, the fibula, tarsal navicular, and the calcaneus. It has no muscular attachments and over 60% of its surface is covered with articular cartilage[17]. Therefore, stability of the talus or a total talus prosthesis, respectively, depends on the integrity of the main ligaments and on the adjacent bones that build up the ankle mortise and the subtalar joint as well as on the anatomical shape of the talus itself[16].
For example, early prosthesis failure occured as a result of size mismatch in two patients in the large series reported by Harnroongroj et al[15]. And according to Ando et al the procedure of total talar replacement carries at least a theoretical risk of anterior instability of the prosthesis, because the anterior talofibular ligament (ATFL) and deep deltoid ligaments are divided during the procedure[16]. To address this problem of ligamentous stabilization, Stevens proposed the addition of porous coating at the main ligamentous attachment sites of the ATFL and the deltoid ligament to provide improved stability[17].
According to this, most important factors for long time survival of a prosthesis for total talus replacement are degree of congruence of articular surfaces and ligamentous stability of the ankle and subtalar joint.
Therefore, to further improve the idea of total talus replacement, our aim was to develop an optimized custom made prosthesis for total talus replacement providing maximum possible congruence of the articular surfaces and a maximum high level of primary stability immediately after implantation. We introduced the use of preoperative magnetic resonance imaging (MRI) data and an internal bracing technique we had been using successfully for augmentation of severe ligamentous injuries of the foot and ankle since several years[22,23]. The primary aim of this so called InternalBraceTM technique is reconstruction or repair of vital tissue rather than replacement with non-vital tendon transplants. The InternalBraceTM acts as a corner stone or check-rein to stability allowing physiological and limiting pathologic motion. Thereby this method applies the classical AO principles to soft tissues.
A custom made hemiprosthesis was developed considering the relevant anatomical principles and the technology for the development of the S.T.A.R. total ankle prosthesis (Stryker)[24-28].
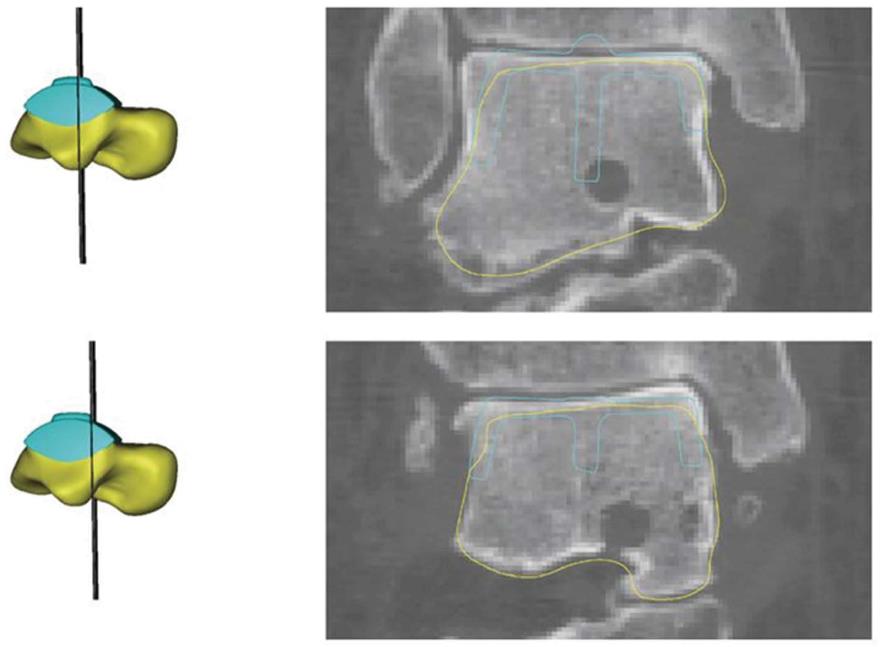
As we believed that a computed tomography (CT) might underestimate the real dimensions of the talus to be replaced, we added an MRI and calculated the mean dimensions of the talus between CT and MRI data to approximate the real dimensions of the talus as accurately as possible (Figures 2 and 3). Most other authors only used X-rays and CT scans for designing the prosthesis[10,15-18].
In cases where the index talus has been severely destructed the prosthesis can be designed alternatively using the mirror image of the CT and MRI data from the contralateral ankle.
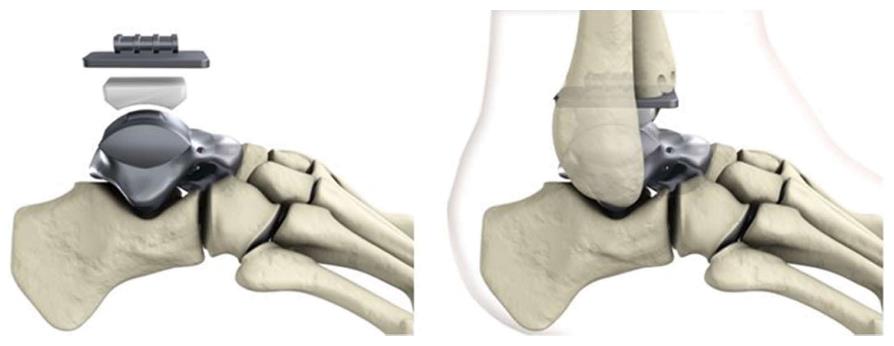
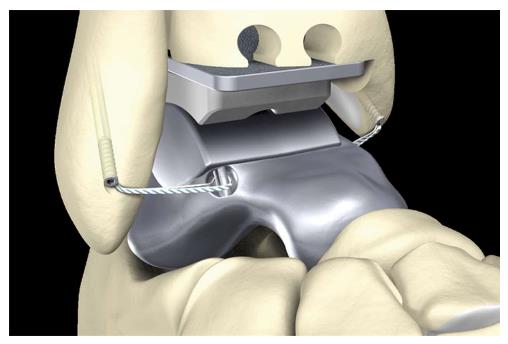
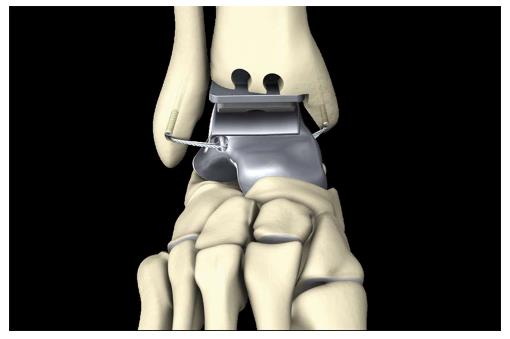
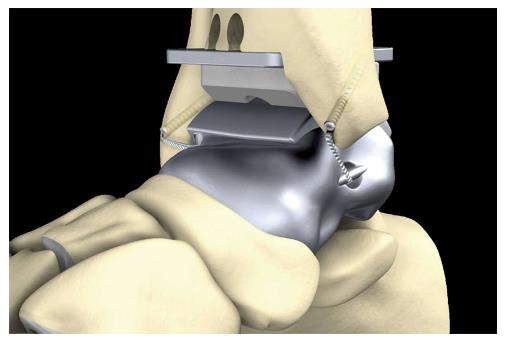
Additionally, four eyelets for fixation of artificial ligaments (Figures 4-8) were added at the correspondent footprints of the ATFL, the deltoid ligament and the interosseous talocalcaneal ligament (ITCL). We used artificial ligaments called FiberTape® (Arthrex, Naples, United States) for performing the InternalBraceTM technique at surgery (Figures 9-13).
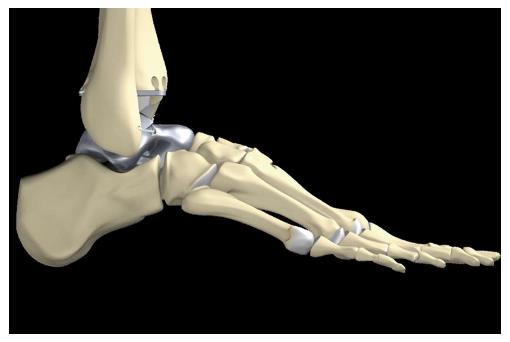
Two modifications can be provided according to the clinical requirements: A tri-articular hemiprosthesis in case of completely intact surrounding articular surfaces or a bi-articular hemiprosthesis combined with the tibial component of the S.T.A.R. total ankle replacement system in case of additional damage to the articular surface of the tibial pilon (Figure 3).
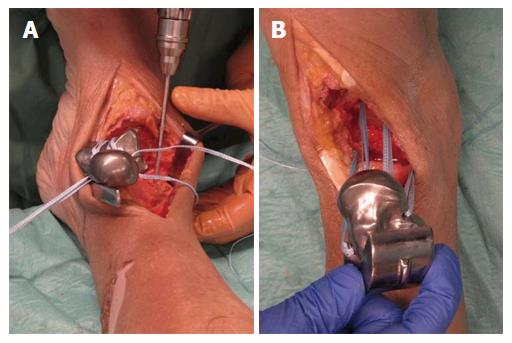
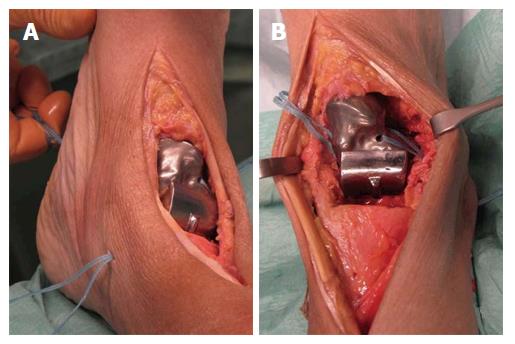
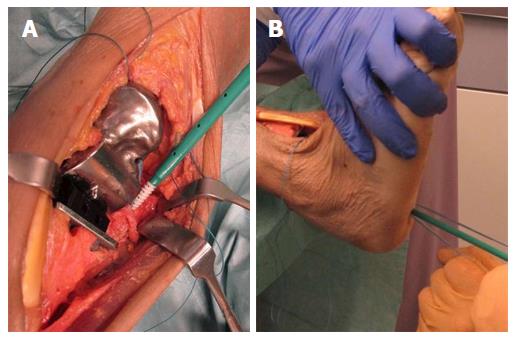
A feasibility study was performed using a 36-year-old male fresh frozen whole leg human cadaver. In the supine position, a straight skin incision was made according to a standard anterior approach to the ankle joint for total ankle arthroplasty (Figure 14). After opening the anterior capsule of the ankle joint, the talus was divided into five main parts by use of a chisel and completely resected. Then the bone tunnels were prepared at the anatomic footprints of the native ligaments for fixation of the artificial ligaments (Figure 14). All four artificial ligaments for replacement of the ATFL, the deltoid ligament and the ITCL were shuttled through the eyelets of the prosthesis and through the bone tunnels before insertion of the prosthesis (Figure 14). Then the prosthesis was inserted from anterior while steady tensioning the four artificial ligaments (Figure 15). Figure 15B shows a view from anterior on the pre-tensioned ATFL.
After insertion of the tibial component and the inlay of the S.T.A.R. total ankle prosthesis we performed an aperture fixation of the correctly pre-tensioned artificial ligaments at the anatomic footprints with an interference screw (SwiveLock® 4.75 mm, Arthrex, Naples, United States). Direct aperture fixation of the artificial ATFL and the deltoid ligament was performed at the distal fibula (Figure 16A) and the medial malleolus, respectively, and retrograde indirect aperture fixation of the ITCL at the calcaneus was performed percutaneously from plantar (Figure 16B).
Correct implantation of the internally braced total talar prosthesis was technically possible via a standard anterior approach to the ankle and using standard instruments. Malleolar osteotomies were not required.
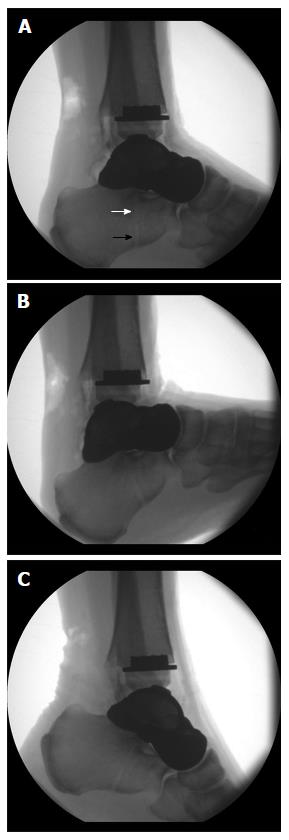
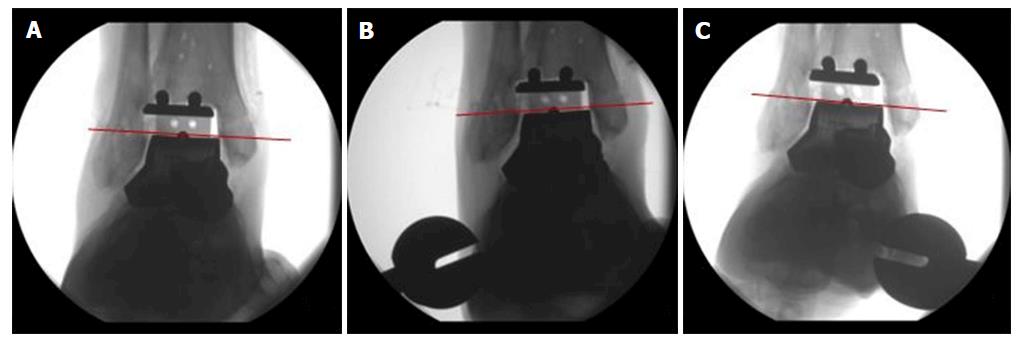
Radiographic examination of the maximum range of motion of the ankle joint after internally bracing of the customized hemiprosthesis was performed. Figure 17 shows neutral position (Figure 17A), maximum dorsiflexion of 22° (Figure 17B), and maximum plantarflexion of 28° (Figure 17C). Note the visible bone tunnel in the calcaneus (black arrow) with the interference screw inside the proximal part of the tunnel (white arrow) to prevent tunnel widening by indirect aperture fixation at the subtalar joint performed percutaneously from plantar.
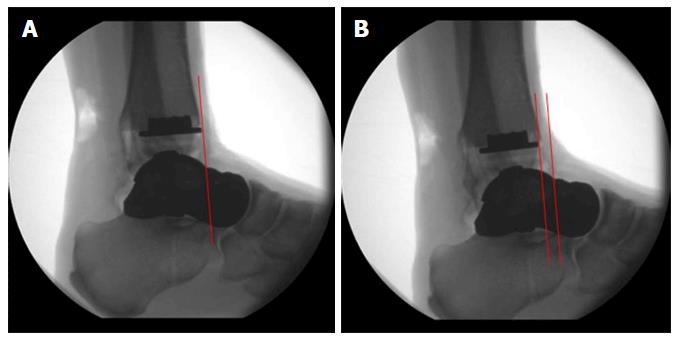
Primary ligamentous stability of the internally braced total talar prosthesis was evaluated by use of standard X-rays after application of varus, valgus or sagittal stress with 150 N: maximum anterior displacement of the talus was 6 mm (Figure 18), maximum varus tilt 3 degrees and maximum valgus tilt 2 degrees (Figure 19) resembling quite physiological ankle function.
Application of an improved internally braced custom made prosthesis for total talus replacement in humans is technically feasible, and first experimental results show a very high primary stability of the implanted prosthesis. Based on our results the described procedure might be a reasonable treatment option in carefully selected cases with no better alternatives left. Due to the quite low frequency of adequate cases, multicentric evaluation seems to be necessary to provide high quality scientific data of outcome results and possible complications after implantation of an internally braced prosthesis for total talus replacement.
Manuscript source: Invited manuscript
Specialty type: Orthopedics
Country of origin: Germany
Peer-review report classification
Grade A (Excellent): A, A
Grade B (Very good): 0
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Mattos BSC, Peng BG S- Editor: Qiu S L- Editor: A E- Editor: Li D
| 1. | Assal M, Stern R. Total extrusion of the talus. A case report. J Bone Joint Surg Am. 2004;86-A:2726-2731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 41] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 2. | Koller H, Assuncao A, Kolb K, Holz U. Reconstructive surgery for complete talus extrusion using the sandwich block arthrodesis: a report of 2 cases. J Foot Ankle Surg. 2007;46:493-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 3. | Lee HS, Chung HW, Suh JS. Total talar extrusion without soft tissue attachments. Clin Orthop Surg. 2014;6:236-241. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 4. | Smith CS, Nork SE, Sangeorzan BJ. The extruded talus: results of reimplantation. J Bone Joint Surg Am. 2006;88:2418-2424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 36] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 5. | Apostle KL, Umran T, Penner MJ. Reimplantation of a totally extruded talus: a case report. J Bone Joint Surg Am. 2010;92:1661-1665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Gross CE, Sershon RA, Frank JM, Easley ME, Holmes GB. Treatment of Osteonecrosis of the Talus. JBJS Rev. 2016; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 7. | Gross CE, Haughom B, Chahal J, Holmes GB. Treatments for avascular necrosis of the talus: a systematic review. Foot Ankle Spec. 2014;7:387-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 8. | Lee KB, Cho SG, Jung ST, Kim MS. Total ankle arthroplasty following revascularization of avascular necrosis of the talar body: two case reports and literature review. Foot Ankle Int. 2008;29:852-858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 9. | Cohen MM, Kazak M. Tibiocalcaneal Arthrodesis With a Porous Tantalum Spacer and Locked Intramedullary Nail for Post-Traumatic Global Avascular Necrosis of the Talus. J Foot Ankle Surg. 2015;54:1172-1177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 10. | Tsukamoto S, Tanaka Y, Maegawa N, Shinohara Y, Taniguchi A, Kumai T, Takakura Y. Total talar replacement following collapse of the talar body as a complication of total ankle arthroplasty: a case report. J Bone Joint Surg Am. 2010;92:2115-2120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 11. | Rzesacz EH, Culemann U, Illgner A, Reilmann H. [Homologous talus replacement after talectomy in infection and septic talus necrosis. Experiences with 3 cases]. Unfallchirurg. 1997;100:497-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 12. | Lampert C. [Ankle joint prosthesis for bone defects]. Orthopade. 2011;40:978-983. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 13. | Magnan B, Facci E, Bartolozzi P. Traumatic loss of the talus treated with a talar body prosthesis and total ankle arthroplasty. A case report. J Bone Joint Surg Am. 2004;86-A:1778-1782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 42] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 14. | Taniguchi A, Takakura Y, Sugimoto K, Hayashi K, Ouchi K, Kumai T, Tanaka Y. The use of a ceramic talar body prosthesis in patients with aseptic necrosis of the talus. J Bone Joint Surg Br. 2012;94:1529-1533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 62] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 15. | Harnroongroj T, Harnroongroj T. The Talar Body Prosthesis: Results at Ten to Thirty-six Years of Follow-up. J Bone Joint Surg Am. 2014;96:1211-1218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 16. | Harnroongroj T, Vanadurongwan V. The talar body prosthesis. J Bone Joint Surg Am. 1997;79:1313-1322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 81] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 17. | Stevens BW, Dolan CM, Anderson JG, Bukrey CD. Custom talar prosthesis after open talar extrusion in a pediatric patient. Foot Ankle Int. 2007;28:933-938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 18. | Ando Y, Yasui T, Isawa K, Tanaka S, Tanaka Y, Takakura Y. Total Talar Replacement for Idiopathic Necrosis of the Talus: A Case Report. J Foot Ankle Surg. 2015;55:1292-1296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 44] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 19. | Taniguchi A, Takakura Y, Tanaka Y, Kurokawa H, Tomiwa K, Matsuda T, Kumai T, Sugimoto K. An Alumina Ceramic Total Talar Prosthesis for Osteonecrosis of the Talus. J Bone Joint Surg Am. 2015;97:1348-1353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 81] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 20. | Angthong C. Anatomic total talar prosthesis replacement surgery and ankle arthroplasty: an early case series in Thailand. Orthop Rev (Pavia). 2014;6:5486. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 21. | Ketz J, Myerson M, Sanders R. The salvage of complex hindfoot problems with use of a custom talar total ankle prosthesis. J Bone Joint Surg Am. 2012;94:1194-1200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 22. | Giannini S, Cadossi M, Mazzotti A, Ramponi L, Belvedere C, Leardini A. Custom-Made Total Talonavicular Replacement in a Professional Rock Climber. J Foot Ankle Surg. 2015;55:1271-1275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 23. | Acevedo J, Vora A. Anatomical reconstruction of the spring ligament complex: “internal brace” augmentation. Foot Ankle Spec. 2013;6:441-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 62] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 24. | Merian M, Glisson RR, Nunley JA. J. Leonard Goldner Award 2010. Ligament balancing for total ankle arthroplasty: an in vitro evaluation of the elongation of the hind- and midfoot ligaments. Foot Ankle Int. 2011;32:S457-S472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 25. | Tochigi Y, Rudert MJ, Brown TD, McIff TE, Saltzman CL. The effect of accuracy of implantation on range of movement of the Scandinavian Total Ankle Replacement. J Bone Joint Surg Br. 2005;87:736-740. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 57] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 26. | Leardini A, O’Connor JJ, Catani F, Giannini S. Mobility of the human ankle and the design of total ankle replacement. Clin Orthop Relat Res. 2004;39-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 53] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 27. | Pellegrini MJ, Glisson RR, Wurm M, Ousema PH, Romash MM, Nunley JA, Easley ME. Systematic Quantification of Stabilizing Effects of Subtalar Joint Soft-Tissue Constraints in a Novel Cadaveric Model. J Bone Joint Surg Am. 2016;98:842-848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 28. | Coetzee JC, Castro MD. Accurate measurement of ankle range of motion after total ankle arthroplasty. Clin Orthop Relat Res. 2004;27-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 93] [Article Influence: 4.4] [Reference Citation Analysis (0)] |