Published online Jul 28, 2016. doi: 10.4329/wjr.v8.i7.656
Peer-review started: January 23, 2016
First decision: March 24, 2016
Revised: April 11, 2016
Accepted: June 1, 2016
Article in press: June 3, 2016
Published online: July 28, 2016
Processing time: 184 Days and 16.1 Hours
Ultrasound is an invaluable imaging modality in the evaluation of pediatric gastrointestinal pathology; it can provide real-time evaluation of the bowel without the need for sedation or intravenous contrast. Recent improvements in ultrasound technique can be utilized to improve detection of bowel pathology in children: Higher resolution probes, color Doppler, harmonic and panoramic imaging are excellent tools in this setting. Graded compression and cine clips provide dynamic information and oral and intravenous contrast agents aid in detection of bowel wall pathology. Ultrasound of the bowel in children is typically a targeted exam; common indications include evaluation for appendicitis, pyloric stenosis and intussusception. Bowel abnormalities that are detected prenatally can be evaluated after birth with ultrasound. Likewise, acquired conditions such as bowel hematoma, bowel infections and hernias can be detected with ultrasound. Rare bowel neoplasms, vascular disorders and foreign bodies may first be detected with sonography, as well. At some centers, comprehensive exams of the gastrointestinal tract are performed on children with inflammatory bowel disease and celiac disease to evaluate for disease activity or to confirm the diagnosis. The goal of this article is to review up-to-date imaging techniques, normal sonographic anatomy, and characteristic sonographic features of common and uncommon disorders affecting the gastrointestinal tract in children.
Core tip: Ultrasound is increasingly utilized to evaluate gastrointestinal disorders in children. Recent improvements in ultrasound technique allow detailed evaluation of bowel pathology. We present a comprehensive review of bowel pathology in children with emphasis on ultrasonographic technique and findings. This review will describe the variety of sonographic techniques available to optimize assessment of bowel disease and sonographic features of normal bowel will be described. Common and uncommon disorders of bowel in children will include congenital, acquired, inflammatory and neoplastic processes.
- Citation: Gale HI, Gee MS, Westra SJ, Nimkin K. Abdominal ultrasonography of the pediatric gastrointestinal tract. World J Radiol 2016; 8(7): 656-667
- URL: https://www.wjgnet.com/1949-8470/full/v8/i7/656.htm
- DOI: https://dx.doi.org/10.4329/wjr.v8.i7.656
Ultrasound is an ideal imaging modality in the pediatric population because it is a real-time, non-invasive, relatively low cost examination without ionizing radiation that requires no sedation. Several recent reviews have emphasized the utility of ultrasound in the evaluation of pediatric bowel pathology[1-3]. Ultrasound of the bowel in children is typically a targeted examination, designed to answer a specific question, and common indications include evaluation for appendicitis, intussusception, and pyloric stenosis. Other focused examinations include evaluation of congenital abnormalities detected prenatally, confirmation of suspected hernia, and problem solving in the patient with necrotizing enterocolitis (NEC). Unsuspected bowel abnormalities may be found during screening for non-specific abdominal pain, including foreign body, tumor, infection, or bowel hematoma. A more comprehensive examination of the entire bowel is used at some centers to evaluate inflammatory bowel disease (IBD) and celiac disease in children.
Ultrasound examinations are typically performed with the patient supine without any preparation. Recent improvements in ultrasound technology, including high-resolution linear probes (12-15 MHz) and harmonic and panoramic imaging, improve image quality[3,4]. Color Doppler evaluation can detect increased perfusion in inflamed loops of bowel. Ultrasound cine clips document bowel motility, and graded compression assesses compressibility and improves resolution by displacing air from the bowel lumen. Oral administration of non-carbonated fluid 30 min prior to the examination will reduce air in the bowel[4]. Other promising newer techniques include oral contrast agents, such as iso-osmolar polyethylene glycol (PEG), to improve bowel distension, referred to as small-intestine contrast enhanced ultrasound[5]. Intravenous contrast agents are not approved for children but are increasingly utilized off-label, particularly in pediatric patients with IBD[4,6]. The pattern of contrast enhancement has been useful to assess disease activity and adjacent inflammatory changes[7]. Lastly, bowel elastography may have applications in the assessment of bowel wall edema and/or fibrosis, particularly in IBD[8].
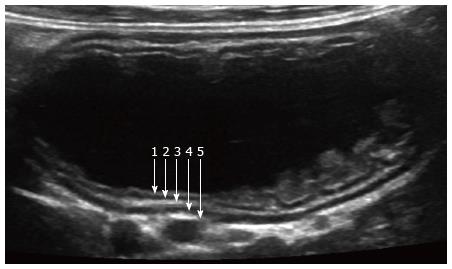
Normal bowel loops have a stratified pattern on high-resolution ultrasound with the following 5 layers: Mucosal interface with lumen (hyperechoic), mucosa (hypoechoic), submucosa (hyperechoic), muscularis (hypoechoic) and serosa (hyperechoic) (Figure 1). Typically, however, only 2 layers are visible on ultrasound, including an inner hyperechoic layer and outer hypoechoic layer. In normal children, small bowel loops are compressible, show minimal vascularity, and have wall thickness < 2.5 mm[9]. Jejunal loops have more folds and peristalse more than ileum, and the colon contains more air, fewer folds, and wall thickness is < 2 mm[9].
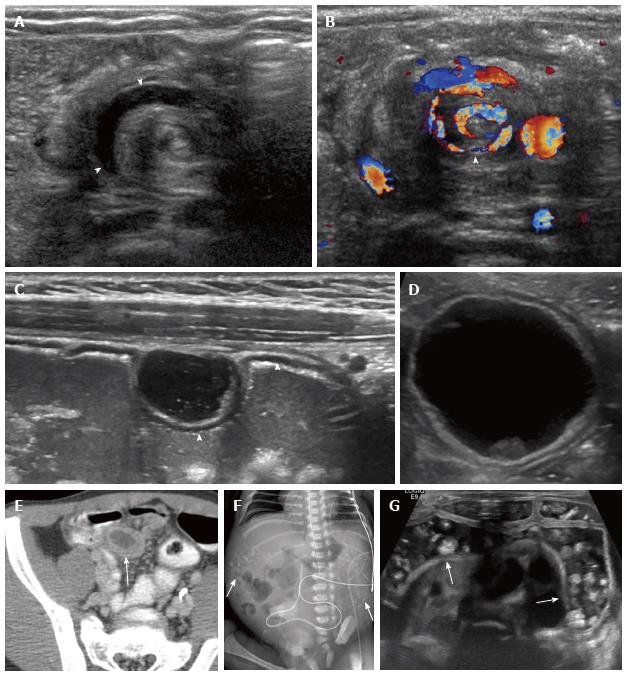
Intestinal malrotation: Intestinal malrotation occurs when the midgut does not undergo its expected rotation around the axis of the superior mesenteric artery during fetal development[10]. Symptoms of malrotation are most commonly caused by volvulus or obstructing peritoneal bands, which typically manifest during the first year of life[10]. Ultrasound may be performed in the vomiting infant to evaluate for pyloric stenosis and malrotation may be an unexpected finding (Figure 2). On ultrasound, there is usually reversal of the position of the superior mesenteric artery (SMA) and superior mesenteric vein (SMV). When volvulus is present, transverse sonographic images show dilated fluid-filled duodenum with alternating rings of low and high echogenicity at the base of the mesentery (“concentric circle sign”)[11]. Color Doppler ultrasound can reveal a spiral appearance of the mesenteric vessels, termed the” whirlpool sign”[10,11]. There may be dilatation of the distal SMV[12]. Some authors advocate ultrasound evaluation of the 3rd portion of the duodenum to confirm its location behind the SMA to exclude malrotation, however, this has not found general application[13].
Gastrointestinal duplication cyst: Gastrointestinal duplication cyst is an additional segment of fetal gut that can occur from the esophagus to the rectum, most commonly at the terminal ileum[14-17]. Gastrointestinal duplication cysts demonstrate a connection with the gastrointestinal (GI) tract by a common wall of serous and muscle membrane, usually without luminal communication, and may contain ectopic pancreatic tissue[15-17]. Complications include ulceration, hemorrhage, perforation, and inflammation[15].
On ultrasound, gastrointestinal duplication cysts are fluid-filled structures, typically with a central anechoic component[15]. The mucosal and submuscosal layers are echogenic, and the shared muscularis layer is hypoechoic[15,17] (Figure 2C). Rarely, other abdominal cysts may have a “pseudo gut signature”, including mesenteric cysts and teratomas; high-resolution transducers should delineate multiple bowel wall layers in true duplication cysts[18,19]. Further characterization can be performed with Tc-99m nuclear scintigraphy, which targets parietal cells in gastric mucosa[15].
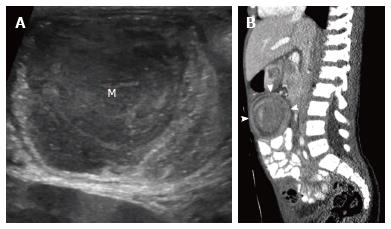
Meckel diverticulum: Meckel diverticulum is the most common malformation of the small bowel, which results from partial or complete failure of involution of the omphalomesenteric duct[10]. It is a true diverticulum that contains all layers of the intestinal wall, and it may contain heterotopic gastric and pancreatic mucosa[10] (Figure 2). It is seen in 0.3%-3% of the population, and approximately 2%-4% of affected patients become symptomatic[10]. Complications include bleeding, small bowel obstruction, inflammation (Meckel diverticulitis), and neoplasm[10]. Sonographic imaging findings are reflective of the specific complication, and can include wall thickening, intussusception, and associated mass[10]. A surrounding hyperemic and echogenic layer is suggestive of associated perforation[20].
Annular pancreas: Annular pancreas is a rare congenital abnormality that can present in childhood with duodenal obstruction or pancreatitis[21]. In the vomiting infant, ultrasound may show narrowing of the 2nd portion of the duodenum, with a surrounding ring of pancreatic tissue. The anomalous branch of the pancreatic duct may be seen on ultrasound coursing obliquely and to the right, anterior to the duodenum[21,22].
Rectourinary fistula: Rectovesical or rectourethral fistula typically occurs in patients with an anorectal malformation such as imperforate anus[23,24]. Neonates with rectourinary fistula may develop enterolithiasis due to mixing of meconium and urine[23,24]. Enterolithiasis appears as calcifications on radiographs, and can be further evaluated with high-frequency, high-resolution real time ultrasound to confirm intraluminal location and distinguish this entity from meconium peritonitis[24] (Figure 2). Enterolithiasis can also be seen in other cases of intestinal obstruction such as ileal stenosis, jejunal atresia, and functional obstruction of the ileum[23,24]. Transperineal ultrasonography can also be performed in patients with anal atresia to identify the internal fistula[25].
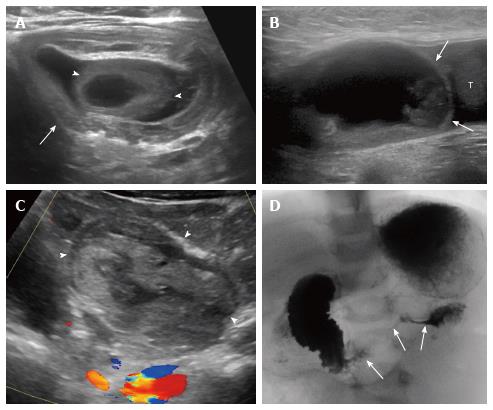
Intussusception: Intussusception is the most common cause of bowel obstruction in children, and it typically occurs between ages 6 mo and 2 years. The most common type is ileocolic, and most cases are idiopathic. Ultrasound is critical for a prompt and accurate diagnosis of intussusception, and has nearly 100% sensitivity for detection[26]. Imaging features of intussusception are characteristic, described as the “pseudokidney” or “donut sign”, with alternating hyperechoic and hypoechoic concentric layers. Fluid trapped between layers of the intussusception and absence of color flow may reflect decreased likelihood of reduction and bowel ischemia. Lead points are typically seen in older children and may be detected by ultrasound, including Meckel diverticulum, duplication cyst, juvenile polyp, and lymphoma (Figure 3A). Ileo-ileocolic intussusception is associated with decreased reduction rate and increased morbidity[27].
It is critical to differentiate ileocolic intussusception from small bowel-small bowel intussusception, as the latter are typically managed conservatively and air reduction is not indicated. A recent review noted that larger intussusception diameter and the presence of lymph nodes within the intussusception favored ileocolic intussusception[28]. In one review, mean AP diameter of ileocolic intussusception was 2.53 cm compared to 1.38 cm of small bowel intussusception[29]. Small bowel intussusceptions have very little fat centrally and occur in older children with bowel disorders such as Henoch-Schonlein Purpura, Crohn disease, and celiac sprue; they are also seen in post-operative patients and in patients with small bowel mass acting as a lead point. Small bowel intussusception length greater than 3.5 cm is a strong predictor of need for surgical intervention[30]. However, most small bowel intussusceptions are idiopathic and transient[31].
Hypertrophic pyloric stenosis: Hypertrophic pyloric stenosis (HPS) is an idiopathic cause of gastric outlet obstruction, which typically occurs during the 2nd to 7th week of life and is more common in boys than girls[1,32,33]. Ultrasound is performed in supine and right lateral decubitus positions with a high-frequency linear-array transducer (12-5 MHz). If sufficient fluid is not present within the stomach to outline the antrum and pylorus, 1-2 ounces of sugar water can be given orally. Axial sonographic images demonstrate the “donut sign”, characterized by a rim of thickened muscle and an echogenic center of mucosa and submucosa[32]. In longitudinal plane, the pylorus remains closed and no fluid passes into the duodenum[32,34]. The mucosa can protrude into the distended distal gastric antrum, creating the “nipple sign”[32,34].
Current guidelines for ultrasound diagnosis of HPS are pyloric muscle thickness > 3 mm, pyloric length > 15 mm, pyloric diameter > 11 mm, and pyloric volume > 12 mL[1,35]. Patient age and weight correlate with pyloric muscle wall thickness, and a lower ultrasound threshold for diagnosis should be used in smaller neonates[36,37]. Imaging the pylorus over time allows the differentiation of HPS and pylorospasm, the latter being a transient phenomenon[3]. Follow-up can be utilized in equivocal cases[3].
Inguinal hernia: Ultrasound is 95.5% accurate for detecting inguinal hernias in boys[38]. The internal ring is measured at rest and with straining (standing, crying, coughing, or bearing down)[38]. In boys of any age, inguinal canal diameter > 4 mm at the internal ring (width of the spermatic cord) is 95% accurate in diagnosing inguinal hernia at surgery[39,40]. Fluid in the processus vaginalis or bowel loops/other peritoneal structures within the inguinal canal are also diagnostic of hernia[39,41]. Contralateral hernias occur in up to 22.4% of patients, and bilateral ultrasonography can guide pre-operative planning[42].
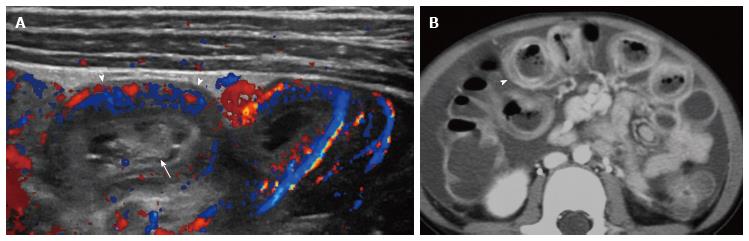
Indirect hernias, the most common type of inguinal hernia in children, occur superolateral to the epigastric vessels, direct hernias occur inferomedial to the epigastric vessels, and femoral hernias occur below the inguinal ligament[38,43]. In the case of herniated bowel loops, ultrasound is used to assess bowel peristalsis, wall thickness, and vascularity. Incarcerated hernias may not show clear continuity with abdominal bowel loops (Figure 3B). Inguinal hernias can compress gonadal vessels and cause testicular hypovascularity and enlargement on ultrasound[44].
Hiatal hernia and gastroesophageal reflux disease: To evaluate for hiatal hernia and gastroesophageal reflux disease (GERD) with ultrasound, the transducer is placed inferior to the xiphoid process in sagittal plane and directed cranially. The diameter of the esophageal hiatus is measured in transverse plane using the liver as an acoustic window[45]. Esophageal hiatal diameters have been shown to be greater in patients with hiatal hernias compared to control subjects[45]. Absence of paraesophageal fat may be a more reliable indicator than hiatal widening because it is not affected by age, obesity, or BMI[46].
Although ultrasound is not recommended for evaluation of GERD, it can be used in cases of unusual posturing or aspiration, because episodes of retrograde passage of gastric contents detected on ultrasound can be correlated temporally with symptoms[47,48]. It can be helpful to detect GERD in an infant with suspected HPS and a normal pylorus. A short intra-abdominal segment of esophagus and/or a wide esophageal angle have been shown to be associated with reflux[48,49].
Duodenal intramural hematoma: Duodenal hematomas in children are typically post-traumatic. If there is no history of trauma, there is a high association with child abuse and additional imaging is warranted (Figure 3). Hematomas may also result from endoscopic biopsy of the duodenum or in children with bleeding disorders[50-52]. Once identified, the hematoma can persist for at least two weeks, typically resolving by 6 wk. On ultrasound, duodenal intramural hematomas appear as a heterogeneous, nonvascularized mass along the course of the duodenum, which can obstruct the duodenal lumen and/or the common bile duct[50-52]. During resolution, the hematoma becomes cystic. Differential diagnostic considerations include duodenal duplication, abscess, pancreatic pseudocyst, or tumor. Ultrasound is also useful for serial follow-up to document either resolution or worsening obstruction requiring intervention.
Appendicitis: Appendicitis is one of the most common surgical emergencies in children, and delay in diagnosis can result in morbidity from an associated complication such as appendiceal rupture or bowel obstruction[53,54]. Non-operative management of acute uncomplicated appendicitis in children is also used in select cases[55,56]. Symptoms of acute appendicitis are variable and can include periumbilical and/or right lower quadrant pain, anorexia, nausea, fever, and leukocytosis[54,57].
Ultrasound is the first imaging choice for suspected appendicitis at most centers[53,57]. Both grayscale and color Doppler imaging are utilized with 5-MHz curved, 9-MHz linear, or 15-MHz linear transducers[58]. Ultrasound is 88% sensitive and 94% specific for the diagnosis of acute appendicitis[59]. Diagnostic criteria for appendicitis include appendiceal diameter > 6 mm (outer wall to outer wall) and associated evidence of inflammation including appendiceal non-compressibility, wall thickening > 2 mm or hyperemia, fluid-filled appendix, increased echogenicity of periappendiceal fat, and/or presence of periappendiceal fluid[58]. Ultrasound diagnosis of perforated appendicitis is made by the presence of marked inflammatory changes in the right lower quadrant with or without visualization of the appendix, an appendicolith without visualization of the appendix, echogenic free fluid, or a fluid collection indicating peritonitis or abscess[58].
Equivocal findings on ultrasound are associated with surgical appendicitis in 12.5%-50% of cases[58,59]. Increasing the size threshold to 7.5-8 mm in equivocal cases has been shown to increase specificity and accuracy[58,60]. Children at low risk for appendicitis with equivocal ultrasound findings are amenable to observation and reassessment[59]. When the patient’s white blood cell count is < 11.0 × 109/L, a non-diagnostic ultrasound or non-visualized appendix on ultrasound are associated with negative predictive values of 95.59% and 96.99%, respectively[61].
Necrotizing enterocolitis: Necrotizing enterocolitis (NEC) is a common cause of morbidity and mortality in premature infants. In NEC, there is bowel necrosis of unknown etiology; mucosal integrity may be compromised, leading to pneumatosis and portal venous gas[62]. The clinical presentation ranges from feeding intolerance or abdominal distension to fulminant shock and death[63]. Indications for surgery in NEC are pneumoperitoneum and deterioration with medical treatment alone. Patients with bowel necrosis may also benefit from surgery, and ultrasound has been shown to be 100% sensitive and 95.4% specific identifying necrosis[63].
Radiographs are the primary imaging tool when evaluating for NEC; ultrasound can be used as a problem-solving tool in select cases when surgery is considered. For diagnosis of NEC, ultrasound evaluates for (1) wall hyperechogenicity (greater than anterior abdominal wall musculature); (2) wall thickening (≥ 3 mm); (3) wall thinning (< 1 mm); (4) intramural gas; (5) hypervascularity; (6) hypovascularity; and (7) aperistalsis[63-66]. The peritoneal cavity is evaluated for ascites and fluid collections, and the portal venous system is evaluated for gas (Figure 4). Small amounts of free air may be more easily seen with ultrasound than with radiography[66]. In one recent review, poor outcome was associated with dilated and fluid-filled bowel, echogenic free fluid, focal fluid collections, increased bowel wall echogenicity, and increased bowel wall thickness[66]. Free intraperitoneal air and focal fluid collection predicted poor outcome in another series[64].
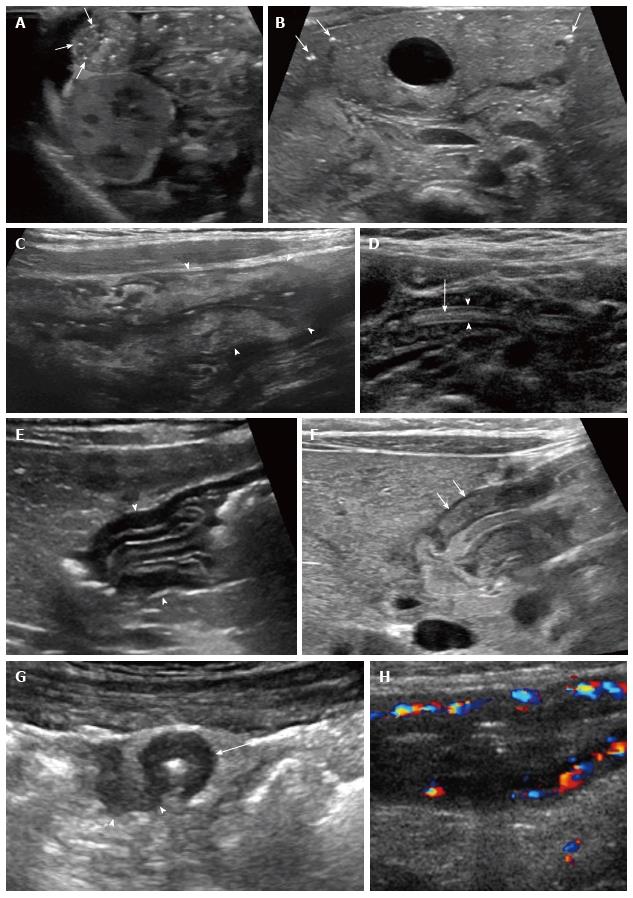
Infectious enteritis/typhlitis: Bacterial enterocolitis may be caused by a variety of pathogens, including Salmonella, Shigella, E. Coli and Campylobacter. Ultrasound findings include bowel wall thickening, hyperechogenicity, and hyperemia, usually in the terminal ileum and cecum[67] (Figure 4C). Adjacent lymph nodes, free fluid, and echogenic mesenteric fat are common. Viral gastroenteritis usually does not demonstrate bowel wall thickening, though ascites and enlarged nodes may be present[67]. Intestinal tuberculosis may show bowel wall thickening, typically with associated hepatosplenomegaly and omental thickening; findings may mimic Crohn disease[68,69]. Typhlitis, or inflammation of the cecum, is more frequently seen in immunocompromised patients and is characterized by marked thickening and hypervascularity; increased thickness of the wall may correlate with a worse prognosis[70,71]. Ascariasis infection can be detected with ultrasound; worms are mobile, tubular hypoechoic intraluminal structures with echogenic walls (Figure 4D). Parallel echogenic line or lines within the worm represent the digestive tract[72].
Allergic gastroenterocolitis: Allergic proctocolitis from cow’s milk allergy is the main cause of rectal bleeding in infants[73,74]. It occurs from early exposure to heterologous proteins such as cow’s milk or cow’s milk proteins derived from maternal breastfeeding. Ultrasound shows colitis with bowel wall thickening (≥ 3 mm) and increased vascularity, especially in the descending and sigmoid colon[73]. Increased Doppler vascularity is measured as 5 or more vessels in the bowel wall in a segment of approximately 2 cm2[73]. The most pronounced thickening is visualized in the mucosa, and the highest number of vessels is seen in the submucosa. In some cases bowel layers are not well defined. Allergic gastritis may mimic HPS on ultrasound; in allergic gastritis the mucosal and submucosal layers are thickened, while in HPS only the muscular layer is thickened[75] (Figure 4). In some patients, allergic gastritis and HPS may coexist[76].
Inflammatory bowel disease: Ultrasound has been found to have a high correlation with MR imaging findings in pediatric small bowel Crohn disease[77]. Ultrasound demonstrates mural thickening with loss of wall stratification, hyperemia, and decreased peristalsis. Fluid collections, fistulae, lymph nodes, and mesenteric inflammation can also be seen[7,9,78] (Figure 4). Strictures may be identified, associated with prestenotic dilatation, hyperperistalsis, and fecalization[9]. Small intestine contrast ultrasonongraphy, using oral administration of iso-osmolar PEG, improves evaluation of the small bowel in patients with Crohn disease[79]. Contrast enhanced ultrasound using IV administration of microbubble contrast shows promising results; the pattern of mural enhancement may aid in assessment of disease activity and/or response to therapy[80]. Ultrasound elastography, a technique that measures tissue stiffness, may help to differentiate inflammation from fibrosis in Crohn disease[77].
Ultrasound also has a role in evaluating ulcerative colitis. In children, the sensitivity and specificity of ultrasound for colonic inflammatory lesions is 88% and 93%, respectively[81]. Characteristic features include colonic and ileal wall thickening (≥ 3 mm), wall hypervascularity, loss of haustra coli, altered stratification of the bowel wall, and enlarged mesenteric lymph nodes[81].
Celiac disease: Celiac disease is an autoimmune malabsorptive enteropathy caused by gluten intolerance[82]. Ultrasound for celiac disease is performed with 5-2 MHz convex and 12-5 MHz linear transducers in the morning after fasting 10 h[82]. All abdominal quadrants are scanned with the lower frequency transducer for a preliminary survey followed by the higher frequency transducer for improved bowel detail[83]. Oral administration of 750 mL isotonic polyethylene glycol can improve visualization of bowel walls and fold pattern[83]. Ultrasound findings include dilated small bowel (> 2.5 cm including the wall), bowel wall thickening (≥ 3 mm), increased or decreased peristalsis, mesenteric lymphadenopathy, ascites, reversed jejunoileal fold pattern (effaced mucosa in the jejunum and thickened folds in the ileum), and small bowel-small bowel intussusception[82-86].
Ultrasound is the preferred study for the initial evaluation of suspected abdominal masses to determine the organ of origin and the characteristics of the mass in the pediatric population. GI tumors are rare in children, and benign tumors are more common than malignant tumors[87]. Benign lesions include polyps, hemangiomas, neurofibromas, leiomyomas, gastrointestinal stromal tumors, lipomas, and neurofibromas. Isolated juvenile polyp is the most common polyp in children; ultrasound (US) may demonstrate a hyperemic intraluminal mass in the bowel[88]. The most common malignant GI tumor in children is lymphoma, typically Burkitt lymphoma. Ultrasound findings are often unsuspected in a child imaged for non-specific abdominal symptoms and may show hypoechoic bowel wall thickening, enlarged mesenteric or retroperitoneal lymph nodes, and intussusception (Figure 5).
Vasculitis: Henoch-Schonlein purpura is the most common vasculitis in children. It is an immune-mediated vasculitis affecting multiple organs, and it typically presents with a palpable purpuric rash and abdominal pain. The jejunum and ileum are commonly involved; ultrasound shows small bowel wall thickening that may reflect hemorrhage, inflammation or infarction. Transient small bowel-small bowel intussusception, obstruction, and pneumatosis intestinalis may be present[89] (Figure 6). Bowel wall hyperemia suggests inflammation, while absent color Doppler flow reflects ischemia and potential risk for perforation[90].
Vascular malformation: Vascular malformations of the small bowel are uncommon in children, but when present can cause hematochezia and anemia[91,92]. Ultrasound findings include bowel wall thickening, luminal narrowing, and tubular anechoic structures within the bowel wall that demonstrate color Doppler flow. Hemangiomas may be seen in isolation or in patients with diffuse hemangiomatosis, Klippel-Trenauney syndrome, or Osler-Weber-Rendu disease[87]. Slow-flow vascular malformations typically show venous waveforms and possibly thrombosis[91].
Linear, high-frequency transducers can be used to evaluate for foreign bodies and can help guide selection of subsequent targeted radiographs[93-97]. Metallic and non-metallic foreign bodies are typically echogenic with posterior shadowing[93]. Administration of water by mouth during the examination can help facilitate detection of foreign bodies within the stomach by serving as an acoustic window. Coins are the most common object ingested, and approximately 2/3 are located at the level of the cricopharyngeus muscle, aortic arch, or lower esophageal sphincter, and will need urgent endoscopic removal. The remaining 2/3 are in the stomach at initial radiologic evaluation and most likely will be spontaneously excreted (94%)[93].
Gastrointestinal bezoars usually form in the stomach, and they can pass into the small bowel and potentially cause obstruction. Children with prior gastric surgery are prone to bezoar development due to impaired gastric emptying. Bezoars are intraluminal masses, which show an echogenic arc-like surface and acoustic shadow[98,99]. Bezoars have been shown to demonstrate twinkle artifact on color Doppler imaging, which can increase diagnostic confidence with ultrasound and help differentiate this entity from intraluminal gas or stool[100].
Ultrasound is increasingly utilized for the evaluation of abdominal disorders in children, given its lack of ionizing radiation or need for patient sedation. Ultrasound is the often the initial modality detecting abnormalities of the GI tract in children, either as part of a targeted exam at the site of symptoms or as an incidental finding. Radiologists interpreting US examinations in children should be familiar with the sonographic appearance of both the normal and abnormal GI tract in order to provide the best care for pediatric patients with abdominal diseases.
Manuscript Source: Invited manuscript
Specialty Type: Radiology, Nuclear Medicine and Medical Imaging
Country of Origin: United States
Peer-Review Report Classification
Grade A (Excellent): A
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Ding XW, Gumustas OG, He ST, Krishnan T S- Editor: Qiu S L- Editor: A E- Editor: Wu HL
| 1. | Arys B, Mandelstam S, Rao P, Kernick S, Kumbla S. Sonography of the pediatric gastrointestinal system. Ultrasound Q. 2014;30:101-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 2. | Anupindi SA, Halverson M, Khwaja A, Jeckovic M, Wang X, Bellah RD. Common and uncommon applications of bowel ultrasound with pathologic correlation in children. AJR Am J Roentgenol. 2014;202:946-959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 3. | Lobo ML, Roque M. Gastrointestinal ultrasound in neonates, infants and children. Eur J Radiol. 2014;83:1592-1600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 4. | Darge K, Anupindi S, Keener H, Rompel O. Ultrasound of the bowel in children: how we do it. Pediatr Radiol. 2010;40:528-536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 5. | Maconi G, Radice E, Bareggi E, Porro GB. Hydrosonography of the gastrointestinal tract. AJR Am J Roentgenol. 2009;193:700-708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 6. | Serra C, Menozzi G, Labate AM, Giangregorio F, Gionchetti P, Beltrami M, Robotti D, Fornari F, Cammarota T. Ultrasound assessment of vascularization of the thickened terminal ileum wall in Crohn’s disease patients using a low-mechanical index real-time scanning technique with a second generation ultrasound contrast agent. Eur J Radiol. 2007;62:114-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 92] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 7. | Chiorean L, Schreiber-Dietrich D, Braden B, Cui XW, Buchhorn R, Chang JM, Dietrich CF. Ultrasonographic imaging of inflammatory bowel disease in pediatric patients. World J Gastroenterol. 2015;21:5231-5241. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 29] [Cited by in RCA: 20] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 8. | Dillman JR, Stidham RW, Higgins PD, Moons DS, Johnson LA, Keshavarzi NR, Rubin JM. Ultrasound shear wave elastography helps discriminate low-grade from high-grade bowel wall fibrosis in ex vivo human intestinal specimens. J Ultrasound Med. 2014;33:2115-2123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 79] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 9. | Biko DM, Rosenbaum DG, Anupindi SA. Ultrasound features of pediatric Crohn disease: a guide for case interpretation. Pediatr Radiol. 2015;45:1557-1566; quiz 1554-1566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 10. | Lee NK, Kim S, Jeon TY, Kim HS, Kim DH, Seo HI, Park DY, Jang HJ. Complications of congenital and developmental abnormalities of the gastrointestinal tract in adolescents and adults: evaluation with multimodality imaging. Radiographics. 2010;30:1489-1507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 60] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 11. | Chen QJ, Gao ZG, Tou JF, Qian YZ, Li MJ, Xiong QX, Shu Q. Congenital duodenal obstruction in neonates: a decade’s experience from one center. World J Pediatr. 2014;10:238-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 12. | Chao HC, Kong MS, Chen JY, Lin SJ, Lin JN. Sonographic features related to volvulus in neonatal intestinal malrotation. J Ultrasound Med. 2000;19:371-376. [PubMed] |
| 13. | Yousefzadeh DK, Kang L, Tessicini L. Assessment of retromesenteric position of the third portion of the duodenum: an US feasibility study in 33 newborns. Pediatr Radiol. 2010;40:1476-1484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 35] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 14. | Srinath A, Wendel D, Bond G, Lowe M. Visual diagnosis: 12-year-old girl with constipation and rectal bleeding. Pediatr Rev. 2014;35:e11-e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 15. | Laskowska K, Gałązka P, Daniluk-Matraś I, Leszczyński W, Serafin Z. Use of diagnostic imaging in the evaluation of gastrointestinal tract duplications. Pol J Radiol. 2014;79:243-250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 16. | Sharma D, Bharany RP, Mapshekhar RV. Duplication cyst of pyloric canal: a rare cause of pediatric gastric outlet obstruction: rare case report. Indian J Surg. 2013;75:322-325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 17. | Gebesce A, Korkmaz M, Keles E, Korkmaz F, Mahmutyazıcıoglu K, Yazgan H. Importance of the ultrasonography in diagnosis of ileal duplication cyst. Gastroenterol Res Pract. 2013;2013:248625. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 18. | Sutcliffe J, Munden M. Sonographic diagnosis of multiple gastric duplication cysts causing gastric outlet obstruction in a pediatric patient. J Ultrasound Med. 2006;25:1223-1226. [PubMed] |
| 19. | Cheng G, Soboleski D, Daneman A, Poenaru D, Hurlbut D. Sonographic pitfalls in the diagnosis of enteric duplication cysts. AJR Am J Roentgenol. 2005;184:521-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 86] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 20. | Baldisserotto M. Color Doppler sonographic findings of inflamed and perforated Meckel diverticulum. J Ultrasound Med. 2004;23:843-848. [PubMed] |
| 21. | Ohno Y, Kanematsu T. Annular pancreas causing localized recurrent pancreatitis in a child: report of a case. Surg Today. 2008;38:1052-1055. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 22. | Vijayaraghavan SB. Sonography of pancreatic ductal anatomic characteristics in annular pancreas. J Ultrasound Med. 2002;21:1315-1318. [PubMed] |
| 23. | Pohl-Schickinger A, Henrich W, Degenhardt P, Bassir C, Hüseman D. Echogenic foci in the dilated fetal colon may be associated with the presence of a rectourinary fistula. Ultrasound Obstet Gynecol. 2006;28:341-344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 24. | Anderson S, Savader B, Barnes J, Savader S. Enterolithiasis with imperforate anus. Report of two cases with sonographic demonstration and occurrence in a female. Pediatr Radiol. 1988;18:130-133. [PubMed] |
| 25. | Kim IO, Han TI, Kim WS, Yeon KM. Transperineal ultrasonography in imperforate anus: identification of the internal fistula. J Ultrasound Med. 2000;19:211-216. [PubMed] |
| 26. | Hryhorczuk AL, Strouse PJ. Validation of US as a first-line diagnostic test for assessment of pediatric ileocolic intussusception. Pediatr Radiol. 2009;39:1075-1079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 108] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 27. | Peh WC, Khong PL, Lam C, Chan KL, Saing H, Cheng W, Mya GH, Lam WW, Leong LL, Low LC. Ileoileocolic intussusception in children: diagnosis and significance. Br J Radiol. 1997;70:891-896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 28. | Lioubashevsky N, Hiller N, Rozovsky K, Segev L, Simanovsky N. Ileocolic versus small-bowel intussusception in children: can US enable reliable differentiation? Radiology. 2013;269:266-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 49] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 29. | Park NH, Park SI, Park CS, Lee EJ, Kim MS, Ryu JA, Bae JM. Ultrasonographic findings of small bowel intussusception, focusing on differentiation from ileocolic intussusception. Br J Radiol. 2007;80:798-802. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 30. | Munden MM, Bruzzi JF, Coley BD, Munden RF. Sonography of pediatric small-bowel intussusception: differentiating surgical from nonsurgical cases. AJR Am J Roentgenol. 2007;188:275-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 45] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 31. | Strouse PJ, DiPietro MA, Saez F. Transient small-bowel intussusception in children on CT. Pediatr Radiol. 2003;33:316-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 60] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 32. | Fonio P, Coppolino F, Russo A, D’Andrea A, Giannattasio A, Reginelli A, Grassi R, Genovese EA. Ultrasonography (US) in the assessment of pediatric non traumatic gastrointestinal emergencies. Crit Ultrasound J. 2013;5 Suppl 1:S12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 33. | Markowitz RI. Olive without a cause: the story of infantile hypertrophic pyloric stenosis. Pediatr Radiol. 2014;44:202-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 34. | Teele RL, Smith EH. Ultrasound in the diagnosis of idiopathic hypertrophic pyloric stenosis. N Engl J Med. 1977;296:1149-1150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 95] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 35. | Rohrschneider WK, Mittnacht H, Darge K, Tröger J. Pyloric muscle in asymptomatic infants: sonographic evaluation and discrimination from idiopathic hypertrophic pyloric stenosis. Pediatr Radiol. 1998;28:429-434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 32] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 36. | Said M, Shaul DB, Fujimoto M, Radner G, Sydorak RM, Applebaum H. Ultrasound measurements in hypertrophic pyloric stenosis: don’t let the numbers fool you. Perm J. 2012;16:25-27. [PubMed] |
| 37. | Leaphart CL, Borland K, Kane TD, Hackam DJ. Hypertrophic pyloric stenosis in newborns younger than 21 days: remodeling the path of surgical intervention. J Pediatr Surg. 2008;43:998-1001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 38. | Chen KC, Chu CC, Chou TY, Wu CJ. Ultrasonography for inguinal hernias in boys. J Pediatr Surg. 1998;33:1784-1787. [PubMed] |
| 39. | Chou TY, Chu CC, Diau GY, Wu CJ, Gueng MK. Inguinal hernia in children: US versus exploratory surgery and intraoperative contralateral laparoscopy. Radiology. 1996;201:385-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 45] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 40. | Erez I, Rathause V, Vacian I, Zohar E, Hoppenstein D, Werner M, Lazar L, Freud E. Preoperative ultrasound and intraoperative findings of inguinal hernias in children: A prospective study of 642 children. J Pediatr Surg. 2002;37:865-868. [PubMed] |
| 41. | Celik A, Ergün O, Ozbek SS, Dökümcü Z, Balik E. Sliding appendiceal inguinal hernia: preoperative sonographic diagnosis. J Clin Ultrasound. 2003;31:156-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 42. | Hata S, Takahashi Y, Nakamura T, Suzuki R, Kitada M, Shimano T. Preoperative sonographic evaluation is a useful method of detecting contralateral patent processus vaginalis in pediatric patients with unilateral inguinal hernia. J Pediatr Surg. 2004;39:1396-1399. [PubMed] |
| 43. | Graf JL, Caty MG, Martin DJ, Glick PL. Pediatric hernias. Semin Ultrasound CT MR. 2002;23:197-200. [PubMed] |
| 44. | Orth RC, Towbin AJ. Acute testicular ischemia caused by incarcerated inguinal hernia. Pediatr Radiol. 2012;42:196-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 45. | Cakmakci E, Celebi I, Tahtabasi M, Tabakci ON, Ozkurt H, Basak M, Karpat Z. Accuracy of ultrasonography in the diagnosis of sliding hiatal hernias. Acad Radiol. 2013;20:453-456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 46. | Cakmakci E, Tahtabasi M, Celebi I, Cakmakci S, Bayram A, Tokgoz S, Dogru M, Basak M. Diagnostic significance of periesophageal fat pad in ultrasonography for sliding hiatal hernias: sonographic fat pad sign. Clin Imaging. 2014;38:170-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 47. | Savino A, Cecamore C, Matronola MF, Verrotti A, Mohn A, Chiarelli F, Pelliccia P. US in the diagnosis of gastroesophageal reflux in children. Pediatr Radiol. 2012;42:515-524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 48. | Westra SJ, Wolf BH, Staalman CR. Ultrasound diagnosis of gastroesophageal reflux and hiatal hernia in infants and young children. J Clin Ultrasound. 1990;18:477-485. [PubMed] |
| 49. | Halkiewicz F, Kasner J, Karczewska K, Rusek-Zychma M. Ultrasound picture of gastroesophageal junction in children with reflux disease. Med Sci Monit. 2000;6:96-99. [PubMed] |
| 50. | Dumitriu D, Menten R, Smets F, Clapuyt P. Postendoscopic duodenal hematoma in children: ultrasound diagnosis and follow-up. J Clin Ultrasound. 2014;42:550-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 51. | Diniz-Santos DR, de Andrade Cairo RC, Braga H, Araújo-Neto C, Paes IB, Silva LR. Duodenal hematoma following endoscopic duodenal biopsy: a case report and review of the existing literature. Can J Gastroenterol. 2006;20:39-42. [PubMed] |
| 52. | Guzman C, Bousvaros A, Buonomo C, Nurko S. Intraduodenal hematoma complicating intestinal biopsy: case reports and review of the literature. Am J Gastroenterol. 1998;93:2547-2550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 17] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 53. | Bachur RG, Levy JA, Callahan MJ, Rangel SJ, Monuteaux MC. Effect of Reduction in the Use of Computed Tomography on Clinical Outcomes of Appendicitis. JAMA Pediatr. 2015;169:755-760. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 55] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 54. | Biçer Ş, Çelik A. Duodenal Obstruction Caused by Acute Appendicitis with Intestinal Malrotation in a Child. Am J Case Rep. 2015;16:574-576. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 55. | Koike Y, Uchida K, Matsushita K, Otake K, Nakazawa M, Inoue M, Kusunoki M, Tsukamoto Y. Intraluminal appendiceal fluid is a predictive factor for recurrent appendicitis after initial successful non-operative management of uncomplicated appendicitis in pediatric patients. J Pediatr Surg. 2014;49:1116-1121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 48] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 56. | Salminen P, Paajanen H, Rautio T, Nordström P, Aarnio M, Rantanen T, Tuominen R, Hurme S, Virtanen J, Mecklin JP. Antibiotic Therapy vs Appendectomy for Treatment of Uncomplicated Acute Appendicitis: The APPAC Randomized Clinical Trial. JAMA. 2015;313:2340-2348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 502] [Cited by in RCA: 527] [Article Influence: 52.7] [Reference Citation Analysis (0)] |
| 57. | Binkovitz LA, Unsdorfer KM, Thapa P, Kolbe AB, Hull NC, Zingula SN, Thomas KB, Homme JL. Pediatric appendiceal ultrasound: accuracy, determinacy and clinical outcomes. Pediatr Radiol. 2015;45:1934-1944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 62] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 58. | Fallon SC, Orth RC, Guillerman RP, Munden MM, Zhang W, Elder SC, Cruz AT, Brandt ML, Lopez ME, Bisset GS. Development and validation of an ultrasound scoring system for children with suspected acute appendicitis. Pediatr Radiol. 2015;45:1945-1952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 46] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 59. | Ramarajan N, Krishnamoorthi R, Gharahbaghian L, Pirrotta E, Barth RA, Wang NE. Clinical correlation needed: what do emergency physicians do after an equivocal ultrasound for pediatric acute appendicitis? J Clin Ultrasound. 2014;42:385-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 60. | Trout AT, Towbin AJ, Fierke SR, Zhang B, Larson DB. Appendiceal diameter as a predictor of appendicitis in children: improved diagnosis with three diagnostic categories derived from a logistic predictive model. Eur Radiol. 2015;25:2231-2238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 44] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 61. | Cohen B, Bowling J, Midulla P, Shlasko E, Lester N, Rosenberg H, Lipskar A. The non-diagnostic ultrasound in appendicitis: is a non-visualized appendix the same as a negative study? J Pediatr Surg. 2015;50:923-927. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 47] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 62. | Garbi-Goutel A, Brévaut-Malaty V, Panuel M, Michel F, Merrot T, Gire C. Prognostic value of abdominal sonography in necrotizing enterocolitis of premature infants born before 33 weeks gestational age. J Pediatr Surg. 2014;49:508-513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 63. | Yikilmaz A, Hall NJ, Daneman A, Gerstle JT, Navarro OM, Moineddin R, Pleasants H, Pierro A. Prospective evaluation of the impact of sonography on the management and surgical intervention of neonates with necrotizing enterocolitis. Pediatr Surg Int. 2014;30:1231-1240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 64. | Silva CT, Daneman A, Navarro OM, Moore AM, Moineddin R, Gerstle JT, Mittal A, Brindle M, Epelman M. Correlation of sonographic findings and outcome in necrotizing enterocolitis. Pediatr Radiol. 2007;37:274-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 101] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 65. | Epelman M, Daneman A, Navarro OM, Morag I, Moore AM, Kim JH, Faingold R, Taylor G, Gerstle JT. Necrotizing enterocolitis: review of state-of-the-art imaging findings with pathologic correlation. Radiographics. 2007;27:285-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 190] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 66. | Muchantef K, Epelman M, Darge K, Kirpalani H, Laje P, Anupindi SA. Sonographic and radiographic imaging features of the neonate with necrotizing enterocolitis: correlating findings with outcomes. Pediatr Radiol. 2013;43:1444-1452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 68] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 67. | Tarantino L. Abdominal Ultrasound in Infectious Enteritis. Eur Med Imaging Review. 2008;1:57-61. |
| 68. | KılıçÖ, Somer A, Hançerli Törün S, Keser Emiroğlu M, Salman N, Salman T, Çelik A, Yekeler E, Uzun M. Assessment of 35 children with abdominal tuberculosis. Turk J Gastroenterol. 2015;26:128-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 69. | Almadi MA, Ghosh S, Aljebreen AM. Differentiating intestinal tuberculosis from Crohn’s disease: a diagnostic challenge. Am J Gastroenterol. 2009;104:1003-1012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 138] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 70. | McCarville MB. Evaluation of typhlitis in children: CT versus US. Pediatr Radiol. 2006;36:890-891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 71. | Mullassery D, Bader A, Battersby AJ, Mohammad Z, Jones EL, Parmar C, Scott R, Pizer BL, Baillie CT. Diagnosis, incidence, and outcomes of suspected typhlitis in oncology patients--experience in a tertiary pediatric surgical center in the United Kingdom. J Pediatr Surg. 2009;44:381-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 44] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 72. | Mahmood T, Mansoor N, Quraishy S, Ilyas M, Hussain S. Ultrasonographic appearance of Ascaris lumbricoides in the small bowel. J Ultrasound Med. 2001;20:269-274. [PubMed] |
| 73. | Epifanio M, Spolidoro JV, Missima NG, Soder RB, Garcia PC, Baldisserotto M. Cow’s milk allergy: color Doppler ultrasound findings in infants with hematochezia. J Pediatr (Rio J). 2013;89:554-558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 74. | Magazzù G, Scoglio R. Gastrointestinal manifestations of cow’s milk allergy. Ann Allergy Asthma Immunol. 2002;89:65-68. [PubMed] |
| 75. | Hümmer-Ehret BH, Rohrschneider WK, Oleszczuk-Raschke K, Darge K, Nützenadel W, Tröger J. Eosinophilic gastroenteritis mimicking idiopathic hypertrophic pyloric stenosis. Pediatr Radiol. 1998;28:711-713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 76. | Blankenberg FG, Parker BR, Sibley E, Kerner JA. Evolving asymmetric hypertrophic pyloric stenosis associated with histologic evidence of eosinophilic gastroenteritis. Pediatr Radiol. 1995;25:310-311. [PubMed] |
| 77. | Dillman JR, Smith EA, Sanchez RJ, DiPietro MA, DeMatos-Maillard V, Strouse PJ, Darge K. Pediatric Small Bowel Crohn Disease: Correlation of US and MR Enterography. Radiographics. 2015;35:835-848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 78. | Casciani E, De Vincentiis C, Polettini E, Masselli G, Di Nardo G, Civitelli F, Cucchiara S, Gualdi GF. Imaging of the small bowel: Crohn’s disease in paediatric patients. World J Radiol. 2014;6:313-328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 14] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 79. | Pallotta N, Civitelli F, Di Nardo G, Vincoli G, Aloi M, Viola F, Capocaccia P, Corazziari E, Cucchiara S. Small intestine contrast ultrasonography in pediatric Crohn’s disease. J Pediatr. 2013;163:778-784.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 50] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 80. | Quaia E. Contrast-enhanced ultrasound of the small bowel in Crohn’s disease. Abdom Imaging. 2013;38:1005-1013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 81. | Civitelli F, Di Nardo G, Oliva S, Nuti F, Ferrari F, Dilillo A, Viola F, Pallotta N, Cucchiara S, Aloi M. Ultrasonography of the colon in pediatric ulcerative colitis: a prospective, blind, comparative study with colonoscopy. J Pediatr. 2014;165:78-84.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 82. | Soresi M, Pirrone G, Giannitrapani L, Iacono G, Di Prima L, La Spada E, Di Fede G, Ambrosiano G, Montalto G, Carroccio A. A key role for abdominal ultrasound examination in “difficult” diagnoses of celiac disease. Ultraschall Med. 2011;32 Suppl 1:S53-S61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 83. | Mirk P, Foschi R, Minordi LM, Vecchioli Scaldazza A, De Vitis I, Guidi L, Bonomo L. Sonography of the small bowel after oral administration of fluid: an assessment of the diagnostic value of the technique. Radiol Med. 2012;117:558-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 84. | Gheibi S. Association between Celiac Disease and Intussusceptions in Children: Two Case Reports and Literature Review. Pediatr Gastroenterol Hepatol Nutr. 2013;16:269-272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 85. | Goyal M, Ellison AM, Schapiro E. An unusual case of intussusception. Pediatr Emerg Care. 2010;26:212-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 86. | Kralik R, Trnovsky P, Kopáčová M. Transabdominal ultrasonography of the small bowel. Gastroenterol Res Pract. 2013;2013:896704. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 87. | Ladino-Torres MF, Strouse PJ. Gastrointestinal tumors in children. Radiol Clin North Am. 2011;49:665-677, v-vi. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 88. | Caiulo S, Moramarco F, Caiulo VA. Juvenile polyp. Eur J Pediatr. 2014;173:125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 89. | Hryhorczuk AL, Lee EY. Imaging evaluation of bowel obstruction in children: updates in imaging techniques and review of imaging findings. Semin Roentgenol. 2012;47:159-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 90. | Teefey SA, Roarke MC, Brink JA, Middleton WD, Balfe DM, Thyssen EP, Hildebolt CF. Bowel wall thickening: differentiation of inflammation from ischemia with color Doppler and duplex US. Radiology. 1996;198:547-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 64] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 91. | Kalmar PI, Petnehazy T, Wießpeiner U, Beer M, Hauer AC, Till H, Riccabona M. Large, segmental, circular vascular malformation of the small intestine (in a female toddler with hematochezia): unusual presentation in a child. BMC Pediatr. 2014;14:55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 92. | Handra-Luca A, Montgomery E. Vascular malformations and hemangiolymphangiomas of the gastrointestinal tract: morphological features and clinical impact. Int J Clin Exp Pathol. 2011;4:430-443. [PubMed] |
| 93. | Salmon M, Doniger SJ. Ingested foreign bodies: a case series demonstrating a novel application of point-of-care ultrasonography in children. Pediatr Emerg Care. 2013;29:870-873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 94. | Piotto L, Gent R, Kirby CP, Morris LL. Preoperative use of ultrasonography to localize an ingested foreign body. Pediatr Radiol. 2009;39:299-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 95. | Jecković M, Anupindi SA, Barbir SB, Lovrenski J. Is ultrasound useful in detection and follow-up of gastric foreign bodies in children? Clin Imaging. 2013;37:1043-1047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 96. | Isaacs DL. Detection of a ballpoint pen in a patient’s abdomen by sonography. J Ultrasound Med. 2006;25:1095-1098. [PubMed] |
| 97. | Chiang TH, Liu KL, Lee YC, Chiu HM, Lin JT, Wang HP. Sonographic diagnosis of a toothpick traversing the duodenum and penetrating into the liver. J Clin Ultrasound. 2006;34:237-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 28] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 98. | Ripollés T, García-Aguayo J, Martínez MJ, Gil P. Gastrointestinal bezoars: sonographic and CT characteristics. AJR Am J Roentgenol. 2001;177:65-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 163] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 99. | Lee KH, Han HY, Kim HJ, Kim HK, Lee MS. Ultrasonographic differentiation of bezoar from feces in small bowel obstruction. Ultrasonography. 2015;34:211-216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 100. | Kim HC, Yang DM, Kim SW, Park SJ, Ryu JK. Color Doppler twinkling artifacts in small-bowel bezoars. J Ultrasound Med. 2012;31:793-797. [PubMed] |