Peer-review started: October 29, 2014
First decision: December 12, 2014
Revised: December 25, 2014
Accepted: January 15, 2015
Article in press: January 19, 2015
Published online: February 28, 2015
Processing time: 106 Days and 22.3 Hours
AIM: To evaluate the changes of intracranial blood flow after carotid artery stenting (CAS), using the flow assessment application “Flow-Insight”, which was developed in our department.
METHODS: Twenty patients treated by CAS participated in this study. We analyzed the change in concentration of the contrast media at the anterior-posterior and profile view image with the flow assessment application “Flow-Insight”. And we compared the results with N-isopropyl-p-[123I] iodoamphetamine-single-photon emission computed tomography (IMP SPECT) performed before and after the treatment.
RESULTS: From this study, 200% of the parameter “blood flow” change in the post/pre-treatment is suggested as the critical line of the hyperperfusion syndrome arise. Although the observed blood flow increase in the digital subtraction angiography system did not strongly correlate with the rate of increase of SPECT, the “Flow-Insight” reflected the rate of change of the vessels well. However, for patients with reduced reserve blood flow before CAS, a highly elevated site was in agreement with the site analysis results.
CONCLUSION: We concluded that the cerebral angiography flow assessment application was able to more finely reveal hyperperfusion regions in the brain after CAS compared to SPECT.
Core tip: Hyperperfusion syndrome is a relatively rare, but potentially serious, complication of carotid revascularization procedures. It is important to detect the excessive increase blood flow after treatment as soon as possible. We found that, although the observed blood flow increase in the digital subtraction angiography system did not strongly correlate with the rate of increase of single-photon emission computed tomography before and after carotid artery stenting, the digital subtraction angiography flow assessment application more finely reflected the rate of change of the vessels well.
- Citation: Wada H, Saito M, Kamada K. Evaluation of changes of intracranial blood flow after carotid artery stenting using digital subtraction angiography flow assessment. World J Radiol 2015; 7(2): 45-51
- URL: https://www.wjgnet.com/1949-8470/full/v7/i2/45.htm
- DOI: https://dx.doi.org/10.4329/wjr.v7.i2.45
Cerebral hyperperfusion after carotid revascularization treatment is defined as a major increase in ipsilateral cerebral blood flow (CBF) well above the metabolic demands of the brain tissue. Cerebral hyperperfusion syndrome is characterized by a unilateral headache, face and eye pain, seizures, and focal symptoms secondary to cerebral edema or intracerebral hemorrhage. The pathological mechanisms of the irreversible changes have been theorized to involve organic dysautoregulation of maximally dilated cerebral vessels. Although the incidence of intracerebral hemorrhage is relatively low, the prognosis for patients with this condition is poor[1,2]. Schroeder et al[3] reported that CBF on N-isopropyl-p-[123I] iodoamphetamine-single-photon emission computed tomography ([123] IMP-SPECT) postoperatively is increased beyond that in the preoperative state[3]; and, in cases of marked hypoperfusion, hyperperfusion after surgical revascularization is known to occur[4].
In this study, we aimed to evaluate the change of intracranial blood flow after carotid artery stenting (CAS) using the flow assessment application “Flow-Insight,” which was developed in our department; and to compare the results with those of IMP-SPECT, which is currently commonly used in clinical practice[5].
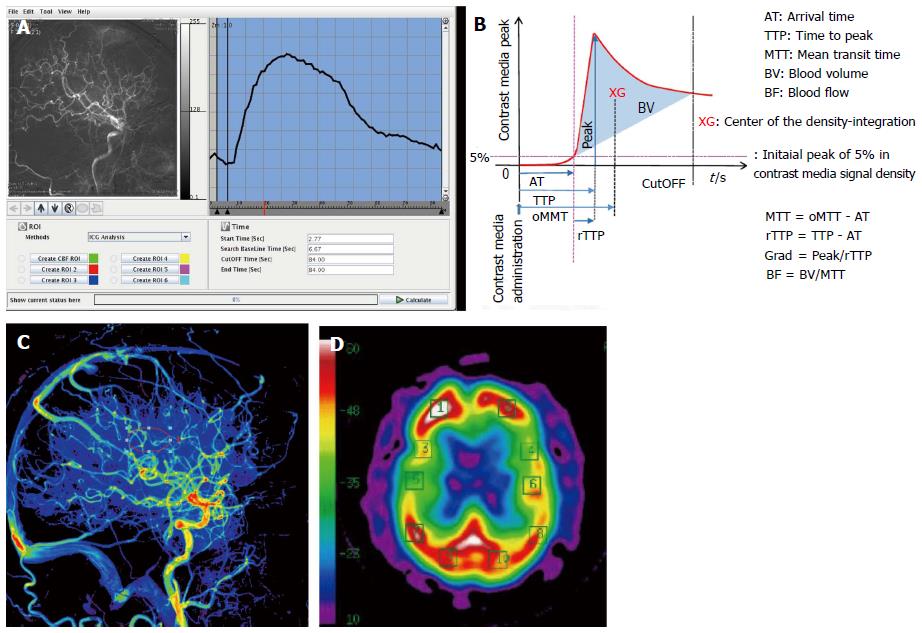
A total of 20 patients (18 men and 2 women), aged 56-82 years (mean, 70.0 ± 5.9 years), without large cortical infarction on conventional magnetic resonance imaging, who underwent CAS between October 2012 and April 2014 were enrolled in the present study. The mean degree of internal carotid artery stenosis was 75.3% ± 15.8% (range, 59%-93%), according to the method of the North American Symptomatic Carotid Endarterectomy Trial[6]. Three patients had symptomatic stenosis and 4 patients had occlusion or stenosis greater than 50% in the contralateral internal carotid artery. All cases of CAS were performed under general anesthesia, using a proximal balloon, distal filter protection, and flow reverse to the femoral vein. Using the location memory of the digital subtraction angiography (DSA) suit Artis zee (Siemens AG Healthcare, Erlangen, Germany), intracranial angiography acquisitions were evaluated in the same position before and after treatment. We analyzed the changes in the concentration of the contrast media as changes in brightness using the novel flow assessment application “Flow-Insight” (Infocom, Tokyo, Japan). The result images were created as an anterior-posterior (AP) view and a profile image. The software converts DSA DICOM image data to 8 bit digital data. It utilizes a mutual information method and phase correction of the two-dimensional Fourier transformation as a motion artifact correction method, and can calculate the integral value of the luminance change of each pixel in the converted data and the peak periods to examine. The calculated parameters include the calculation amount arrival time, time to peak, and mean transit time, with a focus on the time and blood volume, as determined by the accumulated amount of brightness and blood flow. In the result images, the rate of the blood flow is displayed by a color bar representing changes of 50%-200%. In this study, we used paired t test for parameter changes before and after CAS and statistical significance was less than 0.05. We determined the region of interest (ROI) in the result images, and compared the results by the ARG method with one 123I-IMP SPECT before and after CAS, using Spearman’s rank correlation coefficient and simple regression analyses.
The study protocol was performed in accordance with the Declaration of Helsinki and its later amendments. All participants provided written informed consent.
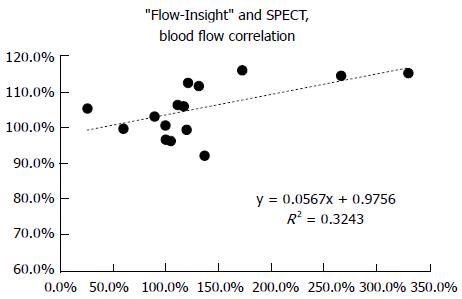
Our results show that the novel “Flow-Insight” application was capable of determining a qualitative value of the blood flow, which was defined as the blood volume divided by the mean transit time (Figure 1B and C) using a central volume principle as previously described[7]. The blood volume and mean transit time were determined using a time density curve (Figure 1A). We next created an ROI and calculated the cerebral blood flow before and after treatment, and compared the increased rate with the corresponding rate evaluated by IMP SPECT. The increased rate was calculated by taking the ratio of the cerebellum and the left and right blood flow communication. We found that the “Flow-Insight” blood flow increase rate of the middle cerebral artery territory correlated with the increase ratio of the same SPECT territory in the cerebellum (Figure 2). The distribution with “Flow-Insight” was close to the 50%-200% range in all patients.
Because of the nature of DSA, it is generally difficult to capture the brain tissue blood perfusion changes. However, the images obtained using the “Flow-Insight” application were found to reflect the changes in the vessels well. To avoid the vessels, we set the ROI as the motor and premotor cortex in the frontal lobe, just above the Sylvian fissure (Figure 1C and D). “Flow-Insight” successfully showed the highly elevated blood flow increase rate areas of patients with reduced vascular reserve areas.
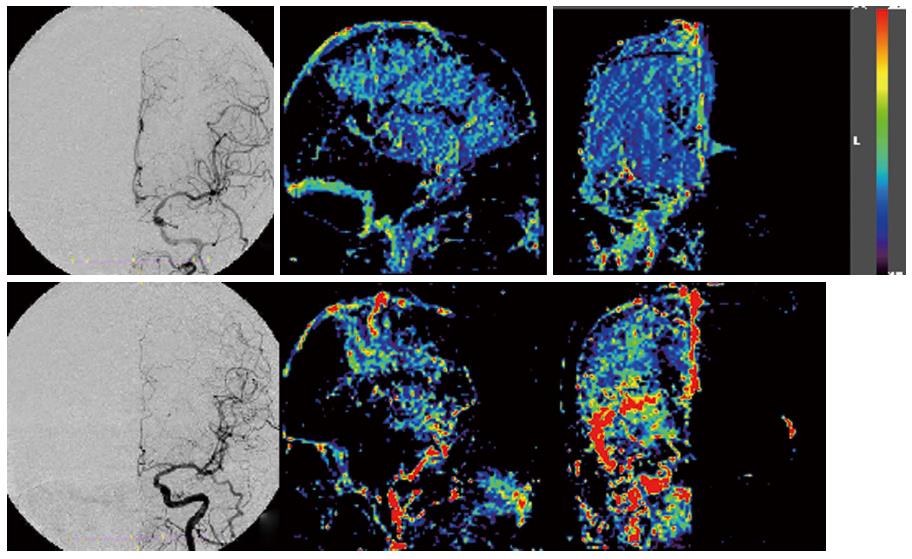
In order to detect the actual increasing area in the human brain, we next created images based on the DSA blood flow increase ratio in the AP and lateral views, by dividing the post-operative CAS images by the pre-operative images (Figures 2 and 3). The result images showed the rate of blood flow, as defined by the color bar at 50%-200%. This range was chosen based on the range from the correlation of blood flow with SPECT. We reduced the image matrix settings to 100 × 100, because the displacement of high-resolution image calculations may lead to overestimation of the results.
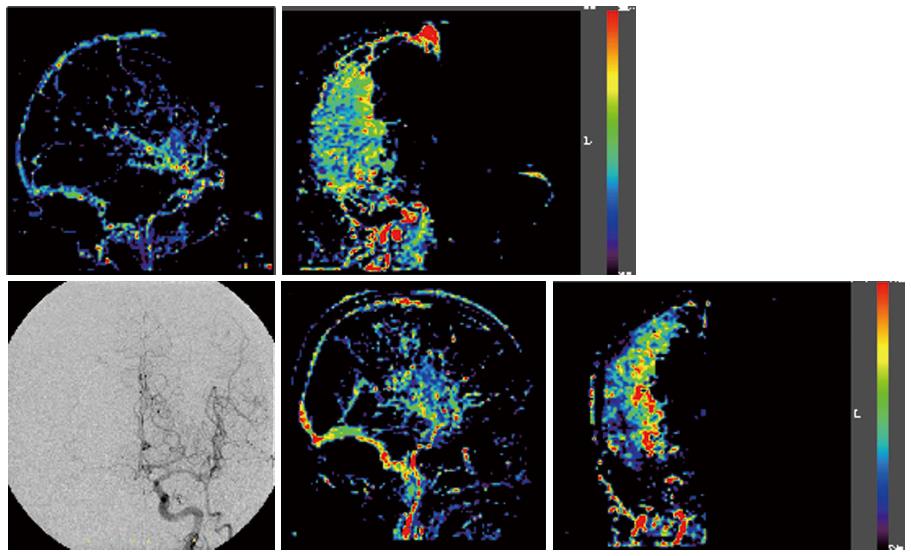
It should be noted that the increased distribution observed after CAS was variable. For example, while Cases 1 and 2 did not show any blood flow from the contra-lateral via the anterior communicating artery, differences in the increased area and in the degree of the increase were noted upon DSA (Figure 3). Conversely, Cases 3 and 4 suffered severe right cervical internal carotid artery stenosis, and our analyses revealed blood flow from the contra-lateral side via the anterior communicating artery at the preoperative DSA (Figure 4). However, while the SPECT for case 4 showed a 114.2% increase ratio, the “Flow-Insight” showed a 265% increase ratio. The patient suffered mild delirium symptoms for 2 d after CAS. On the other hand, Case 3 also showed blood flow via the anterior communicating artery. However, in this case, the “Flow-Insight” blood flow increase ratio was only 131%, while the SPECT also showed a relatively low ratio, at 111.3%, suggesting that the patient did not suffer hyper-perfusion syndrome per se. The fact that the distribution of the intracranial blood flow increase rate after CAS showed quite different results in each case, not only in terms of the presence or absence of the left and right or anterior-posterior communication, but also in terms of the diameter and length, may be due to differences in the degree and area of the increased blood flow. The “Flow-Insight” application successfully revealed the presence of increased blood flow regions, which were not evident upon conventional SPECT.
In the case of carotid endarterectomy, the presence or absence of collateral flow via the anterior or posterior communicating artery deeply affects the risk of intraoperative ischemic complications[8,9]. Moreover, intraoperative measurement of distal internal carotid artery pressure reflects the intracranial vascular reserve, and is important in predicting postoperative hyperperfusion[4]. Reduced blood flow due to internal carotid artery stenosis results in decreased blood flow reserves intracranially, and this has been demonstrated to be significantly influenced by the microscopic vascular bed and the left and right or anterior and posterior communication[10]. Local increases of blood flow after treatment and blood flow buffering through the left-right or anterior-posterior communication may lead to hyperperfusion syndrome. Therefore, hyperperfusion syndrome cannot completely be predicted by the presence or absence of blood flow communication only. Furthermore, local variations at the site of the blood flow increase may cause different symptoms of hyperperfusion syndrome, such as headaches, intracranial hemorrhage (subarachnoid hemorrhage, subcortical hemorrhage, or basal ganglia hemorrhage), delirium, or convulsions[1,2,11].
IMP-SPECT is currently the conventional modality for measuring intracranial blood flow. In this study, when comparing the SPECT and the “Flow-Insight” blood flow increase rates in the whole treated area, no clear correlation was observed. However, when evaluating the correlation restricted to the MCA territory, which is devoid of major vessels and which is the perfusion area of the internal carotid artery, we detected a good correlation. We moreover found that increased blood flow after treatment of internal carotid artery stenosis is not confined to a uniform territory. However, we were unable to draw any firm and conclusions regarding this observation because of the limited size of the study, since only one patient presented with typical hyperperfusion syndrome. This novel evaluation method revealed the increase ratio and its intracranial distribution, and because it strongly reflected the local vascular reserve, as well as the left and right or anterior-posterior artery communication, we believe that is has the potential to predict the presence of hyperperfusion syndrome.
At this time, quantification of cerebral blood flow is not available from the “Flow-Insight”. However, we hypothesized that if there was a high correlation with the results of the post-operative blood flow achieved from the “Flow-Insight” and that obtained from SPECT, the “Flow-Insight” would be able to predict post-operative hyperperfusion syndrome. Accordingly, we found that the “Flow-Insight” blood flow increase rate correlated with the increase ratio of SPECT. However, the correlation, R2, was 0.324, which suggests that the correlation was only moderate. One reason for why no strong correlation was observed could be that the “Flow-Insight” is performed immediately after treatment, whereas SPECT is usually performed approximately 24 h post-operation. After CAS, normalization of cerebral blood flow may spontaneously be initiated. Thus, the results of the flow assessment using DSA will be different from the blood flow measured by SPECT in the following points: first, contrast medium is a non-diffusible tracer. The contrast medium is injected just proximal of the lesion in “Flow-Insight,” indicating that this evaluation method is highly focal. Moreover, it is based on processing of an image acquired by the inhibition of the radiation translucency and subtraction of an X-ray image. Accordingly, “Flow-Insight” reveals slightly different blood flow from that evaluated by conventional SPECT or computed tomography perfusion. In terms of the terminology of this novel method, it should be pointed out that “Flow-Insight” is the flow assessment software used, while cerebral angiography is simply a method used to obtain the black-and-white 2D image, and is used only to evaluate the appearance of the vascular system, such as the form of aneurysms or continuity of vessels. However, a major advantage of DSA is that it has a time scale, which computed tomography angiography and magnetic resonance angiography do not.
It has been previously reported that the right and left venous phase appearance times are symmetrical in the internal carotid artery occlusion test (balloon occlusion test)[12], and this method has been proposed as an alternative to angiographic imaging. However, it was due to the statistical processing taking a partial ROI.
By using the “Flow-Insight” software, it is easy to visualize the intracranial blood flow changes using the color bar, despite the 2D nature of the images. It is hence a useful indicator to understand the entire brain, unlike focal methods evaluating only the ROI. Moreover, the fact that it can be used immediately after treatment unlike other post-operative evaluations such as SPECT or computed tomography perfusion is a great advantage.
Application of flow assessment angiography with indocyanine green in bypass surgery has been previously reported[13-15], and cerebral blood flow assessment using the latest DSA system may also be applied for these patients[16]. The “Flow-Insight” application is based on the same principle as flow assessment angiography and is easy to use with the proper equipment. However, the method of analysis could be improved further, and the application is currently inconvenient to apply in other ways. DSA flow assessment is also associated with a number of issues related to certain factors, such as the head position during data acquisition, the velocity and amount of contrast media used, and the diameter of the catheter. Moreover, it is a direct injection of contrast media, which may cause increased pressure in the arteries, rather than a systemic intravenous administration into the physiological blood flow. Furthermore, since it is based on two-dimensional images, the accumulated value of the 2D images are greatly affected by the three-dimensional volume, such as for example the intracranial venous phase overlapping with the arterial phase, and the image accumulated by subtraction images can be easily affected by motion artifacts. However, the risk of this can be minimized by using general anesthesia to keep the patients stationary, and by the angle and position of the table being memorized by a specialized device during the treatment. In the future, if it becomes possible to perform 3D assessment analyses, it can be expected that the accuracy of this application will dramatically improve.
In the present study, using flow assessment application we were able to reveal more finely hyperperfusion regions in the brain after CAS. Flow assessment application was useful for understanding the pathology of the brain post-CAS. Further studies to determine the optimal angiographic conditions, such as the optimal contrast agent concentration and amount, are warranted.
Flow assessment study derived from digital subtraction angiography (DSA) for the cerebral arterial flow in clinical settings have been proposed, but no exist so much. Hyperperfusion syndrome is a relatively rare, but potentially serious, complication of carotid artery stenting. Therefore, the authors applied to detect the excessive increase blood flow after treatment.
In the present study, using flow assessment application of cerebral angiography the authors were able to reveal more finely hyperperfusion regions in the brain after carotid artery stenting. Then it suggests that any hyperperfusion syndrome variation is come from which brain area of excessive increased flow.
Just after treatment, only usual cerebral angiography, without using other modality, can predict the occurrence of serious complications. Furthermore, it reveals that flow assessment application was aim to be a useful clinical application.
From this study, 200% of the parameter “blood flow” change in the post/pre-treatment is suggested as the critical line of the hyperperfusion syndrome arise. By using the “Flow-Insight” software, it is easy to visualize the intracranial blood flow changes using the color bar, despite the 2D nature of the images. It is hence a useful indicator to understand the entire brain, unlike focal methods evaluating only the region of interest.
Flow assessment application: It analyze focal blood flow and derive parametric imaging maps from DSA. The origin is from Indocyanine green video angiography at the open micro-surgery. Cerebral hyperperfusion syndrome: It is a rare, serious complication either after carotid endarterectomy or carotid stent placement. Impaired cerebral autoregulation. The syndrome is characterized by a unilateral headache, face and eye pain, seizures, and focal symptoms secondary to cerebral edema or intracerebral hemorrhage. It may be fatal once an intracranial hemorrhage occurs.
The paper is well written.
P- Reviewer: Rodriguez GJ, Spalice A, Shen J S- Editor: Ji FF L- Editor: A E- Editor: Wu HL
| 1. | Bernstein M, Fleming JF, Deck JH. Cerebral hyperperfusion after carotid endarterectomy: a cause of cerebral hemorrhage. Neurosurgery. 1984;15:50-56. [PubMed] |
| 2. | Piepgras DG, Morgan MK, Sundt TM, Yanagihara T, Mussman LM. Intracerebral hemorrhage after carotid endarterectomy. J Neurosurg. 1988;68:532-536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 281] [Cited by in RCA: 233] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 3. | Schroeder T, Sillesen H, Sørensen O, Engell HC. Cerebral hyperperfusion following carotid endarterectomy. J Neurosurg. 1987;66:824-829. [PubMed] |
| 4. | Yoshimoto T, Shirasaka T, Yoshizumi T, Fujimoto S, Kaneko S, Kashiwaba T. Evaluation of carotid distal pressure for prevention of hyperperfusion after carotid endarterectomy. Surg Neurol. 2005;63:554-557; discussion 557-558. [RCA] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 5. | Iida H, Itoh H, Nakazawa M, Hatazawa J, Nishimura H, Onishi Y, Uemura K. Quantitative mapping of regional cerebral blood flow using iodine-123-IMP and SPECT. J Nucl Med. 1994;35:2019-2030. [PubMed] |
| 6. | North American Symptomatic Carotid Endarterectomy Trial Collaborators. Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. N Engl J Med. 1991;325:445-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6331] [Cited by in RCA: 5730] [Article Influence: 168.5] [Reference Citation Analysis (0)] |
| 7. | Meier P, Zierler KL. On the theory of the indicator-dilution method for measurement of blood flow and volume. J Appl Physiol. 1954;6:731-744. [PubMed] |
| 8. | Lopez-Bresnahan MV, Kearse LA, Yanez P, Young TI. Anterior communicating artery collateral flow protection against ischemic change during carotid endarterectomy. J Neurosurg. 1993;79:379-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 9. | Schwartz RB, Jones KM, LeClercq GT, Ahn SS, Chabot R, Whittemore A, Mannick JA, Donaldson MC, Gugino LD. The value of cerebral angiography in predicting cerebral ischemia during carotid endarterectomy. AJR Am J Roentgenol. 1992;159:1057-1061. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 31] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 10. | Kuroda H, Ogasawara K, Hirooka R, Kobayashi M, Fujiwara S, Chida K, Ishigaki D, Otawara Y, Ogawa A. Prediction of cerebral hyperperfusion after carotid endarterectomy using middle cerebral artery signal intensity in preoperative single-slab 3-dimensional time-of-flight magnetic resonance angiography. Neurosurgery. 2009;64:1065-1071; discussion 1071-1072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 11. | Solomon RA, Loftus CM, Quest DO, Correll JW. Incidence and etiology of intracerebral hemorrhage following carotid endarterectomy. J Neurosurg. 1986;64:29-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 144] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 12. | Abud DG, Spelle L, Piotin M, Mounayer C, Vanzin JR, Moret J. Venous phase timing during balloon test occlusion as a criterion for permanent internal carotid artery sacrifice. AJNR Am J Neuroradiol. 2005;26:2602-2609. [PubMed] |
| 13. | Li J, Lan Z, He M, You C. Assessment of microscope-integrated indocyanine green angiography during intracranial aneurysm surgery: a retrospective study of 120 patients. Neurol India. 2009;57:453-459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 40] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 14. | Oda J, Kato Y, Chen SF, Sodhiya P, Watabe T, Imizu S, Oguri D, Sano H, Hirose Y. Intraoperative near-infrared indocyanine green-videoangiography (ICG-VA) and graphic analysis of fluorescence intensity in cerebral aneurysm surgery. J Clin Neurosci. 2011;18:1097-1100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 15. | Uchino H, Nakamura T, Houkin K, Murata J, Saito H, Kuroda S. Semiquantitative analysis of indocyanine green videoangiography for cortical perfusion assessment in superficial temporal artery to middle cerebral artery anastomosis. Acta Neurochir (Wien). 2013;155:599-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 16. | Struffert T, Deuerling-Zheng Y, Engelhorn T, Kloska S, Gölitz P, Bozzato A, Kapsreiter M, Strother CM, Doerfler A. Monitoring of balloon test occlusion of the internal carotid artery by parametric color coding and perfusion imaging within the angio suite: first results. Clin Neuroradiol. 2013;23:285-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |