Published online May 15, 2016. doi: 10.4291/wjgp.v7.i2.199
Peer-review started: July 30, 2015
First decision: September 28, 2015
Revised: October 21, 2015
Accepted: March 7, 2016
Article in press: March 9, 2016
Published online: May 15, 2016
Processing time: 289 Days and 17.6 Hours
Since the discovery of the Hedgehog (Hh) pathway in drosophila melanogaster, our knowledge of the role of Hh in embryonic development, inflammation, and cancerogenesis in humans has dramatically increased over the last decades. This is the case especially concerning the pancreas, however, real therapeutic breakthroughs are missing until now. In general, Hh signaling is essential for pancreatic organogenesis, development, and tissue maturation. In the case of acute pancreatitis, Hh has a protective role, whereas in chronic pancreatitis, Hh interacts with pancreatic stellate cells, leading to destructive parenchym fibrosis and atrophy, as well as to irregular tissue remodeling with potency of initiating cancerogenesis. In vitro and in situ analysis of Hh in pancreatic cancer revealed that the Hh pathway participates in the development of pancreatic precursor lesions and ductal adenocarcinoma including critical interactions with the tumor microenvironment. The application of specific inhibitors of components of the Hh pathway is currently subject of ongoing clinical trials (phases 1 and 2). Furthermore, a combination of Hh pathway inhibitors and established chemotherapeutic drugs could also represent a promising therapeutic approach. In this review, we give a structured survey of the role of the Hh pathway in pancreatic development, pancreatitis, pancreatic carcinogenesis and pancreatic cancer as well as an overview of current clinical trials concerning Hh pathway inhibitors and pancreas cancer.
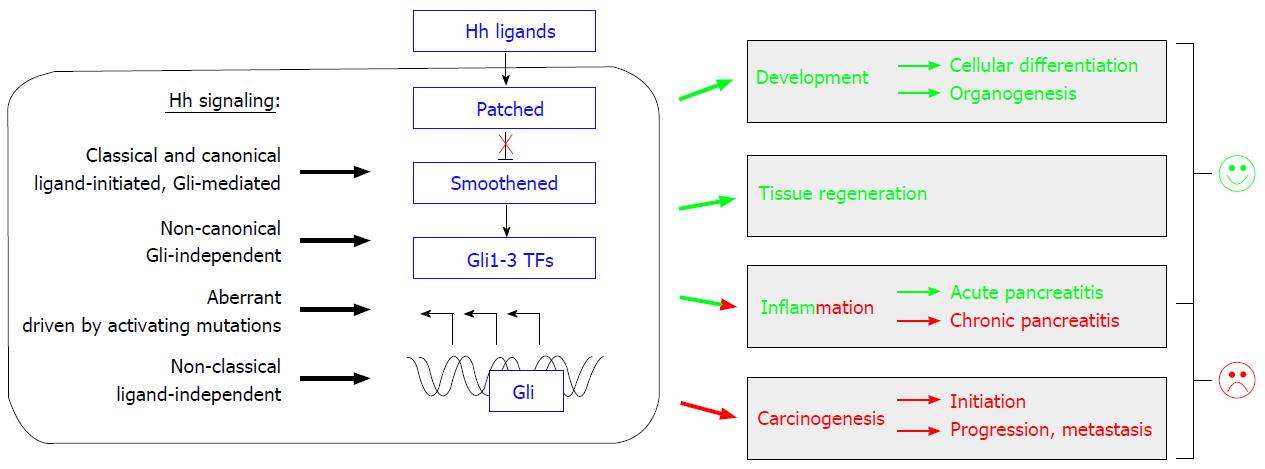
Core tip: The Hedgehog (Hh) pathway is a ligand-dependent and evolutionary conserved cellular signaling mechanism with various physiologic (development) and pathogenetic functions (especially carcinogenesis). Concerted Hh signaling is essential for human pancreatic development and homeostasis of the gastrointestinal tract. Aberrant expression within the Hh signaling pathway results in malformations like annular pancreas. The Janus aspect of Hh in pancreatitis is reflected by the protective role of Hh in acute pancreatitis vs the disease-progressive function of Hh in chronic pancreatitis (CP), whereby CP is linked to pancreatic cancerogenesis via pancreatic intraepithelial neoplasia (PanIn). Starting with PanIn and ending up at metastatic disease, Hh pathway is expressed in ductal pancreatic cancer thereby influencing and being paracrine influenced by the tumor microenvironment.
- Citation: Klieser E, Swierczynski S, Mayr C, Jäger T, Schmidt J, Neureiter D, Kiesslich T, Illig R. Differential role of Hedgehog signaling in human pancreatic (patho-) physiology: An up to date review. World J Gastrointest Pathophysiol 2016; 7(2): 199-210
- URL: https://www.wjgnet.com/2150-5330/full/v7/i2/199.htm
- DOI: https://dx.doi.org/10.4291/wjgp.v7.i2.199
Hedgehog (Hh) genes were originally identified in droso-phila melanogaster as regulators of body patterning during embryonic development[1]. Today it is known that the Hh pathway plays a central role in diverse biological processes in mammals, such as embryonic development, cell proliferation, differentiation, tissue repair and maintenance of stem cell status in the adult[2].
In general, activation of the Hh pathway relies on the binding of a secreted ligand to its receptor. Three ligand homologues are known in mammals: Desert hedgehog (Dhh), Indian hedgehog (Ihh) and Sonic hedgehog (Shh). The ligands are produced as precursors and are secreted after extensive modifications to bind to their membrane bound receptor, called Patched. In mammals, two homologues exist, Patched1 (Ptch1) and Patched2 (Ptch2). After signal transduction via the co-receptor Smoothened (Smo), the executing transcription factors of the Hh pathway are the Gli proteins, of which three homologues are known in mammals: Gli1, Gli2 and Gli3[3]. Using a simplified model, the canonical Hh signaling can be described as follows[2,4]: In the absence of a Hh ligand, Ptch inactivates Smo - probably by preventing its localization into the primary cilium, a cell organelle that is thought to be essential for proper Hh signaling[5,6]. As a consequence, the Gli proteins are processed in such a way that they act as transcriptional repressors of the Hh target genes. However, upon binding of the Hh ligand to the receptor Ptch, inactivation of Smo is ended, allowing Smo to translocate to the primary cilium and initiate a cascade of events that ultimately lead to the conversion of Gli factors into their active form. The latter then shuttle into the nucleus and enable transcription of Hh target genes, including components of the pathway itself, such as Ptch and Gli1, indicating a built-in feedback loop within the Hh signaling cascade[2]. In addition to the “classical and canonical” Hh signaling described above, also non-canonical (Gli-independent), non-classical (ligand-independent) and aberrant Hh signaling (driven by activating mutations) have been identified at different stages of carcinogenesis (Figure 1)[7].
The pancreas is a fundamental organ of the digestive system with specialized endocrine and exocrine functions. The acinar cells within the exocrine pancreatic compartment produce and secrete numerous digestive enzymes into the duodenum. In the endocrine compartment, specialized cells produce hormones and directly release them into the blood stream - most importantly to control and regulate the blood glucose concentration. It is known from previous studies that physiologic Hh pathway signaling is crucial for correct development of the pancreas[8,9]. With this review, we give an overview of the current understanding of the role of Hh signaling in pancreatic development, cell differentiation and functional specialization. In a second part, the pathomechanistic implications of deregulated Hh signaling are discussed for the clinically most important pancreatic pathologies.
Pancreatic development is based on: (1) The fusion of two evaginations of the foregut to one single organ; and (2) endodermal growth by dichotomy branching. According the classical Carnegie stages[10,11], in stage 13, the dorsal pancreatic bud arises at first as a thickening of the endodermal tube, which proliferates, into the dorsal mesogastrium. In close, in stage 14, the ventral pancreatic bud evaginates to the liver primordium. As a result of differential growth of the duodenum, which rotates 90 degrees clockwise and becomes “C”-shaped, the ventral pancreatic bud comes to lie below and behind the dorsal pancreatic bud in stage 15. Until stage 17, both pancreatic buds have fused: The ventral pancreatic bud forms the posterior part of the head and the posterior part of the uncinated process, whereas the rest of the pancreas is formed by the dorsal pancreatic bud (the anterior part of the head, the body and the tail). Failure of the ventral pancreatic bud to migrate will result in an annular pancreas with consequent duodenal stenosis[12]. The main pancreatic duct (of Wirsung) is formed by the fusion of the distal part of the dorsal pancreatic duct and the entire ventral pancreatic duct and enters the duodenum combined with the bile duct at the major papilla. Until the postnatal period, the proximal portion of the dorsal pancreatic duct either obliterates or persists as an accessory duct (of Santorini), entering the duodenum at the minor papilla (10% adults), so-called pancreatic divisum.
Differentiation and early specification of pancreatic endoderm is induced by fibroblast growth factor 2 and activin [a transforming growth factor beta (TGF-β) family member], both produced by the notochord and endothelium of the dorsal aorta. Both repress the expression of the transcription factor Shh locally in the gut endoderm, destined to form the dorsal pancreatic bud. Endoderm lying caudally to the pancreatic region does not respond to those signals[13]. The ventral bud is induced by upregulation of the pancreatic and duodenal homeobox 1 (PDX1) gene from the splanchnic mesoderm.
From 10th to 15th week, the primitive endodermal ductal epithelium provides the stem cell population for all the secretory cells, which are initially located in the duct walls or in the buds, from which they arise. Islet differentiation proceeds in two phases[13]: Phase I (9th-15th week) is characterized by the proliferation of polyhormonal cells, whereas the differentiation of monohormonal cells is seen from week 16 onwards, referred to as phase II. Later, these endocrine cells accumulate in pancreatic islets (of Langerhans) and scatter throughout the pancreas, starting with insulin and amylin secretion by β-cells approximately at the 5th month until neonatal period. The dorsal bud gives rise mostly to α-cells, which produce glucagon; however, most of the pancreatic polypeptide producing γ-cells develop from the ventral bud. After week 30, somatostatin-producing δ-cells are seen. The remaining primitive duct cells will either differentiate into definitive duct cells with microvilli and cilia or into acinar cells in which zymogen granules or acinar cell markers can be detected at weeks 12-16[13].
Correct ductal branching pattern and formation of acinar structures is determined by pancreatic mesenchyme which gives rise to connective tissue between the ducts resulting in pancreatic proliferation and maintaining the relative proportions of acinar, α-and β-cells. Additionally, it provides cell lines for smooth muscle within the pancreatic tissue, and angiogenic mesenchyme produces blood and lymphatic vessels.
Pancreas development is regulated by the activation/inactivation of Hh signaling members, which are ex-/repressed either within pancreatic tissue (e.g., Ihh) or in adjacent tissue (e.g., Shh)[14]. Initial absence of Shh signaling is required for regular pancreatic development, because ectopic expression of Shh leads to transformation of pancreatic mesoderm into intestinal mesenchyme in mice[15]. In single mutant mice (i.e., Shh-/- or Ihh-/-), gastrointestinal defects of the developing endoderm like annular pancreas or other malformations have been reported, suggesting similarities to human gut malformations[8,16].
It was shown that the graded response to Hh-signaling controls regular pancreatic development in mice, in which Hh signaling occurs at low levels during early organogenesis to ensure the correct establishment of organ boundaries and tissue architecture, and is up-regulated at later developmental stages to promote proliferation and maturation of the tissue[9,17-19]. Nielsen et al[20] confirmed the suggested concerted Hh signaling also in human pancreatic organogenesis: In early pancreatic development (7.5 wk), Gli3 was highly expressed in developing pancreatic ducts - while Smo and Gli2 were absent. In contrast, Smo and Gli2 were highly expressed between weeks 14 to 18, whereas the expression of Gli3 was reduced.
PDX1 (a pancreatic-promoting transcription factor; syn.: Insulin promotor factor 1) is also expressed in the preduodenal endoderm - including the sites of dorsal and ventral pancreatic bud formation. Total absence of the pancreas is observed in homozygous PDX1 mutant mice that suggest that PDX1 is necessary for the formation of the pancreas and may be essential in the differentiation of pancreatic precursor cells[21,22]. Although all of the involved downstream effectors of human pancreas development have not been determined in detail yet, it appears that expression of the paired homeobox genes PAX4 and PAX6 specifies the endocrine cell lineage: Cells expressing both become β-, δ-, and γ-cells; whereas those expressing only PAX6 become α-cells.
The cellular and molecular processes in acute pancreatitis (AP) and chronic pancreatitis (CP) were intensively elucidated in the last years providing valuable detailed insights which could be important in the next years for a further therapeutical approach in this partially lethal disease entity (reviewed in detail in[23-25]). In short, in the phase of AP, the major cellular key players are neutrophils, monocytes and macrophages which interact by building and secretion of cytokines and inflammatory mediators, mainly tumor necrosis factor α, interleukin (IL) 1β and 6, and monocyte chemotactic protein. In the phase of CP, pancreatic stellate cells (PSC), tissue infiltrating myeloid cells, and particularly macrophages are coming to the fore by induced and increased progressive fibrosing of the pancreas tissue, being mediated mainly by nuclear factor (NF)-κB[26,27]. Finally, the crosstalk of the mentioned cells is linked to T-subsets (CD-8+/central memory cells as well as T-regulator cells) which are involved in the pathogenesis of CP[28,29]. Additionally, CP is commonly regarded as a relevant risk factor for ductal pancreatic cancer (DPC) by irregular ductal changes leading to acino-ductal metaplasia and pancreatic intraepithelial neoplasia (PanIn)[30,31].
Focusing on the linkage between the Hh pathway and AP as well CP, experimental investigations demonstrated that the members of the Hh pathway could be detected in different amounts in AP and CP, whereby the definitive functional role of Hh in AP and CP seems to be very different. Additionally, in the process of CP forward to DPC an irregular expression pattern of the Hh members are observed compared to the normal and structured embryonic development of the pancreas[9,32,33].
Compared to CP, the role of Hh in AP has been dealt with only in few studies. Summarizing these data, activation of the Hh signaling is linked to injury and repair using the cerulean-mediated model, whereby the unequivocal conclusion of the available experimental data is that the Hh has protective function in AP. In 2008, Fendrich et al[33] presented a functional analysis of the Hh pathway in AP using pharmacologic and genetic techniques (like Ptch1-LacZ reporter mice and two different Cre-driven pancreas-specific depletion mice models of Smo) demonstrating that Hh is essentially involved in effective regeneration of the exocrine pancreas. By this approach, Shh, Ihh, and Gli1 are increasingly expressed in caerulein treated mice, whereby the pharmacologic and genetic inhibition lead to persistence of PDX1 expressing metaplastic intermediates and impaired tissue repair. Additionally, the group of Zhou et al[34] used a Cerulein-induced AP model in mice to show elegantly that: (1) Shh, not Ihh or Dhh, is involved in this model; (2) Shh-inhibition aggravates the AP; and (3) the anti-inflammatory autocrine effect of Shh is mediated by IL-10. A recent experimental study from 2014 showed that Gli1, the downstream member of the Hh cascade, could essentially influence the inflammatory reaction in the circumstances of remodeling processes of the pancreas. Based on genetic analysis of deletion of a single allele of Gli1, the authors postulated that the canonical Hh pathway, respectively the transcription factor Gli1, is essential for pancreatic recovery in inflammatory processes via Gli1 targeted cytokines, including IL-6, murine homolog of IL8, monocyte chemoattractant protein-1, and Macrophage colony-stimulating factor M-csf, leading to pancreatic tumorgenesis via improper stromal remodeling and persistence of the inflammatory infiltrate[35].
Empiric studies in humans with CP demonstrated a heterogeneous upregulated expression of Ihh, its receptors Ptch and Hedgehog-Interacting Protein, and Smo in different histological distribution and cellular localization of human tissue with CP using Northern blotting, immunohistochemistry and Western-blotting[32,36,37]. Interestingly, the members of the Hh pathway were localized mainly in the islet cells, whereas the Hh signaling members were present in degenerated acinar and tubular complexes of CP[36,37]. In addition, Kayed et al[37] could show that the inhibition of the Hh pathway via Cyclopamin led to growth inhibition of TAKA-1 pancreatic ductal cells through cell cycle arrest in vitro.
Based on cDNA microarrays, Bhanot et al[31] could support the findings, that the Hh pathway is altered in microdissected ectatic ducts of CP whereby dysregulation of Hh could enhance the probability of DPC via duct ectasia, acino-ductal metaplasia or intraepithelial neoplasia as reviewed by Bahnot et al[31] in 2008.
As mentioned above, PSCs are essentially involved in the pathogenesis of the CP, whereby the main question is, how the Hh pathway regulates the activation of these PSCs.
The experimental analysis of the group of Shinozaki et al[38] revealed that Ihh has no evident effect on expression of collagen-1 or alpha-smooth muscle actin or on proliferation of PSCs, but Ihh modulates the migration potency by changing the amount of membrane-type 1 matrix metalloproteinase and its localization on the plasma membrane leading to a pro-migration status of PSCs. Although the Ihh effects are mediated by Gli1, experimental overexpression of Gli1 using an adenovirus-mediated or RNA interference techniques revealed a negatively regulation by Gli1 to Ihh effects in vitro.
But the question remains: Why is Hh pathway upregulated within the fibrogenic process of CP? Based on in vitro and in situ studies with xenografts as well as in humans with pancreatitis, it is postulated that para- and partially autocrine activation of stromal cells by Hh ligands from epithelial components and vice versa are responsible[39-41]. The experimental data of Jung et al[42] are based on transgenic phenotypes in zebrafish with over-expression of either Ihh or Shh along with green fluorescence protein. Consecutive analysis of these transgenic phenotypes using quantitative and qualitative investigations of mRNA and protein levels including PCR, in situ hybridization, and immunohistochemistry revealed that myofibroblasts and ductal cells are activated and proliferate which is triggered by paracrine Hh signaling in a restricted expression of Ptc1, Smo and Gli1/2. Additionally, Hh ligands could induce matrix metallopeptidase 9 and TGF-β1 in this animal model[42].
Recent investigations by Tsang et al[43] could support the published findings of pro-fibrinogenic effects of Hh in CP by using an in vivo model. The application of Rhein, a natural anthraquinone derivative, reduces the activation of PSCs in mice with experimental induced CP. The morphological effect of Rhein in reduced pancreatic fibrosis was paralleled by reduced molecular expression of fibrogenic markers including alpha-smooth muscle actin, fibronectin 1, type I collagen as well as the members of the Hh pathway Shh and Gli1.
Interestingly, the promoting fibrotic effect of Hh signaling is not only existent in pancreas, but also could be observed in other organs like lung, bile duct and liver implicating a tissue independent overriding principle of the Hh pathway in this pathogenesis[44-46].
Since chronic recurrent inflammation has been linked to carcinogenesis, especially in pancreas, some findings of Hh in AP/CP and pancreatic carcinogenesis are presented in the following for supporting this already emphasized linkage[47,48]. First of all, Hh modulates the axis between inflammation and cancerogenesis via activation and production of cytokines by human peripheral CD4+ T cells[49]. Furthermore, experimental studies of Hh in AP and CP revealed morphological changes like ductal metaplasia promoted by Shh, which are per se no pre-tumorous conditions[33,50]. Nevertheless, during progression of CP, morphological changes of the ductal pancreatic tissue like papillary lesions with nuclear atypia resulting in PanIn lesions could be observed which have a high association to aberrant Hh expression and pancreatic cancer[31,50].
In conclusion (summarized in Table 1), members of the Hh pathway have protective properties in case of AP, whereby the face of Hh changes to a progressive and disease-promoting function in CP. Especially in CP, the negative effects of Hh on tissue remodeling and repair favored the possibility of cancerogenesis via de- and trans-differentiation[51-54].
| Acute pancreatitis | Chronic pancreatitis | |
| Pathogenetic effect of Hh | Protective | Progressive |
| Detected members of Hh | ↑Shh (Ihh, Dhh), Gli1 | ↑Ihh (Shh), Ptc, Hip, SMO, Gli1, Gli2 |
| Interactive cells (auto-and paracrine effects) | Acinar/ductal cells with; acute inflammatory cells | Acinar/ductal cells with; PSC |
| (Immune) mediators of inflammation | IL-10, IL-6, mIL-8, Mcp-1, and M-csf (Csf1) | MT1-MMP, MMP9, TGF-β1, smooth muscle actin, fibronectin 1, type I collagen |
| Association to cancerogenesis | No | Yes, possibly via ADM and PanIn |
Hh signaling in the normal pancreas and in pancreatic ductal adenocarcinoma is exclusively paracrine with expression of Shh (tumor cell and stroma signal circle as shown in Figure 2)[55]. The silencing of Smo in pancreatic cancer epithelium in mice showed no altered tumor spread or development, so the Hh signaling does not occur in an autocrine way[55].
In paracrine signaling, the Hh ligand sends signals directly to the stroma and provides a selective tumor growth advantage. This was established through a pancreatic cancer model where Hh signal was needed for overall tumor growth while the particular tumor cells themselves did not respond to Hh ligand[56].
The existence of cancer stem cells (CSC) in different tumors, including pancreatic cancer, offers an explanation why some therapy assessments are ineffective[57,58]. Therefore, a good knowledge base for new therapies, which target pancreatic CSCs, is very important. The Hh signaling pathway plays a vital role in pancreatic and embryonic development; autocrine or paracrine secreted Shh activates a signal transduction cascade that includes other Hh members like Ptch and Smo, which then activates the canonical Hh pathway through Gli.
This leads to transcription of multiple targets like Nanog, Cyclin D1, Ptch, Gli1 and Gli2. Activation of Shh signaling seems to precede the transformation of pancreatic tissue stem cells to cancerous stem cells. This was shown in mice, which were treated with sulforaphane to inhibit the growth of these stem cells. Sulforaphane is a natural compound found in cruciferous vegetables like broccoli that as an inhibitor acts on various receptors and pathways with anti-cancerous properties like apoptosis induction and cell proliferation[59]. This experimental study showed that human pancreatic stem cells need the activity of the Hh-Gli pathway for proliferation, survival, self-renewal and tumorigenicity[60].
In 2002, Chen et al[61] modulated mammalian embryonic pancreas development in vitro using cyclopamine treated pancreatic explants. A recombinant form of Shh was added to pancreatic buds to activate the Hh signaling pathway. The fluorescently labeled epithelium of the pancreatic explants underwent extensive growth and branching when treated by cyclopamine, which indicates that Hh inhibition did not block branching in the epithelium[61].
Walter et al[62] isolated pancreatic fibroblasts from benign and malignant primary pancreatic resection specimens by immunohistochemistry marker selection through vimentin. Together with two fibroblast cell lines, SC2 and SC3 (from non-neoplastic pancreas), the cancer-associated fibroblasts (CAF) were characterized for Hh activity. The fibroblast cell lines and the isolated CAFs where treated with Shh ligands to observe any expression changes on Gli mRNA. As a result they detected overexpression of Smo in pancreatic CAFs, which could transduce the Shh signal followed by Gli1 activation. The Hh pathway has been identified as activated in cancer associated stromal fibroblasts in mouse models of pancreatic cancer. CAFs can actively transduce the Hh signal to induce Gli expression. CAFs expressing Smo respond to exogenous Hh ligand, whereas control fibroblasts lacking Smo expression are unresponsive to Hh ligand, and downregulation of Smo in CAFs inhibits transduction of the Hh signal[62].
In human tumor xenografts, expression of Shh by tumor cells correlated with increased expression of GLi1 and Ptch1 in the stromal compartment. Pathway inhibition affected only stromal Gli1 and Ptch1 expression and resulted in decreased tumor growth exclusively in Hh ligand-expressing tumors[63].
Tian et al[64] demonstrated that the expression of an oncogenic allele of Smo (SmoM2) in mouse pancreas neither activated Hh signaling in epithelial cells nor promoted their neoplastic transformation. In murine pancreatic cancer models as well as in human pancreatic cancer specimens, activation of the Hh pathway was observed only in stromal cells surrounding Hh ligand-expressing tumor cells[64].
Tumors of the pancreas can develop either from ductal, neuroendocrine or acinar cell populations. Due to a lack of information about the role of the Hh signaling pathway in acinar and neuroendocrine tumors of the pancreas, the following paragraphs will concentrate on DPC.
Among all cancers, DPC has one of the worst prognoses among all cancers with an overall 5-year survival rate of less than 5%[65]. Chemo- and radiotherapy are largely ineffective; furthermore, metastatic spread frequently occurs even after complete surgical resection[66]. The Hh pathway is one highly promising signaling transduction pathway for a better understanding of the origin of DPC.
Expression of Hh pathway members is usually not present in healthy adult pancreatic tissue[67]. In 2008, a global sequencing analysis revealed that the Hh pathway is one of the core signaling pathways that undergoes somatic alterations in nearly all pancreatic cancers[68]. Kayed et al[37] showed an aberrant activity of the Hh pathway in chronic pancreatitis and pancreatic cancer. Later on, it was recognized that Shh expression enhances the proliferation of pancreatic duct epithelial cells[69] and is not only up-regulated in the setting of pancreatic injury, but also in noninvasive precursor lesions of DPC: (1) PanIn; and (2) intraductal papillary mucinous neoplasia (IPMN) starting with rising expression values up to Hh pathway persistence in metastatic state[67,70,71]. Additionally, it was stated that up-regulation of the Shh ligand is sufficient to misdirect the pancreatic ductal epithelium towards a gastrointestinal metaplastic phenotype, which explains the involvement in IPMN formation[50,63].
However, dysfunction, or rather re-activation of the Hh pathway is not the only reason for the development of PanIn and DPC. Lauth et al[72] described a synergistic molecular crosstalk between Hh pathway and activated V-Ki-ras2 Kirsten rat sarcoma viral oncogene homolog (K-RAS) signaling pathway[72]. Over 90% of patients suffering from DPC showed a K-RAS mutation, thus identifying the K-RAS pathway as another key mediator of pancreatic carcinogenesis[68,73]. Patients with a K-RAS mutation developed PanIn; and an additional P53 loss of function leads to subsequent development of the lesion towards DPC[74]. According to various studies, the crosstalk between Hh and K-RAS takes place via the RAF/MEK/MAPK pathway[75,76].
In 2013, Mills et al[77] were able to identify Gli1 as an effector of K-RAS at early stages of pancreatic carcinogenesis. They showed in a mouse model that loss of Gli1 impairs K-RAS-induced carcinogenesis. Although the mice still developed PanIn, the incidence of PanIn decreased and as a result, no mice suffered from DPC[77].
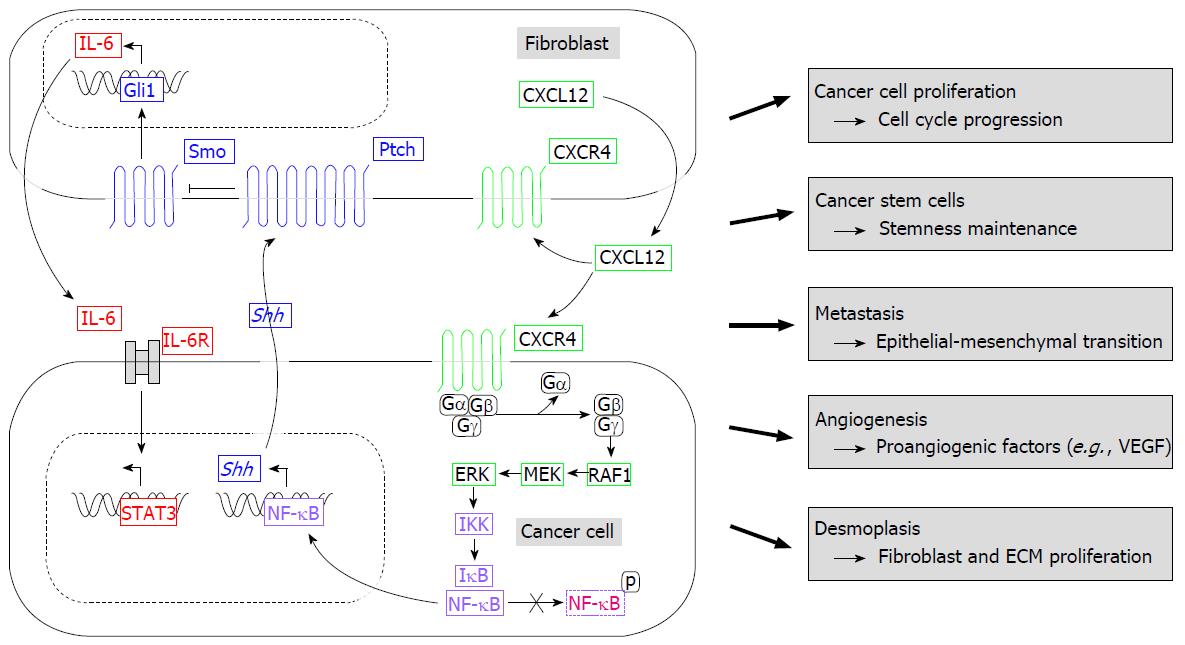
In recent studies, another central role in pre-neoplastic lesions of the pancreas is awarded to the signal transducer and activator of transcription 3 (STAT3) and its upstream cytokine IL-6. It is supposed that STAT3 activation is involved in driving early changes in the microenvironment promoting PanIn formation in the presence of oncogenic K-RAS[78,79]. Mills et al[77] stated that Gli1 also acts on CAF by paracrine regulation of the IL-6/STAT3 pathway in stromal cells of the tumor microenvironment (TME) and, thus, regulating the progression of precursor lesions and tumor formation (Figure 2).
DPC pathogenesis is characterized by a desmoplastic reaction to invading tumor cells, including a dense extracellular matrix that was recently shown to be the result of epithelial to mesenchymal transition (EMT)[80,81]. The epithelial-mesenchymal interaction, especially in the paracrine model of the Hh pathway, plays a distinctive role in different carcinoma entities as well as in DPC. Deregulated Hh pathway in PanIn and DPC leads to the secretion of Hh ligands Shh and Ihh, followed by a paracrine activation of CAFs in the surrounding stroma leading to expansion and desmoplasia[40,82,83]. In detail, neoplastic epithelium secretes Shh, which binds to the cognate Ptch-receptor on stromal cells, followed by desmoplastic stromal expansion and microenvironment remodeling. Moreover, supporting the paracrine action model of Hh pathway in DPC, Yauch et al[83] showed that treatment with Hh pathway antagonist results in downregulation of Hh target genes only in the tumor stroma but not in the epithelial cancer cell. In the same way, Smo expression decreases in mesenchymal cells in the pancreas resulting in Hh pathway activation. However, Lee et al[80] described that Hh pathway activity controls the balance between epithelial and stromal elements: Pathway activation causes stromal hyperplasia and reduced epithelial growth, whereas Hh inhibition causes accelerated growth of epithelial elements and suppression of desmoplasia.
It is suggested that the TME and extensive desmoplasia are partly responsible for chemoresistance in DPC by creating a “fence” around the tumor cells, which protects them against therapeutic compounds[84]. Therefore, tearing down this barrier could be a promising strategy to improve therapeutic approaches. Singh et al[85] already showed that inhibition of Hh pathway depleted tumor-associated stromal tissue.
There are many other different tumor specific characteristics that are influenced by the interrelation of Hh pathway and the TME. Bailey et al[86] identified paracrine Shh-mediated fibroblasts within the TME as source of Hypoxia-inducible factor 1 alpha (HIF-1α), which is known to be a regulator of angiogenesis and metastasis in cancer. Another example is the CXC-motif-chemokine 12/CXC chemokine receptor type 4 (CXCL12/CXCR4) pathway, which is on the one hand critical for normal cellular processes, but on the other hand contributes to metastasis, growth, survival and stem cell characteristics of cancer cells[87-89]. CXCL12, the sole ligand for CXCR4, is produced by tumor-associated stromal cells, is increased in DPC; and after binding to its receptor CXCR4, leads to activation of extracellular signal-regulated kinases resulting in release and nuclear translocation of NF-κB, which then directly binds the Shh promotor[85,90,91]. In summary, Hh pathway acts in a predominantly paracrine manner, thereby influencing and being influenced by the TME (for an overview of Hh-dependent interactions between tumor and stroma cells in DPC, Figure 2).
CSC, also called tumor initiating cells are suggested to be responsible for cancer initiation, progression and chemo-resistance in several malignancies including DPC[92]. The transcription factors Nanog, octamer-binding transcription factor 4 and BMI1 Proto-Oncogene, Polycomb Ring Finger (BMI-1) are essential for the “stemness”, including characteristics like self-renewal of CSC[93-95]. The Hh pathway is implicated in the maintenance of pancreatic CSCs: For example, Li et al[96] stated that Shh expression was 46-fold greater in pancreatic CSCs (CD24+/CD44+/ESA+) as in other DPC cells (CD24-/CD44-/ESA-). Additionally, Gli1 is known to up-regulate genes that are crucial for many properties for stemness of CSC - like Nanog and BMI-1[97-99].
Recapitulating this chapter, Hh pathway plays an important role in DPC, beginning from PanIn precursors to progressed metastatic disease. Hh signaling cross talks with a variety of other signaling pathways, like K-RAS, requires the interaction with the EMT in particular via paracrine pathway stimulation in order to contribute to the development of DPC (Figure 1).
At present, clinical trials using Hh inhibitors enroll patients with pancreatic malignancies including advanced, metastatic, recurrent or resectable pancreatic cancer. Currently, no trials are listed within the United States National Institutes of Health database (http://www.clinicaltrials.gov) which target pancreatitis or other pancreatic non-neoplastic conditions. As summarized in Table 2, most trials in the phase 1 or 2 setting use GDC-0449 (vismodegib) which is a small molecular weight inhibitor of Smo[100] thereby interfering with Hh signaling at the plasma membrane level similarly to cyclopamine, a naturally occurring Smo antagonist[101]. Other Hh-targeting drugs in current clinical trials on pancreatic cancer are the Smo-inhibitors LDE-225 (Sonidegib)[102] and IPI-926 (Saridegib)[103].
| Drug | Combination | Phase | Status | Trial ID |
| GDC-0449 | Gemcitabine | 0 | N | NCT01713218 |
| 1/2 | A | NCT01195415 | ||
| 2 | A | NCT01064622 | ||
| Erlotinib, gemcitabine | 1 | A | NCT00878163 | |
| Gemcitabine, nab-paclitaxel | 2 | A | NCT01088815 | |
| LDE-225 | Gemcitabine, nab-paclitaxel | 1/2 | R | NCT01431794 |
| 1/2 | R | NCT02358161 | ||
| BKM120 | 1 | C | NCT01576666 | |
| IPI-926 | Gemcitabine | 1/2 | C | NCT01130142 |
For the latter, a preclinical study on pancreatic cancer in mice demonstrated that IPI-926 depletes tumor-associated stromal tissue and facilitated the delivery and increased the intratumoral concentration of gemcitabine[84]. In line with these results, all currently ongoing clinical trials combine selective Hh antagonists with established chemotherapies (gemcitabine, paclitaxel) or other targeted drugs (erlotinib epidermal growth factor receptor inhibitor) or BKM120 (Phosphatidylinositol-4,5-bisphosphate 3-kinase inhibitor) to investigate possible therapeutic benefits of these drug combinations. Taken together, current clinical studies employ inhibitors of the Smo co-receptor in combination with established chemotherapeutic drugs. Novel experimental inhibitors targeting the Hh pathway at the level of the transcriptional regulation (e.g., Gant-61, Gant-58) have not yet entered the stage of clinical evaluation[104].
Besides its physiologic functions in human pancreatic development, the Hh pathway is activated in numerous pathological conditions, including carcinogenesis. However, the data on its functional aspects currently available draw a more nuanced picture. Progression from pancreatic cancer precursors lesions (PanIn) to DPC and metastatic disease is strongly influenced by a paracrine Hh signal modulating the interaction between DPC cells and CAFs. This Hh driven signaling predominantly includes the IL-6/STAT3 and the CXCL12/CXCR4 pathways resulting in disease progression by invasion, angiogenesis, metastasis formation and chemoresistance as well as gaining of stem cell like characteristics. Therefore, therapeutic targeting of the Hh pathway may provide new therapeutic approaches to improved disease control and prognosis for both, chronic pancreatitis and pancreatic carcinogenesis.
P- Reviewer: Boros M, Morini S S- Editor: Kong JX L- Editor: A E- Editor: Li D
| 1. | Nüsslein-Volhard C, Wieschaus E. Mutations affecting segment number and polarity in Drosophila. Nature. 1980;287:795-801. [PubMed] [DOI] [Full Text] |
| 2. | Teglund S, Toftgård R. Hedgehog beyond medulloblastoma and basal cell carcinoma. Biochim Biophys Acta. 2010;1805:181-208. [PubMed] [DOI] [Full Text] |
| 3. | Lees C, Howie S, Sartor RB, Satsangi J. The hedgehog signalling pathway in the gastrointestinal tract: implications for development, homeostasis, and disease. Gastroenterology. 2005;129:1696-1710. [PubMed] [DOI] [Full Text] |
| 4. | Heretsch P, Tzagkaroulaki L, Giannis A. Modulators of the hedgehog signaling pathway. Bioorg Med Chem. 2010;18:6613-6624. [PubMed] [DOI] [Full Text] |
| 5. | Huangfu D, Liu A, Rakeman AS, Murcia NS, Niswander L, Anderson KV. Hedgehog signalling in the mouse requires intraflagellar transport proteins. Nature. 2003;426:83-87. [PubMed] [DOI] [Full Text] |
| 6. | May SR, Ashique AM, Karlen M, Wang B, Shen Y, Zarbalis K, Reiter J, Ericson J, Peterson AS. Loss of the retrograde motor for IFT disrupts localization of Smo to cilia and prevents the expression of both activator and repressor functions of Gli. Dev Biol. 2005;287:378-389. [PubMed] [DOI] [Full Text] |
| 7. | Shevde LA, Samant RS. Nonclassical hedgehog-GLI signaling and its clinical implications. Int J Cancer. 2014;135:1-6. [PubMed] [DOI] [Full Text] |
| 8. | Hebrok M. Hedgehog signaling in pancreas development. Mech Dev. 2003;120:45-57. [PubMed] [DOI] [Full Text] |
| 9. | Lau J, Kawahira H, Hebrok M. Hedgehog signaling in pancreas development and disease. Cell Mol Life Sci. 2006;63:642-652. [PubMed] [DOI] [Full Text] |
| 10. | Hill MA. Early human development. Clin Obstet Gynecol. 2007;50:2-9. [PubMed] [DOI] [Full Text] |
| 11. | Yi H, Xue L, Guo MX, Ma J, Zeng Y, Wang W, Cai JY, Hu HM, Shu HB, Shi YB. Gene expression atlas for human embryogenesis. FASEB J. 2010;24:3341-3350. [PubMed] [DOI] [Full Text] |
| 12. | Etienne D, John A, Menias CO, Ward R, Tubbs RS, Loukas M. Annular pancreas: a review of its molecular embryology, genetic basis and clinical considerations. Ann Anat. 2012;194:422-428. [PubMed] [DOI] [Full Text] |
| 13. | Howard ER, Stringer MD, Colombani PM. Surgery of the Liver, Bile Ducts and Pancreas in Children. London: Arnold Publishers; 2002; 239-246. |
| 14. | Hebrok M, Kim SK, St Jacques B, McMahon AP, Melton DA. Regulation of pancreas development by hedgehog signaling. Development. 2000;127:4905-4913. [PubMed] |
| 15. | Apelqvist A, Ahlgren U, Edlund H. Sonic hedgehog directs specialised mesoderm differentiation in the intestine and pancreas. Curr Biol. 1997;7:801-804. [PubMed] [DOI] [Full Text] |
| 16. | Ramalho-Santos M, Melton DA, McMahon AP. Hedgehog signals regulate multiple aspects of gastrointestinal development. Development. 2000;127:2763-2772. [PubMed] |
| 17. | Kawahira H, Scheel DW, Smith SB, German MS, Hebrok M. Hedgehog signaling regulates expansion of pancreatic epithelial cells. Dev Biol. 2005;280:111-121. [PubMed] [DOI] [Full Text] |
| 18. | Cano DA, Hebrok M, Zenker M. Pancreatic development and disease. Gastroenterology. 2007;132:745-762. [PubMed] [DOI] [Full Text] |
| 19. | van den Brink GR. Hedgehog signaling in development and homeostasis of the gastrointestinal tract. Physiol Rev. 2007;87:1343-1375. [PubMed] [DOI] [Full Text] |
| 20. | Nielsen SK, Møllgård K, Clement CA, Veland IR, Awan A, Yoder BK, Novak I, Christensen ST. Characterization of primary cilia and Hedgehog signaling during development of the human pancreas and in human pancreatic duct cancer cell lines. Dev Dyn. 2008;237:2039-2052. [PubMed] [DOI] [Full Text] |
| 21. | Jonsson J, Carlsson L, Edlund T, Edlund H. Insulin-promoter-factor 1 is required for pancreas development in mice. Nature. 1994;371:606-609. [PubMed] [DOI] [Full Text] |
| 22. | Offield MF, Jetton TL, Labosky PA, Ray M, Stein RW, Magnuson MA, Hogan BL, Wright CV. PDX-1 is required for pancreatic outgrowth and differentiation of the rostral duodenum. Development. 1996;122:983-995. [PubMed] |
| 23. | Thrower E, Husain S, Gorelick F. Molecular basis for pancreatitis. Curr Opin Gastroenterol. 2008;24:580-585. [PubMed] [DOI] [Full Text] |
| 24. | Zheng L, Xue J, Jaffee EM, Habtezion A. Role of immune cells and immune-based therapies in pancreatitis and pancreatic ductal adenocarcinoma. Gastroenterology. 2013;144:1230-1240. [PubMed] [DOI] [Full Text] |
| 25. | Gukovsky I, Li N, Todoric J, Gukovskaya A, Karin M. Inflammation, autophagy, and obesity: common features in the pathogenesis of pancreatitis and pancreatic cancer. Gastroenterology. 2013;144:1199-209.e4. [PubMed] [DOI] [Full Text] |
| 26. | Omary MB, Lugea A, Lowe AW, Pandol SJ. The pancreatic stellate cell: a star on the rise in pancreatic diseases. J Clin Invest. 2007;117:50-59. [PubMed] [DOI] [Full Text] |
| 27. | Treiber M, Neuhöfer P, Anetsberger E, Einwächter H, Lesina M, Rickmann M, Liang S, Kehl T, Nakhai H, Schmid RM. Myeloid, but not pancreatic, RelA/p65 is required for fibrosis in a mouse model of chronic pancreatitis. Gastroenterology. 2011;141:1473-1485. [PubMed] [DOI] [Full Text] |
| 28. | Schmitz-Winnenthal H, Pietsch DH, Schimmack S, Bonertz A, Udonta F, Ge Y, Galindo L, Specht S, Volk C, Zgraggen K. Chronic pancreatitis is associated with disease-specific regulatory T-cell responses. Gastroenterology. 2010;138:1178-1188. [PubMed] [DOI] [Full Text] |
| 29. | Grundsten M, Liu GZ, Permert J, Hjelmstrom P, Tsai JA. Increased central memory T cells in patients with chronic pancreatitis. Pancreatology. 2005;5:177-182. [PubMed] [DOI] [Full Text] |
| 30. | Becker AE, Hernandez YG, Frucht H, Lucas AL. Pancreatic ductal adenocarcinoma: risk factors, screening, and early detection. World J Gastroenterol. 2014;20:11182-11198. [PubMed] [DOI] [Full Text] |
| 31. | Bhanot UK, Möller P. Mechanisms of parenchymal injury and signaling pathways in ectatic ducts of chronic pancreatitis: implications for pancreatic carcinogenesis. Lab Invest. 2009;89:489-497. [PubMed] [DOI] [Full Text] |
| 32. | Kayed H, Kleeff J, Osman T, Keleg S, Büchler MW, Friess H. Hedgehog signaling in the normal and diseased pancreas. Pancreas. 2006;32:119-129. [PubMed] [DOI] [Full Text] |
| 33. | Fendrich V, Esni F, Garay MV, Feldmann G, Habbe N, Jensen JN, Dor Y, Stoffers D, Jensen J, Leach SD. Hedgehog signaling is required for effective regeneration of exocrine pancreas. Gastroenterology. 2008;135:621-631. [PubMed] [DOI] [Full Text] |
| 34. | Zhou X, Liu Z, Jang F, Xiang C, Li Y, He Y. Autocrine Sonic hedgehog attenuates inflammation in cerulein-induced acute pancreatitis in mice via upregulation of IL-10. PLoS One. 2012;7:e44121. [PubMed] [DOI] [Full Text] |
| 35. | Mathew E, Collins MA, Fernandez-Barrena MG, Holtz AM, Yan W, Hogan JO, Tata Z, Allen BL, Fernandez-Zapico ME, di Magliano MP. The transcription factor GLI1 modulates the inflammatory response during pancreatic tissue remodeling. J Biol Chem. 2014;289:27727-27743. [PubMed] [DOI] [Full Text] |
| 36. | Kayed H, Kleeff J, Esposito I, Giese T, Keleg S, Giese N, Büchler MW, Friess H. Localization of the human hedgehog-interacting protein (Hip) in the normal and diseased pancreas. Mol Carcinog. 2005;42:183-192. [PubMed] [DOI] [Full Text] |
| 37. | Kayed H, Kleeff J, Keleg S, Büchler MW, Friess H. Distribution of Indian hedgehog and its receptors patched and smoothened in human chronic pancreatitis. J Endocrinol. 2003;178:467-478. [PubMed] |
| 38. | Shinozaki S, Ohnishi H, Hama K, Kita H, Yamamoto H, Osawa H, Sato K, Tamada K, Mashima H, Sugano K. Indian hedgehog promotes the migration of rat activated pancreatic stellate cells by increasing membrane type-1 matrix metalloproteinase on the plasma membrane. J Cell Physiol. 2008;216:38-46. [PubMed] [DOI] [Full Text] |
| 39. | Kolterud A, Grosse AS, Zacharias WJ, Walton KD, Kretovich KE, Madison BB, Waghray M, Ferris JE, Hu C, Merchant JL. Paracrine Hedgehog signaling in stomach and intestine: new roles for hedgehog in gastrointestinal patterning. Gastroenterology. 2009;137:618-628. [PubMed] [DOI] [Full Text] |
| 40. | Bailey JM, Swanson BJ, Hamada T, Eggers JP, Singh PK, Caffery T, Ouellette MM, Hollingsworth MA. Sonic hedgehog promotes desmoplasia in pancreatic cancer. Clin Cancer Res. 2008;14:5995-6004. [PubMed] [DOI] [Full Text] |
| 41. | Wicking C, Smyth I, Bale A. The hedgehog signalling pathway in tumorigenesis and development. Oncogene. 1999;18:7844-7851. [PubMed] [DOI] [Full Text] |
| 42. | Jung IH, Jung DE, Park YN, Song SY, Park SW. Aberrant Hedgehog ligands induce progressive pancreatic fibrosis by paracrine activation of myofibroblasts and ductular cells in transgenic zebrafish. PLoS One. 2011;6:e27941. [PubMed] [DOI] [Full Text] |
| 43. | Tsang SW, Zhang H, Lin C, Xiao H, Wong M, Shang H, Yang ZJ, Lu A, Yung KK, Bian Z. Rhein, a natural anthraquinone derivative, attenuates the activation of pancreatic stellate cells and ameliorates pancreatic fibrosis in mice with experimental chronic pancreatitis. PLoS One. 2013;8:e82201. [PubMed] [DOI] [Full Text] |
| 44. | Lin N, Tang Z, Deng M, Zhong Y, Lin J, Yang X, Xiang P, Xu R. Hedgehog-mediated paracrine interaction between hepatic stellate cells and marrow-derived mesenchymal stem cells. Biochem Biophys Res Commun. 2008;372:260-265. [PubMed] [DOI] [Full Text] |
| 45. | Omenetti A, Porrello A, Jung Y, Yang L, Popov Y, Choi SS, Witek RP, Alpini G, Venter J, Vandongen HM. Hedgehog signaling regulates epithelial-mesenchymal transition during biliary fibrosis in rodents and humans. J Clin Invest. 2008;118:3331-3342. [PubMed] [DOI] [Full Text] |
| 46. | Stewart GA, Hoyne GF, Ahmad SA, Jarman E, Wallace WA, Harrison DJ, Haslett C, Lamb JR, Howie SE. Expression of the developmental Sonic hedgehog (Shh) signalling pathway is up-regulated in chronic lung fibrosis and the Shh receptor patched 1 is present in circulating T lymphocytes. J Pathol. 2003;199:488-495. [PubMed] [DOI] [Full Text] |
| 47. | Blaylock RL. Cancer microenvironment, inflammation and cancer stem cells: A hypothesis for a paradigm change and new targets in cancer control. Surg Neurol Int. 2015;6:92. [PubMed] [DOI] [Full Text] |
| 48. | Ling SB, Feng TT, Jia KQ, Tian Y, Li Y. Inflammation to cancer: The molecular biology in the pancreas (Review). Oncol Lett. 2014;7:1747-1754. [PubMed] [DOI] [Full Text] |
| 49. | Stewart GA, Lowrey JA, Wakelin SJ, Fitch PM, Lindey S, Dallman MJ, Lamb JR, Howie SE. Sonic hedgehog signaling modulates activation of and cytokine production by human peripheral CD4+ T cells. J Immunol. 2002;169:5451-5457. [PubMed] |
| 50. | Strobel O, Rosow DE, Rakhlin EY, Lauwers GY, Trainor AG, Alsina J, Fernández-Del Castillo C, Warshaw AL, Thayer SP. Pancreatic duct glands are distinct ductal compartments that react to chronic injury and mediate Shh-induced metaplasia. Gastroenterology. 2010;138:1166-1177. [PubMed] [DOI] [Full Text] |
| 51. | Neureiter D, Zopf S, Dimmler A, Stintzing S, Hahn EG, Kirchner T, Herold C, Ocker M. Different capabilities of morphological pattern formation and its association with the expression of differentiation markers in a xenograft model of human pancreatic cancer cell lines. Pancreatology. 2005;5:387-397. [PubMed] [DOI] [Full Text] |
| 52. | Quint K, Stintzing S, Alinger B, Hauser-Kronberger C, Dietze O, Gahr S, Hahn EG, Ocker M, Neureiter D. The expression pattern of PDX-1, SHH, Patched and Gli-1 is associated with pathological and clinical features in human pancreatic cancer. Pancreatology. 2009;9:116-126. [PubMed] [DOI] [Full Text] |
| 53. | DI Fazio P, Montalbano R, Quint K, Alinger B, Kemmerling R, Kiesslich T, Ocker M, Neureiter D. The pan-deacetylase inhibitor panobinostat modulates the expression of epithelial-mesenchymal transition markers in hepatocellular carcinoma models. Oncol Lett. 2013;5:127-134. [PubMed] [DOI] [Full Text] |
| 54. | Quint K, Tonigold M, Di Fazio P, Montalbano R, Lingelbach S, Rückert F, Alinger B, Ocker M, Neureiter D. Pancreatic cancer cells surviving gemcitabine treatment express markers of stem cell differentiation and epithelial-mesenchymal transition. Int J Oncol. 2012;41:2093-2102. [PubMed] [DOI] [Full Text] |
| 55. | Honselmann KC, Pross M, Jung CM, Wellner UF, Deichmann S, Keck T, Bausch D. Regulation mechanisms of the hedgehog pathway in pancreatic cancer: a review. JOP. 2015;16:25-32. [PubMed] [DOI] [Full Text] |
| 56. | Marini KD, Payne BJ, Watkins DN, Martelotto LG. Mechanisms of Hedgehog signalling in cancer. Growth Factors. 2011;29:221-234. [PubMed] [DOI] [Full Text] |
| 57. | Allegra A, Alonci A, Penna G, Innao V, Gerace D, Rotondo F, Musolino C. The cancer stem cell hypothesis: a guide to potential molecular targets. Cancer Invest. 2014;32:470-495. [PubMed] [DOI] [Full Text] |
| 58. | O’Connor ML, Xiang D, Shigdar S, Macdonald J, Li Y, Wang T, Pu C, Wang Z, Qiao L, Duan W. Cancer stem cells: A contentious hypothesis now moving forward. Cancer Lett. 2014;344:180-187. [PubMed] [DOI] [Full Text] |
| 59. | Chinembiri TN, du Plessis LH, Gerber M, Hamman JH, du Plessis J. Review of natural compounds for potential skin cancer treatment. Molecules. 2014;19:11679-11721. [PubMed] [DOI] [Full Text] |
| 60. | Li SH, Fu J, Watkins DN, Srivastava RK, Shankar S. Sulforaphane regulates self-renewal of pancreatic cancer stem cells through the modulation of Sonic hedgehog-GLI pathway. Mol Cell Biochem. 2013;373:217-227. [PubMed] [DOI] [Full Text] |
| 61. | Chen JK, Taipale J, Cooper MK, Beachy PA. Inhibition of Hedgehog signaling by direct binding of cyclopamine to Smoothened. Genes Dev. 2002;16:2743-2748. [PubMed] [DOI] [Full Text] |
| 62. | Walter K, Omura N, Hong SM, Griffith M, Vincent A, Borges M, Goggins M. Overexpression of smoothened activates the sonic hedgehog signaling pathway in pancreatic cancer-associated fibroblasts. Clin Cancer Res. 2010;16:1781-1789. [PubMed] [DOI] [Full Text] |
| 63. | Rosow DE, Liss AS, Strobel O, Fritz S, Bausch D, Valsangkar NP, Alsina J, Kulemann B, Park JK, Yamaguchi J. Sonic Hedgehog in pancreatic cancer: from bench to bedside, then back to the bench. Surgery. 2012;152:S19-S32. [PubMed] [DOI] [Full Text] |
| 64. | Tian H, Callahan CA, DuPree KJ, Darbonne WC, Ahn CP, Scales SJ, de Sauvage FJ. Hedgehog signaling is restricted to the stromal compartment during pancreatic carcinogenesis. Proc Natl Acad Sci USA. 2009;106:4254-4259. [PubMed] [DOI] [Full Text] |
| 65. | Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277-300. [PubMed] [DOI] [Full Text] |
| 66. | Li J, Wientjes MG, Au JL. Pancreatic cancer: pathobiology, treatment options, and drug delivery. AAPS J. 2010;12:223-232. [PubMed] [DOI] [Full Text] |
| 67. | Thayer SP, di Magliano MP, Heiser PW, Nielsen CM, Roberts DJ, Lauwers GY, Qi YP, Gysin S, Fernández-del Castillo C, Yajnik V. Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature. 2003;425:851-856. [PubMed] [DOI] [Full Text] |
| 68. | Jones S, Zhang X, Parsons DW, Lin JC, Leary RJ, Angenendt P, Mankoo P, Carter H, Kamiyama H, Jimeno A. Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. Science. 2008;321:1801-1806. [PubMed] [DOI] [Full Text] |
| 69. | Morton JP, Mongeau ME, Klimstra DS, Morris JP, Lee YC, Kawaguchi Y, Wright CV, Hebrok M, Lewis BC. Sonic hedgehog acts at multiple stages during pancreatic tumorigenesis. Proc Natl Acad Sci USA. 2007;104:5103-5108. [PubMed] [DOI] [Full Text] |
| 70. | Hingorani SR, Wang L, Multani AS, Combs C, Deramaudt TB, Hruban RH, Rustgi AK, Chang S, Tuveson DA. Trp53R172H and KrasG12D cooperate to promote chromosomal instability and widely metastatic pancreatic ductal adenocarcinoma in mice. Cancer Cell. 2005;7:469-483. [PubMed] [DOI] [Full Text] |
| 71. | Feldmann G, Dhara S, Fendrich V, Bedja D, Beaty R, Mullendore M, Karikari C, Alvarez H, Iacobuzio-Donahue C, Jimeno A. Blockade of hedgehog signaling inhibits pancreatic cancer invasion and metastases: a new paradigm for combination therapy in solid cancers. Cancer Res. 2007;67:2187-2196. [PubMed] [DOI] [Full Text] |
| 72. | Lauth M, Bergström A, Shimokawa T, Tostar U, Jin Q, Fendrich V, Guerra C, Barbacid M, Toftgård R. DYRK1B-dependent autocrine-to-paracrine shift of Hedgehog signaling by mutant RAS. Nat Struct Mol Biol. 2010;17:718-725. [PubMed] [DOI] [Full Text] |
| 73. | Hingorani SR, Petricoin EF, Maitra A, Rajapakse V, King C, Jacobetz MA, Ross S, Conrads TP, Veenstra TD, Hitt BA. Preinvasive and invasive ductal pancreatic cancer and its early detection in the mouse. Cancer Cell. 2003;4:437-450. [PubMed] [DOI] [Full Text] |
| 74. | Collins MA, Bednar F, Zhang Y, Brisset JC, Galbán S, Galbán CJ, Rakshit S, Flannagan KS, Adsay NV, Pasca di Magliano M. Oncogenic Kras is required for both the initiation and maintenance of pancreatic cancer in mice. J Clin Invest. 2012;122:639-653. [PubMed] [DOI] [Full Text] |
| 75. | Merchant A, Joseph G, Wang Q, Brennan S, Matsui W. Gli1 regulates the proliferation and differentiation of HSCs and myeloid progenitors. Blood. 2010;115:2391-2396. [PubMed] [DOI] [Full Text] |
| 76. | Ji Z, Mei FC, Xie J, Cheng X. Oncogenic KRAS activates hedgehog signaling pathway in pancreatic cancer cells. J Biol Chem. 2007;282:14048-14055. [PubMed] [DOI] [Full Text] |
| 77. | Mills LD, Zhang Y, Marler RJ, Herreros-Villanueva M, Zhang L, Almada LL, Couch F, Wetmore C, Pasca di Magliano M, Fernandez-Zapico ME. Loss of the transcription factor GLI1 identifies a signaling network in the tumor microenvironment mediating KRAS oncogene-induced transformation. J Biol Chem. 2013;288:11786-11794. [PubMed] [DOI] [Full Text] |
| 78. | Fukuda A, Wang SC, Morris JP, Folias AE, Liou A, Kim GE, Akira S, Boucher KM, Firpo MA, Mulvihill SJ. Stat3 and MMP7 contribute to pancreatic ductal adenocarcinoma initiation and progression. Cancer Cell. 2011;19:441-455. [PubMed] [DOI] [Full Text] |
| 79. | Lesina M, Kurkowski MU, Ludes K, Rose-John S, Treiber M, Klöppel G, Yoshimura A, Reindl W, Sipos B, Akira S. Stat3/Socs3 activation by IL-6 transsignaling promotes progression of pancreatic intraepithelial neoplasia and development of pancreatic cancer. Cancer Cell. 2011;19:456-469. [PubMed] [DOI] [Full Text] |
| 80. | Lee JJ, Perera RM, Wang H, Wu DC, Liu XS, Han S, Fitamant J, Jones PD, Ghanta KS, Kawano S. Stromal response to Hedgehog signaling restrains pancreatic cancer progression. Proc Natl Acad Sci USA. 2014;111:E3091-E3100. [PubMed] [DOI] [Full Text] |
| 81. | Rhim AD, Mirek ET, Aiello NM, Maitra A, Bailey JM, McAllister F, Reichert M, Beatty GL, Rustgi AK, Vonderheide RH. EMT and dissemination precede pancreatic tumor formation. Cell. 2012;148:349-361. [PubMed] [DOI] [Full Text] |
| 82. | Nakashima H, Nakamura M, Yamaguchi H, Yamanaka N, Akiyoshi T, Koga K, Yamaguchi K, Tsuneyoshi M, Tanaka M, Katano M. Nuclear factor-kappaB contributes to hedgehog signaling pathway activation through sonic hedgehog induction in pancreatic cancer. Cancer Res. 2006;66:7041-7049. [PubMed] [DOI] [Full Text] |
| 83. | Yauch RL, Gould SE, Scales SJ, Tang T, Tian H, Ahn CP, Marshall D, Fu L, Januario T, Kallop D. A paracrine requirement for hedgehog signalling in cancer. Nature. 2008;455:406-410. [PubMed] [DOI] [Full Text] |
| 84. | Olive KP, Jacobetz MA, Davidson CJ, Gopinathan A, McIntyre D, Honess D, Madhu B, Goldgraben MA, Caldwell ME, Allard D. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science. 2009;324:1457-1461. [PubMed] [DOI] [Full Text] |
| 85. | Singh AP, Arora S, Bhardwaj A, Srivastava SK, Kadakia MP, Wang B, Grizzle WE, Owen LB, Singh S. CXCL12/CXCR4 protein signaling axis induces sonic hedgehog expression in pancreatic cancer cells via extracellular regulated kinase- and Akt kinase-mediated activation of nuclear factor κB: implications for bidirectional tumor-stromal interactions. J Biol Chem. 2012;287:39115-39124. [PubMed] [DOI] [Full Text] |
| 86. | Bailey JM, Mohr AM, Hollingsworth MA. Sonic hedgehog paracrine signaling regulates metastasis and lymphangiogenesis in pancreatic cancer. Oncogene. 2009;28:3513-3525. [PubMed] [DOI] [Full Text] |
| 87. | Sun X, Cheng G, Hao M, Zheng J, Zhou X, Zhang J, Taichman RS, Pienta KJ, Wang J. CXCL12/CXCR4/CXCR7 chemokine axis and cancer progression. Cancer Metastasis Rev. 2010;29:709-722. [PubMed] [DOI] [Full Text] |
| 88. | Hermann PC, Huber SL, Herrler T, Aicher A, Ellwart JW, Guba M, Bruns CJ, Heeschen C. Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell. 2007;1:313-323. [PubMed] [DOI] [Full Text] |
| 89. | Mayr C, Neureiter D, Pichler M, Berr F, Wagner A, Kiesslich T, Namberger K. Cytotoxic effects of chemokine receptor 4 inhibition by AMD3100 in biliary tract cancer cells: Potential drug synergism with gemcitabine. Mol Med Rep. 2015;12:2247-2252. [PubMed] [DOI] [Full Text] |
| 90. | Maréchal R, Demetter P, Nagy N, Berton A, Decaestecker C, Polus M, Closset J, Devière J, Salmon I, Van Laethem JL. High expression of CXCR4 may predict poor survival in resected pancreatic adenocarcinoma. Br J Cancer. 2009;100:1444-1451. [PubMed] [DOI] [Full Text] |
| 91. | Matsuo Y, Ochi N, Sawai H, Yasuda A, Takahashi H, Funahashi H, Takeyama H, Tong Z, Guha S. CXCL8/IL-8 and CXCL12/SDF-1alpha co-operatively promote invasiveness and angiogenesis in pancreatic cancer. Int J Cancer. 2009;124:853-861. [PubMed] [DOI] [Full Text] |
| 92. | Lee CJ, Li C, Simeone DM. Human pancreatic cancer stem cells: implications for how we treat pancreatic cancer. Transl Oncol. 2008;1:14-18. [PubMed] |
| 93. | Kashyap V, Rezende NC, Scotland KB, Shaffer SM, Persson JL, Gudas LJ, Mongan NP. Regulation of stem cell pluripotency and differentiation involves a mutual regulatory circuit of the NANOG, OCT4, and SOX2 pluripotency transcription factors with polycomb repressive complexes and stem cell microRNAs. Stem Cells Dev. 2009;18:1093-1108. [PubMed] [DOI] [Full Text] |
| 94. | Mueller MT, Hermann PC, Witthauer J, Rubio-Viqueira B, Leicht SF, Huber S, Ellwart JW, Mustafa M, Bartenstein P, D’Haese JG. Combined targeted treatment to eliminate tumorigenic cancer stem cells in human pancreatic cancer. Gastroenterology. 2009;137:1102-1113. [PubMed] [DOI] [Full Text] |
| 95. | Mayr C, Neureiter D, Wagner A, Pichler M, Kiesslich T. The role of polycomb repressive complexes in biliary tract cancer. Expert Opin Ther Targets. 2015;19:363-375. [PubMed] [DOI] [Full Text] |
| 96. | Li C, Heidt DG, Dalerba P, Burant CF, Zhang L, Adsay V, Wicha M, Clarke MF, Simeone DM. Identification of pancreatic cancer stem cells. Cancer Res. 2007;67:1030-1037. [PubMed] [DOI] [Full Text] |
| 97. | Inaguma S, Riku M, Hashimoto M, Murakami H, Saga S, Ikeda H, Kasai K. GLI1 interferes with the DNA mismatch repair system in pancreatic cancer through BHLHE41-mediated suppression of MLH1. Cancer Res. 2013;73:7313-7323. [PubMed] [DOI] [Full Text] |
| 98. | Wang X, Venugopal C, Manoranjan B, McFarlane N, O’Farrell E, Nolte S, Gunnarsson T, Hollenberg R, Kwiecien J, Northcott P. Sonic hedgehog regulates Bmi1 in human medulloblastoma brain tumor-initiating cells. Oncogene. 2012;31:187-199. [PubMed] [DOI] [Full Text] |
| 99. | Po A, Ferretti E, Miele E, De Smaele E, Paganelli A, Canettieri G, Coni S, Di Marcotullio L, Biffoni M, Massimi L. Hedgehog controls neural stem cells through p53-independent regulation of Nanog. EMBO J. 2010;29:2646-2658. [PubMed] [DOI] [Full Text] |
| 100. | Robarge KD, Brunton SA, Castanedo GM, Cui Y, Dina MS, Goldsmith R, Gould SE, Guichert O, Gunzner JL, Halladay J. GDC-0449-a potent inhibitor of the hedgehog pathway. Bioorg Med Chem Lett. 2009;19:5576-5581. [PubMed] [DOI] [Full Text] |
| 101. | Cooper MK, Porter JA, Young KE, Beachy PA. Teratogen-mediated inhibition of target tissue response to Shh signaling. Science. 1998;280:1603-1607. [PubMed] [DOI] [Full Text] |
| 102. | Pan S, Wu X, Jiang J, Gao W, Wan Y, Cheng D, Han D, Liu J, Englund NP, Wang Y. Discovery of NVP-LDE225, a Potent and Selective Smoothened Antagonist. ACS Med Chem Lett. 2010;1:130-134. [PubMed] [DOI] [Full Text] |
| 103. | Tremblay MR, Lescarbeau A, Grogan MJ, Tan E, Lin G, Austad BC, Yu LC, Behnke ML, Nair SJ, Hagel M. Discovery of a potent and orally active hedgehog pathway antagonist (IPI-926). J Med Chem. 2009;52:4400-4418. [PubMed] [DOI] [Full Text] |
| 104. | Lauth M, Bergstrom A, Shimokawa T, Toftgard R. Inhibition of GLI-mediated transcription and tumor cell growth by small-molecule antagonists. Proc Natl Acad Sci USA. 2007;104:8455-8460. [PubMed] [DOI] [Full Text] |