INTRODUCTION
Blood Oxygen Level Dependent functional magnetic resonance imaging (BOLD fMRI) is a brain mapping technique using deoxyhemoglobin contained in the blood vessels as an endogenous contrast agent to produce functional activation maps[1].
Neural activation induces a transient increase in regional oxygen extraction from the blood that is coupled with a much larger increase in cerebral blood flow (CBF) and cerebral blood volume (CBV). This influx of oxygenated hemoglobin results in a net decrease in regional deoxyhemoglobin concentration. This drop in paramagnetic deoxyhemoglobin concentration leads to an increase in the magnetic relaxation times T2 and T2*. The mapping of eloquent areas is thus obtained by acquiring T2 or T2*-weighted images consecutively while the subject is at rest or performing a task, and detecting the signal increase related to the local reduction of deoxyhemoglobin concentration accompanying the functional activation relative to baseline.
fMRI studies are carried out using a gradient echo sequence with echo planar readout[2]. This sequence is very sensitive to the static local magnetic field inhomogeneities and therefore is suitable for detection of T2* related signal changes, and at the same time allows scanning of the whole brain within one repetition time (TR) of 2-3 s. The subject performs a cognitive, sensorimotor or visual paradigm inside the bore of the MRI scanner during image acquisition. In a typical clinical fMRI paradigm, a block design is used consisting of 20-30 s blocks (a.k.a., epochs) of rest or control tasks alternating with blocks of active tasks. In this way the subject’s brain activity is usually monitored for a total duration of 200-300 s by acquiring whole brain MR images with a good spatial (typically 3 mm × 3 mm × 3 mm) and temporal resolution (e.g. 2 s). Four dimensional (3D+time) datasets are created where a signal time series is recorded and stored for each voxel. Functional activation maps are obtained by detecting areas of the brain showing a statistically significant signal increase due to the executed active task relative to the resting or control task.
To achieve this goal, a series of processing steps needs to be carried out on the fMRI dataset[3]. First the raw images are initially time shifted so that all the slices in each whole brain acquisition (volume) that occur in one TR result as if they were acquired at the same time. Then all the volumes are registered to a reference volume to correct for small head motions. A further preprocessing step consists of spatially smoothing each voxel signal time series in order to reduce low and high frequency noise.
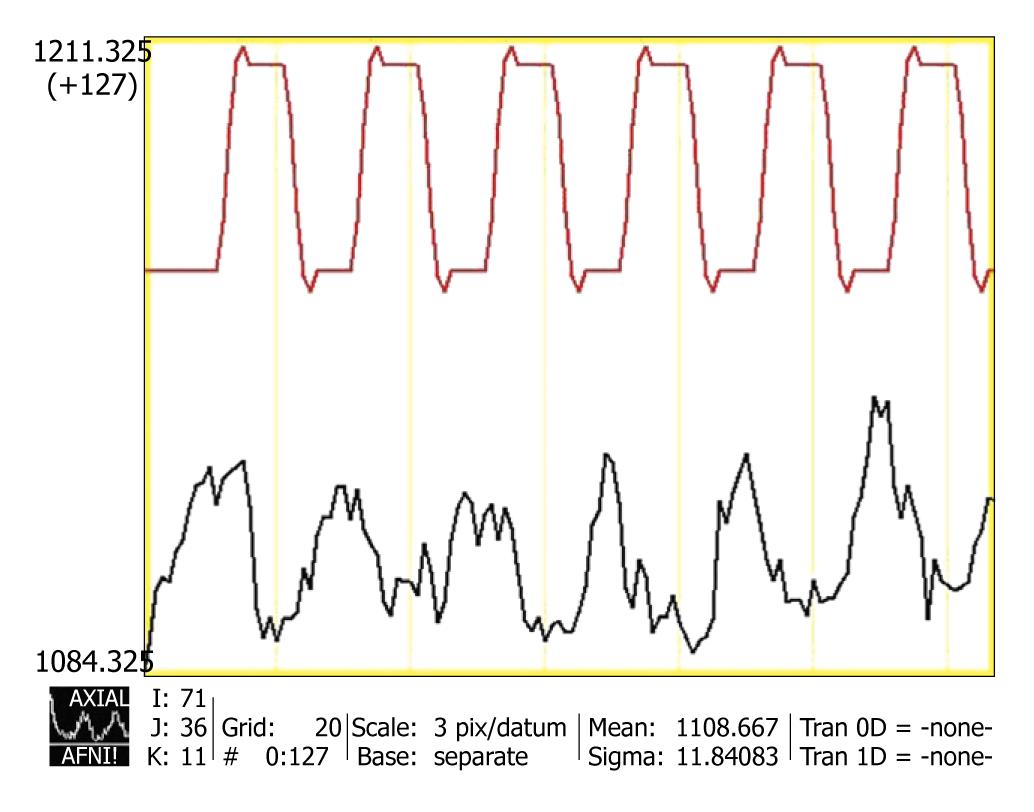
At this point a statistical analysis is carried out that aims to determine, voxel by voxel, the “goodness of the fit” of the signal time series to a theoretical hemodynamic response function obtained by convolving the paradigm timing with an impulse response function (Figure 1). The “goodness of the fit” can be expressed through several statistical parameters, such as P-value, Z or t score or cross correlation coefficient.
Figure 1 Magnetic resonance signal time series (black line) in an activated voxel.
Subject was engaged in a language phonemic task over a time period of 260 s. The red line curve represents the ideal hemodynamic response function the voxel time series was fitted to. Very good correlation between the two curves can be easily seen. AFNI software (afni.nimh.nih.gov) was used for data processing.
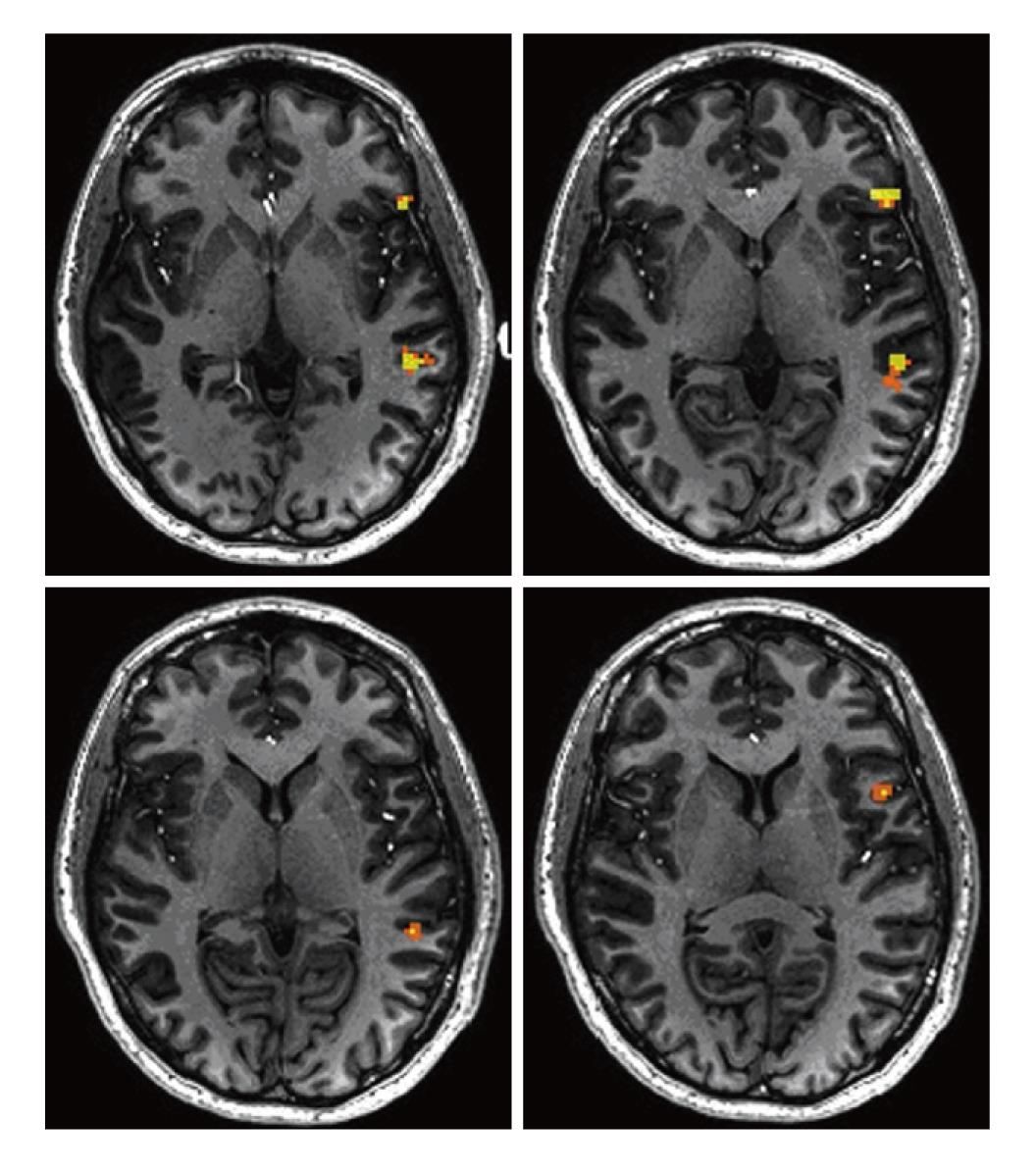
Activation maps are generated choosing a threshold (significance) on the statistical score. The suprathreshold regions are visualized as “hot spots,” often overlaid in color and coregistered on a higher resolution anatomical MR image (Figure 2).
Figure 2 Activation (colored voxels) in the Broca’s and Wernicke’s areas in a normal subject performing a phonemic fluency task.
The t-score map was thresholded at 4.0 value (P < 0.001) and superimposed on a high resolution T1 weighted MPRAGE image. AFNI software (afni.nimh.nih.gov) was used for data processing.
The neurobehavioral paradigms used for functional MRI studies can be divided into two categories, block-design and event-related design. Block design paradigms, which are more commonly used clinically, utilize consistent and repeated blocks of stimuli (active task) and rest (control task), often of the same duration.
In the event-related paradigm design single events are used as stimuli instead of epochs of consecutively administered multiple stimuli. Each trial is considered separately as being time locked to the beginning of the stimulus, and signal changes are explored in relation to the onset of the event generated by the trial.
Block design paradigms provide higher sensitivity for detection of statistically significant signal changes between the control and active conditions as well as allow for better patient compliance, and for these reasons are generally preferred for clinical fMRI studies[4].
Introduced in the early nineties by Ogawa et al[5], BOLD fMRI has become an extensively used imaging technique in the neuroscience community.
BOLD fMRI has been applied to the study of a broad spectrum of brain functions from simple motor and visual activities to complex language, memory and emotion and even higher-level reasoning tasks such as abstract mathematical reasoning[6].
In the last decade it has made the transition from a purely research imaging technique to a viable clinical technique used primarily for presurgical planning in patients with brain tumors and other resectable brain lesions.
CLINICAL FMRI FOR PRESURGICAL PLANNING
The goal of surgical treatment of brain lesions is the maximization of lesion resection with simultaneous minimization of damage to eloquent surrounding normal brain tissue in order to minimize the risk of postoperative neurological deficits[7,8]. Several studies have demonstrated that patient survival and quality of life improve with the extent of resection, provided that there is an absence of surgically-induced permanent neurologic deficits[9]. For this reason it is of paramount importance to identify the eloquent cortical areas within the lesions or in close spatial proximity to its borders that are at risk of being damaged during surgery, as well as to avoid the interruption of “eloquent” white matter fiber tracts that can also lead to loss of neurological function. These areas are usually identified using more invasive brain mapping techniques, such as the gold standard ECS (intraoperative electrocortical stimulation mapping) for localization of critical functional areas during awake or asleep craniotomy, the implantation of a subdural grid for brain mapping using electrocorticography[10], the recording of intraoperative somatosensory or motor evoked potentials. The somewhat less invasive Wada test used for the identification of the dominant language hemisphere (but not eloquent cortical localization) is also not without risks. In contrast, BOLD fMRI is performed preoperatively, is completely non-invasive and safe, allows for whole brain mapping and in conjunction with high resolution MR structural images can depict with accuracy the spatial relationship between the margin of a lesion and functionally viable brain tissue.
BOLD fMRI for presurgical planning provides the neurosurgeon with a valuable tool because it allows for accurate assessment of risk of postoperative motor, cognitive (including language), visual and somatosensory deficits, which is very useful for both surgical decision-making with respect to extent of resection and informed discussions with patients regarding risks and benefits of surgical resection[11]. Furthermore, in cases for which a decision in favor of resection has been made, BOLD fMRI can determine the safest surgical trajectory to the lesion. Preoperative fMRI activation maps can guide the intraoperative cortical stimulation (ICS) mapping, reducing the total surgical time and associated risk of morbidity related to invasive mapping as reported in the literature[12,13].
Performance of clinical fMRI studies in patients suffering from brain tumors or other resectable brain structural lesions, who are often neurologically impaired, requires special considerations with respect to patient preparation, neurobehavioral evaluation and paradigm selection, which would not be necessary in normal volunteer research fMRI studies.
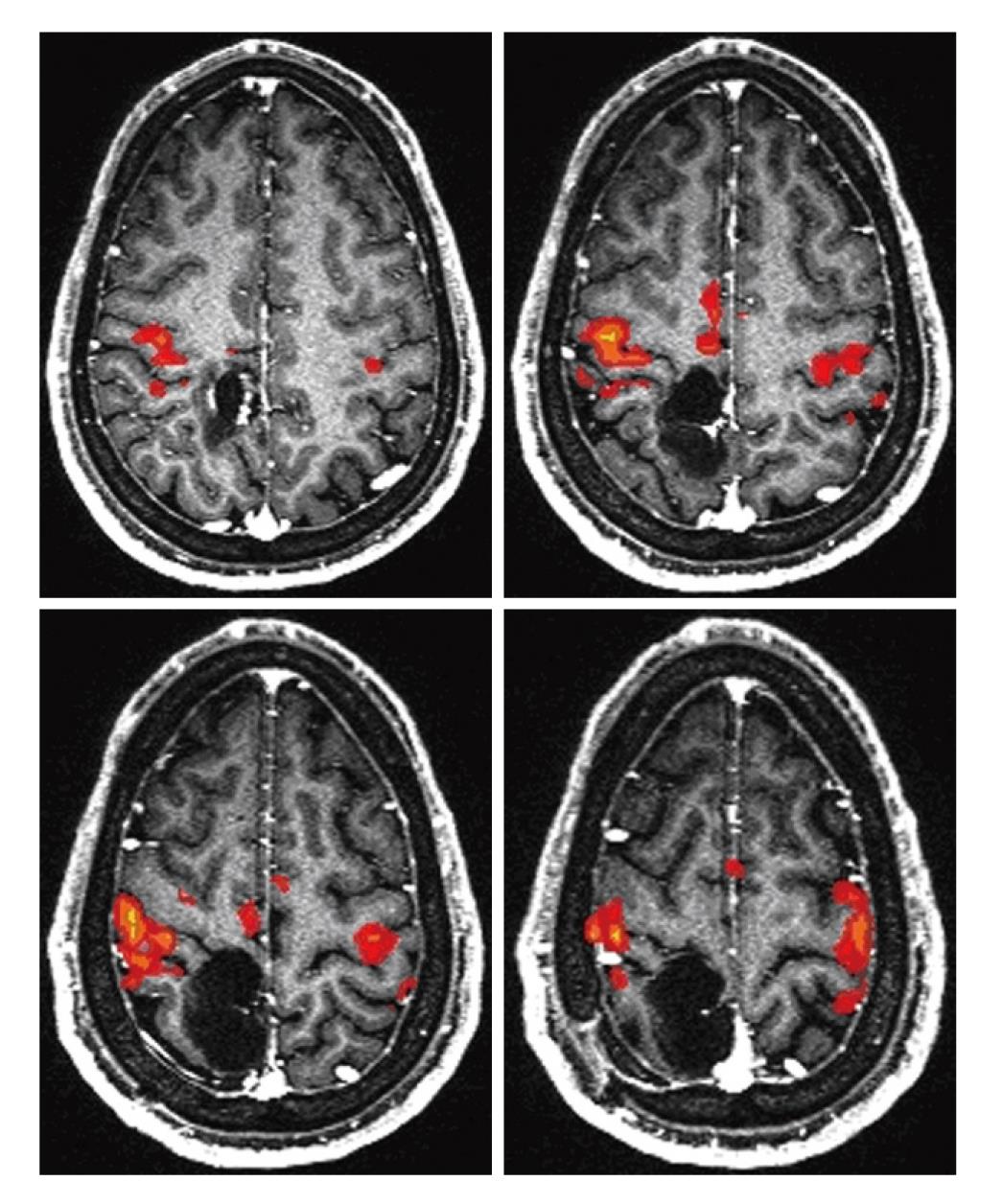
The choice of paradigms depends mainly on the location of the lesion and the patient’s ability to adequately perform individual tasks[14]. For example if a brain tumor is adjacent to the inferior frontal gyrus, one or more expressive language tasks should be performed by the patient in order to determine the proximity of functional Broca’s area to the lesion. Similarly, if the lesion is perirolandic in location, tasks that are known to reproducibly activate the primary motor cortex (PMC) should be used (Figure 3). In both cases, the exact task (s) chosen will depend on the patient’s ability to adequately perform each of a battery of available tasks in each category, as assessed during a prescan training session.
Figure 3 Hand Motor mapping in a patient with a lesion located in the right perirolandic region.
Images were acquired on a 3 T magnetic resonance imaging scanner. AFNI software (afni.nimh.nih.gov) was used for data processing.
A combination of neuroradiologist review of available prior structural brain MR imaging studies and prescan interview by a neuroradiologist or other physician (through which medical/neurological history is obtained and gross functional deficits are determined) determines the battery of likely useful BOLD fMRI paradigms for a particular patient. Once this battery has been selected, it is necessary to perform a prescan training session in which the patient is instructed regarding each task and asked to practice each task to objectively assess performance capability and assess for subtle cognitive or sensorimotor deficits that might impair task performance.
Currently, clinical BOLD fMRI for presurgical planning is used to map the motor, language and the visual eloquent cortex utilizing a variety of well-documented paradigms from the fMRI literature and multicenter clinical experience that have demonstrated consistent efficacy in mapping eloquent cortex[15]. Below a brief description of the most commonly used paradigms for presurgical mapping by fMRI is reported.
SENSORIMOTOR PARADIGMS
Paradigms to map the sensorimotor areas have been used since the first days of clinical fMRI. Most patients are able to perform these tasks, and these tasks do not require special equipment. These paradigms are used during a clinical fMRI exam when brain lesions are in proximity to the PMC. They are usually implemented with a simple block design in which blocks of complete absence of any body movement alternate with blocks of continuous movement of the specific part of the body whose cortical representation area needs to be determined. A unilateral or bilateral finger tapping task is most often used for the localization of the PMC because the hand representation area comprises a large portion of the motor homunculus and yields a robust BOLD signal[16]. These paradigms also yield robust activation of the supplementary motor area (SMA) and the premotor cortex. A hand grasp task can also be run for the same purpose. Furthermore, if the lesion is anatomically located more superomedially along the PMC in a parasagittal or midline convexity location, a toe or ankle movement task is needed in order to determine the foot representation area of the PMC[17], whereas if the lesion extends more inferiorly, involving the frontal opercular region, a tongue or lip movement task can be performed to localize the face representation area of the PMC. It is worth mentioning that these motor paradigms also elicit activation in the ipsilateral cerebellar hemisphere.
LANGUAGE PARADIGMS
The main goals of language presurgical mapping by fMRI are to provide the neurosurgeon with information about cerebral hemispheric language lateralization as well as precise localization of critical eloquent language cortex with respect to the margins of potentially resectable brain lesions. A large number of effective research-level language activation paradigms have been reported in the literature, and given the variety of approaches used for fMRI language mapping across different institutions, there still remains a need for standardization for clinical use[18]. However, we will describe in this article a series of paradigms that have been frequently used in the clinical setting.
Phonemic verbal fluency or phonological tasks are administered mainly with the purpose of localizing Broca’s area in the frontal lobe which is responsible for speech production. Silent Word Generation or Verb Generation are two tasks well documented in the clinical fMRI literature that activate expected expressive language cortex including the dorso-lateral prefrontal cortex, inferior frontal gyrus, variably cingulate language regions, SMA, premotor and motor regions[19]. In performing these tasks patients are asked to produce nouns or verbs associated, respectively, with a presented letter or noun. Control tasks for these paradigms usually consist of simple fixation. The Rhyming paradigm is an interesting example of a dual choice phonological processing task[20]. During the active block of this task, pairs of words are displayed visually and patients are required to press a button on a response pad if the words rhyme, whereas during the control block two rows of stick figures are shown and patients are required to press the button if the two rows match. The verbal fluency and rhyming tasks have also been demonstrated to effectively lateralize hemispheric language function[21,22].
Another expressive task often used both for presurgical fMRI and intraoperative mapping is object naming, which requires the patient to name presented pictures in blocks alternating with periods of fixation. This task is a poor lateralizing task because of its tendency to activate nonessential as well as essential eloquent cortex, often in a bihemispheric pattern representative of the global language network.
Receptive paradigms are useful to localize Wernicke’s area (posterior aspect of the superior temporal gyrus) and other eloquent cortical areas in the dominant temporal-parietal region such as supramarginal gyrus, angular gyrus, inferior temporal gyrus and middle temporal gyrus. These paradigms generally include language comprehension tasks such as sentence reading or listening comprehension and can be designed to be forced choice paradigms in which patient performance can be monitored using recordings of button presses on a response keypad. Another very useful receptive language task is a passive story listening task, in which patients are asked simply to passively listen to blocks of garbled speech alternating with blocks of meaningful stories[23].
Semantic paradigms constitute another category of language tasks. They are designed to be useful for language lateralization and localization of inferior frontal as well as temporoparietal language areas in the dominant cerebral hemisphere. Typical semantic paradigms require the association between a noun and a category (e.g. dog-animal) or a noun and a verb (e.g. dog-bark)[20]. These paradigms are also designed to be forced choice in order to monitor patients response. During the control block, patients are engaged in dual choice tasks not involving language processing.
VISUAL PARADIGMS
The most efficient and complete method to map the visual cortex is to use expanding (or contracting) checkered rings to activate retinal locations at successively greater eccentricities and to use a rotating checkered wedge or hemifield to map successive angular positions[24].
In doing so, each quadrant of the visual field can be identified and the central vision can be distinguished from the peripheral vision, the latter being essential for reading.
Neurons responding preferentially to visual stimulation at different locations in the visual field are activated at different times during each of the stimulus sequences. Activation maps can be displayed color coding the delay in activation relative to each quadrant of the visual field and/or the relative eccentricities. Many other visual stimulation tasks can be used to simply activate the primary visual cortex without providing sensitive retinotopic mapping; most of these tasks incorporate some kind of flashing checkerboard-type stimulus.
CLINICAL VALIDATION AND SURGICAL IMPACT
The early era of clinical functional imaging validation studies were conducted comparing the results of preoperative fMRI with those obtained using intraoperative ‘gold standard’ brain mapping techniques. Excellent concordance has been reported between the pattern of activation revealed by fMRI and ICS mapping for motor mapping[25-30]. A high degree (between 80% and 100% in most series) of concordance between these two techniques has also been found for language mapping[31-33]. For example, a recent study by Bizzi and colleagues found 83% sensitivity and 82% specificity for BOLD imaging localization when compared to ICS[34]. Moreover, several studies have demonstrated the effectiveness of fMRI for determining language lateralization because of the agreement (90%-100% in most series) between the results of fMRI and the Wada test or neuropsychological testing[31].
BOLD fMRI can have a remarkable impact on surgical management of brain tumor patients. Petrella and colleagues demonstrated how the results of presurgical fMRI altered the surgical treatment plan in 19 out of 39 patients (P < 0.05), allowing in most cases a more aggressive approach than that which would have been taken without access to the results of fMRI, and surgical time was also reduced in the majority of cases[12]. A similar study by Medina et al[13] suggested an impact of preoperative fMRI on not just the overall surgical plan but specifically the intraoperative mapping plan in 52% of cases. fMRI also eliminated the need for performance of further preoperative diagnostic studies, including the Wada test. In another study the positive impact of fMRI on patient outcome was demonstrated in a group of 22 patients who underwent preoperative motor fMRI; only 6 experienced mild postoperative neurological deficits but they all fully recovered within 3 mo[35].
BOLD fMRI offers several advantages compared to ICS. However, these two techniques play a complementary role because of their different nature and the respective limitations. BOLD fMRI is a positive activation technique, whereas ICS allows more specific detection of only critical, but not nonessential participatory, eloquent cortical regions by inducing the arrest or disruption of normal function. Thus, fMRI is more capable of depicting the global language network at the expense of producing ‘false positives’ with respect to essential eloquent cortical mapping when compared to ICS, which only indicates such essential functional cortex[36].
LIMITATIONS OF BOLD FMRI AND ROLE OF BOLD CEREBROVASCULAR REACTIVITY MAPPING
The significance of neurovascular uncoupling
Activation maps obtained by BOLD fMRI are based on the coupling between neuronal firing and hemodynamic changes such as increased blood flow, blood volume and oxygenation occuring in the vasculature adjacent to activation areas. However, in many brain diseases the neurovascular coupling has been reported to be altered. Examples include, but are not limited to carotid occlusion, transient global ischemia, penumbra of cerebral ischemia, subarachnoid hemorrhage, epilepsy and Alzheimer’s disease (AD)[37-42]. In brain tumors this relation is often severely altered because it has been demonstrated both in angiographic as well as in MRI studies that tumor vasculature responds much less vigorously to physiological stimuli than vessels in normal cerebral cortex[43-45]. The inability of such aberrant microvasculature to display a normal BOLD response to neural stimulation is referred to as neurovascular uncoupling (NVU). Hence, in the setting of NVU, false negatives can be seen on activation maps, thus limiting the ability of fMRI to adequately define areas of eloquent cortex in the vicinity of or actually within a lesion where loss of normal cerebrovascular autoregulation occurs. It is not uncommon to observe eloquent cortex in regions of impaired cerebrovascular reactivity (CVR), and multiple published studies have reported unreliable results of presurgical mapping, both in terms of reduced activation measured in the hemisphere ipsilateral to the tumor and in terms of incorrect assessment of language dominance, which can be attributed to NVU[45-47]. Decreased BOLD fMRI activation volumes in the tumor ipsilateral hemisphere have also been shown to correlate with increased rCBV in patients with high grade gliomas demonstrating the loss of autoregulatory capability in tumor neovasculature[48]. This is a critical issue in presurgical mapping using BOLD fMRI, since false negative activation may result in inadvertent surgical resection of functional cortex, with resultant permanent postoperative neurological deficits. Since the aim of neurosurgical intervention is the maximization of resection of diseased (e.g. tumor or epileptogenic zone) brain tissue with simultaneous maximal preservation of eloquent cortex, surgical decision-making based on preoperative fMRI activation maps relies heavily on the absence of false negative activation. NVU potential must, therefore, be adequately assessed in order to provide accurate activation maps for surgical planning purposes.
CVR mapping
Since the relaxation of arterial smooth muscle, which strongly depends on the blood CO2 partial pressure (PCO2), is thought to be responsible for maintaining the blood flow in the microcirculation distal to large feeding vessels, the cerebrovascular autoregulation can be investigated by manipulating the PCO2 level in the blood. The CVR is then defined as the change in blood flow per unit change in PCO2.
CVR can be measured by different imaging modalities, including Transcranial Doppler, Single Photon Emission Computed Tomography, Positron Emission Tomography and MRI. Early imaging-based measurements of CVR detected the magnitude of blood flow change in response to the administration of a contrast agent[49-51].
However, MRI has become the preferred choice because of its noninvasiveness and good spatial resolution. Furthermore, qualitative and quantitative CVR measurements can be obtained by using BOLD MR sequences identical to the ones used in functional MRI that do not require use of exogenous contrast agents.
As mentioned in the introduction of this review, the BOLD contrast arises from the decrease of deoxyhemoglobin (dHb) resulting from neuronal activation. Since increases in PCO2 cause dilation of cerebral blood vessels without increasing the metabolic rate of brain parenchyma, and changes the dHb concentration in the cerebral vasculature, BOLD MR imaging can be used to test vascular reactivity following a hypercapnia challenge[52].
Hypercapnia can be induced by various approaches. In general, these methods can be categorized into two types[53-61]: breath-hold (BH), and inhalation of CO2-manipulated air. For the first category, the subject is instructed via visual or auditory cues to hold his breath for a short period (normally 10-20 s), which is interleaved with normal breathing periods for a total paradigm duration of approximately 4-5 min. The best reproducibility has also been reported for a breath hold task of 15-20 s duration compared to longer and shorter periods[62]. Data are pre-processed similar to BOLD functional imaging data, and maps of percentage signal change or cross correlation with an expected hemodynamic response function are provided for assessment of CVR. The typical hemodynamic response used for neuronal activity-based signal change can be used to model BH related signal change; however, a more accurate model has recently been developed accounting for a slower signal change for BOLD-related respiratory response[63]. The hypercapnia condition is established a few seconds into the BH period, and increased BOLD signal can be detected in normal brain regions. After BH, the subject quickly recovers to normocapnia during the normal breathing period. A brief inspiration or expiration is commonly performed before the start of BH. In general, there is no significant advantage found for inspiration or expiration BH techniques[64]. It has been suggested that inspiration BH is preferable for easier patient tolerance and larger BOLD signal change[64]. Nevertheless, at ultra-high field, it is found that altered oxygen concentration in the nasal cavity during end-inspiration may result in artifactual signal changes, which confound the interpretation of the CVR measurement[65]. The main advantage of the BH method is that it does not require sophisticated equipment and can be easily performed during a routine clinical MR imaging session, but the main drawback of this method is the impossibility of quantitation of CVR using this approach.
The second category of methods involves administration of air with altered CO2 or O2 levels[55,57,59-61]. The main advantage of this approach is that it allows precise control of the content of inhaled air, and end tidal (ET) CO2 can be monitored during the experiments, which furnishes a quantitative measure of the induced hypercapnia condition that can enable a quantitative assessment of CVR. In addition, the detected BOLD signal is less likely to be contaminated by BOLD signal changes related to neuronal activation secondary to visual or auditory processing that would typically affect BH-based methods. On the other hand, it normally takes longer to build up a hypercapnia condition that produces sufficient BOLD signal changes with these types of stimuli. The equipment setup time and patient training needed for use of such gas delivery systems could be relatively problematic for routine clinical use, especially with debilitated patients, but a few groups have proposed and built specialized systems for clinical applications[55,57]. These methods typically require the use of a facial mask and the setup of experimental apparatus to control the CO2 delivery, but it offers the advantage that by continously monitoring the change of ET CO2 level in the blood, quantitative CVR measurements can be performed[66]. Protocols to detect CVR by a CO2 inhalation task usually include the administration through a mask of air and a CO2/air mixture for induction of artificial hypercapnia, with CO2 concentration varying from 5% to 10%[67-69]. The ET-CO2 levels are monitored by using infrared devices. The paradigm design for alternate delivery of air and gas mixture is similar to the block design used to study brain activation. Still there is lack of standardization of epoch duration; however, a recent study by Yezhuvath et al[57] demonstrated that a paradigm with air breathing for 1 min interleaved with 1 min CO2 breathing, repeated three times is sensitive enough for CVR measurement because the results were not significantly different from the ones obtained with longer epochs of air and CO2 breathing. More importantly, a recent study has suggested that inspiration of 4% CO2, 21% O2 and balanced N2 is the preferred method to induce hypercapnia, because BH and inhalation of CO2/O2 mixture without N2 could elicit unwanted metabolic activities in the brain[55].
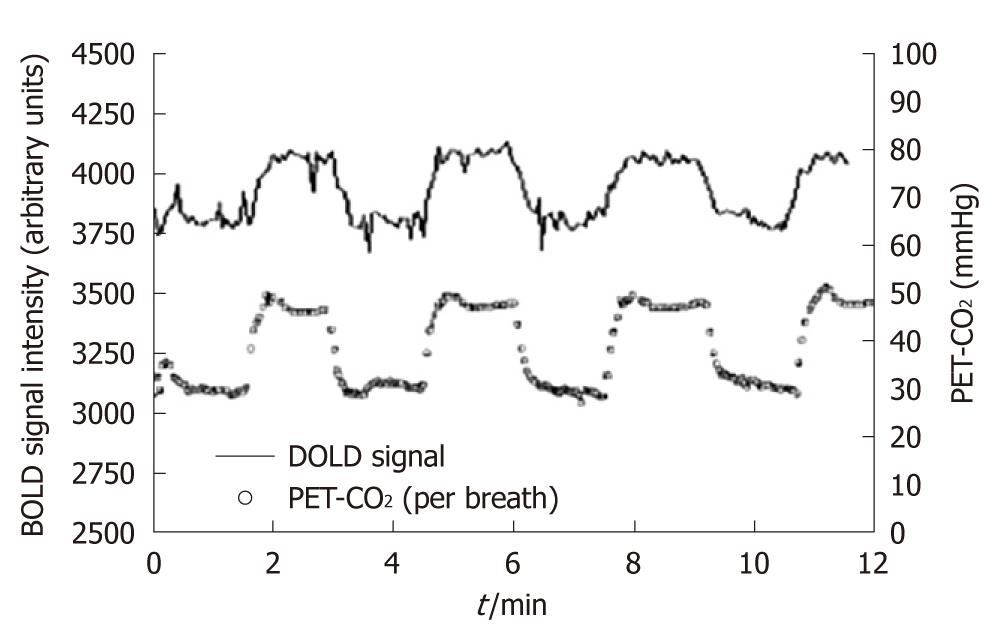
Following standard BOLD fMRI preprocessing steps and correcting for dephasing between the BOLD signal and the recorded pressure of end-tidal CO2 (PET-CO2), signal quantitative CVR maps are computed voxel by voxel by the ratio between MR signal change due to CO2 inhalation over the related pCO2 changes and are expressed in units of percentage MR signal change/mmHg CO2 (Figure 4). The rationale for this definition comes from the proportionality between the BOLD signal and CBF, as well as the definition of CVR as the change in CBF per unit change in PCO2. However, the simple administration through a mask of air and CO2 is more likely to provide semiquantitative rather than purely quantitative CVR maps. It is, indeed, necessary to control and modify the CO2 level rapidly and precisely in order to gain the required square wave changes in PET-CO2, and when patients are simply inhaling CO2 through a mask they hyperventilate in order to blow off the CO2. For this reason, long experimental times are also needed to reach a steady PET-CO2 level before hypercapnia measurements can be effectively performed.
Figure 4 Example of cerebrovascular reactivity measurement using administration of air with altered CO2 or O2 levels: after time shifting the Blood Oxygen Level Dependent signal pattern closely follows pressure of end-tidal CO2 (end tidal pressure of carbon dioxide) pattern (from Vesely et al[69]).
Blood Oxygen Level Dependent magnetic resonance imaging and pressure of end-tidal CO2 (PET-CO2) signals are reported in arbitrary units. BOLD: Blood Oxygen Level Dependent.
In order to overcome the above-mentioned problems relating to standard, a special device has recently been developed that offers some advantages towards a more accurate measurement of CVR[70]. This device consists of a rebreathing circuit that rapidly changes the PCO2 between two steady levels keeping constant the ET oxygen levels (ETO2) during BOLD image acquisition[69]. In this way, a controlled reproducible physiologic stimulus is applied that allows truly quantitative CVR measurement. Experimental applications of this tool have been carried out in patients affected with Moyamoya and steno-occlusive diseases and the results have been validated by comparison with different imaging modalities[55,71,72]. However, to date, no such applications of such a device have been performed in brain tumor patients, and concerns have been raised regarding such patients’ ability to tolerate such methods.
CLINICAL EXAMPLE OF NVU DETECTED WITH BH CVR MAPPING
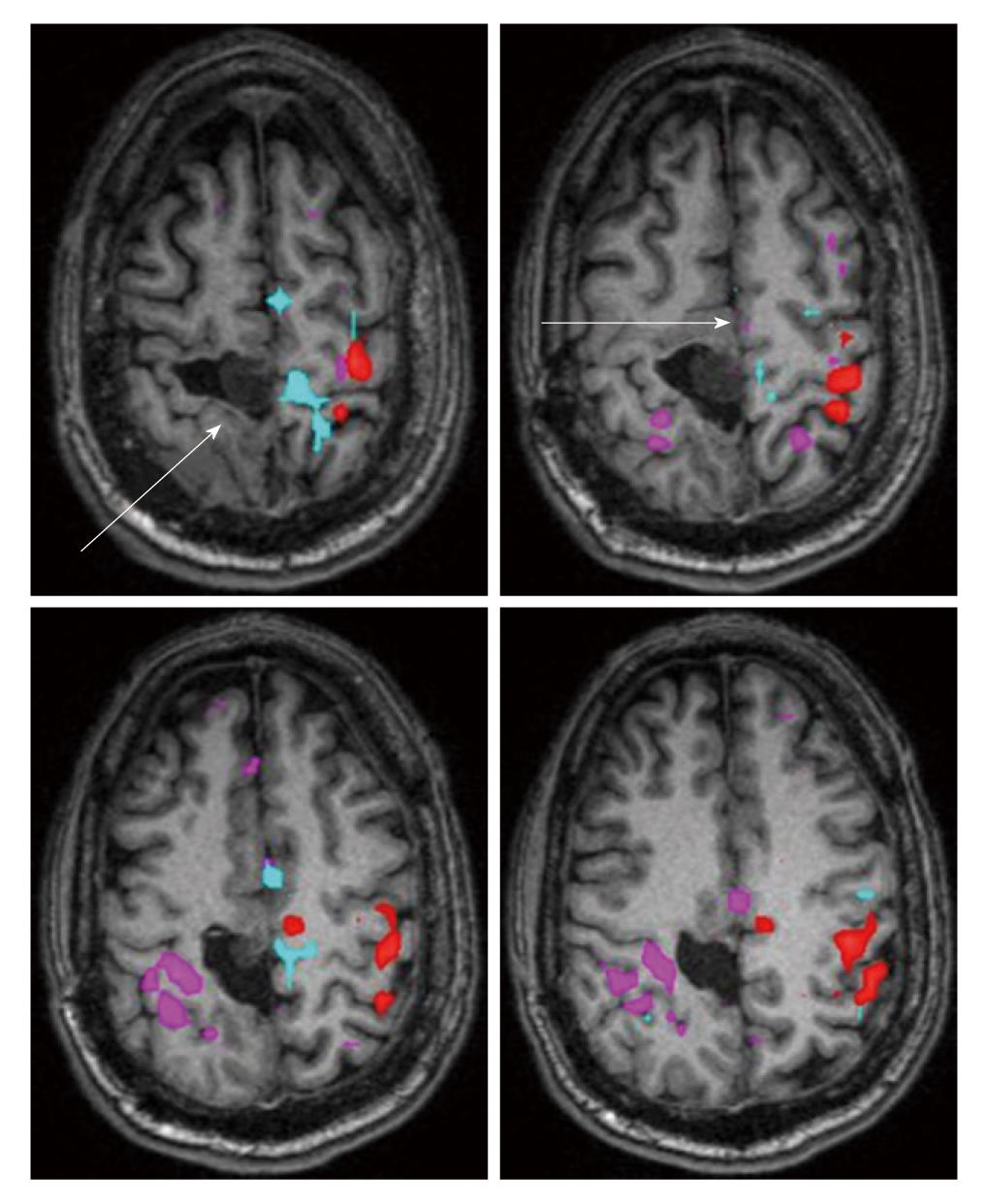
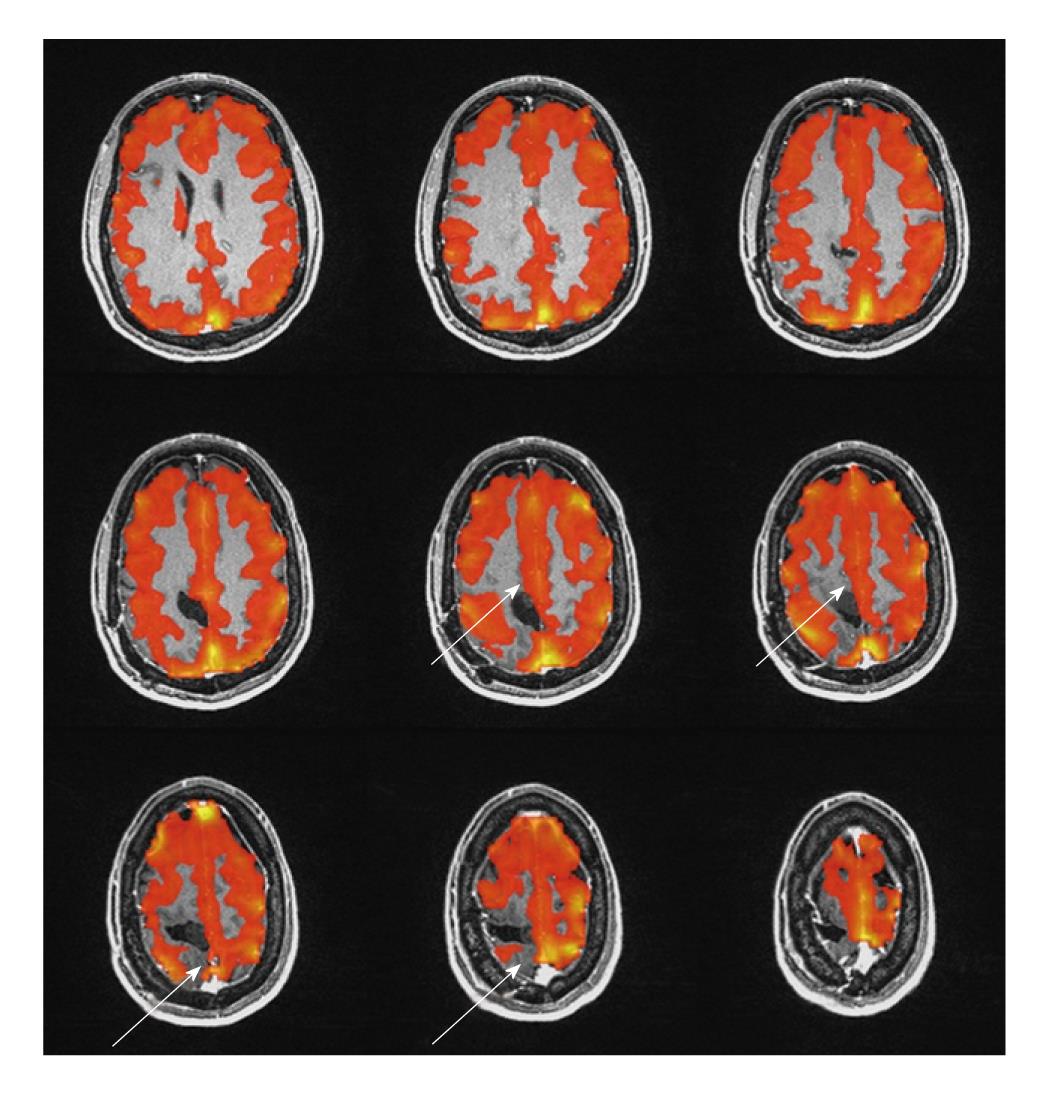
A 39-year-old male with previous surgical resection of a grade II well differentiated diffuse astrocytoma presented with tumor recurrence at the surgical bed. As shown on the axial precontrast T2 FLAIR and postcontrast T1 weighted 3D MPRAGE images, a nonenhancing, FLAIR hyperintense nodular mass is seen along the medial aspect of the surgical resection cavity. The patient presented with distal left lower extremity weakness, with specifically left foot weakness with respect to plantar flexion and extension, but did not demonstrate any upper extremity or facial weakness. Presurgical BOLD fMRI demonstrates expected activation in the hand representation area of the PMC, but absent activation is seen in the foot representation area of the PMC more superomedially (Figure 5). The corresponding BH CVR map demonstrates abnormally reduced regional CVR in this area, suggesting NVU and high risk of false negative activation (Figure 6). In this case, susceptibility artifact from the previous surgery is also likely to be at least partially contributory, but NVU likely represents the main reason for false negative activation. Given the presence of intact motor strength in the left lower extremity including some degree of preserved mobility of the foot, this does not represent true negative activation due to complete absence of eloquent cortex and resultant complete loss of function, but rather represents false negative activation; although functional cortex remains, it is impossible to elicit BOLD activation in the foot representation area of the PMC due to NVU. Based on this detection of NVU, a decision was made by the neurosurgeon to approach the resection of the recurrent lesion cautiously and to perform intraoperative cortical stimulation mapping for adequate determination of the location of the foot representation area of the PMC. The activation maps were useful, however, to delineate the hand representation area, and this did affect the extent of craniotomy needed for adequate intraoperative mapping.
Figure 5 Color coding of Blood Oxygen Level Dependent activation map, overlaid on noncontrast T1-weighted 3D MPRAGE axial images, is as follows: magenta refers to left hand motor task activation (hand opening/closing), thresholded at a T value of 4.
6, while red refers to the right hand motor task, thresholded at a T value of 4.5, and cyan/light blue refers to bilateral foot motor task (toe flexion/extension), thresholded at a clustered cross-correlation value of 0.50. Note the striking absence of any activation on the foot motor task in the foot representation area of the right (ipsilesional) primary motor cortex as well as absence of activation in the superior frontal gyrus (supplementary motor area), while the contralateral homologous regions demonstrate expected activation, as seen color-coded in cyan.
Figure 6 Blood Oxygen Level Dependent breath-hold cerebrovascular reactivity map, overlaid on postcontrast 3D MPRAGE anatomic axial images.
The Blood Oxygen Level Dependent cerebrovascular reactivity (CVR) map was thresholded at positive 0.25% signal change. Note the absence of CVR just anterior and posterior to the surgical resection cavity in the right frontal convexity (in the expected location of the foot representation area of the primary motor cortex, as well as decreased CVR along the medial right frontal convexity, corresponding to the supplementary motor area.
OTHER CLINICAL APPLICATIONS OF CVR MAPPING
While CVR is considered a sensitive indicator for assessing the brain’s ability to dynamically adjust its energy supply, the clinical applications of CVR mapping (aside from presurgical mapping) are yet to reach their full potential, mainly due to technical constraints. In recent years, with the development of various advanced approaches, viable clinical applications of CVR mapping have emerged.
CVR mapping in cerebrovascular disease is emerging as a promising tool for clinicians involved in the evaluation of patients at risk for stroke. A few studies have been reported using hypercapnia induced BOLD MRI signal response to probe CVR in patients with arterial steno-occlusive diseases such as carotid artery stenosis[55,71,72]. For example, a patient with right middle cerebral artery occlusion and Moyamoya phenomenon secondary to aplastic anemia showed paradoxical CVR values in the right middle cerebral artery territory in the cortex[55].
CVR mapping is also a powerful method to detect abnormal neurovascular coupling and physiological effects of pharmacological agents in the brain. For instance, quantitative CVR approaches have been used to monitor CVR changes in the brain from caffeine consumption[73-75]. The coupling ratio between CBF and the cerebral metabolic rate for oxygen was found to be significantly decreased in both visual and motor cortices[73-75].
Another important application of CVR mapping is in the research of aging and neurodegenerative diseases. The blood vessels’ ability to dilate, which is reflected by CVR, may be a more sensitive biomarker than resting state perfusion. A recent study has demonstrated that CVR declined with age in a healthy adult cohort[76]. The decrease in CVR was shown to be steeper and spatially more extensive in the brain than resting state perfusion alterations. CVR in a mild AD patient group and age-matched controls has also been assessed[77]. Significant brain CVR deficits were found in early AD subjects. The spatial distribution of CVR reduction was different from that of resting perfusion alterations, but appeared to be consistent with in vivo amyloid mapping findings. The magnitude of CVR reduction correlated well with the volume of leukoaraiosis.
Peer reviewer: Guido Cavaletti, MD, Associate Professor, Department Neuroscienze e Tecnologie Biomediche - Università di Milano “Bicocca”, v. Cadore 48 - 20052 Monza, Italy
S- Editor Tian L L- Editor Webster JR E- Editor Zheng XM